Abstract
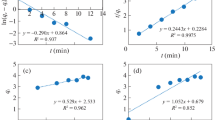
In recent years, arsenic contamination in our surface water sources has become a major problem. Therefore, water treatment is the key to alleviate this problem. Adsorption as one of the polluted water remediation methods has proven to be efficient and rapid. Its underlying mechanisms can be ascertained through the study of isotherm, kinetics and thermodynamics parameters of the adsorption process itself. Therefore, in the present paper, existing data of arsenate (As(V)) adsorption mechanisms by multiple samples of adsorbents will be interpreted by modelling the data. The data were obtained from experiments conducted previously involving humic acid-coated magnetite nanoparticles (NP). The effect of variables such as the synthesis temperatures and humic acid concentrations on the adsorption was thoroughly investigated via the experiments. In this paper, both linear and non-linear models were applied, and the results were compared. The non-linear model fitting was done with Microsoft Excel 2016 solver. The equilibrium adsorption data was fitted to the Freundlich, Langmuir and Temkin isotherms as well as the kinetic pseudo-first order (PFO) and pseudo-second order (PSO) models. Linear coefficient of determination was used for linear regression while non-linear regressions was done through the non-linear error function chi-square (χ2). The best model that fits the experimental data were decided accordingly for the different adsorbent samples.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Wołowiec M, Komorowska-Kaufman M, Pruss A, Rzepa G, Bajda T (2019) Removal of heavy metals and metalloids from water using drinking water treatment residuals as adsorbents: a review. Minerals 9(8):487. https://doi.org/10.3390/min9080487
López-Luna J, Ramírez-Montes LE, Martinez-Vargas S, Martínez AI, Mijangos-Ricardez OF, González-Chávez, Carrillo-González R, Solís-Domínguez FA, Cuevas-Díaz, Vázquez-Hipólito V (2019) Linear and nonlinear kinetic and isotherm adsorption models for arsenic removal by manganese ferrite nanoparticles. SN Appl Sci 1(8):N/A. https://doi.org/10.1007/s42452-019-0977-3
Rahdar S, Taghavi M, Khaksefidi R, Ahmadi S (2019) Adsorption of arsenic (V) from aqueous solution using modified saxaul ash: isotherm and thermodynamic study. Appl Water Sci 9(4):87–96. https://doi.org/10.1007/s13201-019-0974-0
Water Quality Association (2014) Arsenic fact sheet [Fact Sheet]. International Headquarters & Laboratory
Devaraj NK, Elghazali SR, Ganapathe LS, Mukter-Uz-Zaman ASM, Wong HY (2019) As(V) Adsorption kinetics of humic acid-coated magnetite particles. Appl Mech Mater 892:72–78
Arsenic Contamination of Groundwater—an overview | ScienceDirect Topics. Arsenic Contamination of Groundwater. https://www.sciencedirect.com/topics/pharmacology-toxicology-and-pharmaceutical-science/arsenic-contamination-of-groundwater#:%7E:text=Over%20137%20million%20people%20in,locations%20in%20Thailand%20and%20China. Last accessed 05 June 2020
Edet UA, Ifelebuegu AO (2020) Kinetics, isotherms, and thermodynamic modeling of the adsorption of phosphates from model wastewater using recycled brick waste. Processes 8(6):665. https://doi.org/10.3390/pr8060665
Ayawei N, Ebelegi AN, Wankasi D (2017) Modelling and interpretation of adsorption isotherms. J Chem 2017:1–11. https://doi.org/10.1155/2017/3039817
Saruchi, Kumar V (2019) Adsorption kinetics and isotherms for the removal of rhodamine B dye and Pb+2 ions from aqueous solutions by a hybrid ion-exchanger. Arab J Chem 12(3):316–329. https://doi.org/10.1016/j.arabjc.2016.11.009
Hokkanen S, Bhatnagar A, Koistinen A, Kangas T, Lassi U, Sillanpää M (2017) Comparison of adsorption equilibrium models and error functions for the study of sulfate removal by calcium hydroxyapatite microfibrillated cellulose composite. Environ Technol 39(8):952–966. https://doi.org/10.1080/09593330.2017.1317839
Palanivell P, Ahmed OH, Latifah O, Abdul Majid NM (2020) Adsorption and desorption of nitrogen, phosphorus, potassium, and soil buffering capacity following application of chicken litter biochar to an acid soil. Appl Sci 10(1):295
Chowdhury S, Mishra R, Saha P, Kushwaha P (2011) Adsorption thermodynamics, kinetics and isosteric heat of adsorption of malachite green onto chemically modified rice husk. Desalination 265(1–3):159–168. https://doi.org/10.1016/j.desal.2010.07.047
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Nature Singapore Pte Ltd.
About this paper
Cite this paper
Mubarak bin Hamzah, A.A., Devaraj, N.K. (2022). Mathematical Modelling of As(V) Adsorption by Humic Acid-Coated Magnetite Nanoparticles. In: Alfred, R., Lim, Y. (eds) Proceedings of the 8th International Conference on Computational Science and Technology. Lecture Notes in Electrical Engineering, vol 835. Springer, Singapore. https://doi.org/10.1007/978-981-16-8515-6_29
Download citation
DOI: https://doi.org/10.1007/978-981-16-8515-6_29
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-16-8514-9
Online ISBN: 978-981-16-8515-6
eBook Packages: Intelligent Technologies and RoboticsIntelligent Technologies and Robotics (R0)