Summary
Entomopathogenic infections by protozoans predominantly, the microsporidians, are of paramount significance in being ineradicable that caused subtle pathologies to induce reproductive productivity decline and shortening of life spans. These have been considered incredibly fit as long-term regulators of pests for effective checks of disease outbreaks. Protozoans are an important constituent of biopesticides with a narrow target range, hence used for a specific problem; their slow-action characteristics rejuvenate suppression of pests with limited field persistence, and devoid of residues. The detection and diagnostic potential for the length of survival of spores in the habitat has been facilitated by the application of advanced biochemical techniques. The critical inputs regulate mutualism after invasion of microsporidia into intracellular environment of the host-cell and influence protein synthesis, growth and endomitosis during merogony. A determined impact of lessening noxious pest hosts with the assistance of non-chemical application techniques in the field attributed commercial significance to the protists (microsporidia) and a variety of other protozoa. Vertical transmission comes into effect once the microsporidians invade gonads of insect pests, followed by the contamination of eggs. Therefore, not only that spores adhere to eggshell surface, and egg contents comprise spores and developmental stages yet the larvae hatched from internally contaminated eggs are often not infected. Microsporidia with reduced mitochondria, that is mitosomes, are sans mitochondrial genome, and the energy for their functioning is extracted from the host’s intracellular environment. Mitosomes, devoid of cristae, might be dependent on ATP produced by glycolytic pathway because oxidative phosphorylation was dysfunctional. Pest suppression characteristics of biocontrol agent are a factor dependent on host’s ecology.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Entomopathogenic
- Protozoa
- Microsporidia
- Biocontrol
- Molecular
- Strategy
- Transovarial transmission
- Environmental
8.1 Introduction
The protozoans belonging to certain phyla are pathogenic to insects. Some of these insects are important pests of various agricultural crops. Thus, these protozoans may act as biocontrol agents and play a significant role to regulate the population of insect pests. The protozoans, unicellular eukaryotes, particularly under the Sub Kingdom Protozoa, viz., neogregarines and microsporidia, are relatively more important in this context. The potential of protozoans to invade the body of their hosts in huge numbers, and kill them by inducing dysfunction of organs or depleting the host of essential reserves, has earned these organisms a unique position in their own way. The refinement of the concept of management of pests incorporating conservation efforts in coherence with refurbishing enhanced populations of the existing natural control agents has been of primary interest in the application of protozoans in the biocontrol of insects. The issues of application of protozoan organisms as important elements of economic entomology have been frequently reviewed (Brooks 1974, 1979; Mclaughlin 1971, 1973; Tanada 1976). Initially it was believed that the actions by protozoans were neither rapid nor useful for a broader range of effectivity. The slower-paced performance of the pathogenic protozoan forms deprived them of their penetrative capabilities to cause mortality into higher abundance of host pest on their own. Their effective use is, therefore, implied in high threshold pest populations to cause damage or else when they are required as a component of pathogen-plus-chemical formulation or in a multiple pathogen combination to act upon pests. In addition, in Lambornella clarki Corliss and Coats, an endoparasitic ciliate of the tree-hole mosquito, Aedes sierrensis (Ludlow), exhibited remarkable potential as a biocontrol agent to keep effective check on it, to prevent outbreak of dangerous diseases. The armament of cuticular cysts is deployed by the ciliates of the genus Lambornella that penetrate to cause damage by attaching to the cuticle of culicine mosquitoes, particularly tree-hole breeding species, for a remarkable mosquito control. Clark and Brandl (1976) gave an illustrated account of the mechanism of dissolution of the cuticle of larval mosquitoes to facilitate penetration into epidermis for its arrival in haemocoele. Two mosquito-associated species have been reported, viz., Lambornella stegomyiae Keilin and L. clarki Corliss & Coats. Egeter et al. (1986) and Washburn and Anderson (1986) also emphasized its effective role as a potential biocontrol agent of the container breeding mosquitoes.
The typical cyst forming ability of this endoparasitic ciliate comes in handy for it to survive over the years to pass over unfavourable conditions. The property of resistance to the cystic framework is the typical characteristic of such protozoans that equipped these pathogenic organisms with appreciable resistance against desiccation. This unique ability of the cysts to withstand adversities during the stages of development enabled the organism encased within, to overwinter or cross over to the next weather circle stage of the following year. Taking a cue out of such apparent characteristics of the cysts of this endoparasite, Anderson et al. (1986a, b) advocated field trials and perused in vitro methods to determine the efficacy and feasibility of their use in field experiments. The association of a variety of protozoans, particularly microsporidians, with mosquitoes and other arthropods of medicinal significance, is known (Lacey and Undeen 1986). The exposition of their practical utility has been restricted because of their complex life cycle, as well as in vivo technology to be employed for maintenance. The application of novel innoculative technique to ensure the enhanced productivity oriented release highlighted its functional significance in the dynamics of mosquitoes, under specific habitat conditions. The report of Flint and Dreistadt (1998) compiled microorganisms, particularly entomophilic protozoans as pathogens in coordination with a variety of bacteria, fungi, nematodes and viruses that possessed the natural potential to infect and kill the hosts if the congregated pest populations flourished under conditions of high humidity. Certain protozoans, viz. the microsporidia, Nosema pyrausta (Fig. 8.5) and Vairimorpha necatrix (Fig. 8.6), are the typical examples that demonstrated regulation of lepidopteran pest populations. The abundance of pest populations would be monitored the moderate to low pathogenicity because the inverse relationship between their pathogenicity and prevalence was obvious. While V. necatrix operated through the insect gut wall invasion as well as disruption due to the bacterial septicaemia. Though this ensured survival and propagation of larval populations, yet adulthood was never attained. Thus, this microsporidian could rarely register its occurrence in natural populations as it was deprived of the transovarial transmission. Under the circumstances, therefore, its potential as an important constituent of being a microbial pesticide, on a short-term control operation, has been realized. On the other hand, the natural companion in the lepidopteran pests, N. pyrausta (Paillot 1927) that parasitized a single host, that is, the European corn borer, with acquired lower pathogenicity in the stressful environment, succeeded to kill larvae. Resultantly, therefore, the adulthood was achieved by most survivors but whose power to reproduce was adversely affected and length of life curtailed. This microsporidian was encountered with higher prevalence in natural precincts, equipped with the virtues of being transmitted transovarially. In such ways though not being an organism that showed severe pathogenicity, it still has been attributed a significant constituent of the mechanism regulating natural pest populations.
8.2 Protozoan Assessment as Biocontrol Agent
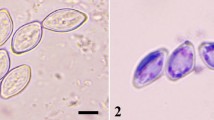
The microsporidia (Fig. 8.1) of various protozoan organisms encountered in a variety of insects in terrestrial as well as aquatic ecosystems have been summarized in Table 8.1. The occurrence of microsporidia in the insect pests has been listed by insect host taxonomic order in Table 8.2. The insect orderwise enumeration of microsporidian fauna is presented in Table 8.3. The reported species of insects frequently occurring in the insect pests in the forest areas have been compiled in Table 8.4. The common as well as specific names of insect pests, that occurred in forests and microsporidia harboured by them are listed in Table 8.5. The precise details of spore wall proteins of microsporidia and their subcellular location are summarily presented in Table 8.6.
Internal structure (Diagrammatic) of a microsporidian spore (Courtesy: Cali and Takvorian, 2014) (Reprinted from Han et al. 2020) (A, Anchoring disc; Dn, Diplokaryon; En, Endospore; Ex, Exospore; Lp, Lamellar Ploroplast; P, Unit membrane; Pt, Polar tube; Pv, Posterior vacuole; R, Ribosome; Sp, Sporoplasm; Tp, Tubular Ploroplast)
8.2.1 Phylum Ciliophora
Though not recognized as obligate endoparasitic forms, a few strains, like T. pyriformis of genus Tetrahymena spp. that otherwise are free-living forms, possessed the traits of parasitism. Corliss and Coats (1976) enlisted parasites of genus Tetrahymena Furgason and Lambornella Keilin of the haemocoele of culicine mosquitoes. The mortality-induced infectivity in the haemocoele of mosquito hosts by both Tetrahymena and Lambornella (Table 8.1) was taken into account, although the exact data on the rates of mortality were evading.
The evidence of culturability of Tetrahymena (Table 8.1) and Lambornella being available, the findings of Clark and Brandl (1976) to conclude prevalence of L. clarki in Aedes sierrensis to the level of 50% strengthened the contrary view of low control potential of ciliophorans. This was because of lower infectivity or abstained natural epizootics of ciliates underlined by McLaughlin (1971) and Henry (1981) that could respond to the likelihood of decimation of infected lot of individuals in the population due to severe pathogenicity. WHO released the document WHO/VBC/81.803 briefing the details of results to give evidence.
8.2.2 Phylum Sarcomastigophora
The evidence is in favour of the spread of these parasitic protozoa due to the faecal ingestion, which is not necessarily associated with the formation of cysts. But the possibility of their being pathogenic is a rarer one. Therefore, in a review published by Wallace (1979), a large number of flagellates belonging to the group of kinetoplastic flagellates, commonly being harboured by a variety of invertebrates, including arthropods, were not considered to be biocontrol organisms.
8.2.3 Phylum Apicomplexa
At least three modes were employed to explore the role of gregarines (sub-class Gregarinia) and some adeleine coccidia (sub-class Coccidia, sub-order Adeleina) of the Apicomplexa in biological control of insects.
8.2.3.1 Laboratory Experiments
A gregarine, D. immitis exhibited the potential to damage Malpighian tubules of A. aegypti (Table 8.1) and other culicine mosquitoes in the laboratory experiments conducted by Sneller (1979).
8.2.3.2 Field Trials
Sneller (1979) also reported parasitization by D. immitis and damage in process, of the Malphigian tubules, by this gregarine. Similarly, the neogregarines and adeleine coccidia, the common intracellular parasitic forms sustained one or more cycles of schizogony, to produce them in enormous numbers. The destructive abilities of the latter and excessive consumption of reserves in the host body led to the greater number of death of host bodies, with decline in reproductive ability in the colonies of insects maintained in the laboratory. McLaughlin (1971) highlighted success of propagation of Mattesia grandis McLaughlin (Table 8.1), a parasite of cotton boll weevil, Anthonomus grandis as a biocontrol agent of insect pest, in field trials. Their application was successful on their engagement as supplement in the role of feed stimulant.
8.2.3.3 Stored Grain Pests
The use of pheromones in the stored grain pest, Trogoderma glabrum, which lured insects as a tool (Shapas et al. 1977), has partially resolved the problem of outreach of infective spores of Mattesia trogodermae (Canning). Several other protozoan mixed formulations have also been applied to utilize Adelina tribolii and Farino cystistribolii against flour beetles, Tribolium spp.
8.2.4 Phylum Microspora
Microsporidia are among the largest group of successful biocontrol organisms under Protozoa. The widely spread out resistant spores initiate infections into pest populations. The spores hatch after ingestion, and penetration (Fig. 8.2) of its polar filament into a gut epithelial cell occurred, pushing through the peritrophic membrane barrier. However, the cytoplasm of the host cell received inoculation of sporoplasm in cases where polar filament was longer enough to have reached up to the cytoplasm of the host cell after passing through the gut wall and penetrating haemocytes directly (Malone 1990). The spores were resultantly produced by sporogony that was preceded by frequently occurring binary or multiple fissions, after initial invasion by the tube of polar filament. It is also known that the fatality, as well as the decline in reproductive potential of the host, and their life span dawned upon because the tissues of the host were replaced by the invasive infective spores in heavier numbers (Fig. 8.3).
Invasion by microsporidia into a cell of the host (a model) (Reprinted from Han et al. 2020)
A typical microsporidial developmental cycle (Fig. 8.3) can be divided into three phases. Phase I, the infective/environmental phase, is the only extracellular part of the cycle. It is represented by mature spores shed into the environment from previously infected hosts. Under appropriate conditions, the spores germinate (e.g., if the spores are ingested by an appropriate host). They are activated by the digestive tract environment; this results in the explosive expulsion of the polar filament (which ‘everts’ becoming a hollow tube). If the polar tube pierces a host cell, the spore contents, the sporoplasm, are injected into it and phase II begins. Phase II is the proliferative phase, the first phase of intracellular development. During this part of the microsporidian life cycle, organisms are usually in direct contact with the host cell cytoplasm or in a parasitophorous vacuole as they increase in number. The transition to phase III, the sporogonic phase, represents the organisms’ commitment to spore formation. In many life cycles, this is morphologically indicated by parasite secretions through the plasmalemma producing a ‘thickened’ membrane (many also form a surrounding sporophorous vesicle, SPOV). The number of cell divisions that follow varies, depending on the genus in question, and results in sporoblast cells that develop into spores (Reprinted from Cali et al. 1999)
It has been emphasized that the negativity features of greater time lag between initiation of infection by a microsporidia and the resultant tissue destruction, which might extend to several weeks, affected the potential of these infective agents that are detrimental to pest, to facilitate their usefulness in vector control. However, the characteristic of transovarial transmission negates this ineptitude to overcome by attaining of the retention of the potential of spread full to capacity of the infective microsporidia.
The activated transferability, sustainability of infective stages, and severe ferocity of invasive penetrative capability equip a biocontrol agent to achieve success. A long-term strategic management scheme encompassed the manipulation of several alternative methods. These included field trials to test the methods to ensure availability of bulk volumes of microsporidia and neogregarines, incorporating tests for ensuring their safety during the process of production, as well as testing of bait formulations to suit appropriate mass production. This has resulted into the registration of a microsporidia as a microbial pesticide with the United States Environmental Protection Agency.
A lot of efforts have been put in to explore novel methods of introduction, keeping an eye on the evaluation of their pathogenicity and the potential of their wider spread. While, on the other hand, the groups like ciliates, flagellates, amoebae and the less pathogenic Apicomplexa have been under focus to investigate their prevalence during random surveys as well as the descriptions of their newer taxa, if any.
The concerns have been expressed to verify the viability of cyst formation activity under experimental conditions (Fig. 8.4), though the cyst forming protozoans of the genus Lambornella, the free-swimming ciliates were amply encountered in natural conditions. This is yet to be tested whether Lambornella organisms could be cultured in the laboratory, and promoted for invasion into the aptly suited mosquito hosts, under natural habitats, by forming cuticular cysts. Therefore, most of the studies illustrating appropriate protozoan organisms for biocontrol of insects depended heavily on microsporidians and neogregarines.
Nucleotide acquisition and metabolism in host cells and microsporidia
Schematic illustration showing nucleotide metabolism in a typical microsporidian parasite within an infected host cell. Host cells can make nucleotides via de novo biosynthesis and regenerate ATP by oxidative phosphorylation – these pathways are absent in microsporidia. Host purine nucleotides can be stolen using microsporidia NTT transporters and then efficiently used and recycled by the parasites. Key: (a) E. cuniculi physically tethers mitochondria using an unidentified protein. (b) Only EcNTT3 has been found in the mitosome. (c) Nematocida may secrete a hexokinase into the host cell to stimulate host nucleotide production. (d) Nucleoside kinases are apparently absent from some microsporidian genomes but are present in Trachypleistophora hominis. (e) Thymidine kinase is present in some microsporidia but not all. (f) The microsporidian RNA degradation pathway is shown in the figure below.
(Reprinted from: Archibald, J.M., Simpson, A.G.B. & Slamovits, C.H. 2020. Handbook of the Protists. Springer. second Ed., p.1657)
8.3 Pathogenicity
The manifestation of pathogenic effects of the cysts (Fig. 8.4) harboured by pests was illustrated by the declining pest population growth emanated as a result of reduced fecundity as well as their longevity. This was implied as the long-term control measure. However, the short-term measure to restrict pest population growth, that attained critical peaks, emerged to be the mortality of larvae that were to be employed to rescue and ensure relief measures against adult mosquitoes.
The efforts have also been made to reveal mystery of short-term and long-term population control of pests that comprised proven record of N. pyrausta (Paillot) (Tables 8.3, 8.4) and V. necatrix (Kramer) (Tables 8.3, 8.4) as the most suited candidates for being agents of biocontrol.
8.3.1 N. Pyrausta (Paillot) (Fig. 8.5)
The European corn borers, Ostrinia nubilalis, introduced into the United States of America in 1917, along with N. pyrausta (Paillot) (Fig. 8.5), parasitizing their Malpighian tubules, comprised one of the most extensively studied moth host-microsporidian systems. The European microsporidian introduced sporadically in 1951 into the USA (Steinhaus 1951) was the subject of investigation (Zimmack et al. 1954) conducted on its distribution in 1954 in seven of the North Central states. The studies dealing with relative fecundity status of female moths revealed striking differences between moths that showed marked differences in their infectivity status. Fewer eggs were laid by the infected moths, whose longevity was short and the non-infected moths, who lived longer, laid numerous eggs (Zimmack and Brindley 1957). Simultaneously, faster-paced growth was hampered in the former, coupled with dim survival prospects.
Diagrammatic representation of the interactions between Nosema-type microsporidia and their hosts. Healthy larvae become infected by ingesting microsporidian spores which are present in the faeces and/or silk of infected individuals. Larvae infected by ingesting spores may die from the infection if they consume many spores at an early larval stage, but many infected larvae can develop into infected adults. Mortality may occur during pupation and emergence as adults. Much of the mortality caused by Nosema-type microsporidia (Tables 8.3, 8.4) occurs in the transovarially infected offspring in infected females. Transovarially infected larvae may be heavily infected and die in early larval stadia
(Reprinted from Maddox et al. 1998. Microsporidia affecting forest Lepidoptera. (In: (Eds.) Maddox, J.V., McManus, M.L. & Solter, L.F. Proceedings: Population Dynamics, Impacts, & Integrated Management of Forest Defoliating Insects.). USDA Forest Service General Technical Report NE-247.).
The evidence of invasive spores of microsporidian organisms was traced by Zimmack and Brindley (1957) in the yolk accompanying embryonated oocytes in the ovary. This was compiled in histological analysis. The infected female moths laid embryonated eggs with spores and the shallow foveae on the egg shell were found embedded with spores by Kramer (1959a). Several other reports have illustrated noticeable prevalence of infection in larvae, pupae as well as adult insects.
The concept of stress-induced effect of fatality to the insect pests infected with N. pyrausta was formalized by Kramer (1959b). The stress of winter was under investigation. The natural populations of O. nubilalis with reduced longevity and fecundity, under influence of the infection by N. pyrausta, were thus regulated, as reported by Van Denburgh and Burbutis (1962). The natural populations of O. nubilalis infected by the larvae of N. pyrausta in two counties of Nebraska between 1957 and 1972 were correlated with the density of parasites by Hill and Gary (1979). The two epizootic waves of Nosema (Tables 8.4, 8.5, 8.6) occurred at the onset of crash in borer populations that co-occurred with peak in parasite prevalence. The event could thus be correlated with the introduction of mortality on augmentation of prevalence by N. pyrausta in larvae of O. nubilalis.
Hill and Gary (1979) provided evidence of the eventuality that the parasitic forms did not act directly to the detriment of pest populations, as those due to a combination of environmental factors that regulated fecundity. The intricacies of regulatory dynamics put forth by Anderson and May (1981) could explain that most of the features of the population cycles concluded could actually conform to the host/parasite association model, derived earlier by Anderson and May (1980).
On the basis of the review of long-term observations, the effect of environmental factors emerged to be the significant operator that induced a supportive role to the dynamics of infections by N. pyrausta, which on its own was unable to cause significant damage to the pest, but, in combination with environmental attributes, was much more destructive.
8.3.2 V. Necatrix (Kramer) (Fig. 8.6)
The utility of V. necatrix (Kramer) as a stronger pathogen than N. pyrausta and V. necatrix (Kramer) is established, and its application as a microbial insecticide has been illustrated effectively (Tanada and Chang 1962). Its initial recovery from the army worms, Mythimna (Pseudaletia) unipuncta (Haworth) in Hawaii from more than half of the dead larvae, was ensured from a decimated pest population. The alarming signals emanated from it due to its uninhibited substantial prevalence the following year, at the same site again, while the population density of the army worms declined (Tanada 1964). Though its prevalence in the routine field crops is minimal, its outbreak in certain forest insects has been substantial (Maddox et al. 1981). A noticeable occurrence of V. necatrix has been encountered in several major agricultural pests, particularly a variety of phytophagous Lepidoptera (Maddox et al. 1981).
Diagrammatic representation of the interactions between Vairimorpha-type microsporidia and their hosts. Healthy larvae become infected by ingesting microsporidian spores which are released when infected larvae die and disintegrate. Spores are seldom present in the faeces or silk of infected larvae. Most Vairimorpha-type microsporidia are very pathogenic; individuals infected during the larval stage seldom develop into infected adults. These infected individuals usually die as larvae
(Reprinted from Maddox et al. 1998. Microsporidia affecting forest Lepidoptera. (In: (Eds.) Maddox, J.V., McManus, M.L. & Solter, L.F. Proceedings: Population Dynamics, Impacts, & Integrated Management of Forest Defoliating Insects.). USDA Forest Service General Technical Report NE-247.).
As the ruptured gut due to polar filament extrusion to reach at haemocoele triggered bacterial septicaemia drawing source bacteria from the midgut lumen, cent percent mortality level was attained following infection. The domain of this disease is with a larval base, in which the sites of infections are primarily the fat bodies. Adult insects evade infections because of the non-survival of larvae under the influence of disease as well as non-occurrence of transovarial transmission. This disease primarily being fatal to larvae than adult insects, it has been recognized as a larvicide for quickened depletion of the pest population.
The entry of protozoan organisms into the body of their hosts is most of the time through ingestion of their cysts. It is very rare that any other route is employed by protozoans to attack the susceptible pest hosts; for instance, the penetrative cysts of the ciliate Lambornella invade the mosquito’s cuticle. It is though intriguing that the life cycle of the flagellate, Trypanosoma sp., commonly parasitizing insects (Wallace 1979), does not comprise true cysts, but the transmittance of infection was suggested to have occurred by ingestion of contaminated faecal material. This infection is never passed on from mother to offspring, but it is transmitted from individual to individual.
8.3.3 Endoreticulatus Schubergi (Zwolfer) (Fig. 8.7)
The protists (microsporidia) in Endoreticulatus group (Table 8.2) were entirely distinguished from the other two, namely N. pyrausta and V. necatrix, described as above. Restricted to midgut epithelial sites, their phylogenetic interrelationships segregated these from others in the other two groups, while simultaneously their life cycles and decimated pathogenicity were also the differentiating features. They were capable of producing only a uninucleate environmental spore, enveloped in a cover, that encompassed 16, 32 or more spores. The most commonly encountered Endoreticulatus schubergi (Fig. 8.7) never adopted the transovarial pathway to reach at the site of infection in larval populations of forest Lepidoptera. It, however, had the potential to produce chronic infections. Though their vertical transmission on the surface of eggs existed (transovum transmission) to a minimal extent, this pathway was even a subdued one when compared to the actual vertical transmission within the environment of the egg (transovarial transmission). The mechanism of transmission of Endoreticulatus schubergi and associated protists are illustrated in Fig. 8.7, identifying the stages at which mortality occurred. A large variety of Lepidopteran pest hosts harboured Endoreticulatus spp. quite frequently, but the impact was felt to the maximum extent of a ‘sublethal’ type only, because of the chronic nature of infection by this species (Maddox et al. 1998).
Diagrammatic representation of the interactions between Endoreticulatus – type microsporidia and their hosts. Healthy larvae became infected by ingesting microsporidian spores which are present in the faeces of infected individuals. The mortality rate is very low in individuals infected with Endoreticulatus-type microsporidia. Infected larvae develop more slowly than healthy larvae and produce faeces contaminated with spores throughout their larval development
(Reprinted from Maddox et al. 1998. In: (Eds.) McManus, M.L. & Liebhold, A.M. Proceedings: Population Dynamics, Impacts, & Integrated Management of Forest Defoliating Insects. USDA Forest Service General Technical Report NE-247, p.187–197.)
8.4 Transmission
Protozoa normally enter their hosts when encysted stages are ingested and rarely by any other route (e.g. the use of invasion cysts of the ciliate Lambornella (Table 8.3) to penetrate the cuticle of mosquitoes). True cysts are unknown in some trypanosomatid flagellates of insects (Wallace 1979) but even in these cases the mode of infection is probably by ingestion of contaminated faecal material. Transmission is defined as horizontal when the pathogen is passed from individual to individual within and between generations but not directly from parent to offspring, whatever the mode of entry. When a parasite infects the gonads of its host, vertical transmission from parent to progeny is possible. This may be transovum, when the infective stages contaminate the external surface of the egg and are consumed by the larvae at eclosion, or transovarial, when the infection is located within the eggshell. Occasionally the infected parent is the male. The different explored routes summarizing transmission pathways of a large variety of protozoans in the insects were reviewed earlier by Fine (1975). Two specific modes of transmission, (i) Horizontal alone and the other (ii) Horizontal as well as Vertical, were concluded. Though the horizontal transfer of cysts has been a natural component of the life cycle’s pathway, the transfer of protozoan organism or their cysts from parents to the offspring is likely facilitated only by the invasion by a parasite or its developing entities into the reproductive bodies, like gonads, or eggs. This could be termed transovarial transmission.
The uniformly adopted strategy by a majority of protozoans to effectively propagate individual to individual transmission essentially required maintenance of parasites throughout the year, including the potential of their reproductive entities, particularly cystic bodies to survive during the unfavourable environment. In such a manner, these bodies enabled overwintering of the developing stages, for example, cysts, diapausing larvae, pupae or non-feeding adults, so as to ensure continuation of the progeny.
8.4.1 Horizontal Alone
Most Protozoa are maintained this way in invertebrate host populations. There is no strong evidence that any amoebae, flagellates, ciliates or Apicomplexa are transmitted by the vertical route. Restriction of transmission to the horizontal route requires that the parasites are maintained in the living hosts throughout the year or that they can survive in the environment through periods when the host is quiescent and non-feeding, that is, as eggs, diapausing larvae, pupae or non-feeding adults. In seasonal pests, it is likely that the initial generation of the new season will have a low prevalence of disease because infected hosts may have a poor over-wintering survival rate, and the number of available infective stages may be low. The latter could have resulted due to loss of viability with time and their ultimate removal from the environment. Infections will multiply in young larvae, releasing infective stages at death to spread through the new generation. Spread will be enhanced if spores are released continuously from live hosts, as would occur with infections of the gut, Malpighian tubules and salivary or silk glands. In multivoltine host, prevalences would increase with each generation. The characteristics of V. necatrix are noticeably remarkable to have a rapid spread within a generation. But its virulence (effecting rapid removal of the parasitized sector from the population), coupled with the absence of a vertical route of transmission and low survival rate of spores in the environment, is sufficient to account for the low prevalences and rare sightings of this parasite in nature.
8.4.2 Horizontal Alone As Well as Vertical & Horizontal Routes
The other alternative mode of transmission could be that of vertical transmission in a way that the invasion of microsporidians into the contents of the eggs could result into hatching of larvae from the contaminated egg material (Kramer 1959a; Brooks 1968). The winter moth, Operophtera brumata (L.) (Canning and Lom 1986), population in an oakwood habitat recorded augmented prevalences of microsporidia. The spread of as many as three species of microsporidia was noticed (Canning 1982). The possibility of occupation of microsporidia on the surface of egg shells as well as inside the contents of the eggs (Kramer 1959b; Brooks 1968) opened up interesting avenues. The rising proportion of incidence of spores and developmental stages of the three species of microsporidia harboured by the winter moth, O. brumata (L.), were encountered in the newly laid, non-embryonated eggs examined by Canning and Lom (1986).
Prior to eclosion, the non-occurrence of parasites either in the larval tissues contained in eggs, or in larvae post 1 day of hatching necessitated the location of site of survival of spores during embryogenesis. The spores were shelved in polar plug, meconium, that represented remnants of yolk entering into the lumen of the gut, during late embryogenesis that facilitated merger of stomodaeum with proctodaeum to form midgut. This not only extended disinfecting protection to larvae but also ensured transovarial transfer for vertical transmission. Sections of larvae within the eggs showed that spores were present only in a plug of material, occupying the lumen of the gut. This plug, the meconium, represents the remnants of the yolk taken into the gut lumen at the time when the anterior and posterior parts of the gut, stomodaeum and proctodaeum, join up via the midgut late in embryogenesis. This suggested itself as a remarkable adaptation to ensure that the larvae did not succumb to overwhelming infections while still within the egg. If the larvae were to die, the purpose for which transovarial transmissions has evolved would be defeated. Therefore, it remained to be ascertained whether the infections in newly hatched larvae are initiated in the epithelial cells of gut due to sporoplasm inoculation after hatching of spores, or whether these spores are passed in the meconium and are eaten by, or accidentally contaminate, the larvae. Thomson (1958) confirmed the occurrence of a similar phenomenon for N. fumiferanae (Thomson)-spruce budworm, Choristoneura fumiferana (Table 8.3) system. N. fumiferanae were fatal only in the actively feeding larvae post-hatching.
Gaugler and Brooks (1975) emphasized that the failure of heavier egg infections of N. heliothidis (Lutz & Splendore) to reduce egg hatch in corn earworms, Heliothis zea, and account for the normal, embryonic development and lack of disease symptoms in newly hatched larvae apparently occurred due to the failure of the egg stages to penetrate larval tissues during embryogenesis. Therefore, the mystery of the mechanism of attained infections by eggs in larvae in the microsporidian—host systems remained to be cracked. The sex-wise differentiation in the larval infestation of the dimorphic diptera, namely Amblyospora (Table 8.2) and Thelohania, revealed heavier infections in males than females (Hazard and Oldacre 1976).
8.5 Protozoans as Biocontrol Agents to Restrict Grasshoppers and Locusts
The density of the pests, attributes of the environment, as well as the conditions of plants are, in fact, instrumental to attract the wrath of the grasshoppers that might be recognized as chronic pests (Watts et al. 1982). Though due to behavioural advantage to the grassland songbirds, utilizing feeding on grasshoppers resulted into extensive breeding of the former (Martin et al. 1997), yet colossal loss was caused to the crops and vegetation, due to chewing and clipping by even lower densities of pests that consumed the precious crops of farmers and poor people. Although the protozoans were considered to be the slow-acting control agents, which affected rapid breeding and feeding speed of acridid organisms, yet their potential for action fell short of comparably rapidly acting fungi and certain chemical insecticides. Bidochka and Khachatourians (1991), Brooks (1988), Mclaughlin (1971), Henry (1990), Maddox (1987), and Raina et al. (1987) put forth extensive reviews to succinctly account for the protozoan potential as biocontrol agents of insect pests. A number of earlier workers (King and Taylor 1936) have been assertive about the enfeebling effect of a variety of protozoa on grasshoppers. Microsporidia have spearheaded the targeted detriment of acridid grasshoppers, but besides Phylum Microspora, a great variety of Amoebida, and Eugregarinida, Phylum Apicomplexa of Neogregarinida as well as Phylum Ciliophora have succeeded as a biocontrol agent. The adverse influence of Malameha locustae (King and Taylor 1936) harboured by Locusta pardalina (Walker) resulted into effect on fecundity of these locusts. The proof of their ferocious attacks on the colonies too, of several susceptible species (Pickford and Randell 1969), maintained in the laboratory was obtained by Jackson et al. (1968). The common occurrence of the pathogen on the colonies of Schistocerca gregaria (Forskil) including the nondiapause strain of Melanoplus sanguinipes was noticed. Interestingly, simultaneous enhanced resistance against insecticides in the cyst-harboured Malpighian tubules (Hinks and Ewen 1986) co-occurred in certain extreme cases, whose account of life history and pathogenesis has been recorded by Braun et al. (1988). Several Orthoptera commonly harboured gregarines in nature as well as in certain poorly maintained colonies of S. gregaria (Harry 1970). Certain well-known protozoans that could succeed in making field crickets physiologically dysfunctional as well as brought disturbances in the ecology of field crickets (Zuk 1987) were brought on record. The taxonomic status of Gregarinida, placed under Apicomplexa, was reviewed by Levine et al. (1980). These were reportedly encountered in the gastro-intestinal tract of more than 30 species of grasshoppers. Though not thoroughly analyzed, gregarines existed in the relationship of symbionts with grasshoppers but their role as potential pathogens is being actively investigated world over (Johnson et al. 1997). However, serious efforts are being made to employ microsporidia as candidates for the potential control of grasshoppers and locusts (Henry and Oma 1981). Based on the characteristics of rapid transmission, fecundity and ease of formulation, N. locustae are the favoured candidates for use in the biocontrol of locusts and grasshoppers. A wide spectrum of Nosema spp. were encountered in the field populations of Locusta migratoria migratorioides (Reiche and Fairnaire) at the Imperial College, London, by Canning (1953, 1962b). Northern Great Plains and Prairies of North America were the native sites of N. locustae. Its spores were also frequently harboured by M. bivittatus, M. mericanzs Saus. (M. sanguinipes) and M. dawsoni (Scudder). This has commonly been recognized as a common inhabitant of Acrididae with more than 80 species having frequently harboured spores of N. locustae (Henry 1969; Henry 1985; van der Paauw et al. 1990; Bomar et al. 1993). The life cycle of N. locustae illustrated that unless its infective spores have arrived in the lumen of the gut through ingestion of N. locustae, they shall not germinate to release binucleate sporoplasm into the live cells of host pests by rapid extrusion of polar tubes. Therefore, through this mechanism the contact of sporoplasms with the epithelial cells of the midgut is successfully established or the sporoplasms reach directly into the gut cells (Raina and Ewen 1979). The invasion of the yellow organ, the principal fat body organ performing the function of storage of energy, by the sporoplasm enabled generation of developmental entities, like meronts, sporonts, sporoblasts and spores in succession (Canning 1962a). A starved energy reserve of the pest due to microsporidial infection and the ensuing hypertrophy, thereafter, disrupted the dynamicity of intermediate metabolism in the fat cells of the principal fat body, and weakened the body of the pest to induce debilitating conditions. In certain other cases, the virulent pathogeny of microsporidians, greater than N. locustae, induced mortality occurred in acridids. As for example, the pathogenicity by N. cuneatum Henry enhanced susceptibility to protozoan infections with effective hindrance to development resulting into frequent mortality at an even greater rate. The experiment enumerated by Mussgnug (1980) that the effective outcome on control of pathogenicity by application of N. locustae alone was at a relatively lower level than when pathogens combined in the formulations of Malathion- N. locustae were applied.
8.6 Forest and Tree Crop Pests
The typical example of vertical transmission of microsporidia of N. fumiferanae brought to the fore the primary pathway of disease transmission through consumption of its spores by the spruce bud worm, Choristoneura fumiferana (Table 8.3), in the forest and tree crops. Virtually, the fourth and fifth instars received a spore inoculum from the frass and cadavers that were generated after the death of larvae, which were naturally infected from parenteral infections, but the latter could not attain maturity. On the other hand, the larvae that did not receive infections from their parents, and instead their fourth and fifth instar stages received the spore inoculum from the frass and cadavers, attained maturity as well as harboured infections. Thus microsporidia are maintained in the pest hosts by vertical transmission, once the consumption of contaminated spores triggers the augmented incidence of infection.
The spores of Nosema were illustrated to be germinated in gut and live cells of gut, fat body and other tissues of Lepidoptera, beetles, locusts and a variety of Orthoptera. They multiply to trigger sporulation that culminated with destruction of organs resulting into being chronic and debilitating. The protozoans were transported by rotten food stuff, cannibalistic behaviour of the organisms through the transovarial pathway (Capinera and Hibbard 1987). The living insects happen to be the fertile ground for the spores of protozoans that sustain with appreciably reduced virulence. It was demonstrated that specifically Nosema locustae has had a restricted role in the biocontrol of grasshoppers. Certain formulations of other protozoans, like Nosema combined with Bacillus thuringiensis, were utilized as dry baits sprinkled over the surface of the soil against other pests, but their availability was restricted commercially.
8.7 Observations on the Sporoplasm
The transport of sporoplasm into the cells of the host pests preceded by the microsporidia infection of host cells involved expedited ejection of the polar tube (Fig. 8.2) (Weidner 1970; Frixione et al. 1992; Takvorian et al. 2005; Han et al. 2017). Weidner (1972) and Han et al. (2017) elucidated that the event of the onset of the infection triggered sticking of microsporidian spores to the cells of the host pests or the surrounding tissues. The indomitable association of the polar tube during infection of the host cells by microsporidia to target spore germination was propagated by Cali et al. (2017). It was also asserted by Takvorian et al. (2013) and Vavra and Larsson (2014) that the transfer of infection is possible by the outflow of infectious sporoplasm, to reach polar tube after its passage from spores. The mature spore formation occurred after initiation of a reproduction cycle, that terminates into meronts’ emergence, once the sporoplasm invaded into the host cell (Cali et al. 2017; Han and Weiss 2017), and the formation of proliferative forms, meronts was preceded by the production of constituents of the cycle, like sporonts and sporoblasts. The microsporidia not only were devoid of functional mitochondria, but also did not comprise the full complement of genes capable of ATP generation, yet could contribute to the completion of glycolysis. The protein coding gene complement of the microsporidial genome was restricted to ~3000.
Three of the total four nucleotide transport proteins (NTT1–4), identified to date from certain microsporidia namely, Encephalitozoon cuniculi and Trachipleistophora hominis, have been known to be the constituents of the membrane of sporoplasm. It has been illustrated in the literature dealing with microsporidia evolution, enumerating the findings of Tsaousis et al. (2008), Heinz et al. (2014) and Dean et al. (2018) that the horizontal gene transfer was instrumental in the displacement of these nucleotide transport proteins from bacteria to microsporidia. Resultantly, these nucleotide transport proteins illustrated their potential to transport ATP, GTP, NAD+ and purine nucleotides from the cytoplasm of the host. The mechanism to transport energy as well as nucleotides from the cells of the hosts is also undertaken by microsporidia major facilitator superfamily (MFS), which is an alternative to the nucleotide transport proteins (NTT1–4), being a sporoplasm surface located protein family. These were ThMFS1–4, of which the location of ThMFS1 and ThMFS3 was traced to be in the sporoplasm plasma membrane during infection. These were also reported (Major et al. 2019) to be performing the similar function of transportation of ATP, GTP and purine, identical to those by NTTs. But it is remarkable that as yet the pyrimidine nucleotide import system of NTTs nor ThMFS remained undeciphered (Heinz et al. 2014; Dean et al. 2018; Major et al. 2019).
Microsporidia are supposed to possess appreciably reduced mitochondria, called mitosome, sans mitochondrial genome as well as ATP generation capacity (Williams et al. 2002; Goldberg et al. 2008). Typically, mitosomes constitute an essential constituent of Microsporidia, Diplomonads, Amoebozoa and Apicomplexa (Tovar et al. 1999, 2003; Williams et al. 2002; Keithly et al. 2005) that are a double membrane bound entity. The role of nucleotide transport proteins to ensure the import of nuclear encoded proteins for the functional activity of mitosomes, as well as the maintenance of organelles, to compensate the absence of cristae as well as their own DNA in the morphologically reduced sized mitosomes than mitochondria is well recognized (Burri et al. 2006; Hans-Peter Braun 2009; Tachezy 2019). The energy produced through glycolytic pathway becomes available for use by microsporidian mitosomes because of the process of oxidative phosphorylation to produce ATP being dysfunctional. However, the former process of energy generation operated only in spores, but became inactivated within the cytosol of pests, while intracellular growth and replication were initiated (Dolgikh et al. 2011; Heinz et al. 2012; and Williams et al. 2014). Han et al. (2019) demonstrated aggregation of mitochondria around microsporidia of the cells of their host pests, for energy supply, as confirmed in their experimentation on Encephalitozoonidae harboured by a parasitophorous vacuole of the cells of host pests.
8.8 Molecular Characterization of the Organisms of Biocontrol
The validity of the names of species of microsporidians was conducted in recent years by using chromosomal DNA comparisons (Munderloh et al. 1990). The control basis for vectors of Nosema spp. has been analyzed to highlight their molecular characteristics (Strett & Henry 1985). The N. Locustae spore structure profiling for polypeptide configuration was done to record pre-eminent polypeptides of exospores and spores post-buffer treatment comprising 4% sodium dodecyl sulphate and 0.02 M dithiothreitol (Heckmann 2020). An electron optically oriented recent approach to characterize a variety of biological control agents was introduced by Avery and Anthony (1983) to crack the mysteries of the molecular basis for biocontrol. In addition, the cell culture revelation could be helpful to elucidate the elements of biology of the candidates for microbial biocontrol. The biology of microsporidians in the in vitro cultures comprising continuous cell lines was demonstrated by Raina et al. (1987). The successful elements required to conduct such studies were aseptic spores in adequate number; applicable stimulus for germination; convenient accessibility to cell lines, along with a sensitive collaborators to conduct research (Kurtti et al. 1990; Heckmann 2020). The apparent proximity between the two microsporidians, N. payrausta and N. furnacalis, naturally parasitizing the European corn borer, Ostrinia nubilalis, and the Asian corn borer, O. furnacalis, respectively, was test cultured on experimentally reared caterpillars of O. nubilalis and centrifugally purified. This experimental illustration reassuringly proved the closeness of the two strains of N. payrausta and N. furnacalis, with even greater strength than it was previously demonstrated (Munderloh et al. 1990). Since the intricacies of distribution and interactions of microsporidia in the intracellular environment have long remained an enigma, certain advanced methods of detection, for example, ELISA (Enzyme-linked Immunosorbent Assay) of these spores, have been applied. These have helped to assess potential length of survival of spores in the habitat, in addition to their detection in the microhabitat. The facts that have emerged from the interactions of microsporidia of N. fumiferana infesting Choristoneura fumiferana (Table 8.3) revealed influence on the nutritional physiology of the hosts, which rendered them nutritionally deficient. This provided comparative data on nutritional efficiencies that revealed noticeable decline in Consumptive Index (CI), Nitrogen Consumptive Index (NCI), Relative Growth Rate (RGR) and gross (ECI) and net (ECD) production efficiencies in the infected than non-infected larvae. The approximate digestibility (AD), N utilization efficiency (NUE) and larval moisture content are taken into account to assess that of the healthy and diseased insects, reared on 2.5%N and 4.5%N diets, the enhanced CI in the former with deficient NCI than those reared on the latter diet, were the significant findings. However, no effect of dietary N was seen on the mortality of healthy insects. The outcome thus indicated sharing of products of host cell metabolism for parasitic metabolism and productivity requirements. A presumptive improvement in the growth and survival of host as well as biocontrol agent at the augmented levels of nitrogen was concluded (Heckmann 2020).
8.9 Molecular Mechanism in Pathogeny
The aspects of physiology, as well as biochemistry of microsporidians, have been scarcely worked upon, and information on the operative Kreb’s cycle in these has been wanting. Isotopic labelling and analysis of intensities of radiolabelled bands have come in handy to crack the mysteries of metabolism in the microsporidia. The cell fractionation achieved by segregating molecular entities based on differentiating isoelectric points, electrophoretic analysis of the enzymes encased within microsporidia, HPLC of the routine precursors or labelled metabolites of biologically significant molecules, functional localization of the specific enzymes in the cellular environment by chemical applications could reveal successfully the details of metabolic pathways within microsporidia (Heckmann 2020). It was presumptively postulated that the pre-emptive regulation of cyclic events within the host cell provided alternative energy pathway for microsporidia, as mitochondria and endoplasmic reticulum aggregated intracellularly, in the vicinity of the former. The greater variety of species diversity and their origin could be deciphered by the knowledge on aspects of evolution of form within the built environment of microsporidian organism, but the information on these was conclusively unavailable (Heckmann 2020). The microsporidia has essentially to survive within the host cell itself after invasion into it, especially following the principle of mutualism by triggering critical inputs during host–cell protein synthesis, growth and endomitosis during merogony. Soon thereafter, at the onset of sporogony, the microsporidia attained the characteristics of a pathogen, after this organism enveloped as a separate entity within the environment of host environment, and finally lysed the cell (Bulla Jr. and Cheng 1976).
The microsporidians predominantly being intracellular parasitic organisms, it’s only at the cellular level that their response could be manifested. But a variety in these manifestations was obvious; for instance, in the genus Pleistophora-type of event the interiors of the host cell are systematically engulfed completely. Once the invader started division to ensure growth, the manifestation into destruction of the cell was evident. Once the microsporidia occupied the interior of a cell, the lytic action to convert the cytoplasm into an array of vesicles, endoplasmic reticulum, cisternae and ribosomes jumbled into an unorganized mass (Bulla Jr. and Cheng 1976). No pathogenesis was encountered beyond the infected region in this event. However, in other genera, exorbitant hypertrophy in the infected cell under influence from the microsporidian resulted. The connective tissue cells and those of mesenchyme origin, reaching up to 14 mm size comprised the hypertrophic cells (Weiser 1969). These are termed xenoma. The physiological and morphological integration of the cell of the host whose internal environment and composition are totally altered, mainly because of the enhanced sized microsporidian, contributed significantly to the formation of xenoma, as a separate entity that was nurtured depleting natural constituents of the host cell. With the gradual growth of xenoma noticeable alterations in its wall triggered inflammation as well as proliferation. This resulted into replacement of xenoma by the granuloma, whose tissues invaded into the former, but with a diminished centrally placed spore mass. The molecular mechanism operating to express mitochondrial association with microsporidia has been a mystery even today (Lom and DyKova 1992). Though the secondary xenomas emanated from the reminiscent spores within, yet these never protruded or were liberated to the exterior. It is obvious that the granulation tissue invasion brought about changes into the wall of xenoma, and thus isolated the microsporidian with restricted auto-infection probability. The viable spores thus could only dehisce out if the host perished or else these were engulfed by the fibroblasts. Thus the mechanism of defence at the cellular level is strengthened with the central role of phagocytes that diminish the macrophage-engulfed spores, if their initial disintegration failed within granuloma. The gradual disintegration of the contents of the spore in the interior of phagocytes left an emptied folded membrane, that itself disintegrated ultimately. The evidence of chitinase enzyme activity was thus concluded by Heckmann (2020) confirming complete disintegration of the phagocytised spores that are redundant not to trigger any fresh infection activity episode further. The molecular level implications of the entry of microsporidians into the live cells were dealt with by Heckmann (2020) to emphasize that to analyze response of the host to the invader, such interactions would have to be understood at the level of cell.
The indications of interactions involving a surface protein 1 (EhSSP1) on the surface of the microsporidial sporoplasm of E. hellem with mitochondria from the cell of the host pest have been expressed (Han et al. 2019). All the three forms of voltage-dependent anion selective channels (VDAC1–3) being expressed in the cytoplasm of the outer membrane of mitochondria interact with EhSSP1. Han et al. (2019) asserted that the strength of the association of the mitochondria with microsprodian parasitophorous vacuole would weaken on account of this interaction being disrupted. Han et al. (2019) demonstrated that the energy retention encountered by the microsporidia in the cell of its host was presumably facilitated by the interaction of EhSSP1 with VDAC. It was peculiar to note that an unidentified cell protein within the host pest incorporated with the constituents of an invasion synapse (sporoplasm and polar tube in the cellular environment, form a synapse) also succeeded to interact with EhSSP1. This could trigger a novel response while sporoplasm is transported into the host cell cytoplasm. Further additional investigations could be conducted to explain this phenomenon. The flagellates, Trypanosoma sp., followed distinctively segregated life cycle pathway sans cysts, and instead, transfer of developmental stages occurred through direct ingestion of excretory matter. Naturally occurring DNA catenanes (Waraich et al. 2020) encountered primarily in the mitochondrial DNA isolated from HeLa cell lines (Hudson and Vinograd 1967) were assigned the task of the predominant method of organizing DNA in the mitochondria of trypanosomes. The network of the latter mitochondrial DNA, also called kinetoplast DNA (kDNA) (Englund et al. 1982), comprised two types of DNA circles that are interlinked to form an intricate network of maxicircles (20–40 kbp) and mini circles (̴ 2.5 kbp) (Lukes et al. 2002).
The soil-dwelling amoeba of genus Dictyostelium was the first free-living protozoan genome to be fully sequenced. It provided opportunity to examine activity of mutants of pathogens, as attenuation of their virulence resulted into depletion of their virulence in Drosophila as well (Han et al. 2020). This meant that Dictyostelium provided a valid tool to examine the virulence mechanism of pathogens of insect pests. Han et al. (2020) carried out genetic manipulation in Dictyostelium that could illustrate the application of autophagy at relative ease during infections by pathogens. The cysteine protease, Paracaspase, was found in Dictyostelium. But sufficient data is not available to examine molecular function of these caspase-like proteins or their role in programmed cell death. Such detailed investigations could also lead to development of treatment therapies against parasitic protozoans (Han et al. 2020).
8.10 Microsporidia Invasion
The transport of sporoplasm into the cells of the host pests preceded by the Microsporidia infection of host cells involved expedited ejection of the polar tube (Fig. 8.2) (Weidner 1970; Frixione et al. 1992; Takvorian et al. 2005; Han et al. 2017). Weidner (1972) and Han et al. (2017) elucidated that the event of the onset of the infection triggered sticking of microsporidian spores to the cells of the host pests or the surrounding tissues. The significant contribution to these initial steps during infection is those of spore wall proteins, that is, SWPs, as illustrated by Southern et al. (2007). The investigations revealed a number of SWPs interacting with host cells due to stronger affinity to the heparin-binding motif (HBM) and sulphated glycosaminoglycans (GAGs) associated with the surface of cells of the host pests on Nosema bombycis, (Table 8.6) Encephalitozoon spp. and Antonospora locustae (Table 8.6) (Hayman et al. 2001; Hayman et al. 2005: Southern et al. 2007; Li et al. 2009; Wu et al. 2009; Chen et al. 2017). A simultaneous report of the critical involvement of the integrin constituent of the host cell into the infection process by the microsporidium, Encephalitozoon intestinalis. E. intestinalis has also been on record in recent years (Leonard and Hayman 2017).
The canonical integrin-binding motif, arginine-glycine-aspartic acid (RGD), that regulated the binding of extracellular matrix (ECM) proteins with host cell integrins, were supposed to be present within a large number of hypothetical proteins, as revealed by the E. intestinalis genome analysis. A variety of pathogenic microbes, viz., several parasites, bacteria and viruses, that are reported to have stuck to the host cells were attributed to comprise proteins conducting interactions with host cell integrins (Patti et al. 1994; Bartlett and Park 2010). It was further demonstrated that the microsporidial spore attachment and host cell infection were barred by the incubation of host cells with RGD-peptides or recombinant alpha3 beta1 and alpha 5 beta 1 human integrin proteins (Leonard and Hayman 2017). It, therefore, conclusively suggested that, for the germination for production of spores and subsequent host cell invasion to succeed, the attachment of spores was an important part of the whole mechanism (Leonard and Hayman 2017).
8.11 Environmental Interactions
The body constituents of microsporidia are usually very soft and fragile that are usually susceptible to a variety of environmental attributes, but its spores can withstand adversities, externally to the cellular environment. The protozoans, with bacteria (decomposers), and ciliates (bacterivores) entered into effective interspecific and intraspecific interactions, in association with mosquito larvae (primary consumers) succeeded to influence characteristics, like cell size, cyst production and growth rate (TerHorst 2010, 2011). According to Maddox and Solter (1996), the survival period of life cycle stages of microsporidia, that belonged to the terrestrial category, distinct from the aquatic ones, was normally over 30 years, in liquid nitrogen storage conditions. But the usual survival time was from 1 month to a year, under natural environmental conditions, when these were under protection from UV radiation and other co-occurring microbes or degradation factors (Maddox 1973; Maddox et al. 1981; Brooks 1980, 1988; Goertz and Hoch 2008). However, microsporidia from aquatic environment were not that sturdy, and could easily be under the influence of environmental degradation (Becnel and Johnson 2000). It was quite an unusual characteristic of microsporidia that it could coordinate its presence or absence while their hosts are unavailable under variable conditions of seasonality or wide fluctuations in their population density. Subsequently, their unique potential to develop in a suitable host species, which was related to its original host species (Lange and Azzaro 2008) or in an alternate or intermediate host, when their routine hosts were not available, was an advantageous adaptation (Micieli et al. 2009).
The vertical transmission in certain types of infections by Edharzardia aedis in the host, Aedis aegypti, (Table 8.1) might not be so ferocious to the extent as to facilitate its inoculation to the local environment with spores for horizontal transmission among larvae of the mosquitoes (Koella et al. 1998). The most common instance of adoption of survival strategy by N. pyrausta microsporidia in diapause stages of the host, as well as to pass over the overwintering period through its fifth instar stage in O. nubilalis (Andreadis 1986; Siegel et al. 1988), and the relatively more common transovarial transmission through infected eggs of N. Portugal (Maddox et al. 1999) are available. This, of course, is quite imperative that the infection stress employed under additional physiological alterations during diapause could as well be fatal to the host; yet the protection of the infecting agent in the surviving host’s environment is facilitated to enable these to remain available, till the breeding of the forthcoming next generation of mosquito pest (Andreadis 1986).
8.12 Advantages & Disadvantages
8.12.1 Advantages
The management of biodiversity in a variety of ecosystems could only be sustained when the insect pest hosts remained under the stress of natural enemies (protists, i.e. microsporidia) to achieve biocontrol. It was, therefore, obvious that the protozoan pathogens that acted to the detriment of such pests be conserved for the safety of human beings, and in this way, the nontarget organisms would also succeed to contribute for the benefit of a balanced ecosystem. The formal recognition to the only species, namely, P. locustae, the pathogen of a grasshopper species, registered by the US Protection Agency (USEPA), as a microbial insecticide, is a distinctive step towards the advantage to human beings under the biocontrol programme, after a passage of half a century of expeditious researches world over in this field.
8.12.2 Disadvantages
The typically recognized slow-acting potential of microsporidia to control insect pests quite often leads to the prolonged wait to record detectable impacts on the pest host populations. Under such an eventuality, newer methods of appropriate formulations, keeping in view the ecological fate of protozoan spores, would be required to be developed. An eye would have to be kept on the cost-effective aspects of economy for the management of protozoan organisms, particularly microsporidia to care for their potential of biocontrol in the context of IPM.
8.13 Production and Storage
8.13.1 Production
Culture media are generally available for ciliates and trypanosomatid flagellates, and there is reason to be optimistic that only minor modifications would be needed to support the growth of species, which are endoparasitic. Since the freshwater environment of the host mosquitoes would be normal habitats for the infective stages of Lambornella and Tetrahymena, the cultured ciliates could be introduced directly. The problems of preventing the ciliates being consumed by the mosquitoes, as observed by Clark and Brandl (1976), would have to be overcome. Cultured flagellates cannot be introduced in this way as they neither form cysts nor have free-swimming stages, which can survive the physical and physiological changes as do ciliates. So far no species of flagellate has been deemed pathogenic enough to warrant efforts being addressed to the problem of introduction. Other Protozoa harboured by invertebrates, suitable for development as biocontrol agents, are all obligate intracellular parasites with resistant spores or cysts for transmission. None can be cultured free of living cells, and methods for mass production are limited at present to growth in natural or experimental hosts. In vitro culture in cell lines has been achieved for a few species, but spore production is well below that which is obtained in living hosts.
8.13.1.1 In Vivo Production
The episodes of production and efficacy of the insect infested microsporidia and neogregarines were reviewed by Brooks (1980). It was hypothesized that the optimum replication rates to produce hosts and to minimize their losses should be ensured by keeping an eye on the dose applied, age of the host and the time of incubation from the standpoint of economic production. Brooks (1980) summarily described a large variety of protozoans that were produced in live hosts, but strikingly in pretty lower numbers. The protozoan infective agents, namely, Malpighamoeba locustae, were encountered in day-to-day faecal matter collections from the host pests. The turnover was substantial in case of V. necatrix from Helicoverpa zea. The cost analysis report by McLaughlin and Bell (1970) presented a comparable account of protozoan organisms, viz., Mattesia grandis (McLaughlin) and N. gasti (McLaughlin), from an experimental report involving pest hosts, that is, cotton boll weevils, A. grandis. Schwalbe et al. (1974) contemplated production of spores of Melanoplus trogodermae to assert biocontrol of T. glabrum in the live environment to store the stored grains. To envisage the production of supernumerary instars of the host pest, the use of an insect growth regulator was suggested by Brooks (1980) to trigger production of spores/larvae en masse. The efforts to produce the protozoan, N. locustae Canning, from the commercial standpoint in grasshoppers, Melanoplus bivittatus, were recognized by the US Environment Protection Agency (EPA) (Henry and Oma 1981).
8.13.1.2 In Vitro Production
The procedures have been developed to date to grow suspension cultures or monolayers of cells for the enhanced output of a variety of microsporidia as well as to ensure simultaneously the availability of the spores of these microsporidia at the required period of intervals from the culture medium. But their status of product output as on to the level of cost-effectiveness could not be ensured. Several earlier investigators (Gupta 1964; Kurtii and Brooks 1971; Bayne et al. 1975; Sohi and Wilson 1976) utilized an already infected host resource to establish primary cell cultures of these microsporidia.
The uninfected cultures were maintained to introduce sterile harvested spores. It was also not necessary to grow the microsporidia or other cells that could only exhibit closer affiliation to their host cells, whether those of invertebrate or vertebrate origin. Primary cultures of silkworms, Bombyx mori, were used by Ishihara and Sohi (1966) and Ishihara (1969) for growth of N. bombycis Nageli (Table 8.6) and cell lines were used by Undeen (1975) for N. algerae in pig kidney cells by Kurtii and Brooks (1977) for N. disstriae and by Atwell et al. (1985) for H. zea cells. The findings of several earlier investigators did not find consistent growth and replication rates in cultures of different kinds, though they did not report comparisons with in vivo systems to record the rates of replication beyond a few days. An improvement in the technique for infection of cells in culture was introduced by Barker et al. (1980), who centrifuged the cultures when the spores were introduced. The close contact between spores and cells, during the process of polar filament eversion, gave a higher level of infection from which the parasites could spread to other cells. Sohi and Wilson (1976) found that spore production of N. disstriae was depressed in culture, although the numbers of parasites remained high. This would be a serious impediment to mass production, and a similar effect has been reported for cultures of N. algerae, in which spore production stopped after six passages (Streett et al. 1980).
8.13.2 Storage
The resistivity of the entities that participate in transmission of certain Protozoa has a greater role to play but the most convenient and safest form of these could be to keep the cysts or spores of these in dry state. Storage presents no insurmountable problem for Protozoa with resistant stages of transmission, but the simplest method of keeping the cysts or spores in the dry state can only be used for certain categories. Dry storage is essential to prevent extrusion of the polar filaments, which occurs automatically in water. The protrusion of polar filaments can be prevented by maintaining these in dry form, which otherwise is attained automatically in the moist state in N. whitei Weiser, a microsporidium of flour beetles Tribolium spp. No apparent loss of viability was observed by Milner (1972) in the spores of Tribolium spp., if stored in a dry flour–yeast mixture at 4 °C to the extent of 15 months. On the other hand, the exposure to even shorter periods of desiccation was fatal to some of the microsporidia. The dried spores of N. algerae lost their potential to infect the larvae of Anopheles stephensi Liston in a study by Alger and Undeen (1970). However, an optimal survival of most of the microsporidia and a fewer variety of neogregarines were attained when these were stored in a sterile water suspension kept at a temperature above freezing point. The spores of N. bombycis Nageli could survive viable for as long as up to a decade, as recorded by Oshima (1964). The treatment by antibiotics was applied to maintain viability of these microsporidia as it deteriorated rapidly under the influence of these contaminants. Contrary to this, the treatment by the required concentrations of antifungal mixtures, needed to deactivate yeasts or growth of hyphae, proved damaging to a larger chunk of the microsporidia in a manner that a span of storage of N. algarae for 5 weeks in natamycin, fungizone and nystatin resulted in a loss of their viability (Lai 1980). Simultaneously, appreciable issues of rearing to maintain insect colonies during conduct of experiments were faced when the antibiotics, like benomyl and fumagillin, were applied to keep check on the growth and spread of microsporidia (Vavra and Maddox 1976). The noticeable reduction in viability of microsporidian spores ensued at the higher temperatures that could incapacitate these in as much as that it was reduced in most species, to weeks, hours or minutes by temperatures that have risen to 40 °C (Maddox 1977). Considerable loss in viability of microsporidian spores of a large number of protozoan species, like N. locustae, resulted at lowered thermal regime (3 years in distilled water at −10 °C), as well as in case these were stored dry (Henry and Oma 1981). The imminent requirement to resolve the problem of storage of such magnitude was appreciated because these protozoans have been recognized as a workable biopesticide over the years by now. But the issues like availability of these biocontrol liquids in huge quantities for applications in the fields kept the farmers baffled, because of large areas under attack by grasshoppers and locusts, and simultaneously methods to be adopted for the delivery of these biopesticides over large farm areas were cumbersome. Thus, the riddles of cost-effectiveness kept the farmers away from immediate adoption of such measures, and instead these were adopted only in cases of extreme emergencies. However, the outcome of a large number of investigations (Kramer 1970a, b; Brooks 1980) illustrated a storage regime at the freezing temperature (i.e. 4 °C) was appropriately helpful for the growth of several species. The need has been felt to enhance adequate quantities of storage of N. locustae at the commercial scale. The methods of lyophilization and cryopreservation in liquid nitrogen, employed to achieve long-term viability (Vavra and Maddox 1976), were not found to be cost-effective because of the high cost of liquid nitrogen.
Later, however, some apprehensions were expressed about the loss of viability of spores to a small extent immediately followed by lyophilization, but the spores that survived the onslaught retained infectivity for longer periods. The loss of viability of a negligible magnitude of Nosema apis Zander post-lyophilization was concluded by Bailey (1972), and, therefore, its prolonged use for storage of viable microsporidia was recommended.
8.14 Future Prospects of Protozoans Biocontrol of Insect Pests
The pests, to the extent of 98%, are known to have been controlled by biocontrol agents. Certain ecological invaders comprising newer predators and parasitoids could conform to the significant constituents of biocontrol that regulate a variety of pests. According to the modern trend, the utilization of emerging strains, biotypes, parasitic hybrids, protozoan and fungal biopesticides required utmost attention for various applications in the management of pests. The principal beneficiaries of the application of protozoan control methods to regulate populations of pests have been the aquaculturists and farmers, particularly those dedicated to modernized aquacultural techniques, like cage culture etc., organic farming and other modern agricultural methods. Newer substitutes would be added day by day by the application of modernized researches to develop formulations of newer substances. Further advancements would target commercialization and adaptability of biopesticides. These efforts would also restrict cost escalation as more and more natural means of biocontrol become acceptable to the public at large. Once more and more advancements in the development of promising candidates among natural products emanated from the farmed products, the test of their efficacy and reliability in application could be ensured by extensive checks on the target pests in a variety of cropping systems.
It is a challenging task to conduct detailed investigations on biochemical pathways in the intracellular organisms that lack mitochondria but are the potential candidates to be employed in biocontrol of insect pests.
8.15 Conclusions
Microsporidia (Protists) belong to the significant group of biocontrol agents that had the potential to keep noticeable benefits of human populations conserved as against the potential of insect pest hosts that could keep the former divested of noticeable economic gains. The unique mechanism of invasion adopted by these pathogens incorporated the use of a specialized apparatus whose functional details are yet not completely available. They are important pathogens of economically important insects and animals. It is noticeable that considerable information is available to assign (i) vertical, (ii) horizontal, as well as (iii) transovarial transmission pathways in the dynamics of the life cycle of microsporidia. But additional information on their mode of entry into the cells of insect pests, along with the mechanism of its emergence post-replicative cycle, would provide solution to the riddle of incomprehensible process of impact on the pest host. The limitations of the excessive applicability of chemicals to control or kill the insect pests have indeed not led us too far in as much as to pave safer pathways down the lane. This is, in particular, due to the well-known adverse ill-effects on the environment, nontarget species as well as public health. This does not necessarily mean that only a fast action strategy could serve meaningful purpose, particularly in the wake of a virtual slow-action reactivity exhibited by most of the known protists (microsporidia). The elements of reliable non-chemical control method could safely have the potential to long-term suppression and maintenance of low densities of insect pests, like grasshoppers and locusts, and these could well be the critical elements of control by natural enemies in terms of application of protists as tools of biocontrol. The judicious mix of ecologically safe biocides utilizing microsporidian protozoans as its constituents, to replace hazardous uneconomical chemicals for control of insect pests, is propagated.
8.16 Points to Remember
-
Biocontrol elements in pathogenic interactions, other than non-chemical and other methods of control, encompassing the tiny protista (Microsporidia) and other protozoans comprise unique entomopathogenic organisms.
-
The invasion of these minute pathogens primarily in the form of cyst into the cells of insect pest hosts is a commonly known event. These infective cysts have the potential to survive over unfavourable conditions as well as overwinter to survive withering conditions, during seasonal change.
-
Pathogens have, of late, been recognized as suppressor of rapid breeding and feeding speed of acridid and a variety of other insect pest hosts, although their action was labelled initially as slow-paced. This could well be a successful biocontrol strategy.
-
The microsporidian invaders made inroads into the intracellular environment of insect pests through cysts. Amongst Nosema-type group of microsporidia, the transmittance of the flagellate, Trypanosoma sp., did not occur by direct ingestion of true cysts but through the ingestion of faecal material. Endoreticulatus group (Table 8.2) was differentiated from the other two, that is, Nosema type and Vairimorpha type, phylogenetically as well as in the status of pathogenicity.
-
The horizontal transmission of microsporidians is a unique pathway in which entry of cysts through eggs, diapausing larvae, pupae or non-feeding adults is attained. The alternative vertical mode facilitated invasion of microsporidians into the contents of the eggs that terminated the process at the hatching of larvae from the contaminated egg contents.
References
Alger NE, Undeen AH (1970) The control of a microsporidian Nosema sp., in an anopheline colony by an egg-rinsing technique. J Invertebr Pathol 15:321–327
Anderson RM, May RM (1980) Infectious diseases and population cycles of forest insects. Science 210:658–661
Anderson RM, May RM (1981) The population dynamics of microparasites within invertebrate populations. Philos Trans R Soc B 291:451–452
Anderson JR, Washburn JO, Egerter DE (1986a) Life cycle of the pathogen Lambornella clarki and its impact on Aedes sierrensis. Mosquito control research. Annual report. University of California, Davis, pp 20–21
Anderson JR, Washburn JO, Egerter DE (1986b) Mass production, storage and field release of Lambornella clarki, a pathogen of Aedes sierrensis. Mosquito Control Research. Annual Report. University of California, Davis, p. 21–22
Andreadis TG (1986) Dissemination of Nosema pyrausta in feral population of the European corn borer Ostrinia nubilalis. J Invertebr Pathol 48:335–343
Atwell BJ, Thomson CJ, Greenway H, Ward G, Waters I (1985) A study of the impaired growth of roots of Zea mays seedlings at low oxygen concentrations. Plant Cell Environ 8:179–188
Avery SW, Anthony DW (1983) Ultrastructural study of early development of Nosema algerae in Arropheles albimanus. J Invertebr Pathol 42:87
Bailey L (1972) The preservation of infective microsporidian spores. J Invertebr Pathol 20:252–254
Barker RJ, Smith J, Lai PF (1980) The culture of the microsporidian parasites Nosema algerae and N. eurytremae in vertebrate cells. J Protozool 27:55
Bartlett AH, Park PW (2010) Proteoglycans in host–pathogen interactions: molecular mechanisms and therapeutic implications. Expert Rev Mol Med 12:e5. https://doi.org/10.1017/S1462399409001367
Bayne CJ, Owczarzak A, Noonan WE (1975) In vitro cultivation of cells and a microsporidian parasite of Biomphalaria glabrata (Pulmonata, Basommatophora). Ann N Y Acad Sci 266:513–527
Becnel JJ, Johnson MA (2000) Impact of Edhazardia aedis (microsporidia : Culicosporidae). On a seminatural population of Aedes aegypti. (Diptera : Culicidae). Biol Control 18:39–48
Bidochka MJ, Khachatourians GG (1991) Review article: microbial and protozoan pathogens of grasshoppers and locusts as potential biocontrol agents. Biocontrol Sci Tech:243–259
Bohne W, Ferguson DJ, Kohler K, Gross U (2000) Developmental expression of a tandemly repeated, glycine-and serine-rich spore wall protein in the microsporidian pathogen Encephalitozoon cuniculi. Infect Immun 68:2268–2275. https://doi.org/10.1128/iai.68.4.2268-2275.2000
Bomar CR, Lockwood JA, Pomerinke MA, French JD (1993) Multiyear evaluation of the effects of Nosema ktcustae (Microsporida: Nosematidae) on rangeland grasshopper (Othoptera: Acrididae) population density and natural biological controls. Environ Entomol 22:489–491
Braun L, Ewen AB, Gillott C (1988) The life cycle of Malameba locustae (King and Taylor) (Amoebidae) in the migratory grasshopper Mektnoplus sanguinipes (F.) (Acrididae). Can Entomol 120:759–772
Brooks WM (1968) Transovarian transmission of Nosema heliothidis in the corn earworm Heliothis zea. J Invertebr Pathol 11:511–512
Brooks WM (1974) Chapter 5. Protozoan infections. In: Cantwell GE (ed) Insect diseases, vol 1. Marcel Dekker, New York, pp 237–300
Brooks WM (1979) Production and efficacy of protozoa. Biotechnol Bioeng 22:91–116
Brooks WM (1980) Production and efficacy of protozoa. Biotechnol Bioeng 22:1415–1440
Brooks WM (1988) Entomogenous protozoa. In: Ignoffo CM (ed) CRC handbook of natural pesticides. Microbial insecticides. Part A. Entomogenous protozoa and fungi, vol 5. CRC Press, pp 1–149
Brosson D, Kuhn L, Prensier G, Vivares CP, Texier C (2005) The putative chitin deacetylase of Encephalitozoon cuniculi: a surface protein implicated in microsporidian spore-wall formation. FEMS Microbiol Lett 247:81–90. https://doi.org/10.1016/j.femsle.2005.04.031
Bulla LA Jr, Cheng TC (1976) Comparative pathobiology, 1. In: Biology of the microsporidia. Plenum Press, New York
Burri L, Williams BA, Bursac D, Lithgow T, Keeling PJ (2006) Microsporidian mitosomes retain elements of the general mitochondrial targeting system. Proc Natl Acad Sci U S A 103:15916–15920. https://doi.org/10.1073/pnas.0604109103
Cali A, Becnel JJ, Takvorian PM (2017) Microsporidia. Hand Protists:1559–1618. https://doi.org/10.1007/978-3-319-28149-0_27
Canning EU (1953) A new microsporidian, Nosema locustae n. sp., from the fat body of the African migratory locust, Locusta migratorioides R & F. Parasitology 43:287–290
Canning EU (1962a) The life cycle of Nosema locustae Canning in Locusta migratoria migratorioides (Reiche & Fairmaire), and its infectivity to other hosts. J Insect Pathol 4:237–241
Canning EU (1962b) The pathogenicity of Nosema locustae Canning. J Insect Pathol 4:248–256
Canning EU (1982) An evaluation of protozoal characteristics in relation to biological control of pests. Parasitology 84:119–149
Canning EU, Lom J (1986) The microsporidia of the vertebrates. Academic Press Inc, New York City, NY, p 289
Canning EU Barker RJ (1981) Transmission of microsporidia between generations of winter moth Operophtera brumata. Parasitology (in press)
Cali A, Takvorian PM (1999) Developmental morphology and life cycles of the microsporidia. In: Wittner M, Weiss LM (eds) The microsporidia and microsporidiosis. AMS Press, Washington, DC, pp 85–128
Canning EU, Wigley PJ, Barker RJ (1983) The taxonomy of three species of microsporidia (Protozoa: Microspora) from an oakwood population of winter moths Operophtera brumata (L.) (Lepidoptera: Geometridae). Syst Parasitol 5:147–159
Cali A, Takvorian PM (2014) Developmental morphology and life cycles of the Microsporidia. In: Weiss LM, Becnel JJ (eds) Microsporidia-pathogens of opportunity. Wiley Blackwell Press, Hoboken NJ, pp 71–133. https://doi.org/10.1002/9781118395264.ch2
Capinera JL, Hibbard BE (1987) Bait formulations of chemical and microbial insecticides for suppression of crop-feeding grasshoppers. J Agric Entomol 4:337–344
Chen J, Geng L, Long M, Li T, Li Z, Yang D et al (2013) Identification of a novel chitin-binding spore wall protein (NbSWP12) with a BAR-2 domain from Nosema bombycis (microsporidia). Parasitology 140:1394–1402. https://doi.org/10.1017/S0031182013000875
Chen L, Li R, You Y, Zhang K, Zhang L (2017) A novel spore wall protein from Antonospora locustae (microsporidia: Nosematidae) contributes to sporulation. J Eukaryot Microbiol 64:779–791. https://doi.org/10.1111/jeu.12410
Clark TB, Brandl DG (1976) Observations on the infection of Aedes sierrensis by a tetrahymenine ciliate. J Invertebr Pathol 28:341–349
Corliss JO, Coats DW (1976) A new cuticular cyst-producing tetrahymenid ciliate, Lambornella clarki n.sp. and the current status of ciliatosis in culicine mosquitoes. Trans Am Microsc Soc 95:725–739
Dean P, Sendra K, Williams T, Watson A, Major P, Nakjang S, Kozhevnikova E, Goldberg AV, Kunji ERS, Hirt RP, Embley TM (2018) Transporter gene acquisition and innovation in the evolution of microsporidia intracellular parasites. Nat Commun 9:1709. https://doi.org/10.1038/s41467-018-03923-4
Dolgikh VV, Senderskiy IV, Pavlova OA, Naumov AM, Beznoussenko GV (2011) Immunolocalization of an alternative respiratory chain in Antonospora (Paranosema) locustae spores: mitosomes retain their role in microsporidial energy metabolism. Eukaryot Cell 10:588–593. https://doi.org/10.1128/EC.00283-10
Egeter DE, Anderson JR, Washburn JO (1986) Dispersal of the parasitic ciliate Lambornella clarki: implications for ciliates in the biological control of mosquitoes. Proc Natl Acad Sci U S A 83:7335–7339
Englund PT, Hajduk SL, Marini JC (1982) The molecular biology of trypanosomes. Annu Rev Biochem 51:695–726
Fine PEM (1981) Vertical transmission of pathogens of invertebrates. In: Bulla LA, Cheng TC (eds) Comparative pathobiology, vol 6, 178 pp
Fine PEM (1975) Vectors and vertical transmission: an epidemiologic perspective. Ann N Y Acad Sci 266:173–194
Flint ML, Dreistadt SH (eds) (1998) Natural enemies handbook: the illustrated guide to biological pest control. University of California, Division of Agriculture, National Research Publication, Oakland, p 3386
Franz JM, Huger AM (1971) Microsporidia causing the collapse of an outbreak of the green trotrix (Tortrix viridana L.) in Germany. Proc 4th Int Colloq Insect Pathol:48–53
Frixione E, Ruiz L, Santillán M, De Vargas LV, Tejero JM, Undeen AH (1992) Dynamics of polar filament discharge and sporoplasm expulsion by microsporidian spores. Cell Motil Cytoskeleton 22:38–50. https://doi.org/10.1002/cm.970220105
Gaugler RR, Brooks WM (1975) Sublethal effects of infection by Nosema heliothidis in the corn earworm, Heliothis zea. J Invertebr Pathol 26:57–63
Goertz D, Hoch G (2008) Vertical transmission and overwintering of microsporidia in the gypsy moth, Lymantria dispar. J Invertebr Pathol 99(1):43–48
Goldberg AV, Molik S, Tsaousis AD, Neumann K, Kuhnke G, Delbac F, Vivares CP, Hirt RP, Lill R, Embley TM (2008) Localization and functionality of microsporidian iron–sulphur cluster assembly proteins. Nature 452:624–628. https://doi.org/10.1038/nature06606
Gupta KS (1964) Cultivation of Nosema mesnili Paillot (microsporidia) in vitro. Curr Sci India 33:407–408
Han B, Weiss LM (2017) Microsporidia: obligate intracellular pathogens within the fungal kingdom. Microbiol Spectrum 5:97–113
Han B, Polonais V, Sugi T, Yakubu R, Takvorian PM, Cali A, Maier K, Long M, Levy M, Tanowitz HB, Guoging P, Delbac F, Zhou Z, Weiss LM (2017) The role of microsporidian polar tube protein 4 (PTP4) in host cell infection. PLoS Pathog 13:e1006341. https://doi.org/10.1371/journal.ppat.1006341
Han B, Ma Y, Tu V, Tomita T, Mayoral J, Williams T, Horta A, Huang H, Weiss LM (2019) Microsporidia interact with host cell mitochondria via voltage-dependent anion channels using sporoplasm surface protein 1. MBio 10:e001944–e001919. https://doi.org/10.1128/mBio.01944-19
Han B, Takyorian PM, Weiss LM (2020) Invasion of host cells by microsporidia. Front Microbiol Rev. https://doi.org/10.3389/fmicb.2020.00172
Hans-Peter Braun RB (2009) Mitochondria. Ed. Immo E. Scheffler. Q Rev Biol 84:103. https://doi.org/10.1086/598289
Harry OG (1970) Gregarina: their effect on growth in the desert locust (Schistocerca gregaria). Nature 225:964–966
Hayman JR, Hayes SF, Amon J, Nash TE (2001) Developmental expression of two spore wall proteins during maturation of the microsporidian Encephalitozoon intestinalis. Infect Immun 69:7057–7066. https://doi.org/10.1128/iai.69.11.7057-7066.2001
Hayman JR, Southern TR, Nash TE (2005) Role of sulfated glycans in adherence of the microsporidian Encephalitozoon intestinalis to host cells in vitro. Infect Immun 73:841–848. https://doi.org/10.1128/iai.73.2.841-848.2005
Hazard EI, Oldacre SVV (1976) Revision of microsporidia (protozoa) close to Thelohania with descriptions of one new family, eight new genera and thirteen new species. United States Department of Agriculture Technical Bulletin No. 1530, pp 1–104
Heckmann RA (2020) E-book: ISBN9780367811006, molecular biology of protozoa for biological control of harmful insects, Chapter 9. In: Gunasekharan M et al (eds) 1995, molecular biology of the biological control of pests and diseases of plants. Taylor & Francis, London, p 18. https://doi.org/10.1201/9780367811006
Heinz E, Williams TA, Nakjang S, Noël CJ, Swan DC, Goldberg AV, Harris SR, Weinmaier T, Markert S, Becher D, Bernhardt J, Dagan T, Hacker C, Lucocq JM, Schweder T, Rattei T, Hall N, Hirt RP, Embley TM (2012) The genome of the obligate intracellular parasite Trachipleistophora hominis: new insights into microsporidian genome dynamics and reductive evolution. PLoS Pathog 8:e1002979. https://doi.org/10.1371/journal.ppat.1002979
Heinz E, Hacker C, Dean P, Mifsud J, Goldberg AV, Williams TA, Nakjang S, Gregory A, Hirt RP, Lucocq JM, Kunji ERS, Embley TM (2014) Plasma membrane-located purine nucleotide transport proteins are key components for host exploitation by microsporidian intracellular parasites. PLoS Pathog 10:e1004547. https://doi.org/10.1371/journal.ppat.1004547
Henry JE (1969) Extension of the host range of Nosema locustae in Orthoptera. Ann Entomol Soc Am 62:452–453
Henry JE (1981) Natural and applied control of insects by protozoa. Annu Rev Entomol 26:49–73
Henry JE (1985) Effect of grasshopper species, cage density, light intensity, and method of inoculation on mass production of Nosema locustae (Microsporida: Nosematidae). J Econ Entomol 78:1245–1250
Henry JE (1990) Control of insects by protozoa. In: New directions in biological control: alternatives for suppressing agricultural pests and diseases. A.R. Liss, Inc, New York, pp 161–176, 837
Henry JE, Oma EA (1981) Protozoa: pest control by Nosema locustae, a pathogen of grasshoppers and crickets. In: Burges HD (ed) Microbial control of pests and plant diseases, 1976–1980. Academic Press, New York, pp 573–586
Hill RE, Gary WJ (1979) Effects of the microsporidium, Nosema pyrausta, on field populations of European corn borers in Nebraska. Environ Entomol 8:91–95
Hinks CF, Ewen AB (1986) Pathological effects of the parasite Malameba locustae in males of the migratory grasshopper Melanoplus sanguinipes and its interaction with the insecticide, cypermethrin. Entomol Erp Appl 42:39–44
Hudson B, Vinograd J (1967) Catenated circular DNA molecules in HeLa cell mitochondria. Nature 216(5116):647–652
Hylis M, Pilarska DK, Oborní KM, Vávra J, Solter LF, Weiser J, Linde A, ML MM (2006) Nosema chrysorrhoeae n. sp. (Microsporidia), isolated from browntail moth (Euproctis chrysorrhoea L.) (Lepidoptera, Lymantriidae) in Bulgaria: characterization and phylogenetic relationships. J Invertebr Pathol 91:105–114
Ishihara R (1969) The life cycle of Nosema bombycis as revealed in tissue culture cells of Bombyx mori. J Invertebr Pathol 14:316–322
Ishihara R, Sohi SS (1966) Infection of ovarian tissue culture of Bombyx mori by Nosema bombycis spores. J Invertebr Pathol 8:538–540
Jackson LL, Baker GL, Henry JE (1968) Effect of Malamoeba locustae infection on the egg lipids of the grasshopper Melanoplus hivitattus. J Insect Physiol 14:1773–1778
Jaroenlak P, Boakye DW, Vanichviriyakit R, Williams BA, Sritunyalucksana K, Itsathitphaisarn O (2018) Identification, characterization and heparin binding capacity of a spore-wall, virulence protein from the shrimp microsporidian. Enterocytozoon hepatopenaei (EHP). Parasit Vectors 11:177. https://doi.org/10.1186/s13071-018-2758-z
Johnson MA, Becnel JJ, Undeen AH (1997) A new sporulation sequence in Edhazardia aedis (Microsporidia: Culicosporidae), a parasite of the mosquito Aedes aegypti (Diptera: Culicidae). J Invertebr Pathol 70:69–75
Keithly JS, Langreth SG, Buttle KF, Mannella CA (2005) Electron tomographic and ultrastructural analysis of the Cryptosporidium parvum relict mitochondrion its associated membranes and organelles. J Eukaryot Microbiol 52:132–140
Knell JD, Allen GE (1978) Morphology and ultrastructure of Unikaryon minutum (Microsporidia: Protozoa), a parasite of the southern pine beetle, Dendroctonus frontalis. Acta Protozool 17:271–278
King RL, Taylor AB (1936) Malpighamoeba locustae sp. n. (Amoebidae), a protozoan parasitic in the malpighian tubes of grasshoppers. Trans Am Microscopists Soc 55:6–10
Koella JC, Sorensen FL, Anderson RA (1998) The malaria parasite, Plasmodium falciparum, increases the frequency of multiple feeding of its mosquito vector, Anopheles gambiae. Proc Biol Sci 265(1398):763–768. https://doi.org/10.1098/rspb.1998.0358
Kohlmayr B, Weiser J, Wegensteiner R, Handel U, Zizka Z (2003) Infection of Tomicus piniperda (Col., Scolytidae) with Canningia tomici sp. n. (Microsporidia, Unikaryonidae). Anz Schädlingskunde 76:65–73
Kramer JP (1959a) Some relationships between Perezia pyraustae Paillot (Sporozoa, Nosematidae) and Pyrausta nubilalis (Hiibner) (Lepidoptera, Pyralidae). J Insect Pathol 1:25–33
Kramer JP (1959b) Observations on the seasonal incidence of microsporidiosis in European corn borer populations in Illinois. Entomophaga 4:37–42
Kramer JP (1970a) Longevity of microsporidian spores with special reference to Octosporea muscaedomesticae flu. Acta Protozool 8:217–224
Kramer JP (1970b) Longevity of microsporidian spores with special reference to Octosporea muscae domesticae. Flu. Acta Protozool 8:127–135
Kurtii TJ, Brooks MA (1971) Growth of a microsporidian parasite in cultured cells of tent caterpillars (Malacosoma). Curr Top Microbiol Immunol 55:204–208
Kurtii TJ, Brooks MA (1977) The rate of development of a microsporidian in moth cell culture. J Invertebr Pathol 29:126–123
Kurtti TJ, Munderloh UG, Noda H (1990) Vairimorpha necatrix: infectivity for the development in a lepidopteran cell line. J Invertebr Pathol 55:61
Kyei-Poku G, Gauthier D, Schwarz R, van Frankenhuyzen K (2011) Morphology, molecular characteristics and prevalence of a Cystosporogenes species (Microsporidia) isolated from Agrilus anxius (Coleoptera: Buprestidae). J Invertebr Pathol 107:1–10
Lacey LA, Undeen AH (1986) Microbial control of blackflies and mosquitoes. Annu Rev Entomol 31:265–296
Lai PF (1980) Microsporidia in Schistosoma mansoni and their propagation in vivo and in vitro. Ph.D. thesis, University of London, p 252
Lange CE, Azzaro FG (2008) New case of long-term persistence of Paranosema locustae (microsporidia) in melanopline grasshoppers (Orthoptera: Acrididae) of Argentina. J Invertebr Pathol 99:357–359
Leonard CA, Hayman JR (2017) Role of host cell integrins in the microsporidium Encephalitozoon intestinalis adherence and infection in vitro. Fed Eur Microbio Soc Microbiol Lett 364:fnx169. https://doi.org/10.1093/femsle/fnx169
Levine ND, Corliss JO, Cox FEG, Deroux G, Grain J, Honigberg BM, Leedale GF, Loeblich AR, Lom J, Lynn D, Merinfeld EG, Page FC, Poljansky G, Sprague V, Vavra J, Wallace FG (1980) A newly revised classification of the protozoa. J Protozool 27:37–58
Li Y, Wu Z, Pan G, He W, Zhang R, Hu J, Zhou Z (2009) Identification of a novel spore wall protein (SWP26) from microsporidia Nosema bombycis. Int J Parasitol 39:391–398. https://doi.org/10.1016/j.ijpara.2008.08.011
Li Z, Pan G, Li T, Huang W, Chen J, Geng L et al (2012) SWP5, a spore wall protein, interacts with polar tube proteins in the parasitic microsporidian Nosema bombycis. Eukaryot Cell 11:229–237. https://doi.org/10.1128/EC.05127-11
Lipa JJ (1976) Microsporidians parasitizing the green tortrix in Poland and their role in the collapse of the tortrix outbreak in Puszcza Niepoilonicka during 1970–1974. Acta Protozool 1(5):529–536
Lom J, DyKova I (1992) Protozoan parasites of fishes. In: Developments in aquaculture and fisheries science, vol 26. Elsevier Publishers, New York, pp 125–154
Lukes J, Guilbride DL, Votypka J, Zikova A, Benne R, Englund PT (2002) Kinetoplast DNA network: evolution of an improbable structure. Eukaryot Cell 1(4):495–502
Maddox JV (1973) The persistence of the microsporidia in the environment. Misc Publ Entomol Soc Am 9:99–104
Maddox JV (1977) Stability of entomopathogenic protozoa. Misc Publ Entomol Soc Am 10:3–18
Maddox JV (1987) Protozoan diseases. In: Fuxa James R, Tanada Y (eds) Epizootiology of insect diseases. John Wiley & Sons, New York, pp 417–452
Maddox JV, Solter L (1996) Infective microsporidian spores in liquid nitrogen. J Eukaryot Microbiol 43(3):221–225. https://doi.org/10.1111/j.1550-7408.1996.tb01395.x
Maddox JV, Brooks WM, Fuxa JR (1981) Vairimorpha necatrix a pathogen of agricultural pests: potential for pest control. In: Burges HD (ed) Microbial control of pests and plant diseases 1976–1980. New York, Academic Press, pp 587–594
Maddox JV, McManus ML, Solter LF (1998) Microsporidia affecting forest Lepidoptera. In: McManua ML, Liebhold AM (eds) Proceedings of population dynamics, impacts, and integrated management of forest defoliating insects, USDA Forest Service general technical report NE-247, pp 187–197
Maddox JV, Baker MD, Jeffords MR, Kuras M, Linde A, Solter LF, McManus ML, Vavra J, Vossbrinck CR (1999) Nosema Portugal, n.sp., isolated from gypsy moth (Lymantria dispar L.) collected in Portugal. J Invertebr Pathol 73:1–14
Major P, Sendra KM, Dean P, Williams TA, Watson AK, Thwaites DT, Embley TM, Hirt RP (2019) A new family of cell surface located purine transporters in microsporidia and related fungal endoparasites. elife 8:e47037. https://doi.org/10.7554/eLife.47037
Malone LA (1990) In vitro spore hatching of two microsporidia, Nosema costelytrae and Vavraiaon coperae, from New Zealand pasture insects. J Invertebr Pathol 55:441–443. https://doi.org/10.1016/0022-2011(90)90091-j
Martin PA, Johnson DL, Forsyth DJ, Hill BD (1997) Indirect effects of the pyrethroid insecticide, deltamethrin on reproductive success of chestnut-collared longspurs. Ecotoxicology 7:89–97
Mclaughlin RE (1971) Use of protozoa for microbial control of insects. In: Burges HD et al (eds) Microbial control of insects and mites. Academic Press, New York, NY, p 861
McLaughlin RE (1973) Protozoa as microbial control agents. Misc Publ Entomol Soc Am 9:95–98
McLaughlin RE, Bell MR (1970) Mass production in vivo of two protozoan pathogens, Mattesia grandis and Glugea gasti, of the boll weevil, Anthonomis grandis. J Invertebr Pathol 16:84–88
McManus ML, Solter L (2003) Microsporidian pathogens in European gypsy moth populations. In: Proceedings: ecology, survey, and management of forest insects. USDA Forest Service, Northeast Research Station Gen Tech Rep: NE-311, pp 44–51
Micieli MV, Garcia JJ, Andreadis TG (2009) Factors affecting horizontal transmission of the microsporidium Amblyospora albifasciati to its intermediate copepod host Mesocyclops annulatus. J Invertebr Pathol 101:228–233
Milner RJ (1972) Nosema whitei, a microsporidan pathogen of some species of Tribolium: III. Effect on T. castaneum. J Invertebr Pathol 19:248–255. https://doi.org/10.1016/0022-2011(72)90216-9
Munderloh VG, Kuritti TJ, Ross SE (1990) Electrophoretic characterizations of chromosomal DNA from two microsporidia. J Invertebr Pathol 55:243
Mussgnug GL (1980) Integration of Nosema locustae with chemical insecticides and entomopox virus for control of grasshoppers. Ph.D. thesis, Montana State University, p 133
Nordin GL, Maddox JV (1974) Microsporida of the fall webworm Hyphantria cunea. 1. Identification, distribution, and comparison of Nosema sp. with similar Nosema spp. from other Lepidoptera. J Invertebr Pathol 24(1–1):3
Oshima K (1964) Method of gathering and purifying active spores of Nosema bombycis and preserving them in good condition. Annotationes Zoologicae Japonenses 37:94–101
Paillot A (1927) Sur deux protozoaires nouveaux parasites des chenilles de Pyrausta nubilalis Hubner. CR Acad Sci Paris 185:673–675
Patti JM, Allen BL, Mcgavin MJ, Höök M (1994) MSCRAMM-mediated adherence of microorganisms to host tissues. Annu Rev Microbiol 48:585–617
Peuvel-Fanget I, Polonais V, Brosson D, Texier C, Kuhn L, Peyret P et al (2006) EnP1 and EnP2, two proteins associated with the Encephalitozoon cuniculi endospore, the chitin-rich inner layer of the microsporidian spore wall. Int J Parasitol 36:309–318. https://doi.org/10.1016/j.ijpara.2005.10.005
Pickford R, Randell RL (1969) A non-diapause strain of the migratory grasshopper, Melanoplus sanguinipes (Orthoptera: Acrididae). Can Entomol 101:894–896
Polonais V, Mazet M, Wawrzyniak I, Texier C, Blot N, El Alaoui H et al (2010) The human microsporidian Encephalitozoon hellem synthesizes two spore wall polymorphic proteins useful for epidemiological studies. Infect Immun 78:2221–2230. https://doi.org/10.1128/IAI.01225-09
Purrini K (1975) On the distribution of microorganisms infecting the larvae of Euproctis chrysorrhoea L. (Lep., Lymantriidae) near Kosova, Yugoslavia. Anz Schädlingskde Pflanzenschutz Umweltschutz 48:182e183
Purrini K, Weiser J (1975) Natural enemies of Euproctis chrysorrhoea in orchards in Yugoslavia. Anz Schädlingskde Pflanzenschutz Umweltschutz 48:11–12
Purrini K, Weiser J (1985) Ultrastructural study of the microsporidian Chytridiopsis typographi (Chytridiopsida: Microspora) infecting the bark beetle, Ips typographus (Scolytidae: Coleoptera), with new data on spore dimorphism. J Invertebr Pathol 45:66–74
Quednau FW (1968) Natural control of larch sawfly at Carleton Bonaventure County, Quebec Province, Canada. (Thelohania, Pristophorae). Can Dept Forest Rural Develop Bi-Mon Res Notes 24:14
Raina SK, Rai MM, Khurad AM (1987) Grasshopper and locust control using microsporidian insecticides. In: Maramorosch K (ed) Biotechnology in invertebrate pathology and cell culture. Academic Press, New York, pp 345–365
Raina SK, Ewen AB (1979) In vitro infection of the fat body cells of the grasshopper Melanoplus sanguinipes (Fab.) by the microsporidian Nosema locustae Canning. In: Proceedings of the Vth international conference on invertebrate tissue culture, developments and applications. Rigi Kalbad, Switzerland
Schwalbe CP, Burkholder WE, Boush GM (1974) Mottesia trogodermae infection rates as influenced by mode of transmission, dosage and host species. J Stored Prod Res 10:161–166
Shapas TJ, Burkholder WE, Boush GM (1977) Population suppression of Trogoderma glabrum by using pheromone luring for protozoan pathogen dissemination. J Econ Entomol 70:469–474
Siegel JP, Maddox JV, Ruesink WG (1988) Seasonal progress of Nosema pyrausta in the European corn borer, Ostrinia nubilalis. J Invertebr Pathol 52:130–136
Smirnoff WA (1966) Thelohaniu pristiphorae sp. n., a microsporidian parasite of the larch sawfly, Prktiphorn erichsonii (Hymenoptera: Tenthredinidae). J Invertebr Pathol 8:360–364
Smirnoff WA (1967) Diseases of the larch sawfly, Pristiphora erichsonii, in Quebec. J Invertebr Pathol 10:417–424
Sneller VP (1979) Inhibition of Dirofilaria immitis in gregarine-infected Aedes aegypti: preliminary observations. J Invertebr Pathol 34:62–70
Sohi SS, Wilson GG (1976) Persistent infection of Malacosoma disstria (Lepidoptera, Lasiocampidae) cell cultures with Nosema (Glugea) disstriae (microsporidia: Nosematidae). Can J Zool 54:336–342
Southern TR, Jolly CE, Lester ME, Hayman JR (2007) EnP1, a microsporidian spore wall protein that enables spores to adhere to and infect host cells in vitro. Eukaryot Cell 6:1354–1362. https://doi.org/10.1128/ec.00113-07
Steinhaus EA (1951) Report on diagnoses of diseased insects 1944–1950. Hilgardia 20:629–678
Streett DA, Ralph D, Hink WF (1980) Replication of Nosema algerae in three insect cell lines. J Protozool 27:113–117
Tachezy J (2019) Hydrogenosomes and mitosomes: mitochondria of anaerobic eukaryotes. Springer, Berlin
Takyorian P, Weiss L, Cali A (2005) The early events of Brachiola algerae (microsporidia) infection: spore germination, sporoplasm structure, and development within host cells. Folia Parasitol 52:118–129
Takyorian PM, Buttle KF, Mankus D, Mannella CA, Weiss LM, Cali A (2013) The multilayered interlaced network (min) in the sporoplasm of the microsporidium anncaliia algerae is derived from golgi. J Eukaryot Microbiol 60:166–178. https://doi.org/10.1111/jeu.12019
Tanada Y (1964) Incidence of microsporidiosis in field population of the armyworm, Pseudaletia unipuncta (Haworth). Proc Hawaiian Entomol Soc 3:435
Tanada Y (1976) Epizootiology and microbial control. In: Bulla LA Jr, Cheng TC (eds) Biology of the microsporidia, vol 1. Plenum, New York, pp 127–135
Tanada Y, Chang GY (1962) An epizootic resulting from a microsporidian and two virus infections in the army-worm, Pseudaletia unipuncta (Haworth). J Insect Pathol 4:129–131
TerHorst CP (2010) Evolution in response to direct and indirect ecological effects in pitcher plant inquiline communities. Am Nat 176:675–685
TerHorst CP (2011) Experimental evolution of protozoan traits in response to interspecific competition. J Evol Biol 24:36–46
Thomson HM (1955) Perezia fumiferanae n. sp., a new species of microsporidia from the spruce budworm Choristoneura fumiferana (Clem.). J Parasitol 41:416–423
Thomson HW (1958) The effect of a microsporidian parasite on the development, reproduction and mortality of the spruce budworm, Choristoneura fumiferana (Clem). Can J Zool 36:499–511
Thomson HM (1959) A microsporidian parasite of the forest tent caterpillar Malacosoma disstria Hbn. Can J Zool 37:217–221
Tovar J, Fischer A, Clark CG (1999) The mitosome, a novel organelle related to mitochondria in the mitochondrial parasite Entamoeba histolytica. Mol Microbiol 32:1013–1021. https://doi.org/10.1046/j.1365-2958.1999.01414.x
Tovar J, León-Avila G, Sánchez LB, Sutak R, Tachezy J, Van Der Giezen M, Hernandez M, Muller M, Lucocq JM (2003) Mitochondrial remnant organelles of giardia function in iron-sulphur protein maturation. Nature 426:172. https://doi.org/10.1038/nature01945
Tsaousis AD, Kunji ER, Goldberg AV, Lucocq JM, Hirt RP, Embley TM (2008) A novel route for ATP acquisition by the remnant mitochondria of Encephalitozoon cuniculi. Nature 141:553. https://doi.org/10.1038/nature06903
Undeen AH (1975) Growth of Nosema algerae in pig kidney cell cultures. J Protozool 22:107–110
Van Denburgh RS, Burbutis PB (1962) The host parasite relationship of the European corn borer, Ostrinia nubilalis, on the protozoan, Perezia pyraustae, in Delaware. J Econ Entomol 55:65–67
Van der Paauw (1990) The microsporidia and microsporidiosis. In: Wittner M, Weiss LM (eds) American Soc Microbiol, Washington, DC, USA, 1999, pp 129–171
van der Paauw H, Johnson DL, Maiga B (1990) Evaluation of a large-scale application of Nosema locustae bait against grasshoppers in the Malian Sahel. Report to the U.S. Agency for International Development, Service Nationale de la Protection des Vegetaux, Mali, and Ciba-Geigy AG, Basel, p 116
Van Frankenhuyzen K, Ebling P, McCron B, Ladd T, Gauthier D, Vossbrinck C (2004) Occurrence of Cystosporogenes sp. (Protozoa: Microsporidia) in a multispecies insect production facility and its elimination from a colony of the eastern spruce budworm, Choristoneura fumiferanae (Clem.) (Lepidoptera: Tortricidae). J Invertebr Pathol 87:16–28
Vavra J, Larsson JIR (2014) Structure of the microsporidia. In: Becnel LM, Weiss JJ (eds) Microsporidia pathogens of opportunity. Wiley Blackwell, Oxford, pp 1–70. https://doi.org/10.1002/9781118395264.ch1
Vavra J, Maddox JV (1976) Methods in microsporidiology. In: Bulla LA et al (eds) Comparative pathobiology, biology of the microsporidia, vol 1, pp 281–231
Wang Y, Dang X, Ma Q, Liu F, Pan G, Li T et al (2015) Characterization of a novel spore wall protein NbSWP16 with proline-rich tandem repeats from Nosema bombycis (microsporidia). Parasitology 142:534–542. https://doi.org/10.1017/S0031182014001565
Wallace FG (1979) Biology of the Kinetoplastida of arthropods. In: Lumsden WHR et al (eds) Biology of the Kinetoplastida, vol 2, pp 213–240
Waraich NF, Jain S, Colloms SD, Stark WM, Burton NP, Maxwell A (2020) A novel decatenation assay for DNA topoisomerases using a singly-linked catenated substrate. Biotechniques. https://doi.org/10.2144/btn-2020-0059
Washburn JO, Anderson JR (1986) Distinction of Lambornella clarki (Ciliophora: Tetrahymenidae) and other mosquito parasites in California treeholes. J Invertebr Pathol 48:296–309
Watts JG, Huddleston EW, Owens JC (1982) Rangeland entomology. Annu Rev Entomol 27:283–311
Wegensteiner R, Weiser J, Führer E (1996) Observations on the occurrence of pathogens in the bark beetle Ips typographus L (Col., Scolytidae). J Appl Entomol 120:199–204
Weidner E (1970) Ultrastructural study of microsporidian development. 1. Nosema sp. Sprague, 1965 in Callinectes sapidus Rathbun. Z Zellforsch 105:33–54
Weidner E (1972) Ultrastructural study of microsporidian invasion into cells. Z Parasitenkd 40:227–242
Weiser J (1969) Phylum microsporida sprague. In: Lee JJ, Hunter SH, Boree EC (eds) Illustrated guide to the protozoa. Allen Press, Lawrence, KS, pp 375–383
Weiser J (1970) Three new pathogens of the Douglas fir beetle, Dendroctonus pseudotsugae: Nosema dendroctoni n. sp., Ophryocystic dendroctoni n. sp., and Chytridiopsis typographi n. comb. J Invertebr Pathol 16:436–441
Weiser J (1971) Three new pathogens of the Douglas fir beetle, Dendroctonus pseudotsugae: Nosema dendroctoni n. sp., Ophryocystic dendroctoni n. sp., and Chytridiopsis typographi n. comb. J Invertebr Pathol 16:436–441
Weiser J, Veber J (1975) Die mikrosporidie Thelohania hyphantriae Weiser des Weissen barenspinner (Hyphantria cunea) und anderer mitghiden seiner luaconose. Z Angew Ent 40:55–70
Weiser J, Wegensteiner R, Zizka Z (1997) Ultrastructures of Nosema typographi Weiser 1955 (Microspora: Nosematidae) of the bark beetle Ips typographus L. (Coleoptera; Scolytidae). J Invertebr Pathol 70:156–160
Weiser J, Holusa J, Zizka Z (2006) Larssoniella duplicati n.sp. (Microsporidia, Unikaryonidae), a newly described pathogen infecting the double-spined spruce bark beetle, Ips duplicatus (Coleoptera, Scolytidae) in the Czech Republic. J Pest Sci 79:127–135
Williams BAP, Hirt RP, Licocq JM, Embley M (2002) A mitochondrial remnant in the microsporidian Trachipleistophora hominis. Nature 418:865–869
Williams BA, Dolgikh VV, Sokolova YY (2014) Microsporidian biochemistry and physiology. In: Weiss LM, Becnel JJ (eds) Microsporidia: pathogens of opportunity. Wiley Blackwell, Hoboken, NJ, pp 245–260. https://doi.org/10.1002/9781118395264.ch9
Wilson GG (1975) Occurrence of Thelohania sp. and Pleistophora sp. (Microsporida: Nosematidae) in Choristoneura fumiferana (Lepidoptera: Tortricidae). Can J Zool 53:1799–1802
Wilson GG, Burke JM (1971) Nosema thomsoni n. sp., a microsporidian from Choristoneura conflictana (Lepidoptera: Tortricidae). Can J Zool 49:786e788
Wilson GG, Burke JM (1978) Microsporidian parasites of Archips cerasivoranus (Fitch) in the district of Algoma, Ontario. Proc Entomol Soc Ontario 109:84e85
Wu Z, Li Y, Pan G, Tan X, Hu J, Zhou Z et al (2008) Proteomic analysis of spore wall proteins and identification of two spore wall proteins from Nosema bombycis (Microsporidia). Proteomics 8:2447–2461. https://doi.org/10.1002/pmic.200700584
Wu Z, Li Y, Pan G, Zhou Z, Xiang Z (2009) SWP25, a novel protein associated with the Nosema bombycis endospore. J Eukaryot Microbiol 56:113–118. https://doi.org/10.1111/j.1550-7408.2008.00375.x
Xu J, Pan G, Fang L, Li J, Tian X, Li T, Zhou Z, Xiang Z (2006) The varying microsporidian genome: existence of long terminal repeat retrotransposon in domesticated silkworm parasite Nosema bombycis. Int J Parasitol 36:1049–1056
Yang D, Dang X, Peng P, Long M, Ma C, Qin JJG et al (2014) NbHSWP11, a microsporidia Nosema bombycis protein, localizing in the spore wall and membranes, reduces spore adherence to host cell BME. J Parasitol 100:623–633. https://doi.org/10.1645/13-286.1
Zimmack HL, Brindley TA (1957) The effect of the protozoan parasite Perezia pyraustae Paillot on the European corn borer. J Econ Entomol 50:637–640
Yang D, Pan G, Dang X, Shi Y, Li C, Peng P et al (2015) Interaction and assembly of two novel proteins in the spore wall of the microsporidian species Nosema bombycis and their roles in adherence to and infection of host cells. Infect Immun 83:1715–1731. https://doi.org/10.1128/IAI.03155-14
Zimmack HL, Arbuthnot KD, Brindley TA (1954) Distribution of the European corn borer parasite Perezia pyraustae and its effect on the host. J Econ Entomol 47:641–645
Zuk M (1987) The effects of gregarine parasites on longevity, weight loss, fecundity and developmental time in the field crickets Gryllus veletis and G. pennsybanucus. Ecol Entomol 12:349–354
Acknowledgements
AY is thankful to The Principal, and Dr. (Mrs.) Neerja Kapoor, Head of the Department of Zoology, C.M.P.P.G. College, Allahabad, for facilities and interactions on the manuscript. NJ is grateful to Head of the Department of Zoology, Baba Bhimrao Ambedkar University, Lucknow, for facilities.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 The Author(s), under exclusive license to Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Yadav, A., Jaiswal, N., Malviya, S., Malhotra, S.K. (2021). Entomopathogenic Protozoa. In: Omkar (eds) Microbial Approaches for Insect Pest Management. Springer, Singapore. https://doi.org/10.1007/978-981-16-3595-3_8
Download citation
DOI: https://doi.org/10.1007/978-981-16-3595-3_8
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-16-3594-6
Online ISBN: 978-981-16-3595-3
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)