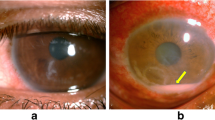
Abstract
Bacterial keratitis remains a leading cause of visual impairment and global blindness. Major risk factors include trauma, underlying ocular surface health, and contact lens wear. Etiological agents differ by geographic regions, populations sampled, antibiotic exposure, prior surgery, and laboratory workup. Nevertheless, overall pathogen spectrum has remained similar over the last 25 years, with gram-positive organisms Staphylococci (S. aureus, S. epidermidis) and Streptococci (S. pneumoniae) being the most frequently recovered and reported microorganisms worldwide. Pseudomonas aeruginosa remains the most frequently recovered and reported gram-negative pathogen among contact lens wearers and noncontact lens wearers.
Current treatment recommendations include initial therapy with a fluoroquinolone (ofloxacin, levofloxacin, moxifloxacin, gatifloxacin), followed by combination/fortified therapy, guided by laboratory workup in case of failure or nonresponsiveness. Increasing fluoroquinolone resistance among ocular isolates worldwide may require a reevaluation of this recommendation for empirical and initial therapy with this class of antibiotic.
Despite the ability of topical antibiotics to achieve high tissue concentrations to eradicate the invading organism, keratitis outcomes remain poor, complicated by corneal melting, scarring, perforation, and additional surgery (corneal transplant). A new paradigm is needed.
Improved clinical and laboratory diagnostic tools are required to augment more accurate and faster laboratory turnaround time in patients presenting with bacterial keratitis. Clinicians are looking towards artificial intelligence and advanced imaging tools to assist them in making a more definitive and quicker diagnosis, while microbiologists and researchers are increasingly using advanced molecular tools to assist them in detection and identification of common and unique pathogens.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
6.1 Introduction
Bacterial keratitis is an infection of the cornea initiated by the invasion and multiplication of bacteria [1]. It is a leading cause of corneal ulceration, opacification, visual morbidity, and blindness and remains a major healthcare burden worldwide [2]. The true burden is not known. Estimates in the United States range from 25,000 to 71,000 cases annually, while global rates may exceed 2.0–3.5 million cases each year [2, 3]. These rates may not reflect the true prevalence. These estimates are based on data collected and analyzed for unique populations more than 15 years ago.
The main function of the cornea is to bend, refract, and focus light. It must remain clear and free of scars to accomplish this task [1, 4]. Although constantly exposed to environmental insults, including microbes, the cornea is well protected by anatomical and local ocular surface epithelial, tear film, and immune defenses [5]. These work together to prevent microbial adherence and invasion. Once there is a breach in the ocular surface immune defense via trauma, contact lens wear, and topical antibiotics/steroids, any organism may gain access, multiply, and establish sight-threatening disease [1, 4, 5]. Active invasion of the corneal epithelium and stroma is considered an ocular emergency. The etiological agent and appropriate antimicrobial therapy must be rapidly identified and implemented to preserve vision [1].
6.2 Etiology
Bacteria are the most common cause of infectious keratitis [1, 4, 6]. Gram-positive organisms, Staphylococci, Streptococci, and Corynebacterium, constitute greater than 70% of reported corneal ulcers worldwide. Pseudomonas species and other gram-negative rods are more often associated with contact lens, surgery, and/or trauma [2, 6, 7].
Etiology may differ by geography, patient populations, culture frequency, and laboratory expertise and practice. In a recent meta-analysis by Teweldemedhin et al., coagulase-negative staphylococci (4.4–66.6%), Pseudomonas aeruginosa (6.8–55%), and Staphylococcus aureus (2–22.4%) were identified as the most commonly reported culture-derived corneal pathogens [8]. Streptococcus pneumoniae (1.3–24.7%) and Streptococcus viridans (0.4–14.3%) were also among the top five reported pathogens [8].
6.3 Risk Factors
Contact lens wear is the most common risk factor for bacterial keratitis in urbanized populations (Table 6.1) [3]. In the United States, 41 million patients are at risk [3]. Contact lens wear risks include trauma in the form of mini-abrasions, ocular surface integrity interruption (hypoxia, tear film disruption), and changes to the ocular surface microbiota [1, 9]. Forty percent of the culture-proven cases have been associated with sleeping and/or swimming in contact lens [3, 8].
Other risk factors associated with this population include trauma with contaminated water, soil, and vegetative matter [1, 4]. Exposure to contaminated soil, plant matter, and water can deliver heavy microbial loads, foreign bodies, and a diverse microbiota to the ocular surface including Pseudomonas aeruginosa, Stenotrophomonas maltophilia, and Achromobacter xylosoxidans. This group of organisms has a large arsenal of virulence factors (biofilm formation, toxins, proteases, resistance genes) that aid in virulence, invasion, and drug resistance [10,11,12].
Disruption of the ocular surface integrity via surgery, keratoprosthesis placement, and/or trauma provides easy access of microorganisms to corneal tissue and disruption of the tear film [1, 4]. Both mechanisms predispose to bacterial invasion and disease. Chronic ocular surface disorders increase the risk of tear film deficiency, altered immune defenses (immunosuppression, steroids), and ocular microbiota (microbiome) dysbiosis (antibiotics, anesthetics, steroids) which lead to increased risk of bacterial infection [1, 4].
In nonurbanized populations, environmental/agrarian trauma, chronic ocular surface disease, malnutrition, and poor and/or delayed access to appropriate ophthalmic care are the major predisposing factors for bacterial keratitis and corneal opacification/blindness [2, 4].
6.4 Role of Ocular and Gut Dysbiosis in Bacterial Keratitis
The ocular surface microbiota plays an important role in the protection of the ocular surface from invasion by opportunistic and/or true bacterial pathogens [13, 14]. Core members include Firmicutes (predominantly, Staphylococci, Streptococci), Actinobacteria (Corynebacteria), and Proteobacteria (Pseudomonas, Haemophilus, Neisseria). They engage in crosstalk with the corneal and conjunctival epithelial cells to provide and coordinate immune, spatial, and chemical protection of the ocular surface [13, 15]. The major outcomes from this partnership include: (a) barrier preservation, (b) inhibition of inflammation, (c) accelerated tissue repair, and (d) exclusion of pathogens.
Increasing metagenomic studies support this role [5]. Dysbiosis of the ocular microbiome by contact lens wear, instillation of topical antibiotics, surgery, and corticosteroid use can lead to increase susceptibility to bacterial virulence and invasion [15, 16].
As the ocular surface is part of the systemic mucosal system, it is in communications with and impacted by the health and dysbiosis of this organ [14, 17]. Kugadas et al. demonstrated in mice that there was an interconnection between gut and ocular surface dysbiosis and increased susceptibility to bacteria (Pseudomonas) induced keratitis [18]. In health, the ocular surface microbiota strengthens the ocular innate immune barriers by significantly increasing the concentration of immune effectors in the tear film. When depleted by administration of gentamicin, the normally resistant Swiss Webster mice became susceptible to the infection. Both the ocular and the gut microbiota contributed to the maintenance of the barrier protection with the ocular surface having a moderate but significant effect [18].
Jayasudha and colleagues also documented a connection between gut dysbiosis and susceptibility to bacterial keratitis [19]. In a recent study, they compared the fecal samples from normal controls and patients with bacterial keratitis and found a distinct difference in the relative abundance of protective anti-inflammatory genera in the gut versus microbial recovered from patients with bacterial keratitis [19].
Taken together, ocular microbiome dysbiosis may be an important unrecognized and underappreciated risk factor in bacterial keratitis.
6.5 Bacterial Keratitis in Children
Bacterial keratitis is a rare condition in children, with children contributing <15% of all cases [20,21,22]. The condition is difficult to treat in this population, as children may not be able to convey symptoms accurately and cooperation for evaluation and treatment may be limited. Additionally, there is a high risk of poor visual outcomes in keratitis, not only due to the disease itself but also due to the sequelae of corneal scarring leading to deprivational and/or anisometropic amblyopia.
The mean age for children presenting with bacterial keratitis typically ranges between 10 and 13 years old with a relatively even distribution between males and females [20,21,22,23,24,25]. Although these patient demographics have remained stable over time, the risk factors for bacterial keratitis in children have changed and vary with geographic location. Studies published in the 1980s and 1990s found that the main risk factor for bacterial keratitis was trauma [25, 26], whereas more recent publications indicate that contact lens wear is the most common factor contributing 40.7–77.6% of cases [20, 21, 23]. Geographic location also appears to affect risk, as in contrast to these recent studies from the United States and Taiwan, studies from Brazil and Mexico found that ocular trauma remains the most common risk factor [25, 27]. It is possible that the age of the patient population plays a role in the difference, as one study of 81 eyes reported trauma and ocular disease were significantly more common in children <12 years old whereas contact lens wear was more common in older children [20].
The causative organism in bacterial keratitis in children differs based upon the most common risk factor in a given patient population. In the studies that reported contact lens wear as the most common risk factor, Pseudomonas aeruginosa was the most common organism representing 30.6–46.2% of isolates [20, 21, 23]. In studies where trauma was the most common risk factor, gram-positive organisms such as coagulase-negative Staphylococcus [24] or Staphylococcus epidermidis [25, 27] represented the majority (23.4–28.6%) of organisms isolated.
Regardless of the pathologic organism, studies published in the 1990s reported that surgical intervention for eye salvage or visual rehabilitation would be indicated in 14–28% of pediatric keratitis cases [22, 26]. For instance, Cruz et al. found that 7 of 51 eyes (14%) required surgery [26]. However, more recent studies showed this percentage to be much lower, ranging from 0 to 6% [23, 27, 28]. In a series of 107 cases, Rossetto et al. found that zero penetrating keratoplasties were performed to treat perforation [23].
Overall, bacterial keratitis in children remains a potentially serious cause of ocular morbidity. The trend towards increasing incidence in older children who wear contact lenses is likely a reflection of global management of refractive error. As the prevalence of myopia is increasing, contact lens use is more prevalent and techniques such as orthokeratology are being employed more frequently. Future studies investing this trend will be useful.
6.6 Pathology
The ocular surface is well protected against microbial insult and invasion [4, 5]. Local immune defenses of the tight epithelial barrier coupled with the wide array of antimicrobial substances in the tear film and a stable microbiome help to maintain ocular surface integrity and health [5, 13, 17]. Disturbance or disruption of any of these can lead to imbalance and ocular surface disease including bacterial keratitis. Any organism gaining access to corneal tissue can establish disease.
In general, bacterial keratitis progress in stages [1]. These include infiltration, ulceration, regression, and healing. Patient outcome for all four stages is dependent on the bacterial species, ocular surface health, host defense response, diagnostic (microbiological, clinical) accuracy, and rapid, appropriate therapy [1, 4].
6.7 Clinical Assessment
6.7.1 Signs and Symptoms
There is no consensus of characteristics that identify infectious keratitis as bacterial in origin [1, 4, 27]. Signs and symptoms are dependent on a combination of the ocular surface health, invading pathogen and host defenses and response. Some common and suggestive signs and symptoms may include conjunctival injection and chemosis, decreased vision, pain, photophobia, tearing, and purulent discharge [1, 4]. A detailed history coupled with a complete ophthalmic examination is the foundation for an accurate and informative clinical assessment in the differential diagnosis of bacterial keratitis [1, 4, 21].
6.7.2 History
The history checklist should include onset of symptoms, recent and past ocular trauma, contact lens wear, and associated activities such as swimming, sleeping, lens changing schedule, and cleaning regimen [1]. In addition, information on previous eye disorders, ocular surgeries, and medications should be recorded. An updated medical history including allergies, systemic medications, family history, and organ system review should also be obtained [1].
6.7.3 Physical Examination
A thorough physical examination checklist should include slit-lamp photos and assessments for vision, intraocular pressure, and pupil evaluation [1, 4]. The location, size, and depth of the corneal infiltrate plus the anterior chamber reaction (cells, flare, fibrin, or hypopyon) must be recorded. Additional predisposing factors for infections such as foreign bodies, blepharitis, entropion, trichiasis, or lagophthalmos should be documented [1, 4].
6.7.4 Laboratory Assessment
Ideally, all corneal ulcers should be cultured for definitive identification of the causative agent(s) before the administration of topical, broad-spectrum antibiotics [6]. However, the most recent American Academy of Ophthalmology Bacterial Keratitis Practice Patterns guidelines indicate microbiological workup only for sight-threatening ulcers (large, central) and/or severe keratitis involving atypical appearance and/or those unresponsive to empirical therapy [1]. Smears should be collected and examined for rapid diagnosis and early implementation of therapy [1].
Current treatment recommendations for small infiltrates with no stromal involvement are to treat first with a broad spectrum commercially available (fluoroquinolone, aminoglycoside) topical antibiotic and culture later if at all. Most corneal ulcers are managed this way both in the community and by corneal specialists [1, 4, 29, 30].
6.7.5 Laboratory Workup
Rapid laboratory identification and in vitro susceptibility profiles are essential for providing appropriate and effective antibacterial therapy. Cultures and smears may offer the best support for (1) central, large ulcers with significant stromal involvement, (2) ulcers following surgery, (3) chronic ulcers, nonresponsive to empirical treatment, (4) ulcers with multiple infiltrates, and (5) ulcers with atypical clinical features [1].
Routine media and smears for microbiological assessment are outlined in Tables 6.2 and 6.3. Corneal scrapings may be obtained using a spatula, blade, and/or flocked swab.
The reported culture-dependent frequency, diversity, and relative abundance of the major bacterial corneal pathogens have remained essentially the same over the last 30 years in South Florida and worldwide. Frequency, diversity, and relative abundance differ however by geographic locations, patient populations, time periods, culture positive criteria, and practice locations [2, 6]. Data collected are highlighted in Tables 6.4, 6.5, 6.6, and 6.7 (Figs. 6.1 and 6.2).
The most commonly reported bacteria corneal pathogens include gram-positive species belonging to two major phyla (Firmicutes and Actinobacteria) [2, 6, 10, 17]. The frequency and abundance vary with patient populations, time period, culture frequency, and empirical treatment.
6.7.5.1 Firmicutes (Gram-Positive Cocci and Rods)
The Firmicutes are a phylum or group of bacteria with characteristics associated with gram-positive cell wall structure (Table 6.4). They usually stain purple with the gram stain due to a thick peptidoglycan layer [10]. They are found in a variety of habitats including the skin and mucus membranes of humans. These are the most reported bacterial species recovered from culture dependent and culture independent bacterial keratitis surveys. Staphylococci, Streptococci, Micrococcus, Lactobacillus, Bacilli, and Clostridia are among the most common genera among this group [6, 10]. This was the second most common phylum (42.4%) reported at our Institute and included 36 different species across 10 genera.
Staphylococci are among the leading causes of bacterial keratitis in the United States and worldwide. Reported prevalence ranges from 1 to 45% [2, 6]. Infections are diverse and include contact lens-associated keratitis (Fig. 6.3) and marginal keratitis. Coagulase-negative Staphylococci constitutes the major corneal pathogen recovered from patients with ocular surface disease including bullous keratopathy, chronic herpetic keratitis, and atopic keratoconjunctivitis [1]. Currently, there are more than 30 species of coagulase-negative Staphylococci [31]. However, S. epidermidis is the most commonly identified and reported species, especially in older patients, bandage contact lens wears, and those with a compromised ocular surface, altered immune response, and microbiome dysbiosis. This group of Firmicutes is among the most frequent bacteria identified by both culture-dependent and culture-independent studies as core members of the ocular surface microbiome [6, 7, 10, 17].
Coagulase-positive species, mainly S. aureus, are the second most frequently reported cause of bacterial keratitis in several reports from the United States and Europe, with rates ranging from 3 to 49% (Fig. 6.4) [6]. Both methicillin-sensitive (MSSA) and methicillin resistant (MRSA) Staphylococcus aureus isolates are frequently recovered from healthcare exposure. In a recent report from South Florida, 81.3% of the 75 evaluated isolates were healthcare–associated following corneal transplantation, contact lens wear, and/or cataract surgery. There was no significant difference between MRSA (n = 47, 74.5%) and MSSA (n = 28, 92.8%), p-0.0507 [32]. Similar results were reported from Taiwan [33].
Streptococci including S. pneumoniae are among the most common gram-positive cocci reported from Asia and are frequently associated with patients with chronic ocular surface disorders (Fig. 6.5). The group includes the beta-hemolytic Streptococci, S. pyogenes (Group A), S. agalactiae (Group B), the alpha-hemolytic species including S. pneumoniae and the viridans group (i.e., Streptococcus mitis, Streptococcus oralis). Nonhemolytic (gamma) Streptococci and nutritionally deficient Streptococci (i.e., Abiotrophia defective) are also in this group. This group appears in chains and/or pairs in gram stain and/or Giemsa smears [6].
Streptococcus pneumoniae was the top corneal pathogen recovered in the Steroids for Corneal Ulcer Trial (SCUT) study and may be the most common gram-positive isolate in some parts of India [34]. S. pneumoniae isolates were recovered in less than 10% of the corneal isolates in South Florida.
The Streptococcus viridans group is frequently involved in infectious crystalline keratopathy, described as gray-white opacities (Fig. 6.6). It most commonly develops in patients on long-term steroid use, especially following penetrating keratopathy. These bacteria are difficult to culture because they are encased in a thick biofilm which protects them from removal and antibiotic therapy [10, 35].
6.7.5.2 Actinobacteria (Gram-Positive Rods)
Actinobacteria are gram-positive rods found in the environment including soil and water and on human skin (Table 6.5). Genera include the Corynebacterium and Cutibacterium (Propionibacterium, Mycobacteria, and the aerobic Actinomycetes). Corynebacterium and Cutibacterium are considered part of the ocular “core” microbiota but are also recovered in bacterial keratitis among contact lens wears, the elderly, and patients with compromised ocular surface disease and/or immunosuppression [36, 37]. Mycobacteria and Nocardia corneal ulcers are discussed in Chaps. 7 and 8 respectively. We have observed a decline in the number of culture dependent Corynebacterium associated corneal ulcers in the last 5 years, but an increase in Nocardia and Mycobacteria species.
6.7.5.3 Proteobacteria (Gram-Negative Cocci, Rods)
Reported prevalence of infectious keratitis due to gram-negative bacteria ranges from 1 to 50% [1, 2, 6]. The majority of these are members of the Proteobacteria (Table 6.6). Common members among these included Pseudomonas aeruginosa, Serratia marcescens, and Moraxella species [4, 6,7,8, 15]. This was the largest and most diverse group of microorganisms recovered in our cultures. Pseudomonas aeruginosa was the most frequent isolate and constituted 65% of the Proteobacteria and 54.3% of the total gram-negative isolates in general. Bacteroidetes were recovered in less than 1% of bacterial corneal ulcers.
Pseudomonas species, predominantly, Pseudomonas aeruginosa, are leading causes of gram-negative bacterial keratitis. It is the leading cause associated with cosmetic contact lenses wear in the United States, the United Kingdom, and France [1, 4, 6]. It is also recovered from patients exposed to trauma with contaminated water, soil, and/or vegetation. In both conventional and metagenomic surveys, it has been recovered from healthy and diseased corneas as well as contact lenses from asymptomatic and symptomatic patients [15]. It presents as an acute, virulent, and progressive infection that can lead to corneal melting and perforation within 48 h (Fig. 6.7). It has an array of virulent factors including toxins, proteases, and antibiotic-resistant genes that aids it in establishing and maintaining corneal infection [1, 4].
6.7.5.3.1 Bacteroidetes
Members of this group are recovered from patients with chronic ocular surface disease and/or chronic local/systemic ocular disease (Table 6.7). These are found in a wide variety of environmental niches (soil, water) and in the gut and on the skin of humans and animals [10]. These ulcers can be indolent and difficult to treat.
6.8 Use of Metagenomics to Detect Bacterial Keratitis
Although several investigators have attempted to use shotgun metagenomics to reduce turnaround time and improved identification of the etiological agent in bacterial keratitis, challenges exist [38]. Currently, there are no standard protocols for specimen collection, storage, or genomic platform. Availability is also an issue.
The main challenge, however, is coordinating and selecting the true ocular pathogen from the diversity of microorganisms recovered and associated with this sensitive method and/or documenting the roles of the other community members to the disease process. In a recent report by Seitzman et al., they confirmed that shotgun metagenomics could amplify the etiological agent identified by culture in a proof-of-concept exercise [39]. This was done by eliminating “contaminating and background taxa.” However, 15 of the 20 (75%) most common taxa have been recovered from bacteria. The most challenging task in establishing an etiological agent interpreting metagenomic results. No culture-negative cases were included in the study. As with culture results, metagenomic analysis must be coordinated with the clinical picture.
6.8.1 Antimicrobial Therapy
Empirical therapy, with broad spectrum, commercially available topical antibiotics Empirical therapy is the current standard of care for treating bacterial keratitis [1]. Both the AAO’s current Bacterial Keratitis Practice guidelines and the Royal College of Ophthalmologists Focus, United Kingdom guidelines recommend monotherapy with a fluoroquinolone as the initial therapy for bacterial keratitis. Alternatives include combination/fortified therapy with a cephalosporin and aminoglycosides. Vancomycin should be reserved for documented multidrug-resistant gram-positive isolates. Efficacy and susceptibility profiles may be significantly different according to location, patient populations, and dispensing frequency [1, 40].
Evidence and support for these recommendations were provided by a 2014 Cochrane-like review that found no difference in the effectiveness between monotherapy with a fluoroquinolone and combination therapy with commonly used fortified antibiotics (cephalosporins and aminoglycosides) [41, 42].
However, increasing drug resistance to the fluoroquinolones and emerging resistance to commonly used fortified drops in the United States have been documented by two national surveys since 2005. The Ocular Tracking Resistance in US Today (TRUST) was the first national surveillance study tracking emerging antibiotic resistance among Staphylococcus aureus, Streptococcus pneumoniae, and Haemophilus influenzae ocular isolates collected over a 3-year period 2005–2008. Susceptibility rates among the fluoroquinolones for MSSA and MRSA were less than 90% and 20% respectively. Susceptibility for S. pneumoniae and Haemophilus influenzae remained at >90% [43].
The Antibiotic Resistance Monitoring in Ocular Microorganisms (ARMOR) replaced and expanded the TRUST study in 2009. Data from the cumulative ARMOR (2009–2018) report confirms the continuation of increasing fluoroquinolone resistance among ocular Staphylococcal isolates and increasing resistance among both Pseudomonas aeruginosa and Streptococcus pneumoniae. Nonsusceptibility rates to the aminoglycosides ranged from 2.9% P. aeruginosa to 17.5% coagulase-negative Staphylococci. The rate for S. aureus was 15.6%. For MSSA and CoNMSSA nonsusceptible rates were less than 7%, which methicillin resistance was associated with rates higher than 25% [44]. Multidrug-resistant isolates to ciprofloxacin and ofloxacin have been reported from several developing countries [45]. Wilcox reported resistance rates ranging from 10 to 24% of isolates from Africa, Asia, and South America [46].
In general, bacterial ocular isolates recovered from keratitis in the United States remain susceptible to combination therapy with fortified antimicrobials, cephalosporins and aminoglycosides and/or aminoglycosides and vancomycin [6, 47].
Increasing or sustained fluoroquinolone resistance (ciprofloxacin, ofloxacin, levofloxacin, and moxifloxacin) among methicillin susceptible and resistant Staphylococci and emerging resistance among Pseudomonas aeruginosa, Serratia marcescens, and Streptococcus pneumoniae remain worrisome [6]. The current recommendation of initial fluoroquinolone treatment for bacterial keratitis may need to be revisited for this location and regions with rates greater than 20% resistance. Antibiotic drug selection should be based on clinical impression, probable pathogens, antibiotic exposure history, and current local susceptibility profiles.
In vitro susceptibility testing may improve antimicrobial therapy. Although there is no current and/or standard ocular breakpoints, Oldenburg et al. documented a correlation with general MIC breakpoints and outcomes for keratitis for moxifloxacin [48]. The conclusion from the SCUT study was that moxifloxacin mediates the relationship between causative organisms and clinical outcomes in bacterial keratitis and is likely on the causal pathway between the organism and outcome [34, 48].
6.9 Steroids and Bacterial Keratitis
The use of corticosteroids as adjunctive therapy in the management of bacterial keratitis is still somewhat controversial. Some clarity about their benefit was provided by the Steroids for Corneal Ulcers Trial (SCUT), a large, randomized, double-blinded, placebo study developed to evaluate the role of corticosteroids treatment of bacterial corneal ulcers [49].
In general, topical corticosteroids appeared safe but provided no significant improvement in the treatment of bacterial keratitis. They did appear to be beneficial for ulcers that were central, deep or large, non-Nocardia or classically invasive Pseudomonas aeruginosa, patients with low baseline vision, and when started early after the initiation of antibiotics [49, 50].
6.10 Conclusions
Bacterial keratitis remains a common, major global and public health concern. Despite improvements in surgical techniques, topical antibiotic spectrum, and patient and provider education, minimal progress has been achieved in reducing the incidence and/or prevalence of bacterial keratitis in urban and nonurban populations.
Local, regional, and international rates and pathogen spectrum have remained relatively stable over the last 30 years with little improvement in clinical assessment/diagnosis or rapid laboratory detection. Increasing exposure to healthcare (contact lens—cosmetic, therapeutic; ocular surgeries) coupled with the aggressive use of broad-spectrum antibiotics, corticosteroids, and other immunosuppressive agents increase the risk of bacterial keratitis. This strategy does not seem to be effective in reducing the antimicrobial resistance. Rates or complications associated with this current “standard of care.”
A new paradigm is needed. It needs to include the integration of improved clinical and laboratory techniques/technology for rapid and accurate diagnosis with the understanding of the dynamic interactions at the ocular surface health, between the ocular and gut microbiome diversity and immune defenses in cornea health and disease. Two new opportunities to reach this goal include the use of artificial intelligence and selection of next generation sequencing.
6.10.1 Artificial Intelligence and Neural Networks
The use of artificial intelligence and neural networks could serve as an adjunct to supplement both the clinical and laboratory diagnosis of bacterial keratitis. A combination of imaging (confocal, slit lamp), microbiology slides as well as integration of clinical and microbiology laboratory data from electronic medical records and/or microbiology laboratory database could be integrated into a neural network to generate a computer-assisted diagnosis of bacterial keratitis.
6.10.2 Next Generation Sequencing
Use of molecular techniques such as shotgun metagenomics and standard bioinformatics interpretations could provide information on the interaction of the ocular microbiome, ocular surface epithelium and local immune (tears) defenses and their contribution to patient diverse outcomes in bacterial keratitis.
References
Lin A, Rhee MK, Akpek EK, Amescua G, Farid M, Garcia-Ferrer FJ, Varu DM, Musch DC, Dunn SP, Mah FS, American Academy of Ophthalmology Preferred Practice Pattern Cornea and External Disease Panel. Bacterial keratitis preferred practice pattern. Ophthalmology. 2019;126(1):P1–P55.
Ung L, Bispo PJM, Shanbhag SS, Gilmore MS, Chodosh J. The persistent dilemma of microbial keratitis: global burden, diagnosis, and antimicrobial resistance. Surv Ophthalmol. 2019;64(3):255–71.
Collier SA, Gronostaj MP, MacGurn AK, Cope JR, Awsumb KL, Yoder JS, Beach MJ. Estimated burden of keratitis—United States, 2010. MMWR Morb Mortal Wkly Rep. 2014;63(45):1027–30.
Hong A, Shute T, Huang A. Bacterial keratitis. In: Mannis M, Holland E, editors. Cornea, fundamentals, diagnosis and management. 4th ed. New York: Elsevier; 2017. p. 875–901.
Boost M, Cho P, Wang Z. Disturbing the balance: effect of contact lens use on the ocular proteome and microbiome. Clin Exp Optom. 2017;100(5):459–72.
Miller D. Update on the epidemiology and antibiotic resistance of ocular infections. Middle East Afr J Ophthalmol. 2017;24(1):30–42.
Karsten E, Watson SL, Foster LJR. Diversity of microbial species implicated in keratitis: a review. Open Ophthalmol J. 2012;6:110–24.
Teweldemedhin M, Gebreyesus H, Atsbaha AH, Asgedom SW, Saravanan M. Bacterial profile of ocular infections: a systematic review. BMC Ophthalmol. 2017;17(1):212.
Fleiszig SM, Evans DJ. Pathogenesis of contact lens-associated microbial keratitis. Optom Vis Sci. 2010;87(4):225–32.
Ryan KJ. Sherris medical microbiology. 7th ed. New York: McGraw-Hill Education; 2018.
Spierer O, Miller D, O’Brien TP. Comparative activity of antimicrobials against Pseudomonas aeruginosa, Achromobacter xylosoxidans and Stenotrophomonas maltophilia keratitis isolates. Br J Ophthalmol. 2018;102(5):708–12.
Wiley L, Bridge DR, Wiley LA, Odom JV, Elliott T, Olson JC. Bacterial biofilm diversity in contact lens-related disease: emerging role of Achromobacter, Stenotrophomonas, and Delftia. Invest Ophthalmol Vis Sci. 2012;53(7):3896–905.
Miller D, Iovieno A. The role of microbial flora on the ocular surface. Curr Opin Allergy Clin Immunol. 2009;9(5):466–70.
Cavuoto KM, Banerjee S, Galor A. Relationship between the microbiome and ocular health. Ocul Surf. 2019;17(3):384–92.
Ozkan J, Willcox MD. The ocular microbiome: molecular characterisation of a unique and low microbial environment. J Current Eye Res. 2019;44(7):685–94.
Thomason CA, Mullen N, Belden LK, May M, Hawley DM. Resident microbiome disruption with antibiotics enhances virulence of a colonizing pathogen. Sci Rep. 2017;7(1):16177.
Lu LJ, Liu J. Human microbiota and ophthalmic disease. Yale J Biol Med. 2016;89(3):325–30.
Kugadas A, Christiansen SH, Sankaranarayanan S, Surana NK, Gauguet S, Kunz R, Fichorova R, Vorup-Jensen T, Gadjeva M. Impact of microbiota on resistance to ocular pseudomonas aeruginosa-induced keratitis. PLoS Pathog. 2016;12(9):e1005855.
Jayasudha R, Chakravarthy SK, Prashanthi GS, Sharma S, Garg P, Murthy SI, Shivaji S. Alterations in gut bacterial and fungal microbiomes are associated with bacterial keratitis, an inflammatory disease of the human eye. J Biosci. 2018;43(5):835–56.
Hsiao CH, Yeung L, Ma DH, Chen YF, Lin HC, Tan HY, Huang SC, Lin KK. Pediatric microbial keratitis in Taiwanese children: a review of hospital cases. Arch Ophthalmol. 2007;125(5):603–9.
Lee YS, Tan HY, Yeh LK, Lin HC, Ma DH, Chen HC, Chen SY, Chen PY, Hsiao CH. Pediatric microbial keratitis in Taiwan: clinical and microbiological profiles, 1998-2002 versus 2008-2012. Am J Ophthalmol. 2014;157(5):1090–6.
Ormerod D, Murphree AL, Gomez DS, Schanzlin DJ, Smith RE. Microbial keratitis in children. Ophthalmology. 1986;93(4):449–55.
Rossetto JD, Cavuoto KM, Osigian CJ, Chang TCP, Miller D, Capo H, Spierer O. Paediatric infectious keratitis: a case series of 107 children presenting to a tertiary referral centre. Br J Ophthalmol. 2017;101(11):1488–92.
Yu MC, Höfling-Lima AL, Furtado GH. Microbiological and epidemiological study of infectious keratitis in children and adolescents. Arq Bras Oftalmol. 2016;79(5):289–93.
Kunimoto DY, Sharma S, Reddy MK, Gopinathan U, Jyothi J, Miller D, Rao GN. Microbial keratitis in children. Ophthalmology. 1998;105(2):252–7.
Cruz OA, Sabir SM, Capo H, Alfonso EC. Microbial keratitis in childhood. Ophthalmology. 1993;100(2):192–6.
Chirinos-Saldaña P, Bautista de Lucio VM, Hernandez-Camarena JC, Navas A, Ramirez-Miranda A, Vizuet-Garcia L, Ortiz-Casas M, Lopez-Espinosa N, Gaona-Juarez C, Bautista-Hernandez LA, Graue-Hernandez EO. Clinical and microbiological profile of infectious keratitis in children. BMC Ophthalmol. 2013;13:54.
Al-Otaibi AG. Non-viral microbial keratitis in children. Saudi J Ophthalmol. 2012;26(2):191–7.
Park J, Lee KM, Zhou H, Rabin M, Jwo K, Burton WB, Gritz DC. Community practice patterns for bacterial corneal ulcer evaluation and treatment. Eye Contact Lens. 2015;41(1):12–8.
Austin A, Schallhorn J, Geske M, Mannis M, Lietman T, Rose-Nussbaumer J. Empirical treatment of bacterial keratitis: an international survey of corneal specialists. BMJ Open Ophthalmol. 2016;2:e000047.
Becker K, Heilmann C, Peters G. Coagulase-negative staphylococci. Clin Microbiol Rev. 2014;27(4):870–926.
Peterson JC, Durkee H, Miller D, Maestre-Mesa J, Arboleda A, Aguilar MC, Relhan N, Flynn HW Jr, Amescua G, Parel JM, Alfonso E. Molecular epidemiology and resistance profiles among healthcare- and community-associated Staphylococcus aureus keratitis isolates. Infect Drug Resist. 2019;12:831–43.
Hsiao CH, Ong SJ, Chuang CC, Ma DHK, Huang YC. A comparison of clinical features between community-associated and healthcare-associated methicillin-resistant Staphylococcus aureus keratitis. J Ophthalmol. 2015;2015:923941.
Lalitha P, Srinivasan M, Manikandan P, Bharathi MJ, Rajaraman R, Ravindran M, Cevallos V, Oldenburg CE, Ray KJ, Toutain-Kidd CM, Glidden DV, Zegans ME, McLeod SD, Acharya NR, Lietman TM. Relationship of in vitro susceptibility to moxifloxacin and in vivo clinical outcome in bacterial keratitis. Clin Infect Dis. 2012;54(10):1381–7.
Porter AJ, Lee GA, Jun AS. Infectious crystalline keratopathy. Surv Ophthalmol. 2018;63(4):480–99.
Das S, Rao AS, Sahu SK, Sharma S. Corynebacterium spp as causative agents of microbial keratitis. Br J Ophthalmol. 2016;100(7):939–43.
Ovodenko B, Seedor JA, Ritterband DC, Shah M, Yang R, Koplin RS. The prevalence and pathogenicity of Propionibacterium acnes keratitis. Cornea. 2009;28(1):36–9.
Borroni D, Romano V, Kaye SB, Somerville T, Napoli L, Fasolo A, Gallon P, Ponzin D, Esposito A, Ferrari S. Metagenomics in ophthalmology: current findings and future prospectives. BMJ Open Ophthalmol. 2019;4:e000248.
Seitzman GD, Hinterwirth A, Zhong L, Cummings S, Chen C, Driver TH, Lee MD, Doan T. Metagenomic deep sequencing for the diagnosis of corneal and external disease infections. Ophthalmology. 2019;126(12):1724–6.
Tuft S, Burton M. Microbial keratitis. Royal College Ophthalmologists, Autumn; 2013.
McDonald EM, Ram FS, Patel DV, McGhee CN. Topical antibiotics for the management of bacterial keratitis: an evidence-based review of high quality randomised controlled trials. Br J Ophthalmol. 2014;98(11):1470–7.
Austin A, Lietman T, Rose-Nussbaumer J. Update on the management of infectious keratitis. Ophthalmology. 2017;124(11):1678–89.
Asbell PA, Colby KA, Deng S, McDonnell P, Meisler DM, Raizman MB, Sheppard JD Jr, Sahm DF. Ocular TRUST: nationwide antimicrobial susceptibility patterns in ocular isolates. Am J Ophthalmol. 2008;145(6):951–8.
Asbell PA, Sanfilippo CM, Sahm DF, DeCory HH. Trends in antibiotic resistance among ocular microorganisms in the United States from 2009 to 2018. JAMA Ophthalmol. 2020;138(5):439–50.
Fernandes M, Vira D, Medikonda R, Kumar N. Extensively and pan-drug resistant Pseudomonas aeruginosa keratitis: clinical features, risk factors, and outcome. Graefes Arch Clin Exp Ophthalmol. 2016;254(2):315–22.
Willcox MD. Management and treatment of contact lens-related Pseudomonas keratitis. Clin Ophthalmol. 2012;6:919–24.
Thomas RK, Melton R, Asbell PA. Antibiotic resistance among ocular pathogens: current trends from the ARMOR surveillance study (2009-2016). Clin Optom. 2019;11:15–26.
Oldenburg CE, Lalitha P, Srinivasan M, Manikandan P, Bharathi MJ, Rajaraman R, Ravindran M, Mascarenhas J, Nardone N, Ray KJ, Glidden DV. Moxifloxacin susceptibility mediates the relationship between causative organism and clinical outcome in bacterial keratitis. Invest Ophthalmol Vis Sci. 2013;54(2):1522–6.
Srinivasan M, Mascarenhas J, Rajaraman R, Ravindran M, Prajna L, Glidden DV, Ray KJ, Hong KC, Oldenburg CE, Lee SM, Zegans ME, McLeod SD, Lietman TM, Nisha R, Acharya NR, Steroids for Corneal Ulcers Trial Group. Corticosteroids for bacterial keratitis: the Steroids for Corneal Ulcers Trial (SCUT). Arch Ophthalmol. 2012;130(2):143–50.
Ni N, Srinivasan M, McLeod SD, Acharya NR, Lietman TM, Rose-Nussbaumer J. Use of adjunctive topical corticosteroids in bacterial keratitis. Curr Opin Ophthalmol. 2016;27(4):353–7.
Acknowledgments
With contributions from Nayef K. Alshammari, Feras Mohder, Anne-Marie E. Okoduwa, and Mike Zein.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Miller, D., Cavuoto, K.M., Alfonso, E.C. (2021). Bacterial Keratitis. In: Das, S., Jhanji, V. (eds) Infections of the Cornea and Conjunctiva. Springer, Singapore. https://doi.org/10.1007/978-981-15-8811-2_6
Download citation
DOI: https://doi.org/10.1007/978-981-15-8811-2_6
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-15-8810-5
Online ISBN: 978-981-15-8811-2
eBook Packages: MedicineMedicine (R0)