Abstract
Of late, a spurt in the general awareness about the biological aspects of nutrition has been witnessed. The changing trend demands for high nutritional value products that can easily and rapidly be produced at large scales in a cost-effective manner. Microalgae constitutes a distinct group of unicellular photosynthetic organisms and a broad variety of eukaryotic algae containing a plethora of beneficial compounds such as carbohydrate, proteins, fatty acids, vitamins, carotenoids, phycobiliproteins, astaxanthin, and lutein. These compounds find application in the production of high-quality nutraceuticals that provide health benefits such as controlling blood pressure, boosting immune system, reducing coronary heart diseases, serving as anticancer agents, and acting as antioxidants. Besides, the benefits of using microalgae are its high productivity on arable and nonarable land, thus posing no threat to the agricultural crop production. Although the nutritional value and its commercialization is still in nascent stage, intense efforts are underway all over the world to explore untapped potential of microalgae that could lead to the solution of several problems through green technologies and open gateway to a multibillion dollar industry. This chapter gives an overview of microalgae and its diversity, nutritional value, and current challenges on its use as nutraceutical product.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
5.1 Introduction
The holistic development of any nation depends on the good health and well-being of its public. The frequent consumption of packaged and junk foods, fast eating food habitats, long-scheduled work, sedentary lifestyle, etc. has resulted in a spurt in the prevalence of various diseases like carcinomas and heart-related disorders (Subudhi 2017). Therefore, there is an increasing global concern on the implementation of healthy foods in diet that will help to maintain a healthy lifestyle (Ranga et al. 2017). However, huge investment and capacity building are required to affirm to the food and nutritional security that often requires huge amount of farming land, livestock, and high density of natural resources. However, the current health policies demand the consumers to fend for themselves and entice them to attain maximum health benefits by spending minimum money (Lenoir-Wijnkoop et al. 2010). Therefore, a paradigm shift from traditional agriculture toward more sustainable solutions is the need of hour. To reduce the possibility of health problems, consumers prefer to use foods rich in adequate nutrients or nutraceuticals. Nutraceutical is a combination of two terms “nutrition” and “pharmaceutical,” a food stuff either in the form of fortified food or dietary supplement that has the capability to impart health benefits by strengthening the body’s response to ward off the infections and to get rid of diseases. Conventionally, these products are available in varied formulations. Nutraceutical are way more advantageous than the medicines as they are devoid of side effect, natural sources, easily available, and affordable. The term “nutraceutical” comprises several products such as vitamins, minerals, enzymes, antioxidants, probiotics, prebiotics, polyunsaturated fats, polyphenols, and spices (Chauhan et al. 2013). Depending upon their functions, nutraceuticals are grouped as dietary supplements, medicinal food, functional foods, etc. (Dillard and German 2000). On the basis of source of their origin, these are classified as products obtained from plants (vitamins, phytochemicals), animal (polysaccharides), or microorganisms (poly amino acids) (Pattanaik et al. 2019). Among the microorganisms, microalgae present a sustainable powerful reserve of number of functional ingredients for wide food applications (Pulz and Gross 2004). In general, the term “algae” circumscribes photosynthetic eukaryotic organisms. On the basis of their size, they can be classified as micro- and macroalgae. Macroalgae or seaweeds are multicellular organisms that include benthic marine organisms with variable sizes extending up to several meters (Markou et al. 2012). In contrast, the term microalgae, or microphytes, are microscopic photosynthetic algae and bacteria or cyanobacteria. Microalgae also have variable sizes, which can be as small as picometers (Waterbury 2006; Barsanti and Gualtiery 2006). The ecology of microalgae is also diverse, which ranges from marine to freshwater forms, and their photosynthetic mechanism resembles that of land-based plants. The microalgae have received an appreciable interest as sources of natural food, polysaccharides, fatty acids, and biomass due to its potentially simple and cost-effective cultivation techniques (Borowitzka 2013; Leu and Boussiba 2014; Aziz et al. 2017). Another major thrust for commercialization of microalgae is their generally regarded as safe (GRAS) status that has important relevance for products intended to be used for consumption purposes (Gangl et al. 2015). In contrast to plants, microalgae have higher growth rates (e.g., one to three doublings per day) and do not pose any competition for resources as they can be grown easily in open environment (Ng et al. 2015). In recognition of innumerable advantages of microalgae, several efforts are underway to promote them at biotechnological platforms, especially from genetic engineering point of view.
5.2 Indian Microalgal Diversity
India owing to its rich biodiversity, diverse ecologies, agronomic practices, and multitude of soil types is considered to be ideal for microalgal growth. The biodiversity-rich parts of the country like Eastern Ghats (Jena et al. 2005, 2008; Prasanna and Kaushik 2005; Rath and Adhikary 2005; Samantraray et al. 2002) and Chilika Lake in Odisha are also home to diverse species of microalgae (Adhikary 2000; Ratha et al. 2003; Rath and Adhikary 2005) and have been widely investigated. The investigation by Ratha et al. (2012) on qualitative distribution of microalgae in diverse ecological habitats from major biodiversity hotspots of India reported Cyanophyceae and Chlorophyceae as the abundantly found algal groups. Suresh et al. (2012) studied the microalgal diversity in Western and Eastern Ghats of Tamil Nadu and reported 97 species of microalgae. Kharkongar and Ramanujam (2014) reported a total of 85 taxa, including cyanobacteria and algal species belonging to diverse classes of algae with highest subaerial algal biodiversity in sacred grove compared to those of plantation and open disturbed forest. It is well reported in literature that the subaerial microhabitats are predominantly habitated by members belonging to Trentepohliales and cyanobacteria since they can easily adapt to adverse environmental conditions by adopting different survival mechanisms, such as production of carotenoids in Trentepohliales and extracellular polymeric substances (EPS) sheath by cyanobacteria as a protective sheath (Urzi and Realini 1998; Tomaselli 2003). Singh and Sharma (2014) reported 19 microalgal taxa belonging to Cyanophyta (8 taxa), Chlorophyta (7 taxa), and Bacillariophyta (4 taxa) from Sheer Khad (stream), which is a tributary of Sutlej River, Himachal Pradesh. Severes et al. (2018) identified microalgae from Western Ghats regions of India. The panorama depicted by the microalgal biodiversity suggests their potential application in diverse fields, nutraceutical one of them, since they are proficient in producing high-value compounds even in the harsh environmental conditions.
5.3 Microalgae as Nutraceutical
The consumption of microalgae as a human food source or nutritional supplements is not new, but their use dates back to prehistoric times. The species of Nostoc find use in various parts across the world, where they are consumed in traditional ways. In Japan, cyanobacterium Aphanothece sacrum is consumed as a popular delicacy known as “suizenji-nori.” The microalgal biomass has gained immense popularity for the production of health-based food products, where it is being used exclusively for this purpose for over past few decades. Depending upon the organic composition, an array of products under different names are present in the market as nutraceuticals like dietary supplements, herbal preparations/products, traditional medicine, food supplements, or botanical supplements (Nicoletti 2012; Rajasekaran et al. 2008). The first-generation food supplements consist of primary metabolites, whereas other substances, relevant to nutraceuticals, are secondary metabolites. The seaweeds contradict this simplified version of classification as they contain both the type of substances. Therefore, a new terminology “superfoods” was suggested (Bishop and Zubeck 2012). A third type of food supplement called as pharmafoods or functional foods is emerging because of their resemblance with ordinary foods in terms of physiological properties. These pharmafoods or functional foods probably can best harness the potential of microalgae in the future for a wide variety of different products after doing away with associated limitation such as poor aesthetics in the current utilizations (Nicoletti 2012).
5.4 Nutritional Components of Microalgae
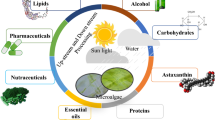
Microalgae are repertoire of valuable compounds such as proteins, carbohydrates, polyunsaturated fatty acids (PUFAs), minerals, and vitamins that can not only heighten the nutritional content of food but also serve to provide benefits to the consumer (Table 5.1 and Fig. 5.1).
5.4.1 Microalgal Proteins
The essential amino acids (EAA) composition of algae is in sync with FAO requirements. Chlorella vulgaris, highly rich in protein content and desirable EAA composition, is popularly used as a food supplement (Becker 2007; Chronakis and Madsen 2011). Microalgae-based proteins have several advantages over other conventional protein sources, and they have scant land utilization compared to animal-based proteins (de Vries and de Boer 2010; Van Krimpen et al. 2013; Smetana et al. 2017), low water requirement, and ability to grow in saline water, among others (FAO 2010). Many species of microalgae produce proteins on par with egg, meat and milk, etc. (Gouveia et al. 2008a). Notably, red species of algae contain low concentrations of leucine and isoleucine, while brown algae species are often limited in methionine, cysteine, and lysine (Dawczynski et al. 2007; Mišurcová et al. 2014).
5.4.2 Microalgal Peptides and Protease Inhibitors
The bioactive peptides produced by the microalgae are known to possess anticancer, antiviral, antioxidant, and immunomodulatory effects. Proteases are a group of proteolytic enzymes with fine applications in food, detergents, and pharmaceutical industries. Their deregulation can result in serious health-related issues, and that is why both proteases and protease inhibitors find applications as therapeutic agents (Drag and Salvensen 2010).
Protease inhibitors are small molecules, and usually, peptides mimic the structure of a substrate and bind to enzymes. Chlorella vulgaris produces pepsin-hydrolyzed peptide with strong antioxidant activity and is resistant against gastrointestinal enzymes (Sheih et al. 2009a), besides containing a peptide with antiproliferative activity (Sheih et al. 2009b). Cyanobacteria also produce a set of potent metabolites in minute quantities (Janssen 2019). Microcystis has been known to produce aeruginosins, which helps to curtail the thromboembolic disorders by efficiently binding to serine proteases (Ersmark et al. 2008; Wang et al. 2009). The brackish water cyanobacterium Nodularia spumigena (Mazur-Marzec et al. 2013) and Anabaena compacta (Anas et al. 2012) are known to produce Spumigins, which inhibits trypsin-like serine proteases. The anabaenopeptins-cyclic hexapeptides act against exopeptidase, carboxypeptidase A (Murakami et al. 2000). The microginins have been shown to inhibit various exopeptidases (Welker and von Dohren 2006). Microginins are considered to be potential candidates for the development of new drugs for cardiovascular diseases as they are known to inhibit angiotensin-converting enzyme (ACE-I) and leucyl aminopeptidase (LAP) (Bagchi et al. 2016). The aerucyclamides have been reported to show activity against cancer cells and parasitic infections (Ishida et al. 2000; Portmann et al. 2008). The cyanopeptolins and aeruginosins show inhibitory action against serine proteases (Gademann and Portmann 2008; Hanessian et al. 2006). Ribulose-1,5-bisphosphate carboxylase/oxygenase (RuBisCO), a heterohexadecamer structured protein, possesses functionally bioactive peptides known to cure cardiovascular diseases, diabetes, neurodegenerative disorders, and oxidative stress (Selvaraj et al. 2017).
5.4.3 Microalgal Carbohydrates
Carbohydrates constitute a wide variety of sugars or polysaccharides. Depending on the species, the microalgae produce discrete carbohydrates, e.g., glycogen, floridean starch, and amylopectin-like polysaccharides (Nakamura et al. 2005). Because of the absence of hemicelluloses and lignin, the algal biomass is amenable to easy digestion (Mussgnug et al. 2010). The cultivation and environmental conditions as well as the microlagal species determine the biomass carbohydrate content. Microalgal polysaccharides are recognized to promote gut microflora growth and regulation of blood glucose as they constitute a part of prebiotic supplements (Ibañez and Cifuentes 2013). Many cyanobacteria and green algae are surrounded by a special mucilaginous covering around their cells or filaments composed of exopolysaccharides (EPS) and are termed as slimes, sheaths, and/or capsules depending upon the species (Kumar and Adhikary 2018). EPS have received wide attention currently for their antimicrobial and anticarcinogenic roles (Mahendran et al. 2013, Bafanaa 2013). The cell wall polysaccharides from Chlorella vulgaris contain β-(1,3)-glucose, while the microalgal species contain heteropolysaccharides with different substituents (Raposo et al. 2014). The polysaccharides from P. cruentum are inhibitory against viruses, as well as bacteria (Huang et al. 2005; Raposo et al. 2014). The extracellular polysaccharides of Rhodella reticulata exhibit free radical scavenging and antioxidant activity (Chen et al. 2010a). Besides, EPS from red microalgae and Arthrospira platensis show antimicrobial and antioxidant activities (Rafika et al. 2011).
5.4.4 Microalgal Polyunsaturated Fatty Acids
Polyunsaturated fatty acids (PUFAs) which contain three or more double bonds play integral role in maintaining tissue integrity and imparting beneficial health effects, especially n-3 PUFAs, which are found to be effective in prevention or treatment of several ailments (e.g., heart-related disorders, various malignancies, and many more). Until recently, omega-3 and omega-6 fatty acids, the integral fatty acids, were mainly being derived from fish oil, but due to several concerns like overexploitation of marine sources, detection of toxic compounds in fishes, awful smell and taste, and oxidative instability, the interest has been deviated toward the exploitation of microalgae as an alternative source of PUFAs (Garcia et al. 2017). The three chief types of omega-3 fatty acids implicated in human health and physiology are α-linolenic acid (ALA), eicosapentaenoic acid (EPA), and docosahexaenoic acid (DHA). The microalgal species Arthrospira produce α-linolenic acid (ALA) and Nannochloropsis, Phaeodactylum, and Nitzschia produce EPA, while Crypthecodinium and Schizochytrium are the producers of DHA. The Crypthecodinium cohnii produces all the enzymes necessary for de novo synthesis of 22:6ω-3 (Henderson and Mackinlay 1999). Nannochloropsis is another promising candidate for pharmaceutical-based applications because it accumulates high levels of PUFA (Udayan et al. 2017). Arachidonic acid (AA), derivative of omega-6 fatty acid is considered as a precursor of prostaglandin and leucotriene synthesis, which has major role to play in circulatory and CNS functions (Medina et al. 1997).
5.4.5 Microalgal Sterols
Sterols, which are required to regulate the membrane fluidity, reduce the LDL-cholesterol levels and promote cardiovascular health (Piironen et al. 2000; Volkman 2003; Silvestro et al. 2013; Alsenani et al. 2015). Microalgae are recognized to produce both saturated and unsaturated sterols such as brassicasterol, sitosterol, and stigmasterol (Kohlhase and Pohl 1988; Volkman 2003). Glaucocystophyte has been reported to produce sitosterol, campesterol, and stigmasterol (Leblond et al. 2011), dinoflagellates produce 4α-methyl sterols and 24-propylidenecholesterol (Thomson et al. 2004; Giner et al. 2009), and Dunaliella tertiolecta and D. salina produce sterols with in vivo neuromodulatory activities (Francavilla et al. 2012).
5.4.6 Microalgal Pigments
Microalgal pigments like chlorophylls (a, b, and c), phycobiliproteins, phycocyanin, phycoerythrin, β-carotene, lutein, and astaxanthin are important bioactive compounds (Zhang et al. 2016). Chlorophyll or its derived products are widely been used for health benefit characteristics such as antioxidant, therapeutic properties, neuroprotective action, and protection against chronic diseases (Pangestuti and Kim 2011; Galasso et al. 2019). Chlorophyll a and its mixture with chlorophyll b exhibit chemopreventive effects, antioxidant activity, promotion of cell arrest, and apoptosis (Mishra et al. 2011). Some of the most important carotenoids with health benefits are ß-carotene, astaxanthin, lutein, and canthaxanthin. The antioxidant and therapeutic potentials of carotenoids include their role in prevention of diabetes, aging, cancer, obesity, and stroke, with higher provitamin A activity of ß-carotene and lipid peroxidation activity of astaxanthin (Chidambara-Murthy et al. 2005; Lin et al. 2016; Raposo et al. 2001, 2013). β-Carotene and astaxanthin strongly prevent oxidative stress through scavenging of free radicals and also exhibit anticancer effect (Cuvelier 2001; Demming-Adams and Adams 2002; Uttara et al. 2009; Lobo et al. 2010). β-Carotene produced by D. salina and D. bardawil lower plasma cholesterol and atherogenesis (García-González et al. 2005). The vitamins C and E, β-carotene, and zinc have proved to be effective against age-related macular degeneration, which is responsible for causing blindness (Taylor et al. 2002). Astaxanthin by Haematococcus pluvialis and Chlorella zofingiensis prevents obesity and fatty liver disease (Lorenz and Cysewski 2000; Ikeuchi et al. 2007). Astaxanthin has protective effects against many diseases, like cancers, and prevents against Helicobacter pylori (Olaizola 2005; Ikeuchi et al. 2007; Kamath et al. 2008; Satoh et al. 2009; Yuan et al. 2011). Dried biomass of Haematococcus pluvialis which is a rich source of astaxanthin has been commercialized owing to its strong antioxidant activity. Astaxanthin-rich Haematococcus available as dietary supplement (Lorenz and Cysewski 2000) has strong antioxidant activity (Higuera-Ciapara et al. 2006). Zeaxanthin by Microcystis aeruginosa, Nannochloropsis, and D. salina helps in vision and antioxidant protection of the body (Jin et al. 2003; Chen et al. 2005). Lutein produced by Chlorella zofingiensis, Chlorella protothecoides, and Muriellopsis sp. has protective antioxidative effect (Kleinegris et al. 2010). Studies found that chances of cataract decreases on consumption of lutein-/zeaxanthin-rich foods. Fucoxanthin is another microalgal pigment with nutraceutical abilities, which is associated with weight loss management (Abidov et al. 2010). The antioxidant and anti-inflammatory potential of phycocyanin and other pigments from red algae is also well documented (Kumar et al. 2014). Phycobiliproteins produced by cyanobacteria and rhodophyta (Eriksen 2008; Watanabe and Ikeuchi 2013; Mulders et al. 2014) reduce oxidative stress by neutralizing the reactive oxygen species (ROS), which is possible due to their chemical structures and chelating properties (Roy et al. 2007; Eriksen, 2008; Stengel et al. 2011; Rodriguez-Sanchez et al. 2012; de Jesus Raposo et al. 2013). Phycocyanin and phycoerythrin are two well-known commercially important phycobiliproteins produced by Spirulina sp. and Porphyridium sp., respectively (Plaza et al. 2009; Rodriguez-Sanchez et al. 2012; Borowitzka 2013). Microalgae produce chlorophylls a, b, c, d, and f with variable absorption spectra and tonality (Chen et al. 2010b; Roy et al. 2011). Studies have shown that the antimutagenic effect of microalgae is conferred by chlorophylls (Ferruzi and Blakeslee 2007; Gouveia et al. 2008a).
5.4.7 Microalgal Vitamins
Microalgae form a part of human nutrition as it is a rich source of a number of vitamins. It has been reported that vitamin B12 has a role to play in DNA repair and histone methylation, and it reduces the risk of breast cancer (Gruber 2016). Nannochloropsis oculata, Chaetoceros calcitrans, Porphyridium cruentum, and Haslea ostrearia are rich source of vitamin E (tocopherols) (Durmaz 2007; Bong and Loh, 2013; Santiago-Morales et al. 2018). Ascorbic acid is not only used as a food additive but also effective against diseases such as cancer and several infections (Boyera et al. 1998; Nunes-Alves et al. 2014). Porphyridium cruentum produces high quantities of vitamins E and C (ascorbic acid), as well as β-carotene (vitamin A) (Mus et al. 2013). The microalga D. salina produces vitamins A, B1, B2, B3, B6, B7, and E (Hosseini Tafreshi and Shariati 2009). Microalgae is also known to contain vitamins D2 and D3 (Takeuchi et al. 1991; Rao and Raghuramulu 1996), together with provitamin D3. Vitamin D and its metabolites are implicated in chemoprevention activities (Giammanco et al. 2015).
5.4.8 Microalgal Minerals
The mineral composition of microalgae which is determined by geographical range and environmental conditions varies extensively among marine and freshwater microalgae on a strain, species, and generic basis (Fox and Zimba 2018). Minerals are present either in the form of compounds or elemental form such as Zn, Ca, and Mg and have important role to play (Nose 1972). The mineral content of microalgae is sufficient to fulfill the recommended daily intake for adults (Alsenani et al. 2015). Significant quantities of Zn, K, Na, Fe, P, Mg, and Mn is produced by Isochrysis sp., Sochrisis sp., Chlorella sp., and Dunaliella sp. (Fabregas and Herrero 1986; Tokuşoglu and Üunal 2003). The mineral content produced by the microalgae fulfills the Recommended Dietary Allowances (RDA), prescribed for various minerals (USDA 2002; Tokuşoglu and Üunal 2003).
5.4.9 Microalgal Phenolic and Volatile Compounds
Phenolic compounds (PCs) or polyphenolics are secondary metabolites produced by microalgae in response to stress conditions (Cabrita et al. 2010; La Barre et al. 2010). Phenolic compounds like caffeic acid, ferulic acid, and p-coumaric acid, which are used as dietary supplements as they represent important classes of natural antioxidants (Stengel et al. 2011). Chlorella and Spirulina are known to produce an array of phenolic compounds in appreciable quantities like phloroglucinol, p-coumaric acid, ferulic acid, and apigenin, which are produced by Chlorella at 74,000 ng/g, 540 ng/g, 0.63 ng/g, and 9.9 ng/g and by Spirulina at 51,000 ng/g, 920 ng/g, 0.67 ng/g, and 6.0 ng/g, respectively (Goiris et al. 2014). The other microalgal species known to produce phenolic compounds are Nostoc sp., Chlorella sp., Anabaena sp., Tolypothrix sp., and Chlamydomonas sp.(Li et al. 2007; Hajimahmoodi et al. 2010) with phenolic contents of par or more than fruits and vegetables (Ismail et al. 2004; Hassimotto et al. 2005; Lin and Tang 2007).
The volatile compounds are also the secondary metabolites that are responsible for imparting characteristic odors to the water (Abd El-Baky et al. 2002). They have diverse structures and biological activities such as antibacterial, antifungal, antiviral, and anticancer. The biologically active volatile compounds are aldehydes, ketones, fatty acids, and isoprenylated and brominated hydroquinones (Mathew et al. 1995; Morinoto et al. 1995; Borowitzka 1997). The heptadecane and tetradecane produced by Spirulina are known to have antibacterial capacity (Ozdemir et al. 2004). Dunaliella salina produces antimicrobial compounds like β-cyclocitral, α- and β-ionone, neophytadiene, and phytol (Herrero et al. 2006).
5.4.10 Microalgal UV-Absorbing Compounds
The sizeable loss of ozone layer and consequent increment in ultraviolet (UV) radiation have resulted in a host of skin problems like extrinsic skin aging and wrinkles, mottled hyperpigmentation, dilated blood vessels, and loss of skin tone. All these skin-related disorders have ameliorated the interest in quest for natural photoprotective compounds (Lee et al. 2015). In aquatic environments, where microalgae figure prominently, the presence and role of UV-absorbing compounds like sporopollenin, scytonemin, and mycosporine-like amino acids (MAAs) have been documented. Their presumed antioxidant and skin protective strategies raise the interest for possible medicinal and cosmetic applications (Dionisio-Sese 2010). Xiong et al. (1997) observed that UV-B-tolerant chlorophyte species of Characium terrestre, Enallax coelastroides, Scenedesmus sp., Scotiella chlorelloidea, and Spongiochloris spongiosa contain produce large amounts of sporopollenin. The biopolymer has been reported to be present also in Dunaliella salina zygotes (Komaristaya and Gorbulin 2006). Microalgal scytonemin appears restricted to cyanobacteria, specifically in the extracellular polysaccharide sheath of Chlorogloeopsis sp., Scytonema sp., and Rivularia sp. (Sinha et al. 1998). It is reportedly the most important UV-absorbing compound in Lyngbya cf. aestuarii, where its area content seems to follow the seasonal fluctuation of solar intensity (Karsten et al. 1998). MAAs are UV radiation-absorbing molecules and possess high molar extinction coefficients. Unlike sporopollenin and scytonemin, which are found in the cell wall or extracellular sheath of the microalgae, MAAs in microalgae are mostly intracellular. The presence of several MAAs in a species was observed in different chlorophytes, haptophytes, diatoms, and dinoflagellates grown in cultures or collected in a broad variety of aquatic habitats (Llewellyn and Airs 2010). MAAs also have been reported in several microalgae-invertebrate (sea anemone, coral, ascidian) symbiotic associations. Among the Chlorophytes, asterina and shinorine are produced by Ankistrodesmus spiralis and Chlorella minutissima; palythine, porphyra, and shinorine are produced by Chlorella sorokiniana and Enallax coelastroides (Xiong et al. 1999); and mycosporine-glycine is produced by Pyramimonas parkeae (Hannach and Sigleo 1998). Among the halophytes, mycosporine-glycine is produced by Isochrysis sp. and Pavlova gyrans (Hannach and Sigleo 1998); among the Diatoms, porphyra and shinorine are produced by Chaetoceros sp., Corethron criophilumporphyra, Cosinodiscus centralisporphyra, Thalassiosira tumidaporphyra, and Porosira glacialis (Helbling et al. 1996; Riegger and Robinson 1997). Among the Dinoflagellates, mycosporine-glycine, palythene, palythine, porphyra, and shinorine are produced by Scrippsiella sweeneyae (Taira et al. 2004); asterina, mycosporine-glycine, palythene, palythenic acid, palythine, palythinol, porphyra, shinorine, and usujirene are produced by Alexandrium catenella, A. excavatum, A. minutum, and A. tamarense (Carreto et al. 1990).
5.4.11 Microalgal Toxic Metabolites
Microalgal species are known to produce toxins called as microcystins; homo- and anatoxin-a, also known as Very Fast Death Factor (VFDF); and saxitoxins. The most prevalent are the microcystins, produced by the blooming Microcystis aeruginosa. The most toxic amino acid known is microcystin LR produced by M. aeruginosa, which is lethal for both animals and human (Khan et al. 2018); its degree of toxicity is determined by the length of amino acid chain (Jungblut and Neilan 2006). Cytotoxins are pharmacologically active compounds with various biological activities such as anticancer, antimicrobial, antiplasmodial, and immunosuppressive (Abdo et al. 2012; Rath and Priyadarshani 2013; Malathi et al. 2014; Mukund and Sivasubramanian 2014; Semary and Fouda 2015; Shaieb et al. 2014). Amphidinium sp. produce an active antimycotic and antiprotozoal agent called as karatungiols (Washida et al. 2006). Dinophysis sp. is known to produce a potent neurotoxin called as okadaic acid for the treatment of cognitive disorders (He et al. 2005).
5.4.12 Microalgae as Food/Single Cell Protein
Microalgae has been consumed for its high protein content since thousands of the year. For example, Nostoc and Arthrospira or Spirulina have been used in Asia and Africa, respectively (Chacòn -Lee and Gonźalez-Maríno 2010). The most commonly consumed microalgae and with GRAS status are Spirulina, Chlorella, Dunaliella, Haematococcus, and Schizochytrium (Hayes et al. 2017). The microalgae-based foods are available as different formulations (Pulz and Gross 2004) or as fortified foods in the forms of confectionaries, refreshments, cereals, and beverages (Liang et al. 2004). Microalgae also serve as feed for animals (Gouveia et al. 2008a). Due to growing popularity of health foods, various food products are supplemented with microalgae. Chlorella and Spirulina are the popularly used microalgal species for the supplementation of pasta. Supplementation of pasta with C. vulgaris results in better nutritional value, enhanced sensorial properties, increased firmness, and improved swelling and water absorption (Fradique et al. 2010, Gouveia et al. 2007). Isochrysis galbana is another widely used species used for the supplementation of the food products. Supplementation of cookies with I. galbana is carried out to provide ω-3 PUFAs, besides exhibiting thermal resistance (Gouveia et al. 2008b). Phycocyanin extracts and whole A. platensis incorporation into cookies enhance the protein as well as fiber content (Singh et al. 2015). Supplementation of cookies with Haematococcus pluvialis provides antioxidant potential to the food product and also lowers the glycemic response (Hossain et al. 2017). Spirulina platensis is widely used in the supplementation of bread due to its antimicrobial activity, besides improving the protein content and mineral profile of the food product (Ak et al. 2016). Arthrospira is another popular supplement of the bread that helps to increase its protein content (Dinu et al. 2012; Achour et al. 2014; Ak et al. 2016). The techno-functional properties of microalgae are exploited as additives in food products (Caporgno and Mathys 2018). The property of microalgae to mimic fat molecules is utilized for its incorporation into emulsion resulting in reduction of percentage of oil as well as enhanced resistance to oxidation (Gouveia et al. 2006) and incorporation of microalgae into vegetarian desserts as coloring agents (Batista et al. 2008).
5.4.13 Microalgal Biological Activities
The microalgal metabolites have the following interesting biological activities:
5.4.13.1 Antioxidant Activity
Antioxidants are compounds that defend the body from the damage caused by potentially harmful molecules called as free radicals. The free radicals such as reactive oxygen and nitrogen species attack biomolecules like DNA and proteins, causing several lethal diseases like carcinoma, cardiovascular, and brain-related disorders (Ngo et al. 2006). Microalgae produce several metabolites that have strong radical scavenging action, e.g., chlorophyll a (Cho et al. 2011); phycoerythrobilin (Yabuta et al. 2010); pigment fucoxanthin and its derivatives, auroxanthin, produced by Undaria pinnatifida (Sachindra et al. 2007); and carotenoids produced by Dunaliella salina (El-Baz et al. 2017).
5.4.13.2 Anticancer Activity
Cancer, also called as malignancy, is caused by uncontrolled growth and division of cells. There are more than 100 different cancer types, a few of them lethal in nature due to their metastatic activity (Kevin et al. 2018). Microalgae produce different types of carotenoids with significant anticancer activities, β-carotene, lutein, astaxanthin, violaxanthin, and fucoxanthin. Significant inhibition of the growth of human colon cancer cell lines and LoVo colon carcinoma cells by β-carotene has been reported by Palozza et al. (2005) and Pham et al. (2013). The inhibitory effect of astaxanthin on the growth of cancer cell lines and colorectal cancer (CRC) cell lines has been studied by Palozza et al. (2009). The antiproliferative effects of lutein and violaxanthin on human colon cell line HCT-116 was studied by Shi and Chen (2002), Cha et al. (2008), Pasquet et al. (2011), Fu et al. (2013), and Talero et al. (2015). Kumar et al. (2013a, b) have reported the antiproliferative effects of fucoxanthin against different cells lines such as SK-Hep-1 and BNL CL.2. Depending on the culturing conditions, e.g., nutrients, temperature (Ingebrigtsen et al. 2016; Lauritano et al. 2016), and growth phase (Ribalet et al. 2007), the bioactivity of microalgae may vary. Lauritano et al. (2016) demonstrated that diatom Skeletonema marinoi shows anticancer activity under nitrogen starvation conditions only. Microalgae produce a repertoire of polysaccharides with anticancer activities (Raposo et al. 2015). Many microalgae present a high content of therapeutic proteins and peptides (Talero et al. 2015).
5.4.13.3 Anti-Angiogenic Activity
Angiogenesis is completely a normal process, but under certain conditions, it may become pathological and cause malignancies, cardiovascular diseases, and many other lethal diseases (Cherrington et al. 2000; Armstrong et al. 2011). Fucoxanthin and fucoxanthinol suppress the angiogenesis process in rats (Sugawara et al. 2006) and significantly inhibit blood cell multiplication. Fucoxanthin also acts as therapeutic agent as it is effective against diabetes, impedes melanin formation (Shimoda et al. 2010), prevents photooxidation of DNA (Heo and Jeon 2009), and has also been reported to promote the synthesis of arachidonic acid and DHA content in mouse livers (Tsukui et al. 2009). Siphonaxanthin, an antiangiogenic agent, is also produced by some species of algae (Ganesan et al. 2010). Aerucyclamide, an antiplasmodial agent isolated from M. aeruginosa, is also a potential candidate to be used in pharmaceutical products (Pen et al. 2012).
5.4.13.4 Anti-obesity Activity
Obesity, an abnormal or excessive fat accumulation, is a major epidemic. It is associated with a number of metabolic syndromes, type 2 diabetes, cardiovascular disease, cancer, and aging (Kopelman 2000). The underlying mechanism that causes obesity is overgrowth of adipose tissue (Wang et al. 2008). Microalgae are known to produce anti-hyperlidimic and fat-lowering agents. The fucoxanthin and fucoxanthinol predominantly produced by Cylindrotheca closterium and Phaeodactylum tricornutum not only inhibit differentiation of 3T3- L1 cells to adipocytes but also adipocyte differentiation (Hayato et al. 2006). The other biological activities fucoxanthin shows are anticancer, anti-oxidant, etc. (Maeda et al. 2007a; Kim et al. 2012). The fat in mouse feeds has been reported to be lowered by the activity of neoxanthin and fucoxanthin (Okada et al. 2008; Maeda et al. 2007b). The long-chain omega-3 fatty acid-rich oil obtained from Aurantiochytrium sp. KRS101 has been reported to decrease weight of obese animals treated with microalgal oil (Yook et al. 2015). Koo et al. (2019) documented that Phaeodactylum extract containing fucoxanthin exerts anti-obesity effects by promoting lipolysis and inhibiting lipogenesis.
5.4.13.5 Antimicrobial Activity
The antimicrobial activity of Chlorella was first demonstrated by Pratt in 1944 (Pratt et al. 1944). Microalgae owes its antimicrobial activity to several compounds like indoles, phenols, and fatty acids (Mayer and Hamann 2005; Mendiola et al. 2007). Microalgae also produce antifungal compounds like okadaic acid, ciguatoxin, and karatungiols produced by Prorocentrum lima, Gambierdiscus toxicus, and dinoflagellate Amphidinium, respectively (Washida et al. 2006). Microcystis aeruginosa possess both antifungal and antibacterial activity (Khalid et al. 2010). Dunaliella salina and Dunaliella primolecta show inhibition to a large number of microorganisms like Staphylococcus aureus, Escherichia coli, Candida albicans, Pseudomonas aeruginosa, and Klebsiella pneumonia (Mendiola et al. 2008; Pane et al. 2015). Mudimu et al. (2014) observed an in vitro antifungal activity against C. albicans by Heterochlorella luteoviridis and Porphyridium purpureum. Ginsberg et al. (1947) first demonstrated the antiviral activity of algal polysaccharides against influenza B and mumps viruses. The cell wall sulfated polysaccharide of Porphyridium sp. is reported to act against Herpes simplex viruses types 1 and 2 (HSV 1, HSV 2) and Varicella zoster virus (VZV). Scenedesmus obliquus cells and protein extracts which were rich in Arg, Lys, Asp, Ala, and His have been found to be effective against Coxsackie B3 virus (Afify et al. 2018).
5.4.13.6 Antiapoptotic Activity
Apoptosis is programmed cell death that helps the body to get rid of damaged or infected cells. It is mediated by intracellular proteolytic cascade, besides the activation and downregulation of proapoptotic and antiapoptotic genes, respectively (Nappo et al. 2012), and stated that diethyl ether extract from marine diatoms Cocconeis scutellum resulted in 89.2% apoptosis. Suh et al. (2017) reported significant inhibition of carcinogenesis and induction of cellular apoptosis through upregulation of apoptotic genes by the ethanol extract of Botryidiopsidaceae sp.
5.4.13.7 Cytotoxic Activity
Microalgae produce several groups of compounds that act as cytotoxic agents. Cyanophora paradoxa (Cp) produce pigments that can efficiently inhibit various carcinomas. The Cp water and ethanol microalgal extracts significantly inhibited the growth of cancer cell lines in vitro at 100 μg mL−1 (Baudelet et al. 2013). Ávila-Román (2019) reported the cytotoxic activity of oxylipins (OXLs) isolated from Chlamydomonas debaryana (13-HOTE) and Nannochloropsis gaditana (15-HEPE) against UACC-62 (melanoma).
5.5 Safety, Market Potential, and Current Challenges
Notwithstanding the nutritional and health aspects of microalgae, high content of nucleic acids associated with them is a major concern. The metabolic conversion of nucleic acid to uric acid may lead to the development of gout or renal calculus (Gantar and Svirčev 2008). The other important issue is that genetic characteristics and technological approaches adopted for biomass productions determine the digestibility and overall nutritional value of microalgae. For instance, toxin production, i.e., hepatotoxins and neurotoxins, by some cyanobacteria when grown in open conditions, poses a threat of microalgal biomass getting contaminated with toxins and other contaminants (Grobbelaar 2003).
Notwithstanding the acceptance of the use of microalgal biomass or myriad metabolites obtained from it, a small number of products can be seen in market. There are still a large number of avenue lying unexplored in this area and numerous challenges to be addressed to come up with innovative solutions. The most important challenge is to achieve high rate of microalgae production at economical scale. The commercial production of microalgae is being carried out in unsophisticated, low-productive man-made structures like artificial open ponds (Chisti 2007). Only a few species of microalgae such as Spirulina, Chlorella, and Dunaliella can be cultured in sustained open ponds. Despite the success of open systems, further advancements are required for closed system cultivation of microalgae. Closed photobioreactors such as tubular, flat panel, air lift, and bubble column which have been around for a long time are difficult to scale up and require high auxiliary energy, investment, and operation costs. The recovery and preservation of microalgae also require considerations. Microalgae cultures are usually very dilute suspensions with concentrations between less than 1 g/L (ponds) and 3–15 g/L (tubular or flat panel reactors) (Wijffels 2019). Thus, the recovery is a challenging task and is achieved by combining various operations such as sedimentation, flotation, filtration, and centrifugation followed by preservation of microalgal cells in order to maintain the protein quality of the biomass and the activity of other compounds of interest (Table 5.2).
5.6 Future Direction of Research
Although many compounds of high biological value and health benefits have already been discovered, microalgae still remain one of the most unexplored groups of organisms in the world, as around 97% of marine microalgal compounds are yet to be isolated and characterized (Guedes et al. 2011). This lack of information demands for intensive research in the area of bioprospecting that involves isolation, identification, and growth optimization of new and locally available microalgal strains. The discovery of hyper-producing strains and novel metabolites with various health benefits helps to improve the economics of nutraceutical production. Among the around 10,000 algae species that are believed to exist, only a few thousand strains are kept in collections, a few hundred are investigated for chemical content, and just handful microalgae, e.g., Chlorella, Dunaliella salina, and Haematococcus pluvialis, are cultivated in industrial quantities (Spolaore et al. 2006). The development of a number of transgenic algal strains boasting recombinant protein expression, engineered photosynthesis, and enhanced metabolism encourages the prospects of engineered microalgae (Rosenberg et al. 2008). The other key area of further research is the complete genome sequencing of high nutraceutical-producing microalgae. Although C. reinhardtii and 30 other organellar and whole algal genomes have reportedly been sequenced (Guarnieri and Pienkos 2015), but in order to understand and characterize the genes and enzymes involved in the production of nutraceuticals and the mechanisms that trigger the production of these metabolites, such as nutrient deprivation and biotic and abiotic stress, information on whole genome sequences is integral. Major advancements are also required to construct bioreactors or open pond systems, which are made of inexpensive and environmentally friendly materials, and development of less energy-demanding and inexpensive harvesting and extraction techniques as extraction and purification of microalgal biomass and nutraceutical compound add up to the major costs of microalgae production plants (Garcia et al. 2017). Intensive research is also required for the identification of phenolic and volatile compounds from microalgae, and furthermore, advanced techniques are required for the isolation of such compounds, since this information is missing till now due to the complexity of their isolation. Despite being rich in proteins, the dried form of microalgae as food or food substitute has failed to attract the consumers due to the fishy smell and dark green color associated with these food products. To harness the benefits of nutritious proteins from microalgae, essential strategies are needed, which would be helpful to cover its smell or taste, for instance, microencapsulation techniques (Chacón-Lee and González-Mariño 2010).
5.7 Conclusion
Microalgae are known for their potential to produce food ingredients and bioactive compounds since ancient times. The microalgal biomasses are being widely cultivated to make the commercial formulation of functional foods and nutraceutical application. Microalgae contain many valuable compounds, which includes omega-3 fatty acids, vitamins, and pigment-protein complexes besides anticancer, antioxidative, cytotoxic, and anti-obesity activities. As research interests and investment in microalgae continue to grow in various parts of the world, their role in providing health benefits and nutrition will keep expanding, and these minuscule biofactories may bring about revolutionary changes in nutraceuticals in the future.
References
Abd El-Baky HH, Shallan MA, El- Baroty G, El-Baz FK (2002) Volatile compounds of the microalga Chlorella vulgaris and their phytotoxic effect. Pak J Biol Sci 5(1):61–65
Abdo SM, Mona HH, Waleed ME, Rawheya ASED, Gamila HA (2012) Antiviral activity of freshwater algae. J Appl Pharm Sci 2:21–25
Abe K, Hattori H, Hirano M (2007) Accumulation and antioxidant activity of secondary carotenoids in the aerial microalga Coelastrella striolata var. multistriata. Food Chem 100:656–661
Abidov M, Ramazanov Z, Seifulla R, Grachev S (2010) The effects of xanthigen in the weight management of obese premenopausal women with non-alcoholic fatty liver disease and normal liver fat. Diabetes Obes Metab 12(1):72–81
Achour HY, Doumandji A, Sadi S, Saadi S (2014) Evaluation of nutritional and sensory properties of bread enriched with Spirulina. Ann Food Sci Technol 15:270–275
Adarme-Vega TC, Thomas-Hall SR, Schenk PM (2014) Towards sustainable sources for omega-3 fatty acids production. Curr Opin Biotechnol 26:14–18
Adhikary SP (2000) A preliminary survey of algae of estuaries and coastal areas in Orissa. Seaweed Res Util 22:1–5
Afify AEMR, El Bbaroty GS, El Baz FK, Abd El Baky HH, Murad AA (2018) Scendesmus obliquus: antioxidant and antiviral activity of proteins hydrolysed by three enzymes. J Genet Eng Biotechnol 16(2):399–408
Ak B, Avsaroglu E, Isık O, Ozyurt G, Kafkas E, Etyemez M, Uslu L (2016) Nutritional and physicochemical characteristics of bread enriched with microalgae Spirulina platensis. Int J Eng Res Appl 6:30–38
Algatech (2019) Corporate Website. http://www.algatech.com/. Accessed on 30 June 2019
Alsenani F, Ahmed F, Schenk PM (2015) Nutraceuticals from microalgae. In: Bagchi D, Preuss HG, Swaroop A (eds) Nutraceuticals and functional foods in human health and disease prevention. CRC Press, Boca Raton, pp 673–684
Anas AR, Kisingi T, Umezawa T, Matsuda F, Campiteli MR, Quinn RJ, Okino T (2012) Thrombin inhibitors from the freshwater cyanobacterium Anabaena compacta. J Nat Prod 75:1546–1552
Arisz SA, van Himbergen JA, Musgrave A, van den Ende H, Munnik T (2000) Polar glycerolipids of Chlamydomonas moewusii. Phytochemistry 53:265–270
Armstrong AW, Voyles SV, Armstrong EJ, Fuller EN, Rutledge JC (2011) Angiogenesis and oxidative stress: common mechanisms linking psoriasis with atherosclerosis. J Dermatol Sci 63:1–9
Asthana RK, Tripathi D, Srivastava MK, Singh A, Singh AP et al (2009) Isolation and identification of a new antibacterial entity from the Antarctic cyanobacterium Nostoc CCC 537. J Appl Phycol 21:81–88
Ávila-Román J, Taleroa E, de los Reyesb C, Zubíab E, Motilvaa V, García-Mauriñoc S (2019) Cytotoxic activity of microalgal-derived oxylipins against human cancer cell lines and their impact on ATP levels. Nat Prod Commun 11:1871–1875
Aziz N, Prasad R, Ibrahim AIM, Ahmed AIS (2017) Promising applications for the production of biofuels through algae. In: Patra JK, Vishnuprasad CN, Das G (eds) Microbial biotechnology. Springer Nature Singapore Pte Ltd, Singapore, pp 81–103
Bafanaa A (2013) Characterization and optimization of production of exopolysaccharide from Chlamydomonas reinhardtii. Carbohydr Polym 95:746–752
Bagchi SN, Sondhi S, Agrawal KM, Banerjee S (2016) An angiotensin-converting enzyme-inhibitory metabolite with partial structure of microginin in cyanobacterium Anabaena fertilissima CCC597, producing fibrinolytic protease. J Appl Phycol 28:177–180
Banker R, Carmeli S (1998) Tenuecyclamides A-D, cyclic hexapeptides from the cyanobacterium Nostoc spongiaformae var tenue. J Nat Prod 61:1248–1251
Barbosa MJ, Zijffers JW, Nisworo A, Vaes W, van Schoonhoven J, Wijffels RH (2005) Optimization of biomass, vitamins, and carotenoid yield on light energy in a flat-panel reactor using the A-stat technique. Biotechnol Bioeng 89:233–242
Barsanti L, Gualtiery P (2006) Algae: anatomy, biochemistry and biotechnology. CRC Taylor & Francis, New York
Batista AP, Gouveia L, Nunes MC, Franco JM, Raymundo A (2008) Microalgae biomass as a novel functional ingredient in mixed gel systems. In: Williams PA, Phillips GO (eds) Gums and stabilisers for the food industry. RSC Publishing, Cambridge, p 14
Baudelet PH, Gagez AL, Berard JB, Juin C, Bridian N, Kaas R, Thiery V, Cadoret JP, Picot L (2013) Antiproliferative activity of Cyanophora paradoxa pigments in melanoma, breast and lung cancer cells. Mar Drugs 11:4390–4406
Becker W (2004) Microalgae in human and animal nutrition. In: Richmond A (ed) Microalgal culture. Handbook. Blackwell, Oxford, pp 312–351
Becker EW (2007) Micro-algae as a source of protein. Biotechnol Adv 25(2):207–210
Bhadury P, Wright PC (2004) Exploitation of marine algae: biogenic compounds for potential antifouling applications. Planta 219:561–578
Bhosale P, Bernstein PS (2005) Microbial xanthophylls. Appl Microbiol Biotechnol 68:445–455
BioReal (2019) Corporate Website. http://www.bioreal.se/index.php?page=1&id=5. Accessed on 30 June 2019
Bishop WM, Zubeck HM (2012) Evaluation of microalgae for use as nutraceuticals and nutritional supplements. J Nutr Food Sci 2:147
Blanco AM, Moreno J, Del Campo JA, Rivas J, Guerrero MG (2007) Outdoor cultivation of lutein-rich cells of Muriellopsis sp. in open ponds. Appl Microbiol Biotechnol 73:1259–1266
Bleakley S, Hayes M (2017) Algal proteins: extraction, application, and challenges concerning production. Foods 6(5):33
Blue Biotech (2019) Blue Biotech Corporate Website. http://www.bluebiotech.de/. Accessed on 02 July 2019
Bong SC, Loh SP (2013) A study of fatty acid composition and tocopherol content of lipid extracted from marine microalgae, Nannochloropsis oculata and Tetraselmis suecica, using solvent extraction and supercritical fluid extraction. Int J Food Res 20:721–729
Borowitzka MA (1988) Vitamins and fine chemicals from micro-algae. In: Borowitzka MA, Borowitzka LJ (eds) Micro-algal biotechnology. Cambridge University Press, Cambridge, pp 153–196
Borowitzka MA (1997) Microalgae for aquaculture opportunities and constraints. J Appl Phycol 9:393–401
Borowitzka MA (2013) High-value products from microalgae – their development and commercialisation. J Appl Phycol 25:743–756
Boyera N, Galey I, Bernanrd BA (1998) Effect of vitamin C and its derivatives on collagen synthesis and cross-linking by normal human fibroblasts. Int J Cosmet Sci 20:151–158
Cabrita MT, Vale C, Rauter AP (2010) Halogenated compounds from marine algae. Mar Drugs 8(8):2301–2307
Caporgno MP, Mathys A (2018) Trends in microalgae incorporation into innovative food products with potential health benefits. Front Nutr 5:58. https://doi.org/10.3389/fnut.2018.00058
Carballo-Cardenas EC, Tuan PM, Janssen M, Wijffels RH (2003) Vitamin E (alpha-tocopherol) production by the marine microalgae Dunaliella tertiolecta and Tetraselmis suecica in batch cultivation. Biomol Eng 20:139–147
Cardozo KH, Guaratini T, Barros MP, Falcão VR, Tonon AP, Lopes NP, Campos S, Torres MA, Souza AO, Colepicolo P, Pinto E (2007) Metabolites from algae with economical impact. Comp Biochem Physiol C Toxicol Pharmacol 146:60–78
Carlsson AS, van Beilen JB, Möller R, Clayton D (2007) Micro- and macro-algae: utility for industrial applications. In: Bowles D (ed) Outputs from the EPOBIO: realising the economic potential of sustainable resources – bioproducts from non-food crops project. CNAP/CPL Press/University of York, York
Carreto JI, Carignan MO, Daleo G, de Marco SG (1990) Occurrence of mycosporine-like amino acids in the red tide dinoflagellate Alexandrium excavatum: UV-protective compounds. J Plankton Res 12:909–921
Ceron MC, Campos I, Sánchez JF, Acién FG, Molina E, Fernández-Sevilla JM (2008) Recovery of lutein from microalgae biomass: development of a process for Scenedesmus almeriensis biomass. J Agric Food Chem 56:11761–11766
Cha KH, Koo SY, Lee DU (2008) Antiproliferative effects of carotenoids extracted from Chlorella ellipsoidea and Chlorella vulgaris on human colon cancer cells. J Agric Food Chem 56:10521–10526
Chacòn -Lee TL, Gonźalez-Maríno GE (2010) Microalgae for “healthy” foods—possibilities and challenges. Compr Rev Food Sci Food Saf 9:655–675
Chattopadhyay P, Chatterjee S, Sen SK (2008) Biotechnological potential of natural food grade biocolorants. Afr J Biotechnol 7:2972–2985
Chauhan B, Gopal Kumar G, Kalam N, Ansari SH (2013) Current concepts and prospects of herbal nutraceutical: a review. J Adv Pharm Technol Res 4(1):4–8
Chen F (1998) Bioreactor technology for production of valuable algal products. Chin J Oceanol Limnol 16:84–90
Chen F, Li HB, Wong RNS, Ji B, Jiang Y (2005) Isolation and purification of the bioactive carotenoid zeaxanthin from the microalga Microcystis aeruginosa by high-speed counter-current chromatography. J Chromatogr 1064(2):183–186
Chen B, You W, Huang J, Yu Y, Chen W (2010a) Isolation and antioxidant property of the extracellular polysaccharide from Rhodella reticulata. World J Microbiol Biotechnol 26:833–840
Chen M, Schliep M, Willows RD, Cai ZL, Neilan BA, Scheer H (2010b) A red-shifted chlorophyll. Science 329:1318–1319
Chen J, Wang Y, Benemann JR, Zhang X, Hu H, Qin S (2015) Microalgal industry in China: challenges and prospects. J Appl Phycol 28(2):715–725. https://doi.org/10.1007/s10811-015-0720-4
Cherrington JM, Strawn LM, Shawver LK (2000) New paradigms for the treatment of cancer: the role of anti-angiogenesis agents. Adv Cancer Res 79:1–38
Chiang IZ, Huang WY, Wu JT (2004) Allelochemicals of Botryococcus braunii (Chlorophyceae). J Phycol 40:474–480
Chidambara-Murthy KN, Vanitha A, Rajesha J, Mahadeva-Swamy M, Sowmya PR, Ravishankar GA (2005) In vivo antioxidant activity of carotenoids from Dunaliella salina—a green microalga. Life Sci 76:1382–1390
Chisti Y (2007) Biodiesel from microalgae. Biotechnol Adv 25:294–306
Cho M, Lee H, Kang I, Won M, You S (2011) Antioxidant properties of extract and fractions from Enteromorpha prolifera, a type of green seaweed. Food Chem 27:999–1006
Chronakis IS, Madsen M (2011) Algal proteins. In: Phillips GO, Williams PA (eds) Handbook of food proteins. Woodhead Publishing Series in Food Sciences, Technology Nutrition, Sawston, pp 353–394
Chu WL (2012) Biotechnological applications of microalgae. Int e-J Sci Med Educ 6:24–37
Coesel SN, Baumgartner AC, Teles LM, Ramos AA, Henriques NM, Cancela L, Varela JC (2008) Nutrient limitation is the main regulatory factor for carotenoid accumulation and for Psy and Pds steady state transcript levels in Dunaliella salina (Chlorophyta) exposed to high light and salt stress. Mar Biotechnol 10:602–611
Cuvelier M-E (2001) Antioxidants. In: Morais R (ed) Functional foods: an introductory course. Escola Superior de Biotecnologia/UCP, Porto, pp 97–108
Cyanotech (2019) Cyanotech annual report. www.cyanotech.com. Accessed on 30 June 2019
D’Souza FML, Loneragan NR (1999) Effects of monospecific and mixed algae diets on survival, development and fatty acid composition of penaeid prawn (Penaeus spp.) larvae. Mar Biol 133:621–633
Dawczynski C, Schubert R, Jahreis G (2007) Amino acids, fatty acids, and dietary fibre in edible seaweed products. Food Chem 103:891–899
Day AG, Brinkmann D, Franklin S, Espina K, Rudenko G, Roberts A, Howse KS (2009) Safety evaluation of a high-lipid algal biomass from Chlorella protothecoides. Regul Toxicol Pharmacol 55:166–180
de Jesus Raposo MF, de Morais RM, De Morais AM (2013) Health applications of bioactive compounds from marine microalgae. Life Sci 93(15):479–486
de Morais MG, da Silva Vaz B, de Morais EG, Vieira Costa JA (2015) Biologically active metabolites synthesized by microalgae. Biomed Res Int 2015:835761. https://doi.org/10.1155/2015/835761
de Vries M, de Boer IJM (2010) Comparing environmental impacts for livestock products: a review of life cycle assessments. Livest Sci 128:1–11
Delattre C, Pierre G, Laroche C, Michaud P (2016) Production, extraction and characterization of microalgal and cyanobacterial exopolysaccharides. Biotechnol Adv 34:1159–1179
Demming-Adams B, Adams WW (2002) Antioxidants in photosynthesis and human nutrition. Science 298:2149–2153
Dillard CJ, German JB (2000) Phytochemicals: nutraceuticals and human health. J Sci Food Agric 80:1744–1756
Dinu M, Vlasceanu G, Dune A, Rotaru G (2012) Researches concerning the growth of nutritive value of the bread products through the Spirulina adding. J Environ Prot Ecol 13:660–665
Dionisio-Sese ML (2010) Aquatic microalgae as potential sources of UV-screening compounds. Philipp J Sci 139(1):5–16
Domínguez-Bocanegra AR, Guerrero Legarreta I, Martinez Jeronimo F, Tomasini Campocosio A (2004) Influence of environmental and nutritional factors in the production of astaxanthin from Haematococcus pluvialis. Bioresour Technol 92:209–214
Drag M, Salvensen GS (2010) Emerging principles in protease-based drug discovery. Nat Rev Drug Discov Perspect 9:690–701
Durmaz Y (2007) Vitamin E (α-tocopherol) production by the marine microalgae Nannochloropsis oculata (Eustigmatophyceae) in nitrogen limitation. Aquaculture 272:717–722
Duval B, Shetty K, Thomas WH (2000) Phenolic compounds and antioxidant properties in the snow alga Chlamydomonas nivalis after exposure to UV light. J Appl Phycol 11:559–566
Earthrise (2019) Earthrise company website. www.earthrise.com. Accessed on 30 June 2019
Egeland ES, Eikrem W, Throndsen J, Wilhelm C, Zapata M, Liaaen-Jensen S (1995) Carotenoids from further prasinophytes. Biochem Syst Ecol 23:747–755
Egeland ES, Guillard RRL, Liaaen-Jensen S (1997) Additional carotenoid prototype representatives and a general chemosystematic evaluation of carotenoids in Prasinophyceae (Chlorophyta). Phytochemistry 44:1087–1097
EID Parry (2019) EID Parry Annual Report. www.eidparry.com. Accessed on 30 June 2019
EL-Baz FK, Aly HF, Khalil WKB, Booles H, Ali GH (2017) Antineurodegenerative activity of microalgae Dunaliella salina in rats with Alzheimer’s disease. Asian J Pharm Clin Res 10(1):134–139
Eriksen NT (2008) Production of phycocyaninda pigment with applications in biology, biotechnology, foods and medicine. Appl Microbiol Biotechnol 80:1–14
Ersmark K, Del Valle JR, Hanessian S (2008) Chemistry and biology of the aeruginosin family of serine protease inhibitors. Angew Chem Int Ed 47:1202–1223
Fabregas J, Herrero C (1986) Marine microalgae as a potential source of minerals in fish diets. Aquaculture 51:237–243
Ferruzi MG, Blakeslee J (2007) Digestion, absorption, and cancer preventive activity of dietary chlorophyll derivatives. Nutr Res 27:1–12
Flora Health (2019) Corporate Website. http://www.florahealth.com/home_usa.cfm. Accessed on 30 June 2019
Food and Agriculture Organization of the United Nations (2010) Algae-based biofuels: applications and co-products. Food and Agriculture Organization of the United Nations, Rome
Fox JM, Zimba PV (2018) Minerals and trace elements in microalgae. In: Microalgae in health and disease prevention. Academic, Cambridge, pp 177–193
Fradique M, Batista AP, Nunes MC, Gouevia L, Bandarra NM, Raymundo A (2010) Incorporation of Chlorella vulgaris and Spirulina maxima biomass in pasta products. Part 1. Preparation and evaluation. J Sci Food Agric 90:1656–1664
Francavilla M, Colaianna M, Zotti MG, Morgese M, Trotta P, Tucci P, Schiavone S, Cuomo V, Trabace L (2012) Extraction, characterization and in vivo neuromodulatory activity of phytosterols from microalga Dunaliella tertiolecta. Curr Med Chem 19(18):3058–3067
Fried A, Tietz A, Benamotz A, Eichenberger W (1982) Lipid-composition of the halotolerant alga Dunaliella bardawil. Biochim Biophys Acta 713:419–426
Fu W, Gudmundsson O, Paglia G, Herjolfsson G, Andrésson OS, Palsson BØ, Brynjolfsson S (2013) Enhancement of carotenoid biosynthesis in the green microalga Dunaliella salina with light-emitting diodes and adaptive laboratory evolution. Appl Microbiol Biotechnol 97:2395–2403
Fuji Chemicals (2019) Corporate website. http://www.fujichemicals.co.jp/English/life_science_/about_astareal/index.htm. Accessed on 02 July 2019
Fujii K, Nakajima H, Ann Y (2008) Potential of Monoraphidium sp. GK12 for energy-saving astaxanthin production. J Chem Technol Biotechnol 83:1578–1584
Gademann K, Portmann C (2008) Secondary metabolites from cyanobacteria: complex structures and powerful bioactivities. Curr Org Chem 12:326–341
Galasso C, Gentile A, Orefice I, Ianora A, Bruno A, Noonan DM et al (2019) Microalgal derivatives as potential nutraceutical and food supplements for human health: a focus on cancer prevention and interception. Nutrients 11:1226. https://doi.org/10.3390/nu11061226
Ganesan P, Matsubara K, Ohkubo T, Tanaka Y, Noda K, Sugawara T, Hirata T (2010) Anti-angiogenic effect of siphonaxanthin from green alga, Codium fragile. Phytomedicine 17:1140–1144
Gangl D, Zedler JAZ, Rajkumar PD, Martinez EMR, Riseley A et al (2015) Biotechnological exploitation of microalgae. J Exp Bot 66(22):6975–6990
Gantar M, Svirčev Z (2008) Microalgae and cyanobacteria: food for thought. J Phycol 44:260–268
Garcia JL, de Vicenete M, Glan B (2017) Microalgae, old sustainable food and fashion nutraceuticals. Microbial biotechnology. John Wiley & Sons Ltd and Society for Applied Microbiology, Hoboken
García-González M, Moreno J, Manzano JC, Florencio FJ, Guerrero MG (2005) Production of Dunaliella salina biomass rich in 9-cis-b-carotene and lutein in a closed tubular photobioreactor. J Biotechnol 115(1):81–90
Giammanco M, Di Majo D, La Guardia M, Aiello S, Crescimannno M, Flandina C et al (2015) Vitamin D in cancer chemoprevention. Pharm Biol 53:1399–1434
Giner JL, Zhao H, Boyer GL, Satchwell MF, Andersen RA (2009) Sterol chemotaxonomy of marine pelagophyte algae. Chem Biodivers 26(7):1111–1130
Ginsberg RH, Goebel WF, Horsfall FL (1947) Inhibition of mumps virus multiplication by a polysaccharide. Proc Soc Exp Biol Med 66:99–100
Goiris K, Muylaert K, Voorspoels S, Noten B, Paepe DD et al (2014) Detection of flavonoids in microalgae from different evolutionary lineages. J Phycol 50(3):485–492
Gouveia L, Oliveira AC (2009) Microalgae as a raw material for biofuels production. J Ind Microbiol Biotechnol 36:269–274
Gouveia L, Raymundo A, Batista AP, Sousa I, Empis J (2006) Chlorella vulgaris and Haematococcus pluvialis biomass as colouring and antioxidant in food emulsions. Eur Food Res Technol 222:362–367
Gouveia L, Batista AP, Miranda A, Empis J, Raymundo A (2007) Chlorella vulgaris biomass used as coloring source in traditional butter cookies. Innovative Food Sci Emerg Technol 8:433–436
Gouveia L, Batista AP, Sousa I, Raymundo A, Bandarra NM (2008a) Microalgae in novel food products. In: Papadopoulos KN (ed) Food chemistry research developments. Nova Science Publishers Inc., Hauppauge, pp 75–112
Gouveia L, Coutinho C, Mendonça E, Batista AP, Sousa I, Bandarra NM et al (2008b) Functional biscuits with PUFA- ω 3 from Isochrysis galbana. J Sci Food Agric 88:891–896
Grobbelaar J (2003) Quality control and assurance: crucial for the sustainability of the applied phycology industry. J Appl Phycol 15:209–215
Gruber BM (2016) BMB-group vitamins: chemoprevention? Adv Clin Exp Med 25:561–568
Guarnieri MT, Pienkos PT (2015) Algal omics: unlocking bioproduct diversity in algae cell factories. Photosynth Res 123:255–263
Guedes AC, Amaro HM, Malcata FX (2011) Microalgae as sources of carotenoids. Mar Drugs 9(4):625–644
Hajimahmoodi M, Faramarzi MA, Mohammadi N, Soltani N, Oveisi MR, Nafissi-Varcheh N (2010) Evaluation of antioxidant properties and total phenolic contents of some strains of microalgae. J Appl Phycol 22(1):43–50
Hanessian S, Del Valle JR, Xue Y, Blomberg N (2006) Total synthesis and structural confirmation of chlorodysinosin A. J Am Chem Soc 128(32):10491–10495
Hannach G, Sigleo AC (1998) Photoinduction of UV-absorbing compounds in six species of marine phytoplankton. Mar Ecol Prog Ser 174:207–222
Hassimotto NMA, Genovese MI, Lajolo FM (2005) Antioxidant activity of dietary fruits, vegetables, and commercial frozen fruit pulps. J Agric Food Chem 53(8):2928–2935
Hayashi T, Hayashi K, Maeda M, Kojima I (1996) Calcium spirulan, an inhibitor of enveloped virus replication, from a blue-green alga Spirulina platensis. J Nat Prod 59:83–87
Hayashi K, Kanekiyo K, Ohta Y, Lee JB, Takenaka H, Hayashi T (2008) Anti influenza A virus activity of an acidic polysaccharide from a blue green alga Nostoc flagelliforme. Planta Med 74:PA34. https://doi.org/10.1055/s-0028-1084032
Hayato M, Masashi H, Tokutake S, Nobuyuk T, Teruo K, Kazuo M (2006) Fucoxanthin and its metabolite, fucoxanthinol, suppress adipocyte differentiation in 3T3-L1 cells. Int J Mol Med 18:147–152
Hayes M, Skomedal H, Skjanes K, Mazur-Marzec H, Torunska-Sitarz A, Catala M, Hosoglu MI, García-Vaquero M (2017) Microalgal proteins for feed, food and health. In: Gonzalez-Fernandez C, Munoz R (eds) Microalgae-based biofuels and bioproducts. Woodhead publishers Elsevier, Sawston, pp 348–368
He J, Yang Y, Xu H, Zhang X, Li X-M (2005) Olanzapine attenuates the okadaic acid-induced spatial memory impairment and hippocampal cell death in rats. Neuropsychopharmacology 30(8):1511–1520
Hejazi MA, Wijffels RH (2003) Effect of light intensity on beta-carotene production and extraction by Dunaliella salina in two-phase bioreactors. Biomol Eng 20:171–175
Helbling EW, Chalker BE, Dunlap WC, Osmund HH, Villafane VE (1996) Photoacclimation of Antarctic diatoms to solar ultraviolet radiation. J Exp Mar Biol Ecol 204:85–101
Henderson RJ, Mackinlay EE (1999) Polyunsaturated fatty acid metabolism in the marine dinoflagellate Crypthecodinium cohnii. Phytochemistry 30(6):1781–1787
Heo SJ, Jeon YJ (2009) Protective effect of fucoxanthin isolated from Sargassum siliquastrum on UV-B induced cell damage. J Photochem Photobiol B 95:101–107
Herrero M, Ibáñez E, Cifuentes A, Reglero G, Santoyo S (2006) Dunaliella salina microalga pressurized extracts as potential antimicrobials. J Food Prot 69:2471–2477
Higuera-Ciapara I, Felix-Valenzuela L, Goycoolea FM (2006) Astaxanthin: a review of its chemistry and applications. Crit Rev Food Sci Nutr 46:185–196
Hossain AKM, Brennan MA, Mason SL, Guo X, Zeng XA, Brennan CS (2017) The effect of astaxanthin-rich microalgae Haematococcus pluvialis and wholemeal flours incorporation in improving the physical and functional properties of cookies. Foods 6(57):1–10
Hosseini Tafreshi A, Shariati M (2009) Dunaliella biotechnology: methods and applications. J Appl Microbiol 107(1):14–35
Huang J, Chen B, You W (2005) Studies on separation of extracellular polysaccharide from Porphyridium cruentum and its anti-HBV activity in vitro. Chin J Mar Drugs 24:18–21
Ibañez E, Cifuentes A (2013) Benefits of using algae as natural sources of functional ingredients. J Sci Food Agric 93(4):703–709
Ibáñez E, Mendiola JA, Rodríguez-Meizoso I, Señořans FS, Reglero G, Cifuentes A (2008) Antioxidants in plant foods and microalgae extracted using compressed fluids. Elec J Env Agricult Food Chem 7(8):3301–3309
Ikeuchi M, Koyama T, Takahashi J, Yazawa K (2007) Effects of astaxanthin in obese mice fed a high-fat diet. Biosci Biotechnol Biochem 71(4):893–899
Ingebrigtsen RA, Hansen E, Andersen JH, Eilertsen HC (2016) Light and temperature effects on bioactivity in diatoms. J Appl Phycol 28:939–950
Ishida K, Nakagawa H, Murakami M (2000) Microcyclamide, a cytotoxic cyclic hexapeptide from the cyanobacterium Microcystis aeruginosa. J Nat Prod 63(9):1315–1317
Ismail A, Marjan ZM, Foong CW (2004) Total antioxidant activity and phenolic content in selected vegetables. Food Chem 87(4):581–586
Jaime L, Mendiola JA, Ibañez E, Martin-Alvarez PJ, Cifuentes A, Reglero G, Señorans FJ (2007) β-carotene isomer composition of sub- and supercritical carbon dioxide extracts. Antioxidant activity measurement. J Agric Food Chem 55:10585–10590
Janssen MLE (2019) Cyanobacterial peptides beyond microcystins – a review on co-occurrence, toxicity, and challenges for risk assessment. Water Res 151:488–499
Jena M, Ratha SK, Adhikary SP (2005) Algal diversity changes in Kathajodi river after receiving sewage of Cuttack and its ecological implication. Indian Hydrobiol 8:67–74
Jena M, Ratha SK, Adhikary SP (2008) Algal diversity in Rushikulya river, Orissa from origin till confluence to the sea. Indian Hydrobiol 11:9–24
Jin E, Feth B, Melis A (2003) A mutant of the green alga Dunaliella salina constitutively accumulates zeaxanthin under all growth conditions. Biotechnol Bioeng 81(1):115–124
Jin E, Lee CG, Polle JEW (2006) Secondary carotenoid accumulation in Haematococcus (Chlorophyceae): biosynthesis, regulation, and biotechnology. J Microbiol Biotechnol 16:821–831
Jungblut AD, Neilan BA (2006) Molecular identification and evolution of the cyclic peptide hepatotoxins, microcystin and nodularin, synthetase genes in three orders of cyanobacteria. Arch Microbiol 185:107–114
Kamath BS, Srikanta BM, Dharmesh SM, Sarada R, Ravishankar CA (2008) Ulcer preventive and antioxidative properties of astaxanthin from Haematococcus pluvialis. Eur J Pharmacol 590:387–395
Kang CD, Lee JS, Park TH, Sim SJ (2005) Comparison of heterotrophic and photoautotrophic induction on astaxanthin production by Haematococcus pluvialis. Appl Microbiol Biotechnol 68:237–241
Karsten U, Maier J, Garcia-Pichel F (1998) Seasonality in UV-absorbing compounds of cyanobacterial mat communities from an intertidal mangrove flat. Aquat Microb Ecol 16:37–44
Karsten U, Lembcke S, Schumann R (2007) The effects of ultraviolet radiation on photosynthetic performance, growth and sunscreen compounds in aeroterrestrial biofilm algae isolated from building facades. Planta 225:991–1000
Katircioglu H, Akin BS, Atici T (2004) Microalgal toxin(s): characteristics and importance. Afr J Biotechnol 3(12):667–674
Kevin A, Andrade M, Lauritano C, Romano G, Ianora A (2018) Marine microalgae with anti-cancer properties. Mar Drugs 16(5):165. https://doi.org/10.3390/md16050165
Khalid MN, Shameel M, Ahmad VU, Shahzad S, Leghari SM (2010) Studies on the bioactivity and phycochemistry of Microcystis aeruginosa (Cyanophycota) from Sindh. Pak J Bot 42:2635–2646
Khan MI, Shin JH, Kim JD (2018) The promising future of microalgae: current status, challenges, and optimization of a sustainable and renewable industry for biofuels, feed, and other products. Microb Cell Factories 17:36. https://doi.org/10.1186/s12934-018-0879-x
Kharkongar D, Ramanujam P (2014) Diversity and species composition of subaerial algal communities in forested area of Meghalaya, India. Int J Biodiversity 2014:1–10. https://doi.org/10.1155/2014/456202
Kim SM, Jung YH, Kwon O, Cha KH, Um BH (2012) A potential commercial source of fucoxanthin extracted from the microalga Phaeodactylum tricornutum. Appl Biochem Biotechnol 166:1843–1855
Kleinegris DM, van Es MA, Janssen M, Brandenburg WA, Wijffels RH (2010) Carotenoid fluorescence in Dunaliella salina. J Appl Phycol 22(5):645–649
Kohlhase M, Pohl P (1988) Saturated and unsaturated sterols of nitrogen-fixing blue-green algae (cyanobacteria). Phytochemistry 27:1735–1740
Komaristaya VP, Gorbulin OS (2006) Sporopollenin in the composition of cell walls of Dunalliela salina Teod. (Chlorophyta) zygotes. Int J Algae 8:43–52
Koo SY, Hwang JH, Yang SH, Um JI, Hong KW, Kang K, Pan CH, Hwang KT, Kim SM (2019) Anti-obesity effect of standardized extract of microalga Phaeodactylum tricornutum containing fucoxanthin. Mar Drugs 17:311. https://doi.org/10.3390/md17050311
Kopelman PG (2000) Obesity as a medical problem. Nature 404:635–643
Kumar D, Adhikary SP (2018) Exopolysaccharides from cyanobacteria and microalgae and their commercial application. Curr Sci 115(2):234–231
Kumar M, Tripathi MK, Srivastava A, Gour JK, Singh RK, Tilak R, Asthana RK (2013a) Cyanobacteria, Lyngya aestuarii and Aphanothece bullosa as antifungal and antileishmanial drug resources. Asian Pac J Trop Biomed 3:458–463
Kumar SR, Hosokawa M, Miyashita K (2013b) Fucoxanthin: a marine carotenoid exerting anti-cancer effects by affecting multiple mechanisms. Mar Drugs 11:5130–5147
Kumar D, Dhar DW, Pabbi S, Kumar N, Walia S (2014) Extraction and purification of C-phycocyanin from Spirulina platensis (CCC540). Indian J Plant Physiol 19(2):184–188
La Barre S, Potin P, Leblanc C, Delage L (2010) The halogenated metabolism of brown algae (Phaeophyta), its biological importance and its environmental significance. Mar Drugs 8(4):988–1010
Lauritano C, Andersen JH, Hansen E, Albrigtsen M, Escalera L, Esposito F, Helland K, Hanssen KØ, Romano G, Ianora A (2016) Bioactivity screening of microalgae for antioxidant, anti-inflammatory, anticancer, anti-diabetes and antibacterial activities. Front Mar Sci 3:1–12. https://doi.org/10.3389/fmars.2016.00068
Leblond JD, Timofte HI, Roche SA, Porter NM (2011) Sterols of glaucocystophytes. Phycol Res 59:129–134
Lee JH, Kim HS, Seo HH, Song MY, Kulkarni A, Choi YH, Kim KW, Moh SH (2015) Antiaging effects of algae-derived mycosporine-like amino acids (MAAs) on skin. In: Farage M, Miller K, Maibach H (eds) Textbook of aging skin. Springer, Berlin
Lenoir-Wijnkoop I, Dapoigny M, Dubois D, van Ganse E, Gutiérrez-Ibarluzea I, Hutton J, Jones P, Mittendorf T, Poley MJ, Salminen S, Nuijten MJ (2010) Nutrition economics–characterising the economic and health impact of nutrition. Br J Nutr 105(1):157–166
Leu S, Boussiba S (2014) Advances in the production of high-value products by microalgae. Indus Biotechnol 10:169–183
Leya T, Rahn A, Lütz C, Remias D (2009) Response of arctic snow and permafrost algae to high light and nitrogen stress by changes in pigment composition and applied aspects for biotechnology. FEMS Microbiol Ecol 67:432–443
Li HB, Cheng KW, Wong CC, Fan KW, Chen F, Jiang Y (2007) Evaluation of antioxidant capacity and total phenolic content of different fractions of selected microalgae. Food Chem 102(3):771–776
Liang S, Liu X, Chen F, Chen Z (2004) Current microalgal health food R & D activities in China. In: Ang PO (ed) Asian Pacific Phycol. 21st century Prospect. Challenges. Springer, Dordrecht, pp 45–48
Lin JY, Tang CY (2007) Determination of total phenolic and flavonoid contents in selected fruits and vegetables, as well as their stimulatory effects on mouse splenocyte proliferation. Food Chem 101(1):140–147
Lin J, Huang L, Yu J, Xiang S, Wang J, Zhang J, Yan X, Cui W, He S, Wang Q (2016) Fucoxanthin, a marine carotenoid, reverses scopolamine-induced cognitive impairments in mice and inhibits acetylcholinesterase in vitro. Mar Drugs 14:67. https://doi.org/10.3390/md14040067
Liu J, Chen F (2016) Biology and industrial applications of Chlorella: advances and prospects. Adv Biochem Eng Biotechnol 153:1–35
Liu BH, Lee YK (2000) Secondary carotenoids formation by the green alga Chlorococcum sp. J Appl Phycol 12:301–307
Llewellyn CA, Airs RL (2010) Distribution and abundance of MAAs in 33 species of microalgae across 13 Classes. Mar Drugs 8(4):1273–1291
Lobo V, Patil A, Phatak A, Chandra N (2010) Free radicals, antioxidants and functional foods: impact on human health. Pharmacogn Rev 4(8):118–126
Lorenz RT, Cysewski GR (2000) Commercial potential for Haematococcus microalgae as a natural source of astaxanthin. Trends Biotechnol 18:160–167
Luo X, Su P, Zhang W (2015) Advances in microalgae- derived phytosterols for functional food and pharmaceutical applications. Mar Drugs 13:4231–4254
Maeda H, Hosokawa M, Sashima T, Funayama K, Miyashita K (2007a) Effect of medium-chain triacylglycerols on anti-obesity effect of fucoxanthin. J Oleo Sci 56(12):615–621
Maeda H, Hosokawa M, Sashima T, Miyashita K (2007b) Dietary combination of fucoxanthin and fish oil attenuates the weight gain of white adipose tissue and decreases blood glucose in obese/diabetic KK-Ay mice. J Agric Food Chem 55:7701–7706
Mahendran S, Saravanan S, Vijayabaskar P, Anandapandian KTK, Shankar T (2013) Antibacterial potential of microbial exopolysaccharide from Ganoderma lucidum and Lysinibacillus fusiformis. Int J Recent Sci Res 4:501–505
Malathi T, Babu MR, Mounika T, Snehalatha D, Rao BD (2014) Screening of cyanobacterial strains for antibacterial activity. Phykos 44:6–11
Markou G, Angelidaki I, Georgakakis D (2012) Microalgal carbohydrates: an overview of the factors influencing carbohydrates production, and of main bioconversion technologies for production of biofuels. Appl Microbiol Biotechnol 95:631–645. https://doi.org/10.1007/s00253-012-4398-0
Martinez-Fernandez E, Acosta-Salmon H, Southgate PC (2006) The nutritional value of seven species of tropical microalgae for blacklip pearl oyster (Pinctada margaritifera, L.) larvae. Aquaculture 257:491–503
Mathew BR, Sankaranarayanan P, Nair C, Varghese T, Smanathon B et al (1995) Evaluation of chemoprevention of oral cancer with Spirulina fusiformis. Nutr Cancer 24:197–202
Matsukawa R, Hotta M, Masuda Y, Chihara M, Karube I (2000) Antioxidants from carbon dioxide fixing Chlorella sorokiniana. J Appl Phycol 12:263–267
Mayer AMS, Hamann MT (2005) Marine pharmacology in 2001–2002: marine compounds with anthelmintic, antibacterial, anticoagulant, antidiabetic, antifungal, anti-inflammatory, antimalarial, antiplatelet, antiprotozoal, antituberculosis, and antiviral activities; affecting the cardiovascular, immune and nervous systems and other miscellaneous mechanisms of action. Comp Biochem Physiol Part C Toxicol Pharmacol 140:265–286
Mazur-Marzec H, Kaczkowska MJ, Blaszczyk A, Ackaalan R, Spoof L, Meriluoto J (2013) Diversity of peptides produced by Nodularia spumigena from various geographical regions. Mar Drugs 11:1–19
Medina AR, Grima EM, Giměnez AG, González MJI (1997) Lipase-catalyzed esterification of glycerol and polyunsaturated fatty acids from fish and microalgae oils. Biotechnol Adv 16:517–580
Mendes RL, Nobre BP, Cardoso MT, Pereira AP, Palavra AF (2003) Supercritical carbon dioxide extraction of compounds with pharmaceutical importance from microalgae. Inorg Chim Acta 356:328–334
Mendiola JA, Maŕın FR, Herńandez SF, Arredondo BO, Sěnoŕans FJ, Ibánez E, Reglero G (2005) Characterization via liquid chromatography coupled to diode array detector and tandem mass spectrometry of supercritical fluid antioxidant extracts of Spirulina platensis microalga. J Sep Sci 28:1031–1038
Mendiola JA, Torres CF, Martín-Alvarez PJ, Santoyo S, Toré A, Arredondo BO, Señoráns FJ, Cifuentes A, Ibáñez E (2007) Use of supercritical CO2 to obtain extracts with antimicrobial activity from Chaetoceros muelleri microalga. A correlation with their lipidic content. Eur Food Res Technol 224:505–510
Mendiola JA, Santoyo S, Cifuentes A, Reglero G, Ibanez E, Senorans FJ (2008) Antimicrobial activity of sub- and supercritical CO2 extracts of the green alga Dunaliella salina. J Food Prot 71:2138–2143
Merapharma (2019) Corporate website. https://www.merapharma.com/about-mera.html. Accessed on 02 July 2019
Mishra VK, Bacheti RK, Husen A (2011) Medicinal uses of chlorophyll: a critical overview. In: Le H, Salcedo E (eds) Chlorophyll: structure, function and medicinal. Uses Publisher, Nova Science Publishers, Inc, Hauppauge
Mišurcová L, Buňka F, Vávra Ambrožová J, Machů L, Samek D, Kráčmar S (2014) Amino acid composition of algal products and its contribution to RDI. Food Chem 151:120–125
Mobin S, Alam F (2017) Some promising microalgal species for commercial applications: a review. Energy Procedia 110:510–517
Moore RE, Patterson GML, Carmichael WW (1988) New pharmaceuticals from cultured blue green algae. In: Fautin DG (ed) Biomedical importance of marine organisms. California Academy of Sciences, San Francisco, pp 143–150
Morinoto T, Nagatu A, Murakami N (1995) Anti-tumor promoting glyceroglycolipid from green alga Chlorella vulgaris. Phytochemistry 40:1433–1437
Mudimu O, Rybalka N, Bauersachs T, Born J, Friedl T, Schulz R (2014) Biotechnological screening of microalgal and cyanobacterial strains for biogas production and antibacterial and antifungal effects. Metabolites 4:373–393
Mukund S, Sivasubramanian V (2014) Anticancer activity of Oscillatoria terebriformis, cyanobacteria in human lung cancer cell line A549. Int J Appl Biol Pharm Technol 5:34–45
Mulders KJM, Lamers PP, Martens DE, Wijffels RH (2014) Phototropic pigment production with microalgae: biological constraints and opportunities. J Phycol 50:229–242
Murakami M, Suzuki S, Itou Y, Kodani S, Ishida K (2000) New anabaenopeptins, potent carboxypeptidase-A inhibitors from the cyanobacterium Aphanizomenon flos-aquae. J Nat Prod 63:1280–1282
Mus F, Toussaint JP, Cooksey KE, Fields MW, Gerlach R, Peyton BM et al (2013) Physiological and molecular analysis of carbon source supplementation and pH stress- induced lipid accumulation in the marine diatom Phaeodactylum tricornutum. Appl Microbiol Biotechnol 97:3625–3642
Mussgnug JH, Klassen V, Schlüter A, Kruse O (2010) Microalgae as substrates for fermentative biogas production in a combined biorefinery concept. J Biotechnol 150(1):51–56
Nakamura Y, Takahashi J, Sakurai A, Inaba Y, Suzuki E, Nihei S et al (2005) Some cyanobacteria synthesize semiamylopectin type α-Polyglucans instead of glycogen. Plant Cell Physiol 46(3):539–545
Nappo M, Berkov S, Massucco C, Di Maria V, Bastida J, Codina C et al (2012) Apoptotic activity of the marine diatom Cocconeis scutellum and eicosopentaenoic acid in BT20 cells. Pharm Biol 50:529–535
Ng DHP, Ng YK, Shen H, Lee YK (2015) Microalgal biotechnology: the way forward. In: Kim SK (ed) Handbook of marine microalgae. Elsevier, Amsterdam, pp 69–80
Ngo DN, Kim MM, Kim SK (2006) Chitin oligosaccharides inhibit oxidative stress in live cells. Carbohydr Polym 74:228–234
Nicoletti M (2012) Nutraceuticals and botanicals: overview and perspectives. Int J Food Sci Nutr 63:2–6
Nose T (1972) Mineral requirements. Suisan Zoshoku 20:289–300
Nunes-Alves C, Booty MG, Carpenter SM, Jayaraman P, Rothchild AC, Behar SM (2014) In search of a new paradigm for protective immunity to TB. Nat Rev Microbiol 12:289–299
Okada T, Nakai M, Maeda H, Hosokawa M, Sashima T, Miyashita K (2008) Suppressive effect of neoxanthin on the differentiation of 3T3-L1 adipose cells. J Oleo Sci 57(6):345–351
Olaizola M (2005) The health benefits of Haematococcus astaxanthin: cardiovascular health. Agro Food Ind Hi-Tech 16:35–37
Ozdemir G, Karabay NU, Dalay MC, Pazarbasi B (2004) Antibacterial activity of volatile component and various extracts of Spirulina platensis. Phytother Res 18(9):754–757
Palozza P, Serini S, Maggiano N, Tringali G, Navarra P, Ranelletti FO, Calviello G (2005) Beta-carotene downregulates the steady-state and heregulin-alpha-induced COX-2 pathways in colon cancer cells. J Nutr 135:129–136
Palozza P, Torelli C, Boninsegna A, Simone R, Catalano A, Mele MC, Picci N (2009) Growth-inhibitory effects of the astaxanthin-rich alga Haematococcus pluvialis in human colon cancer cells. Cancer Lett 283:108–117
Pane G, Cacciola G, Giacco E, Mariottini GL, Coppo E (2015) Assessment of the antimicrobial activity of algae extracts on bacteria responsible of external Otitis. Mar Drugs 13(10):6440–6452
Pangestuti R, Kim SK (2011) Biological activities and health benefit effects of natural pigments derived from marine algae. J Funct Foods 3(4):255–266
Parry Nutraceuticals (2019) Parry Nutraceuticals website. http://www.parrynutraceuticals.com/food-ingredients.Aspx
Pasquet V, Morisset P, Ihammouine S, Chepiedm A, Aumailley L, Berard JB et al (2011) Antiproliferative activity of violaxanthin isolated from bioguided fractionation of Dunaliella tertiolecta extracts. Mar Drugs 9:819–831
Patil V, Källqvist T, Olsen E, Vogt G, Gislerød HR (2007) Fatty acid composition of 12 microalgae for possible use in aquaculture feed. Aquac Int 15:1–9
Pattanaik A, Sukla LB, Pradhan D, Shukla V (2019) Artificial intelligence and virtual environment for microalgal source for production of nutraceuticals. Biomed J Sci Tech Res 13(5):4362–4367. https://doi.org/10.26717/BJSTR.2019.13.002459
Pelah D, Sintov A, Cohen E (2004) The effect of salt stress on the production of canthaxanthin and astaxanthin by Chlorella zofingiensis grown under limited light intensity. World J Microbiol Biotechnol 20:483–486
Pen S, Scarone L, Manta E, Stewart L, Yardley V et al (2012) Synthesis of a Microcystis aeruginosa predicted metabolite with antimalarial activity. Bioorg Med Chem Lett 22(15):4994–4997
Pereira H, Barreira L, Figueiredo F, Custodio L, Vizetto-Duarte C, Polo C et al (2012) Polyunsaturated fatty acids of marine macroalgae: potential for nutritional and pharmaceutical applications. Mar Drugs 10:1920–1935
Pham DNT, Leclerc D, Levesque N, Deng L, Rozen R (2013) β-Carotene 15,15′-monooxygenase and its substrate β-carotene modulate migration and invasion in colorectal carcinoma cells. Am J Clin Nutr 98:413–422
Piironen V, Lindsay DG, Miettinen TA, Toivo J, Lampi AM (2000) Plant sterols: biosynthesis, biological function and their importance to human nutrition. J Sci Food Agric 80:939–966
Piorreck M, Baasch KH, Pohl P (1984) Biomass production, total protein, chlorophylls, lipids and fatty acids of freshwater green and bluegreen algae under different nitrogen regimes. Phytochemistry 23:207–216
Plaza M, Herrero M, Cifuentes A, Ibanez E (2009) Innovative natural functional ingredients from microalgae. J Agric Food Chem 57:7159–7170
Plaza M, Santoyo S, Jaime L, Reina GG, Herrero M, Senorans FJ, Ibanez E (2010) Screening for bioactive compounds from algae. J Pharm Biomed Anal 51:450–455
Ploutno A, Carmeli S (2000) Nostocyclyne A. A novel antimicrobial cyclophane from the cyanobacterium Nostoc sp. J Nat Prod 63:1524–1526
Poerschmann J, Spijkerman E, Langer U (2004) Fatty acid patterns in Chlamydomonas sp. as a marker for nutritional regimes and temperature under extremely acidic conditions. Microb Ecol 48:78–89
Ponomarenko LP, Stonik IV, Aizdaicher NA, Orlova TY, Popovskaya GI, Pomazkina GV, Stonik VA (2004) Sterols of marine microalgae Pyramimonas cf. cordata (Prasinophyta), Attheya ussurensis sp. nov. (Bacillariophyta) and a spring diatom bloom from Lake Baikal. Comp Biochem Physiol B Biochem Mol Biol 138:65–70
Portmann C, Blom JF, Gademann K, Juttner F (2008) Aerucyclamides A and B: isolation and synthesis of toxic ribosomal heterocyclic peptides from the cyanobacterium Microcystis aeruginosa PCC 7806. J Nat Prod 71(7):1193–1196
Prasanna R, Kaushik BD (2005) Algal diversity from morphology to molecules. In: Satyanarayana T, Johri BN (eds) Microbial diversity: current perspectives and potential applications. IK International, New Delhi, pp 323–344
Pratt R, Daniels TC, Eiler JJ, Gunnison JB, Kumler WD, Oneto JF et al (1944) Chlorellin, an antibacterial substance from Chlorella. Science 99:351–352
Pro Aigen Biotech (2019) https://www.indiamart.com/proalgen-biotech-limited/. Accessed on 30 June 2019
Pulz O, Gross W (2004) Valuable products from biotechnology of microalgae. Appl Microbiol Biotechnol 65(6):635–648
Rabbani S, Beyer P, Lintig J, Hugueney P, Kleinig H (1998) Induced beta carotene synthesis driven by triacylglycerol deposition in the unicellular alga Dunaliella bardawil. Plant Physiol 116:1239–1248
Rafika C, Lamia T, Rym BD, Omeya EA, Ali Y, Khamissa G, Jihen BA, Hela O, Ouada HB (2011) Evaluation of cytotoxicity and biological activities in extracellular polysaccharides released by cyanobacterium Arthrospira platensis. Braz Arch Biol Technol 54:831–838
Rajasekaran A, Sivagnanam G, Xavier R (2008) Nutraceuticals as therapeutic agents: a review. Res J Pharm Technol 1:328–340
Ranga RA, Vijaya RD, Ravishankar GA (2017) Secondary metabolites from algae for nutraceutical application. Novel Tech Nutr Food Sci 1(1). https://doi.org/10.31031/NTNF.2017.01.000503
Rao SD, Raghuramulu N (1996) Food chain as origin of vitamin D in fish. Comp Biochem Physiol Part A Physiol 114:15–19
Raposo MFJ, Mendes-Pinto MM, Morais R (2001) Carotenoids, foodstuff and human health. In: Morais R (ed) Functional foods an introductory course. Universidade Católica Portuguesa—Escola Superior de Biotecnologia, Porto
Raposo MFJ, de Morais RMSC, Bernardo de Morais AMM (2013) Bioactivity and applications of sulphated polysaccharides from marine microalgae. Mar Drugs 11(1):233–252
Raposo MF, de Morais AM, de Morais RM (2014) Influence of sulphate on the composition and antibacterial and antiviral properties of the exopolysaccharide from Porphyridium cruentum. Life Sci 101:56–63
Raposo MFJ, Morais AMB, Morais RMS (2015) Marine polysaccharides from algae with potential biomedical applications. Mar Drugs 13:2967–3028
Rath J, Adhikary SP (2005) A check list of algae from Chilika Lake, Orissa. Nelumbo Bull Bot Surv India 47:101–114
Rath B, Priyadarshani I (2013) Antibacterial and antifungal activity of marine cyanobacteria from Odisha coast. Int J Curr Trends Res 2:248–251
Ratha SK, Naik K, Padhi SB (2003) Epiphytic algal diversity associated with different aquatic macrophytes of freshwater ponds in and around Berhampur University campus, Orissa. Nat Environ Pollut Technol 2:205–208
Ratha SC, Prasanna R, Gupta V, Dhar DW, Saxena AK (2012) Bioprospecting and indexing the microalgal diversity of different ecological habitats of India. World J Microbiol Biotechnol 28:1657–1667
Reitan KI, Rainuzzo JR, Olsen Y (1994) Effect of nutrient limitation on fatty acid and lipid content of marine microalgae. J Phycol 30:972–979
Remias D, Lütz-Meindl U, Lütz C (2005) Photosynthesis, pigments and ultrastructure of the alpine snow alga Chlamydomonas nivalis. Eur J Phycol 40:259–268
Ribalet F, Wichard T, Pohnert G, Ianora A, Miralto A, Casotti R (2007) Age and nutrient limitation enhance polyunsaturated aldehyde production in marine diatoms. Phytochemistry 68:2059–2067
Riegger L, Robinson D (1997) Photoinduction of UV-absorbing compounds in Antarctic diatoms and Phaeocystis antarctica. Mar Ecol Prog Ser 160:13–25
Rodriguez-Garcia I, Guil-Guerrero JL (2008) Evaluation of the antioxidant activity of three microalgal species for use as dietary supplements and in the preservation of foods. Food Chem 108:1023–1026
Rodriguez-Sanchez R, Ortiz-Butron R, Blas-Valdivia V, Hernandez-Garcia A, Cano-Europa E (2012) Phycobiliproteins or C-phycocyanin of Arthrospira (Spirulina) maxima protect against HgCl2-caused oxidative stress and renal damage. Food Chem 135:2359–2365
Rosenberg JN, Oyler GA, Wilkinson L, Betenbaugh MJ (2008) A green light for engineered algae: redirecting metabolism to fuel a biotechnology revolution. Curr Opin Biotechnol 19:430–436
Roy KR, Arunasree KM, Dhoot A, Aparna R, Reddy GV, Vali S, Reddanna P (2007) C-phycocyanin inhibits 2-acetylaminofluorene-induced expression of MDR1 in mouse macrophage cells: ROS mediated pathway determined via combination of experimental and in silico analysis. Arch Biochem Biophys 459:169–177
Roy S, Llewellyn CA, Egeland ES, Johnson G (2011) Phytoplankton pigments: characterization, chemotaxonomy, and applications in oceanography. Cambridge University Press, New York
Sachindra N, Sato E, Maeda H, Hosokawa M, Niwano Y, Kohno M et al (2007) Radical scavenging and singlet oxygen quenching activity of marine carotenoid fucoxanthin and its metabolites. J Agric Food Chem 55:8516–8522
Samantraray SM, Mallick AK, Padhy SB (2002) Chilika environment and its algal vegetation. Seaweed Res Util 24:13–17
Sanchez JF, Fernández-Sevilla JM, Acién FG, Cerón MC, Pérez-Parra J, Molina-Grima E (2008) Biomass and lutein productivity of Scenedesmus almeriensis: influence of irradiance, dilution rate and temperature. Appl Microbiol Biotechnol 79:719–729
Santiago-Morales IS, Trujillo-Valle L, Márquez-Rocha FJ, López Hernández JF (2018) Tocopherols, phycocyanin and superoxide dismutase from microalgae as potential food antioxidants. Appl Food Biotechnol 5:19–27
Sathasivam R, Kermanee P, Roytrakul S, Juntawong N (2012) Isolation and molecular identification of β-carotene producing strains of Dunaliella salina and Dunaliella bardawil from salt soil samples by using species-specific primers and internal transcribed spacer (ITS) primers. Afr J Biotechnol 11(34):8425–8432
Satoh A, Tsuji S, Okada Y, Murakami N, Urami M, Nakagawa K, Ishikura M, Katagiri M, Koga Y, Shirasawa T (2009) Preliminary clinical evaluation of toxicity and efficacy of a new astaxanthin-rich Haematococcus pluvialis extract. J Clin Biochem Nutr 44:280–284
Selvaraj G, Kaliamurthi S, Cakmak ZE, Cakmak T (2017) RuBisCO of microalgae as potential targets for nutraceutical peptides: a computational study. Biotechnology 16(4–6):130–144
Semary NAE, Fouda M (2015) Anticancer activity of Cyanothece sp. strain extracts from Egypt: first record. Asian Pac J Trop Biomed 5:992–995
Severes A, Nivas S, D’Souza L, Hegde S (2018) Diversity study of freshwater microalgae of some unexplored water bodies of a rapidly developing industrial region in India. J Algal Biomass Util 9(2):31–40
Shaieb FA, Issa AA, Meragaa A (2014) Antimicrobial activity of crude extracts of cyanobacteria Nostoc commune and Spirulina platensis. Arch Biomed Sci 2:34–41
Sheih IC, Fang TJ, Wu TK (2009a) Isolation and characterisation of a novel angiotensin I-converting enzyme (ACE) inhibitory peptide from the algae protein waste. Food Chem 115(1):279–284
Sheih IC, Fang TJ, Wu TK, Lin PH (2009b) Anticancer and antioxidant activities of the peptide fraction from algae protein waste. J Agric Food Chem 58(2):1202–1207
Shi XM, Chen F (2002) High-yield production of lutein by the green microalga Chlorella protothecoides in heterotrophic fed-batch culture. Biotechnol Prog 18:723–727
Shi X, Wu Z, Chen F (2006) Kinetic modeling of lutein production by heterotrophic Chlorella at various pH and temperatures. Mol Nutr Food Res 50:763–768
Shimoda H, Tanaka J, Shan S, Maoka T (2010) Antipigmentary activity of fucoxanthin and its influence on skin mRNA expression of melanogenic molecules. J Pharm Pharmacol 62:1137–1145
Silvestro D, Andersen TG, Schaller H, Jensen PE (2013) Plant sterol metabolism. Δ7-sterol-C5-desaturase (STE1/DWARF7), Δ5,7-sterol-Δ7-reductase (DWARF5) and Δ24-sterol-Δ24-reductase (DIMINUTO/DWARF1) show multiple subcellular localizations in Arabidopsis thaliana (Heynh) L. PLoS One 8:e564529. https://doi.org/10.1371/journal.pone.0056429
Singh UB, Sharma C (2014) Microalgal diversity of Sheer Khad (stream): a tributary of Sutlej River, Himachal Pradesh, India. J Res Plant Sci 3(1):235–241
Singh P, Singh R, Jha A, Rasane P, Gautam AK (2015) Optimization of a process for high fibre and high protein biscuit. J Food Sci Technol 52:1394–1403
Sinha RP, Klisch M, Gröniger A, Häder DP (1998) Ultraviolet-absorbing/screening substances in cyanobacteria, phytoplankton and macroalgae. J Photochem Photobiol B Biol 47:83–94
Smetana S, Sandmann M, Rohn S, Pleissner D, Heinz V (2017) Autotrophic and heterotrophic microalgae and cyanobacteria cultivation for food and feed: life cycle assessment. Bioresour Technol 245(Pt A):162–170. https://doi.org/10.1016/j.biortech.2017.08.113
Sonani RR, Rastogi RP, Patel R, Madamwar D (2016) Recent advances in production, purification and applications of phycobiliproteins. World J Biol Chem 7:100–109
Spolaore P, Joannis-Cassan C, Duran E, Isambert A (2006) Commercial applications of microalgae. J Biosci Bioeng 101(2):87–96
Stengel DB, Connan S, Popper ZA (2011) Algal chemodiversity and bioactivity: sources of natural variability and implications for commercial application. Biotechnol Adv 29:483–501
Subudhi S (2017) Bioprospecting for algal based nutraceuticals and high value added compounds. J Pharm Pharm Sci 4(2):145–150. https://doi.org/10.15436/2377-1313.17.16511313.17.1651
Sugawara T, Matsubara K, Akagi R, Mori M, Hirata T (2006) Antiangiogenic activity of brown algae fucoxanthin and its deacetylated product, fucoxanthinol. Agric Food Chem 54:9805–9810
Suh SS, Kim SM, Kim JE, Hong JM, Lee SG, Youn UJ, Han SJ, Kim IC, Kim S (2017) Anticancer activities of ethanol extract from the Antarctic freshwater microalga, Botryidiopsidaceae sp. BMC Complement Altern Med 17:509. https://doi.org/10.1186/s12906-017-1991-x
Suresh A, Kumar RP, Dhanasekaran D, Thajuddin N (2012) Biodiversity of microalgae in Western a and Eastern Ghats, India. Pak J Biol Sci 15(19):919–928
Taira H, Aoki S, Yamanoha B, Taguchi S (2004) Daily variation in cellular content of UV-absorbing compounds mycosporine-like amino acids in the marine dinoflagellate Scrippsiella sweeneyae. J Photochem Photobiol B Biol 75:145–155
Takeuchi A, Okano T, Tanda M, Kobayashi T (1991) Possible origin of extremely high contents of vitamin D3 in some kinds of fish liver. Comp Biochem Physiol A Physiol 100:483–487
Talero E, García-Mauriño S, Ávila-Román J, Rodríguez-Luna A, Alcaide A, Motilva V (2015) Bioactive compounds isolated from microalgae in chronic inflammation and cancer. Mar Drugs 13(10):6152–6209
Taylor HR, Tikellis G, Robman LD, Mc Carty CA, Mc Neil JJ (2002) Vitamin E supplementation and macular degeneration: randomised controlled trial. BMJ 325(7354):11. https://doi.org/10.1136/bmj.325.7354.11
Thomson PG, Wright SW, Bolch CJS, Nichols PD, Skerratt JH, McMinn A (2004) Antarctic distribution, pigment and lipid composition, and molecular identification of the brine dinoflagellate Polarella glacialis (Dinophyceae). J Phycol 40:867–873
Tokuşoglu Ö, Üunal MK (2003) Biomass nutrient profiles of three microalgae: Spirulina platensis, Chlorella vulgaris, and Isochrisis galbana. J Food Sci 68:1144–1148
Tomaselli L (2003) Biodeterioration processes on inorganic substrata. Coalition 6:5–9
Tramroy P (2011) Microalgae market outlook report website. CBDM.T-Market and Business Intelligence. http://www.microalgae-market.com/
Tsukui T, Baba N, Hosokawa M, Sashima T, Miyashit K (2009) Enhancement of hepatic docosahexaenoic acid and arachidonic acid contents in C57BL/6J mice by dietary fucoxanthin. Fish Sci 75:261–263
Tukaj Z, Matusiak-Mikulin K, Lewandowska J, Szurkowski J (2003) Changes in the pigment patterns and the photosynthetic activity during a light-induced cell cycle of the green alga Scenedesmus armatus. Plant Physiol Biochem 41:337–344
Udayan A, Arumugam M, Pandey A (2017) Nutraceuticals from algae and cyanobacteria. In: Algal green chem. Elsevier, Amsterdam, pp 65–89
Uhlik DJ, Gowans CS (1974) Synthesis of nicotinic acid in Chlamydomonas eugametos. Int J Biochem 5:79–84
Urzi C, Realini M (1998) Colour changes of Noto’s calcareous sandstone as related to its colonisation by microorganisms. Int Biodeterior Biodegrad 42(1):45–54
US Nutra (2019) Corporate website. http://www.usnutra.com/. Accessed on 21 July 2019
USDA (2002) USA: dietary reference intakes and recommended dietary allowances. Nat Acad of Sciences. Available at: http://arborcom.com/frame/nutrc.htm. Accessed on 21 July 2019
Uttara B, Singh AV, Zamboni P, Mahajan R (2009) Oxidative stress and neurodegenerative diseases: a review of upstream and downstream antioxidant therapeutic options. Curr Neuropharmacol 7(1):65–74. https://doi.org/10.2174/157015909787602823
Valenzuela-Espinoza E, Millan-Nuñez R, Núñez-Cebrero F (2002) Protein, carbohydrate, lipid and chlorophyll a content of Isochrysis aff. galbana (clone T-Iso) cultured with a low cost alternative to the f/2 medium. Aquac Eng 25(4):207–216
Van Krimpen M, Bikker P, Van der Meer I, Van der Peet-Schwering C, Vereijken J (2013) Cultivation, processing and nutritional aspects for pigs and poultry of European protein sources as alternatives for imported soybean products. Wageningen UR Livestock Research, Lelystad
Vilchez C, Garbayo I, Lobato MV, Vega JM (1997) Microalgae-mediated chemicals production and wastes removal. Enzyme Microb Technol 20:562–572
Volkman JK (2003) Sterols in microorganisms. Appl Microbiol Biotechnol 60(5):495–506
Wang L, Pan B, Sheng J, Xu J, Hu Q (2007) Antioxidant activity of Spirulina platensis extracts by supercritical carbon dioxide extraction. Food Chem 105:36–41
Wang H, Peiris TH, Mowery A, Le Lay J, Gao Y, Greenbaum LE (2008) CCAAT/enhancer binding protein-beta is a transcriptional regulator of peroxisome-proliferator-activated receptor-gamma coactivator-1alpha in the regenerating liver. Mol Endocrinol 22:1596–1605
Wang G, Goyal N, Hopkinson B (2009) Preparation of L-proline based aeruginosin 298-A analogs: optimisation of the P1-moiety. Bioorg Med Chem Lett 19:3798–3803
Washida K, Koyama T, Yamada K, Kitab M, Urmura D et al (2006) Karatungiols A and B two novel antimicrobial polyol compounds, from the symbiotic marine dinoflagellate Amphidinium sp. Tetrahedron Lett 47(15):2521–2525
Watanabe M, Ikeuchi M (2013) Phycobilisome: architecture of a light-harvesting supercomplex. Phytother Res 116:265–276
Waterbury JB (2006) The cyanobacteria—isolation, purification and identification. In: Dworkin M (ed) The prokaryotes—a handbook on the biology of bacteria, vol 4. Springer, New York, pp 1053–1073
Welker M, von Dohren H (2006) Cyanobacterial peptides—nature’s own combinatorial biosynthesis. FEMS Microbiol Rev 30:530–563
Wijffels RH (2019) Algae for energy, http://www.knaw.nl/agenda/pdf/wijffels.pdf. Accessed on 02 July 2019
Wu Z, Dejtisakdi W, Kermanee P, Ma C, Arirob W, Sathasivam R, Juntawong N (2016) Outdoor cultivation of Dunaliella salina KU 11 using brine and saline lake water with raceway ponds in northeastern Thailand. Biotechnol Appl Biochem 64(6):938–943
Xiao R, Zheng Y (2016) Overview of microalgal extracellular polymeric substances (EPS) and their applications. Biotechnol Adv 34:1225–1244
Xiong F, Komenda J, Kopecky J, Nedbal L (1997) Strategies of ultraviolet-B protection in microscopic algae. Physiol Plant 100:378–388
Xiong FS, Kopecky J, Nedbal L (1999) The occurrence of UV-B absorbing mycosporine-like amino acids in freshwater and terrestrial microalgae (Chlorophyta). Aquat Bot 63:37–49
Yaakob Z, Ali E, Zainal A, Mohamad M, Takriff MS (2014) An overview: biomolecules from microalgae for animal feed and aquaculture. J Biol Res (Thessaloniki) 21(1):6. http://www.jbiolres.com/content/21/1/6/6
Yabuta Y, Fujimura H, Kwak CS, Enomoto T, Wata-nabe F (2010) Antioxidant activity of the phycoeryth-robilin compound formed from a dried Korean purple laver (Porphyra sp.) during in vitro digestion. Food Sci Technol Res 16:347–351
Yook JS, Kim KA, Park JE, Lee SH, Cha YS (2015) Microalgal oil supplementation has an anti-obesity effect in c57bl/6j mice fed a high fat diet. Prev Nutr Food Sci 20(4):230–237
Yuan JP, Chen F, Liu X, Li XZ (2002) Carotenoid composition in the green microalga Chlorococcum. Food Chem 76:319–325
Yuan JP, Peng J, Yin K, Wang JH (2011) Potential health-promoting effects of astaxanthin: a high-value carotenoid mostly from microalgae. Mol Nutr Food Res 55:150–165
Zhang CW, Cohen Z, Khozin-Goldberg I, Richmond A (2002) Characterization of growth and arachidonic acid production of Parietochloris incisa comb. Nov (Trebouxiophyceae, Chlorophyta). J Appl Phycol 14:453–460
Zhang D, Wan M, del Rio-Chanona EA, Huang J, Wang W, Li Y, Vassiliadis VS (2016) Dynamic modelling of Haematococcus pluvialis photoinduction for astaxanthin production in both attached and suspended photobioreactors. Algal Res 13:69–78
Zhekisheva M, Zarka A, Khozin-Goldberg I, Cohen Z, Boussiba S (2005) Inhibition of astaxanthin synthesis under high irradiance does not abolish triacylglycerol accumulation in the green alga Haematococcus pluvialis (Chlorophyceae). J Phycol 41:819–826
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Kaur, P. (2020). Microalgae as Nutraceutical for Achieving Sustainable Food Solution in Future. In: Singh, J., Vyas, A., Wang, S., Prasad, R. (eds) Microbial Biotechnology: Basic Research and Applications. Environmental and Microbial Biotechnology. Springer, Singapore. https://doi.org/10.1007/978-981-15-2817-0_5
Download citation
DOI: https://doi.org/10.1007/978-981-15-2817-0_5
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-15-2816-3
Online ISBN: 978-981-15-2817-0
eBook Packages: Earth and Environmental ScienceEarth and Environmental Science (R0)