Abstract
The use of macular laser treatment to attempt to prevent CNV is reviewed in relation to age-related macular degeneration (AMD), the principal cause of visual loss due to CNV. The causes of age-related CNV and ways in which macular laser may interact and influence with those processes for good or ill are reviewed and discussed. In particular, progressive dysfunction of the retinal pigment epithelium (RPE) and the development of self-perpetuating chronic inflammatory neurodegeneration are noted as central to the development of all chronic progressive retinopathies, including AMD. By activating restorative repair, retinal laser treatment can reverse these processes, reducing the risks of visual loss including CNV. Laser-induced retinal damage (LIRD) is wholly adverse, negating many treatment benefits and worsening outcomes. By maximizing therapeutic effects and precluding the adverse effects from LIRD, modern retinal laser therapy, epitomized by low-intensity/high-density subthreshold diode micropulse laser (SDM), can slow AMD progression and reduce the risk of visual loss due to the development of new CNV. The benefits of laser sublethal to the retinal pigment epithelium such as SDM are greatest in eyes than stand to benefit most from treatment—those with high-risk AMD. These are the same eyes worsened by the LIRD associated with short-pulse continuous wave and nanosecond lasers inherently damaging to the RPE. The underlying principles of disease progression and laser prevention in AMD may also apply to other chronic progressive retinopathies and other causes of macular CNV.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
For most of the modern history of ophthalmology, laser treatment has been the primary treatment for macular disease [1]. Until the relatively recent advent of vascular endothelial growth factor (VEGF) inhibitors, ablative photocoagulation was the only treatment option for most eyes with choroidal neovascularization (CNV) [1,2,3,4]. Due to the risks of treatment-associated visual loss, location, and often poor definition of macular neovascular lesions, few patients with macular CNV were candidates for treatment. Of those, fewer still enjoyed any long-term treatment benefits [5].
While anti-VEGF medications have revolutionized the treatment of CNV, both due to their effectiveness and minimal risks of treatment-associated visual loss, other than Age-Related Eye Disease Study (AREDS) vitamin supplements, there remains no effective treatment, pharmacologic or otherwise, to prevent macular CNV [6, 7]. The history of macular laser treatment to prevent CNV is as long and as disappointing as the treatment of CNV itself. Recent studies indicate a change in this regard [8,9,10,11]. Spurred by a new understanding of retinal laser treatment, new information from both the laboratory and clinic suggests that laser for prevention of CNV should be effective, and is [11]. To understand the past failure, and future promise, of laser treatment to prevent CNV it is essential to understand exactly what is meant by “laser” treatment, and all that follows from it [8]. Our discussion of laser CNV prevention will focus on CNV complicating age-related macular degeneration (AMD). This is for two reasons: first, because AMD is the most important cause of visual loss due to CNV; and second, because only in AMD have notable efforts been made to prevent CNV by any means, and in particular, with laser [12, 13].
For over 50 years, photocoagulation was universally presumed to be the necessary and sufficient cause of all therapeutic benefits of retina laser treatment [14,15,16]. Low-intensity/high-density subthreshold diode micropulse laser (SDM) proved this presumption to be false by demonstrating therapeutically effective treatment in the total absence of laser-induced retinal damage (LIRD) [17,18,19,20,21]. As a watershed development in the history of retinal laser treatment, this discovery led to two key advances in our understanding of retinal laser treatment. These bear heavily on the following examination of the potential for the laser to prevent CNV. First, as both unnecessary and the sole source of all risks, adverse treatment effects, and treatment limitations, we now understand photocoagulation (for other than cautery) to be a complication of retinal laser treatment. Second, abandonment of photocoagulation as the necessary precondition for therapy has led to, for the first time, a satisfying and useful understanding of the mechanism of action of retinal laser treatment as a physiologic “reset” phenomenon [22, 23]. Unlike prior unsuccessful attempts to explain the effects of retinal laser arising from photocoagulation, reset theory accounts for all clinically observed retinal laser effects. Further, in a fundamental test of any theory, reset theory has accurately predicted a new retinal laser application never conceived of in the photocoagulation era. These include reversal of anti-VEGF drug tolerance in neovascular AMD (NAMD); improved retinal and visual function following panmacular laser treatment in dry AMD, inherited retinopathies, and open-angle glaucoma; and neuroprotective effects in glaucomatous optic neuropathy [22,23,24,25].
Critical to a clear understanding of the retinal laser literature is a clear understanding of various retinal laser modes and their effects [8]. Complicating this task is historical imprecision and frequent misuse, unintentional and otherwise, of key clinical terminology that can obfuscate and mislead readers not keenly aware of these issues. The history of laser prevention of NAMD is particularly illustrative in this regard. Essential differences in the conception, design, expectations, claimed and actual effects of various laser modes account for both the past failures and future promise of retinal laser for the prevention of CNV. To draw out the key distinctions, the following discussion will divide the topic into two main headings: Laser for drusen; and laser not for drusen.
2 Laser for Drusen
As the hallmark of dry AMD, drusen are an important risk factor for progression and visual loss, particularly due to the development of CNV and NAMD [25,26,27,28,29,30]. As such, drusen are a clinically useful indicator of the degree of age-related macular dysfunction. The risk of age-related visual loss generally parallels the number, size, and proximity of drusen to the fovea [25,26,27,28,29,30]. Thus, drusen reduction has been a natural target for therapeutic intervention in dry AMD [31, 32].
Inflammatory disturbances of the macula in eyes with drusen, such as the development of CNV, other exudations, and focal chorioretinitis, including the iatrogenic chorioretinitis of macular photocoagulation, have long been noted to result in local disappearance of drusen in and around the inflammatory lesion [27, 30, 33, 34]. This suggested macular photocoagulation might be used therapeutically to reduce drusen and the risks of visual loss due to AMD [33, 35].
At the time of the first attempts to use photocoagulation to reduce drusen, the standard for photocoagulation intensity was established by the Early Treatment of Diabetic Retinopathy Study (ETDRS) and Macular Photocoagulation Study Group (MPSG) reports, which employed intense suprathreshold treatment resulting in clinically obvious white, full-thickness retinal burns [36, 37]. Recognition that drusen occurred at the level of the RPE suggested that less intense and thus less obviously clinically visible photocoagulation lesions (limited to the RPE and outer retina, or “threshold” intensity), barely visible, or even initially non-ophthalmoscopically “subthreshold” photocoagulation lesions, might be effective and reduce adverse treatment effects by reducing neurosensory retinal damage [32]. These less-severe lower intensity retinal burns were described, in comparison to the starkly white ETDRS and MPSG lesions, as “invisible” [38]. However, photocoagulation lesions acutely invisible or difficult to see ophthalmoscopically are virtually always immediately visible by fundus fluorescein angiography (FFA) and become clinically visible minutes to weeks after treatment. The contrast in lesion severity with conventional ETDRS and MPSG suprathreshold photocoagulation, however, led investigators to incorrectly assume a difference in substance, rather than style. This was (1) because LIRD was assumed to be essential to the therapeutic benefits of retinal laser treatment in general, and drusen reduction in particular could not be done without; and (2) over-estimation of the risk and adverse effect reduction associated with lower intensity retinal photocoagulation [31, 32, 35, 38].
The initial reports of macular laser treatment to reduce drusen were positive and encouraging [31, 32]. Treatment, directed at the drusen themselves, was effective in reducing drusen numbers and density, and improving visual acuity [31, 32, 35, 38]. With time, however, treated eyes began to demonstrate an increased propensity to develop new CNV thus converting to NAMD; worsening, rather than improving, the long-term visual prognosis [39,40,41]. Subsequent studies found that eyes demonstrating both the greatest drusen reduction and the greatest likelihood of CNV were those receiving the most intense macular photocoagulation [42, 43]. This realization brought about the effective end of interest in conventional (millisecond) continuous wave (CW) laser treatment for drusen [44].
It was recognized that LIRD to retinal architecture, particularly the Bruch’s membrane/RPE complex, was a likely key predisposing factor to CNV, but possibly not necessary to achieve effective drusen reduction [9]. What to do? By shortening laser pulse duration the thermal effects of retinal laser treatment could be more selectively concentrated in the RPE [45, 46]. Microsecond CW exposures demonstrated the ability to selectively damage the outer retina and the homogeneously pigmented RPE of animals in laboratory studies [47,48,49,50]. Further shortening of the laser pulse to the nanosecond range can result in selective heating of the RPE melanosomes themselves [50]. However, shortening of pulse duration results in narrowing of the therapeutic range (TR), such that at nanosecond exposures, there is no TR. Instead, the threshold for RPE cell death is exceeded before the threshold for HSP activation (the cell dying before the HSPs can activate), the prime mediator of therapeutic retinal laser effects [8, 49]. In practice, this means that the effect of nanosecond laser (NSL) exposure is either no effect at all (below the TR threshold); or—at minimum—photodisruptive killing of the RPE by intracellular vaporization of RPE melanosomes. In this volatile, literally explosive setting, excess laser energy or RPE pigment density will necessarily result in collateral damage extending to the retina and Bruch’s membrane [8]. Again, next to the severity of the suprathreshold full-thickness retinal burns from conventional standard photocoagulation in the ETDRS and MPSG reports, however, the LIRD resulting from short-pulse CW lasers, generally confined to the RPE and outer retina, still considered essential, seemed also negligible in comparison. By confining LIRD to the RPE and outer retina, it was hoped that drusen could be reduced without incurring either neurosensory retinal damage or increasing the risk of CNV, led to studies of both microsecond and nanosecond lasers, more selective for the RPE, as preventative treatments for AMD, and thus age-related CNV.
In 1999, Roider and coworkers reported two patients with soft drusen treated with 1.7-microsecond exposures of 527 nm laser with identical treatment parameters (“selective retinal laser therapy”, or SRT, Lutronic, Billerica MA, USA), both clinically subthreshold, without visible laser spots at the time of treatment [51]. Both produced LIRD visible by FFA. However, in one eye the LIRD became visible clinically following treatment. In this eye, the drusen disappeared. In the other patient, no clinically visible LIRD developed later and there was no drusen reduction. The authors duly note that these eyes demonstrate the clinical variability of short pulse laser effects, a clinical manifestation of the narrow CW laser TR.
As noted above, NSL takes the quest for selective RPE destruction a step further. By shortening the pulse further, NSL maximizes the thermal energy uptake to the RPE melanosomes themselves, causing explosive vaporization of the melanosomes with internal cavitation of the RPE causing cell death (2RT®; Ellex Pty Ltd., Adelaide, Australia). In its idealized form, 2RT NSL seeks to limit LIRD to selective killing of the RPE, which they term “rejuvenation,” without causing damage to adjacent structures such as photoreceptors and Bruch’s membrane. Pilot studies of NSL demonstrated both drusen reduction and improvement in visual acuity without a notable increase in CNV [9, 10]. At 24 months follow-up in a randomized clinical trial of comparing NSL to sham for dry AMD, the LEAD study found no overall effect of treatment [52]. Subgroup analyses showed drusen reduction in eyes with early, low-risk AMD, without an increase in CNV incidence, suggesting the success of the trial hypothesis. However, the 2RT NSL treatment response in eyes with high-risk AMD, those with the greatest risk of visual loss and with thus the most to gain from effective preventive treatment, was negative [52]. In these eyes, often identified by the presence of reticular pseudodrusen (RPD), NSL caused rapid progression of AMD and visual loss, particularly due to the development and/or progression of geographic atrophy. Of note is that LIRD was not considered an adverse treatment effect in the LEAD study. This is despite the fact that LEAD trial was performed to assess the effect of “non-damaging” 2RT NSL in AMD, a stated differentiator of the LEAD study from prior studies of laser for drusen (https://www.ellex.com/us/products/2rt/). Unfortunately, no fundus images accompanied the published report [52]. However, in a meeting presentation of LEAD study results by the investigators, all posttreatment fundus photographs presented demonstrated laser damage due to the NSL applications, clinically indistinguishable from conventional CW photocoagulation lesions (Guymer R, Marshall J, et al. The LEAD Study, presentation to the European Society of Retina specialists, Sept. 22, 2018, Vienna, Austria). The LEAD study authors did not explicitly correlate worsening of high-risk AMD with LIRD, but concluded that 2RT NSL was contraindicated in eyes with high-risk AMD [52].
Despite disappointing results, the LEAD study findings offer important insights. While it supports the concept that LIRD is necessary for drusen reduction (without establishing a benefit) it also suggests that avoidance or minimization of damage to Bruch’s membrane may reduce the risk of CNV as an adverse treatment effect. However, it also demonstrates that that NSL is unreliable for this purpose, resulting in significant collateral damage in many, if not most, treated eyes leading to significant worsening of high-risk eyes. As with other studies of laser for drusen reduction, the LEAD study challenges the presumption that drusen reduction is a desirable end. Rather, the acceleration of visual loss due to age-related geographic atrophy (ARGA) in the LEAD study suggests 2RT appears to cause rapid decompensation of the most vulnerable eyes with high-risk AMD. This may be due to added physiologic stresses associated with the inflammation and healing response to the LIRD caused by NSL in these already tenuous eyes. Transmacular shock-waves produced by explosive photodisruption of the RPE, unique to NSL, may also contribute to RPE decompensation as far as 2 mm from an NLS application site (Chang DB, Luttrull JK; unpublished data, March 2019). The worsening of AMD, and particularly the progression of ARGA following laser for drusen in the LEAD study is not entirely unexpected. In the Cochrane meta-analysis of laser for drusen studies, Virgili and associates noted that, while little information could be gleaned from prior studies in this regard, available data suggested that ARGA worsened after laser for drusen [44]. In addition, a prospective clinical trial of microsecond SRT laser hoping to slow ARGA progression was abandoned early, due to the finding that treatment, designed to produce LIRD at the margins of the ARGA lesions, increased the rate of ARGA progression by 50% compared to untreated controls [53].
In sum, efforts to reduce the risks of visual loss in AMD with laser treatment have been driven by the recognized necessity of LIRD to achieve drusen reduction and the presumption of a resulting benefit. The failure of these efforts results from this very same LIRD, and casts doubt on the presumption of benefits from drusen reduction.
3 Laser Not for Drusen
The only treatment as yet proven to reduce the risk of developing age-related CNV in a multicenter prospective clinical trial is AREDS vitamin supplementation [6]. Interestingly, challenging the presumption of the need for drusen reduction, the success of AREDS treatment was not associated with any notable treatment effects on drusen or other morphologic features of AMD. The treatment benefits were consistent over time, reducing progression to advanced AMD by about 4% per year over the course of the study. Unlike short pulse lasers, the greatest benefit from AREDS supplements was seen in eyes at the greatest risk for visual loss [6]. As 90% of eyes with advanced AMD have CNV, it was a reduction in new CNV that accounted for most of the AREDS treatment benefits as the progression of ARGA was not affected. Thus, it was nutritionally derived improvement in retinal physiology alone that accounted for the benefits of AREDS study, and it did so, via CNV reduction, without reducing drusen or effecting any other alteration in macular anatomy [6].
All laser modes are capable of causing LIRD. Some, like NSL, cause it necessarily. For others, such as CW lasers, LIRD is prohibitively difficult to avoid and still accomplish effective treatment. Only a micropulsed laser (MPL) operated at a low duty-cycle (DC) can reliably and predictably preclude LIRD while maintaining therapeutic effectiveness [9]. The epitome of low-DC MPL reliably sublethal to the RPE is “low-intensity/high-density subthreshold diode micropulse laser” (SDM). SDM both established and defined the fundamental principles of modern retinal laser therapy. First; by employing a low-DC (5%) 810 nm near-infrared laser, SDM is reliably sublethal to the RPE (“low-intensity”), which it selectively targets. Despite this safety, it is an effective activator of RPE HSPs, the essential mediators of the therapeutic laser response. Second; the therapeutic response is amplified, maximized, and made clinically effective by confluent and complete treatment aimed at recruiting large areas of diseased retina (“high-density”) to the normalizing process [9, 45, 46, 49, 50, 54, 55]. This is in contrast to the traditional focal or local application strategy used in conventional photocoagulation, which are abandoned in modern retinal laser therapy [37, 38]. Over time, the SDM treatment paradigm has evolved to the employment of just two treatment fields: “panmacular” treatment, consisting of confluent treatment of the entire retina between the major vascular arcades including the fovea; and “panretinal” treatment, consisting of confluent and complete treatment of all the retina outside the major vascular arcades [11, 20, 22,23,24,25, 46, 56, 57]. Panmacular treatment is used for all treatment indications. Panretinal treatment is added for generalized retinopathies, such as diabetic retinopathy or early retinitis pigmentosa, resulting in treatment of the entire retina, and thus all of the retinopathy, just as would be treated by medication (Figs. 30.1 and 30.4).
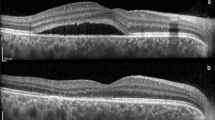
Intravenous fundus fluorescein angiograms (FFA) of diabetic retinopathy (DR) following total retinal SDM laser (panmacular and panretinal) in three patients (a), (b) and (c). No VEGF inhibitors, steroids, or other medical treatment for DR was used. Left photo, before treatment; right photo after treatment. Note reduction in macro and microvascular leakage along with reversal/diminution of retinopathy severity in each case
Initial studies of SDM demonstrated effective treatment for complications of diabetic retinopathy in the complete absence of LIRD [18,19,20, 46]. Like AREDS supplements, the absence of LIRD with SDM indicates, by exclusion, that SDM treatment benefits arise entirely from laser-induced improvements in retinal physiology elicited by sublethal thermal laser stimulation of the RPE [58]. Whereas the TR of CW laser is only 10× ANSI MPE, the same ANSI data show the TR of micropulse lasers, like SDM, to be much wider, 100×MPE or more [47]. SDM is thus safe and without any known adverse treatment effects [45, 47, 58]. Because of the broad SDM TR, treatment intensity need not be subjectively titrated on a per eye basis (effectively turning each treated eye into a dosimetry experiment), as decades of clinical experience have identified safe and effective SDM parameters that can be used effectively in all eyes of all patients without regard for pathology or pigment variation [8, 18,19,20, 45, 47, 57, 58]. Employment of “fixed” SDM laser parameters enhances treatment safety by eliminating the possibility for surgeon misjudgment, the most common cause of visual loss from modern retinal laser treatment [8].
As noted, SDM led to a new understanding of the therapeutic benefits of retinal laser treatment stemming from a physiologic “reset” effect on the RPE, normalizing retinal function largely independent of the underlying cause of dysfunction [23,24,25]. Making no unique contribution to treatment efficacy and solely responsible for all risks, limitations, and adverse effects of retinal laser treatment, photocoagulation, and all other forms of LIRD are now considered necessary complications of treatment [8, 21,22,23,24, 59].
Reset theory indicated that SDM should improve retinal function not just in the conventional retinal laser indications such as diabetic macular edema, proliferative diabetic retinopathy, central serous chorioretinopathy, and retina vein occlusions; but in any chronic progressive retinopathy (CPR) of any cause [23,24,25]. In short, this is because all CRPs are neurodegenerations, and thus share many key features that are addressed by the reset effects of laser treatment [23, 24, 54, 55]. By addressing those fundamental commonalities, the reset phenomenon acts as a “non-specific trigger of disease-specific repair,” improving both retinal and visual function in all CPRs, including AMD [23, 24].
In a pilot study of a large cohort of 547 eyes of 363 patients with dry AMD, panmacular SDM treatment was followed by a much lower than expected rate of new CNV [11]. In the absence of LIRD or any other adverse treatment effects, the annual incidence of new CNV following panmacular SDM in dry AMD was 0.87%, compared to 4% in the AREDS [6]. This 80% reduction in expected CNV was noted despite significantly higher risk factors for CNV in the panmacular SDM-treated group compared to the AREDS, particularly age (median 84 vs. 69 years) [6]. Adjusting only for the risk factor age, SDM treated eyes had an incidence of new CNV 93–98% lower than expected compared to the AREDS and other natural history studies of AMD [6, 11]. In a subsequent study of 111 eyes of 70 patients with ARGA from the same dry AMD cohort, SDM reduced the radial velocity of ARGA progression 55% per year compared to untreated controls (p = 0.0002) (pretreatment observation period avg. 2.7 years, posttreatment avg. 1.9 years) [56]. Echoing the AREDS, other than reducing the incidence of new CNV and slowing ARGA progression, panmacular SDM prophylaxis of dry AMD was not associated with any notable short-term effects on drusen or other macular morphology [11, 56]. The findings of these studies will inform the design of future confirmatory studies. However, the robust results of these pilot studies of panmacular SDM for dry AMD indicate that retinal laser treatment designed to preclude, rather than cause, LIRD may become an important tool in the prevention of visual loss due to AMD and age-related CNV in particular [11].
In sum, while retina-damaging laser increases the risk of CNV, non-damaging laser sublethal to RPE reduces that risk. While damaging laser appears to cause rapid progression of high-risk AMD; non-damaging laser, sublethal to the RPE, appears to slow progression significantly. Thus, contrary to traditional thinking (but as predicted by reset theory) it is successful avoidance of LIRD, and thus avoidance of drusen reduction, that appears key to the success or failure of laser for prevention of CNV in AMD. How do we account for this? First, by understanding AMD as a chronic progressive neurodegenerative disease; second, by taking into account the effects of retinal laser on retinal integrity and RPE function; third, by recognizing how the classical dynamics of wound repair are engaged by treatment; and fourth, by understanding the ability of laser to modulate both acute and chronic inflammation—one the healer, the other the driver, of the chronic disease process (Figs. 30.1, 30.2, 30.3, 30.4, 30.5).
Pseudophakic cystoid macular edema (CME). Unresponsive to combination of topical steroid and nonsteroidal anti-inflammatory drops for 1 year. Drops discontinued and panmacular SDM performed. (a) Infrared (IR), (b) late phase FFA and (c) Spectral-domain optical coherence tomography (OCT) prior to treatment with severe CME. Visual acuity (VA) 20/200. (d) IR and (e) OCT 1 month following panmacular SDM. CME resolved. VA 20/30
Idiopathic retinal vasculitis, optic neuritis, and vitriitis. (a) FFA before treatment. (b) FFA 1 month following total retinal SDM laser. No other local or systemic medical treatment given. Note the decrease in inflammatory dye leakage from retinal vessels and optic nerve. VA prior to treatment 20/70; after, 20/50
Bevacizumab- and ranibizumab-resistant serous macular detachment associated with diabetic macular edema. No steroid therapy was given. Top: Before panmacular SDM. VA 20/200. Bottom: Three months after panmacular SDM. Note the decrease in macular edema and resolution of inflammatory serous macular detachment. VA 20/70
4 Dry AMD as a Model of Chronic Disease and Inflammaging
The association of aging with chronic inflammation, often referred to as “inflammaging,” is now recognized as a significant component of virtually all chronic, age-related diseases, including AMD [60]. Unlike acute inflammation, inflammaging is low grade, chronic, persistent, and self-perpetuating, and leads to tissue degeneration [61]. To understand the mechanisms by which inflammaging is generated, we must first understand the essential role of the immune system in the maintenance of normal tissue function and homeostasis.
In healthy cells, there is constant surveillance and repair to maintain normal cell function and homeostasis [49, 62, 63]. However, in disease intracellular abnormalities often either escape repair and/or exceed the cell’s ability to manage them successfully leading to a loss of normal function that is generally characteristic of the primary underlying disease process. Further, both normal and diseased tissues produce waste, or “self-debris,” that includes damaged cells and macromolecules. In disease, the accumulation of this waste is excessive and thus progressive, ultimately compromising tissue structure and function [60]. At the tissue level, the mechanism employed to repair this damage and remove this waste is inflammatory-mediated (inflammation being a prerequisite of repair) and is dependent on resident macrophages and mast cells [64]. With aging this “housekeeping” function becomes less efficient, due mainly to the combination of increased generation of self-debris and inefficient removal, requiring additional inflammatory input to maintain the tissue in a physiologic (normal) or near physiologic working state, a process that is mediated but ultimately compromised by assembly of the “inflammasome.” The inflammasome is a multiprotein stimulus-dependent oligomer that activates the inflammatory process by promoting secretion of pro-inflammatory cytokines and interleukins with dysregulation of inflammasomes being a feature of all chronic diseases [65]. This heightened inflammatory state, between basal physiologic inflammation and pathologic inflammation, is referred to as “para-inflammation” [64]. With further aging the inflammatory stakes continue to rise, eventually escalating to require mobilization of a systemic immune response that includes the recruitment of additional leukocytes and expression of systemic pro-inflammatory cytokines [66]. Thus, maintenance of tissue homeostasis in the aging human requires an increasing inflammatory response to address increased reparative demands that eventually moves beyond para-inflammation to a self-perpetuating and degenerative chronic inflammatory state referred to as “inflammaging” [60, 66].
A classic example of a disease with self-debris is AMD. In dry AMD, this debris is the phenotypic marker of disease and a clinically useful biomarker of the heightened inflammatory state within the retina, classically represented by drusen. Ultrastructurally, this is reflected by priming or activation of the NLRP3 inflammasome and the specific proteins known to be associated with inflammasome assembly [65]. Experimentally, identified proteins include C1q complement component in drusen extracts from AMD donor tissue, carboxyethylpyrrole-adducted proteins from the aging retina, and Alu RNA transcripts from the RPE of patients with ARGA [67,68,69,70,71]. This inflammasome-mediated damage to the retina in AMD may be further aggravated by inflammatory pyroptotic and apoptotic effects resulting from IL-1β and IL-18 expression [70]. In those individuals who have the genetic risk variant for complement factor H (CFH) or other complement risk variants, regulation of these factors is abnormal and excessive quantities of inflammatory complement components are generated and deposited in the retina. Thus, the chronic inflammatory state we recognize as AMD is driven by a number of factors, including complement dysfunction and inflammasome-mediated inflammation, with genetic and environmental (such as smoking) modifiers [72,73,74,75,76,77,78,79,80,81,82,83,84,85].
5 Acute Inflammation—Prelude to Repair
It is a basic tenet of biologic repair that an acute inflammatory response is necessary to activate the correct cascade of molecular events to generate successful and complete repair [81, 82]. In contrast, the molecular signature of all acquired age-related diseases, such as AMD, is the presence of chronic inflammation. Chronic inflammation, by definition, indicates that normal repair is not proceeding [61, 65, 81, 82, 86, 87]. Unimpeded, this can lead to tissue degeneration and vision loss via the end-stage AMD phenotypes we recognize clinically as ARGA and disciform scarring due to CNV. Thus, a desired goal of any therapy would be to intervene preventatively, prior to anatomic derangement and visual loss (end-stage disease markers), when the disease process is manifest only by the earliest detectable physiologic dysfunction and most amenable to repair [23]. If the chronic inflammatory disease bed that is AMD could be converted to an acute inflammatory lesion, would the resulting corrective molecular cascades allow for healing and repair sufficient to permit restoration and normalization of retinal function?
6 Laser—Prelude to Acute Inflammation
As discussed previously, clinical retinal laser effects can be generally divided into two types: damaging (lethal to at least the RPE) and non-damaging (sublethal to the RPE). These divisions reflect progression in our understanding of the mechanism of retinal laser treatment. As discussed, while photocoagulation was once the goal of retinal laser treatment, it is now clear that photocoagulation and indeed all forms and degrees of LIRD are unnecessary and detrimental; complications of treatment rather than the necessary and sufficient cause of therapeutic retinal laser effects. Within the context of LIRD there is a spectrum of damage that is also instructive. An extreme example of LIRD is the inadvertent rupture of Bruch’s membrane. This can be seen with excessively intense photocoagulation, or even as the result of micro- or nanosecond laser damage limited to the outer retina, such as that reported in the LEAD study [52]. Clinically acute rupture of Bruch’s membrane is heralded by the instantly recognizable audible “pop” and subretinal gas-bubble formation, with the simultaneous localized appearance of subretinal hemorrhage. Clinically undesirable as promoting the development of CNV, this observation has led to the use of laser rupture of Bruch’s membrane to experimentally promote CNV animal models [88].
By any definition, whether clinically inadvertent or experimentally deliberate, laser-induced rupture of Bruch’s membrane is a good example of an acute, brief, and once-off traumatic event that will result in activation of a classical tissue repair response [89]. As an extreme example of sorts, it also offers an important insight into the prior failures of laser treatment associated with LIRD to prevent CNV.
Classical tissue repair involves a sequence of key interactions that can be divided into the three overlapping phases of (1) inflammation, (2) tissue formation, and (3) tissue remodeling; involving cells, cytokines, and the extracellular matrix (ECM) [82, 90,91,92].
Ultimately, the degree of tissue remodeling and resultant scar tissue deposition is directly proportional to the severity of the initial injury: the greater the injury, the greater the degree of acute inflammation induced and the greater the degree of irreversible tissue damage and scar tissue formation. Scar tissue formation represents the restoration of tissue integrity, but without restoration of normal tissue function. Thus, we can ultimately say that, based on the canons of wound healing, a therapeutically ideal laser treatment causes acute injury without structural damage. This in turn generates the desirable endpoint of complete repair without scar tissue formation, thus restoration and normalization of tissue function. LIRD violates this maxim.
This new understanding holds that any degree of LIRD lethal to the target RPE is not only undesirable but a complication counterproductive and detrimental to the goal of therapeutically effective retinal laser treatment. “Non-damaging” laser can thus only properly refer to the complete absence of LIRD at the histopathologic level at any point postoperatively, as the RPE is affected but not killed by laser exposure. Functional retina is lost, RPE cells, the mediators of laser response, are destroyed rather than revitalized and the therapeutic response thus diminished. Finally, as the previously discussed clinical studies illustrate, violation of tissue integrity, no matter how small, can “light the fuse” for the eventual development of CNV by promoting angiogenesis and/or compromising the natural barrier to vascularization presented by the healthy and intact RPE/Bruch’s membrane complex [39,40,41,42, 44].
7 Laser: Mechanism of Action
As stated, laser efficacy derives from its ability to generate a sublethal injury to the RPE leading to repair [8]. At the cellular level, this results in normalization of RPE function, or “homeotrophy.” By improving RPE function, retinal function and autoregulation are improved via normalized expression of, and response to, RPE-derived factors such as cytokines and interleukins. Photoreceptor toiletry and waste processing are improved and thus debris accumulation diminished [26, 29, 30, 33,34,35, 93,94,95,96]. At the tissue level, this leads to improved retinal and visual function via activation of reparative laser-induced acute inflammation which is inherently antagonistic to disease-driving chronic inflammation. Therefore, for the laser to be therapeutically effective, it must cause an acute, brief, and once-off “injury” to the RPE to activate tissue repair and to be maximally beneficial, the laser must be “tissue-sparing” and thus sublethal to the RPE. MPL such as panmacular SDM stands alone in its ability to predictably and reliably fulfil both criteria [8]. So, in the absence of LIRD, how does laser work to elicit retinal repair and functional restoration?
Heat shock proteins (HSPs) are a family of proteins that are constitutively expressed in all cells, having critical roles in maintaining homeostasis and normal cell function, but, as noted above, are also significantly upregulated in response to acute insults, such as heat and oxidative stress, perceived by the cell to be existential threats [97]. HSPs are typically grouped into different subfamilies according to their molecular weights in kilo Daltons (hsp100, hsp90, hsp70, hsp60, hsp40, and small HSPs (sHSP), which includes α-crystallins). Each has a particular role. The hsp 70 family is of particular relevance to the current discussion [98]. HSP activation is especially sensitive to the acuity and severity of a cellular threat. Thus, the insidiously progressive dysfunctions of chronic disease are poor triggers of HSP-mediated repair [18, 22, 23, 49, 57, 58, 62, 63]. In chronic disease, the homeostatic functions of HSPs may become taxed to the point of failure, leading to failure of the HSP system itself and further accumulation of damage, debris, chronic inflammation, cell death, and tissue failure [49, 99,100,101,102,103].
As noted above, thermal laser exposure is an effective trigger of HSP activation [49, 97, 104]. The attributes of SDM which are ideal in this regard arise from the facility of SDM to controllably photocoagulate only a small fraction of intracellular proteins sufficient to activate HSP-mediated cellular repair, but insufficient to cause cell death [8, 105]. Most importantly, this laser-induced HSP activation response (salvific and homeotrophic, rather than baseline and homeostatic) results in the repair of not only the acute laser-induced damage but also proceeds to indiscriminate repair of the accumulated damage from the underlying chronic disease process that has escaped HSP surveillance and would otherwise lead to progressive cellular dysfunction and ultimate death. Independence of the salvific HSP repair process from the cause(s) of the accumulated cell damage and resultant dysfunction particular to the underlying disease process makes the reset phenomenon both powerful and elegant, and the basis for the description of retinal laser in chronic retinopathies as a “non-specific trigger of disease-specific repair” [49, 100,101,102,103].
8 Laser and AMD-Biologic Effects
Laser activation of RPE HSPs triggers a cascade of reparative and modulated inflammatory effects, factors, and processes that improve cell and retinal function. These include decreased expression of angiogenesis promoters VEGF, TGF-β, and bFGF; increased angiogenesis inhibitor pigment epithelial-derived factor expression; improved mitochondrial function; increased retinal nitrous oxide levels; inhibition of apoptosis; modulation of tissue matrix metalloproteinases; reduced free radical species and increased superoxide dismutase activity; increased mRNA expression of cytokine markers of reparative acute inflammation and decreased markers of chronic inflammation; local and systemic immunomodulation including local stem cell activation and monocyte and hematopoietic progenitor cell recruitment to the retina; and improved retinal macro- and microglial function [8, 22, 23, 47, 58, 63, 106,107,108,109,110,111,112,113,114,115,116]. Absent compromise of the RPE/Bruch’s membrane complex integrity and tissue scarring from LIRD that increase the risk of CNV in AMD, any or all of the above responses to laser treatment sublethal to the RPE such as SDM may contribute to a reduced risk of age-related CNV.
As noted, a characteristic abnormality in AMD is the accumulation of metabolic by-products such as lipofuscin, damaged organelles, and nonfunctioning or toxic proteins [117]. This informs us that the RPE in AMD is functioning in an environment of chronic oxidative stress, while still managing to maintain relatively normal visual cycle metabolism and upkeep of Bruch’s membrane. With aging the normal level of homeostatic HSP function is insufficient to maintain repair. This oxidative stress-induced cellular damage also contributes to increased protein misfolding and formation of detrimental protein aggregates within the cytosol, further compromising cell function [118, 119]. Here, activation of additional compensatory processes may mitigate HSPs failure. Two that are particularly important are the proteasome and autophagy pathways. The first involves tagging misfolded proteins with ubiquitin and transferring them to the proteasome degradation pathway, which is a multicatalytic proteolytic complex that recognizes and selectively degrades oxidatively damaged and ubiquitinated proteins. The second, the autophagy pathway is in itself an inbuilt intracellular waste disposal system [120,121,122,123,124].
9 Why Drusen Elimination Might Be Undesirable
HSPs, the proteasome, and cellular autophagy are the three key intracellular pathways that maintain RPE homeostasis. In aging, all are increasingly compromised leading to activation of the nuclear factor-kappaB (NF-kappaB) signalling pathway. NF-kappaB, a transcription factor that plays a critical role in diverse cellular processes associated with proliferation, cell death, and development; as well as innate and adaptive immune responses, is normally sequestered in the cytoplasm by a family of inhibitory proteins known as inhibitors of NF-kappaB. Activation of NF-kappaB leads to the assembly of the NLRP3 inflammasome and activation of a tissue repair response [119].
Clinically, we recognize this stage of the immune response to aging as the typical inflammaging-associated AMD phenotype of soft drusen and pigmentary alterations at the macula. In other words, these clinical characteristics are key biomarkers of what is happening at the cellular and molecular level. As biomarkers they are important because they inform us that the RPE is chronically stressed and functionally compromised, but still viable. From a natural history perspective, the disappearance of these lesions is often associated with the death of RPE leading to ARGA and visual loss [125]. Once begun, the process of ARGA is progressive [96].
Although drusen are a sign of impaired autophagy and inflammaging (and indirectly of impaired proteasomal degradation and HSP function), their presence nonetheless indicates that these pathways are still functional [93]. SDM appears to reduce the risks of visual loss in high-risk AMD from both ARGA and CNV by improving, rather than further stressing and decompensating RPE function; and by preserving the integrity of the RPE/Bruch’s membrane complex [11, 26, 56].
As noted above, the NF kappaB pathway, resulting in the assembly of the NLRP3 inflammasome, appears to be crucial to the pathogenesis of AMD [126]. The biological significance of NLRP3 inflammasome activation is the release of active IL-1β and IL-18 into the extracellular space. The secreted IL-1β facilitates the chronic inflammatory response in the tissues while IL-18 promotes caspase-3 dependent RPE apoptosis, both hallmarks of AMD [65]. Inhibition of IL-1β and IL-18 expression via RPE HSP activation is thus yet another point in the disease process where retinal laser sublethal to the RPE may reduce the risk of CNV in AMD by inhibiting chronic inflammation [127].
Finally, at the tissue level, retinal laser-induced inflammatory recalibration in the direction of restoration and repair and away from progressive degeneration has local microglial and systemic components. These include activation of resident retinal stem cells and recruitment of bone marrow-derived stem cells, a potent combination that can foster repair, regeneration, and functional restoration [54, 55, 111].
While difficult to demonstrate clinically in dry AMD, the images in Figs. 30.1, 30.2, 30.3, 30.4, 30.5 illustrate the anti-inflammatory effects of panmacular and total retinal SDM as monotherapy in various other clinical settings described above. Note also the complete absence of LIRD in each case.
10 Summary
In summary, as the most important cause of CNV and related visual loss, AMD is a complex web of dysfunctional intracellular and extracellular events involving hundreds, if not thousands, of signalling molecules in innumerable interrelations. At our current level of understanding it is hard to envisage a targeted therapy, acting on a specific molecule(s) at a particular point in a particular pathway(s), that might singularly modulate a disease process of such complexity. The complexity of targeted therapeutic alteration of cell physiology dwarfs the comparative simplicity of binding VEGF in the extracellular space accounting for the failure of targeted drug therapy to prevent AMD—and thus CNV—to date. Yet the judicious application of light seems to do exactly that. Rather than attempting to selectively manipulate cell chemistry, retinal laser awakens powerful and fundamental mechanisms of cellular repair and functional restoration harnessing this same biologic complexity to advantage. Acting on a fraction of proteins in a single cell via HSP activation, laser is a catalyst launching a multitude of cascading effects within and far beyond the cell, resulting in physiologic—and thus ideal—functional normalization, “resetting” the RPE. The resultant reversal of the chronic disease process reduces the risks of visual loss—including the development of CNV. Absent LIRD, the benefits of modern laser are unopposed by adverse treatment effects and are greatest in eyes with the highest risks of age-related visual loss [11, 56]. Conversely, LIRD is adverse to retinal integrity and retinal and visual function. In AMD, LIRD may accelerate retinal degeneration, further increasing the risk of visual loss from ARGA and CNV, especially in the sickest, most compromised and vulnerable eyes [44, 52, 53]. Thus drusen elimination requiring LIRD appears undesirable, especially in the most functionally compromised high-risk eyes, and of uncertain long-term benefit in eyes with early AMD. In contrast, retinal laser treatment sublethal to the RPE, and thus wholly therapeutic, appears to hold great promise in the prevention of visual loss from chronic progressive retinopathies in general, and from CNV in AMD in particular. Further study will be illuminating.
Key Learning Points
-
1.
While CNV may result from focal damage to the macula from a number of causes, the main cause is chronic progressive disease, principally age-related macular degeneration.
-
2.
All chronic progressive retinopathies (CPRs) are neurodegenerations, and as such have much in common despite disparate etiologies and phenotypes.
-
3.
Dysfunction of the retinal pigment epithelium is a key commonality of all CRPs, leading to the second key commonality of chronic, self-perpetuating, degenerative inflammation. These are the key predisposing factors to CNV in AMD.
-
4.
The therapeutic effects of retinal laser treatment improve RPE and thus retinal function, and are antagonistic to chronic inflammation, resulting in repair and functional restoration.
-
5.
Laser-induced retinal damage (LIRD) is the cause of all adverse treatment effects and compromises the therapeutic benefits of laser treatment while offering no unique benefits over modern retinal laser therapy which is reliably sublethal to the RPE. LIRD increases AMD progression and the risks of visual loss, including CNV, particularly in high-risk eyes.
-
6.
By eliminating LIRD and maximizing therapeutic laser effects, modern retinal laser, epitomized by low-intensity/high-density subthreshold diode micropulse laser (SDM), safely slows disease progression, reducing the risks of visual loss and CNV in AMD, especially in the highest risk eyes.
-
7.
By addressing the commonalities of retinal neurodegenerations via non-targeted, pathoselective disease-specific repair, SDM may offer similar benefits in other retinopathies that predispose to macular CNV.
Financial Disclosures
Dr. Luttrull:
Ojai Retinal Technologies, LLC—Management, Patent, Equity.
Retinal Protection Sciences, LLC—Management, Equity.
Replenish, Inc.—Patent, Equity.
Dr. Kent:
None.
References
Jepson CN, Wetzig PC. Photocoagulation in disciform macular degeneration. Am J Ophthalmol 1969;67(6):920–930. Epub 1969/06/01. PubMed PMID: 5785852.
Argon laser photocoagulation for senile macular degeneration. Results of a randomized clinical trial. Arch Ophthalmol 1982;100(6):912–918. Epub 1982/06/01. PubMed PMID: 7046707.
Avila MP, Weiter JJ, Jalkh AE, Trempe CL, Pruett RC, Schepens CL. Natural history of choroidal neovascularization in degenerative myopia. Ophthalmology 1984;91(12):1573–1581. Epub 1984/12/01. PubMed PMID: 6084222.
Francois J, De Laey JJ, Cambie E, Hanssens M, Victoria-Troncoso V. Neovascularization after argon laser photocoagulation of macular lesions. Am J Ophthalmol 1975;79(2):206–210. Epub 1975/02/01. PubMed PMID: 1167736.
Recurrent choroidal neovascularization after argon laser photocoagulation for neovascular maculopathy. Macular Photocoagulation Study Group. Arch Ophthalmol 1986;104(4):503–512. Epub 1986/04/01. PubMed PMID: 2420315.
A randomized, placebo-controlled, clinical trial of high-dose supplementation with vitamins C and E, beta carotene, and zinc for age-related macular degeneration and vision loss: AREDS report no. 8. Arch Ophthalmol. 2001;119(10):1417–36. Epub 2001/10/23. PubMed PMID: 11594942; PubMed Central PMCID: PMC1462955.
Heier JS, Antoszyk AN, Pavan PR, Leff SR, Rosenfeld PJ, Ciulla TA, et al. Ranibizumab for treatment of neovascular age-related macular degeneration: a phase I/II multicenter, controlled, multidose study. Ophthalmology. 2006;113(4):633.e1–4. Epub 2006/02/18. https://doi.org/10.1016/j.ophtha.2005.10.052. PubMed PMID: 16483659.
Chhablani J, Roh YJ, Jobling AI, Fletcher EL, Lek JJ, Bansal P, et al. Restorative retinal laser therapy: present state and future directions. Surv Ophthalmol. 2017. Epub 2017/10/11. https://doi.org/10.1016/j.survophthal.2017.09.008. PubMed PMID: 28987614.
Guymer RH, Brassington KH, Dimitrov P, Makeyeva G, Plunkett M, Xia W, et al. Nanosecond-laser application in intermediate AMD: 12-month results of fundus appearance and macular function. Clin Exp Ophthalmol 2014;42(5):466–479. Epub 2013/10/15. https://doi.org/10.1111/ceo.12247. PubMed PMID: 24118741.
Jobling AI, Guymer RH, Vessey KA, Greferath U, Mills SA, Brassington KH, et al. Nanosecond laser therapy reverses pathologic and molecular changes in age-related macular degeneration without retinal damage. FASEB J 2015;29(2):696–710. Epub 2014/11/14. https://doi.org/10.1096/fj.14-262444. PubMed PMID: 25392267.
Luttrull JK, Sinclair SH, Elmann S, Glaser BM. Low incidence of choroidal neovascularization following subthreshold diode micropulse laser (SDM) in high-risk AMD. PLoS One 2018;13(8):e0202097. Epub 2018/08/24. https://doi.org/10.1371/journal.pone.0202097. PubMed PMID: 30138455.
Friedman DS, O'Colmain BJ, Munoz B, Tomany SC, McCarty C, de Jong PT, et al. Prevalence of age-related macular degeneration in the United States. Arch Ophthalmol 2004;122(4):564–572. Epub 2004/04/14. https://doi.org/10.1001/archopht.122.4.564. PubMed PMID: 15078675.
Wong WL, Su X, Li X, Cheung CM, Klein R, Cheng CY, et al. Global prevalence of age-related macular degeneration and disease burden projection for 2020 and 2040: a systematic review and meta-analysis. Lancet Glob Health 2014;2(2):e106–e116. Epub 2014/08/12. https://doi.org/10.1016/s2214-109x(13)70145-1. PubMed PMID: 25104651.
Molnar I, Pournaras CJ, Tsacopoulos M, Gilodi N, Leuenberger PM. [Panphotocoagulation. Mechanism of action and early indications]. Ophtalmologie: organe de la Societe francaise d’ophtalmologie 1987;1(4):449–452. Epub 1987/10/01. PubMed PMID: 3153915.
Shimizu K. Light coagulation in diabetic retinopathy and the mechanism of its effect. Ganka Ophthalmol 1971;13(11):1001–1006. Epub 1971/11/01. PubMed PMID: 5169175.
Stefansson E. Oxygen and diabetic eye disease. Graefes Arch Clin Exp Ophthalmol 1990;228(2):120–123. Epub 1990/01/01. PubMed PMID: 2186971.
Brader HS, Young LH. Subthreshold diode micropulse laser: a review. Semin Ophthalmol 2016;31(1–2):30–39. Epub 2016/03/10. https://doi.org/10.3109/08820538.2015.1114837. PubMed PMID: 26959127.
Chen G, Tzekov R, Li W, Jiang F, Mao S, Tong Y. Subthreshold micropulse diode laser versus conventional laser photocoagulation for diabetic macular Edema: a meta-analysis of randomized controlled trials. Retina 2016;36(11):2059–2065. Epub 2016/10/25. https://doi.org/10.1097/iae.0000000000001053. PubMed PMID: 27096529.
Luttrull JK, Musch DC, Mainster MA. Subthreshold diode micropulse photocoagulation for the treatment of clinically significant diabetic macular oedema. Br J Ophthalmol 2005;89(1):74–80. Epub 2004/12/24. https://doi.org/10.1136/bjo.2004.051540. PubMed PMID: 15615751; PubMed Central PMCID: PMC1772486.
Luttrull JK, Musch DC, Spink CA. Subthreshold diode micropulse panretinal photocoagulation for proliferative diabetic retinopathy. Eye (Lond) 2008;22(5):607–612. Epub 2007/02/13. https://doi.org/10.1038/sj.eye.6702725. PubMed PMID: 17293791.
Luttrull JK, Spink CJ. Serial optical coherence tomography of subthreshold diode laser micropulse photocoagulation for diabetic macular edema. Ophthalmic Surg Lasers Imaging 2006;37(5):370–377. Epub 2006/10/05. PubMed PMID: 17017196.
Luttrull JK, Chang DB, Margolis BW, Dorin G, Luttrull DK. Laser resesitization of medically unresponsive neovascular age-related macular degeneration: efficacy and Implications. Retina 2015;35(6):1184–1194. Epub 2015/02/05. https://doi.org/10.1097/iae.0000000000000458. PubMed PMID: 25650711.
Luttrull JK, Margolis BW. Functionally guided retinal protective therapy for dry age-related macular and inherited retinal degenerations: a pilot study. Invest Ophthalmol Vis Sci 2016;57(1):265–275. Epub 2016/01/29. https://doi.org/10.1167/iovs.15-18163. PubMed PMID: 26818793.
Luttrull JK. Improved retinal and visual function following panmacular subthreshold diode micropulse laser for retinitis pigmentosa. Eye (Lond). 2018;32(6):1099–110. Epub 2018/02/17. https://doi.org/10.1038/s41433-018-0017-3. PubMed PMID: 29449615; PubMed Central PMCID: PMCPMC5997672.
Luttrull JK, Samples JR, Kent D, Lum BJ. Panmacular subthreshold diode micropulse laser (SDM) as neuroprotective therapy in primary open-angle glaucoma. In: Samples JR, Kneppper PA, editors. Glaucoma research 2018–2020. Amsterdam: Kugler Publications; 2018. p. 281–94.
Abdelsalam A, Del Priore L, Zarbin MA. Drusen in age-related macular degeneration: pathogenesis, natural course, and laser photocoagulation-induced regression. Surv Ophthalmol 1999;44(1):1–29. Epub 1999/08/31. PubMed PMID: 10466585.
Bressler NM, Munoz B, Maguire MG, Vitale SE, Schein OD, Taylor HR, et al. Five-year incidence and disappearance of drusen and retinal pigment epithelial abnormalities. Waterman Stud Arch Ophthalmol 1995;113(3):301–308. Epub 1995/03/01. PubMed PMID: 7534060.
Bressler SB, Maguire MG, Bressler NM, Fine SL. Relationship of drusen and abnormalities of the retinal pigment epithelium to the prognosis of neovascular macular degeneration. The Macular Photocoagulation Study Group. Arch Ophthalmol 1990;108(10):1442–1447. Epub 1990/10/01. PubMed PMID: 1699513.
Chamberlin JA, Bressler NM, Bressler SB, Elman MJ, Murphy RP, Flood TP, et al. The use of fundus photographs and fluorescein angiograms in the identification and treatment of choroidal neovascularization in the Macular Photocoagulation Study. The Macular Photocoagulation Study Group Ophthalmology 1989;96(10):1526–1534. Epub 1989/10/01. PubMed PMID: 2479899.
Smiddy WE, Fine SL. Prognosis of patients with bilateral macular drusen. Ophthalmology 1984;91(3):271–277. Epub 1984/03/01. PubMed PMID: 6201789.
Ho AC, Maguire MG, Yoken J, Lee MS, Shin DS, Javornik NB, et al. Laser-induced drusen reduction improves visual function at 1 year. Choroidal Neovascularization Prevention Trial Research Group. Ophthalmology 1999;106(7):1367–1373.; discussion 74. Epub 1999/07/16. PubMed PMID: 10406624.
Olk RJ, Friberg TR, Stickney KL, Akduman L, Wong KL, Chen MC, et al. Therapeutic benefits of infrared (810-nm) diode laser macular grid photocoagulation in prophylactic treatment of nonexudative age-related macular degeneration: two-year results of a randomized pilot study. Ophthalmology 1999;106(11):2082–2090. Epub 1999/11/26. https://doi.org/10.1016/s0161-6420(99)90487-6. PubMed PMID: 10571341.
Dithmar S, Pollithy S, Ach T. Disappearance of central confluent soft drusen following vitrectomy and ILM peeling. Eye (Lond). 2013;27(6):779–81. Epub 2013/04/06. https://doi.org/10.1038/eye.2013.35. PubMed PMID: 23558209; PubMed Central PMCID: PMCPMC3682349.
Lim LT, Holicki J, Walker JD. Disappearance of extramacular drusen after chronic retinal detachment. Oman journal of ophthalmology. 2012;5(1):67. Epub 2012/05/05. https://doi.org/10.4103/0974-620x.94793. PubMed PMID: 22557885; PubMed Central PMCID: PMCPMC3339683.
Duvall J, Tso MO. Cellular mechanisms of resolution of drusen after laser coagulation. An experimental study. Arch Ophthalmol 1985;103(5):694–703. Epub 1985/05/01. PubMed PMID: 4039560.
Argon laser photocoagulation for neovascular maculopathy. Three-year results from randomized clinical trials. Macular Photocoagulation Study Group. Arch Ophthalmol. 1986;104(5):694–701. Epub 1986/05/01. PubMed PMID: 2423061.
Treatment techniques and clinical guidelines for photocoagulation of diabetic macular edema. Early Treatment Diabetic Retinopathy Study Report Number 2. Early Treatment Diabetic Retinopathy Study Research Group. Ophthalmology. 1987;94(7):761–74. Epub 1987/07/01. PubMed PMID: 3658348.
Akduman L, Olk RJ. Subthreshold (invisible) modified grid diode laser photocoagulation in diffuse diabetic macular edema (DDME). Ophthalmic Surg Lasers 1999;30(9):706–714. Epub 1999/11/26. PubMed PMID: 10574491.
Laser treatment in patients with bilateral large drusen: the complications of age-related macular degeneration prevention trial. Ophthalmology. 2006;113(11):1974–86. Epub 2006/11/01. https://doi.org/10.1016/j.ophtha.2006.08.015. PubMed PMID: 17074563.
Owens SL, Bunce C, Brannon AJ, Wormald R, Bird AC. Prophylactic laser treatment appears to promote choroidal neovascularisation in high-risk ARM: results of an interim analysis. Eye (Lond) 2003;17(5):623–627. Epub 2003/07/12. https://doi.org/10.1038/sj.eye.6700442. PubMed PMID: 12855972.
Owens SL, Bunce C, Brannon AJ, Xing W, Chisholm IH, Gross M, et al. Prophylactic laser treatment hastens choroidal neovascularization in unilateral age-related maculopathy: final results of the drusen laser study. Am J Ophthalmol 2006;141(2):276–281. Epub 2006/02/07. https://doi.org/10.1016/j.ajo.2005.08.019. PubMed PMID: 16458680.
Kaiser RS, Berger JW, Maguire MG, Ho AC, Javornik NB. Laser burn intensity and the risk for choroidal neovascularization in the CNVPT Fellow Eye Study. Arch Ophthalmol 2001;119(6):826–832. Epub 2001/06/19. PubMed PMID: 11405833.
Rodanant N, Friberg TR, Cheng L, Aurora A, Bartsch D, Toyoguchi M, et al. Predictors of drusen reduction after subthreshold infrared (810 nm) diode laser macular grid photocoagulation for nonexudative age-related macular degeneration. Am J Ophthalmol 2002;134(4):577–585. Epub 2002/10/18. PubMed PMID: 12383815.
Virgili G, Michelessi M, Parodi MB, Bacherini D, Evans JR. Laser treatment of drusen to prevent progression to advanced age-related macular degeneration. The Cochrane database of systematic reviews. 2015;(10):Cd006537. Epub 2015/10/24. https://doi.org/10.1002/14651858.CD006537.pub3. PubMed PMID: 26493180; PubMed Central PMCID: PMCPmc4733883.
Dorin G. Subthreshold and micropulse diode laser photocoagulation. Semin Ophthalmol 2003;18(3):147–153. Epub 2004/10/30. https://doi.org/10.1076/soph.18.3.147.29812. PubMed PMID: 15513476.
Luttrull JK, Sramek C, Palanker D, Spink CJ, Musch DC. Long-term safety, high-resolution imaging, and tissue temperature modeling of subvisible diode micropulse photocoagulation for retinovascular macular edema. Retina 2012;32(2):375–386. Epub 2011/10/06. https://doi.org/10.1097/IAE.0b013e3182206f6c. PubMed PMID: 21971077.
Lavinsky D, Wang J, Huie P, Dalal R, Lee SJ, Lee DY, et al. Nondamaging Retinal Laser Therapy: Rationale and Applications to the Macula. Invest Ophthalmol Vis Sci. 2016;57(6):2488–500. Epub 2016/05/10. https://doi.org/10.1167/iovs.15-18981. PubMed PMID: 27159441; PubMed Central PMCID: PMCPMC5995023.
Roider J, Brinkmann R, Wirbelauer C, Laqua H, Birngruber R. Subthreshold (retinal pigment epithelium) photocoagulation in macular diseases: a pilot study. Br J Ophthalmol. 2000;84(1):40–7. Epub 1999/12/28. PubMed PMID: 10611098; PubMed Central PMCID: PMCPMC1723228.
Sramek C, Mackanos M, Spitler R, Leung LS, Nomoto H, Contag CH, et al. Non-damaging retinal phototherapy: dynamic range of heat shock protein expression. Invest Ophthalmol Vis Sci 2011;52(3):1780–1787. Epub 2010/11/20. https://doi.org/10.1167/iovs.10-5917. PubMed PMID: 21087969.
Sramek CK, Leung LS, Paulus YM, Palanker DV. Therapeutic window of retinal photocoagulation with green (532-nm) and yellow (577-nm) lasers. Ophthalmic Surg Lasers Imaging 2012;43(4):341–347. Epub 2012/05/17. https://doi.org/10.3928/15428877-20120426-05. PubMed PMID: 22589338.
Roider J, Brinkmann R, Wirbelauer C, Birngruber R, Laqua H. Variability of RPE reaction in two cases after selective RPE laser effects in prophylactic treatment of drusen. Graefes Arch Clin Exp Ophthalmol 1999;237(1):45–50. Epub 1999/02/10. PubMed PMID: 9951641.
Guymer RH, Wu Z, Hodgson LAB, Caruso E, Brassington KH, Tindill N, et al. Subthreshold nanosecond laser intervention in age-related macular degeneration: the LEAD randomized controlled clinical trial ophthalmology 2018. Epub 2018/09/24. https://doi.org/10.1016/j.ophtha.2018.09.015. PubMed PMID: 30244144.
Prahs P, Walter A, Regler R, Theisen-Kunde D, Birngruber R, Brinkmann R, et al. Selective retina therapy (SRT) in patients with geographic atrophy due to age-related macular degeneration. Graefes Arch Clin Exp Ophthalmol 2010;248(5):651–658. Epub 2009/12/22. https://doi.org/10.1007/s00417-009-1208-1. PubMed PMID: 20024687.
Midena E, Pilotto E. Emerging Insights into Pathogenesis. Dev Ophthalmol 2017;60:16–27. Epub 2017/04/21. https://doi.org/10.1159/000459687. PubMed PMID: 28427061.
Vujosevic S, Micera A, Bini S, Berton M, Esposito G, Midena E. Proteome analysis of retinal glia cells-related inflammatory cytokines in the aqueous humour of diabetic patients. Acta Ophthalmol 2016;94(1):56–64. Epub 2015/08/14. https://doi.org/10.1111/aos.12812. PubMed PMID: 26268591.
Luttrull JK. Slowed progression of age-related geographic atrophy following subthreshold diode micropulse laser—in review. 2019.
Luttrull JK, Sinclair SH. Safety of transfoveal subthreshold diode micropulse laser for fovea-involving diabetic macular edema in eyes with good visual acuity. Retina 2014;34(10):2010–2020. Epub 2014/05/20. https://doi.org/10.1097/iae.0000000000000177. PubMed PMID: 24837050.
Luttrull JK, Dorin G. Subthreshold diode micropulse laser photocoagulation (SDM) as invisible retinal phototherapy for diabetic macular edema: a review. Curr Diabetes Rev 2012;8(4):274–284. Epub 2012/05/17. PubMed PMID: 22587512; PubMed Central PMCID: PMC3412206.
Lavinsky D, Cardillo JA, Melo LA, Jr., Dare A, Farah ME, Belfort R, Jr. Randomized clinical trial evaluating mETDRS versus normal or high-density micropulse photocoagulation for diabetic macular edema. Invest Ophthalmol Vis Sci 2011;52(7):4314–4323. Epub 2011/02/25. https://doi.org/10.1167/iovs.10-6828. PubMed PMID: 21345996.
Franceschi C, Campisi J. Chronic inflammation (inflammaging) and its potential contribution to age-associated diseases. J Gerontol A Biol Sci Med Sci 2014;69 Suppl 1:S4–S9. Epub 2014/05/17. https://doi.org/10.1093/gerona/glu057. PubMed PMID: 24833586.
Howcroft TK, Campisi J, Louis GB, Smith MT, Wise B, Wyss-Coray T, et al. The role of inflammation in age-related disease. Aging (Albany NY). 2013;5(1):84–93. Epub 2013/03/12. https://doi.org/10.18632/aging.100531. PubMed PMID: 23474627; PubMed Central PMCID: PMC3616233.
Baldwin RL. Energetics of protein folding. J Mol Biol 2007;371(2):283–301. Epub 2007/06/22. https://doi.org/10.1016/j.jmb.2007.05.078. PubMed PMID: 17582437.
Kregel KC. Heat shock proteins: modifying factors in physiological stress responses and acquired thermotolerance. J Appl Physiol (1985). 2002;92(5):2177–86. Epub 2002/04/19. https://doi.org/10.1152/japplphysiol.01267.2001. PubMed PMID: 11960972.
Medzhitov R. Origin and physiological roles of inflammation. Nature 2008;454(7203):428–435. Epub 2008/07/25. https://doi.org/10.1038/nature07201. PubMed PMID: 18650913.
Ambati J, Atkinson JP, Gelfand BD. Immunology of age-related macular degeneration. Nat Rev Immunol 2013;13(6):438–451. Epub 2013/05/25. https://doi.org/10.1038/nri3459. PubMed PMID: 23702979; PubMed Central PMCID: PMC3941009.
Xu H, Chen M, Forrester JV. Para-inflammation in the aging retina. Prog Retin Eye Res 2009;28(5):348–368. Epub 2009/06/30. https://doi.org/10.1016/j.preteyeres.2009.06.001. PubMed PMID: 19560552.
Doyle SL, Campbell M, Ozaki E, Salomon RG, Mori A, Kenna PF, et al. NLRP3 has a protective role in age-related macular degeneration through the induction of IL-18 by drusen components. Nat Med 2012;18(5):791–798. Epub 2012/04/10. https://doi.org/10.1038/nm.2717. PubMed PMID: 22484808; PubMed Central PMCID: PMC3984677.
Hollyfield JG, Bonilha VL, Rayborn ME, Yang X, Shadrach KG, Lu L, et al. Oxidative damage-induced inflammation initiates age-related macular degeneration. Nat Med 2008;14(2):194–198. Epub 2008/01/29. https://doi.org/10.1038/nm1709. PubMed PMID: 18223656; PubMed Central PMCID: PMC2748836.
Kaneko H, Dridi S, Tarallo V, Gelfand BD, Fowler BJ, Cho WG, et al. DICER1 deficit induces Alu RNA toxicity in age-related macular degeneration. Nature 2011;471(7338):325–330. Epub 2011/02/08. https://doi.org/10.1038/nature09830. PubMed PMID: 21297615; PubMed Central PMCID: PMC3077055.
Tarallo V, Hirano Y, Gelfand BD, Dridi S, Kerur N, Kim Y, et al. DICER1 loss and Alu RNA induce age-related macular degeneration via the NLRP3 inflammasome and MyD88. Cell 2012;149(4):847–859. Epub 2012/05/01. https://doi.org/10.1016/j.cell.2012.03.036. PubMed PMID: 22541070; PubMed Central PMCID: PMC3351582.
Tseng WA, Thein T, Kinnunen K, Lashkari K, Gregory MS, D’Amore PA, et al. NLRP3 inflammasome activation in retinal pigment epithelial cells by lysosomal destabilization: implications for age-related macular degeneration. Invest Ophthalmol Vis Sci 2013;54(1):110–120. Epub 2012/12/12. https://doi.org/10.1167/iovs.12-10655. PubMed PMID: 23221073; PubMed Central PMCID: PMC3544415.
Anderson DH, Radeke MJ, Gallo NB, Chapin EA, Johnson PT, Curletti CR, et al. The pivotal role of the complement system in aging and age-related macular degeneration: hypothesis re-visited. Prog Retin Eye Res 2010;29(2):95–112. Epub 2009/12/08. https://doi.org/10.1016/j.preteyeres.2009.11.003. PubMed PMID: 19961953.
Clark SJ, Perveen R, Hakobyan S, Morgan BP, Sim RB, Bishop PN, et al. Impaired binding of the age-related macular degeneration-associated complement factor H 402H allotype to Bruch's membrane in human retina. J Biol Chem 2010;285(39):30192–30202. Epub 2010/07/28. https://doi.org/10.1074/jbc.M110.103986. PubMed PMID: 20660596; PubMed Central PMCID: PMC2943316.
Johnson PT, Betts KE, Radeke MJ, Hageman GS, Anderson DH, Johnson LV. Individuals homozygous for the age-related macular degeneration risk-conferring variant of complement factor H have elevated levels of CRP in the choroid. Proc Natl Acad Sci USA 2006;103(46):17456–17461. Epub 2006/11/03. https://doi.org/10.1073/pnas.0606234103. PubMed PMID: 17079491; PubMed Central PMCID: PMC1859950.
Khandhadia S, Cipriani V, Yates JR, Lotery AJ. Age-related macular degeneration and the complement system. Immunobiology 2012;217(2):127–146. Epub 2011/08/27. https://doi.org/10.1016/j.imbio.2011.07.019. PubMed PMID: 21868123.
Lachmann PJ. The amplification loop of the complement pathways. Adv Immunol 2009;104:115–149. Epub 2009/01/01. https://doi.org/10.1016/s0065-2776(08)04004-2. PubMed PMID: 20457117.
Laine M, Jarva H, Seitsonen S, Haapasalo K, Lehtinen MJ, Lindeman N, et al. Y402H polymorphism of complement factor H affects binding affinity to C-reactive protein. J Immunol 2007;178(6):3831–3836. Epub 2007/03/07. PubMed PMID: 17339482.
Richards A, Kavanagh D, Atkinson JP. Inherited complement regulatory protein deficiency predisposes to human disease in acute injury and chronic inflammatory states the examples of vascular damage in atypical hemolytic uremic syndrome and debris accumulation in age-related macular degeneration. Adv Immunol 2007;96:141–177. Epub 2007/11/06. https://doi.org/10.1016/s0065-2776(07)96004-6. PubMed PMID: 17981206.
Sjoberg AP, Trouw LA, Clark SJ, Sjolander J, Heinegard D, Sim RB, et al. The factor H variant associated with age-related macular degeneration (His-384) and the non-disease-associated form bind differentially to C-reactive protein, fibromodulin, DNA, and necrotic cells. J Biol Chem 2007;282(15):10894–10900. Epub 2007/02/13. https://doi.org/10.1074/jbc.M610256200. PubMed PMID: 17293598.
Tuo J, Grob S, Zhang K, Chan CC. Genetics of immunological and inflammatory components in age-related macular degeneration. Ocul Immunol Inflamm 2012;20(1):27–36. Epub 2012/02/14. https://doi.org/10.3109/09273948.2011.628432. PubMed PMID: 22324898; PubMed Central PMCID: PMC3292785.
Clark RA. Basics of cutaneous wound repair. J Dermatol Surg Oncol 1993;19(8):693–706. Epub 1993/08/01. PubMed PMID: 8349909.
Singer AJ, Clark RA. Cutaneous wound healing. N Engl J Med 1999;341(10):738–746. Epub 1999/09/02. https://doi.org/10.1056/nejm199909023411006. PubMed PMID: 10471461.
Hageman GS, Anderson DH, Johnson LV, Hancox LS, Taiber AJ, Hardisty LI, et al. A common haplotype in the complement regulatory gene factor H (HF1/CFH) predisposes individuals to age-related macular degeneration. Proc Natl Acad Sci USA 2005;102(20):7227–7232. Epub 2005/05/05. https://doi.org/10.1073/pnas.0501536102. PubMed PMID: 15870199; PubMed Central PMCID: PMC1088171.
Haines JL, Hauser MA, Schmidt S, Scott WK, Olson LM, Gallins P, et al. Complement factor H variant increases the risk of age-related macular degeneration. Science 2005;308(5720):419–421. Epub 2005/03/12. https://doi.org/10.1126/science.1110359. PubMed PMID: 15761120.
Klein RJ, Zeiss C, Chew EY, Tsai JY, Sackler RS, Haynes C, et al. Complement factor H polymorphism in age-related macular degeneration. Science 2005;308(5720):385–389. Epub 2005/03/12. https://doi.org/10.1126/science.1109557. PubMed PMID: 15761122; PubMed Central PMCID: PMC1512523.
Bonafe M, Storci G, Franceschi C. Inflamm-aging of the stem cell niche: breast cancer as a paradigmatic example: breakdown of the multi-shell cytokine network fuels cancer in aged people. BioEssays 2012;34(1):40–49. Epub 2011/11/17. https://doi.org/10.1002/bies.201100104. PubMed PMID: 22086861.
Varadhan R, Yao W, Matteini A, Beamer BA, Xue QL, Yang H, et al. Simple biologically informed inflammatory index of two serum cytokines predicts 10 year all-cause mortality in older adults. J Gerontol A Biol Sci Med Sci 2014;69(2):165–173. Epub 2013/05/22. https://doi.org/10.1093/gerona/glt023. PubMed PMID: 23689826; PubMed Central PMCID: PMC4038244.
Tobe T, Ortega S, Luna JD, Ozaki H, Okamoto N, Derevjanik NL, et al. Targeted disruption of the FGF2 gene does not prevent choroidal neovascularization in a murine model. Am J Pathol 1998;153(5):1641–1646. Epub 1998/11/12. https://doi.org/10.1016/s0002-9440(10)65753-7. PubMed PMID: 9811357; PubMed Central PMCID: PMC1853405.
Kent D, Sheridan C. Choroidal neovascularization: a wound healing perspective. Mol Vis. 2003;9:747–55.
Clark RAF. Wound repair: overview and general considerations. In: Clark RAF, editor. The molecular and cellular biology of wound repair. Second ed. New York: Plenum; 1996. p. 1–50.
Hunt TK. Basic principles of wound healing. J Trauma 1990;30(12 Suppl):S122–S128. Epub 1990/12/01. PubMed PMID: 2254971.
Nathan C, Sporn M. Cytokines in context. J Cell Biol. 1991;113(5):981–6.
Blasiak J, Pawlowska E, Szczepanska J, Kaarniranta K. Interplay between Autophagy and the Ubiquitin-Proteasome System and Its Role in the Pathogenesis of Age-Related Macular Degeneration. International journal of molecular sciences. 2019;20(1). Epub 2019/01/11. https://doi.org/10.3390/ijms20010210. PubMed PMID: 30626110; PubMed Central PMCID: PMCPMC6337628.
Bornstein P. Diversity of function is inherent in matricellular proteins: an appraisal of thrombospondin 1. J Cell Biol. 1995;130(3):503–6.
Mones J, Garcia M, Biarnes M, Lakkaraju A, Ferraro L. Drusen Ooze: a novel hypothesis in geographic atrophy. Ophthalmol Retina 2017;1(6):461–473. Epub 2017/01/01. https://doi.org/10.1016/j.oret.2017.02.006. PubMed PMID: 31047436.
Schmitz-Valckenberg S, Sadda S, Staurenghi G, Chew EY, Fleckenstein M, Holz FG. Geographic atrophy: semantic considerations and literature review. Retina. 2016;36(12):2250–64. Epub 2016/08/24. https://doi.org/10.1097/iae.0000000000001258. PubMed PMID: 27552292; PubMed Central PMCID: PMCPMC5115977.
Pockley AG. Heat shock proteins, inflammation, and cardiovascular disease. Circulation 2002;105(8):1012–1017. Epub 2002/02/28. PubMed PMID: 11864934.
Fort PE, Lampi KJ. New focus on alpha-crystallins in retinal neurodegenerative diseases. Exp Eye Res. 2011;92(2):98–103. Epub 2010/12/01. https://doi.org/10.1016/j.exer.2010.11.008. PubMed PMID: 21115004; PubMed Central PMCID: PMCPmc4454605.
Dudeja V, Vickers SM, Saluja AK. The role of heat shock proteins in gastrointestinal diseases. Gut. 2009;58(7):1000–9. Epub 2009/06/13. https://doi.org/10.1136/gut.2007.140194. PubMed PMID: 19520890; PubMed Central PMCID: PMCPMC2896385.
Lu RC, Tan MS, Wang H, Xie AM, Yu JT, Tan L. Heat shock protein 70 in Alzheimer's disease. BioMed research international. 2014;2014:435203. Epub 2014/11/29. https://doi.org/10.1155/2014/435203. PubMed PMID: 25431764; PubMed Central PMCID: PMCPMC4241292.
Morimoto RI, Santoro MG. Stress-inducible responses and heat shock proteins: new pharmacologic targets for cytoprotection. Nat Biotechnol 1998;16(9):833–838. Epub 1998/09/22. https://doi.org/10.1038/nbt0998-833. PubMed PMID: 9743115.
Srivastava K, Narang R, Bhatia J, Saluja D. Expression of heat shock protein 70 gene and its correlation with inflammatory markers in essential hypertension. PLoS One. 2016;11(3):e0151060. Epub 2016/03/19. https://doi.org/10.1371/journal.pone.0151060. PubMed PMID: 26989902; PubMed Central PMCID: PMCPMC4798713.
Xu Q, Metzler B, Jahangiri M, Mandal K. Molecular chaperones and heat shock proteins in atherosclerosis. Am J Physiol Heart Circ Physiol. 2012;302(3):H506–14. Epub 2011/11/08. https://doi.org/10.1152/ajpheart.00646.2011. PubMed PMID: 22058161; PubMed Central PMCID: PMCPMC3353778.
Santoro MG. Heat shock factors and the control of the stress response. Biochem Pharmacol 2000;59(1):55–63. Epub 1999/12/22. PubMed PMID: 10605935.
Lanneau D, de Thonel A, Maurel S, Didelot C, Garrido C. Apoptosis versus cell differentiation: role of heat shock proteins HSP90, HSP70 and HSP27. Prion. 2007;1(1):53–60. Epub 2007/01/01. PubMed PMID: 19164900; PubMed Central PMCID: PMCPmc2633709.
De Cilla S, Vezzola D, Farruggio S, Vujosevic S, Clemente N, Raina G, et al. The subthreshold micropulse laser treatment of the retina restores the oxidant/antioxidant balance and counteracts programmed forms of cell death in the mice eyes. Acta Ophthalmol. 2018. Epub 2018/12/27. https://doi.org/10.1111/aos.13995. PubMed PMID: 30585429.
Gao X, Xing D. Molecular mechanisms of cell proliferation induced by low power laser irradiation. Journal of biomedical science. 2009;16:4. Epub 2009/03/11. https://doi.org/10.1186/1423-0127-16-4. PubMed PMID: 19272168; PubMed Central PMCID: PMCPMC2644974.
Iwami H, Pruessner J, Shiraki K, Brinkmann R, Miura Y. Protective effect of a laser-induced sub-lethal temperature rise on RPE cells from oxidative stress. Exp Eye Res 2014;124:37–47. Epub 2014/05/08. https://doi.org/10.1016/j.exer.2014.04.014. PubMed PMID: 24800654.
Karu TI, Kolyakov SF. Exact action spectra for cellular responses relevant to phototherapy. Photomed Laser Surg 2005;23(4):355–361. Epub 2005/09/08. https://doi.org/10.1089/pho.2005.23.355. PubMed PMID: 16144476.
Lund DJ, Sliney DH. A new understanding of multiple-pulsed laser-induced retinal injury thresholds. Health Phys 2014;106(4):505–515. Epub 2014/02/25. https://doi.org/10.1097/HP.0b013e3182a2a837. PubMed PMID: 24562071.
Caballero S, Kent DL, Sengupta N, Li Calzi S, Shaw L, Beli E, et al. Bone marrow-derived cell recruitment to the neurosensory retina and retinal pigment epithelial cell layer following subthreshold retinal phototherapy. Invest Ophthalmol Vis Sci. 2017.
Flaxel C, Bradle J, Acott T, Samples JR. Retinal pigment epithelium produces matrix metalloproteinases after laser treatment. Retina 2007;27(5):629–634. Epub 2007/06/15. https://doi.org/10.1097/01.iae.0000249561.02567.fd. PubMed PMID: 17558327.
Hattenbach LO, Beck KF, Pfeilschifter J, Koch F, Ohrloff C, Schacke W. Pigment-epithelium-derived factor is upregulated in photocoagulated human retinal pigment epithelial cells. Ophthalmic Res 2005;37(6):341–346. Epub 2005/09/15. https://doi.org/10.1159/000088263. PubMed PMID: 16158012.
Inagaki K, Shuo T, Katakura K, Ebihara N, Murakami A, Ohkoshi K. Sublethal photothermal stimulation with a micropulse laser induces heat shock protein expression in ARPE-19 cells. J Ophthalmol 2015;2015:729792. Epub 2015/12/24. https://doi.org/10.1155/2015/729792. PubMed PMID: 26697211; PubMed Central PMCID: PMC4677213.
Karu T. Photobiology of low-power laser effects. Health Phys 1989;56(5):691–704. Epub 1989/05/01. PubMed PMID: 2651364.
Midena E, Bini S, Martini F, Enrica C, Pilotto E, Micera A, et al. Changes of aqueous humor muller cells’ biomarkers in human patients affected by diabetic macular edema after subthreshold micropulse laser treatment. Retina. 2018. Epub 2018/10/10. https://doi.org/10.1097/iae.0000000000002356. PubMed PMID: 30300267.
Kaarniranta K, Tokarz P, Koskela A, Paterno J, Blasiak J. Autophagy regulates death of retinal pigment epithelium cells in age-related macular degeneration. Cell biology and toxicology. 2017;33(2):113–28. Epub 2016/12/03. https://doi.org/10.1007/s10565-016-9371-8. PubMed PMID: 27900566; PubMed Central PMCID: PMCPMC5325845.
Kaarniranta K, Elo M, Sironen R, Lammi MJ, Goldring MB, Eriksson JE, et al. Hsp70 accumulation in chondrocytic cells exposed to high continuous hydrostatic pressure coincides with mRNA stabilization rather than transcriptional activation. Proc Natl Acad Sci USA. 1998;95(5):2319–24. Epub 1998/04/16. PubMed PMID: 9482883; PubMed Central PMCID: PMCPMC19331.
Kaarniranta K, Salminen A, Eskelinen EL, Kopitz J. Heat shock proteins as gatekeepers of proteolytic pathways-Implications for age-related macular degeneration (AMD). Ageing Res Rev 2009;8(2):128–139. Epub 2009/03/12. PubMed PMID: 19274853.
Buchner J. Supervising the fold: functional principles of molecular chaperones. FASEB J 1996;10(1):10–19. Epub 1996/01/01. PubMed PMID: 8566529.
Johnson LV, Leitner WP, Rivest AJ, Staples MK, Radeke MJ, Anderson DH. The Alzheimer’s a beta-peptide is deposited at sites of complement activation in pathologic deposits associated with aging and age-related macular degeneration. Proc Natl Acad Sci USA 2002;99(18):11830–11835. Epub 2002/08/22. https://doi.org/10.1073/pnas.192203399. PubMed PMID: 12189211; PubMed Central PMCID: PMC129354.
Kopito RR. Aggresomes, inclusion bodies and protein aggregation. Trends Cell Biol 2000;10(12):524–530. Epub 2000/12/21. PubMed PMID: 11121744.
Ryhanen T, Hyttinen JM, Kopitz J, Rilla K, Kuusisto E, Mannermaa E, et al. Crosstalk between Hsp70 molecular chaperone, lysosomes and proteasomes in autophagy-mediated proteolysis in human retinal pigment epithelial cells. J Cell Mol Med. 2009;13(9b):3616–31. Epub 2008/11/20. https://doi.org/10.1111/j.1582-4934.2008.00577.x. PubMed PMID: 19017362; PubMed Central PMCID: PMCPMC4516511.
Ryhanen T, Viiri J, Hyttinen JM, Uusitalo H, Salminen A, Kaarniranta K. Influence of Hsp90 and HDAC inhibition and tubulin acetylation on perinuclear protein aggregation in human retinal pigment epithelial cells. J Biomed Biotechnol. 2011;2011:798052. Epub 2010/10/29. https://doi.org/10.1155/2011/798052. PubMed PMID: 20981255; PubMed Central PMCID: PMCPMC2963810.
Mones J, Garcia M, Biarnes M, Lakkaraju A, Ferraro L. Drusen Ooze: a novel hypothesis in geographic atrophy. Ophthalmol Retina. 2017;1:461–73.
Gao J, Liu RT, Cao S, Cui JZ, Wang A, To E, et al. NLRP3 inflammasome: activation and regulation in age-related macular degeneration. Mediators Inflamm. 2015;2015:690243. Epub 2015/02/24. https://doi.org/10.1155/2015/690243. PubMed PMID: 25698849; PubMed Central PMCID: PMCPMC4324923.
Martine P, Chevriaux A, Derangere V, Apetoh L, Garrido C, Ghiringhelli F, et al. HSP70 is a negative regulator of NLRP3 inflammasome activation. Cell Death Disease. 2019;10(4):256. Epub 2019/03/16. https://doi.org/10.1038/s41419-019-1491-7. PubMed PMID: 30874540; PubMed Central PMCID: PMCPMC6420651.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Luttrull, J.K., Kent, D. (2020). Laser for Prevention of Choroidal Neovascularization. In: Chhablani, J. (eds) Choroidal Neovascularization. Springer, Singapore. https://doi.org/10.1007/978-981-15-2213-0_30
Download citation
DOI: https://doi.org/10.1007/978-981-15-2213-0_30
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-15-2212-3
Online ISBN: 978-981-15-2213-0
eBook Packages: MedicineMedicine (R0)