Abstract
Enteric methane emission in ruminants in addition to being an environmental pollutant causes a loss of 10–11% of the total gross energy intake of the animal. The various strategies available to mitigate enteric methane emission include management strategies, feeding strategies, rumen manipulation and advanced strategies. Feeding strategies are practical approaches to mitigate enteric methane emission and can be practiced with ease by farmers under field conditions. Various approaches that cause rumen manipulation and thereby reduce enteric methane emission are supplementation of bacteriocins, ionophores, fats, oils, organic acids, probiotics, prebiotics, sulphate, halogenated methane analogues, nitroxy compounds, fungal metabolite, secondary plant metabolites, microalgae and exogenous enzymes. In the Indian context, ruminant livestock are grazed in wastelands or fed with poor-quality agricultural waste, whose digestibility is low, and the nutritional requirement of the animals is not met resulting in poor productivity. Improving per animal productivity is a potent tool to reduce enteric methane emission per unit of product produced, and this can be achieved through ration balancing. As regards the greenhouse gas abatement opportunities, measures to reduce enteric methane production have immense economic and ecological benefits to the farmers.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Ruminants
- Enteric methane emission
- Mitigation
- Livestock management
- Manure management
- Pasture management
- Feed management
- Rumen manipulation
- Secondary metabolites
8.1 Introduction
The “agriculture, forestry and other land use (AFOLU)” sector releases 10–12 Gt CO2e per annum. Greenhouse gas emissions from agricultural activities (Fig. 8.1) include “land use changes”; enteric methane emission from ruminants; lowland rice cultivation; N2O emissions from nitrogenous fertilizer use; and crop residue burning (Lipper et al. 2014; Smith et al. 2014; Venkatramanan and Shah 2019). Methane is a very significant greenhouse gas in view of its global warming potential and atmospheric lifetime. The causes for “increases in CH4 concentration include natural wetlands emissions (177–284 Tg CH4 yr–1), agriculture and waste (187–224 Tg CH4 yr–1), fossil fuel related emissions (85–105 Tg CH4 yr–1), other natural emissions (61–200 Tg CH4 yr–1), and biomass and biofuel burning (32–39 Tg CH4 yr–1)” (Venkatramanan and Shah 2019). Livestock are one of the largest single sources of anthropogenic methane emission in the world. Amongst livestock, ruminants (cattle, buffaloes, sheep and goats) are the principal sources of enteric methane emission.
About 30% of methane emissions at the global level are released from the ruminant activity. In other words, globally, 2.7 Gt of CO2e of enteric CH4 per annum are released from the ruminants. As regards the livestock methane emissions, 2.1 Gt (77%) of CO2e is contributed by cattle, 0.37 Gt (14%) of CO2e by buffaloes and 0.26 Gt CO2e by small ruminants like sheep and goats (FAO 2017).
In India, 49.1% of enteric methane was contributed by cattle, 42.8% by buffaloes, 5.38% by goats and 2.59% by sheep. Importantly, during 1961–2010, the increase in methane emissions (70.6%) from livestock population of India is much greater than the increase in methane emissions from livestock population worldwide (54.3%). It is reported that by 2050, about 15.7% of enteric CH4 emission at the global level will be contributed by the Livestock population of India (Patra 2014). One of the reasons for high enteric methane emission in India is the larger bovine population which emits more methane than any other livestock species. On an average, cattle and buffaloes aggregately emit more than 90% of the total enteric methane emission of the country. The contribution from small ruminants is relatively small and constitutes only 7.7%. Rest of the methane emissions arise from the species such as yak and mithun, which are scattered to specific states only. Enteric methane emission from crossbred cattle is comparatively much more than the emissions from indigenous cattle (46 versus 25 kg/animal/year). Enteric methane emission from livestock is not uniform across the states and varies considerably according to the livestock numbers, species, type of feed and fodders, etc. (Malik et al. 2015).
The National Institute of Animal Nutrition and Physiology, India, has developed a statewise inventory for enteric methane emission from Indian livestock. Their estimates revealed that Uttar Pradesh was the largest enteric methane emitting state and other major methane emitting states were Rajasthan, Madhya Pradesh, Bihar, West Bengal, Maharashtra, Karnataka and Andhra Pradesh. These states altogether have 66% of the total livestock population and are responsible for 68% enteric methane emissions (Bhata et al. 2016).
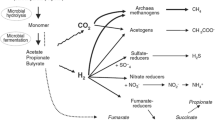
Enteric methane emission in livestock is a consequence of ruminal/hindgut fermentation. In the reticulo rumen or hindgut of livestock, simple and complex carbohydrates are hydrolyzed to 5 or 6 carbon sugars by enzymes secreted by microbes. These sugars are then fermented to volatile fatty acids through metabolic pathways that produce metabolic hydrogen. The metabolic hydrogen is converted to hydrogen by hydrogenase expressing bacterial species (Methanogens), and the hydrogen is converted to methane by archaea. In fact, the dissolved hydrogen through a negative feedback restrains fermentation pathways occurring in the rumen. Incidentally, the non-removal of dissolved hydrogen can lessen the degradation of carbohydrate, growth rate of microbes, and also the production of microbial protein (Wolin 1974; McAllister and Newbold 2008). Methanogenesis can be regarded as an evolutionary adaptation that enables the rumen ecosystem to dispose of hydrogen, which may otherwise accumulate and inhibit fibre degradation.
Enteric methane emission in ruminants in addition to being an environmental pollutant causes a loss of 10–11% of the total gross energy intake of the animals. Methane mitigation options are therefore associated with improved efficiency of animal production. Thus enteric methane mitigation measures would be advantageous both environmentally and nutritionally.
8.2 Strategies to Mitigate Enteric Methane Emission
There are various strategies available to mitigate enteric methane emission. The strategies may be broadly grouped as management strategies, feeding strategies, rumen manipulation and advanced strategies.
8.2.1 Management Strategies
Management strategies that could be adopted to control enteric methane emission are reducing the ruminant livestock population, breeding management and manure management.
8.2.1.1 Reducing the Ruminant Livestock Population
In underdeveloped and developing nations, agriculture and livestock rearing is a way of life and indeed contributes greatly to the income of the farmers. Nevertheless, unable to maintain and rear high producing ruminants, the small-scale farmers primarily meet their demands by maintaining a large number of livestock which are low producing ruminants (Tarawali et al. 2011). This can increase the total enteric methane emission in a given area. Thus the most effective methane mitigating strategy will be to increase livestock productivity, which may permit reduction in livestock numbers to provide the same product output at a reduced enteric methane emission. Higher animal performance also reduces methane emission because this can lead to a reduction in the number of animals in the production system.
8.2.1.2 Breeding Management
Adopting a superior breeding program to enhance per animal productivity is one of the best options to reduce enteric methane emission as the production of enteric methane is greatly reduced on account of improvement in the productivity of livestock. It is thus important to consider the enteric methane production per unit of the livestock product produced (kg of milk, meat, or wool).
Another school of thought is that genetics influences enteric methane emission in ruminants (Robertson and Waghorn 2002; Pinares-Patino et al. 2007). Different amounts of methane emission per unit intake of the same quality ration at the same level of performance were recorded, and accordingly livestock were categorized as “high” and “low” methane emitters (Pinares-Patino et al. 2007). Genetic selection and breeding of such animals that consume less feed and produce less methane per unit of feed may be one of the management strategies for reduction in methanogenesis.
8.2.1.3 Manure Management
Animal manure from both ruminant and non-ruminant livestock contributes to methane emission. Stored manure is an important source of methane emission (Priano et al. 2014). Manures are organic compounds which on decomposition by anaerobic bacteria transform their carbon skeleton into methane. Production of methane from manure from dairy cattle, beef cattle and dairy ewe was reported to be 33.2, 2.0 and 0.3 kg/head/year, respectively (Merino et al. 2011).
Many factors indeed influence the emission of methane from manure. Physical properties of manure like density, humidity, amount of digestible material and temperature influence methane emission (Priano et al. 2014; Saggar et al. 2004). The type of ration fed to the animal influences the manure quality and the quantity of methane emitted from it. Methane emissions from manure storage arise only when animals are intensively reared. When animals are managed on grazing this scenario does not arise. Methane emitted from manures can be greatly reduced by minimizing the duration of storage of manure and giving lesser time for the “microbial fermentation” to take place. Anaerobic digesters are recommended as a mitigation strategy for methane, and are a source of renewable energy as well and provide sanitation opportunities for developing countries.
8.2.2 Feeding Strategies
Feeding strategies are practical approaches to mitigate enteric methane emission and can be practiced with ease by farmers under field conditions. These may include pasture improvement, feed processing, increasing concentrate in ration and strategic supplementation.
8.2.2.1 Pasture Improvement
Enteric methane emission is influenced by the type of forage the animal feeds upon. Animals grazing on mixed pastures, having leguminous forages, emit less methane than animals grazing on grass alone pastures. This effect can be explained by the presence of condensed tannins in legumes (Waghorn 2007).
There is also variation in enteric methane emission of ruminants grazing on tropical or temperate pastures. C4 grasses in tropical pastures can produce more methane per kg of dry matter ingested than temperate grasses with C3 photosynthesis. This is due to the poor digestibility of C4 grasses compared to C3 grasses (Archimède et al. 2011).
In temperate pastures itself, Boadi et al. (2002) reported less energy loss as methane in steers grazing on pastures during the early period of the grazing season than the mid- and late-grazing period. Pastures rich in grasses with high sugar content are suitable for animal grazing for a simple reason that high sugar-rich grasses promote the growth of rumen microbes resulting in the increased synthesis of microbial protein and decreasing the production of methane. Hence improving pasture quality is another important way by which enteric methane emission can be reduced.
8.2.2.2 Feed Processing
The processing of feeds especially roughages can improve the feeding value by increasing its digestibility and/or by increasing feed intake. Any attempt to increase feed digestibility/intake has potential to reduce enteric methane emission. Improving the nutritive value of low-quality forages/feeds that are fed to ruminants could increase their productivity and consequently reduce methane emissions.
Feed processing techniques may include chaffing and grinding of straws, alkali/ammonia treatment of straws and feed residues and urea-molasses blocks. These processing techniques are reported to check the methane emission from rumen by 10%. The reduction of forage particle size has significant benefits like increasing feed intake and digestibility, increasing microbial activity due to increase in the surface area of the substrate and consequent improvement in the productivity of livestock (Gerber et al. 2013). Ammoniation or protein supplementation of low-quality forages or crop residues increased methane losses in proportion to digestibility; however methane production per unit of product produced was reduced (Johnson and Johnson 1995). Beauchemin et al. (2008), had stated that methanogenesis was lower on feeding silage compared to feeding hay and was also lower in finely ground feed or pellets compared to roughly processed feed.
8.2.2.3 Increasing Concentrate in Ration
Altering the roughage concentrate ratio of ruminant ration by increasing concentrates at the expense of roughage causes reduction in methane emission. Methane production can be reduced to 3% from 6.5% by feeding ruminants more concentrates (Beauchemin and McGinn 2005).
Higher concentrate feeding decreases fibre (cellulose and hemicellulose) and increases starch which results in significant modifications in both the physiochemical conditions in the rumen and microbial populations. Increase of amylolytic bacteria results in a change in short chain fatty acid production, promoting a proportional increase of propionate and a reduction of acetate which reduces the hydrogen availability in the rumen, causing reduced methane emission (Pereira et al. 2015). Beauchemin and McGinn (2005) reported that beef cattle, fed diets containing mainly corn grain produce about 30% less methane than when fed diets containing mainly barley grain. Methane emission can be reduced by enhancing the nutritive value and digestibility of feed by balancing the ration with addition of concentrate for grazing animals.
This strategy of increasing concentrate in ration to reduce methane emission has its own limitations. In high concentrate rations, increased production of lactic acid could trigger ruminal acidosis, reduce milk fat and shorten the productive life span of animals. Moreover, its economic viability is also questionable. Increasing the concentrate proportion will have negative impact on fibre digestibility which could cause potential loss in production and will also result in increased concentration of fermentable organic matter in manure and is likely to increase methane emissions from stored manure (Lee et al. 2012).
8.2.2.4 Strategic Supplementation
In tropical countries, where ruminant livestock are grazed in wastelands or on very poor-quality pastures or fed with poor-quality agricultural waste, the digestibility of the diet is low and the nutritional requirement of the animals is not met. In this context protein supplementation in the diets increases the nutrient digestibility and significantly decreases methane production in the rumen (Mehra et al. 2006). Balanced diet with inclusion of protein supplements when fed to the lactating cows and buffaloes caused a marked reduction in the methane emission and an observable increase in both the production of milk and fat content in milk (Kannan and Garg 2009). Similar findings were observed when lactating crossbred cows are fed with balanced diet with respect to calcium, phosphorus and protein (Kannan et al. 2011).
8.2.3 Rumen Manipulation
Enteric methane emission from ruminant livestock can be reduced by various approaches that cause rumen manipulation viz., supplementation of bacteriocins, ionophores, fats, oils (Beauchemin et al. 2008), organic acids (Newbold et al. 2005; Wallace et al. 2006), probiotics, prebiotics, sulphate, halogenated methane analogues, nitroxy compounds (Morgavi et al. 2010), fungal metabolites and secondary plant metabolites (Patra et al. 2006; Wallace et al. 2002; Calsamiglia et al. 2007), microalgae, exogenous enzymes, etc.
8.2.3.1 Bacteriocins
Bacteriocins are naturally occurring bacterial products with a bactericidal activity. They are effective as they directly inhibit methanogens and redirect hydrogen to other reductive rumen bacteria such as propionate producer or acetogens. Callaway et al. (1997) have shown that nisin, a food additive, reduces methanogenesis by 36%. However, the organisms developed resistance quickly. This problem can be overcome by the use of bacteriocin of rumen origin. Lee et al. (2002) found that semi-purified bacteriocin inhibits 50% of methane production in vitro and methanogens did not show any adaptation to these bacteriocins.
8.2.3.2 Ionophores
Ionophores are generally used as feed additives in order to improve the efficiency of digestion in ruminants. Ionophores include tetronasin, monensin, lasalocid, salinomycin, narasin, lysocellin, etc. These ionophore antibiotics are carboxylic polyether compounds produced by various strains of Streptomyces, e.g. monensin by S. cinnamonensis and lasalocid by S. lasoliensis. Monensin is moderately active against gram-positive bacteria, certain mycobacteria and coccidia, while lasalocid is specifically active against hydrogen producing bacteria and results in higher propionate production which in turn is related to low methane production (Kobayashi et al. 1992).
Ionophores increase the proportion of gram-positive bacteria in the rumen, resulting in a shift in fermentation acids from acetate and butyrate to propionate, and hence decrease the methane production. Several studies were conducted to know the efficacy of monensin in rumen modification. Few studies found the positive mitigating potential of monensin in enteric methane emission. Nevertheless, the ionophore effects on enteric methane emission are ephemeral as the microbes adapt themselves (Kataria 2015).
8.2.3.3 Fats/Oils
Adding fats to the diet can reduce methane emission by lowering ruminal fermentability and also through hydrogenation of unsaturated fats (Johnson and Johnson 1995). As methane is produced in the rumen to act as a hydrogen sink during the fermentation of carbohydrate, polyunsaturated fatty acids (PUFA) having double or triple bonds have potential to be used as hydrogen sinks, because these bonds will get saturated by hydrogen and less hydrogen will be available for methane production. Adding 4.6% canola oil, a source of unsaturated fat, to a high-forage diet was an effective suppressant of methane, with daily methane emissions decreasing by 32% and methane emissions as a percentage of GE intake decreasing by 21% (Beauchemin and McGinn 2006).
Fat feeding has also been shown to reduce methane through a reduction in protozoal numbers. Methanogenic bacteria are metabolically associated with ciliate protozoa (Newbold et al. 1995), and feeding oil can cause substantial decreases in protozoan populations (Ivan et al. 2004). Rumen protozoans were found to be responsible for higher hydrogen production and also acting as a habitat for the methanogens present in the rumen (Newbold et al. 1995). In defaunated ruminants, the methanogenic bacteria do not get the symbiotic partner and methane synthesis is partially inhibited. On defaunation, the methane production is reduced by 20–50% (Van Nevel and Demeyer 1996) depending on the various factors in the diet of the animal.
8.2.3.4 Organic Acids
Within the rumen, methane represents a terminal hydrogen sink. Propionate production represents an alternative hydrogen sink in normal rumen fermentation, provided sufficient precursors are available. The precursors to propionate production are “pyruvate”, “oxaloacetate”, “malate”, “fumarate” and “succinate”. The propionate can also be produced from pyruvate through an important pathway called acrylate pathway. Organic acids mentioned above have potential to jettison reducing power and thereby methane production can be reduced. Dietary supplementation of dicarboxylic organic acids such as malate, fumarate, aspartate etc. reduces methane production (Martin 1998). These organic acids are converted to succinate or propionate by reduction process, and less hydrogen will be available for methane production.
Addition of fumaric acid decreased methane emissions in vitro (Asanuma et al. 1999) and in vivo (Bayaru et al. 2001) but not in all studies (McGinn et al. 2004), possibly because of the level of supplementation used. Malate, a potent methane inhibitor, is present in animal feeds like alfalfa (2.9–7.5% of DM) and Bermuda grass (1.9–4.5%), but its level varies with variety and stage of maturity. In vitro, continuous rumen culture experiments have shown that adding graded levels of fumarate to fermentations of rye grass pasture substrate linearly depressed methane production while linearly increased propionate production (Kolver et al. 2004). The long-term effects of organic acids on methane mitigation have not been demonstrated. Moreover, the high price of organic acids makes its use uneconomical.
8.2.3.5 Probiotics
Probiotics such as yeast cultures are used to stimulate bacterial activity in the rumen. The probiotics have been shown to stabilize the rumen pH, increase propionate levels and decrease the amount of acetate, methane and ammonia production. Probiotics cause the redirection of hydrogen ions away from methanogenesis and also decrease production of hydrogen during feed fermentation. Addition of Saccharomyces cerevisiae reduced methane production in vitro (Mutsvangwa et al. 1992). Lactic acid utilizing bacteria like Megasphaera elsdenii and Propionibacterium spp. and yeast like S. cerevisiae have major effects on methanogenesis by decreasing methane (Seo et al. 2010). Chaucheyras et al. (2012) suggested that live yeast cells can stimulate the use of hydrogen by acetogenic strains of ruminal bacteria thereby enhancing the formation of acetate and decreasing the formation of methane.
8.2.3.6 Prebiotics
Prebiotic compounds such as mannan-oligosaccharide (MOS), fructo-oligosaccharide (FOS), galacto-oligosaccharide (Mwenya et al. 2004) enhances propionate production by stimulating Selenomonas, Succinomonas and Megasphera with inhibition of acetate producers such as Ruminococcus and Butyrivibrio. The administration of galacto-oligosaccharides has brought about reduction of methane production up to 11%.
8.2.3.7 Sulphate
Sulphate reducing bacteria have the highest affinity to utilize hydrogen in the rumen, even better than methanogens, but the availability of sulphate in the rumen appears to be a limitation. Hegarty (1999) showed that to reduce methane emissions by 50%, about 0.75 moles of sulphate ingestion per day is required. Kamra et al. (2004) observed that sulphate supplementation helps in increasing the production of fibre degrading enzymes and fibre degradation in the rumen. Therefore, for a high fibre ration, sulphate/sulphite supplementation improves fibre degradability and inhibits methanogensis. However, a proper dose needs to be optimized, as toxic levels of sulphide can be generated on sulphate reduction.
8.2.3.8 Halogenated Methane Analogues
Various halogenated methane analogues have been tried as methane inhibitors. They include carbon tetrachloride, chloral hydrate, trichloroacetamide, DDT, trichloroacetaldehyde, bromochloromethane, chloroform, methylene chloride, methylene bromide, nitrapyrin, hemiacetal of chloral and starch (Haque 2001). Favourable effects on supplementation of these have been reported only in those animals fed on high roughage diets. Bromochloromethane inhibits methane production by reacting with reduced form of vitamin B12 which inhibits methanogenesis.
8.2.3.9 Nitrooxy Compounds
The “3-nitrooxypropanol (NOP)” and “ethyl-3-NOP” were found to possess an important anti-methanogenic property which indeed aids in reducing methane production. In fact, “3-nitrooxypropanol (NOP)” through interference with a co-enzyme (methyl-coenzyme M reductase) of methanogens checks the methanogen growth and thereby reduces methane production. Also, 3-NOP has inhibitory effects against methanogenic archaea without inducing any effects on growth of non-methanogenic bacteria in the rumen (Duin et al. 2016).
8.2.3.10 Fungal Metabolites
Lovastatin which is a secondary fungal metabolite is found to limit the activity of “3-hydroxy-3-methyl glutaryl coenzyme A reductase”. HMG-CoA reductase is an important enzyme involved in the biosynthesis of cholesterol (Jahromi et al. 2013). Rice straw fermented with Aspergillus terreus was found to possess lovastatin which was observed to reduce the population of methanogens and production of methane. Saprophytic fungal strains Mortierella wolfii were found to be a suitable candidate for the inhibition of the methanogenesis process (Cosgrove et al. 2012). The fungal metabolites, viz. “pravastatin” and “mevastatin”, were observed for reducing the ratio of ruminal acetate to propionate and also reducing the methanogen population (Morgavi et al. 2013).
8.2.3.11 Secondary Plant Metabolites
Plants produce chemicals collectively called as secondary plant metabolites that help in the self-defence of the plant species concerned. Most of these secondary plant metabolites have anti-microbial and anti-methanogenic activity. There are numerous plant extracts each with varying effects on ruminal fermentation and feed digestion (Kamra et al. 2005).
Garlic oil extracted from Allium sativum is a complex mixture of many secondary plant products including allicin, diallyl sulfide, diallyl disulfide and allyl mercaptan and a decrease in methane production was observed on supplementing garlic oil (Busquet et al. 2005). Ethanol and water extracts of the bulb of Allium sativum (garlic) reduced methane production significantly in the rumen liquor of buffaloes (Patra et al. 2006).
Methanol extract of seed pulp of Terminalia chebula (kadukka) reduced methane production significantly in the rumen liquor of buffaloes (Patra et al. 2006). The presence of tannins in Terminalia chebula might be responsible for reduction in methane emission. Phenolic acids such as p-coumaric acids, ferulic acids, cinnamic acids and phloretic acids and some monomeric phenolics have been found to decrease methane, acetate and propionate production (Asiegbu et al. 1995). Tannin, an important secondary plant metabolite, was found to be toxic against rumen organisms like protozoans, methanogens and other microbes that degrade the fibrous material. There are reports indicating a decrease in methane emission with dietary addition of condensed tannins or inclusion of condensed tannin containing forage (Carulla et al. 2005). Negative effects on ruminal fibre digestion, which may relate to decreased number of cellulolytic bacteria, formation of condensed tannin complexes that are resistant to enzymatic digestion and impaired substrate adhesion by fibrinolytic microbes, would reduce hydrogen availability to lessen methanogenesis (Carulla et al. 2005). Patra et al. (2006) showed that adding water extracts of neem seeds decreased total rumen volatile fatty acid (VFA) digestibility and also reported antiprotozoal activity.
Essential oils beneficially affect ruminal fermentation, causing an increase in VFA (Castillejos et al. 2005) and decrease in the rate of amino acid deamination (McIntosh et al. 2003). The antimicrobial activity of essential oils and secondary plant metabolites is highly specific, which raises the possibility that these compounds can be used to target methanogens. McIntosh et al. (2003) reported that growth of the methanogen Methanobrevibacter smithii was inhibited when the concentration of essential oil in supplemental product exceeded 1000 ppm.
Saponins have been found to have anti-protozoal and anti-methanogenic activities (Sliwinski et al. 2002). There are plants, which contain saponins such as Alfalfa (3–5%), Sapindus rarak, Sapindus mokorossi, Yucca schidigera (4%), Quillaja saponaria (10%), etc. that cause a decrease in methane production by 20–60%. Saponins reduce the protozoal population which reduces the inter species hydrogen transfer to the methanogenic bacteria attached to the protozoa, thereby decreases the hydrogen availability to the methanogens.
8.2.3.12 Microalgae
Asparagopsis, a genus of microalgae, even at 2% of total substrate organic matter, was reported to reduce methane production by 99% in, in vitro condition. Similarly, Oedogonium, a member of filamentous algae, was found to decrease methane production (Machado et al. 2014). Further, Cystoseira trinodis and Dictyota bartayresii, members of brown algae, are found under in vitro conditions to inhibit methane production (Dubois et al. 2013).
8.2.3.13 Exogenous Enzymes
The role of exogenous enzymes in mitigating enteric methane emission has been reviewed with contradictory results. Exogenous enzymes increase fibre digestibility, improves feed efficiency and decreases fermentable organic matter in manure, thus reducing overall methane emission from ruminant production system. However, Chung et al. (2012), reported increased enteric methane emission per unit dry matter intake or milk yield by 10–11% on exogenous enzyme supplementation with endoglucanase and xylanase activity.
8.2.3.14 Defaunation
Ten per cent decrease in enteric methane emission has been reported on defaunation. However, defaunation may have a negative impact on feed digestibility, animal productivity and milk fat content. Practically, it is not possible to maintain defaunated livestock as possibility of the animals getting refaunated always exists.
8.2.4 Advanced Strategies
Advanced strategies can be adopted in technologically advanced livestock farms to mitigate enteric methane emission. Some of the advanced strategies are precision feeding, vaccines that target methanogens, genetic transformation of rumen bacteria and transferring rumen microbiome of low methane emitting ruminants.
8.2.4.1 Precision Feeding
Precision feeding is feeding the livestock with the right feed at the right time, so that it satisfies its nutritional requirement at that point of time. Precision feeding combines the genetics of the animal with feed management. Precision feeding will improve feed digestibility, feed efficiency and livestock productivity and hence indirectly reduce enteric methane emission. Precision feeding can be adopted only in high value livestock farms that are highly advanced technologically.
8.2.4.2 Vaccines that Target Methanogens
Vaccines have been developed that specifically target methanogens in the rumen. There are immunogenic fractions on the surface of methanogens and antibodies developed against these could curtail methanogenic activity in ruminants (Wedlock et al. 2013). However, the majority of the vaccines have failed due to poor efficacy and low cross-reactivity. The reverse vaccinology technology has provided solution for identifying conserved vaccines. Moreover sequencing the genome of Methanobacterium ruminantium has provided data for identifying conserved vaccine targets.
8.2.4.3 Genetic Transformation of Rumen Bacteria
Genetic modification of rumen microorganisms is aimed to alter their fermentation characteristics. Extensive characterization of the molecular genetics of rumen bacteria with high precision is needed for the success of this genetic modification. Even then the viability of the altered organisms in the natural environment is doubtful. Moreover, the product obtained/organism used has to be approved by both national and international regulatory standards.
8.2.4.4 Transferring Rumen Microbiome of Low Methane Emitting Ruminants
Another possibility of mitigating enteric methane emission is through transferring the microbiome of low methane emitters to the rumen of high methane producing ruminants. Such intervention when made in the early stage of life of the calf may have greater stability. Better understanding of host microbiome relations is needed for the success of such a venture.
8.2.4.5 Farmer-Friendly Approaches to Mitigate Enteric Methane Emission
As discussed earlier in this chapter, feeding strategies such as pasture improvement, feed processing, increasing concentrate in ration and strategic supplementation are practical approaches to mitigate enteric methane emission. Farmers can be advised to chaff the forage before feeding it to their livestock. Chaff cutters are available with the farmers, or farmers can purchase them at subsidized cost. Increasing concentrate in ration and strategic supplementation can be achieved through ration balancing. Ration balancing is considered as the practical approach to mitigate enteric methane in the Indian context.
Indian livestock are grazed on very poor-quality tropical pastures or fed on crop residues having a very low nutritive value. Hence the productivity of animals is far below their genetic potential. Improving per animal productivity is a potent tool in reducing enteric methane emission per unit of product produced. Feeding balanced diets reduced enteric methane emission in terms of g/d and g/kg milk yield by 10.1 and 13.5% respectively, which further reduced the part of dietary gross energy loss as methane by 10.3%. Intestinal flow of microbial nitrogen increased by 51.4 g/d whereas, faecal nitrogen excretion reduced by 19.4% (Garg et al. 2013). Ration balancing shifts the rumen fermentation more towards propionate production and microbial protein production; thus enteric methane emission is reduced. Rumen fermentation efficiency can be improved through critical nutrient supplementation.
8.3 Conclusion
Mitigating enteric methane emissions in ruminants will assist not only in the achievement of international commitments that benefit climate change but also in the improvement of energy utilization efficiency and the performance of livestock. Increasing livestock productivity across production systems increases food security and improves livelihood of farmers. In the perspective of GHG emission management, enteric methane emission reduction through significant gain in productivity is construed as a “lowest-cost option”, and it has positive economic repercussions to the farmers dependent on livestock (Gerber et al. 2013). A country like India cannot afford the energy loss as methane in livestock, as it demands additional feed resources to compensate the loss (Malik et al. 2016).
Livestock farming in India is a low productivity venture. But the demand for livestock products is ever rising. In order to meet the consumer demand for livestock products, farmers rear a large number of livestock, larger than the number that would be economically viable with the available feed resources. More pressure is thus created on availability of feed, and livestock productivity declines further. In this context any increase in productivity resulting in an increased livestock product will lead to increase in employment in livestock enterprises for marketing and distribution of livestock products.
References
Archimede H, Eugène M, Marie-magdeleine C, Boval M, Martin C, Morgavi D, Lecomte P, Doreau M (2011) Comparison of methane production between C3 and C4 grasses and legumes. Fuel Energy Abstr 166:59–64
Asanuma N, Iwamoto M, Hino T (1999) Effect of the addition of fumarate on methane production by ruminal microorganism in vitro. J Dairy Sci 82:780–787
Asiegbu FO, Paterson A, Morrison IM, Smith JE (1995) Effect of cell wall phenolics and fungal metabolites on methane and acetate production under in vitro conditions. J Gen Appl Microbiol 41:475–485
Bayaru E, Kanda S, Kamada T, Itabashi H, Andoh S, Nishida T, Ishida M, Itoh T, Nagara K, Isobe Y (2001) Effect of fumaric acid on methane production, rumen fermentation and digestibility of cattle fed roughage alone. J Anim Sci 72:139–146
Beauchemin KA, McGinn SM (2005) Methane emissions from feedlot cattle fed barley or corn diet. J Anim Sci 83:6
Beauchemin KA, McGinn SM (2006) Methane emissions from beef cattle: effects of fumaric acid, essential oil, and canola oil. J Anim Sci 84:1489–1496
Beauchemin KA, Kreuzer M, O’Mara F, McAllister TA (2008) Nutritional management for enteric methane abatement: a review. Aust J Exp Agric 48:21–27
Bhata R, Malik PK, Kolte AP, and Gupta R (2016) Annual progress report on outreach project on methane. NIANP, Bangalore
Boadi D, Wittenberg KM, McCaughey WP (2002) Effects of grain supplementation on methane production of grazing steers using the sulphur (SF6) tracer gas technique. Can J Anim Sci 82:151–157
Busquet M, Calsamiglia S, Ferret A, Carro MD, Kamel C (2005) Effect of garlic oil and four of its compounds on rumen microbial fermentation. J Dairy Sci 88:4393–4404
Callaway TR, Carneiro De Melo AMS, Russell JB (1997) The effect of nisin and monensin on ruminal fermentations in vitro. Curr Microbiol 35:90–96
Calsamiglia M, Busquet P, Cardozo W, Castillejos L, Ferret A (2007) Invited review: essential oils as modifiers of rumen microbial fermentation. J Dairy Sci 90:2580–2595
Carulla JE, Kreuzer M, Machmuller A, Hess HD (2005) Supplementation of Acacia mearnsii tannins decreases methanogenesis and urinary nitrogen in forage-fed sheep. Aust J Agric Res 56:961–970
Castillejos L, Calsamiglia S, Ferret A, Losa R (2005) Effects of a specific blend of essential oil compounds and the type of diet on rumen microbial fermentation and nutrient flow from a continuous culture system. Anim Feed Sci Technol 119:29–41
Chaucheyras-Durand F, Chevaux E, Martin C, Forano E (2012) Use of yeast probiotics in ruminants: effects and mechanisms of action on rumen pH, fibre degradation, and microbiota according to the diet. Chapter 7. In: Rigobelo EC (ed) Probiotic in animals. Intech, Rijeka. https://doi.org/10.5772/50192:119-152
Chung YH, Zhou M, Holtshausen L, Alexander TW, McAllister TA (2012) A fibrolytic enzyme additive for lactating Holstein cow diets: ruminal fermentation, rumen microbial populations, and enteric methane emissions. J Dairy Sci 95:1419–1427
Ciais P, Sabine C, Bala G et al (2013) Carbon and other biogeochemical cycles. In: Stocker TF, Qin D, Plattner G-K, Tignor M, Allen SK, Boschung J, Nauels A, Xia Y, Bex V, Midgley PM (eds) Climate change 2013: The physical science basis. Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change. Cambridge University Press, Cambridge/New York
Cosgrove GP, Muetzel S, Skipp R, Mace WJ (2012) Effects of endophytic and saprophytic fungi on in vitro methanogenesis. N Z J Agric Res 55:293–307
Dubois B, Tomkins NW, Kinley RD, Bai M, Seymour S, Paul NA (2013) Effect of tropical algae additives on rumen in vitro gas production and fermentation characteristics. Am J Plant Sci 4:34–43
Duin EC, Wagner T, Shima S, Prakash D, Cronin B, Yanez-Ruiz DR, Duval S, Rumbeli R, Stemmler RT, Thauer RK, Kindermann M (2016) Mode of action uncovered for the specific reduction of methane emissions from ruminants by the small molecule 3-nitrooxypropanol. Proc Natl Acad Sci 113:6172–6177
FAO (2017) Reducing Enteric Methane for improving food security and livelihoods. www.fao.org/in-action/enteric-methane/background/why-is-enteric-methane.../en/
Garg MR, Sherasia PL, Phondba BT, Sheike SK, Patel CT (2013) Effect of feeding balanced ration on milk production enteric methane emission and metabolic profile in cross bred cows under field conditions. Indian J Dairy Sci 66:113–119
Gerber PJ, Steinfeld H, Henderson B, Mottet A, Opio C, Dijkman J, Falcucci A, Tempio G (2013) Tackling climate change through livestock–A global assessment of emissions and mitigation opportunities. Food and Agriculture Organization of the United Nations (FAO), Rome
Haque N (2001) Environmental implication of methane production: diet and rumen ecology. Short course, CAS in Animal Nutrition, IVRI, Izatnagar
Hegarty RS (1999) Mechanisms for competitively reducing ruminal methanogenesis. Aust J Agric Res 50:1299–1305
Ivan M, Koenig KM, Teferedegne B, Newbold CJ, Entz T, Rode LM, Ibrahim M (2004) Effects of the dietary Enterolobium cyclocarpum foliage on the population dynamics of rumen ciliate protozoa in sheep. Small Rumin Res 52(1–2):81–91
Jahromi FM, Liang JB, Ho YW, Mohamad R, Goh YM, Shokryazdan P (2013) Lovastatin in Aspergillus terreus: fermented rice straw extracts interferes with methane production and gene expression in Methanobrevibacter smithii. Biomed Res Int
Johnson KA, Johnson DE (1995) Methane emissions from cattle. J Anim Sci 73:2483–2492
Kamra DN, Agarwal N, Yadav MP (2004) Methanogenesis in the rumen and the Greenhouse effect on the environment. Livest Int 8(9):2–8
Kamra DN, Agarwal N, Chaudhary LC (2005) Inhibition of ruminal methanogenesis by tropical plants containing secondary plant compounds. In: Soliva CR, Takahash J, Kreuzer M (eds) Proceeding of the 2nd international conference of greenhouse gases and animal agriculture. ETH Zurich, Zurich, pp 102–111
Kannan A, Garg MR (2009) Effect of ration balancing on methane emission reduction in lactating animals under field conditions. Indian J Dairy Sci 62:292–296
Kannan A, Garg MR, Mahesh Kumar BV (2011) Effect of ration balancing on milk production, microbial protein synthesis and methane emission in crossbred cows under field conditions in Chittoor district of Andhra Pradesh. Indian J Anim Nutr 28:117–132
Kataria RP (2015) Use of feed additives for reducing green house gas emissions from dairy farms. Microbiol Res 6:6120
Kobayashi Y, Wakita M, Hoshino S (1992) Effects of ionophore salinomycin on nitrogen and long-chain fatty acid profiles of digest in the rumen and the duodenum of sheep. Anim Feed Sci Technol 36:67–76
Kolver ES, Aspin PW, Jarvis GN, Elborough KM, Roche JR (2004) Fumarate reduces methane production from pasture fermented in continuous culture. Proc N Z Soc Anim Prod 64:155–159
Lee SS, Hsu JT, Mantovani HC, Russell JB (2002) The effect of bovicin HC5, a bacteriocin from Streptococcus bovis HC5, on ruminal methane production in vitro. FEMS Microbiol Lett 217:51–55
Lee C, Hristov AN, Dell CJ, Feyereisen GW, Kaye J, Beegle D (2012) Effect of dietary protein concentration on ammonia and greenhouse gas emissions from dairy manure. J Dairy Sci 95:1930–1941
Lipper L, Thornton P, Campbell B et al (2014) Climate-smart agriculture for food security. Nat Clim Chang 4:1068–1072. https://doi.org/10.1038/nclimate2437
Machado L, Magnusson M, Paul NA, De Nys R, Tomkins N (2014) Effects of marine and freshwater macroalgae on in vitro total gas and methane production. PLoS One 9:e85289. https://doi.org/10.1371/journal.pone.0085289
Malik PK, Bhatta R, Soren NM, Sejian V, Mech A, Prasad KS, Prasad CS (2015) Feed based approaches in enteric Methane Amelioration. livestock production and climate change. CAB International, Wallingford, pp 336–359
Malik PK, Kolte AP, Dhali A, Seijan V, Thirumalisamy G, Gupta R, Bhatta R (2016) GHG emissions from livestock challenges and ameliorative measures to counter adversity. Intech Open. https://doi.org/10.5772/6488J
Martin SA (1998) Manipulation of ruminal fermentation with organic acids: a review. J Anim Sci 76:3123–3132
McAllister A, Newbold CJ (2008) Redirecting rumen fermentation to reduce methanogenesis. Aust J Exp Agric 48:7–13
McGinn SM, Beauchemin KA, Coates T, Colombatto D (2004) Methane emissions from beef cattle: effects of monensin, sunflower oil, enzymes, yeast, and fumaric acid. J Anim Sci 82:3346–3356
McIntosh FM, Williams P, Losa R, Wallace RJ, Beever DA, Newbold CJ (2003) Effects of essential oils on ruminal microorganisms and their protein metabolism. Appl Environ Microbiol 69:5011–5014
Mehra UR, Khan MY, Lal M, Hasan QZ, Das A, Har R, Verma AK, Dass RS, Singh P (2006) Effect of sources of supplementary protein on intake, digestion and efficiency of energy utilization in buffaloes fed wheat straw based diets. Asian-Aust J Anim Sci 19(5):638–644
Merino P, Ramirez-Fanlo E, Arriaga H, Del Hierro O, Artetxe A (2011) Regional inventory of methane and nitrous oxide emission from ruminant livestock in the Basque country. Anim Feed Sci Technol 166–167:628–640
Morgavi DP, Forano E, Martin C, Newbold CJ (2010) Microbial ecosystem and methanogenesis in ruminants. Animal 4:1024–1036
Morgavi DP, Martin C, Boudra H (2013) Fungal Secondary metabolites from Monascus spp reduce rumen methane production in vitro and in vivo. J Anim Sci 91:848–860
Mutsvangwa T, Edward IE, Topp JH, Peterson GF (1992) Anim Prod 55:35–40
Mwenya B, Santanso B, Sar C, Gano Y, Kobayashi T (2004) Effects of including beta 1-4 galacto oligosacharides, lactic acid bacteria or yeast culture on methanogenesis as well as energy and nitrogen metabolism in sheep. Anim Feed Sci Technol 118:19–20
Newbold CJ, Lassalas B, Jouany JP (1995) The importance of methanogens associated with ciliate protozoa in ruminal methane production in vitro. Lett Appl Microbiol 21:230–234
Newbold CJ, López S, Nelson N, Ouda JO, Wallace RJ (2005) Propionate precursors and other metabolic intermediates as possible alternative electron acceptors to methanogenesis in ruminal fermentation in vitro. Br J Nutr 94(1):27–35
Patra AK (2014) Trends and projected estimates of GHG emissions from Indian livestock in comparison with GHG emissions from world and developing countries. Asian Australas J Anim Sci 27(4):592–599
Patra AK, Kamra D, Agarwal N (2006) Effect of plant extract on in vitro methanogenesis, enzyme activities and fermentation of feed in rumen liquor of buffalo. Anim Feed Sci Technol 128:276–291
Pinares-Patiño CS, D’Hour P, Jovana JP, Martin C (2007) Effect of stocking rate on methane and carbon dioxide emissions from grazing cattle. Agric Ecosyst Environ 121:30–46
Priano ME, Fuse VS, Ger JI, Berkovic AM, Williams KE (2014) Strong differences in the CH4 emission from feces of grazing steers submitted to different feeding schedules. Anim Feed Sci Technol 194:145–150
Ribeiro Pereira LG, Machado FS, Campos MM, Guimaraes Júnior R, Tomich TR, Reis LG, Coombs C (2015) Enteric methane mitigation strategies in ruminants: a review. Revista Colombiana de Ciencias Pecuarias 28(2):124–143
Robertson LJ, Waghorn GC (2002) Dairy industry perspectives on methane emissions and production from cattle fed pasture or total mixed rations in New Zealand. Proc N Z Soc Anim Prod 62:213–218
Saggar S, Bolan NS, Bhandral R, Hedley CB, Luo J (2004) A review of emissions of methane, ammonia, and nitrous oxide from animal excreta deposition and farm effluent application in grazed pastures. N Z J Agric Res 47(4):513–544
Seo JK, Kim SW, Kim MH, Upadhaya SD, Kam DK (2010) Direct fed microbials for ruminant animals. Asian-Australas J Anim Sci 23(12):1657–1667
Sliwinski BJ, Soliva CR, Machmuller A, Kreuzer M (2002) Efficacy of plant extracts rich in secondary constituents to modify rumen fermentation. Anim Feed Sci Technol 101:101–114
Smith P, Bustamante M, Ahammad H et al (2014) Agriculture, forestry and other land use (AFOLU). In: Edenhofer O, Pichs-Madruga R, Sokona Y, Farahani E, Kadner S, Seyboth K, Adler A, Baum I, Brunner S, Eickemeier P, Kriemann B, Savolainen J, Schlömer S, von Stechow C, Zwickel T, Minx JC (eds) Climate change 2014: Mitigation of climate change. Contribution of working group III to the fifth assessment report of the intergovernmental panel on climate change. Cambridge University Press, Cambridge/New York
Tarawali S, Herrero M, Descheemaeker K, Grings E, Blümmel M (2011) Pathways for sustainable development of mixed crop livestock systems: taking a livestock and pro-poor approach. Livest Sci 139:11–21
Van Nevel CJ, Demeyer DI (1996) Control of rumen methanogenesis. Environ Monit Assess 42:73–97
Venkatramanan V, Shah S (2019) Climate smart agriculture technologies for environmental management: the intersection of sustainability, resilience, wellbeing and development. In: Shah S et al (eds) Sustainable green technologies for environmental management. Springer Nature Singapore Pte Ltd., Singapore, pp 29–51. https://doi.org/10.1007/978-981-13-2772-8_2
Waghorn GC (2007) Beneficial and detrimental effects of dietary condensed tannins for sustainable sheep and goat production: progress and challenges. Anim Feed Sci Technol 147:116–139
Wallace RJ, McEwan NR, McIntosh FM, Teferedegne B, Newbold CJ (2002) Natural products as manipulators of rumen fermentation. Asian-Australas J Anim Sci 10:1458–1468
Wallace RJ, Wood TA, Rowe A, Price J, Yanez DR (2006) Encapsulated fumaric acid as a means of decreasing ruminal methane emissions. Int Congr Ser 1293:148–151
Wedlock DN, Janssen PH, Leahy SC, Shu D, Buddle BM (2013) Progress in the development of vaccines against rumen methanogens. Animal 7(S2):244–252
Wolin MJ (1974) Metabolic interactions among intestinal microorganisms. Am J Clin Nutr 27:1320–1328
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Valli, C. (2020). Mitigating Enteric Methane Emission from Livestock Through Farmer-Friendly Practices. In: Venkatramanan, V., Shah, S., Prasad, R. (eds) Global Climate Change and Environmental Policy. Springer, Singapore. https://doi.org/10.1007/978-981-13-9570-3_8
Download citation
DOI: https://doi.org/10.1007/978-981-13-9570-3_8
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-13-9569-7
Online ISBN: 978-981-13-9570-3
eBook Packages: Earth and Environmental ScienceEarth and Environmental Science (R0)