Abstract
The main objective of the present study was to evaluate the phytopathogen biomass of Phytophthora capsici and Colletotrichum gloeosporioides as inducers of antifungal metabolites from Trichoderma asperellum. The experiment was carried out through a solid-state fermentation using corncob as support/substrate. Water, ethanol, and toluene were used to recover the antifungal metabolites. The strain of P. capsici was inhibited by the metabolites recovered from all extracts; however C. gloeosporioides resist them and develop a normal growth. The bioactive extracts were fractioned using Amberlite XAD16®, and each fraction was analyzed by LC-ESI-MS. LC-ESI-MS analysis showed two major compounds; an unknown compound (1) was detected as [M + H]− (m/z 478) while dihydroxybergamotene (2) as [M + H]− (m/z 260). In addition, other four compounds were detected: viridepyronone (3), koninginin D (4), acetyltetrahydroxyanthraquinone (5), and virone or gliotoxin (6). The results suggest that the biomass of P. capsici worked as an inducer of antibiotic compounds in T. asperellum.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
Trichoderma is a prominent and well-studied biocontrol agent, due to its capabilities to control and kill several phytopathogen pests, such as Phytophthora tropicalis, Phytophthora palmivora, Alternaria solani, Bipolaris oryzae, Pyricularia oryzae, and Sclerotinia sclerotiorum, among others (Prabhakaran et al. 2015; Singh et al. 2016, 2017; Sriwati et al. 2015). The competence of space and nutrients, mycoparasitism, and the production of antibiotics are the main mechanisms of Trichoderma for the biocontrol of pests (Ghorbanpour et al. 2018; Jeleń et al. 2014; Keswani et al. 2014). The term antibiosis is related to secretion of chemicals with biological activity, such as cell wall-degrading enzymes, siderophores, chelating iron, and volatile and nonvolatile metabolites (El-Debaiky 2017; Mutawila et al. 2016; Keswani 2015). It is well known that Trichoderma has the ability to produce different secondary metabolites, such as alcohols, ketones, alkanes, furans, and mono- and sesquiterpenes, in order to inhibit the growth of phytopathogens (including fungi, bacteria, yeast) and also promote plant growth (Hu et al. 2017; Jeleń et al. 2014). The production and release of secondary metabolites from fungi are activated by the presence of an organism that represents a threat to its survival (Sun et al. 2016). The production of antibiotic compounds depends on several factors, such as fungal growth phase and nutritional, biological, and environmental conditions, and they could be induced or activated by other organisms when they are invading the space in the soil, competing for available nutrients, among others (Arinbasarova et al. 2017; Vinale et al. 2016). There are several reports focusing on the identification of antibiotic compounds produced by biological control agents (BCAs), particularly by Trichoderma spp. Most of them are reported compounds effective against various phytopathogens (Angel et al. 2016; Yamazaki et al. 2016). The majority of the reports about the production of antibiotic compounds are focused on the use of liquid cultures; however, filamentous fungi seem to be more adapted to solid environments (Shakeri and Foster 2007; Viniegra-González 2014). In literature it is possible to find a lot of information about solid-state fermentation and fungi in order to produce all kinds of enzymes, antioxidants, bioactive compounds, and biomass, among others (El-Gendy et al. 2017; Elegbede and Lateef 2017; Mohamed et al. 2016). Therefore, the objective of the present study was focused on the evaluation of the phytopathogen biomass of Phytophthora capsici and Colletotrichum gloeosporioides as inducers of antifungal metabolites from Trichoderma asperellum through solid-state fermentation conditions.
2 Materials and Methods
2.1 Chemicals and Reagents
HPLC grade acetonitrile and acetic acid were purchased from Sigma-Aldrich. Ultrapure water (Milli-Q) was generated by the Millipore System (Bedford, USA). SPE cartridges and Oasis MAX 96-well plate 30 lm (30 mg) were obtained from Waters (Milford, MA, USA).
2.2 Microorganism and Culture Conditions
The strains of Trichoderma asperellum and Phytophthora capsici were kindly proportioned by the Agricultural Parasitology Department of the UAAAN (Universidad Autónoma Agraria Antonio Narro, Saltillo, México). Colletotrichum gloeosporioides was proportioned by the Food Research Department of the UAdeC (Universidad Autónoma de Coahuila). Fungal strains were cultivated and preserved in a milk-glycerol 8.5% solution. Potato dextrose agar (PDA) was used to reactivate all fungi strains. The incubation was done at 28 °C during 5 days; then the preservation was at ±4 °C.
2.3 Phytopathogen Biomass Production
A cornmeal medium (17 g/L) was used to produce phytopathogen biomass. This medium was maintained under shaking for 1 h at 58 °C. Then, it was filtrated and sterilized (15 min at 115 °C). The inoculation of phytopathogens was as follows: C. gloeosporioides (1 × 106 spores/mL) and P. capsici (10 PDA plugs from a culture of 7 days old). The incubation was at 28 °C during 7 days under shaking (200 rpm).
2.4 Substrates
The corncob was proportioned by the Mexican Institute of Maize, UAAAN Coahuila, México. The material was dried, ground, fractioned (300–1680 μm), and stored under low moisture conditions for further evaluation. This material was used as a substrate on SSF without any pretreatment.
2.5 Culture Conditions
Polyethylene bags were used as a bioreactor in this study. The corncob (30 g) was mixed with phytopathogen biomass (3%). The biomass from each phytopathogen was evaluated in a separate experimental procedure. T. asperellum was inoculated at 1 × 107 spores g−1 of substrate adjusting the relative moisture at 50%. The fermentation was incubated for 5 days at 24 °C. Three different solid-state fermentations were done: (1) substrate mixed with biomass of C. gloeosporioides, (2) substrate mixed with biomass of P. capsici, and (3) substrate without phytopathogen biomass. Not fermented corncob was used as a control.
2.6 Samples Extraction
Water, ethanol, and toluene were the solvents evaluated to recover the metabolites released. The fermented material (20 gm) was eluted with 40 mL of each solvent using a plastic column (60 mL). Toluene extract was concentrated by evaporation at 35 °C, and then it was dissolved in 5 ml of ethanol (Vinale et al. 2009). All samples were passed through a Millipore® nylon membrane (0.45 μm) and then injected in a vial (2 mL).
2.7 Antifungal Assays
The crude extracts obtained in the last section were tested against C. gloeosporioides and P. capsici to evaluate their antibiotic properties. Pathogen plugs (5 mm diameter) from growing edges of colonies were placed at the center of Petri plates containing PDA (Vinale et al. 2006). The crude extract (10 μL) was applied on the top of each plug. Ethanol, toluene, and water were applied alone as a solvent control. The growth of the phytopathogens on PDA without application of solvent or extract was used as a control. The results were presented as a percentage of inhibition growth. A bifactorial arrangement (3 × 2) was made to the antifungal determination. The analysis of variance and means comparison (Tukey) were done in all cases. All treatments were done in triplicate.
2.8 Extracts Fractionation
Only the extracts (ethanolic and aqueous) from the fermentation supplemented with P. capsici biomass showed biological activity and therefore were fractioned. The ionic polymeric resin Amberlite XAD16® was used to perform the fractionation of the extracts. This resin was packed into a glass column (200 mL) and then it was filled with 40 mL of the extract. The aqueous extract was eluted first with distilled water (aqueous fraction); then, absolute ethanol was used to recover the compounds adsorbed in the resin (ethanolic fraction). The ethanolic extract was eluted first with absolute ethanol (ethanolic fraction); then, absolute methanol was used to recover the compounds adsorbed in the resin (methanolic fraction) (Ruiz-Martínez et al. 2011). At the end of the fractionation, nine samples were obtained.
2.9 LC-ESI-MS Analysis
The analyses by reversed-phase high-performance liquid chromatography were performed on a Varian HPLC system including an autosampler (Varian ProStar 410, USA), a ternary pump (Varian ProStar 230I, USA), and a PDA detector (Varian ProStar 330, USA). A liquid chromatograph ion trap mass spectrometer (Varian 500-MS IT Mass Spectrometer, USA) equipped with an electrospray ion source also was used. Samples (5 μL) were injected onto a Denali C18 column (150 mm × 2.1 mm, 3 μm, Grace, USA). The oven temperature was maintained at 30 °C. The eluents were formic acid (0.2%, v/v; solvent A) and acetonitrile (solvent B). The following gradient was applied: initial, 3% B; 0–5 min, 9% B linear; 5–15 min, 16% B linear; and 15–45 min, 50% B linear. The column was then washed and reconditioned. The flow rate was maintained at 0.2 mL/min, and elution was monitored at 245, 280, 320, and 550 nm. The whole effluent (0.2 mL/min) was injected into the source of the mass spectrometer, without splitting. All MS experiments were carried out in the negative mode [M-H]−. Nitrogen was used as nebulizing gas and helium as damping gas. The ion source parameters were spray voltage 5.0 kV; capillary voltage and temperature were 90.0 V and 350 °C, respectively. Data were collected and processed using MS Workstation software (V 6.9). Samples were firstly analyzed in full scan mode acquired in the m/z range 50–2000. MS/MS analyses were performed on a series of selected precursor ions.
3 Results
3.1 Antifungal Assays
The antifungal activity of the toluene, ethanol, and water extracts from the SSF by T. asperellum is presented in Table 6.1. The extracts obtained from the SSF added with P. capsici biomass showed important effects on the reduction of P. capsici growth rate compared with the control (no extract added). On the other hand, any extract obtained from the SSF added with C. gloeosporioides biomass showed important values of activity against the growth rate of C. gloeosporioides. The growth of phytopathogen strain P. capsici was reduced by the three extracts evaluated. The activity showed by toluene extract resulted in a very low value. However, the best results were shown by the ethanol and the water extract, with values of 9.6 and 6.1%, respectively.
3.2 HPLC Analyses
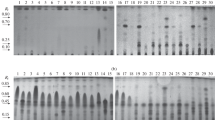
The three extracts with antifungal activities were analyzed by high-pressure liquid chromatography (HPLC), in order to identify important signals and its retention times. The water extract showed two signals differenced and more intense than the controls. The first peak was observed at 9.0 min and the second one at 18.9 min of retention time (RT) (Fig. 6.1). In the ethanolic extracts, it was possible to observe one signal in the chromatogram, different than the controls. This compound was observed with high intensity at 18.7 of RT (Fig. 6.2). The toluene extract showed exactly the same profile than the controls (Fig. 6.3).
3.3 LC-ESI-MS Analysis
The LC-ESI-MS analysis revealed the presence of six compounds from the extracts of the SSF by T. asperellum. The major compounds detected correspond to an unknown compound (1) and dihydroxybergamotene (2), with a molecular mass of [M + H]− (m/z 478) and [M + H]− (m/z 260), respectively. In addition, other four compounds were detected: viridepyronone (3), koninginin D (4), acetyltetrahydroxyanthraquinone (5), and virone or gliotoxin (6) (Table 6.2). All molecular masses obtained in the present study were compared with microbial bioactive metabolites reported in literature.
4 Discussion
In the present study, the induction of bioactive compounds through a solid-state fermentation by T. asperellum was observed. Several studies have been focused on the production of antifungal metabolites, such as Jeerapong et al. (2015), who reported the extraction of metabolites from a culture of T. harzianum F031 in a submerged fermentation (PDB) showing a reduction of 76.6% growth rate on C. gloeosporioides by an agar dilution assay. Also, several metabolites obtained from a liquid culture of T. harzianum have been reported as inhibitors of the growth rate of Fusarium oxysporum (Saravanakumar et al. 2016).
In the present study, dry biomass (evaluating P. capsici and C. gloeosporioides) was added in the fermentation process in order to evaluate a possible induction of specific antifungal compounds. A low percentage of inhibition was achieved by the extracts from the SSF with P. capsici biomass as inducer, and on the other hand, negligible activity was detected by the extracts from the SSF with C. gloeosporioides biomass as an inducer. It has been reported that the behavior of Trichoderma with other microorganisms who represent a competence in a microenvironment. Normally, several signals are sent and perceived between the microorganisms involved (Albuquerque and Casadevall 2012; Hogan 2006). The share of this information causes a response on each microorganism secreting enzymes or specific secondary metabolites. Low inhibition activity could be due to the use of inert phytopathogen biomass as inducer (no signaling). Therefore, these results suggest the necessity of Trichoderma to secrete its compounds as a reaction to the presence of a living organism, as it happened with the induction of several metabolites by the effect of a co-cultivation of Trichoderma sp. and Acinetobacter johnsonii (Zhang et al. 2017).
The fractionation of the crude extracts indicates the high polarity of the compounds released by Trichoderma and its potential role in phytopathogen inhibition. Metabolites 1 and 2 were detected in the chromatogram of the water extract showing a phytopathogen inhibition of 6.11%. However, in the ethanolic extract, only metabolite 2 was detected resulting in a phytopathogen inhibition of 9.6%. This result suggests that metabolite 2 could be more potent to act as antifungal compound working alone than in a synergy with compound 1.
Most of the bioactive metabolites from fungi reported in the literature have been obtained in liquid culture without any process of specific induction (Li et al. 2016). Those metabolites are commonly secreted by the fungal strain evaluated in each investigation. It is possible to find Trichoderma bioactive compounds with capacity to inhibit the growth of phytopathogenic microorganisms and also working as a plant growth promoting, such as harzianic acid (Vinale et al. 2013), azaphilone (Mevers et al. 2016), harzianolide (Mazzei et al. 2016), T39 butenolide (Keswani et al. 2017), and harzianopyridone (Ahluwalia et al. 2015), among others, mainly produced by using potato dextrose broth (PDB) (Saravanakumar et al. 2018). Several microorganisms have been reported as sensitive to the reduction of their growth, such as Gaeumannomyces graminis, Rhizoctonia solani, Pythium ultimum, P. irregular, and Sclerotinia sclerotiorum, due to the high antibiotic activity of the metabolites produced by Trichoderma spp. (Vinale et al. 2006, 2009). Frequently, the culture of T. harzianum in PDB is used by several authors to the production of bioactive metabolites (Shakeri and Foster 2007; Shentu et al. 2013).
Six metabolites obtained in the present study have been already reported by other authors, such as viridepyronone (3) reported by Evidente et al. (2003), showing antifungal inhibition against Sclerotium rolfsii. Koninginin D (4) was identified by (Kang et al. 2011), from the culture of T. koningii on malt extract agar (MEA), affecting the growth of several fungal phytopathogens (Zhou et al. 2014). The family of anthraquinones, particularly the acetyl-tetra-hydroxy-anthraquinone (5) shows a low effectG. graminis. However, good results are reported reducing the gray mold severity caused by B. cinerea (Vinale et al. 2008). Blight and Grove (1986) reported the production of virone (6) as the major metabolite by G. virens.
In the present study, it is reported the identification of a molecular mass (m/z 478) corresponding to a compound (1) not reported yet. The second compound denominated dihydroxybergamotene (2) was identified by Zhang et al. (2009), using a fungal strain of Acremonium sp. reporting anti-inflammatory as the main activity of this metabolite.
5 Conclusion
Six compounds were identified in the extracts obtained from a SSF with T. asperellum and the use of phytopathogen biomass as inducer under SSF culture conditions. Under the present conditions, two major compounds were detected, an unknown compound (1) and dihydroxybergamotene (2), respectively. Both compounds were obtained with water and ethanol suggesting the high polarity of them and the facilities to further extractions and applications. Biomass of C. gloeosporioides as inducer on the SSF did not show any effect on phytopathogen inhibition. Biomass of P. capsici as inducer on the SSF showed little effect on phytopathogen inhibition. The results suggest a further research focused on the possibilities to increase the quantity of phytopathogen biomass (more than 3%) expecting major induction of antifungal compounds and also the possibility to enhance the induction of metabolites evaluating co-culture conditions under SSF.
References
Ahluwalia V, Kumar J, Rana VS, Sati OP, Walia S (2015) Comparative evaluation of two Trichoderma harzianum strains for major secondary metabolite production and antifungal activity. Nat Prod Res 29(10):914–920
Albuquerque P, Casadevall A (2012) Quorum sensing in fungi–a review. Med Mycol 50(4):337–345
Angel LPL, Yusof MT, Ismail IS, Ping BTY, Mohamed Azni INA, Kamarudin NH, Sundram S (2016) An in vitro study of the antifungal activity of Trichoderma virens 7b and a profile of its non-polar antifungal components released against Ganoderma boninense. J Microbiol 54(11):732–744
Arinbasarova AY, Baskunov BP, Medentsev AG (2017) A low-molecular mass antimicrobial peptide from Trichoderma aureoviride Rifai VKM F-4268D. Microbiology 86(2):289–291
Betina V, Sedmera P, Vokoun J, Podojil M (1986) Anthraquinone pigments from a conidiating mutant of Trichoderma viride. Experientia 42(2):196–197
Blight MM, Grove JF (1986) Viridin. Part 8. Structures of the analogues virone and wortmannolone. J Chem Soc, Perkin Trans 1:1317–1322
El-Debaiky SA (2017) Antagonistic studies and hyphal interactions of the new antagonist Aspergillus piperis against some phytopathogenic fungi in vitro in comparison with Trichoderma harzianum. Microb Pathog 113:135–143
Elegbede JA, Lateef A (2017) Valorization of corn-cob by fungal isolates for production of xylanase in submerged and solid state fermentation media and potential biotechnological applications. Waste Biomass Valoriz 9:1–15
El-Gendy MMAA, SHM A-Z, El-Bondkly AMA (2017) Construction of potent recombinant strain through intergeneric protoplast fusion in endophytic fungi for anticancerous enzymes production using rice straw. Appl Biochem Biotechnol 183:1–21
Evidente A, Cabras A, Maddau L, Serra S, Andolfi A, Motta A (2003) Viridepyronone, a new antifungal 6-substituted 2H-Pyran-2-one produced by Trichoderma viride. J Agric Food Chem 51(24):6957–6960
Ghorbanpour M, Omidvari M, Abbaszadeh-Dahaji P, Omidvar R, Kariman K (2018) Mechanisms underlying the protective effects of beneficial fungi against plant diseases. Biol Control 117:147–157
Hogan DA (2006) Talking to themselves: autoregulation and quorum sensing in fungi. Eukaryot Cell 5(4):613–619
Hu M, Li QL, Yang YB, Liu K, Miao CP, Zhao LX, Ding ZT (2017) Koninginins R-S from the endophytic fungus Trichoderma koningiopsis. Nat Prod Res 31(7):835–839
Jeerapong C, Phupong W, Bangrak P, Intana W, Tuchinda P (2015) Trichoharzianol, a new antifungal from Trichoderma harzianum F031. J Agric Food Chem 63(14):3704–3708
Jeleń H, Błaszczyk L, Chełkowski J, Rogowicz K, Strakowska J (2014) Formation of 6-n-pentyl-2H-pyran-2-one (6-PAP) and other volatiles by different Trichoderma species. Mycol Prog 13(3):589–600
Kang D, Kim J, Choi J, Liu K, Lee C (2011) Chemotaxonomy of Trichoderma spp. using mass spectrometry-based metabolite profiling. J Microbiol Biotechnol 21(1):5–13
Keswani C (2015) Ecofriendly management of plant diseases by biosynthesized secondary metabolites of Trichoderma spp. J Brief Idea. https://doi.org/10.5281/zenodo.15571
Keswani C, Mishra S, Sarma BK, Singh SP, Singh HB (2014) Unravelling the efficient application of secondary metabolites of various Trichoderma spp. Appl Microbiol Biotechnol 98:533–544
Keswani C, Bisen K, Chitara MK, Sarma BK, Singh HB (2017) Exploring the role of secondary metabolites of Trichoderma in tripartite interaction with plant and pathogens. In: Singh J, Seneviratne G (eds) Agro-environmental sustainability. Springer, Cham, pp 63–79
Li Y, Sun R, Yu J, Saravanakumar K, Chen J (2016) Antagonistic and biocontrol potential of Trichoderma asperellum zjsx5003 against the maize stalk rot pathogen Fusarium graminearum. Indian J Microbiol 56(3):318–327
Mazzei P, Vinale F, Woo SL, Pascale A, Lorito M, Piccolo A (2016) Metabolomics by proton high-resolution magic-angle-spinning nuclear magnetic resonance of tomato plants treated with two secondary metabolites isolated from Trichoderma. J Agric Food Chem 64(18):3538–3545
Mevers E, Saurí J, Moser A, Varlan M, Martin G, Clardy J (2016) Chemical warfare: the battle between termite-associated actinobacteria and Trichoderma harzianum, a fungal pathogen. Planta Med 82(01):SL5
Mohamed SA, Saleh RM, Kabli SA, Al-Garni SM (2016) Influence of solid state fermentation by Trichoderma spp. on solubility, phenolic content, antioxidant, and antimicrobial activities of commercial turmeric. Biosci Biotechnol Biochem 80(5):920–928
Mutawila C, Vinale F, Halleen F, Lorito M, Mostert L (2016) Isolation, production and in vitro effects of the major secondary metabolite produced by Trichoderma species used for the control of grapevine trunk diseases. Plant Pathol 65(1):104–113
Prabhakaran N, Prameeladevi T, Sathiyabama M, Kamil D (2015) Screening of different Trichoderma species against agriculturally important foliar plant pathogens. J Environ Biol 36(1):191–198
Ruiz-Martínez J, Ascacio J, Rodríguez R, Morales D, Aguilar C (2011) Phytochemical screening of extracts from some Mexican plants used in traditional medicine. J Med Plant Res 5(13):2791–2797
Saravanakumar K, Yu C, Dou K, Wang M, Li Y, Chen J (2016) Synergistic effect of Trichoderma-derived antifungal metabolites and cell wall degrading enzymes on enhanced biocontrol of Fusarium oxysporum f. sp. cucumerinum. Biol Control 94:37–46
Saravanakumar K, Chelliah R, Ramakrishnan SR, Kathiresan K, Oh DH, Wang MH (2018) Antibacterial and antioxidant potentials of non-cytotoxic extract of Trichoderma atroviride. Microb Pathog 115:338–342
Shakeri J, Foster HA (2007) Proteolytic activity and antibiotic production by Trichoderma harzianum in relation to pathogenicity to insects. Enzym Microb Technol 40(4):961–968
Shentu X, Liu W, Zhan X, Yu X, Zhang C (2013) The elicitation effect of pathogenic fungi on trichodermin production by Trichoderma brevicompactum. Sci World J 2013:6
Singh HB, Sarma BK, Keswani C (eds) (2016) Agriculturally important microorganisms: commercialization and regulatory requirements in Asia. Springer, Singapore
Singh HB, Sarma BK, Keswani C (eds) (2017) Advances in PGPR. CABI, Wallingford
Sriwati R, Melnick RL, Muarif R, Strem MD, Samuels GJ, Bailey BA (2015) Trichoderma from Aceh Sumatra reduce Phytophthora lesions on pods and cacao seedlings. Biol Control 89:33–41
Sun J, Pei Y, Li E, Li W, Hyde KD, Yin WB, Liu X (2016) A new species of Trichoderma hypoxylon harbours abundant secondary metabolites. Sci Rep 6:37369
Vinale F, Marra R, Scala F, Ghisalberti EL, Lorito M, Sivasithamparam K (2006) Major secondary metabolites produced by two commercial Trichoderma strains active against different phytopathogens. Lett Appl Microbiol 43(2):143–148
Vinale F, Sivasithamparam K, Ghisalberti EL, Marra R, Barbetti MJ, Li H, Woo SL, Lorito M (2008) A novel role for Trichoderma secondary metabolites in the interactions with plants. Physiol Mol Plant Pathol 72(1):80–86
Vinale F, Flematti G, Sivasithamparam K, Lorito M, Marra R, Skelton BW, Ghisalberti EL (2009) Harzianic acid, an antifungal and plant growth promoting metabolite from Trichoderma harzianum. J Nat Prod 72(11):2032–2035
Vinale F, Nigro M, Sivasithamparam K, Flematti G, Ghisalberti EL, Ruocco M, Varlese R, Marra R, Lanzuise S, Eid A, Woo SL, Lorito M (2013) Harzianic acid: a novel siderophore from Trichoderma harzianum. FEMS Microbiol Lett 347(2):123–129
Vinale F, Strakowska J, Mazzei P, Piccolo A, Marra R, Lombardi N, Manganiello G, Pascale A, Woo SL, Lorito M (2016) Cremenolide, a new antifungal, 10-member lactone from Trichoderma cremeum with plant growth promotion activity. Nat Prod Res 30(22):2575–2581
Viniegra-González G (2014) New horizons for the production of industrial enzymes by solid-state fermentation. In: Guevara-Gonzalez R, Torres-Pacheco I (eds) Biosystems engineering: biofactories for food production in the century XXI. Springer International Publishing, Cham, pp 319–340
Wafaa MH, Mohamed HAA (2002) Enhancement of antifungal metabolite production from gamma-ray induced mutants of some Trichoderma species for control onion white rot disease. 植物病理學會刊 11(1):45–56
Yamazaki H, Rotinsulu H, Takahashi O, Kirikoshi R, Namikoshi M (2016) Induced production of a new dipeptide with a disulfide bridge by long-term fermentation of marine-derived Trichoderma brevicompactum. Tetrahedron Lett 57(51):5764–5767
Zhang P, Bao B, Dang HT, Hong J, Lee HJ, Yoo ES, Bae KS, Jung JH (2009) Anti-inflammatory sesquiterpenoids from a sponge-derived fungus Acremonium sp. J Nat Prod 72(2):270–275
Zhang L, Niaz S, Khan D, Wang Z, Zhu Y, Zhou H, Lin Y, Li J, Liu L (2017) Induction of diverse bioactive secondary metabolites from the mangrove endophytic fungus Trichoderma sp. (Strain 307) by co-cultivation with Acinetobacter johnsonii (Strain B2). Mar Drugs 15(2):35
Zhou XX, Li J, Yang YH, Zeng Y, Zhao PJ (2014) Three new koninginins from Trichoderma neokongii 8722. Phytochem Lett 8:137–140
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
De la Cruz-Quiroz, R., Ascacio-Valdés, J.A., Rodríguez-Herrera, R., Roussos, S., Aguilar, C.N. (2019). Phytopathogen Biomass as Inducer of Antifungal Compounds by Trichoderma asperellum Under Solid-State Fermentation. In: Singh, H., Keswani, C., Reddy, M., Sansinenea, E., García-Estrada, C. (eds) Secondary Metabolites of Plant Growth Promoting Rhizomicroorganisms. Springer, Singapore. https://doi.org/10.1007/978-981-13-5862-3_6
Download citation
DOI: https://doi.org/10.1007/978-981-13-5862-3_6
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-13-5861-6
Online ISBN: 978-981-13-5862-3
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)