Abstract
Taenia, one of the earliest recognized helminths, is a comparatively large genus with 42 known valid species. Three most commonly recognized species of human importance are T. solium and T. asiatica (pork tapeworms) and T. saginata (beef tapeworm). Adult tapeworms reside in human intestine, and the disease is called taeniasis. The larva (cysticercus) of only T. solium can infect internal organs of human, known as cysticercosis; when the larva infects the central nervous system (CNS) of the host, it is known as neurocysticercosis (NCC). NCC is the most severe form of the disease with considerable morbidity and mortality. It is considered as the most common cause of community-acquired epileptic seizure disorders. The life cycle of T. solium involves two hosts: humans are the only definitive host and accidental intermediate host, while pig is the natural intermediate host. The eggs passed through faeces by T. solium carriers contaminate the environment. Both human and pig can get infection (cysticercosis/NCC) through ingestion of eggs, while human acquires taeniasis through consumption of cysticercotic pork. T. solium infection is highly endemic in Africa, Asia and Latin America. Now developed world is also facing this problem due to human migration from Taenia endemic areas. Therapeutic measures for NCC-related active epilepsy include anti-epileptic drug(s) with or without steroids: surgery/placement of shunt is indicated for patients with raised intracranial pressure. Antiparasitic drug to kill the brain cysticerci remains controversial. Treatment of choice for taeniasis is niclosamide; alternatives are praziquantel and albendazole. The following measures such as antiparasitic therapy to eliminate Taenia carriers in endemic populations, health education, toilet facilities and handwash with soap, control on sale of measly pork, restriction on pig roaming and pig vaccination, etc. may help to control the disease.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
Taenia species are among the earliest recognized helminths. Life history and ecology of Taenia species are well studied and drawn considerable attention among the eucestodes. Taenia is a comparatively large genus with 42 known valid species and 3 subspecies (Hoberg 2006). Morphologically, they are ribbon-like structure with series of well-defined segments known as proglottids and therefore the name Taenia (Greek ταίνια meaning ribbon/stripe). The most common species infecting humans are the pork tapeworm T. solium and T. asiatica and the beef tapeworm T. saginata. Infection with adult tapeworms of T. solium, T. asiatica or T. saginata leads to taeniasis in humans. Among all the species of Taenia, the larval form (cysticercus) of only T. solium can infect internal organs of human, and the disease is called cysticercosis. When the larva of T. solium is lodged in the brain, it is known as neurocysticercosis (NCC). NCC is considered as the severe form of the disease with considerable morbidity and mortality. The life cycle of T. solium involves two hosts; viz. human (definitive host) and pig (intermediate host). Human harbours the adult tapeworm, which produces thousands of eggs for years. These eggs are excreted through faeces and disseminated in the environment. When free-roaming pigs ingest egg-loaded human faeces, the hexacanth embryos are liberated from the eggs, which penetrate the mucosal layer of the intestinal wall and enter into the blood circulation and subsequently develop into cysticerci (larval forms) in different internal organs, especially in the muscles of pigs. Human can also develop cysticercosis/NCC through consumption of food (raw vegetables and salads, etc.) and water contaminated by T. solium eggs. When human eats undercooked cysticercotic pork, the larva (cysticercus) develops into an adult worm in human intestine, and the disease is called taeniasis. In brief, both human and swine can get cysticercosis/NCC by ingestion of T. solium eggs excreted by the adult worm carriers. Clinical features of NCC may vary from mild form with little or no symptoms to life-threatening medical emergency. T. solium infection is cosmopolitan but highly endemic in African, Asian and Latin American countries where open-field defaecation practice is common and pigs roam freely with easy access to human faeces, and humans consume undercooked pig meat and hygienic standards are poor.
2 Morphology/Structure
The body of a classical adult eucestode is divided into three distinct parts, namely, scolex, neck and strobila. The scolex, responsible for attachment to the host tissue, is present at the anterior end. The size and morphological features of scolex are used to identify the worms. The neck is present immediately posterior to the scolex. It is the narrowest unsegmented and poorly differentiated region. New segments or proglottids differentiate from the neck region, and they push the older segments gradually posterior forming a chain of proglottids called strobila, which is the last part of the adult parasite. As each proglottid moves posteriorly, its reproductive system starts maturing progressively. Therefore, the posterior most proglottids have the fully developed reproductive systems. The progressive maturity of the reproductive systems divides strobila into immature, mature and gravid proglottids (filled with egg) with often atrophied reproductive organs.
Three species of the Taenia genus, T. solium, T. saginata and T. asiatica, can cause taeniasis (intestinal infection) in humans. However, only the first two species have been considered for centuries except T. asiatica that remained undiscovered till recently. These tapeworms are flat and opaque white or yellowish with long segments measuring 1–12 m in the adult stage. An adult tapeworm has three segments: scolex, narrow neck and large strobila with hundreds of proglottids. The scolex is the organ for attachment with four suckers and a rostellum. Rostellum may be armed with hooks as in T. solium, or unarmed and sunken (T. saginata), or with rudimentary hooklets (T. asiatica) (Fig. 7.1) (Eom and Rim 1993; Flisser 1994). The scolex is a muscular structure with mesenchymal tissues that lodges the main part of the nervous system of the parasite and nephridial canals. Proximal segments are immature, followed by mature and gravid proglottids filled with eggs. Mature proglottids are hermaphrodites and contain thousands of testes. The female sexual organs have one lobulated ovary, connected to an oviduct. Gravid proglottids are full of 50–60 × 103 fertile eggs. The egg-containing uterus develops 7–32 lateral branches; this feature allows identification of species. T. solium has 7–16 and T. saginata has 14–32 branches. The eggs are spherical and their size varies from 31 to 43 μm in diameter. When eggs are excreted, most of them are fully embryonated and infective, while others may be at different stages of maturation. The main features of tapeworms and eggs are shown in Table 7.1 and Fig. 7.2 (Eom and Rim 1993; Flisser 2013; Galan-Puchades and Fuentes 2013).
After 15–48 h of ingestion of eggs, hexacanth embryos (oncopsheres) are liberated in the intestine of the host that pierce the intestine and enter into the blood circulation. When the embryos are lodged in the internal organs, they develop into the larval forms called cysticercus, which is a fluid-filled bladder-like structure with invaginated whitish scolex (de Aluja et al. 1998). When definitive host (human) consumes the viable cysticercus present in the muscles of intermediate host (pork/beef), bladder wall pore widens that allows the scolex and neck to evaginate, and the adult tapeworm develops (de Queiroz and Alkire 1998). The adult tapeworm resides in the small intestine of humans, the only definitive hosts. Stages of taeniid development are as follows: embryo inside the egg, circulating oncosphere or larva, post-oncosphere that is transformed into the cysticercus or metacestode in tissues (the post-larval or pre-adult stage) and the adult worm. Eggs and embryos are microscopic (Fig. 7.3), while cysticerci (Fig. 7.4) and adult worms are macroscopic (Fig. 7.1).
3 Biology/Life Cycle
3.1 T. solium
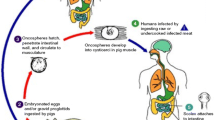
It is commonly known as pork tapeworm. Humans are the only definite host and harbour the adult tapeworm in their intestine. Both swine and human act as the intermediate hosts harbouring larvae in different internal organs (Fig. 7.5). The adult tapeworm residing in the small intestine of human produces thousands of eggs daily which are disseminated into the environment through faeces. The intermediate host (pig) ingests these eggs, which develop into cysticerci in different internal organs like muscles and brain. When human consumes cysticercotic pork, these cysts develop into adult worms residing the human intestine. The gravid proglottids start to separate from the distal end and are excreted in the faeces after 2 months of intestinal infection. Every day around four to five proglottids break off from the adult worm, and each segment has 50–60 × 103 fertile eggs. The eggs are spherical (31–43 μm in diameter) with thick striated cover containing oncosphere. Both swine and man can ingest the eggs, and these eggs reach digestive tract and lose their coat due to action of the gastric acid and pancreatic enzymes resulting in release of hexacanth embryos or oncospheres. Oncospheres pierce the intestinal wall using their hooklets to reach the blood circulation and further reach the different organs such as subcutaneous tissue, skeletal muscles, central nervous system (CNS) and eyes. Now, the oncospheres drop their hooklets, take a vesicular shape and develop into cysticerci by gradual invagination of scolex, and it takes approximately 2 months to develop (Escobar and Nieto 1972). Its life cycle is completed when human consumes the undercooked measly pork. After reaching the small intestine, the scolex evaginates from the cysticerci and attaches itself to the mucosal wall and gradually evolves into the adult tapeworm. However, humans may also be infected with eggs in one of the following ways: (1) by hetero-infection through consumption of food/water contaminated with eggs of Taenia, (2) by exogenous autoinfection caused by ano-oral ingestion in Taenia carriers and (3) by endogenous autoinfection, in which the eggs present in the small intestine reach to the stomach due to reverse peristalsis. Hetero-infection is the most common route, while the last two modes of infection are very uncommon. The eggs of Taenia are sticky, and they may remain stuck to nail and nail beds of Taenia carriers if the hands are not properly washed with soap after defaecation. While handling such carriers can contaminate the food and disseminate the disease (cysticercosis/NCC).
In human, the parasite has a predilection to the brain and spinal cord, and when it gets lodged in these organs, it results in NCC. The larval stage also infects other tissues, especially those with pulsatile or contractile property and enriched in cholinergic innervations like skeletal muscle, diaphragm, heart, tissue and cavities subjected to contractile, rhythmic pulsatile movement by the internal organs like peritoneum, pleura and subcutaneous tissue (Del Brutto and Sotelo 1988).
Other animals have also been reported to harbour cysticerci of T. solium. An Indonesian study revealed that sera from 7 of 64 dogs were highly positive for T. solium infection by ELISA and immunoblot. Subsequent examination of two such dogs showed T. solium cysticerci in their brain and heart muscle. Mitochondrial DNA analyses revealed that cysticerci from these dogs were similar with cysticerci obtained previously from swine and biopsies of local people (Ito et al. 2002). T. solium cysticercus had also been reported from the brain of a cat (Schwan et al. 2002).
3.2 T. saginata
Humans get infection through consumption of raw/undercooked beef infected with larvae of T. saginata (Fig. 7.5). The developmental stages of both T. saginata and T. solium are similar. After 3 months, T. saginata becomes sexually mature and produce gravid proglottids, which are expelled in faeces. Gravid proglottids may contain 50–80 × 103 eggs, with different maturation stages. Eggs are only infective to cattle, the intermediate host; hence T. saginata does not cause cysticercosis in human. When ingested eggs by the intermediate host (cattle) come in contact with gastric and intestinal juices, the active embryos are liberated, which penetrate the intestinal wall and reach the blood circulatory system. Generally, oncospheres develop in to cysticercus in cardiac and skeletal muscles and seldom in fat and visceral organs, and become infective to human by 10 week time. Cysticerci may start degenerating within a few months after infection, and a substantial numbers of cysticerci are usually dead and calcified by about 9 months (Pawlowski and Murell 2000).
3.3 T. asiatica
T. asiatica was first reported in Taiwan and later on in many other Asian countries such as Indonesia, Korea, the Philippines, and Thailand (Eom et al. 1992). It has recently been reported from India (Singh et al. 2016). Morphologically, T. asiatica is related to T. saginata; it has scolex without hooklets, a large number of dichotomous pattern of uterine branches in gravid proglottids and a posterior protuberance (Table 7.1). T. asiatica cysticerci develop in visceral organs of pigs such as the liver, omentum, lungs and serosa; however, T. saginata cysticerci infect only muscles of cattle. In general, T. asiatica life cycle is similar to T. solium; specifically the adult stage develops in humans (Fig. 7.5; Eom et al. 1992). Taeniasis due to T. asiatica occurs in human after consumption of raw/undercooked meat or viscera of pigs infected with cysticerci. Cysticercosis in human due to T. asiatica has not been reported till date.
4 Epidemiology
The incidence of T. solium infection significantly varies according to sanitation, eating habits and pig husbandry practices in a region. However, it is hard to estimate the exact prevalence of T. solium-associated taeniasis, because eggs of different Taenia species are indistinguishable by microscopic method.
Cysticercosis is an under-reported disease because of involvement of different internal organs. However, NCC is considered the severe form of the disease, and epilepsy is the common clinical manifestation of parenchymal NCC. Extra-parenchymal location of the cysticerci in the brain leads to raised intracranial pressure and hydrocephalus. The World Health Organization estimates approximately 50,000 deaths every year due to NCC. Overall, NCC is identified as a cause of active epilepsy in 26.3–53.8% of seizure disorders in the developing world (Del Brutto et al. 2005; Montano et al. 2005; Prasad et al. 2009a, b). It is prevalent in Asian countries such as China, Cambodia, India, Indonesia, Mongolia, Myanmar, Nepal, Thailand and Vietnam. T. solium infection has been eradicated from Japan and South Korea. Seroprevalence studies in Asian countries (Vietnam, China, Korea and Indonesia) indicate high rates of exposure to the parasite ranging from 0.02% to 12.6% (Kong et al. 1993; Margono et al. 2001; Rajshekhar et al. 2003).
Reports have demonstrated a potentially high risk of T. solium infection for inhabitants of many Latin American countries with variable incidence rates suggesting an active transmission involving pig and human cycle in the region (Fleury et al. 2006; Garcia-Noval et al. 1996). Studies from Guatemala, Honduras, Peru and Mexico demonstrated NCC infection in rural populations between 9% and 18% (Flisser et al. 2003). A study from Mexico had shown that up to 50% of patients with evidence of NCC were neurologically and systematically asymptomatic (Chavarria et al. 2003). Swine cysticercosis is also often reported at meat inspection in the slaughterhouses of Latin America.
T. solium is an emerging and expanding zoonosis in African countries like Cameroon, Mozambique, Nigeria, South Africa, Tanzania, Zimbabwe, etc. Data from African countries show low incidence of human infection and a high prevalence in pig populations. This discrepancy may be due to lack of suitable surveillance, monitoring and reporting systems. Moreover, a high prevalence of pig cysticercosis (20–40%) is reported from East and Southern Africa (Phiri et al. 2002).
NCC is now becoming prevalent in developed countries because of increased human travel from endemic areas and migration of tapeworm carriers (Burneo et al. 2009; Schantz et al. 1992). Individuals who have never gone outside the USA as well as those who travel to disease-endemic regions are at risk acquiring infection. Hospital-based data analysis showed that up to 2% of admissions in neurosciences in southern California were due to NCC (McCormick 1985). The USA reports more than 1000 cases of NCC per year (Shandera et al. 1994).
Only few population-based data are available that provide evidence about the community burden, risk factors, and geographical distribution of cysticercosis in India. Studies from India reported that 28.4–48.3% cases of active epilepsy are due to NCC by neuroimaging techniques such as CT and MRI (Prasad et al. 2009a, b; Rajshekhar et al. 2006). In Indian subcontinent, single cyst infection is more frequently reported, and its prevalence ranges from 47.7% to 53.4% of NCC cases (Prabhakaran et al. 2007).
5 Clinical Features
5.1 Clinical Manifestations of Taeniasis
Most people having taeniasis are either asymptomatic or have lowtomoderate symptoms. The most frequent complaint is passage of proglottids with slight discomfort. Other common symptoms are abdominal pain often colicky in nature (frequent in children), loss of appetite, nausea, constipation/diarrhoea, dizziness, headache, weakness, hyper-excitability, etc. These clinical symptoms may appear when the tapeworms become fully mature in human intestine, usually 6–8 weeks after ingestion of measly meat of swine (Ooi et al. 2013). Stomach pain and nausea are more frequent in the morning which is reduced after having small amounts of food. These symptoms are most common in children than adults and often lead to increased or decreased appetite. Infants may have diarrhoea, fever, irritability, vomiting and weight loss. The most severe complication of taeniasis is appendicitis.
5.2 Clinical Manifestations of NCC
The clinical manifestations of NCC vary with location, stage and number of T. solium cysticerci in the brain and immune response of the host to the parasite. It can affect the parenchyma, subarachnoid space or intraventricular systems within the CNS and rarely ocular and spinal region.
5.3 Parenchymal NCC
In parenchymal NCC, the most common clinical manifestation is seizure. It accounts up to 80% of patients (Ndimubanzi et al. 2010). Generalized tonic-clonic or simple partial seizure with motor symptomatology is mostly associated with NCC. However, some patients may have myoclonic, truncal or complex partial seizures (Sotelo et al. 1985). Presence of multiple degenerating parenchymal cysts with massive pericystic oedema may lead to cysticercotic encephalitis. The other most common clinical feature is headache (37% of NCC cases) followed by altered mental state (28% cases), neuro-focal deficits (16% cases), signs of increased intracranial pressure (12% cases) and cognitive decline (5% cases) (Carabin et al. 2011).
5.4 Subarachnoid NCC
Hydrocephalus is the most common clinical presentation of subarachnoid NCC due to increased intracranial pressure followed by stroke in about 12% of cases (Marquez and Arauz 2012). A variety of stroke syndromes have been described in patients with NCC such as cerebral infarctions, intracranial haemorrhages and transient ischemic attacks. Other manifestations are acute meningitis and cranial nerve involvement.
5.5 Ventricular NCC
The main clinical manifestation of intraventricular NCC is obstruction of cerebrospinal fluid (CSF) flow that leads to increased intracranial pressure leading to acute hydrocephalus. It usually happens when freely moving cysticerci reach the third ventricle from lateral ventricles or move up to cerebral aqueduct from fourth ventricle leading to blockage of CSF flow. It is an acute emergency and may need urgent shunt placement. Cysticerci present in the lateral ventricles may compress nearby tissues and are generally associated with focal neurological symptoms. Cysticerci present in the fourth ventricle may cause brainstem dysfunction because of compression of fourth ventricle floor (Madrazo et al. 1983; Sinha and Sharma 2012).
5.6 Spinal Cord NCC
In NCC, spinal cord involvement is very rare (1–5%); however, introduction of MRI has significantly improved the diagnosis. Generally, cysts are single or clusters of multiple cysts and present in the spinal cord parenchyma or spinal cord subarachnoid space, and the clinical symptoms are radicular pain and motor deficits of subacute onset with progressive course (Bandres et al. 1992; Park et al. 2011).
5.7 Ophthalmic Cysticercosis
Intraocular cysticerci (ophthalmic cysticercosis) may be found in the anterior chamber, the lens, the vitreous and the sub-retinal space. Although it is often asymptomatic, inflamed degenerating cysticerci cause progressive decrease in vision with symptoms like proptosis, diplopia, chorioretinitis, retinal detachment or vasculitis (Kruger-Leite et al. 1985; Madigubba et al. 2007).
5.8 Systemic Cysticercosis
Cysticerci can develop in almost any body site but tend to have a predilection for muscle or subcutaneous tissues. Cysticerci at these sites are usually asymptomatic, but the patient may notice subcutaneous, pealike or walnut-sized nodules. Subcutaneous nodules are more common in patients from Asia and Africa than from Latin America. In cases of major muscle involvement, acute myopathy can develop (Sawhney et al. 1976).
6 Pathogenesis of NCC
T. solium cysticercus is a structurally complex helminthic larva, which expresses diverse sets of antigens mounting variable immune response that leads to various clinical manifestations of the disease. Histological investigations on human and pig brain tissues have shown a very low or no inflammatory response around viable cysticerci. Viable cysticerci escape the host’s immune response by blocking/inhibiting the complement system. The viable cysticerci produce excretory/secretory molecules such as paramyosin which inhibits Clq (Laclette et al. 1987); taeniaestatin identified in T. taeniaeformis inhibits both classical and alternate pathways of complement system. Taeniaestatin also decreases IL-1 and IL-2 production and lymphocyte proliferation (White et al. 1992). Cysticerci cell wall is rich in sulphated polysaccharides, which are to a large extent immunologically inert. Moreover, sulphated polysaccharides activate and consume complement and evade its response. Cysticercal cysteine proteinase degrades the host immunoglobulins, and its prostaglandin E2 (PGE2) induces Th2 response and suppresses the inflammation (Garcia et al. 2014; Terrazas 2008). However, when the cysticerci start to die/degenerate, granuloma is formed with inflammation around the dying cysts in both human and pig (Singh et al. 2013, 2015a, b). Immune cells such as eosinophils, lymphocytes, macrophages, and plasma cells infiltrate around the dying cysticerci to form granuloma. These cells secrete cytokines, chemokines and other inflammatory mediators that cause the various symptoms in patients. The infiltrating cells produce high level of Th1 (IFN-γ, TGF-β and IL-18) and lower level of Th2 cytokines (IL-4, IL-13 and IL-10) in the brain tissue surrounding dying cyst (Restrepo et al. 2001). Anti-helminthic treatment also induces Th1 (IFN-γ) and pleiotropic (IL-6) cytokine response with leukocyte infiltration around dead cysticerci in swine (Singh et al. 2015a, b).
The host immune response against cysticerci can be of two types, i.e. humoral and cellular. In humoral immune response, a number of immunoglobulin classes as specific antibodies against the parasite are produced. The most common immunoglobulin is IgG detected in patient’s CSF, serum and saliva which is suggestive of long duration of infection (Grogl et al. 1985; Zini et al. 1990). The immune response against T. solium cysticerci display both Th1 and Th2 type; however, the basic mechanisms are not clear. Probably, the parasite is destroyed by eosinophils, which is supposed to be mediated by Th2 cytokines (Ostrosky-Zeichner and Estanol 1999). The inflammatory response that kills the parasite and leads to resolution of fibrosis is mediated by Th1 cytokines. Peripheral immune response is associated with reduced lymphocytic proliferation, inhibition of granulocyte aggregation and induction of Th2 response (IL-4, IL-5 and IL-13) (White et al. 1997). In chronic NCC, pro-inflammatory cytokines, up-regulated cellular adhesion molecules such as ICAM-1 and activation of MMP-9 and MMP-2 contribute to blood-brain barrier disruption resulting in seizure disorder (Alvarez et al. 2002; Prasad et al. 2009a, b; Verma et al. 2011). It has been reported that individuals with glutathione S-transferase (GST)-M1 and (GST)-T1 deletions (null genotypes) are low producers of GST enzymes and such individuals are at higher risk to develop seizures in NCC. GST enzymes are essential for the protection of cells from damage caused by reactive oxygen species (ROS) generated during inflammation. Hence, higher GST activity may maintain asymptomatic condition, possibly by neutralizing the ROS and free radicals (Singh et al. 2017).
7 Diagnosis
7.1 Diagnosis of Taeniasis
Intestinal Taenia infections in humans are diagnosed by the detection of gravid segments or eggs in faecal samples. However, there are various tools for diagnosis of taeniasis with variable sensitivity and specificity.
7.2 Microscopy
Traditional microscopy to detect Taenia eggs in the stool has poor sensitivity but a very high specificity. However, concentration methods, preferably using sedimentation, increase the sensitivity. Taenia egg appears as a thick radiate cover and hooks under the microscope. However, eggs of different Taenia species (T. solium, T. asiatica and T. saginata) cannot be differentiated by microscopic examination (Garcia and Del Brutto 2003). Detection of gravid segment with its uterine branching is a reliable test to differentiate different Taenia species.
7.3 Antigen Detection in Stool
Copro-antigen detection in stool by capture ELISA has enhanced the sensitivity two to three times for diagnosis of taeniasis. To detect tapeworm antigen in stool, generally polyclonal antibody is used against the adult tapeworm in copro-antigen ELISA (Bustos et al. 2012).
7.4 Molecular Method
The differentiation of the different species of Taenia is possible by mitochondrial DNA (cox1, cob and NADH dehydrogenase 1 gene)-based PCR, and the results are consistent if tapeworm material is available (Gonzalez et al. 2000, 2002; Yamasaki et al. 2004). However, the sensitivity of direct PCR assays in stool samples is yet to be defined.
7.5 Diagnosis of NCC
The clinical diagnosis of NCC is hard due to its polymorphic/nonspecific disease symptoms. However, a precise diagnosis can be done if clinical and epidemiologic data is interpreted together with the neuroimaging findings and the results of specific immunological tests.
7.6 Immunodiagnostic Techniques
In immunodiagnosis, specific antibodies and circulating cysts antigens are detected in serum or CSF. Various techniques such as complement fixation test, ELISA, indirect haemagglutination test, latex agglutination, radioimmunoassay and enzyme-linked immune-electrotransfer blot (EITB) have been used to detect antibodies. The serum-based ELISA to detect antibody is not reliable to diagnose NCC; however, it is very much in use due to simplicity of the technique. EITB is the most specific test so far. This immune-blot uses fractions of glycoproteins (50, 39–42, 24, 21, 18, 14 and 13 kDa) from crude extracts purified using a lentil-lectin column (Tsang et al. 1989).
7.7 Neuroimaging Techniques
Neuroimaging techniques such as computed tomography (CT) and magnetic resonance imaging (MRI) have significantly enhanced the diagnostic precision of NCC. CT is found to be more sensitive for the detection of calcified cysts; however, MRI is the most accurate as it gives information on the number and topography of lesions, stage of the cysts and the intensity of inflammatory response.
MRI has particular advantages such as it is non-invasive, it does not use any ionizing radiation and it has a high soft-tissue resolution. It may also provide both morphological and functional information. MRI is found to be accurate neuroimaging tool for the detection of dying and viable cysts, whereas CT is good for calcified lesions, but it cannot differentiate the stage of the parasite (Garcia and Del Brutto 2003). Moreover, a study has shown that MRI with gradient echo sequence phase imaging is equally good as CT for the detection of the scolex in lesions and also the calcified stage (Gupta et al. 2001).
8 Treatment
8.1 Anti-helminthic Therapy
Although treatment for NCC with anticysticercal drugs such as albendazole, mebendazole, praziquantel, metriphonate and flubendazole has been used, there has been a debate over safety and usefulness of such drugs due to lack of clinical trials to define doses and duration of therapy. Cysts location in the brain is a major factor that decides the success of anti-helminthic treatment. When the parasite infects the brain parenchyma, treatment with these drugs show radiological and clinical improvement in majority of patients with only a low adverse effect in some cases (Carpio et al. 2008; Del Brutto et al. 2006). However, when the parasite infects the subarachnoid basal cisterns, the prognosis is uncertain, and in such cases, either albendazole or praziquantel is preferred (Sotelo et al. 1988). However, controversies still persist about the use antiparasitic drugs in treatment of NCC, especially on seizure occurrence and other neurological disorders that result from inflammatory response triggered by antiparasitic treatment. Albendazole has been found to be more effective than praziquantel in clinical trials (Takayanagui and Jardim 1992). Another benefit of using albendazole is that it can also kill subarachnoid and ventricular cysts and has relatively less interaction with steroids and anti-epileptic drugs. However, increased dose of praziquantel (up to 30 mg/kg/day) or more prolonged or even repeated use has been recommended for treatment of patients with large subarachnoid cyst (Fleury et al. 2011). Further, surgery is also recommended by some clinicians as the best option for this form of infection. Albendazole is effective on 75–90% of cysts in the parenchyma when 15 mg/kg/day of albendazole is administered; however, the duration of treatment in such patients varies from 8 to 30 days (Castro et al. 2009). Further, antiparasitic therapy is recommended only in patients with low number of parenchymal cyst since the dying parasites release antigens to the surrounding tissues that initiate a strong inflammatory immune response, thus aggravating the symptoms. In order to reduce the risk, concomitant corticosteroids should be given, especially if there are large numbers of cysts. However, antiparasitic therapy is not recommended in patients with cysticercotic encephalitis. Recommendations for the treatment of NCC are given in Table 7.2 (Garcia et al. 2002).
8.2 Anti-inflammatory Therapy
Corticosteroids are commonly used to reduce the inflammatory response that leads to various neurological symptoms due to degenerating cysticerci. Drug of choice for this therapy is dexamethasone; dose varies between 4.5 and 12 mg/day. It is also used in chronic cysticercosis arachnoiditis or encephalitis, where up to 32 mg/day of dexamethasone may be given to decrease the brain oedema (Del Brutto et al. 1993). Another drug, prednisone at 1 mg/kg/day, may replace dexamethasone when long-term corticosteroid is given (Suastegui et al. 1996). To reduce the raised acute intracranial pressure secondary to NCC, mannitol (2 g/kg/day) is also recommended as an osmotic agent.
9 Anti-epileptic Drug Therapy
Other drugs generally used to treat symptomatic NCC patients are anti-epileptic drugs (AEDs). Carbamazepine, phenytoin and phenobarbitone are the first-line AEDs more often used for the control of seizures due to NCC. In recurrent severe headache associated with seizures, valproic acid/topiramate is preferred. Addition of praziquantel and albendazole with AED may improve seizure control. One study showed that 83% patients were seizure-free when treated with combination of anti-epileptic and anticysticercal drugs, whereas only 26% patients were seizure-free when treated with AEDs alone over a period of 28 months. However, despite better seizure control, albendazole and praziquantel may not be a definitive therapy for NCC-related active epilepsy, and AEDs need to be continued (Del Brutto et al. 1992). There is no definite time frame how long the anti-epileptic treatment should be given. Generally, AED is prescribed until a 2-year seizure-free period followed by gradual withdrawal.
10 Prevention
Cysticercosis/NCC is recognized as an eradicable disease. Eradication of swine cysticercosis through better animal husbandry and pig meat inspection are the important parameters adopted by the developed countries. These measures helped in breaking the transmission cycle of T. solium infection in the USA and Western Europe (Ferreira et al. 1997). Tapeworm carriers are the main target for anti-helminthic therapy for effective control of T. solium infection, since a small number of such carriers are likely to infect large numbers of healthy individuals. In developing countries the control measures should focus on mass administration of anti-helminthic drugs in endemic regions to cure tapeworm carriers and public awareness programme through health education. Other measures include toilet facilities to discourage open-field defaecation, handwash with soap after defaecation, restriction on sale of measly pork, restriction on pig roaming and pig vaccination, etc. Mass treatment of pigs in highly endemic regions with albendazole for 1 week followed by at least 2 weeks window period before slaughter for human consumption can reduce the incidence of T. solium carriers.
11 Treatment of Taeniasis
The only source of NCC/cysticercosis both for humans and pigs is the adult T. solium carrier individuals; a single tapeworm carrier can infect large number of hosts. Therefore, tapeworm carriers are the appealing target for control of the disvease. Tapeworm carriers can be cured either by individualized or mass treatment of the population in the endemic area with a single oral dose of either niclosamide (2 g in adults) or praziquantel (5–10 mg/kg; Garcia et al. 2007). Albendazole 15 mg/kg is a good alternative to praziquantel especially in the developing countries.
12 Vaccination of Swine
Vaccination/immunization of porcine population in endemic region may be good approach to interrupt the T. solium life cycle thus preventing taeniasis and NCC. Many research groups had evaluated several parasite antigens derived from different developmental stages of the related cestodes (T. crassiceps, T. solium, T. saginata and T. ovis) or of synthetic origin with variable results. Three different protective antigens (TSOL18, TSOL45-1A and TSOL16) were identified from T. solium oncospheres and evaluated as vaccines (Lightowlers 2004). These antigens were found to induce almost complete protection in vaccinated pigs challenged with T. solium in experiments. Studies showed that the two doses of TSOL18 vaccine had given almost complete protection from infection in swine population (Flisser et al. 2004; Gonzalez et al. 2005).
13 Treatment of Infected Swine
Treatment of cysticercotic swine can reduce the taeniasis burden and help in T. solium elimination. Praziquantel and albendazole are proven to be highly effective in treatment of swine cysticercosis. However, oxfendazole at a single oral dose of 30 mg/kg demonstrated 100% effective to kill muscle cysts with no major side effects (Pondja et al. 2012). In naturally infected swine, treatment with albendazole (15 mg/kg) for 2 weeks was found 94% and 100% effective to kill the cysticerci in the brain and muscles, respectively (Singh et al. 2015a, b).
14 Conclusions
T. solium infection (taeniasis and cysticercosis/NCC) is a serious public health issue worldwide, particularly in developing nations including India. Swine cysticercosis is an economic loss to the pig farmers. Pig health and management would produce benefits to pig farmers. Avoidance of open defaecation and practice of using latrines may prevent roaming pigs or piglets from infection. Proper porcine meat inspection in slaughterhouses and other measures such as health education, availability of health-care services, elimination of T. solium carriers by mass treatment and control on sale of cysticerci infected pork may help in decreasing the infection rate in the endemic areas.
References
Alvarez JI, Colegial CH, Castano CA, Trujillo J, Teale JM, Restrepo BI (2002) The human nervous tissue in proximity to granulomatous lesions induced by Taenia solium metacestodes displays an active response. J Neuroimmunol 127:139–144
Bandres JC, White AC Jr, Samo T, Murphy EC, Harris RL (1992) Extraparenchymal neurocysticercosis: report of five cases and review of management. Clin Infect Dis 15:799–811
Burneo JG, Plener I, Garcia HH (2009) Neurocysticercosis in a patient in Canada. CMAJ 180:639–642
Bustos JA, Rodriguez S, Jimenez JA, Moyano LM, Castillo Y, Ayvar V, Allan JC, Craig PS, Gonzalez AE, Gilman RH, Tsang VC, Garcia HH (2012) Detection of Taenia solium taeniasis coproantigen is an early indicator of treatment failure for taeniasis. Clin Vaccine Immunol 19:570–573
Carabin H, Ndimubanzi PC, Budke CM, Nguyen H, Qian Y, Cowan LD, Stoner JA, Rainwater E, Dickey M (2011) Clinical manifestations associated with neurocysticercosis: a systematic review. PLoS Negl Trop Dis 5:e1152
Carpio A, Kelvin EA, Bagiella E, Leslie D, Leon P, Andrews H, Hauser WA (2008) Effects of albendazole treatment on neurocysticercosis: a randomised controlled trial. J Neurol Neurosurg Psychiatry 79:1050–1055
Castro N, Marquez-Caraveo C, Brundage RC, Gonzalez-Esquivel D, Suarez AM, Gongora F, Jara A, Urizar J, Lanao JM, Jung H (2009) Population pharmacokinetics of albendazole in patients with neurocysticercosis. Int J Clin Pharmacol Ther 47:679–685
Chavarria A, Roger B, Fragoso G, Tapia G, Fleury A, Dumas M, Dessein A, Larralde C, Sciutto E (2003) TH2 profile in asymptomatic Taenia solium human neurocysticercosis. Microbes Infect 5:1109–1115
de Aluja AS, Martinez MJ, Villalobos AN (1998) Taenia solium cysticercosis in young pigs: age at first infection and histological characteristics. Vet Parasitol 76:71–79
de Queiroz A, Alkire NL (1998) The phylogenetic placement of Taenia cestodes that parasitize humans. J Parasitol 84:379–383
Del Brutto OH, Sotelo J (1988) Neurocysticercosis: an update. Rev Infect Dis 10:1075–1087
Del Brutto OH, Santibanez R, Noboa CA, Aguirre R, Diaz E, Alarcon TA (1992) Epilepsy due to neurocysticercosis: analysis of 203 patients. Neurology 42:389–392
Del Brutto OH, Sotelo J, Roman GC (1993) Therapy for neurocysticercosis: a reappraisal. Clin Infect Dis 17:730–735
Del Brutto OH, Santibanez R, Idrovo L, Rodriguez S, Diaz-Calderon E, Navas C, Gilman RH, Cuesta F, Mosquera A, Gonzalez AE, Tsang VC, Garcia HH (2005) Epilepsy and neurocysticercosis in Atahualpa: a door-to-door survey in rural coastal Ecuador. Epilepsia 46:583–587
Del Brutto OH, Roos KL, Coffey CS, Garcia HH (2006) Meta-analysis: cysticidal drugs for neurocysticercosis: albendazole and praziquantel. Ann Intern Med 145:43–51
Eom KS, Rim HJ (1993) Morphologic descriptions of Taenia asiatica sp. n. Kor J Parasitol 31:1–6
Eom KS, Rim HJ, Geerts S (1992) Experimental infection of pigs and cattle with eggs of Asian Taenia saginata with special reference to its extrahepatic viscerotropism. Kisaengchunghak Chapchi 30:269–275
Escobar A, Nieto D (1972) Parasitic diseases. In: Minckler J (ed) Pathology of the nervous system. McGraw-Hill, New York, pp 2503–2521
Ferreira AP, Vaz AJ, Nakamura PM, Sasaki AT, Ferreira AW, Livramento JA (1997) Hemagglutination test for the diagnosis of human neurocysticercosis: development of a stable reagent using homologous and heterologous antigens. Rev Inst Med Trop Sao Paulo 39:29–33
Fleury A, Morales J, Bobes RJ, Dumas M, Yanez O, Pina J, Carrillo-Mezo R, Martinez JJ, Fragoso G, Dessein A, Larralde C, Sciutto E (2006) An epidemiological study of familial neurocysticercosis in an endemic Mexican community. Trans R Soc Trop Med Hyg 100:551–558
Fleury A, Carrillo-Mezo R, Flisser A, Sciutto E, Corona T (2011) Subarachnoid basal neurocysticercosis: a focus on the most severe form of the disease. Expert Rev Anti-Infect Ther 9:123–133
Flisser A (1994) Taeniasis and cysticercosis due to Taenia solium. Prog Clin Parasitol 4:77–116
Flisser A (2013) State of the art of Taenia solium as compared to Taenia asiatica. Kor J Parasitol 51:43–49
Flisser A, Sarti E, Lightowlers M, Schantz P (2003) Neurocysticercosis: regional status, epidemiology, impact and control measures in the Americas. Acta Trop 87:43–51
Flisser A, Gauci CG, Zoli A, Martinez-Ocana J, Garza-Rodriguez A, Dominguez-Alpizar JL, Maravilla P, Rodriguez-Canul R, Avila G, Aguilar-Vega L, Kyngdon C, Geerts S, Lightowlers MW (2004) Induction of protection against porcine cysticercosis by vaccination with recombinant oncosphere antigens. Infect Immun 72:5292–5297
Galan-Puchades MT, Fuentes MV (2013) Taenia asiatica: the most neglected human Taenia and the possibility of cysticercosis. Kor J Parasitol 51:51–54
Garcia HH, Del Brutto OH (2003) Imaging findings in neurocysticercosis. Acta Trop 87:71–78
Garcia HH, Evans CA, Nash TE, Takayanagui OM, White AC Jr, Botero D, Rajshekhar V, Tsang VC, Schantz PM, Allan JC, Flisser A, Correa D, Sarti E, Friedland JS, Martinez SM, Gonzalez AE, Gilman RH, Del Brutto OH (2002) Current consensus guidelines for treatment of neurocysticercosis. Clin Microbiol Rev 15:747–756
Garcia HH, Gonzalez AE, Del Brutto OH, Tsang VC, Llanos-Zavalaga F, Gonzalvez G, Romero J, Gilman RH (2007) Strategies for the elimination of taeniasis/cysticercosis. J Neurol Sci 262:153–157
Garcia HH, Rodriguez S, Friedland JS (2014) Immunology of Taenia solium taeniasis and human cysticercosis. Parasite Immunol 36:388–396
Garcia-Noval J, Allan JC, Fletes C, Moreno E, DeMata F, Torres-Alvarez R, Soto de Alfaro H, Yurrita P, Higueros-Morales H, Mencos F, Craig PS (1996) Epidemiology of Taenia solium taeniasis and cysticercosis in two rural Guatemalan communities. Am J Trop Med Hyg 55:282–289
Gonzalez LM, Montero E, Harrison LJ, Parkhouse RM, Garate T (2000) Differential diagnosis of Taenia saginata and Taenia solium infection by PCR. J Clin Microbiol 38:737–744
Gonzalez LM, Montero E, Puente S, Lopez-Velez R, Hernandez M, Sciutto E, Harrison LJ, Parkhouse RM, Garate T (2002) PCR tools for the differential diagnosis of Taenia saginata and Taenia solium taeniasis/cysticercosis from different geographical locations. Diagn Microbiol Infect Dis 42:243–249
Gonzalez AE, Gauci CG, Barber D, Gilman RH, Tsang VC, Garcia HH, Verastegui M, Lightowlers MW (2005) Vaccination of pigs to control human neurocysticercosis. Am J Trop Med Hyg 72:837–839
Grogl M, Estrada JJ, MacDonald G, Kuhn RE (1985) Antigen-antibody analyses in neurocysticercosis. J Parasitol 71:433–442
Gupta RK, Rao SB, Jain R, Pal L, Kumar R, Venkatesh SK, Rathore RK (2001) Differentiation of calcification from chronic hemorrhage with corrected gradient echo phase imaging. J Comput Assist Tomogr 25:698–704
Hoberg EP (2006) Phylogeny of Taenia: species definitions and origins of human parasites. Parasitol Int 55(Suppl):S23–S30
Ito A, Putra MI, Subahar R, Sato MO, Okamoto M, Sako Y, Nakao M, Yamasaki H, Nakaya K, Craig PS, Margono SS (2002) Dogs as alternative intermediate hosts of Taenia solium in Papua (Irian Jaya), Indonesia confirmed by highly specific ELISA and immunoblot using native and recombinant antigens and mitochondrial DNA analysis. J Helminthol 76:311–314
Kong Y, Cho SY, Cho MS, Kwon OS, Kang WS (1993) Seroepidemiological observation of Taenia solium cysticercosis in epileptic patients in Korea. J Kor Med Sci 8:145–152
Kruger-Leite E, Jalkh AE, Quiroz H, Schepens CL (1985) Intraocular cysticercosis. Am J Ophthalmol 99:252–257
Laclette JP, Merchant MT, Willms K (1987) Histological and ultrastructural localization of antigen B in the metacestode of Taenia solium. J Parasitol 73:121–129
Lightowlers MW (2004) Vaccination for the prevention of cysticercosis. Dev Biol (Basel) 119:361–368
Madigubba S, Vishwanath K, Reddy G, Vemuganti GK (2007) Changing trends in ocular cysticercosis over two decades: an analysis of 118 surgically excised cysts. Indian J Med Microbiol 25:214–219
Madrazo I, Garcia-Renteria JA, Sandoval M, Lopez Vega FJ (1983) Intraventricular cysticercosis. Neurosurgery 12:148–152
Margono SS, Subahar R, Hamid A, Wandra T, Sudewi SS, Sutisna P, Ito A (2001) Cysticercosis in Indonesia: epidemiological aspects. Southeast Asian J Trop Med Public Health 32(Suppl 2):79–84
Marquez JM, Arauz A (2012) Cerebrovascular complications of neurocysticercosis. Neurologist 18:17–22
McCormick GF (1985) Cysticercosis – review of 230 patients. Bull Clin Neurosci 50:76–101
Montano SM, Villaran MV, Ylquimiche L, Figueroa JJ, Rodriguez S, Bautista CT, Gonzalez AE, Tsang VC, Gilman RH, Garcia HH (2005) Neurocysticercosis: association between seizures, serology, and brain CT in rural Peru. Neurology 65:229–233
Ndimubanzi PC, Carabin H, Budke CM, Nguyen H, Qian YJ, Rainwater E, Dickey M, Reynolds S, Stoner JA (2010) A systematic review of the frequency of neurocyticercosis with a focus on people with epilepsy. PLoS Negl Trop Dis 4:e870
Ooi HK, Ho CM, Chung WC (2013) Historical overview of Taenia asiatica in Taiwan. Kor J Parasitol 51:31–36
Ostrosky-Zeichner L, Estanol B (1999) Immunopathogenesis of neurocysticercosis: is damage mediated by the host immune response? Int J Parasitol 29:649–650
Park YS, Lee JK, Kim JH, Park KC (2011) Cysticercosis of lumbar spine, mimicking spinal subarachnoid tumor. Spine J 11:e1–e5
Pawlowski Z, Murell KD (2000) Taeniasis and cysticercosis. In: Hui Y, Murrell KD, Nip WK, Stanfield P, Satter SA (eds) Foodborne diseases handbook. Marcel Dekker, New York, pp 217–227
Phiri IK, Dorny P, Gabriel S, Willingham AL 3rd, Speybroeck N, Vercruysse J (2002) The prevalence of porcine cysticercosis in Eastern and Southern provinces of Zambia. Vet Parasitol 108:31–39
Pondja A, Neves L, Mlangwa J, Afonso S, Fafetine J, Willingham AL 3rd, Thamsborg SM, Johansen MV (2012) Use of oxfendazole to control porcine cysticercosis in a high-endemic area of Mozambique. PLoS Negl Trop Dis 6:e1651
Prabhakaran V, Rajshekhar V, Murrell KD, Oommen A (2007) Conformation-sensitive immunoassays improve the serodiagnosis of solitary cysticercus granuloma in Indian patients. Trans R Soc Trop Med Hyg 101:570–577
Prasad A, Prasad KN, Gupta RK, Pradhan S (2009a) Increased expression of ICAM-1 among symptomatic neurocysticercosis. J Neuroimmunol 206:118–120
Prasad KN, Prasad A, Gupta RK, Nath K, Pradhan S, Tripathi M, Pandey CM (2009b) Neurocysticercosis in patients with active epilepsy from the pig farming community of Lucknow district, north India. Trans R Soc Trop Med Hyg 103:144–150
Rajshekhar V, Joshi DD, Doanh NQ, van De N, Xiaonong Z (2003) Taenia solium taeniosis/cysticercosis in Asia: epidemiology, impact and issues. Acta Trop 87:53–60
Rajshekhar V, Raghava MV, Prabhakaran V, Oommen A, Muliyil J (2006) Active epilepsy as an index of burden of neurocysticercosis in Vellore district, India. Neurology 67:2135–2139
Restrepo BI, Alvarez JI, Castano JA, Arias LF, Restrepo M, Trujillo J, Colegial CH, Teale JM (2001) Brain granulomas in neurocysticercosis patients are associated with a Th1 and Th2 profile. Infect Immun 69:4554–4560
Sawhney BB, Chopra JS, Banerji AK, Wahi PL (1976) Pseudohypertrophic myopathy in cysticerosis. Neurology 26:270–272
Schantz PM, Moore AC, Munoz JL, Hartman BJ, Schaefer JA, Aron AM, Persaud D, Sarti E, Wilson M, Flisser A (1992) Neurocysticercosis in an Orthodox Jewish community in New York City. N Engl J Med 327:692–695
Schwan EV, de Scally MP, van Rensburg CL, Durand DT (2002) Cerebral cysticercosis in a cat. J S Afr Vet Assoc 73:219–221
Shandera WX, White AC Jr, Chen JC, Diaz P, Armstrong R (1994) Neurocysticercosis in Houston, Texas. A report of 112 cases. Medicine (Baltimore) 73:37–52
Singh AK, Prasad KN, Prasad A, Tripathi M, Gupta RK, Husain N (2013) Immune responses to viable and degenerative metacestodes of Taenia solium in naturally infected swine. Int J Parasitol 43:1101–1107
Singh AK, Singh SK, Singh A, Gupta KK, Khatoon J, Prasad A, Rai RP, Gupta RK, Tripathi M, Husain N, Prasad KN (2015a) Immune response to Taenia solium cysticerci after anti-parasitic therapy. Int J Parasitol 45:749–759
Singh SK, Singh AK, Prasad KN, Singh A, Rai RP, Tripathi M, Gupta RK, Husain N (2015b) Expression of adhesion molecules, chemokines and matrix metallo- proteinases (MMPs) in viable and degenerating stage of Taenia solium metacestode in swine neurocysticercosis. Vet Parasitol 214:59–66
Singh SK, Prasad KN, Singh AK, Gupta KK, Chauhan RS, Singh A, Rai RP, Pati BK (2016) Identification of species and genetic variation in Taenia isolates from human and swine of North India. Parasitol Res 115:3689–3693
Singh A, Prasad KN, Singh AK, Singh SK, Gupta KK, Paliwal VK, Pandey CM, Gupta RK (2017) Human glutathione s-transferase enzyme gene polymorphisms and their association with neurocysticercosis. Mol Neurobiol 54:2843–2851
Sinha S, Sharma BS (2012) Intraventricular neurocysticercosis: a review of current status and management issues. Br J Neurosurg 26:305–309
Sotelo J, Guerrero V, Rubio F (1985) Neurocysticercosis: a new classification based on active and inactive forms. A study of 753 cases. Arch Intern Med 145:442–445
Sotelo J, Escobedo F, Penagos P (1988) Albendazole vs praziquantel for therapy for neurocysticercosis. A controlled trial. Arch Neurol 45:532–534
Suastegui Roman RA, Soto-Hernandez JL, Sotelo J (1996) Effects of prednisone on ventriculoperitoneal shunt function in hydrocephalus secondary to cysticercosis: a preliminary study. J Neurosurg 84:629–633
Takayanagui OM, Jardim E (1992) Therapy for neurocysticercosis. Comparison between albendazole and praziquantel. Arch Neurol 49:290–294
Terrazas LI (2008) The complex role of pro- and anti-inflammatory cytokines in cysticercosis: immunological lessons from experimental and natural hosts. Curr Top Med Chem 8:383–392
Tsang VC, Brand JA, Boyer AE (1989) An enzyme-linked immunoelectrotransfer blot assay and glycoprotein antigens for diagnosing human cysticercosis (Taenia solium). J Infect Dis 159:50–59
Verma A, Prasad KN, Nyati KK, Singh SK, Singh AK, Paliwal VK, Gupta RK (2011) Association of MMP-2 and MMP-9 with clinical outcome of neurocysticercosis. Parasitology 138:1423–1428
White AC Jr, Tato P, Molinari JL (1992) Host-parasite interactions in Taenia solium cysticercosis. Infect Agents Dis 1:185–193
White AC Jr, Robinson P, Kuhn R (1997) Taenia solium cysticercosis: host-parasite interactions and the immune response. Chem Immunol 66:209–230
Yamasaki H, Allan JC, Sato MO, Nakao M, Sako Y, Nakaya K, Qiu D, Mamuti W, Craig PS, Ito A (2004) DNA differential diagnosis of taeniasis and cysticercosis by multiplex PCR. J Clin Microbiol 42:548–553
Zini D, Farrell VJ, Wadee AA (1990) The relationship of antibody levels to the clinical spectrum of human neurocysticercosis. J Neurol Neurosurg Psychiatry 53:656–661
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Prasad, K.N., Singh, S.K. (2018). Taeniasis and Neurocysticercosis: Emerging Public Health Problems. In: Singh, P. (eds) Infectious Diseases and Your Health. Springer, Singapore. https://doi.org/10.1007/978-981-13-1577-0_7
Download citation
DOI: https://doi.org/10.1007/978-981-13-1577-0_7
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-13-1576-3
Online ISBN: 978-981-13-1577-0
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)