Abstract
EUS is an important procedure for the differential diagnosis of type 1 autoimmune pancreatitis (AIP) from other pancreatic diseases because IgG4-SC is often associated with type 1 AIP. Bile duct wall thickening is considered the EUS finding of IgG4-SC with type 1 AIP. Cholangiocarcinoma (CCA) and primary sclerosing cholangitis (PSC) are important diagnoses that should be differentiated from IgG4-SC. IDUS findings of non-stricture bile duct lesions are different between IgG4-SC and CCA. The thickening of the bile duct wall in non-stricture lesion is the most characteristic IDUS finding of IgG4-SC. The IDUS findings of stricture lesions are also different between IgG4-SC and PSC. IDUS is a useful modality for differentiating IgG4-SC from CCA and PSC, as the details of bile duct wall thickening are easily observable.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Intraductal Ultrasonography (IDUS)
- IDUS Findings
- Bile Duct Wall Thickening
- IgG4-related Sclerosing Cholangitis (IgG4-SC)
- Autoimmune Pancreatitis (AIP)
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
Endoscopic Ultrasonography (EUS)
Endoscopic ultrasonography (EUS) is a reliable diagnostic procedure that provides high-resolution images of nearby organs. Radial scanning and linear array EUS are used for the evaluation of pancreatobiliary diseases. Advanced EUS imaging, including EUS elastography and contrast-enhanced EUS, can be more useful than conventional EUS, which has only B-mode imaging abilities. EUS elastography is an imaging modality used for the evaluation of tissue stiffness and is also used for the differential diagnosis of solid pancreatic masses. Contrast-enhanced EUS uses a contrast agent and Doppler mode for vascularization imaging. Endoscopic ultrasonography-guided fine-needle aspiration (FNA) is now widely accepted as a safe and effective modality for histological evaluation of pancreatic tissue because of its high diagnostic accuracy and low complication rate.
EUS Findings of IgG4-SC
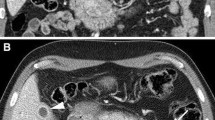
No study has described the detection of IgG4-related sclerosing cholangitis (IgG4-SC) by EUS; however, several studies have evaluated the diagnostic ability of EUS for type 1 autoimmune pancreatitis (AIP). These type 1 AIP findings are important for the diagnosis of IgG4-SC, as most cases of IgG4-SC are associated with type 1 AIP [1]. The characteristic EUS finding for the diagnosis of type 1 AIP is diffuse hypoechoic pancreatic enlargement, sometimes with hyperechoic inclusions. Hoki et al. [2] compared conventional EUS findings between AIP and pancreatic cancer and determined that the incidences of diffuse hypoechoic areas, diffuse enlargement, bile duct wall thickening, and peripancreatic hypoechoic margins are higher for AIP than for pancreatic cancer. Hyodo [3] also reported marked wall thickening in the intrapancreatic common bile duct in all patients. Bile duct wall thickening is considered the EUS finding of IgG4-SC with type 1 AIP (Fig. 11.1).
Using conventional EUS imaging alone, some type 1 AIP cases can be difficult to differentiate from pancreatic cancer. Dietrich et al. [4] reported that EUS elastography of type 1 AIP shows a characteristic stiff elastographic pattern in not only the mass lesion but also in the surrounding pancreatic parenchyma. Hocke et al. [5] reported that type 1 AIP pancreatic parenchyma and the surrounding pancreas show hypervascularization, whereas the lesions of pancreatic cancer are hypovascularized on contrast-enhanced EUS. Imazu et al. [6] used contrast-enhanced harmonic EUS to detect that the peak and maximum intensity gain of mass lesions in type 1 AIP were significantly higher than those of pancreatic cancer. Therefore, advanced EUS imaging techniques are useful tools for differentiating type 1 AIP from other pancreatic diseases.
Histological examination is critical for the definitive diagnosis of type 1 AIP. According to the International Consensus Diagnostic Criteria (ICDC), tissue samples obtained by EUS Trucut biopsy or surgical resection are considered suitable for the histopathological diagnosis of type 1 AIP, but EUS-FNA is not recommended, because it is difficult to obtain an adequate amount of tissue sample for histological evaluation. However, EUS-FNA using a 19-gauge needle has been reported to be useful for diagnosing type 1 AIP [7]. Kanno et al. [8] recently reported that EUS-FNA using a 22-gauge needle reliably provided a histological diagnosis of AIP, as determined by the ICDC, in 20 of 25 patients (80%). These findings suggest that EUS-FNA may provide new opportunities for the histological diagnosis of AIP.
The EUS finding of bile duct wall thickening suggests the presence of IgG4-SC with type 1 AIP. However, detailed images of bile duct wall thickening from EUS have not been reported. The coexistence of type 1 AIP is one of the diagnostic hallmarks for the ICDC diagnosis of IgG4-SC 2012 [9] and has a relatively high incidence. Therefore, the current role of EUS in the diagnosis of IgG4-SC is accurate diagnosis of type 1 AIP.
Intraductal Ultrasonography (IDUS)
After endoscopic retrograde cholangiopancreatography (ERCP), endoscopic transpapillary IDUS is another reliable procedure for the evaluation of bile duct wall thickening. IDUS is performed using a thin-caliber ultrasonic probe that consists of a sheath catheter, transducer, and cable. Biliary sphincterotomy is not necessary to perform this procedure using wire-guided IDUS. IDUS provides high-resolution images of the bile duct wall, which typically consists of inner hypoechoic and outer hyperechoic layers. With the development of optical technologies, IDUS can provide more detailed imagery of the bile duct, pancreatic duct, and adjacent structures. Bile duct wall thickness in patients with and without obstructive jaundice is typically reported to be less than 0.8 mm and 0.6 mm, respectively [10]. IDUS should be performed prior to biliary drainage because of mechanical inflammation that can occur following the procedure. IDUS is a useful modality for evaluation of bile duct stones, differential diagnosis of indeterminate biliary strictures, and superficial spread of bile duct cancer.
IDUS Findings of IgG4-SC
Characteristic biliary imaging findings are one of the four criteria for the clinical diagnosis of IgG4-SC 2012 [9]. Biliary tract imaging can reveal the characteristics associated with bile duct wall thickening, including diffuse or segmental narrowing of the intrahepatic and/or extrahepatic bile ducts. Computed tomography, magnetic resonance imaging, and EUS can also be used to detect bile duct wall thickness, but slight changes in thickness can be clearly observed using IDUS. Thus, IDUS is a useful modality for detailing bile duct wall thickening. IDUS findings of circular-symmetrical wall thickening, a smooth outer margin, a smooth inner margin, and homogeneous internal echo at the stenotic area are useful for the diagnosis of IgG4-SC (Fig. 11.2) [11,12,13,14,15,16]. The thickening of the bile duct wall, which appears normal on a cholangiogram, remains the most characteristic IDUS finding for the diagnosis of IgG4-SC. Bile duct wall thickening spreads continuously from the intrapancreatic bile duct to the upper bile duct in most IgG4-SC cases [11, 13, 17].
Endoscopic retrograde cholangiography (ERC) shows intrahepatic stricture in the patients with IgG4-SC. (a) Intraductal ultrasonography (IDUS) shows wall thickness in the hilar stricture. Symmetry is circular-symmetric. Outer margin is smooth. Inner margin is smooth. Internal echo is homogeneous. (b) IDUS shows wall thickness in the middle CBD in which cholangiogram is normal. (c) IDUS shows wall thickness in intrapancreatic bile duct in which cholangiogram is normal
Comparison of IDUS Findings Between IgG4-SC and Cholangiocarcinoma (CCA)
Type 3 IgG4-SC is characterized by stenosis in the hilar hepatic lesions and the lower common bile duct, and type 4 IgG4-SC presents with bile duct strictures in the hilar hepatic lesions only. The cholangiographic findings of types 3 and 4 IgG4-SC should be differentiated from those associated with CCA. IDUS findings are different between IgG4-SC and CCA in each stricture and non-stricture lesion of the bile duct. IDUS findings for IgG4-SC include circular-symmetric wall thickness, a smooth outer margin, a smooth inner margin, and a homogeneous internal echo in the biliary stricture. For a CCA diagnosis, however, IDUS findings may consist of asymmetric wall thickness, a notched outer margin, a non-smooth inner margin, and homogeneous internal echo in the biliary stricture (Fig. 11.2). The most characteristic IDUS finding of IgG4-SC is wall thickness in non-strictures of the bile duct, which appears normal on a cholangiogram. Wall thickness continuously spreads from the intrapancreatic bile duct to the upper bile duct in most IgG4-SC cases. On the contrary, bile duct wall thickness is not observed in the non-stricture site with CCA because cancer is not present there (Fig. 11.2). According to receiver operating characteristic curve analysis, a bile duct wall thickness of 0.8 mm is the optimal cutoff for differentiating IgG4-SC from CCA, as this thickness appears normal on a cholangiogram. The sensitivity, specificity, and accuracy of IgG4-SC diagnosis using a bile duct wall thickness cutoff of 0.8 mm were 95%, 90.9%, and 93.5%, respectively. Using a cutoff of 1 mm, the sensitivity, specificity, and accuracy were 85%, 100%, and 87%, respectively. No CCA cases have been noted to have a bile duct wall thickness greater than 1 mm; therefore, this cutoff can be used to exclude CCA from IgG4-SC diagnoses completely [11]. A comparison of IDUS findings between IgG4-SC and CCA is summarized in Fig. 11.3.
IDUS Findings of Primary Sclerosing Cholangitis (PSC)
PSC is a chronic cholestatic liver disease of unknown cause characterized by chronic inflammation and obliterative fibrosis of the bile ducts, which leads to diffuse biliary stenosis and increased wall thickness throughout the intra- and extrahepatic bile duct. Type 2 IgG4-SC should be differentiated from PSC, because biliary stenosis is diffusely distributed throughout the intra- and extrahepatic bile ducts in both. Typical IDUS findings related to PSC include circular-asymmetric wall thickness, an irregular inner margin, an unclear outer margin, diverticulum-like outpouching, heterogeneous internal echo, and the disappearance of three layers (Fig. 11.4) [12, 18]. Kubota et al. [12] also observed asymmetric wall thickness and an unpreserved outer margin in stenotic lesions of the bile duct in patients with PSC. The specific ERCP findings for PSC are diverticulum-like outpouching, which is similar to band-like stricture, and a beaded or pruned-tree appearance. Diverticulum-like outpouching is considered to be the most objective finding on ERCP, but it has the lowest sensitivity of the specific findings for PSC. However, the sensitivity for detecting diverticulum-like outpouching is higher for IDUS than for ERCP in patients with PSC [18]. Therefore, IDUS is more useful than ERCP for the early detection of diverticulum-like outpouching, which is specific to PSC.
ERC shows diverticulum-like outpouching in the patient with primary sclerosing cholangitis (PSC). (a) IDUS shows diverticulum-like outpouching (white arrow). Symmetry is circular-asymmetric. Inner margin is irregular. Outer margin is notched. Three layers were disappeared. Internal echo is heterogeneous, and internal foci are observed
Comparison of IDUS Findings Between IgG4-SC and PSC
The thickening of the bile duct wall that spreads continuously from the intrapancreatic bile duct to the upper bile duct is observed in IgG4-SC and PSC. The incidences of circular-asymmetric wall thickness, an irregular inner margin, diverticulum-like outpouching, an unclear outer margin, heterogeneous internal echo, and the disappearance of three layers are higher in PSC than in IgG4-SC [18]. An irregular inner margin, diverticulum-like outpouching, and disappearance of three layers were specific IDUS findings for PSC, as compared with IgG4-SC. Differences in IDUS findings between IgG4-SC and PSC clearly reflect pathological changes in the bile duct. Fibroinflammation is primarily observed in the stroma of the bile duct walls of patients with IgG4-SC, while the bile duct epithelium remains intact. The luminal side of the bile ducts, including the lining of biliary epithelial cells, is preferentially affected in patients with PSC. A comparison of the IDUS findings between IgG4-SC and PSC is summarized in Fig. 11.5.
Conclusions
EUS is important for the differential diagnosis of type 1 AIP from other pancreatic diseases and is useful for the diagnosis of IgG4-SC, which is often associated with type 1 AIP. CCA and PSC are important diagnoses that should be differentiated from IgG4-SC. IDUS findings of non-stricture bile duct lesions are different between IgG4-SC and CCA, and findings of stricture lesions vary between IgG4-SC and PSC. Therefore, IDUS is a useful modality for differentiating IgG4-SC from CCA and PSC, as the details of bile duct wall thickening are easily observable. Further EUS and IDUS improvements will ensure the appropriate and safe diagnosis of IgG4-SC.
References
Tanaka A, Tazuma S, Okazaki K, Nakazawa T, Inui K, Chiba T, et al. Clinical features, response to treatment, and outcomes of IgG4-related sclerosing cholangitis. Clin Gastroenterol Hepatol. 2017;15(6):920–6 e3.
Hoki N, Mizuno N, Sawaki A, Tajika M, Takayama R, Shimizu Y, et al. Diagnosis of autoimmune pancreatitis using endoscopic ultrasonography. J Gastroenterol. 2009;44(2):154–9.
Hyodo N, Hyodo T. Ultrasonographic evaluation in patients with autoimmune-related pancreatitis. J Gastroenterol. 2003;38(12):1155–61.
Dietrich CF, Hirche TO, Ott M, Ignee A. Real-time tissue elastography in the diagnosis of autoimmune pancreatitis. Endoscopy. 2009;41(8):718–20.
Hocke M, Ignee A, Dietrich CF. Contrast-enhanced endoscopic ultrasound in the diagnosis of autoimmune pancreatitis. Endoscopy. 2011;43(2):163–5.
Imazu H, Kanazawa K, Mori N, Ikeda K, Kakutani H, Sumiyama K, et al. Novel quantitative perfusion analysis with contrast-enhanced harmonic EUS for differentiation of autoimmune pancreatitis from pancreatic carcinoma. Scand J Gastroenterol. 2012;47(7):853–60.
Iwashita T, Yasuda I, Doi S, Ando N, Nakashima M, Adachi S, et al. Use of samples from endoscopic ultrasound-guided 19-gauge fine-needle aspiration in diagnosis of autoimmune pancreatitis. Clin Gastroenterol Hepatol. 2012;10(3):316–22.
Kanno A, Ishida K, Hamada S, Fujishima F, Unno J, Kume K, et al. Diagnosis of autoimmune pancreatitis by EUS-FNA by using a 22-gauge needle based on the International Consensus Diagnostic Criteria. Gastrointest Endosc. 2012;76(3):594–602.
Ohara H, Okazaki K, Tsubouchi H, Inui K, Kawa S, Kamisawa T, et al. Clinical diagnostic criteria of IgG4-related sclerosing cholangitis 2012. J Hepatobiliary Pancreat Sci. 2012;19(5):536–42.
Tamada K, Tomiyama T, Oohashi A, Aizawa T, Nishizono T, Wada S, et al. Bile duct wall thickness measured by intraductal US in patients who have not undergone previous biliary drainage. Gastrointest Endosc. 1999;49(2):199–203.
Naitoh I, Nakazawa T, Ohara H, Ando T, Hayashi K, Tanaka H, et al. Endoscopic transpapillary intraductal ultrasonography and biopsy in the diagnosis of IgG4-related sclerosing cholangitis. J Gastroenterol. 2009;44(11):1147–55.
Kubota K, Kato S, Uchiyama T, Watanabe S, Nozaki Y, Fujita K, et al. Discrimination between sclerosing cholangitis-associated autoimmune pancreatitis and primary sclerosing cholangitis, cancer using intraductal ultrasonography. Dig Endosc. 2011;23(1):10–6.
Moon SH, Kim MH. The role of endoscopy in the diagnosis of autoimmune pancreatitis. Gastrointest Endosc. 2012;76(3):645–56.
Tabata T, Kamisawa T, Hara S, Kuruma S, Chiba K, Kuwata G, et al. Differentiating immunoglobulin g4-related sclerosing cholangitis from hilar cholangiocarcinoma. Gut Liver. 2013;7(2):234–8.
Kamisawa T, Ohara H, Kim MH, Kanno A, Okazaki K, Fujita N. Role of endoscopy in the diagnosis of autoimmune pancreatitis and immunoglobulin G4-related sclerosing cholangitis. Dig Endosc. 2014;26(5):627–35.
Kanno A, Masamune A, Shimosegawa T. Endoscopic approaches for the diagnosis of autoimmune pancreatitis. Dig Endosc. 2015;27(2):250–8.
Hirano K, Tada M, Isayama H, Yamamoto K, Mizuno S, Yagioka H, et al. Endoscopic evaluation of factors contributing to intrapancreatic biliary stricture in autoimmune pancreatitis. Gastrointest Endosc. 2010;71(1):85–90.
Naitoh I, Nakazawa T, Hayashi K, Miyabe K, Shimizu S, Kondo H, et al. Comparison of intraductal ultrasonography findings between primary sclerosing cholangitis and IgG4-related sclerosing cholangitis. J Gastroenterol Hepatol. 2015;30(6):1104–9.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Springer Science+Business Media Singapore
About this chapter
Cite this chapter
Naitoh, I., Nakazawa, T., Ohara, H., Joh, T. (2019). Imaging: EUS and IDUS. In: Kamisawa, T., Kim, MH. (eds) IgG4-Related Sclerosing Cholangitis. Springer, Singapore. https://doi.org/10.1007/978-981-10-4548-6_11
Download citation
DOI: https://doi.org/10.1007/978-981-10-4548-6_11
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-10-4547-9
Online ISBN: 978-981-10-4548-6
eBook Packages: MedicineMedicine (R0)