Abstract
Carl Langenbuch first reported the procedure of cholecystectomy in 1882. He used an open approach then which remained unchanged for more than 100 years (Bittner, Langenbecks Arch Surg 389:157–163, 2004). In 1987, Philippe Mouret performed the first video-assisted laparoscopic cholecystectomy (LC) in Lyon, France (Kaiser and Corman, Surg Oncol Clin N Am 10:483–492, 2001). LC was a significant advancement, removing many of the objections that patients had to go under open cholecystectomy. It is now considered world-wide as the “golden standard” because of its unquestionable advantages in terms of smaller incision, reduced postoperative pain, shorter hospital stay, and faster return to normal activities when compared to the traditional open approach (Soper et al., Arch Surg 127:917–921, 1992; Schirmer et al., Ann Surg 213:665–676, 1991; Johansson et al., Br J Surg 92:44–49, 2005). Laparoendoscopic single-site cholecystectomy (SILSC), which emerged as a form of natural orifice surgery, was first described in 1995 (Navarra et al., Br J Surg 84:695, 1997). Hypothetically, the benefits of laparoendoscopic single-site surgery (SILS) would include those of the conventional multiport laparoscopic procedures in association with improved cosmetic results and patient satisfaction as the operation is performed through just one incision.
The application of SILSC is in its infancy, but gaining significant momentum. Experience with single incision laparoscopic technique, is growing rapidly. With advancements in laparoscopic techniques and the development of new instruments, more complex operations are being approached laparoscopically, including resection of malignant intestinal tumors (Lai et al., Arch Surg 144:143–147, 2009; Adani et al., Surgery 145:452, 2009; Sarpel et al., Ann Surg Oncol 16:1572–1577, 2009; Choh and Madura, Surg Clin North Am 89:53–77, 2009). Laparoscopic biliary-enteric anastomosis has been reported for benign biliary tract disease (O’Rourke et al., Am J Surg 187:621–624, 2004; Chokshi et al., J Laparoendosc Adv Surg Tech A 19:87–91, 2009). All of these have made it possible to perform laparoscopic bile duct exploration, bile duct reconstruction, biliary bypass, and resection of congenital choledochal cyst and high cholangiocarcinomas, all through a single incision. Besides that, some complex hepatic procedures such as liver resection has been reported using the SILS technique (Binenbaum et al., Arch Surg 144:734–738, 2009; Hodgett et al., J Gastrointest Surg 13:188–192, 2009; Hong et al., J Laparoendosc Adv Surg Tech A 19:75–78, 2009; Fagotti et al., Fertil Steril 92:1168.e13–1168.e16, 2009; Fader and Escobar, Gynecol Oncol 114:157–161, 2009).
Evidence to support routine use of single incision laparoscopic hepatobiliary surgery is still lacking, but this approach can be considered an alternative and may be applied in a selected group of patients. Despite the rapid uptake of SILS techniques, it has become increasingly clear that there still remain issues that need to be addressed; among which, safety is of utmost importance. SILS hepatobiliary surgery requires a unique technical skill set, with ergonomic and technical demands that are different from conventional laparoscopic skills, exhibiting a learning curve even for expert laparoscopic surgeons. Only through a fully implemented system of proficiency-based progression can competent laparoscopic surgeons be ensured to further advance the technique. It is the intention of this section to detail our current techniques for SILS hepatobiliary surgery.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Cholecystectomy
- Calot’s triangle
- Common bile duct exploration
- Choledochoscope
- J tube
- Cholecystojejunostomy
- Ligament of Treitz
- Jejunojejunostomy
- Cholangiojejunostomy
- Hepatoduodenal ligament
- Roux-en-Y conduit
- Choledochalcystectomy
- Hepaticojejunostomy
- Interposition of the Ileum
- Liver Cyst Fenestration
- Partial Hepatectomy
5.1 Laparoscopic Cholecystectomy
5.1.1 Indications and Case Selection
-
1.
Patients generally have documented cholelithiasis and symptoms attributable to a diseased gallbladder, including cholecystolithiasis, polypoid lesions of gallbladder, chronic cholecystitis, acute cholecystitis with symptomatic relief after treatment, etc.
-
2.
Prophylactic laparoscopic cholecystectomy for asymptomatic cholelithiasis may be justified for certain patients, such as patients complicated with diabetic mellitus, cardiorespiratory dysfunction in stable phase, Sickle cell disease, total parenteral nutrition, chronic immunosuppression, no immediate access to health care facilities (e.g., missionaries, military personnel, Peace Corps workers, relief workers), incidental cholecystectomy for patients undergoing laparoscopic procedure for other indications.
-
3.
Other indications for laparoscopic cholecystectomy include acalculous cholecystitis (e.g., biliary dyskinesia), gallstone pancreatitis, gallbladder polyps greater than 1 cm in diameter and porcelain gallbladder.
5.1.2 Contraindications
-
1.
Absolute contraindications to performing laparoscopic cholecystectomy include the inability to tolerate general anesthesia or pneumoperitoneum, suspicion of carcinoma, refractory coagulopathy.
-
2.
Relative contraindications to performing laparoscopic cholecystectomy include advanced acute cholecystitis, Mirizzi syndrome, previous upper abdominal surgery, cholangitis, diffuse peritonitis, Cirrhosis or portal hypertension, chronic obstructive pulmonary disease, cholecystenteric fistula, morbid obesity, pregnancy (during first and third trimesters).
5.1.3 Major Instruments or Energy Sources
-
1.
Laparoscopy System.
-
2.
Holding forceps.
-
3.
Harmonic scalpel.
5.1.4 Team Setup, Anesthesia and Position
Under general anesthesia, the patient was placed in supine position with the arms adducted along the side. The position of surgeon, assistants and nurses is shown in Fig. 5.1.

Fig. 5.1
5.1.5 Key Steps
-
1.
Establishment of Pneumoperitoneum and Placement of Trocars.
-
2.
Mobilizing the inferior margin of the gallbladder.
-
3.
Mobilizing the superior margin of the gallbladder.
-
4.
Mobilizing the cystic duct and artery.
-
5.
Ligating and cutting off the cystic duct.
-
6.
Taking out gallbladder.
5.1.6 Surgical Techniques
-
1.
Establishment of Pneumoperitoneum and Placement of Trocars.
-
2.
Mobilizing the inferior margin of the gallbladder.
To expose the inferior margin of the gallbladder, the body of the gallbladder was lifted superiorly and medially by a grasper in the surgeon’s left hand. The serosal layer of the inferior margin of the gallbladder was cut open by a harmonic scalpel in the surgeon’s right hand along the borderline between the gallbladder and the liver. A loose connective tissue layer could be found and dissected between the hepatic plate and the gallbladder. Mobilization was performed as much as possible towards the superior margin through the loose connective tissue layer behind the gallbladder from the neck to the fundus (Fig. 5.2a–i).


Fig. 5.2
-
3.
Mobilizing the superior margin of the gallbladder.
To expose the superior margin of the gallbladder, the margin of liver was pushed back by the grasper in the left hand and the fundus of the gallbladder was released drooping freely. The serosal layer of the superior margin of the gallbladder was cut open by the harmonic scalpel in the right hand from the fundus to the neck until reaching the boundary of Calot’s triangle. The mobilization was performed also through the loose connective tissue layer between the hepatic plate and gallbladder until meeting the space previously dissected in the previous step (Fig. 5.3a, b).

Fig. 5.3
-
4.
Mobilizing the cystic duct and artery.
After previous mobilization, the body of gallbladder was freed completely from liver. To expose the cystic duct, the body of gallbladder was vertically pulled upward by grasper in the left hand. And the lateral region of cystic duct was dissected by the harmonic scalpel. Then, the gallbladder was pulled laterally and downward to expose the medial region of cystic duct. In this region, cystic artery could be found and dissected by the harmonic scalpel. In this step, much attention must be paid to avoid bile duct injury. When the gallbladder was pulled upward, the cystic duct and bile duct were in a shape of inverted “T”. The hepatic duct may be pulled up sometimes. In this situation, the bile duct is prone to be injured. The lateral region and medial region may need to be dissected alternately according to specific circumstance and the arrangement of the three instruments should shift accordingly (Fig. 5.4a–n).



Fig. 5.4
-
5.
Ligating and cutting off the cystic duct.
In order to ligate the cystic duct, a 10 mm hemolock forceps was needed to go through the 10 mm trocar. The 10 mm laparoscope and 5 mm harmonic scalpel were removed and a 5 mm laparoscope was inserted through the 5 mm trocar that was used for the harmonic scalpel in previous steps. The hemolock forceps was inserted through the 10 mm trocar. The gallbladder was pulled upward by the grasper in the left hand to expose the cystic duct. The cystic duct was double ligated by two hemolock clips. When the hemolock forceps was replaced by a harmonic scalpel to cut off the cystic duct through the 10 mm trocar, the scalpel went alone the same track as the hemolock forceps (Fig. 5.5a–f).

Fig. 5.5
-
6.
Taking out gallbladder.
A 10 mm specimen forceps was inserted through the 10 mm trocar to grasp the end of the cystic duct. Under the monitoring of 5 mm laparoscope from above, the gallbladder was pulled towards the incision. The 10 mm trocar was removed and the opening end of cystic duct was pulled out of the incision. Bile and stones were cleared under direct vision and then the gallbladder was taken out easily. Three trocars were reinserted and routine inspection and washing were performed before suturing the incision with absorbable stitches (Fig. 5.6a, b).
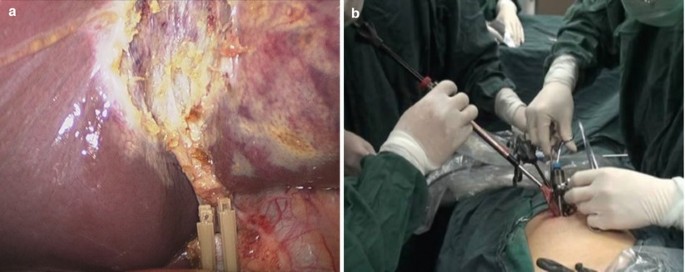
Fig. 5.6
5.1.7 Tips and Tricks
-
1.
The patient is placed in the reversed Trendelenberg position with right side elevated like the conventional laparoscopic cholecystectomy. The operating surgeon and the assistant stands on the left side.
-
2.
The grasper introduced through the right trocar is used to retract the gallbladder up and to the left. The dissection of the gallbladder from the liver bed using harmonic scalpel should start from the right edge of the gallbladder.
-
3.
The cystic duct and the artery are isolated. The segment five of the liver is seen in the window created, with the cystic duct seen as the only structure entering the gallbladder (critical view). The cystic duct is left intact as it serves as an additional point of fixation.
5.2 Laparoscopic Common Bile Duct Exploration
5.2.1 Indications and Case Selection
-
1.
Patients with primary, or secondary extrahepatic bile duct stones complicated with cholecystolithiasis, which should be: (1) Large stones (>10 mm). (2) Multiple stones. (3) Stone location proximal to the cystic duct/common bile duct junction. (4) Narrow/tortuous cystic duct, etc. And it should be with dilated common bile duct (6–10 mm).
-
2.
Failed laparoscopic transcystic exploration or preoperative endoscopic stone extraction.
-
3.
Biliary ascariasis.
5.2.2 Contraindications
-
1.
Inability to tolerate general anesthesia or pneumoperitoneum.
-
2.
Suspicion of carcinoma.
-
3.
Refractory coagulopathy.
-
4.
Residual, or regenerated stone after biliary surgery.
-
5.
Severe adhesion around the bile ducts.
-
6.
Stenosis of bile duct.
-
7.
Intrahepatic bile duct stone, and hepatectomy is considered.
5.2.3 Major Instruments or Energy Sources
-
1.
Laparoscopy System.
-
2.
Holding forceps.
-
3.
Harmonic scalpel.
-
4.
Cholangioscope.
5.2.4 Team Setup, Anesthesia and Position
It is the same as Sect. 5.1.4.
5.2.5 Key Steps
-
1.
Dissection of gallbladder.
-
2.
Incision of common bile duct and removal of the stones.
-
3.
Choledocoscopy exploration and removal of the stones.
-
4.
Placement of J tube.
-
5.
Closure of the common bile duct.
-
6.
Resection of gallbladder.
5.2.6 Surgical Techniques
-
1.
Dissection of gallbladder.
Dissection of gallbladder was performed as described in the cholecystectomy section, with the cyst duct clipped but uncut.
-
2.
Incision of common bile duct and removal of the stones.
The gallbladder was maintained in position, which helped to expose the common bile duct (CBD). After CBD and its diameter were clearly identified, a longitudinal incision of 1–1.5 cm was then made over the anterior wall. Once the duct was opened, nearby stones were grasped and removed directly with grasper or Semm’s spoon forceps (Fig. 5.7a–f).

Fig. 5.7
-
3.
Choledocoscopy exploration and removal of the stones.
A flexible choledochoscope was then passed through the 5-mm port to perform visually guided extraction of any other residual stones. The superior biliary tract and the inferior biliary tract were explored in turn. When a stone was located, a Dormia retrieval basket was used to extract it (Fig. 5.8a–d).

Fig. 5.8
-
4.
Placement of J tube.
Once clearance of CBD stones was confirmed and no presence of edema or inflammation was observed, a guidewire was passed through the instrument channel of the choledochoscope and advanced through the ampulla of Vater into the duodenum, after which the scope was removed, leaving the guidewire in place. A J-tube was then threaded along the guidewire into the CBD, through the ampulla of Vater, and into the duodenum. With one end of the J-tube in the lumen of the duodenum, the guidewire was removed and the J-tube was retained in the CBD. A completion check choledochoscopy was performed to ensure exact positioning of the J-tube before primary choledochorrhaphy (Fig. 5.9a–f).

Fig. 5.9
-
5.
Closure of the common bile duct.
Primary closure of the choledochotomy was performed longitudinally using absorbable 3/0 Vicryl suture(s). If the diameter of CBD was less than 1.5 cm, interrupted suture would work (Fig. 5.10a–f).

Fig. 5.10
-
6.
Resection of gallbladder.
Removal of gallbladder was performed as described in the cholecystectomy section. A drain was pulled in through the incision and placed in the foramen of Winslow (Fig. 5.11).

Fig. 5.11
5.2.7 Tips and Tricks
-
1.
The ENBD, ERBD or J-type tube was introduced into the common bile duct preoperatively or intraoperatively for T-tube-free laparoscopic choledocholithtomy.
-
2.
ENBD and ERBD drainage: A 7Fr ENBD tube (Cook, Winston-Salem, NC, USA) or an 8.5Fr 5-cm ERBD tube (Cook, Winston-Salem, NC, USA) was placed preoperatively in the CBD of patient without sphincterotomy. The ERBD tubes were commonly removed by endoscopy on postoperative week 2. Cholangiography was scheduled to confirm the absence of residual stones in the biliary tract before the ENBD tubes were removed.
-
3.
Pigtail J-tube drainage: The use of the pigtail J-tube is ideal for patients with large single or multiple stones primarily in the CBD. Once clearance of CBD stones is confirmed, a pigtail J-tube can be employed as an internal drainage/stent for biliary tree decompression in the presence of distal ductal edema or spasm. A guidewire was passed through the instrument channel of the choledochoscope and advanced through the ampulla of Vater into the duodenum, after which the scope was removed, leaving the guidewire in place. A pigtail J-tube (HuaMei, Zhangjiagang, Jiangsu, China) was then threaded along the guidewire into the CBD, through the ampulla of Vater, and into the duodenum. With one end of the J-tube in the lumen of the duodenum, the guidewire was removed and the J-tube was retained in the CBD. This was facilitated by the coils on both ends of the J-tube, which provided a self-retaining mechanism. A completion check choledochoscopy was performed to ensure exact positioning of the J-tube before primary choledochorrhaphy with interrupted or continuous 3/0 absorbable Vicryl suture(s). The position of the pigtail J-tube was further confirmed with fluoroscopy before the J-tube was removed and the patient was discharged.
-
4.
No biliary drainage: We performed primary choledochorrhaphy with interrupted 3/0 absorbable Vicryl suture leaving no biliary drainage for the patients who had absence of distal ductal edema or spasm during the completion check choledochoscopy.
5.3 Laparoscopic Cholecystojejunostomy
5.3.1 Indications and Case Selection
-
1.
Obstruction in the inferior segment of common bile duct, e.g. radical resection is improper for carcinoma of head of pancreas in later period.
-
2.
Staging operation for carcinoma of head of pancreas.
-
3.
Malignant obstruction in the inferior segment of common bile duct, in which document dilated cystic duct is identified.
5.3.2 Contraindications
-
1.
Patients with obstruction in cystic duct due to malignant change in later period.
-
2.
Patients with low insertion of cystic duct into the common hepatic duct, which is easily obstructed by tumor.
-
3.
Patients with cholangiocarcinoma in the middle or superior segment.
-
4.
Patients with gallbladder stone or common bile duct stone.
5.3.3 Major Instruments or Energy Sources
-
1.
Laparoscopy System.
-
2.
Holding forceps.
-
3.
Harmonic scalpel.
-
4.
Endoscopic linear stapler.
5.3.4 Team Setup, Anesthesia and Position
Under general anesthesia, the patient was placed in supine position with the legs apart. The position of surgeon, assistants and nurses is shown in Fig. 5.12.

Fig. 5.12
5.3.5 Key Steps
-
1.
Exploration of the lesion.
-
2.
Dissection of upper jejunum.
-
3.
Anastomosis of gallbladder and jejunum.
-
4.
Side-to-side jejunojejunostomy.
-
5.
Irrigation and drainage.
5.3.6 Surgical Techniques
-
1.
Exploration of the lesion.
The procedure began with a general exploration of the abdomen, particularly to confirm the radiological patent hepatocystic junction.
-
2.
Dissection of upper jejunum.
First, an upper jejunal loop (the first loop) was identified by a descending (ligament of Treitz) exploration of the small intestine. A suitable loop (45–50 cm) distally to the ligament of Trietz that could easily and comfortably reach the sub-hepatic space was passed in an anterior colic manner without tension and approximated to the gallbladder (Fig. 5.13a–h).


Fig. 5.13
-
3.
Anastomosis of gallbladder and jejunum.
A small incision was made in the body of the gallbladder, and the bile evacuated (Fig. 5.14a, b).

Fig. 5.14
A similar incision was made on the predesigned anti-mesenteric part of the approximated jejunum. The jaws of the Endo-GIA stapler was introduced through these two incisions and pushed to its full length, closed, and fired (Fig. 5.15a–d).

Fig. 5.15
The two entry sites were combined as one. They were closed by using 3/0 absorbable Vicryl sutures (Fig. 5.16a–f).

Fig. 5.16
-
4.
Side-to-side jejunojejunostomy.
The patient was then placed in the position with a 15° tile to the right. The anti-mesenteric part of the first and second (45–50 cm distally to the anastomosis) loops of jejunum were pulled together for enteroenterostomy (Fig. 5.17a, b).

Fig. 5.17
A 0.5 cm incision was separately made on both anti-mesenteric part of the jejunum (Fig. 5.18a, b).

Fig. 5.18
The jaws of the Endo-GIA stapler was introduced through these two incisions and pushed to its full length, closed, and fired. Making sure there was no mesenteric volvulus (Fig. 5.19a–d).

Fig. 5.19
The common opening left behind after the removal of the stapler was closed by 3/0 absorbable Vicryl sutures (Fig. 5.20a–h).


Fig. 5.20
-
5.
Irrigation and drainage.
After adequate flushing of the surgical field, a drain was placed behind the anastomosis of cholecystojejunostomy through the umbilicus (Fig. 5.21a–d).

Fig. 5.21
5.3.7 Tips and Tricks
-
1.
The patency of the hepatocystic junction, and the junction of the cystic duct with the common hepatic duct was more than 1 cm away from the proximal extent of the tumors should be confirmed.
-
2.
Two percutaneous transfascial retraction sutures (3/0 Prolene. Ethicon, US) could be placed at the right and left costal margins to achieve adequate exposure not only to facilitate anastomosis but also throughout the whole procedure. One suture was placed at one end of the anastomotic line between the fundus of the gallbladder and intestine, and the other was at one end of the anastomotic line between the two segments of intestine
5.4 Laparoscopic Cholangiojejunostomy
5.4.1 Indications and Case Selection
-
1.
Benign stenosis of extrahepatic bile duct.
-
2.
Striction in the terminal of common bile duct.
-
3.
Paraduodenal papilla diverticulum, such causing recurrent pancreatitis and cholangitis.
-
4.
Congenital anomalies of biliary system, e.g. reconstruction of biliary tract after cyst resection for congenital cystic dilatation of common bile duct.
-
5.
Biliary alimentary anastomotic stoma striction.
-
6.
Carcinoma of gallbladder or head of pancreas, which is difficult to resect.
-
7.
Carcinoma of the middle segment of bile duct.
5.4.2 Contraindications
Patients with intrahepatic bile duct striction or stone should be the contraindicated to perform laparoscopic cholangiojejunostomy.
5.4.3 Major Instruments or Energy Sources
-
1.
Laparoscopy System.
-
2.
Holding forceps.
-
3.
Harmonic scalpel.
-
4.
Endoscopic linear stapler.
5.4.4 Team Setup, Anesthesia and Position
Under general anesthesia, the patient was placed in supine position with the legs apart. The position of surgeon, assistants and nurses is shown in Fig. 5.22.

Fig. 5.22
5.4.5 Key Steps
-
1.
Dissection of gallbladder.
-
2.
Skeletonization of hepatoduodenal ligament.
-
3.
Transection of common bile duct.
-
4.
Transection of common hepatic duct.
-
5.
Dissection of jejunum.
-
6.
Cholangiojejunostomy.
-
7.
Jejunojejunostomy.
-
8.
Specimen retraction.
-
9.
Irrigation and drainage.
5.4.6 Surgical Techniques
-
1.
Dissection of gallbladder.
The gallbladder was removed from the liver bed without transecting the cystic duct (Fig. 5.23a, b).

Fig. 5.23
-
2.
Skeletonization of hepatoduodenal ligament.
A percutaneous transfascial retraction suture was placed below the xiphoid process to achieve adequate exposure. Lymph nodes and connective tissues lateral and posterior to the bile duct were mobilized using the 5-mm harmonic scalpel, leading to complete skeletonization of the proper hepatic artery, portal vein and common bile duct (CBD) (Fig. 5.24a–r).



Fig. 5.24
-
3.
Transection of common bile duct.
The CBD was transected at the superior edge of duodenum (2 cm distally to the inferior margin of tumor) (Fig. 5.25a–h).


Fig. 5.25
-
4.
Transection of common hepatic duct.
Dissection was next extended further in the cephalad direction, and the common hepatic duct or extrahepatic right and left bile ducts were divided at least 2 cm away from the superior margin of the tumor. The surgical specimen was removed and included the distal common hepatic duct, the common bile duct with the attached gallbladder, and the regional node-bearing tissue (Fig. 5.26a–i).

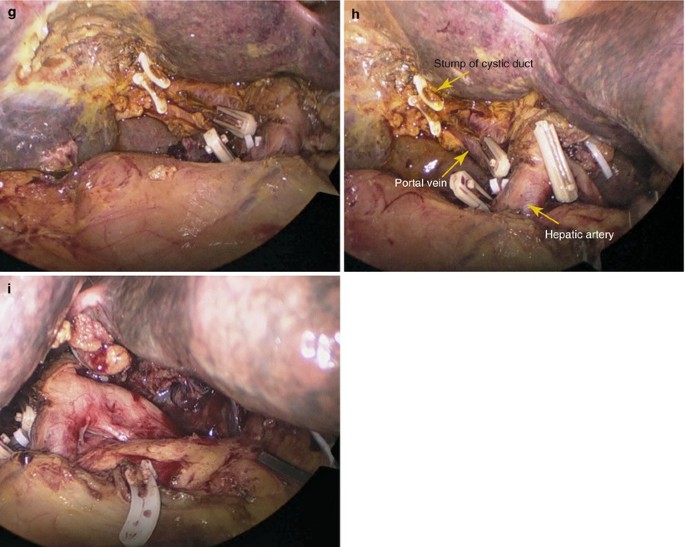
Fig. 5.26
-
5.
Dissection of jejunum.
The first jejunal loop 10–15 cm distally to the ligament of Treitz that comfortably reached the sub-hepatic space was used to create a Roux-en-Y conduit. It was divided using the GIA stapler (Fig. 5.27a–e).

Fig. 5.27
-
6.
Cholangiojejunostomy.
The distal part of the jejunal loop was brought through a window in the transverse mesocolon toward the severed common duct. The end-to-side choledochojejunal anastomosis was constructed on the anti-mesenteric part of the loop (5 cm to the end) by 3/0 absorbable Vicryl suture (Fig. 5.28a–n).



Fig. 5.28
-
7.
Jejunojejunostomy.
The proximal afferent loop was sutured in a side-to-side/end-to-side manner to the ascending limb of jejunum (45 cm distal to the choledochojejunal anastomosis) extracorporeally. Once the jejunojejunostomy was completed, the intestine was sent into the abdominal cavity and pneumoperitoneum was then reestablished (Fig. 5.29a–c).

Fig. 5.29
-
8.
Specimen retraction.
The specimen bag was retracted through the umbilical incision (Fig. 5.30a, b).
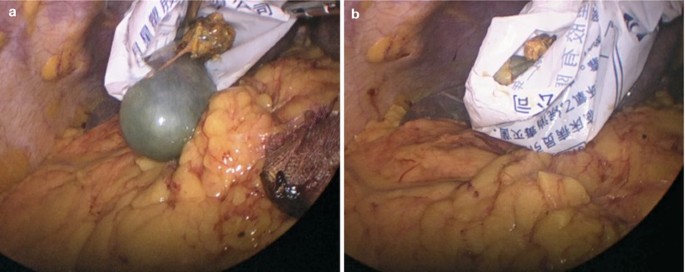
Fig. 5.30
-
9.
Irrigation and drainage.
After adequate flushing the surgical field, a sub-hepatic drain was placed close to the choledochojejunal anastomosis through the umbilicus (Fig. 5.31a–d).

Fig. 5.31
5.5 Laparoscopic Choledochalcystectomy with Jejunal Roux-en-Y Reconstruction
5.5.1 Indications and Case Selection
-
1.
Type I and II choledochalcyst.
-
2.
Relapse of symptoms or complications after cyst internal drainage.
-
3.
Highly suspicious of malignant change of cyst.
5.5.2 Contraindications
-
1.
Any contraindications to open liver resection.
-
2.
Patients who cannot tolerate pneumoperitoneum or general anesthesia.
-
3.
Type III, IV and V choledochalcyst.
5.5.3 Major Instruments or Energy Sources
-
1.
Laparoscopy System.
-
2.
Holding forceps.
-
3.
Harmonic scalpel.
-
4.
Endoscopic linear stapler.
5.5.4 Team Setup, Anesthesia and Position
Under general anesthesia, the patient was placed in supine position with the legs apart. The position of surgeon, assistants and nurses is shown in Fig. 5.32.

Fig. 5.32
5.5.5 Key Steps
-
1.
Dissection of gallbladder.
-
2.
Dissection of cyst.
-
3.
Hepaticojejunostomy and jejunojejunostomy.
5.5.6 Surgical Techniques
-
1.
Dissection of gallbladder.
Dissection of gallbladder was performed as described in the cholecystectomy section (Fig. 5.33a–j).

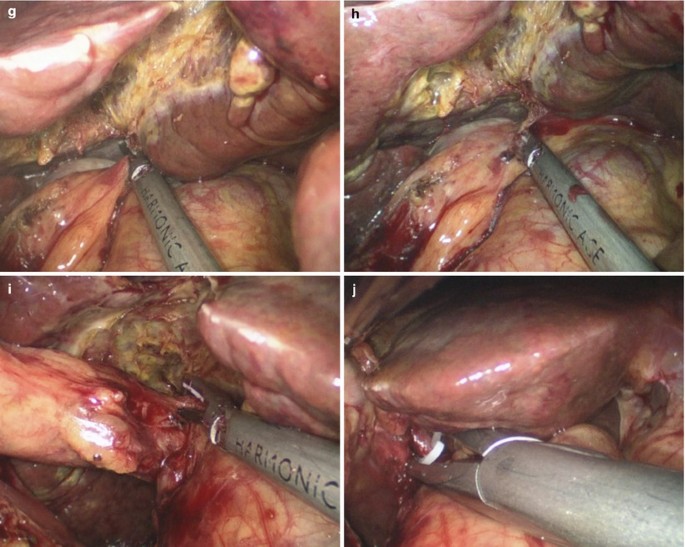
Fig. 5.33
-
2.
Dissection of cyst.
Placement of stay suture below the xiphoid process was performed as described in the fourth section of this chapter. CBD was verified by a puncture in its anterior surface. After confirmation, the hepatic common bile (HCB) was transected, and then the distal portion of HCB was pulled downward. The dissection was extended along the cyst. After confirming the transitional area of choledochal cyst at the head of the pancreas, the CBD was transected distal to the cyst, and the cyst was then completely dissected (Fig. 5.34a–q).



Fig. 5.34
-
3.
Hepaticojejunostomy and jejunojejunostomy.
Hepaticojejunostomy and jejunojejunostomy (Roux-en-Y reconstruction) was performed as described in the fourth section of this chapter (Fig. 5.35a–h).


Fig. 5.35
5.6 Laparoscopic Cholecystojejunostomy with Interposition of the Ileum
5.6.1 Indications and Case Selection
-
1.
Obstruction in the inferior segment of common bile duct, e.g. radical resection is improper for carcinoma of head of pancreas in later period.
-
2.
Staging operation for carcinoma of head of pancreas.
-
3.
Malignant obstruction in the inferior segment of common bile duct, in which document dilated cystic duct is identified.
5.6.2 Contraindications
-
1.
Patients with obstruction in cystic duct due to malignant change in later period.
-
2.
Patients with low insertion of cystic duct into the common hepatic duct, which is easily obstructed by tumor.
-
3.
Patients with cholangiocarcinoma in the middle or superior segment.
-
4.
Patients with gallbladder stone or common bile duct stone.
5.6.3 Major Instruments or Energy Sources
-
1.
Laparoscopy System.
-
2.
Holding forceps.
-
3.
Harmonic scalpel.
-
4.
Endoscopic linear stapler.
5.6.4 Team Setup, Anesthesia and Position
It is the same as Sect. 5.5.4.
5.6.5 Key Steps
-
1.
Exploration of lesion.
-
2.
Dissection of terminal ileum.
-
3.
Creation of an ileal segment with its mesentery pedicle.
-
4.
Anastomosis between gallbladder and ileal segment.
-
5.
Jejuno-ileostomy.
5.6.6 Surgical Techniques
-
1.
Exploration of lesion.
Exploration was performed as described in section three (Fig. 5.36).

Fig. 5.36
-
2.
Dissection of terminal ileum.
A segment of ileum was divided between 15 and 60 cm from the ileocecal junction (Fig. 5.37a–d).

Fig. 5.37
-
3.
Creation of an ileal segment with its mesentery pedicle.
A distal ileal segment of 45 cm was transected along with its mesentery pedicle by using Endo-GIA (Fig. 5.38a–n).



Fig. 5.38
-
4.
Anastomosis between gallbladder and ileal segment.
The ileal segment was pulled upward and side-to-side anastomosis between the gallbladder and proximal ileal segment was performed as described in the cholecystojejunostomy section (Fig. 5.39a–u).




Fig. 5.39
-
5.
Jejuno-ileostomy.
The distal end of transected ileal segment was interposed and anastomosed to the upper jejunal loop (15 cm from the ligament of Treitz) in a side-to-side way as described in the cholecystojejunostomy section. Continuity of small bowel was restored extracorporally by end-to-end ileo-ileal anastomosis (Fig. 5.40a–f).

Fig. 5.40
5.7 Laparoscopic Liver Cyst Fenestration
5.7.1 Indications and Case Selection
-
1.
Patients with symptomatic single cyst with the diameter larger than 5 cm, which is located in the liver superficially.
-
2.
Patients with a limited number of large and superficial cysts that can be individually treated as single cysts to establish a large communication between the cyst and the peritoneal cavity.
-
3.
Patients with cysts treated by alcohol and other non-operative procedures ineffectively.
5.7.2 Contraindications
-
1.
Inability to tolerate general anesthesia or pneumoperitoneum.
-
2.
Refractory coagulopathy
-
3.
Cysts located deep in the liver, which are difficult to manage operatively.
-
4.
There are communications between the cysts and intrahepatic ducts.
-
5.
Cysts complicated with hemorrhage and infection.
5.7.3 Major Instruments or Energy Sources
-
1.
Laparoscopy System.
-
2.
Holding forceps.
-
3.
Harmonic scalpel.
5.7.4 Team Setup, Anesthesia and Position
Under general anesthesia, the patient was placed in supine position with the arms adducted along the side. The position of surgeon, assistants and nurses is shown in Fig. 5.41.

Fig. 5.41
5.7.5 Key Steps
-
1.
Exploration of lesion and identification and aspiration of the cyst.
-
2.
Fenestration of the cyst.
-
3.
Irrigation and check of the abdominal cavity.
-
4.
Drainage and closure of the incision.
5.7.6 Surgical Techniques
-
1.
Exploration of lesion and identification and aspiration of the cyst.
After a brief exploration of the abdomen, the main cyst was punctured. Aspiration was performed to make a definite judgment about the cyst liquid (Fig. 5.42a, b).

Fig. 5.42
-
2.
Fenestration of the cyst.
A suction drain was placed close to the opening and the main part of the liquid was suctioned out of the abdomen. The decompressed cyst was entered and resected with and ultrasonic dissector (Fig. 5.43a–h).

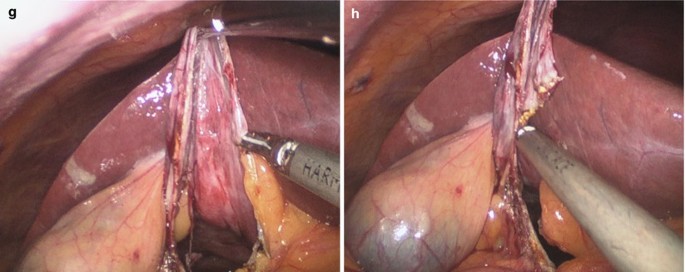
Fig. 5.43
-
3.
Irrigation and check of the abdominal cavity.
A careful control of the internal part of the cyst must be performed in case of bile leakage or hemorrhage. After adequate flushing the surgical field, a drain could be placed if necessary (Fig. 5.44a, b).

Fig. 5.44
5.7.7 Tips and Tricks
Using the harmonic scalpel (harmonic ACE 5 mm, Ethicon Endo-Surgery, Somerville), a liver cyst fenestration was carried out in the typical manner.
5.8 Laparoscopic Partial Hepatectomy
5.8.1 Indications and Case Selection
-
1.
Benign liver lesions.
-
2.
Symptomatic hemangioma.
-
3.
Symptomatic focal nodular hyperplasia.
-
4.
Adenoma.
-
5.
Symptomatic giant hepatic cyst.
-
6.
Malignant liver lesions.
-
7.
Hepatocellular carcinoma.
-
8.
Colorectal carcinoma metastasis.
-
9.
Others, including liver donor hepatectomy for liver transplant, malignant lesions, or indeterminate lesions (cannot rule out cancer).
5.8.2 Contraindications
-
1.
Any contraindications to open liver resection.
-
2.
Patients who cannot tolerate pneumoperitoneum or general anesthesia.
-
3.
Dense adhesions that cannot be lysed laparoscopically.
-
4.
Lesions too close to major vasculature.
-
5.
Lesions too large to be safely manipulated laparoscopically.
-
6.
Resection that requires extensive portal lymphadenectomy.
5.8.3 Major Instruments or Energy Sources
-
1.
Laparoscopy System.
-
2.
Holding forceps.
-
3.
Harmonic scalpel.
-
4.
Endoscopic Linear Stapler.
5.8.4 Team Setup, Anesthesia and Position
Under general anesthesia, the patient was placed in supine position with the arms adducted along the side. The position of surgeon, assistants and nurses is shown in Fig. 5.45.

Fig. 5.45
5.8.5 Key Steps
-
1.
Exploration of abdominal cavity and identification of the lesion.
-
2.
Dissection of hepatic ligament.
-
3.
Resection of liver tissue.
-
4.
Retrieval of the specimen.
-
5.
Irrigation and drainage.
5.8.6 Surgical Techniques
-
1.
Exploration of abdominal cavity and identification of the lesion.
The mass involving the left hepatic lobe was identified and exploration of the peritoneal cavity was performed to rule out the possibility of an extrahepatic cause (Fig. 5.46a–d).

Fig. 5.46
-
2.
Dissection of hepatic ligament.
The patient was positioned in a reverse Trendelenburg position. The left triangular ligament was divided by means of ultrasonic scalpel in order to expose the left lobe (Fig. 5.47a–f).

Fig. 5.47
-
3.
Resection of liver tissue.
The liver parenchymal transection line was marked with cautery hook. Liver transection was performed using ultrasonic scalpel (Fig. 5.48a–f).

Fig. 5.48
Hemostasis and liver division surface coagulation were completed with Ligasure device. Some vascular and bile duct structures could be ligated and transected by Endo-GIA (Fig. 5.49a–k).


Fig. 5.49
-
4.
Retrieval of the specimen.
The resected left hepatic lobe was then put in a plastic bag and completely removed through the umbilical incision (Fig. 5.50a–c).

Fig. 5.50
-
5.
Irrigation and drainage.
After final inspection for the site of bleeding and bile leakage, aided by saline irrigation, the drainage tubes were placed in the foramen of Winslow and near the stump of liver separately (Fig. 5.51a, b).

Fig. 5.51
5.8.7 Tips and Tricks
-
1.
For left lateral segmentectomies, the liver was mobilized from the falciform ligament and left triangular ligament using the Harmonic Scalpel (Ethicon Endo-Surgery, Inc.)
-
2.
Intraoperative ultrasound could be performed using a laparoscopic ultrasound probe. The site of the tumor was confirmed, and the transection plane with appropriate margins was marked using diathermy.
5.9 Complications Analysis and Management
5.9.1 Haemorrhage
The most common reason of haemorrhage in laparoscopic cholecystectomy is sever inflammation in the cystic triangle, which tends to form obscure anatomical structure and injure the cystic artery. But such cases aren’t suitable for SILS. In SILS cholecystectomy, cystic artery can be excellently coagulated by ultrasound scalpel. It is notable that the ultrasound scalpel working time and clamped tissue thickness determine the effect of coagulation. Intraoperative haemorrhage can also be induced by liver parenchyma injury accompanied with deeper wound surface of gallbladder bed. Electrocantery and haemostatic gauze can stop bleeding easily.
In SILS biliary surgery, massive bleeding can be induced by the injury of hepatic artery or portal vein. In SILS surgery, the lack of triangle relationship and limitation of motion may injure the vessels in the hepatoduodenal ligament during the dissection of the extrahepatic bile ducts. In case of massive haemorrhage around the common bile duct in SILS biliary surgery, local compression hemostasis by gauze and converting to open surgery should be decided as soon as possible.
The application of SILS in liver diseases is limited. So, the occurrence of massive bleeding is not high. For intrahepatic vascular systems with different diameters, choosing the proper cutting and coagulating instruments is absolutely essential. The chose of ultrasound scalpel, LigaSure, linear stapler or Hemolock is decided by the diameters of Gllisson’s sheath. After the lobe or tumor had been completely removed, a bipolar electrocauterizer or argon beam coagulator was used for hemostasis of the liver wound.
5.9.2 Biliary Injury and Bile Leakage
In SILS cholecystectomy and biliary exploration, vague anatomical relationship caused by local inflammation can induce laceration of extrahepatic bile duct, cutting and burning of bile duct, partly or completely clamped bile duct and even transaction. In this case, conversion to open surgery is always necessary. So, how to identify correct anatomical structure and choosing proper cases for SILS is the basis of avoiding biliary injury. More attention should be paid to the direction and strength of gallbladder retraction in SILS procedure. Excessive gallbladder retraction can cause jerking and angulating the common bile duct. In this case, gallbladder dissection may injury the sidewall of common hepatic duct and the clamping of cystic duct may cause a partial or complete clamping of the common bile duct. Bile leakage in SILS liver resection is always caused by dissecting the intrahepatic bile duct with unsuitable instruments. For larger bile ducts, clamping with Hemolock is recommended. Careful examination of bile leakage from liver wound and a proper placement of drainage are important methods to prevent postoperative bile leakage. Clamping and local suture can be used to deal with intraoperative bile leakage. For postoperative bile leakage, if the patient doesn’t have abdominal pain, fever and peritonitis symptoms, the drainage is adequate and conservative treatment will be considered.
5.9.3 Secondary Common Bile Duct Stone
Except for missing common bile duct stone before the operation, most postoperative common bile duct stones in SILS cholecystectomy are dropped from gallbladder. No matter what kind of dissecting sequence is used in SILS cholecystectomy, the gallbladder will be inevitable moved before the cystic duct was being clamped. So the possibility of gallbladder stone dropping into the common bile duct is existent. If the gallbladder stones is multiple, and the volume of stone is smaller (less than 3 mm), or the patients have the history of biliary pancreatitis, informed consent on the possibility of secondary common bile duct stones should be given before the operation. Endoscopic sphincterotomy (EST) can be used in the treatment of common bile duct stones.
5.9.4 Bile Duct Residual Stones
In SILS biliary exploration, the choledochoscope and basket enter the abdomen through the umbilical incision. In the surgical field, the choledochoscope and basket are parallel to the common bile duct. Though it is easier to observe and retract the intrahepatic bile duct stones, the stones in the distal end are difficult and often missed easily. Intraoperatively combined with duodenoscope or ERCP can identify the existence of residual stones in common bile duct while confirming whether the distal end of the C tube is in the duodenum.
5.9.5 Bile Duct Stenosis
Excess tissue of bile duct side wall can induce postoperative bile duct stenosis, especially in the case when the common bile duct diameter is less than 1 cm. Do not involved too much of bile duct side wall in the suture. SILS is not suitable in cases when the diameter of common bile duct is less than 5 mm.
References
Bittner R. The standard of laparoscopic cholecystectomy. Langenbecks Arch Surg. 2004;389:157–63.
Kaiser AM, Corman ML. History of laparoscopy. Surg Oncol Clin N Am. 2001;10:483–92.
Soper NJ, Stockmann PT, Dunnegan DL, Ashley SW. Laparoscopic cholecystectomy. The new ‘gold standard’? Arch Surg. 1992;127:917–21.
Schirmer BD, Edge SB, Dix J, Hyser MJ, Hanks JB, Jones RS. Laparoscopic cholecystectomy. Treatment of choice for symptomatic cholelithiasis. Ann Surg. 1991;213:665–76. discussion 677.
Johansson M, Thune A, Nelvin L, Stiernstam M, Westman B, Lundell L. Randomized clinical trial of open versus laparoscopic cholecystectomy in the treatment of acute cholecystitis. Br J Surg. 2005;92:44–9.
Navarra G, Pozza E, Occhionorelli S, Carcoforo P, Donini I. One-wound laparoscopic cholecystectomy. Br J Surg. 1997;84:695.
Lai EC, Tang CN, Ha JP, Li MK. Laparoscopic liver resection for hepatocellular carcinoma. Ten-year experience in a single center. Arch Surg. 2009;144:143–7.
Adani GL, Aprile G, Baccarani U, Risaliti A, De Anna D, Lorenzin D, et al. Benefit of laparoscopy for rectal resection in patients operated simultaneously for synchronous liver metastases: preliminary experience. Surgery. 2009;145:452.
Sarpel U, Hefti MM, Wisnievsky JP, Roayaie S, Schwartz ME, Labow DM. Outcome for patients treated with laparoscopic versus open resection of hepatocellular carcinoma: case-matched analysis. Ann Surg Oncol. 2009;16:1572–7.
Choh MS, Madura 2nd JA. The role of minimally invasive treatments in surgical oncology. Surg Clin North Am. 2009;89:53–77.
O’Rourke RW, Lee NN, Cheng J, Swanstrom LL, Hansen PD. Laparoscopic biliary reconstruction. Am J Surg. 2004;187:621–4.
Chokshi NK, Guner YS, Aranda A, Shin CE, Ford HR, Nguyen NX. Laparoscopic choledochal cyst excision: lessons learned in our experience. J Laparoendosc Adv Surg Tech A. 2009;19:87–91.
Binenbaum SJ, Teixeira JA, Forrester GJ, Harvey EJ, Afthinos J, Kim GJ, et al. Single-incision laparoscopic cholecystectomy using a flexible endoscope. Arch Surg. 2009;144:734–8.
Hodgett SE, Hernandez JM, Morton CA, Ross SB, Albrink M, Rosemurgy AS. Laparoendoscopic single site (LESS) cholecystectomy. J Gastrointest Surg. 2009;13:188–92.
Hong TH, Kim HL, Lee YS, Kim JJ, Lee KH, You YK, et al. Transumbilical single-port laparoscopic appen-dectomy (TUSPLA): scarless intracorporeal appendectomy. J Laparoendosc Adv Surg Tech A. 2009;19:75–8.
Fagotti A, Fanfani F, Marocco F, Rossitto C, Gallotta V, Scambia G. Laparoendoscopic single-site surgery (LESS) for ovarian cyst enucleation: report of first 3 cases. Fertil Steril. 2009;92:1168.e13–6.
Fader AN, Escobar PF. Laparoendoscopic single-site sur-gery (LESS) in gynecologic oncology: technique and initial report. Gynecol Oncol. 2009;114:157–61.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2013 Springer Science+Business Media Dordrecht
About this chapter
Cite this chapter
Wu, S., Fan, Y., Tian, Y. (2013). Hepatobiliary Surgery. In: Wu, S., Fan, Y., Tian, Y. (eds) Atlas of Single-Incision Laparoscopic Operations in General Surgery. Springer, Dordrecht. https://doi.org/10.1007/978-94-007-6955-7_5
Download citation
DOI: https://doi.org/10.1007/978-94-007-6955-7_5
Published:
Publisher Name: Springer, Dordrecht
Print ISBN: 978-94-007-6954-0
Online ISBN: 978-94-007-6955-7
eBook Packages: MedicineMedicine (R0)





































































