Abstract
Growth is an obvious and easily measured response, and is included in many standard toxicity tests as well as in research projects. Reduced growth is frequently traced back to reduced food intake, but even without reduced feeding, it is a logical outcome since organisms must expend energy to defend themselves against and detoxify contaminants. The more energy needed to detoxify pollutants, the less will be available for growth. In addition to overall body growth, molting, regeneration, development of calcified structures (shell and bone), carcinogenesis, and smoltification are other developmental processes that take place after larval stages. These processes are all sensitive to environmental contaminants.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Growth
Growth is the end result of a number of processes including food intake and energy metabolism which are affected negatively by many contaminants. Consequently, growth tends to be reduced in exposed organisms.
1.1 Fishes
Fishes continue growing as juveniles and adults, albeit more slowly over time, throughout their lives, and growth can be affected by contaminants. There have been hundreds of studies of fish growth in response to contaminants, largely in freshwater species and early life stages. Many of these studies are based on dietary exposure, and show that reduced feeding is responsible for growth effects. However, some environmentally important pollutants, including xenoestrogens and metals have been shown to affect growth via hormone-mediated mechanisms, possibly by interfering with the GH receptor and/or GH transcription (Deane and Woo 2009). Other contaminants, such as dioxins, furans, PCBs, polycyclic aromatic hydrocarbons, pesticides, phenols, ammonia, pharmaceuticals and metals, may affect growth and other developmental processes by thyroid disruption (reviewed by Brown et al. 2004; Rolland 2000).
Woltering (1984) reviewed 173 studies to date on fish growth. The tests include exposure to metals, pesticides, unclassified organics, inorganic compounds, detergents and complex effluents. Fry survival was significantly reduced in 57 % of all tests at the lowest effect level and fry growth was reduced in 36 % of the tests. Only 60 % of the tests include exposure of adults; adult growth was seldom the most sensitive effect. Fry survival and growth were very often equally sensitive.
1.1.1 Metals
There have been many studies of effects of metals on juvenile fishes, of which a few will be mentioned. Baker et al. (1998) fed juvenile grey mullet (Chelon labrosus) a basal (4.4 mg Cu/kg dry wt) or high-Cu diet (2,400 mg Cu/kg dry wt) for 10 weeks to assess the relationship between growth and Cu-induced oxidative damage. No mortalities were seen, but growth rate and food intake were reduced by 43 and 29 %, respectively, in the high-Cu group. Aqueous exposure to Cu also affects feeding and growth. Buckley et al. (1982) exposed coho salmon (Oncorhynchus kisutch) to sublethal levels of aqueous copper (0.5 and 0.25 of the LC50.), which caused loss of appetite and decreased growth. These both recovered when fish were returned to clean water.
Juvenile green and white sturgeon (Acipenser medirostris and A. transmontanus) were exposed to dietary meHg, at 25, 50, and 100 mg meHg kg−1 diet for 8 weeks. Higher mortality and lower growth rate were noted in both species in a dose-dependent manner (Lee et al. 2011). Green sturgeon exhibited earlier and more severe effects than white sturgeon. Hg accumulated to the highest concentrations in kidneys of both species.
In a field study, Cd, Cr, Cu, Fe, Pb, and Zn concentrations in the muscle, gill and liver of six fish species (Sparus auratus, Atherina hepsetus, Mugil cephalus, Trigla cuculus, Sardina pilchardus and Scomberesox saurus) from the northeast Mediterranean Sea were measured and the relationships between fish size (length and weight) and metal concentrations were investigated by linear regression analysis (Canli and Atli 2003). In most cases, relationships between metal concentrations and fish size were negative. Highly significant (P < 0.001) negative relationships were found between fish length and Cr concentrations in the liver of A. hepsetus and M. cephalus, and Cr concentrations in the gill of T. cuculus. Cr and Pb concentrations in the liver and Cu concentrations in all the tissues of Scomberesox saurus also showed very significant (P < 0.001) negative relationships.
1.1.2 Organics
Most studies of organic chemicals affecting marine fish growth have been done on oil and its components. Pink salmon fry (Oncorhynchus gorbuscha) fed oil-contaminated prey (0.6, 3.2, and 6.5 mg l−1) had a reduced growth rate with increasing concentrations, which was strongly correlated with decreases in feeding rate (Schwartz 1985). Growth reduction in fry exposed to 0.7 mg l−1 of WSF in the water was greater than those of fry fed the oil-contaminated prey.
Following the Exxon Valdez spill, Heintz et al. (2000) reported delayed effects in pink salmon that had been exposed as embryos to gravel coated with weathered oil. Weathering caused the PAH composition to be dominated by heavier compounds. Survivors that appeared healthy were released to the marine environment with coded-wire tags and their survival evaluated when they returned 2 years later. Fish that had been exposed to an initial concentration of total PAH equal to 5.4 μg l−1 had a 15 % decrease in marine survival compared to unexposed fish. Reduced growth was seen in fish that had had embryonic exposure to 18 μg l−1 PAH. Thus, exposure at sensitive early life stages led to reduced growth and higher mortality later in life.
Rice et al. (2000) exposed polychaetes (Armandia brevis) for 28 days to sediments with benzo(a)pyrene (BaP), para-para dichlorodiphenyldichloroethylene (pp′DDE), Aroclor 1254, or field sediments collected from sites in Puget Sound, Washington, contaminated predominantly with PAHs or chlorinated compounds. Exposed worms were then fed to juvenile English sole (Pleuronectes vetulus) for 10 or 12 days, and fish were measured for length and weight, sacrificed, and preserved. Growth of fish was lower than reference in all but one of eight groups fed contaminated worms, but statistically significant reductions in growth were only observed in three groups, at least in part due to low statistical power. Fish from all exposed groups showed increased expression of CYP1A, and fish exposed to BaP showed evidence of hepatic PAH-DNA adducts.
Cong et al. (2009) investigated effects of the organophosphate insecticide diazinon on juvenile snakehead fish Channa striata, which were exposed twice to 4-day pulses of 0.016, 0.079 or 0.35 mg l−1 of diazinon, separated by a 2 week interval (done to imitate the exposure conditions near rice fields). Fish were then moved to clean water for recovery. Diazinon caused long term inhibition of brain ChE activity, which was still significantly depressed at the termination of the experiment, and the highest diazinon concentration caused a significant 30 % growth inhibition.
1.1.3 Hypoxia
Growth rates of winter flounder Pseudopleuronectes americanus and summer flounder Paralichthys dentatus were reduced as DO decreased, particularly at 50–70 % sat, and as temperature increased. Summer flounder were more tolerant than winter flounder. A significant relationship between feeding rate and growth indicated that reduced food consumption was responsible for growth limitation (Steirhoff et al. 2006).
Effects of moderate hypoxia and oscillating DO on growth of European sea bass (Dicentrarchus labrax) were investigated (Thetmeyer et al. 2001). Fish were exposed to one of three conditions: 40 % sat; oscillations between 40 and 86 % with a period of 770 min, and 86% sat as a control at 22 °C for 1 month. Fish in hypoxia consumed significantly less food, had reduced growth, and lower condition factor. Those in oscillating conditions were intermediate, and not statistically distinguishable from either normoxic or hypoxic groups. Growth was correlated with food intake, suggesting that reduced growth was primarily due to reduced appetite.
When juvenile turbot Scophthalmus maximus and sea bass Dicentrarchus labrax were fed to satiation, food intake and growth were depressed under hypoxia (3.2 and 4.5 mg O2 l−1). Growth was comparable between fish fed to satiation in hypoxia and those reared in normoxia but fed restricted rations (Pichavant et al. 2001). Decreased food thus appears to be a mechanism by which prolonged hypoxia reduces growth, and may be a way to reduce energy and thus oxygen demand.
Killifish (Fundulus heteroclitus) were subjected to severe hypoxia (1.2 mg O2 l−1), moderate hypoxia (3.0 mg O2 l−1), normoxia (7.1 mg O2 l−1) and hyperoxia (10.6 mg O2 l−1) and specific growth rate calculated weekly. Severe hypoxia reduced growth and reduced muscle protein content and RNA: DNA compared with other treatments. However, growth increased significantly during the second 2-week interval, to the rate of normoxic fish (Rees et al. 2012). Neither moderate hypoxia nor hyperoxia affected growth or biochemical variables, showing that F. heteroclitus tolerates hypoxia and, during prolonged severe hypoxia, compensates for the initial negative effects on growth. Its capacity to grow in low DO contributes to its persistence in highly degraded habitats.
1.1.4 Polluted Environment
European flounder (Platichthys flesus) responses to chemical stress were assessed in four estuaries with different patterns of contamination. Fish in contaminated estuaries were characterized by high levels of bioaccumulated contaminants, slow energetic metabolism and reduced growth rate (Evrard et al. 2010).
Juvenile turbot (Scophthalmus maximus) were exposed to sediments contaminated with metals, PAH and PCBs (Kerambrun et al. 2012a). Significant decreases in growth rates, morphometric index, RNA:DNA ratio and lipid storage index were observed with increasing levels of contamination from site A to site C (Fig. 8.1). The decrease in physiological status could be related to the accumulation of metals in gills and of PAH metabolites in bile.
Differences in specific growth rate in weight (GW), length (GL) and recent otolith growth (RG) of juvenile turbot exposed to 5 sediments (Ref. Seine, A,B,C) after 7 (gray) and 21 (black) days (M ± SE). Numbers = significant differences compared to t0 (Reprinted from Kerambrun et al. 2012a: 136, courtesy of Elsevier Publishing Co.)
Juvenile sea bass (Dicentrarchus labrax) and turbot (Scophtalmus maximus) were caged for 38 days at three locations with varying levels of contaminants (Kerambrun et al. 2012b). At the most contaminated station, all the fish died within 2 weeks. At the least contaminated station, fish had higher growth, RNA:DNA ratios and condition index than at the intermediate station. Lipid storage index, based on the ratio of the quantity of triacylglycerols to sterols (TAG:ST), was significantly higher in the less contaminated station. Reduced growth and condition was associated with metal accumulation in gills.
1.2 Mollusks
Growth of mollusks can be investigated by measuring increments in the shell, or by measuring the condition index (weight of soft tissues vs shell), which reflects growth of soft tissues. Studies are quite limited, and many of the studies on contaminant effects on mollusk growth are quite old. Studies on shell growth reflect effects on calcification, which will be discussed later in the chapter.
1.2.1 Metals
Juvenile and adult mollusks are sensitive to metals. Boyden et al. (1975) found that Zn at 0.25 and 0.50 mg l−1 reduced growth of juvenile C. gigas to 78 and 51 % of control values, but after a 5-day depuration period, growth rebounded. Cunningham (1976) found similar retardation of growth in juvenile C. virginica exposed to 0.01 and 0.10 mg l−1 Hg for 47 days, and similar recovery after return to clean water for about a month. Exposure to a mercury-equilibrated algal suspension containing 0.25, 0.42 and 1 Hg l−1 reduced growth and condition of adult limpets Crepidula fornicata over 16 weeks (Thain 1984).
1.2.2 Organics
There has been some work, much of it old, on effects of pesticides and PAHs on clam growth. Butler et al. (1968) investigated effects of the pesticide carbaryl (Sevin) and its breakdown product, 1-naphthol, on clams (Clinocardium nuttalli). Sevin was less toxic than 1-naphthol to juvenile clams, the respective 96-h TLM’s (median tolerance limits) being 3.75 and 2.70 mg l−1. Growth of clams was reduced more by 1-naphthol than by Sevin. The food consumption of clams exposed to 1.6 mg l−1 Sevin was markedly reduced and their food conversion efficiency was impaired.
Small (<30 mm) clams, Protothaca staminea, were marked and placed in the intertidal zone of Sequim Bay, Washington for 1 year to measure effects of oiled sediments on growth (Anderson et al. 1981). Growth in oil-treated sediment was significantly slower than in clean sediment; oil mixed into sediment (10 cm) produced greater tissue contamination and greater growth reduction than a layer (3 cm) of oiled sediment. Effects of residual Exxon Valdez oil on P. staminea, were studied 5–6 years after the spill using reciprocal transplants of clams between unoiled and oiled sites (Fukuyama et al. 2000). Tagged clams were transplanted in 1994 and collected in 1995 and 1996. Greatest mortality and tissue burdens of hydrocarbons were in clams from the oiled site, and the best growth was in clams moved from the oiled to the unoiled site where they could depurate.
1.2.3 Hypoxia
Burrell et al. (2011) examined the relationship between diel-cycling hypoxia and growth and the acquisition and progression of Perkinsus marinus infections in C. virginica. Laboratory and field experiments indicated that diel-cycling hypoxia increased acquisition and progression of infection, and decreased growth. Patterson et al. (2011) found heavier 15N in tissues of oysters under lower DO conditions; this is consistent with reduced feeding and catabolism of tissues, common in stressed bivalves and in keeping with the reduced growth noted previously.
1.2.4 Acidification/Climate Change
Sydney rock oysters, Saccostrea glomerata, were deployed at sites affected and unaffected by acid sulfate soils in two Australian estuaries. After 10 weeks, oysters were transplanted within and across sites and maintained another 10 weeks. Oysters that remained 20 weeks at affected sites grew at about half the rate of oysters at reference sites (Amaral et al. 2012b) Oysters moved from acidified to reference sites grew more than oysters moved from reference to acidified sites or that stayed at reference sites. Dickinson et al. (2012) studied interactive effects of salinity and CO2 on growth and energy homeostasis in C. virginica. Juveniles were exposed for 11 weeks to 30 or 15 psu salinity at current atmosphere (∼400 μatm), or ∼700–800 μatm. Exposure to elevated P CO2 and/or low salinity led to increased mortality, reduced tissue energy stores (glycogen and lipid) and negative soft tissue growth, indicating energy deficiency. In contrast, juvenile mussels, M. galloprovincialis under conditions of −0.3 and −0.6 pH units had increased absorption efficiency and ammonium excretion, and increased scope for growth and tissue dry weight, suggesting that this species tolerates acidification (Fernandez-Reiriz et al. 2012).
1.2.5 Other Pollutants
Growth of oysters (Crassostrea virginica) subjected to chlorine-produced oxidants was studied by Scott and Vernberg (1979). The condition index and gonadal index were reduced by 0.16 mg l−1. This concentration also reduced fecal production. This implies effects on feeding, which was not directly measured. There were seasonal differences in responses, reflecting temperature interacting with toxic effects.
1.3 Crustaceans
Most effects on growth are related to the molt cycle, which is discussed below. However, there is some evidence of effects of selected organic contaminants on somatic growth of crustaceans.
1.3.1 Organics
Guo et al. (2012) investigated long-term effects of PCB 126 on the copepod Tigriopus japonicus. No obvious effects were observed in the first generation at concentrations <100 μg l−1 but in the second generation effects on reproduction were seen at levels over 1 μg l−1 and on growth at concentrations over 0.1 μg/l. Thus, they became more sensitive in the second generation and growth was the most sensitive parameter. The authors concluded that environmental risk assessment of contaminants should be based on a long-term multigenerational exposure to provide realistic measures of pollutant effects.
1.4 Other Taxa
1.4.1 Acidification/Climate Change
Sea urchins, Lytechinus variegatus, were reared in ambient seawater (380 μatm) and two elevated levels of pCO2, 560 and 800 μatm. After 89 days, urchins at ambient pCO2 weighed 12 % more than those at 560 μatm and 28 % more than those at 800 μatm (Albright et al. 2012). Skeletons examined with scanning electron microscopy, showed degradation of spines at 800 μatm. Doropoulos et al. (2012) found that after 2 months’ growth in ambient or elevated pCO2 levels, the linear extension and calcification of coral (Acropora millepora) recruits decreased as CO2 partial pressure (pCO2) increased. When recruits were subjected to incidental fish grazing, their mortality was inversely size dependent. There was an additive effect of pCO2 such that recruit mortality was higher under elevated pCO2 irrespective of size. In elevated pCO2, coral recruits needed to double their size at the highest pCO2 to escape grazing mortality. This general trend was seen with different predators (blenny, surgeonfish, and parrotfish). In contrast, OA-like conditions can enhance the ecological success of non-calcifying anthozoans e.g. sea anemones. Increased growth (abundance and size) of the sea anemone (Anemonia viridis) was observed by Suggett et al. (2012) along a natural pCO2 gradient at Vulcano, Italy. Both gross photosynthesis (PG) and respiration (R) increased with pCO2 indicating that the increased growth was, at least in part, fuelled by bottom up (CO2 stimulation) of metabolism. The increase of PG outweighed that of R and the genetic identity of the symbiotic microalgae (Symbiodinium spp.) remained unchanged. These observations of enhanced productivity with pCO2, may increase fitness and enable non-calcifying anthozoans to thrive in future high CO2 environments.
Reduced growth can have profound ecological consequences for calcifying organisms. Kroeker et al. (2013) investigated species interactions that can modify direct effects of acidification on individual species and showed that altered competitive dynamics between calcareous species and fleshy seaweeds drive significant ecosystem shifts. Although calcareous species recruited and grew in low pH during early successional stages, they were rapidly overgrown by fleshy seaweeds later in succession in low pH. The altered competitive dynamics between calcareous and fleshy seaweeds is probably due to decreased growth of calcareous species, increased growth of fleshy seaweeds, and/or altered grazing rates. Phase shifts towards dominance by fleshy seaweeds are common in many marine ecosystems, and these results suggest that changes in the competitive balance between these groups is a key factor leading to profound ecosystem changes as the pH decreases.
Six Caribbean coral reef sponges—Aiolochroia crassa, Aplysina cauliformis, Aplysina fistularis, Ectyoplasia ferox, Lotrochota birotulata and Smenospongia conulosa—were grown for 24 days in seawater ranging from current summer maxima (28 °C; pH = 8.1) to those predicted for 2100 (31 °C; pH = 7.8). For all species, growth and survival were similar among temperature and pH levels (Duckworth et al. 2012). Sponge attachment, which is important for reef consolidation, was unchanged by pH for all species, and highest at 31 °C for E. ferox, I. birotulata and A. cauliformis. These findings on adult sponges suggest that ecological roles and growth of these species will not be adversely affected by the temperature and pH predicted for the end of the century.
2 Regeneration and Molting
Regeneration of lost tissue is a process that, while extremely limited in birds and mammals, is common among invertebrates and fishes. The processes by which structures are regenerated resemble the cell differentiation that goes on in embryos, although a pool of stem cells must be initially developed from more differentiated tissue near the point of injury. In crustaceans, the regeneration of appendages is tied in with the molt cycle.
2.1 Crustaceans
In order to grow, crustaceans must molt their old exoskeleton to accommodate their larger body. Growth is thus periodic, and depends on molting of the exoskeleton. The frequency of molting and the growth increment are influenced by environmental factors and hormones, particularly ecdysteroids, which are steroids. Molt-inhibiting hormones also play a role. In preparation for molting (proecdysis) ecdysteroid levels in hemolymph increase and the tissue layer under the exoskeleton secretes materials that erode and soften the old cuticle. Calcium is dissolved and moved to interior storage places, softening the old cuticle. The epidermal tissues then produce a thin, soft new exoskeleton underneath it. Then the animal takes in a lot of water, swells up and bursts the old skeleton along specific weak points, and extricates itself from it (ecdysis). Mobility is limited right after a molt (postecdysis) because the new exoskeleton is very soft and not rigid enough to keep the limbs stiff. Animals are very vulnerable until the new exoskeleton expands and hardens. Although most crustaceans molt throughout their entire life, small individuals must molt frequently; some species have a terminal molt after reaching their maximum size. During ecdysis, postecdysis, and intermolt phases, ecdysteroid levels in hemolymph are low.
Some crustaceans can autotomize injured limbs, i.e., break them off at a preformed breakage plane where there is a membrane, thus minimizing tissue damage. Autotomy is an effective anti-predator response, and animals subsequently regenerate lost appendages, in association with the molt cycle. Regeneration begins after a period of tissue re-organization and is first noticeable as a small protrusion at the autotomy plane. The limb bud grows within a covering of cuticle. In crabs, regenerating limbs grow folded and only unfold when the animal molts. In shrimp, the regenerating limbs have joints nestled within one another, so they also do not become functional until the animal molts. Thus, regeneration is closely tied with the molt cycle. Regeneration consists of basal growth, when tissue differentiation occurs and which is independent of the molt cycle, and pro-ecdysial growth, a phase of rapid growth which is dependent on molting hormones. There may be a plateau between basal and proecdysial growth. Multiple autotomy, removal of many limbs, stimulates accelerated regeneration and molting. Many chemicals alter the rate of limb regeneration and/or molting. The two processes may be affected independently, but it is often not possible to distinguish effects on regeneration per se from those on the molt cycle, since they are usually coupled and both processes are affected simultaneously. Some toxicants produce morphological alterations in the regenerated limbs; these may be relatively minor, such as reduced number of pigment cells, setae, or tubercles, or may be major, such as abnormal bending or defects in exoskeleton formation. By molting, crustaceans may be able to reduce their body burdens of contaminants that are contained within the exoskeleton (Keteles and Fleeger 2001; Bergey and Weis 2007).
2.1.1 Metals
The most common effect of metals is retardation of regeneration accompanied by a delay in ecdysis; in some cases regeneration is affected without altering the timing of the molt. A series of studies of metals on regeneration in fiddler crabs was conducted in the 1970s and 1980s by Weis and colleagues; these are reviewed in Weis et al. (1992). Delayed regeneration and molting were observed in Uca pugilator after exposure to HgCl2, meHg, Cd, and Zn at 0.5–1.0 mg l−1. Retardation of regeneration was accompanied with delayed ecdysis so when molting took place limbs were fully formed. MeHg at 0.1 mg l−1 inhibited melanin pigment development in the regenerated limbs and reduced the number of tubercles on regenerated first walking legs of males. While Hg and Cd individually retarded limb regeneration, the presence of meHg reduced the particularly toxic effects of Cd at low salinity. Zn and Hg together were additive, while Zn and Cd interacted antagonistically. Uca pugnax from a contaminated site were less affected by meHg than those from a relatively clean site, i.e., limb regeneration was not as greatly retarded, indicating that they had acquired some tolerance. However, short-term pre-exposure to low concentrations of meHg did not enhance tolerance to higher concentrations.
Juvenile tiger shrimp, Penaeus monodon, shortened the time to first molt and decreased molting frequency after exposure to 0.9 mg l−1 Cr (Chen and Lin 2001). They had a reduced feeding rate. Grass shrimp, P. pugio, telson regeneration was retarded by exposure to Hg or meHg (10 μg l−1) and they had a reduced intermolt period (Kraus and Weis 1988). However, these effects were not seen in shrimp from a contaminated site. Hexavalent Cr inhibited limb regeneration in grass shrimp (Rao et al. 1985). The antifouling agent tributyltin (TBT) retarded limb regeneration and molting in U. pugilator at 0.5 μg l−1, and produced anatomical abnormalities in regenerated chelae of males, in which the regenerated dactyl curved upward, away from the pollex, instead of downward towards it (Fig. 8.2) (Weis et al. 1987b). In P. pugio, 0.5 μg l−1 TBT slowed regeneration rate, but did not produce abnormalities (Khan et al. 1993).
(a) Malformed chela in fiddler crab after regenerating in TBT. Chela has deformity in the dactyl (D). (b) Curled, stunted chela (C) and malformed walking legs 2 and 3 after regeneration in TBT (Reprinted from Weis et al. 1987b: 323, courtesy Springer Publishing Co.)
2.1.2 Organic Contaminants
2.1.2.1 Aromatic Hydrocarbons
Exposure of juvenile blue crabs to 1 mg l−1 benzene or dimethylnaphthalene increased the length of the intermolt cycle, decreased the increment per molt, and retarded limb regeneration (Cantelmo et al. 1981). Affected crabs had slower limb regeneration, a longer plateau stage, and a longer time for regenerated limbs to develop pigmentation. Wang and Stickle (1987) found that the WSF (1.5 or 2.5 mg l−1) of South Louisiana crude oil inhibited growth and molting in blue crabs, reduced the increment at molt, and prolonged the intermolt period.
2.1.2.2 Pesticides, PCBs and Dioxins
The PCB mixture Aroclor 1242 (8 mg l−1) inhibited limb regeneration in U. pugilator, with greater inhibition at high and low salinities than at intermediate salinities (Fingerman and Fingerman 1978). Chlorophenols and dithiocarbamates at 0.1–1.0 mg l−1 inhibited regeneration in P. pugio, generally without affecting the timing of molting. Retarded limb regeneration and molting in P. pugio was seen in response to dithiocarbamates and pentachlorophenol, with the early stages of regeneration more sensitive than later ones (Rao et al. 1979). Other chlorophenols (2,3,4,5-tetrachlorophenol and 2,3,4,6-tetrachlorophenol at 0.3 and 0.7 mg l−1 respectively) inhibited limb regeneration but did not alter the duration of the molt cycle, suggesting a direct effect on limb growth rather than one on hormones (Rao et al. 1981).
Current insect growth-regulating pesticides are very toxic to growth processes in crustaceans. The chitin-synthesis inhibitor, diflubenzuron (Dimilin®) interferes with chitin deposition and produces disturbances in cuticular structure. Treated larvae appear healthy until they molt, at which time they may be unable to shed the old cuticle and die. Exposure to 0.5, 5.0 and 50 μg l−1 produced a dose-dependent retardation of regeneration and molting in U. pugilator; crabs that molted in high concentrations had high mortality (Weis et al. 1987a). Regenerated limbs had blackened areas in which the cuticle had not developed properly. Dimilin also retarded the molt cycle in juvenile fiddler crabs (Cunningham and Myers 1987) and grass shrimp, and 0.11 μg l−1 inhibited limb regeneration (Touart and Rao 1987). This suggests that in addition to affecting cuticle synthesis, it affects molting hormones, mitosis and differentiation of limb buds. Stueckle et al. (2008) found that methoprene, a juvenile hormone mimic, retarded regeneration in U. pugnax, with greater effects on males, which took longer than females at 0.1 μg l−1 and exhibited a greater frequency of abnormalities at 1.0 μg l−1. They had a greater frequency of abnormal limbs that failed to regenerate or had a bent or bulging merus, carpus or propodus, or a hook-shaped dactyl. The frequency of abnormal limbs was greatest at intermediate concentrations, producing inverted U-shaped curves, reminiscent of other endocrine disrupting effects (Fig. 8.3).
However, not all toxic chemicals retard limb regeneration and molting. DDT (10 μg l−1) accelerated limb regeneration in U. pugilator (Weis and Mantel 1976). In the case of crabs with multiple autotomy, the time to ecdysis was shortened as well. These responses may have been due to heightened excitation of the nervous system and secretion of neuroendocrine factors promoting molting.
2.1.3 Hypoxia
Shrimps (Penaeus semisulcatus) kept for 17 days at an oxygen level of 2 mg l−1 did not molt, and high mortality was observed. When the DO was increased to 5 mg l−1 the mortalities ceased and many molts took place (Clark 1986).
2.2 Fishes
Teleost fins are composed of connective tissue and bony rays, and can regenerate after amputation. Cells at the cut end dedifferentiate to form a blastema, a mass of undifferentiated stem cells, which then grows and develops new bony rays and connective tissue. Teleosts are also capable of regenerating scales and other structures, including components of the nervous system.
2.2.1 Metals
The rate of caudal fin regeneration in Fundulus heteroclitus was retarded by 0.1 mg l−1 Cd (Weis and Weis 1976) and 0.001 mg l−1 meHg (Weis and Weis 1978), which also retarded regeneration in Mugil cephalus. However, the two metals interacted in an antagonistic fashion so that fish exposed to combinations of meHg and Cd regenerated at rates comparable to controls. In addition, fish pre-exposed to 0.05 mg l−1 Cd acclimated to the metal and their regeneration in 0.1 mg l−1 was not retarded, but was faster than controls, evidence of hormesis (Weis and Weis 1986). In contrast to Hg and Cd, Zn at 1.0, 3.0 and 10.0 mg l−1 accelerated regeneration in a dose-dependent fashion, and could counteract the inhibitory effects of meHg (Weis and Weis 1980).
Exposure to Cd and Zn at 0.1 mg l−1 reduced calcification and reduced Ca/P ratios in regenerating scales of F. heteroclitus (Sauer 1987).
2.2.2 Organics
The pesticides DDT, malathion, parathion, and carbaryl at 10 μg l−1 retarded fin regeneration in F. heteroclitus (Weis and Weis 1975). The effects of DDT were less pronounced than the other three insecticides.
2.2.3 Polluted Environment
There have been a number of field reports of increased prevalence of a fin erosion condition (or “fin rot”) in fishes from highly contaminated sites. While these reports were more common in decades past, new reports continue to be published (Bodammer 2000; Ziskowski et al. 2008). While many of these studies find bacterial infection associated with the condition, the infection may be opportunistic and the condition may also involve poor ability to regenerate fins after injury.
2.3 Other Taxa
Echinoderms are capable of regenerating their arms. This ability was apparently unknown to people who tried to kill nuisance crown-of-thorns starfish by cutting them up. All they accomplished by this was to produce more nuisance starfish.
2.3.1 Metals
Arm regeneration in the brittle star, Ophioderma brevispina was inhibited by 0.1 μg l−1 of TBT or triphenyltin (Walsh et al. 1986). Cadmium-exposed brittle stars, Microphiopholis gracillima, regenerated thinner arms with less soft and skeletal tissue (D’Andrea et al. 1996).
2.3.2 Hypoxia
Arm regeneration in the brittle star Amphiura filiformis was studied in normoxia >8.5 mg O2 l−1 (control) and in two levels of hypoxia, 2.7 and 1.8 mg O2 l−1 (29 and 19 % sat). The treatments were chosen to simulate short term hypoxia (17 days) over a 2-month period. Reduced arm growth was seen in both hypoxic treatments compared with normoxia. Slowest regeneration of arms was observed at 1.8 mg O2 l−1 (65 % arm regeneration compared to controls) (Nilsson and Sköld 1996).
2.3.3 Acidification/Climate Change
Sea stars, Luidia clathrata with two arms excised, were maintained in seawater of pH 8.2 or 7.8. After 97 days, a period of time sufficient for 80 % arm regeneration, the lower pH did not significantly effect whole animal growth, arm regeneration, or biochemical composition (Schram et al. 2011). Wood et al. (2011) investigated responses of the brittlestar, Ophiocten sericeum, a polar species, to a temperature increase of 3.5 °C (ambient, 5–8.5 °C) and reduction in pH to 7.7 or 7.3 (ambient was 8.3). Faster arm regeneration was stimulated by higher temperature but was counteracted by low pH; at pH 7.3 in high temperature, the maintenance of calcified structures reduced the rate of regeneration, possibly due to use of energy reserves.
3 Shell/Bone Formation
Numerous studies have been done on effects of contaminants on formation of skeletal structures. In recent years studies on calcification have been dominated by examination of effects of ocean acidification on different taxa, which vary greatly in susceptibility. A widespread analysis suggests that increased acidity is affecting the size and weight of shells and skeletons, and the trend is widespread across marine species (Watson et al. 2012). Variation in shell thickness and skeletal size was studied in clams, snails, brachiopods, and sea urchins from 12 different environments from tropics to polar regions to get a clearer understanding of similarities and differences among taxa, and to make better predictions of how animals respond to increasing acidity. The study showed that over evolutionary time, animals adapt to environments where calcium carbonate is difficult to obtain by forming lighter skeletons. Low pH makes it harder for marine animals to make shells and skeletons. As the availability of calcium carbonate decreases, skeletons get lighter and account for a smaller part of the animal’s weight. The fact that same effect occurs consistently across taxa suggests the effect is widespread. The effect is greatest at low temperatures; polar species have the smallest and lightest skeletons, suggesting that they may be at greater risk as the ocean becomes more acidic.
3.1 Crustacea
3.1.1 Polluted Sites
Many crustaceans inhabiting degraded estuaries and coastal waters can develop a condition called “shell disease” or shell erosion. For example, lobsters and rock crabs (Homarus americanus and Cancer irroratus) from grossly polluted areas of the NY Bight (a sewage sludge dump site) had erosion on legs, spines and elsewhere. The condition could be produced in the lab by exposing animals to sediments from the sewage sludge or dredge spoil disposal site (Young and Pearce 1975). Shrimp, Crangon septemspinosa, from the vicinity of the dump site had high prevalence of eroded appendages and blackened erosion of the exoskeleton, rarely observed elsewhere (Gopalan and Young 1975); this condition was produced in the laboratory in 50 % of the individuals exposed to sea water from the site. Erosion was progressive and the eroded parts of appendages did not regenerate after molting. Shell erosion is caused by bacteria that break down chitin, with subsequent secondary infection of underlying tissue by other bacteria. These dump sites have been closed, and shell erosion disease has not been reported from the area since then.
3.1.2 Acidification
Ries et al. (2009) investigated responses of various marine organisms to different pCO2 levels. In three crustaceans, blue crabs (Callinectes sapidus), lobsters (Homarus americanus), and shrimps (Penaeus plebejus) net calcification was, surprisingly, greatest under the highest level of pCO2 (2,856 mg l−1). An explanation offered for these unexpected results was that if seawater is the source of an organism’s calcifying fluid, then the concentration of dissolved inorganic carbon (DIC) in this fluid will increase as pCO2 increases. Organisms that can maintain an elevated pH at their site of calcification despite reduced external pH will convert much of this increased DIC to carbonate. They would have a final carbonate concentration at the site of calcification that is only slightly less than (and possibly equal to or greater than) that under present-day pCO2 levels, depending on the efficiency of their proton-regulating mechanism. Furthermore, organisms that accrete shell that is covered and protected by an external organic layer (such as the crustacean epicuticle) can be more resilient to acidification than those whose shell is directly exposed to seawater.
3.2 Mollusks
3.2.1 Metals
The effects of Cu and Zn on the daily shell growth of M. edulis were measured with a laser diffraction technique. Within 3 days 10 μg l−1 Cu inhibited shell growth (Manley et al. 1984); effects became more severe as exposure time increased. Recovery of nearly normal shell growth occurred upon transfer to clean sea water. Effects of zinc were less severe. Oysters (C. gigas) are sensitive to effects of TBT at levels as low as 0.05 μg l−1 Oysters living near marinas developed anomalies in which the shell developed many layers with gel-like material between the layers (Fig. 8.4) (Waldock and Thain 1983). Abnormalities decreased with distance from port areas, and TBT could induce it in the laboratory. Oysters transplanted into port areas developed these abnormalities. After the use of TBT paints was banned, abnormalities decreased (Alzieu et al. 1986).
TBT-exposed oyster shell showing chambers in upper valve (Reprinted from Alzieu 1986, courtesy of IEEE)
Zuykov et al. (2011) studied morphology of the inner shell surface of the mussel Mytilus edulis after short-term exposures to Ag as free-ion (Ag+) and as nanoparticles. The nacre portion of exposed mussels (both treatments) developed doughnut shaped structures of calcium carbonate micrograins covering the surface. Formation of these structures was explained as a disturbance of shell calcification.
3.2.2 Organics
Mollusks in mesocosms with sediment were exposed to oil and oil + dispersant equivalent to 50 mg l−1 oil (Carr et al. 1987). Mya arenaria showed major reduction in shell growth and condition index in oil and in oil + dispersant treatments.
3.2.3 Acidification
Pteropods are shelled pelagic mollusks that play an important role in planktonic food webs. The impact of pH was investigated on Limacina helicina, a key species of Arctic ecosystems. Pteropods were kept under pH conditions corresponding to pCO2 levels of 350 and 760 μatm. The pteropods had a 28 % decrease in calcification at the higher pCO2 (expected for the year 2100), supporting concerns for their future in high CO2, as well as for species dependent on them for food (Comeau et al. 2009). A more comprehensive study (Comeau et al. 2010) examined pCO2 levels of 280, 380, 550, 760 and 1,020 μatm with ambient (control) and elevated (+4 °C) temperatures. Respiration was unaffected by pCO2 at control temperature, but significantly increased as a function of the pCO2 level at elevated temperature. Precipitation of CaCO3 declined as a function of pCO2 at both temperatures. The decrease in CaCO3 precipitation was highly correlated to the aragonite saturation state. Even though the pteropods could precipitate CaCO3 at low aragonite saturation state, their shell production is very sensitive to decreased pH. Comeau et al. (2012) found evidence for shell dissolution in this species at pH of 7.9. Bednarsek et al. (2012) found shell dissolution of this species is already happening. They examined L. helicina antarctica collected from the top 200 m of the water column, where aragonite saturation levels were around 1, and where upwelled deep water mixes with surface water with anthropogenic CO2. They compared the shell structure under a scanning electron microscope with samples from aragonite-supersaturated regions, and found severe shell dissolution in the undersaturated region. Laboratory incubations for 8 days in aragonite saturation levels of 0.94–1.12 produced equivalent levels of shell dissolution. Benthic mollusks are also affected. Gazeau et al. (2007) demonstrated that calcification rates of M. edulis and C. gigas decline linearly with increasing pCO2. Mussel and oyster calcification was projected to decrease by 25 and 10 %, respectively, by the end of the century. Gaylord et al. (2011) found that acidification degrades the mechanical integrity of larval shells of the mussel M. californianus, on rocky shores in the northeastern Pacific. Larvae cultured in seawater with CO2 of 540 or 970 mg l−1 produced weaker, thinner and smaller shells and lower tissue mass than those raised under current conditions. M. edulis adults exposed to elevated pCO2 decreased shell growth and exhibited internal shell corrosion of the nacreous layer at high pCO2 (240 and 405 Pa) (Fig. 8.5) (Melzner et al. 2012). Thus the inner shell surface is vulnerable in mussels, especially under conditions of low food.
(a) Mussel Shell length growth during 7 weeks (b) Internal shell surface dissolution. Low food (white) and high food (black) groups, mean ± SEM (Reprinted from Melzner et al. 2012 (open access))
Nienhuis et al. (2010) tested the effects of increased pCO2 (forecast to occur in 100 and 200 years) on shell deposition and dissolution in the rocky intertidal snail, Nucella lamellosa. Shell weight gain decreased linearly with increasing pCO2 levels. However, this trend was paralleled by weight loss of empty shells, suggesting that the declines in shell weight gain in live snails were due to dissolution of existing shell, rather than reduced production of new shell material. Acidification may therefore have a greater effect on shell dissolution than deposition, at least in this species. Range et al. (2012) found some similar results with mussels. The carbonate chemistry of Rio Formosa water was manipulated by diffusing CO2, to attain two reduced pH levels, by −0.3 and −0.6 pH units, relative to control seawater. After 84 days, no differences were in seen in growth (somatic or shell) of juvenile mussels M. galloprovincialis. The naturally elevated total alkalinity of the seawater prevented under-saturation of CaCO3. However, calcification was reduced in elevated CO2, but most of the loss of shell probably occurred as post-deposition dissolution in the internal aragonite nacre layer. The results show that, even under extreme levels of acidification, juvenile M. galloprovincialis continued to calcify and grow in this coastal lagoon. Melatunan et al. (2013) studied effects pH levels of 8.0 and 7.7 and temperatures of 15 or 20 °C on shell growth of Littorina littorea. Snails in acidified seawater, elevated temperature, or both had reduced shell growth. The increase in shell length was lower for individuals kept in low pH + high temperature, and the shell thickness increase at the growing edge was lower under low pH and combined conditions. ATP was positively correlated with shell thickening and weight, indicating that effects of low pH and elevated temperature may occur in part through metabolic disruption.
Shellfish with weakened shells can become more susceptible to predation and bioerosion. Amaral et al. (2012a) investigated the susceptibility of oysters from acidified areas (receiving runoff from acid sulphate soils) and reference areas to predation by the gastropod Morula marginalba. Shells were significantly weaker at acidified sites than at reference sites (Amaral et al. 2012b), and more oysters were consumed because M. marginalba needed less time to drill through the weaker shells. The boring sponge Cliona celata was grown on scallop (Argopecten irradians) shells in seawater at current pH and that predicted for 2100 (7.8) (Duckworth and Peterson 2013). Lower pH greatly affected shell boring. At pH = 7.8, sponges bored twice the number of holes and removed twice as much shell weight than at pH = 8.1, showing that OA may increase boring rates of C. celata in shellfish with weaker shells. Green crabs Carcinus maenas and periwinkles Littorina littorea were put under ambient conditions or warming and acidification, both separately and in combination, for 5 months and predators, prey, and their interactions were examined (Landes and Zimmer 2012). Acidification reduced the claw strength in C. maenas and weakened the shells of L. littorea. Predator–prey interactions were not changed, indicating that both species were affected equally. One would not expect that to be the case in most predator/prey interactions, however.
In the Pacific Coast of North America, because of patterns of ocean circulation, shellfish are already suffering from changes in ocean chemistry. Colder, more acidic waters well up from the depths and move inshore in bays and estuaries of Oregon, Washington, and British Columbia, causing damage to oysters (Feely et al. 2008). Wild oysters in many sites have failed to reproduce successfully because acidic waters have prevented larvae from forming shells. Oyster hatcheries have adapted to the acidity by buffering the water in which they grow their larvae, by providing the oysters with sodium bicarbonate.
In contrast to the response of most mollusks, calcification rates in the cephalopod Sepia officinalis increased during long-term exposure to elevated pCO2 (Gutowska et al. 2010). The potential negative impact of increased calcification in the cuttlebone of S. officinalis was discussed with regard to its function as a lightweight and porous buoyancy regulation device. The response of the nautilus, a cephalopod with an outer shell, is very different. In Argonauta nodosa, females construct a very thin (225 μm) shell that lacks an outer protective layer and that is used as a brood chamber for developing embryos. Wolfe et al. (2012) immersed shell fragments at different temperatures and pH. Shells incubated in pH 7.8 (projected for 2070) for 14 days had reduced weight due to dissolution, with increased dissolution in warmer and lower pH treatments. Unless living animals respond with increased mineralization, the brood chamber will be susceptible to dissolution under ocean acidification, which could compromise survival of the species, according to the authors.
The diversity of responses among species prevents clear predictions about ecosystem level impacts of acidification. Kroeker et al. (2011) used shallow water CO2 vents as a model system to examine ecosystem responses to acidification in rocky reefs. They found fewer taxa, reduced taxonomic evenness, and lower biomass in low pH zones. However, the number of individuals did not differ, suggesting that there is compensation via population increases of acid-tolerant taxa. The trophic structure shifted to fewer trophic groups and dominance by generalists, suggesting a simplification of food webs.
3.2.4 Polluted Sites
Blue mussels, M. edulis, were exposed to dredged material from a polluted site, Black Rock Harbor, Connecticut (US) at 0–10 mg l−1 of suspended sediment (Nelson 1987). At >1.5 mg l−1 mussels showed reduced scope for growth and shell growth. Exposure in the field to lower concentrations of dredged material produced no noticeable effects.
3.3 Fishes
There have been far fewer papers studying effects of contaminants on bone development in fishes. Of those studies that have been done, many are on freshwater species.
3.3.1 Metals
Lead has particular toxicity causing weakened bones in fishes, but this has been seen primarily in fresh water systems where the pH is low, allowing more Pb to be dissolved and bioavailable to fish (Hamilton and Haines 1989).
3.3.2 Organics
After a spill of the pesticide kepone in the James River Virginia, effects on the skeletal system were seen in spot (Leiostomus xanthurus), croaker (Micropogonias undulatus), and black drum (Pogonias cromis), which had shortened vertebral columns and “broken back” syndrome due to vertebral fractures (Davis 1997). This could be demonstrated in the laboratory (Couch et al. 1979) with kepone, as well as other compounds such as trifluralin, dibutylphthalate, toxaphene, PCBs, and toxaphene (Karen et al. 1998, 2001).
Striped bass (Morone saxatilis) from estuaries along the East Coast were examined for body burdens of selected contaminants and bone strength (vertebral mechanical properties) (Mehrle et al. 1982). Fish from the Hudson River, the most polluted site, had the weakest vertebrae. PCBs were the most prevalent contaminant. Authors proposed a mode of action involving competition for vitamin C between collagen in bone and MFOs (mixed-function oxidases) that detoxify the contaminants. The competition for vitamin C causes a decrease in bone vitamin C and reduces collagen content, with an increase in bone minerals and the ratio of minerals:collagen that renders the backbone more fragile. Sea bass (Dicentrarchus labrax) were exposed to the soluble fraction of light cycle oil (1,136 ng l−1) for 7 days which did not affect the frequency and severity of vertebral abnormalities, but decreased mineralization of vertebrae in a reversible way, indicating that it is an early stress indicator (Danion et al. 2011).
3.4 Other Taxa
3.4.1 Foraminifera
Foraminifera are single-celled protists with a calcified spiral shell. Prazeres et al. (2011) investigated effects of Zn in the symbiont-bearing foraminifer Amphistegina lessonii. Forams were acutely (48 h) exposed to dissolved Zn (9.5–93.4 μg Zn l–1). Many individuals showed white spots and/or dark-brown areas in the test after 24 and 48 h, with a positive correlation between this endpoint and dissolved Zn. Increases in lipid peroxidation and metallothionein-like protein were observed, particularly in pale/partly-bleached individuals. Denoyelle et al. (2012) developed a chronic bioassay by incubating forams for 30 days in seawater with Cd, fuel Oil #2, or drilling muds. Responses included pseudopodal activity and number of newly built chambers. Experiments were conducted in a solution of calcein in seawater, so that foraminiferal tests with newly added chambers could be seen by an epifluorescence microscope. The forams had a strong response to incubation with high concentrations of all tested pollutants.
Not surprisingly, acidification has negative effects on foram shells. McIntyre-Wressnig et al. (2013) assessed effects of elevated pCO2 on the survival, fitness, shell microfabric and growth of Amphistegina gibbosa, a symbiont-bearing, benthic coral-reef species that precipitates low-Mg calcite tests. Specimens were cultured in controlled pCO2 (ambient, 1,000 ppm by volume [ppmv], and 2,000 ppmv) for 6 weeks. Fitness and survival were not affected. While test growth was not affected by elevated pCO2, areas of dissolution were observed in small, well defined patches distributed over the whole test surface. Similar dissolution was observed in offspring produced in the 2,000 ppmv pCO2 treatments.
3.4.2 Echinoderms
Ossicles of the sea urchin Echinus acutus and the sea star Asterias rubens were studied in stations along a metal contamination gradient. Ossicles with major mechanical functions – sea urchin spines and sea star ambulacral plates – were analyzed for metals and mechanical properties. Sea star plates were more contaminated by Cd, Pb, and Zn than sea urchin spines, and their stiffness and toughness decreased in the most contaminated station (Moureaux et al. 2011). This was attributed either to the incorporation of metals in the calcite lattice and/or to deleterious effects of metals during skeletogenesis.
Ries et al. (2009) tested various marine organisms under different pCO2 levels and found that in purple sea urchins (Eucidaris tribuloides) net calcification increased relative to the control under intermediate pCO2 levels (605 and 903 mg l−1), and then declined at the highest pCO2 level (2,856 mg l−1), and spines deteriorated. Urchins accrete shell that is covered by an external organic layer, which may give them more resilience to elevated pCO2 than species producing shell that is directly exposed to ambient water. Wood et al. (2008) found that the brittle star, Amphiura filiformis, increased its rate of metabolism and ability to calcify to compensate for increased seawater acidity. However, the up-regulation of metabolism and calcification, potentially ameliorating some of the effects of increased acidity, came at a substantial cost, namely muscle wastage.
3.4.3 Corals
3.4.3.1 Organics
Jackson et al. (1989) and Guzmán et al. (1994) studied effects of a Panamanian oil spill on injury, growth, and regeneration of corals over 5 years. The spill initially caused bleaching, tissue swelling, mucus production, and dead areas, even in subtidal reefs that had not been in contact with the oil. Corals from heavily oiled reefs had higher levels of injury, faster regeneration but slower growth. Hydrocarbons in reef sediments were correlated with the degree of injury and negatively correlated with growth (Fig. 8.6). The probable cause of persistently high injury was chronic exposure to sediments mixed with partially degraded oil that were exported from mangroves onto reefs. Injury resulted in a reallocation of resources to regeneration and decreased investment in fecundity and growth. There was no evidence of coral recovery 5 years after the spill.
Siderastrea siderea and Porites astreoides. Relation between growth rates and oil in reef sediments in 1986 and 1988. Symbols connote specific reefs (Reprinted from Guzman et al. 1994: 239, courtesy Inter-Research)
Shafir et al. (2007) used a “nubbin assay” on coral fragments to evaluate short- and long-term impacts of dispersed oil from six commercial dispersants, the dispersants themselves, and water-soluble-fractions (WSFs) of Egyptian crude oil, on two branching corals, Stylophora pistillata and Pocillopora damicornis. Survival and growth of nubbins were recorded for up to 50 days after a single, 24 h exposure to various concentrations. Manufacturer-recommended dispersant concentrations were highly toxic and caused mortality for all nubbins. Dispersed oil and the dispersants were significantly more toxic than crude oil WSFs. As corals are particularly susceptible to dispersants and dispersed oil, authors felt their results rule out the use of dispersants near coral reefs.
3.4.3.2 Ocean Acidification/Climate Change
Corals are particularly sensitive to effects of climate change and acidification, and it is predicted by some that within decades the rates of reef erosion will exceed rates of accretion throughout much of the tropics and subtropics. In addition to acidification reducing calcification, rising temperatures trigger bleaching, the loss of symbiotic microalgae from the coral, doubly stressing the corals. Loss of corals reduces habitat for associated reef species and the whole reef community can be degraded. However some reef species may benefit from weakened coral skeletons; bioeroding sponges such as Cliona orientalis normally erode coral skeletons. Wisshak et al. (2012) found a clear relationship between the pH of the seawater and the bioerosion rate of the sponges. Thus, with increasing ocean acidification corals will be exposed to a double stress: calcification will be more difficult, and existing skeletons will be weakened more by bioerosion.
Pandolfi et al. (2011) reviewed historical data on past climate change and found that of the five major episodes of severe “reef crises,” four coincided with periods of ocean acidification. However, there is considerable variation among different types of corals, with some being much more resistant than others. An inventory of coral growth from Pacific Panama shows that declines have occurred in some, but not all species (Manzello 2010). Growth declined significantly in the most important reef builder of the eastern tropical Pacific, Pocillopora damicornis, by nearly one-third from 1974 to 2006. The rate of decline in skeletal extension for P. damicornis from Pacific Panama (0.9 % year−1) was comparable to Porites in the Indo-Pacific over the past 20–30 years (0.89–1.23 % year−1). Branching pocilloporid corals show increased tolerance to thermal stress, but appear to be very susceptible to OA. In contrast, the massive pavonid corals are relatively tolerant to both thermal stress and to OA.
Ries et al., (2010) investigated impacts of OA on the temperate coral Oculina arbuscula by rearing colonies for 60 days in chambers bubbled with air-CO2 gas mixtures of varying pCO2. Measurement of calcification and linear extension revealed that skeletal accretion was minimally impaired; the corals continued accreting new skeletal material, although at reduced rates. Correlation between rates of linear extension and calcification suggests that reduced calcification resulted from reduced aragonite accretion, rather than from dissolution. Accretion of pure aragonite under each condition discounts the possibility that corals will begin producing calcite, a less soluble form of CaCO3, as oceans acidify. The corals’ nonlinear responses and their ability to accrete new skeletal material suggest that they can control the biomineralization process.
Calcification was measured in the Mediterranean cold-water scleractinian coral (CWC) Madrepora oculata in sea water reduced or enriched in pCO2 (Maier et al. 2012). Calcification rates were the same for ambient and elevated pCO2 (404 and 867 μatm) with 0.06 ± 0.06 % day−1, while calcification was 0.12 ± 0.06 % day−1 when pCO2 was reduced to its pre-industrial level (285 μatm), suggesting that present-day CWC calcification in the Mediterranean Sea has already drastically declined by 50 % due to OA.
Physiological data and models of coral calcification indicate that corals utilize a combination of seawater bicarbonate and respiratory CO2 for calcification, rather than seawater carbonate. However, a number of investigators attribute effects of experimental acidification by CO2 or hydrochloric acid additions to a reduction in seawater carbonate ion concentration and thus aragonite saturation state (Jury et al. 2010). Thus, there is a discrepancy between the physiological and geochemical views of coral biomineralization. Furthermore, not all calcifiers respond negatively to decreased pH or saturation state. Madracis corals responded strongly to variation in bicarbonate concentration, but not consistently to carbonate concentration, aragonite saturation state, or pH. Corals calcified at normal or elevated rates under low pH (7.6–7.8) when the seawater bicarbonate was above 1,800 μm. Conversely, corals incubated at normal pH had low calcification rates if the bicarbonate concentration was lowered.
There may be differences in the energetic costs of calcification among different corals. Pandolfi et al. (2011) stated that corals probably utilize all forms of seawater carbon, using enzyme and proton pumps to convert CO2 and HCO3 − to carbonate ions for rapid calcification, but this process is energetically costly; well-nourished corals are better equipped to do this in a high CO2 environment. However, poorly nourished or energetically depleted corals (including bleached ones) are more sensitive to acidification. Evolutionary adaptation and phenotypic plasticity in thermal tolerance may also buffer populations of corals, as will be discussed further in Chap. 11.
The role of zooxanthellae was further investigated by Holcomb et al. (2012) in Astrangia poculata. Elevated pCO2 appeared to have a similar effect on calcification whether or not zooxanthellae were present at 16 °C. However, at 24 °C, corals spawned and there was a sex difference in response. Females exposed to elevated pCO2 had calcification rates 39 % lower than females at ambient pCO2, while males showed a non-significant decline. The increased sensitivity of females may reflect a greater investment of energy in reproduction. Thus, sex and spawning affect the sensitivity to acidification.
Thresher et al. (2011) examined the distribution and skeletal characteristics of coral taxa along a natural deep-sea concentration gradient on seamounts of SW Australia. Carbonate undersaturation had little effect on the depth distribution, growth or skeletal composition of live scleractinians or gorgonians, with corals growing, often abundantly, in waters as much as 20–30 % under-saturated. Evidence for an effect of acidification on the accumulation of reef structure was not clear. Abundant old scleractinian skeletons were present well below the aragonite saturation horizon, although this might have been the result of ferromanganese deposition on exposed skeletons.
The exact responses of coral reefs to pH changes are uncertain. Naturally low pH (6.70–7.30) groundwater normally discharges at localized submarine springs at Puerto Morelos, Mexico, and gives insight into potential long term responses of coral ecosystems to low pH. Crook et al. (2012) found species richness and colony size declined with increasing proximity to low-pH water. Iguchi et al. (2012) found that the massive coral, Porites australiensis decreased calcification and fluorescence yield (reflecting photosynthesis) in acidified seawater, but that zooxanthellar density was unchanged, unlike in Acropora species. Thus responses are quite variable among different corals.
A compilation of studies by Edmunds et al. (2012) showed great variation in calcification as a function of pH, [HCO3 −], and [CO3 2−], and concluded that studies of the effects of OA on corals need to pay closer attention to reducing variance in experimental outcomes to achieve stronger synthetic capacity; coral genera respond in dissimilar ways to pH, [HCO3 −], and [CO3 2−]; and calcification of massive Porites spp. is relatively resistant to short exposures of increased pCO2, similar to that expected within 100 years.
3.4.4 Coccolithophores/Calcareous Algae
Coccolithophores are phytoplankton with calcified tests, and are susceptible to OA. A significant effect of pCO2 and temperature on calcification was found by De Bodt et al. (2010) on Emiliania huxleyi. Coccosphere particles were smaller at higher temperature and CO2. The number of well-formed coccoliths decreased with increasing pCO2 but temperature did not affect morphology. In natural ecosystems, there is a shift in species composition from strongly to weakly calcified species and strains. As OA increases, species that have to invest more energy to form their calcite skeleton may be displaced (Beaufort et al. 2011). However, in the coastal zone of Chile, where currently acidic conditions prevail (pH 7.6–7.9 instead of 8.1 on average), highly calcified individuals were found; a strain of E. huxleyi has evolved there that is resistant to acidification. Furthermore, Smith et al. (2012) studied coccolithophore morphology in the Bay of Biscay and found seasonality in the morphotypes of E. huxleyi, the most abundant species. While pH and CaCO3 saturation are lowest in winter, the E. huxleyi population shifts from <10 % (summer) to >90 % (winter) of the heavily calcified form. The finding that the most heavily calcified form dominates under acidic conditions is contrary to the earlier predictions and raises questions about responses of coccolithophores to high pCO2.
Larger calcareous algae may also be affected by ocean acidification. Price et al. (2011) investigated potential effects on growth, calcification and photophysiology of two species of reef macroalgae, and found Halimeda opuntia had net dissolution and 15 % reduction in photosynthetic capacity, while H. taenicola did not calcify but did not alter its photophysiology. The different responses of the two species may be due to anatomical and physiological differences and could represent a future shift in their relative dominance. Ries et al. (2009) found that calcifying red algae (Neogoniolithon) grew better at 600 mg l−1 than at 300 mg l−1CO2, but exhibited reduced calcification at higher pCO2 levels. Johnson and Carpenter (2012) investigated effects of elevated pCO2 and temperature on calcification of Hydrolithon onkodes, a coralline alga, and subsequent effects on susceptibility to grazing by sea urchins. H. onkodes was exposed for 21 days to a combination of pCO2 (420, 530, 830 μatm) and temperature (26, 29 °C). They found increased calcification in moderately elevated pCO2, similar to Ries et al. (2009), and reduced calcification at higher p CO2. There was a trend for highest calcification at ambient temperature. When H. onkodes was exposed to the sea urchin Echinothrix diadema, grazing removed 60 % more algae grown at high temperature and high pCO2 than at ambient temperature and low pCO2. The increased susceptibility to grazing was considered early evidence of the potential for cascading effects of OA and temperature on coral reef ecosystems. However, the possibility that feeding by E. diadema might also be impaired after spending 21 days in these conditions did not seem to be considered.
Since calcifying algae are impaired by acidification, they can be outcompeted by non-calcifying species. Hofmann et al. (2012) examined Corallina officinalis (calcifying) and Chondrus crispus (noncalcifying) and found an interactive effect of CO2 concentration and exposure time on growth rates of C. officinalis. Community structure changed, as Chondrus crispus cover increased in all treatments (385, 665, and 1,486 μatm pCO2) while C. officinalis cover decreased in both elevated-pCO2 treatments.
4 Carcinogenesis
Cancer is a developmental process gone awry – it is uncontrolled growth of cells. Normal cells multiply when the body needs them, stop dividing when growth is complete, and die when no longer needed. Cancer occurs when cells divide and grow uncontrollably, forming malignant tumors or neoplasms, which may invade nearby parts of the body. The cancer may also spread to more distant parts of the body via cells that break off and enter the blood to be transported to other organs. Not all tumors are cancerous, however. Benign tumors do not grow uncontrollably, do not invade neighboring tissues, and do not spread through the body. The development of cancer is often associated with exposure to certain chemicals, termed carcinogens, in both humans, and marine animals.
4.1 Mollusks
A number of laboratory and field studies have associated environmental chemicals with neoplasms in shellfish (Yevich and Barszcz 1977). The soft shell clam Mya arenaria from chronically oil-polluted sites had elevated levels of gonadal and hematopoeitic neoplasms. Similar findings were seen in soft shell clams from an oil spill site in Massachusetts (Brown et al. 1977), in Macoma balthica from areas of Chesapeake Bay (Christensen et al. 1974), and others (Sindermann 1979). Oysters (C. virginica) developed neoplasms when exposed in the laboratory or field to contaminated sediment from Black Rock Harbor (BRH), Bridgeport, Connecticut (Gardiner et al. 1991). Neoplasia was seen after 30- and 60-days laboratory exposure to a 20 mg l−1 suspension of BRH sediment with postexposure for 0, 30, or 60 days. Composite tumor incidence was 13.6 %. Tumors were most prevalent in the renal epithelium, followed by gill, gonad, gastrointestinal, heart, and neural tissue; tumors did not regress when oysters were placed in clean sediments. Gill neoplasms developed in oysters deployed for 30 days at BRH and 36 days at a BRH dredge material disposal area, and kidney and gastrointestinal neoplasms developed in oysters caged 40 days in Boston Harbor. Exposed oysters accumulated high concentrations of PCBs, PAHs, and chlorinated pesticides. Gonad tumors and siphon anomalies were seen in Mya arenaria at sites in New England with widespread use of the herbicides Tordon 101, 2,4-D,2,4,5-T, and other agrochemicals (Gardner et al. 1991).
The general hypothesis has been that cancers are caused by chemical pollutants. However, when the occurrence of mollusk neoplasms was reviewed by Mix (1988), who examined neoplastic diseases in shellfish from around the world, results supporting this hypothesis were not prevalent in the literature. Some studies found shellfish with no neoplasms in highly polluted environments and others found neoplastic diseases in pristine areas. Mix concluded that more research is needed to better understand the association between chemicals and neoplasia in shellfish. However, there has been little research since then to lead to this understanding. Wolowicz et al. (2005) thought pollution by carcinogens was implicated in the Gulf of Gdańsk, southern Baltic Sea. A higher prevalence of mollusk tumors was observed in infaunal deposit/suspension feeders compared to epifaunal suspension-feeders, providing new ecological insights into the genesis of the neoplasia. They discussed a relationship between sediment factors and the incidence of neoplasia across a range of environmental conditions.
4.2 Fishes
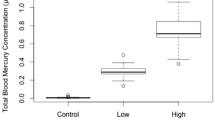
There have been numerous reports of liver tumors in fishes from contaminated sites around the world. Fish in the laboratory can develop tumors after treatment with a variety of chemical carcinogens (Couch and Harshbarger 1985). The field was reviewed by Bauman (1992) who concluded that some types of tumors, but only neoplasms that have chemicals as a portion of their etiology, would be useful in assessing ecosystem health. Lesions which may fit these criteria include liver neoplasms and skin lesions in a variety of primarily benthic fishes, and neural lesions in various drum species and butterflyfish. There is a correlation with habitat degradation and length of time of residence in a contaminated site. Bottom-dwelling fishes in contact with contaminated sediments appear to be most vulnerable. Winter flounder (P. americanus) from degraded east coast estuaries had tumors (Murchelano and Wolke 1985). Winter flounder fed BRH-contaminated (see above) blue mussels contained xenobiotic chemicals analyzed in mussels (PCBs, PAHs, chlorinated pesticides), and developed renal and pancreatic neoplasms and hepatic neoplastic precursor lesions, demonstrating trophic transfer of sediment-bound carcinogens up the food chain (Gardiner et al. 1991). Hepatic lesions in fish from Boston Harbor were correlated with chlorinated hydrocarbons in the liver (Moore et al. 1996). They have been decreasing over time along with the level of pollution in Boston Harbor (Fig. 8.7).
Cumulative graph of winter flounder lesion prevalence 1987–1993 from Deer Island, Boston Harbor. Mean length and age for each year are shown as line plots to indicate between-year comparability of samples (Reprinted from Moore et al. 1996: 464, courtesy Elsevier Publishing Co.)
Tumors in West Coast flatfish (English sole, Parophrys vetulus) were also associated with contaminants (Malins et al. 1984, 1988); Sole with the highest frequencies of liver neoplasms were from the urban Duwamish Waterway (16 %) and Everett Harbor (12 %), while frequencies in sole from other areas ranged from 0 to 5.5 %. Sediment PAH was significantly correlated with tumors. Metals correlated with total (including non-neoplastic) hepatic lesions. Myers et al. (1990) reported that liver lesions of English sole in Puget Sound represent morphologically identifiable steps leading to the development of neoplasms, similar to carcinogenesis in rodents. The view that these lesions are caused by exposure to chemicals in Puget Sound is based on statistical associations between levels of aromatic hydrocarbons (AH) in sediment and prevalence of lesions, the contribution of AHs in accounting for the variability in neoplasm prevalence in a regression model, correlations between lesions and levels of metabolites of aromatic compounds in bile, and experimental induction of lesions in fish injected with extracts of Puget Sound sediments. These induced lesions were structurally identical to those seen in wild fish from the same site. Since remediation (sediment capping) of highly contaminated areas, there has been a trend of decreasing lesions in English sole (Myers et al. 2008). Lesion risk has been consistently low (<0.20), showing that sediment capping was effective in reducing AH exposure and cancers in resident flatfish.
The Atlantic tomcod, Microgadus tomcod, is a bottom-dwelling anadromous species in estuaries including the Hudson River (HR), which contains high concentrations of sediment-bound contaminants including PCBs and PAHs. The HR population was reported to have a high level of liver tumors (up to 90 % in 2-year old fish) compared to only 5 % in fish from other estuaries (Dey et al. 1993). These tomcod have elevated levels of CYP1A mRNA, hydrophobic DNA adducts, and elevated PCBs in their tissues, suggesting that DNA damage by carcinogens is occurring (Wirgin et al. 1994). Even the highly tolerant killifish (mummichog) Fundulus heteroclitus, develops liver tumors if the environment is toxic enough. Fish from a creosote-contaminated site in the Elizabeth River, VA (sediments at 2,200 mg kg−1 dry wt PAHs from a wood treatment facility) had liver tumors (Figs. 8.8 and 8.9) in about one third of the population (Vogelbein et al. 1990). There has been a recent report of skin cancer in fish from Australian waters in the area with the “ozone hole” and thus high exposure to UV light. Sweet et al. (2012) found extensive melanosis and melanoma in wild populations of the commercially-important coral trout, Plectropomus leopardus. The syndrome was similar to previous studies associated with UV induced melanomas in the platyfish, Xiphophorus. Relatively high prevalence rates (15 %) were found at sites in the Great Barrier Reef. Authors concluded that in the absence of microbial pathogens and given the strong similarities to UV-induced melanomas in platyfish, the likely cause was environmental exposure to UV radiation. Further studies are needed to establish the distribution of the syndrome.
Gross liver pathology and normal histology from mummichog from (a) reference site (GB gall bladder), (b) liver of fish from Station 3 exhibiting tan focal lesions (c) liver of fish from Station 3 exhibiting many focal lesions and one 8 mm diameter hepatocellular carcinoma (arrow). (d) normal hepatocellular structure of fish from reference site (Reprinted from Vogelbein et al. 1990: 5980, courtesy American Association for Cancer Research)
Hepatocellular carcinomas in mummichogs from creosote-contaminated site. (a) carcinoma with invasive border, (b) higher magnification of carcinoma (c) less well differentiated hepatocellular carcinoma (d) less well differentiated hepatocellular carcinoma with epithelial-appearing tumor cells (e) poorly differentiated hepatocellular carcinoma (f) poorly differentiated anaplastic hepatocellular carcinoma with epithelial-appearing cells (Reprinted from Vogelbein et al. 1990: 5983, courtesy American Association for Cancer Research)
5 Smoltification
In salmonid fishes that breed in streams, larvae hatch out and live in freshwater for a period of time and then undergo further developmental processes to prepare them for their seaward migration and life in salt water. Newly hatched larvae (alevins) remain in the gravel and utilize the yolk sac as they grow into the fry stage, at which point they emerge from the gravel and feed on insects and zooplankton. As they grow to about 10 cm long, they utilize deeper water and become known as parr, which can remain in freshwater for several years. Then they become smolts, when they begin to prepare for downstream migration. An essential part of this development involves osmoregulation and development of gill enzymes carbonic anhydrase (CA) and Na+K+-ATPase. The regulation of smoltification requires environmental factors such as photoperiod and temperature, and endogenous factors, namely hormones. Thyroid hormones and growth hormone peak during smoltification. Treatment with either hormone can stimulate some of the changes associated with smoltification. Thus, these changes are susceptible to some endocrine disruptors. Smolts have a limited period of readiness (“physiological smolt window”) for entering salt water, and the timing of sea water entry can be adversely affected by pollution.
5.1 Metals
Unlike seawater, freshwater pH is normally close to neutral, around 6.5–7.0. In freshwater systems affected by acid rain, pH decreases and aluminum is released from sediments to which it is usually tightly bound and unavailable. Under these conditions Al becomes toxic. Staurnes et al. (1993) exposed smolting Atlantic salmon (Salmo salar) to acid water (pH 5 or pH 5 plus 50 μg Al l−1), which resulted in osmoregulatory failure and mortality. Al strongly enhanced toxicity. Sensitivity to low pH or low pH + Al exposure greatly increased when fish had developed to seawater tolerant smolts. In Al-exposed fish, gill CA activity decreased. Gill Na+K+-ATPase activity in control fish peaked in mid-May simultaneously with development of seawater tolerance. Fish from both exposed groups had low seawater tolerance and reduced Na+, K+-ATPase activity, which declined to 60 % in acid-exposed fish and even lower in Al-exposed fish. Kroglund et al. (2007) exposed 1-year old hatchery reared S. salar smolts to water of pH 5.8 from 3 to 60 days. Fish exposed to Lake Imsa water (pH > 6.5) acted as controls. Control fish had gill-Al concentrations of 5–10 μg Al g−1 gill dry weight (dw), while Al-exposed fish had gill-Al concentrations >20 μg Al g−1 gill dw prior to seawater release. Gill Na+, K+-ATPase activity was depressed in all groups having >25 μg Al g−1 gill dw. Following exposure, the smolts were released to monitor downstream migration and ocean return rates. Return rates were reduced by 20–50 % in all Al-exposed groups.
5.2 Pesticides and PCBs
Mortensen and Arukwe (2006) studied effects of 1,1-dichloro-2,2-bis(p-chlorophenyl) ethylene (DDE) on the thyroid and steroid-metabolizing system in Atlantic salmon parr. Fish were exposed for 5 days to waterborne DDE and thyroxine (T4), both singly and in combination. Thyroid-stimulating hormone (TSHβ), T4 deiodinase (T4ORD), thyroid receptors (TRα and TRβ), and insulin-like growth factor type 1 receptor (IGF-1R) were analyzed. Results indicated that DDE alters thyroid hormone–dependent genes and hepatic CYP3A and PXR levels, demonstrating possible physiological and endocrine consequences from exposure to endocrine-disrupting chemicals during smoltification.
Many investigators have examined effects of PCBs on smoltification. Folmar et al. (1982) injected yearling coho salmon (Oncorhynchus kisutch) with 150 μg kg−1 of Aroclor 1254 just prior to smoltification. Alterations in the normal developmental patterns of T4 were observed in PCB-injected fish. At sea-water entry, one-half of each group (experimentals and controls) were put in seawater and the other half into seawater with 700 μg l−1 No. 2 fuel oil. Significant mortalities were observed in all treatment groups. Mortalities in the PCB-injected, fuel oil-exposed fish appeared additive compared with PCB-injected-only or fuel oil-exposed-only fish. Arctic charr (Salvelinus alpinus) were orally dosed with 0, 1, or 100 mg Aroclor 1,254 kg−1 body mass (High Dose, HD) in November (Jørgensen et al. 2004). They were then held in freshwater until they smolted in June the next year when they were transferred to seawater. HD charr had reduced plasma growth hormone, insulin-like growth factor-1, and thyroxin and triiodothyronine titers during smoltification. The hormonal alterations corresponded with impaired hyposmoregulatory ability, and reduced growth rate and survival in seawater.
Lerner et al. (2007) examined effects of aqueous Aroclor 1254 (1 μg l−1(PCB-1) or 10 μg l−1 (PCB-10)) on Atlantic salmon after 21 days exposure either as yolk-sac larvae or as juveniles just prior to the parr–smolt transformation. After exposure, yolk-sac larvae were reared at ambient conditions for 1 year, until smolting the following May. Juveniles were sampled immediately after exposure. At smolting, prior exposure to PCB -1 as larvae did not affect behavior, while PCB-10 dramatically decreased preference for seawater. There were no long-term effects on osmoregulation or hormones of fish exposed as larvae. Juveniles exposed to PCB-1 or PCB-10 showed a dose-dependent reduction in preference for seawater. Fish treated with PCB-10 had a 50 % decrease in gill Na+, K+-ATPase. In addition, plasma T3 was reduced 35–50 % and plasma cortisol 58 % in response to either concentration. Thus, effects vary according to developmental stage. Exposure to PCBs in freshwater can inhibit changes that occur during smolting, and reduce marine survival.
5.3 Contaminants of Emerging Concern (CECs)
Bangsgaard et al. (2006) exposed Atlantic salmon parr or pre-smolts to estradiol-17β (E2 conc.: nominal 500 ng l−1/actual 8–16 ng l−1) and tert-octylphenol (OP: nominal 25 μg l−1/actual 4.5–6.5 μg l−1 and OP: nominal 100 μg l−1/actual 10–30 μg l−1) for 26 days in freshwater, and studied effects on parr-smolt transformation. Vitellogenesis was induced by all treatments, and elevated VTG levels were still found 4–5 months after treatment. Smolting was impaired by E2 and OP-100 as judged by reduced gill Na+, K+-ATPase and reduced ability to regulate plasma osmolality and muscle water content in 24-h seawater (SW) challenge tests during smolting. Downstream migration was monitored by implanting passive integrated transponder tags into smolts and placing them in a stream raceway. Downstream movement was initiated in all groups, but E2 and OP-100 fish migrated at lower frequency than controls, suggesting that xenoestrogens reduce physiological and behavioral components of smoltification, even when exposure occurred several months prior to smolting.
Madson et al. (2004) gave Atlantic salmon serial injections over 20 days of 2 μg/g body weight estradiol (E2), 120 μg/g nonylphenol (4-NP) in peanut oil or peanut oil as control. After the last injection, fish were individually tagged. Two days later 100 fish per group were released into a small stream. Migration was evaluated by measuring arrival time at a trap downstream. Serum VTG levels increased several-fold in both male and female E2- and 4-NP-treated fish. Overall, E2- and 4-NP-treatment impaired smolting as judged by reduced gill Na+, K+-ATPase activity and α-subunit Na+, K+-ATPase mRNA level, reduced muscle water content and increased mortality following 24 h SW-challenge. After release, control fish initiated downstream migration immediately, with 50 % of them appearing in the trap within 10 days. E2- and 4-NP-treated fish appeared in the trap, with a delay of 6 and 8 days, respectively. The total number of fish reaching the trap was control (81 %), E2 (53 %), 4-NP (12 %), indicating that short-term exposure to environmental estrogens impaired smoltification, survival, and downstream migration.
Keen et al. (2005) studied 4-week dietary exposure of coho salmon (Oncorhynchus kisutch) to nonylphenol (4-NP) during the parr-smolt transformation. Doses varied between 0 (control) and 2,000 mg/kg after which fish were transferred to sea water, when all groups were fed the control diet. Dietary treatment of 4-NP did not influence growth or smoltification of coho salmon, a result that conflicts to some extent with other reports (e.g. Madson et al., above) in which deleterious effects of 4-NP were linked to disruption of the endocrine system. Differences may be due to species, concentration, route of exposure, timing, or other factors. There have not been studies of other types of CECs besides environmental estrogens.
5.4 Acidification
Effects of low pH were discussed in relation to Al, above. There have also been studies of effects of low pH alone. Saunders et al. (1983) reared Atlantic salmon (Salmo salar) at pH 6.4–6.7 (control) and 4.2–4.7, from Feb. to June, to assess the effect on survival, growth, and smolting under rising (4–8.5 °C) or relatively constant (9.5–10.5 °C) temperature. Survival was lower in low pH under both temperature regimes. Neither group in low pH gained weight while both control groups did. Parr–smolt transformation, as indicated by salinity tolerance and gill Na+, K+-ATPase activity, was impaired by low pH. Controls increased their tolerance to 35 psu salinity between March and May but those in low pH did not. ATPase activity was greater in controls in rising than in constant temperature. Plasma chloride and sodium levels were reduced at low pH, indicating impaired ionic regulation in freshwater. Thyroid hormones (T3 and T4) gave no clear indication of effects, but smoltification did not proceed normally at low pH.
Haya et al. (1985) found effects of low pH on energy metabolism, an indirect way of affecting smoltification. Juvenile S. salar were held for 76 days at pH 4.7 during the time when the final stages of smoltification normally occur. Controls at pH 6.5 had significant increases in weight, length and liver somatic index which were not seen in fish at low pH. After 15 days, ADP and glucose levels were higher and adenylate energy charge (AEC), and glycogen lower in muscles of fish at low pH; differences were maintained until the end of the experiment. ATP and total adenylate concentrations in muscle were lower after 62 days in low pH. Fish at low pH decreased their food intake, reducing growth.
6 Conclusions
This chapter has focused on developmental processes that take place in juvenile or adult animals, and the research findings have shown that developmental processes remain sensitive to contaminant effects throughout life. Effects on growth per se have been studied for a long time, and in many cases, reductions in growth can be traced back to reduced food intake (see Chap. 2). Reduced limb regeneration in crustaceans can be in some cases attributed to endocrine disruption of molting hormones, and impaired smoltification in salmon can also be endocrine-related. Carcinogenesis was a major research area in previous years, but studies have diminished along with much of the gross contamination that produced cancers in highly polluted systems. Reports of tumors in both fish and mollusk populations at contaminated sites have decreased considerably over the past 20 years. This may reflect improving environmental conditions due to reduction of contaminants in effluents and cessation of dumping of wastes into the ocean. However, it may also be that this type of research is not as popular as it once was. Some reports suggest that exposures to some types of contaminants are increasing (Hanson et al. 2009), probably due to increased rain and runoff from land. Recently, considerable attention is being devoted to effects of ocean acidification on deposition of calcareous structures (shells, bones) in a variety of marine species, with mollusks and corals being the taxa that appear to be at greatest risk.
References
Albright R, Bland C, Gillette P, Serafy JE, Langdon C, Capo TR (2012) Juvenile growth of the tropical sea urchin Lytechinus variegatus exposed to near-future ocean acidification scenarios. J Exp Mar Biol Ecol 426–427:12–17
Alzieu C (1986) TBT detrimental effects on oyster culture in France – evolution since antifouling paint restriction. In: Oceans 86, IEEE proceedings, vol 4. Marine Technology Society, Washington, DC, p 1132
Alzieu C, Sanjuan J, Deltreil JP, Borel M (1986) Tin contamination in Arcachon Bay: effects on oyster shell anomalies. Mar Pollut Bull 17:494–498
Amaral V, Cabral HN, Bishop MJ (2012a) Effects of estuarine acidification on predator–prey interactions. Mar Ecol Prog Ser 445:117–127
Amaral V, Cabral HN, Bishop MJ (2012b) Moderate acidification affects growth but not survival of 6-month-old oysters. Aquat Ecol 46:119–127
Anderson JW, Riley RG, Kiesser SL, Thomas BL, Fellingham OW (1981) Natural weathering of oil in marine sediments: tissue contamination and growth of the clam, Protothaca staminea. In: Technical report of the symposium on ocean pollution, Halifax, Nova Scotia, Canada, 19 Oct 1981, 25 pp
Baker RT, Handy RD, Davies SJ, Snook JC (1998) Chronic dietary exposure to copper affects growth, tissue lipid peroxidation, and metal composition of the grey mullet, Chelon labrosus. Mar Environ Res 45:357–365
Bangsgaard K, Madsen SS, Korsgaard B (2006) Effect of waterborne exposure to 4-tert-octylphenol and 17beta-estradiol on smoltification and downstream migration in Atlantic salmon, Salmo salar. Aquat Toxicol 80:23–32
Bauman PC (1992) The use of tumors in wild populations of fish to assess ecosystem health. J Aquat Ecosyst Health 1:135–146
Beaufort L et al (2011) Sensitivity of coccolithophores to carbonate chemistry and ocean acidification. Nature 476:80–83
Bednaršek N, Tarling GA, Bakker DCE, Fielding S, Jones EM, Venables HJ, Ward P, Kuzirian A, Lézé B, Feely RA, Murphy EJ (2012) Extensive dissolution of live pteropods in the Southern Ocean. Nat Geosci 5:881–885. doi:10.1038/ngeo1635
Bergey L, Weis JS (2007) Molting as a mechanism of depuration of metals in the fiddler crab, Uca pugnax. Mar Environ Res 64:556–562
Bodammer JE (2000) Some new observations on the cytopathology of fin erosion disease in winter flounder Pseudopleuronectes americanus. Dis Aquat Organ 40:51–65
Boyden CR, Watling H, Thornton I (1975) Effect of zinc on settlement of the oyster, Crassostrea gigas. Mar Biol 31:227–234
Brown RS, Wolke RE, Saila SB, Brown CW (1977) Prevalence of neoplasia in New England populations of the soft-shell clam (Mya arenaria). Ann N Y Acad Sci 298:522–534
Brown SB, Adams BA, Cyr DG, Eales JG (2004) contaminant effects on the teleost fish thyroid. Environ Toxicol Chem 23:1680–1701
Buckley JT, Roch M, McCarter JA, Rendell CA, Matheson AT (1982) Chronic exposure of coho salmon to sublethal concentrations of copper—I. Effect on growth, on accumulation and distribution of copper, and on copper tolerance. Comp Biochem Physiol C 72:15–19
Burrell RD, Breitburg D, Hondorp D, Keppel A (2011) Breathless nights: Diel-Cycling hypoxia and the prevalence of Perkinsus marinus (dermo) infections in Crassostrea virginica. Presented at Coastal and Estuarine Research Federation meetings, Nov. 2011, Daytona Beach, FL
Butler JA, Millemann RE, Stewart NE (1968) Effects of the insecticide Sevin on survival and growth of the cockle clam Clinocardium nuttalli. J Fish Res Board Can 25:1621–1635
Canli M, Atli G (2003) The relationships between heavy metal (Cd, Cr, Cu, Fe, Pb, Zn) levels and the size of six Mediterranean fish species. Environ Pollut 121:129–136
Cantelmo A, Lazell R, Mantel L (1981) The effects of benzene on molting and limb regeneration in juvenile Callinectes sapidus. Mar Biol Lett 2:333–343
Carr RS, Neff JM, Boehm PD (1987) A study of the fate and effects of chemically and physically dispersed oil on benthic marine communities using large-scale continuous flow exposure systems. In: Vernberg WB, Calabrese A, Thurberg FP, Vernberg FJ (eds) Pollution physiology of estuarine organisms. University of South Carolina Press, Columbia, pp 47–68
Chen J-C, Lin C-H (2001) Toxicity of copper sulfate for survival, growth, molting, and feeding of juveniles of the tiger shrimp Penaeus monodon. Aquaculture 192:55–65
Christensen DJ, Farley CA, Kern FG (1974) Epizootic neoplasms in the clam Macoma balthica (L) from Chesapeake Bay. J Natl Cancer Inst 52:1739–1749
Clark JV (1986) Inhibition of moulting in Penaeus semisulcatus (de Haan) by long-term hypoxia. Aquaculture 52:253–254
Comeau S, Gorsky G, Jeffree R, Teyssié J-L, Gattuso J-P (2009) Impact of ocean acidification on a key Arctic pelagic mollusk (Limacina helicina). Biogeosciences 6:1877–1882
Comeau S, Jeffree R, Teyssié J-L, Gattuso J-P (2010) Response of the Arctic Pteropod Limacina helicina to projected future environmental conditions. PLoS One 5(6):e11362. doi:10.1371/journal.pone.0011362
Comeau S, Alliouane S, Gattuso J-P (2012) Effects of ocean acidification on overwintering juvenile Arctic pteropods, Limacina helicina. Mar Ecol Prog Ser 456:279–284
Cong NV, Phuong NT, Bayley M (2009) Effects of repeated exposure of diazinon on cholinesterase activity and growth in snakehead fish (Channa striata). Ecotoxicol Environ Saf 72:699–703
Couch JA, Harshbarger JC (1985) Effects of carcinogenic agents on aquatic animals: an environmental and experimental overview. Environ Carcinog Rev 3:63–105
Couch JA, Winstead JT, Hanson DJ, Goodman LR (1979) Vertebral dysplasia in young fish exposed to the herbicide trifluralin. J Fish Dis 2:35–42
Crook ED, Potts D, Rebolledo-Vieyra M, Hernandez L, Paytan A (2012) Calcifying coral abundance near low-pH springs: implications for future ocean acidification. Coral Reefs 31:239–245
Cunningham PA (1976) Inhibition of shell growth in the presence of mercury and subsequent recovery of juvenile oysters. Proc Natl Shellfish Assoc 66:1–5
Cunningham PA, Myers LE (1987) Effects of diflubenzuron (Dimilin®) on survival, molting, and behavior of juvenile fiddler crabs, Uca pugilator. Arch Environ Contam Toxicol 16:745–752
D’Andrea AF, Stancyk SE, Chandler GT (1996) Sublethal effects of cadmium on arm regeneration in the burrowing brittlestar, Microphiopholis gracillima. Ecotoxicology 5:115–133
Danion M, Deschamps M-H, Thomas-Guyon H, Bado-Nilles A, Le Floch S, Quentel C, Sire J-Y (2011) Effect of an experimental oil spill on vertebral bone tissue quality in European sea bass (Dicentrarchus labrax L.). Ecotoxicol Environ Saf 74:1888–1895
Davis WP (1997) Evidence for developmental and skeletal responses as potential signals of endocrine disrupting compounds in fishes. In: Rolland RM, Gilbertson M, Peterson RE (eds) Chemically induced alterations in functional development and reproduction of fishes. SETAC Press, Pensacola, pp 61–72
De Bodt C, Van Oostende N, Harlay J, Sabbe K, Chou L (2010) Individual and interacting effects of pCO2 and temperature on Emiliania huxleyi calcification: study of the calcite production, the coccolith morphology and the coccosphere size. Biogeosciences 7:1401–1412
Deane EE, Woo YS (2009) Modulation of fish growth hormone levels by salinity, temperature, pollutants and aquaculture related stress: a review. Rev Fish Biol Fish 19:97–120
Denoyelle M, Geslin E, Jorissen FJ, Cazes L, Galgani F (2012) Innovative use of foraminifera in ecotoxicology: a marine chronic bioassay for testing potential toxicity of drilling muds. Ecol Indic 12:17–25
Dey WP, Peck TH, Smith CE, Kreamer G-L (1993) Epizootiology of hepatic neoplasia in Atlantic tomcod (Microgadus tomcod) from the Hudson River estuary. Can J Fish Aquat Sci 50:1897–1907
Dickinson GH, Ivanina AV, Matoo OB, Pörtner HO, Lannig G, Bock C, Beniash E, Sokolova IM (2012) Interactive effects of salinity and elevated CO2 levels on juvenile eastern oysters, Crassostrea virginica. J Exp Biol 215:29–43
Doropoulos C, Ward S, Marshell A, Diaz-Pulido G, Mumby PJ (2012) Interactions among chronic and acute impacts on coral recruits: the importance of size-escape thresholds. Ecology 93:2131–2138
Duckworth AR, Peterson BJ (2013) Effects of seawater temperature and pH on the boring rates of the sponge Cliona celata in scallop shells. Mar Biol 160:27–35
Duckworth AR, West L, Vansach T, Stubler A, Hardt M (2012) Effects of water temperature and pH on growth and metabolite biosynthesis of coral reef sponges. Mar Ecol Prog Ser 462:67–77
Edmunds PJ, Brown D, Moriarty V (2012) Interactive effects of ocean acidification and temperature on two scleractinian corals from Moorea, French Polynesia. Glob Change Biol 18:2173–2183
Engel DW (1987) Metal regulation and molting in the blue crab Callinectes sapidus: copper, zinc, and metallothionein. Biol Bull 172:69–82
Evrard E, Devaux A, Bony S, Burgeot T, Riso R, Budzinski H, Le Du M, Quiniou L, Laroche J (2010) Responses of the European flounder Platichthys flesus to the chemical stress in estuaries: load of contaminants, gene expression, cellular impact and growth rate. Biomarkers 15:111–127
Feely RA, Sabine CL, Hernandez-Ayon J, Ianson D, Hales B (2008) Evidence for upwelling of corrosive “acidified” water onto the Continental Shelf. Science 320:1490–1492
Fernandez-Reiriz M, Range P, Alvarex-Salgado X, Espinosa J, Labarta U (2012) Tolerance of juvenile Mytilus galloprovincialis to experimental seawater acidification. Mar Ecol Prog Ser 454:65–74
Fingerman S, Fingerman M (1978) Effects of two polychlorinated biphenyls (Aroclors 1242 and 1254) on limb regeneration in the fiddler crab Uca pugilator at different times of the year. Vie Milieu 28–29:69–75
Folmar LC, Dickhoff WW, Zaugg WE, Hodgins HO (1982) The effects of Aroclor 1254 and No. 2 fuel oils on smoltification and sea-water adaptation of coho salmon (Oncorhynchus kisutch). Aquat Toxicol 2:291–300
Fukuyama AK, Shigenaka G, Hoff RZ (2000) Effects of residual Exxon Valdez oil on intertidal Protothaca staminea: mortality, growth, and bioaccumulation of hydrocarbons in transplanted clams. Mar Pollut Bull 40:1042–1050
Gardiner G, Yevich P, Harshbarger J, Malcolm AR (1991) Carcinogenicity of Black Rock Harbor sediment to Eastern Oyster and trophic transfer of Black Rock Harbor carcinogens from the blue mussel to the winter flounder. Environ Health Perspect 90:53–66
Gardner G, Yevich P, Hurst J, Thayer P, Benyi S, Harshbarger JC, Pruell RJ (1991) Germinomas and teratoid siphon anomalies in softshell clams, Mya arenaria, environmentally exposed to herbicides. Environ Health Perspect 90:43–51
Gaylord B, Hill TM, Sanford E, Lenz EA, Jacobs LA, Sato KN, Russell AD, Hettinger A (2011) Functional impacts of ocean acidification in an ecologically critical foundation species. J Exp Biol 214:2586–2594
Gazeau F, Quiblier C, Jansen JM, Gattuso J-P, Middelburg JJ, Heip CHR (2007) Impact of elevated CO2 on shellfish calcification. Geophys Res Lett 34:L07603. doi:10.1029/2006GL028554
Gopalan UK, Young JS (1975) Incidence of shell disease in shrimps in the New York Bight. Mar Pollut Bull 6:149–153
Guo F, Wang L, Wang W-X (2012) Acute and chronic toxicity of polychlorinated biphenyl 126 to Tigriopus japonicus: effects on survival, growth, reproduction and intrinsic rate of population growth. Environ Toxicol Chem 31:639–645
Gutowska MA, Melzner F, Pörtner HO, Meier S (2010) Cuttlebone calcification increases during exposure to elevated seawater pCO2 in the cephalopod Sepia officinalis. Mar Biol 157:1653–1663
Guzmán HM, Burns KA, Jackson JB (1994) Injury, regeneration and growth of Caribbean reef corals after a major oil spill in Panama. Mar Ecol Prog Ser 105:231–241
Hamilton SJ, Haines TA (1989) Bone characteristics and metal concentrations in white suckers (Catostomus commersoni) from one neutral and three acidified lakes in Maine. Can J Fish Aquat Sci 46:440–446
Hanson N, Förlin L, Larsson A (2009) Evaluation of long-term biomarker data from perch (Perca fluviatilis) in the Baltic Sea suggests increasing exposure to environmental pollutants. Environ Toxicol Chem 28:364–373
Haya K, Waiwood BA, Van Eeckhaute L (1985) Disruption of energy metabolism and smoltification during exposure of juvenile Atlantic salmon Salmo salar to low pH. Comp Biochem Physiol C 82:323–330
Heintz RA, Rice SD, Wertheimer AC, Bradshaw RF, Thrower FP, Joyce JE, Short JW (2000) Delayed effects on growth and marine survival of pink salmon Oncorhynchus gorbuscha after exposure to crude oil during embryonic development. Mar Ecol Prog Ser 208:205–216
Hofmann LC, Straub S, Bischof K (2012) Competition between calcifying and noncalcifying temperate marine macroalgae under elevated CO2 levels. Mar Ecol Prog Ser 464:89–105
Holcomb M, Cohen AL, McCorkle DC (2012) An investigation of the calcification response of the scleractinian coral Astrangia poculata to elevated pCO2 and the effects of nutrients, zooxanthellae and gender. Biogeosciences 9:29–39
Iguchi A, Ozake S, Nakamura T, Inoue M, Tanaka Y, Suzuki A, Kawahata H, Sakai K (2012) Effects of acidified seawater on coral calcification and symbiotic algae on the massive coral Porites australiensis. Mar Environ Res 73:32–36
Jackson JB et al (1989) Ecological effects of a major oil spill on Panamanian coastal marine communities. Science 241:37–44
Johnson MD, Carpenter RC (2012) Ocean acidification and warming decrease calcification in the crustose coralline alga Hydrolithon onkodes and increase susceptibility to grazing. J Exp Mar Biol Ecol 434–435:94–101
Jørgensen EH, Aas-Hansen O, Maule AG, Strand JE, Vijayan MM (2004) PCB impairs smoltification and seawater performance in anadromous Arctic charr (Salvelinus alpinus). Comp Biochem Physiol C Toxicol Pharmacol 138:203–212
Jury CP, Whitehead RF, Szmant AM (2010) Effects of variations in carbonate chemistry on the calcification rates of Madracis aure tenra (= Madracis mirabilis sensu Wells, 1973): bicarbonate concentrations best predict calcification rates. Global Change Biol 16:1632–1644
Karen DJ, Draughn R, Fulton M, Ross PE (1998) Bone strength and acetylcholinesterase inhibition as endpoints in chlorpyrifos toxicity to Fundulus heteroclitus. Pestic Biochem Physiol 60:167–175
Karen DJ, Ross PE, Klaine SJ (2001) Further considerations of the skeletal system as a marker of episodic chlorpyrifos exposure. Aquat Toxicol 52:285–296
Keen PL, Higgs DA, Hall KJ, Ikonomou M (2005) Effects of dietary exposure of 4-nonylphenol on growth and smoltification of juvenile coho salmon (Oncorhynchus kisutch). Sci Total Environ 349:81–94
Kerambrun E, Henry F, Perrichon P, Courcot L, Meziane T, Spilmont N, Amara R (2012a) Growth and condition indices of juvenile turbot, Scophthalmus maximus, exposed to contaminated sediments: effects of metallic and organic compounds. Aquat Toxicol 108:130–140
Kerambrun E, Henry F, Courcot L, Gevaert F, Amara R (2012b) Biological responses of caged juvenile sea bass (Dicentrarchus labrax) and turbot (Scophtalmus maximus) in a polluted harbour. Ecol Indic 19:161–171
Keteles KA, Fleeger JW (2001) The contribution of ecdysis to the fate of copper, zinc, and cadmium in grass shrimp, Palaemonetes pugio Holthuis. Mar Pollut Bull 42:1397–1402
Khan AT, Weis JS, Saharig CE, Polo AE (1993) Effect of tributyltin on mortality and telson regeneration of grass shrimp, Palaemonetes pugio. Bull Environ Contam Toxicol 50:152–157
Kraus ML, Weis JS (1988) Differences in the effects of mercury on telson regeneration in two populations of grass shrimp Palaemonetes pugio. Arch Environ Contam Toxicol 17:115–120
Kroeker KJ, Micheli F, Gambi MC, Martz TR (2011) Divergent ecosystem responses within a benthic marine community to ocean acidification. Proc Natl Acad Sci 108:14515–14520
Kroeker KJ, Micheli F, Gambi MC (2013) Ocean acidification causes ecosystem shifts via altered competitive interactions. Nat Clim Change 3:156–159
Kroglund F, Finstad B, Stefansson SO, Nilsen TO, Kristensen T, Rosseland BO, Teien HC, Salbu B (2007) Exposure to moderate acid water and aluminum reduces Atlantic salmon post-smolt survival. Aquaculture 273:360–373
Landes A, Zimmer M (2012) Acidification and warming affect both a calcifying predator and prey, but not their interaction. Mar Ecol Prog Ser 450:1–10
Lee J-W, De Riu N, Lee S, Bai SC, Moniello G, Hung SS (2011) Effects of dietary methylmercury on growth performance and tissue burden in juvenile green (Acipenser medirostris) and white sturgeon (A. transmontanus). Aquat Toxicol 105:227–234
Lerner DT, Björnsson BT, McCormick SD (2007) Effects of aqueous exposure to polychlorinated biphenyls (Aroclor 1254) on physiology and behavior of smolt development of Atlantic salmon. Aquat Toxicol 81:329–336
Madsen S, Skovbølling S, Nielsen C, Korsgaard B (2004) 17-β Estradiol and 4-nonylphenol delay smolt development and downstream migration in Atlantic salmon, Salmo salar. Aquat Toxicol 68:109–120
Maier C, Watremez P, Taviani M, Weinbauer MG, Gattuso JP (2012) Calcification rates and the effect of ocean acidification on Mediterranean cold-water corals. Proc R Soc B Biol Sci 279:1716–1723
Malins DC et al (1984) Chemical pollutants in sediments and diseases of bottom-dwelling fish in Puget Sound, Washington. Environ Sci Technol 13:705–713
Malins DC et al (1988) Neoplastic and other diseases in fish in relation to toxic chemicals: an overview. Aquat Toxicol 11:43–67
Manley AR, Gruffydd LD, Almada-Villela PC (1984) The effect of copper and zinc on the shell growth of Mytilus edulis measured by a laser diffraction technique. J Mar Biol Assoc UK 64:417–427
Manzello DP (2010) Coral growth with thermal stress and ocean acidification: lessons from the eastern tropical Pacific. Coral Reefs 29:749–758
McIntyre- Wressnig A, Bernhard JM, McCorkle DC, Hallock P (2013) Non-lethal effects of ocean acidification on the symbiont-bearing benthic foraminifer Amphistegina gibbosa. Mar Ecol Prog Ser 472:45–60
Mehrle PA, Haines TA, Hamilton S, Ludke JL, Mayer FL, Ribick MA (1982) Relationship between body contaminants and bone development in east-coast striped bass. Trans Am Fish Soc 111:123–130
Melatunan S, Calosi P, Rundle SD, Widdicombe S, Moody AJ (2013) Effects of ocean acidification and elevated temperature on shell plasticity and its energetic basis in an intertidal gastropod. Mar Ecol Prog Ser 472:155–168
Melzner F, Stange P, Trubenbach K, Thomsen J, Casties I, Panknin U, Gorb S, Guowska MA (2012) Food supply and seawater pCO2 impact calcification and internal shell dissolution in the blue mussel Mytilus edulis. PLoS One 6(9):art.e24223.2011
Mix MC (1988) Shellfish diseases in relation to toxic chemicals. Aquat Toxicol 11:29–42
Moore MJ, Shea D, Hillman RE, Stegeman JJ (1996) Trends in hepatic tumours and hydropic vacuolization, fin erosion, organic chemicals, and stable isotope ratios in winter flounder from Massachusetts, U.S.A. Mar Pollut Bull 32:458–470
Mortensen AS, Arukwe A (2006) The persistent DDT metabolite,1,1-dichloro-2,2-bis(p-chlorophenyl) ethylene, alters thyroid hormone-dependent genes, hepatic cytochrome P4503A, and pregnane X receptor gene expressions in Atlantic salmon (Salmo salar) Parr. Environ Toxicol Chem 25:1607–1615
Moureaux C, Simon J, Mannaerts G, Catarino AI, Pernet P, Dubois P (2011) Effects of field contamination by metals (Cd, Cu, Pb, Zn) on biometry and mechanics of echinoderm ossicles. Aquat Toxicol 105:698–707
Murchelano RA, Wolke RE (1985) Epizootic carcinoma in the winter flounder, Pseudopleuronictes americanus. Science 228:587–589
Myers M, Landahl JT, Krahn MM, Johnson LL, McCain BB (1990) Overview of studies on liver carcinogenesis in English sole from Puget Sound; evidence for a xenobiotic chemical etiology I: pathology and epizootiology. Sci Total Environ 94:33–50
Myers MS, Anulacion BF, French BL, Reichert WL, Laetz CA, Buzitis J, Olson OP, Sol SY, Collier TK (2008) Improved flatfish health following remediation of a PAH-contaminated site in Eagle Harbor, Washington. Aquat Toxicol 88:277–288
Nelson WC (1987) A comparison of the physiological condition of the blue mussel, Mytilus edulis, after laboratory and field exposure to dredged material. In: Vernberg WB, Calabrese A, Thurberg FP, Vernberg FJ (eds) Pollution physiology of estuarine organisms. University of South Carolina Press, Columbia, pp 185–205
Nienhuis S, Palmer AR, Harley CDG (2010) Elevated CO2 affects shell dissolution rate but not calcification rate in a marine snail. Proc R Soc Lond B 277:2553–2558
Nilsson HC, Sköld M (1996) Arm regeneration and spawning in the brittle star Amphiura filiformis (O.F. Müller) during hypoxia. J Exp Mar Biol Ecol 199:193–206
Pandolfi JM, Connolly SR, Marshall DJ, Cohen AL (2011) Projecting coral reef futures under global warming and ocean acidification. Science 333:418–422
Patterson HK, Carmichael RH, Boettcher A (2011) Stressed out oysters: measuring sublethal responses using stable isotopes and protein regulation. Presented at Coastal and Estuarine Research Federation meetings, Nov. 2011, Daytona Beach, FL
Pichavant K, Person-Le-Ruyet J, Le Bayon N, Severe A, Le Roux A, Boeuf G (2001) Comparative effects of long-term hypoxia on growth, feeding and oxygen consumption in juvenile turbot and European sea bass. J Fish Biol 59:875–883
Prazeres M, Martins SE, Bianchini A (2011) Biomarkers response to zinc exposure in the symbiont-bearing foraminifer Amphistegina lessonii (Amphisteginidae, Foraminifera). J Exp Mar Biol Ecol 407:116–121
Price NN, Hamilton SL, Tootell JS, Smith JE (2011) Species-specific consequences of ocean acidification for the calcareous tropical green algae Halimeda. Mar Ecol Prog Ser 440:67–78
Range P, Piló D, Ben Hamadou R, Chícharo MA, Matias D, Joaquim S, Oliveira AP, Chícharo L (2012) Seawater acidification by CO2 in a coastal lagoon environment: effects on life history traits of juvenile mussels Mytilus galloprovincialis. J Exp Mar Biol Ecol 424–425:89–98
Rao KR, Fox FR, Conklin PJ, Cantelmo AC, Brannon AC (1979) Physiological and biochemical investigation of the toxicity of pentachlorophenol to crustaceans. In: Vernberg WB, Thurberg FP, Calabrese A, Vernberg FJ (eds) Marine pollution: functional responses. Academic Press, New York, pp 307–340
Rao KR, Fox FR, Conklin PJ, Cantelmo AC (1981) Comparative toxicology and pharmacology of chlorophenols: studies on the grass shrimp Palaemonetes pugio. In: Vernberg FJ, Calabrese A, Thurberg FP, Vernberg WB (eds) Biological monitoring of marine pollutants. Academic Press, New York, pp 37–72
Rao KR, Conklin PJ, Doughtie DG (1985) Physiological and histopathological evaluation of toxicity of hexavalent chromium to the grass shrimp Palaemonetes pugio Holthuis. In: Vernberg FJ, Thurberg FP, Calabrese A, Vernberg WB (eds) Marine pollution and physiology: recent advances. University of South Carolina Press, Columbia, pp 131–183
Rees BB, Targett TE, Ciotti BJ, Tolman CA, Akkina SS, Gallaty AM (2012) Temporal dynamics in growth and white skeletal muscle composition of the mummichog Fundulus heteroclitus during chronic hypoxia and hyperoxia. J Fish Biol 81:148–164
Rice CA, Myers MS, Willis ML, French BL, Casillas E (2000) From sediment bioassay to fish biomarker – connecting the dots using simple trophic relationships. Mar Environ Res 50:527–533
Ries JB, Cohen AL, McCorkle DC (2009) Marine calcifiers exhibit mixed responses to CO2-induced ocean acidification. Geology 37:1131–1134
Ries JB, Cohen AL, McCorkle DC (2010) A nonlinear calcification response to CO2-induced ocean acidification by the coral Oculina arbuscula. Coral Reefs 29:661–674
Rolland R (2000) A review of chemically-induced alterations in thyroid and vitamin A status from field studies of wildlife and fish. J Wildl Dis 36:615–635
Sauer GR (1987) The effect of cadmium and zinc on calcium uptake and scale regeneration in Fundulus heteroclitus. In: Vernberg WB, Calabrese A, Thurberg FP, Vernberg FJ (eds) Pollution physiology of estuarine organisms. University of South Carolina Press, Columbia, pp 373–399
Saunders R, Henderson EB, Harmon PR, Johnston CE, Eales JG (1983) Effects of low environmental pH on smolting of Atlantic Salmon (Salmo salar). Can J Fish Aquat Sci 40:1203–1211
Schram J, McClintock JB, Angus RA, Lawrence JM (2011) Regenerative capacity and biochemical composition of the sea star Luidia clathrata (Say) (Echinodermata: Asteroidea) under conditions of near-future ocean acidification. J Exp Mar Biol Ecol 407:266–274
Schwartz JP (1985) Effect of oil-contaminated prey on the feeding and growth rate of pink salmon fry (Oncorhynchus gorbuscha). In: Vernberg FJ, Thurberg FP, Calabrese A, Vernberg WB (eds) Marine pollution and physiology: recent advances. University of South Carolina Press, Columbia, pp 459–476
Scott GI, Vernberg WB (1979) Seasonal effects of chlorine produced oxidants on the growth, survival, and physiology of the American oyster, Crassostrea virginica (Gmelin). In: Vernberg WB, Thurberg FP, Calabrese A, Vernberg FJ (eds) Marine pollution: functional responses. Academic, New York, pp 415–435
Shafir S, Van Rijn J, Rinkevich B (2007) Short and long term toxicity of crude oil and oil dispersants to two representative coral species. Environ Sci Technol 41:5571–5574
Sindermann CJ (1979) Pollution-associated diseases and abnormalities of fish and shellfish: a review. Fish Bull 76:717–749
Smith HEK et al (2012) Predominance of heavily calcified coccolithophores at low CaCO3 saturation during winter in the Bay of Biscay. Proc Natl Acad Sci USA 109:8845–8849
Staurnes M, Blix P, Reite OB (1993) Effects of acid water and aluminum on parr–smolt transformation and seawater tolerance in Atlantic Salmon, Salmo salar. Can J Fish Aquat Sci 50:1816–1827
Steirhoff KL, Targett T, Miller K (2006) Ecophysiological responses of juvenile summer and winter flounder to hypoxia: experimental and modeling analyses of effects on estuarine nursery quality. Mar Ecol Prog Ser 325:255–266
Stueckle T, Likens J, Foran CM (2008) Limb regeneration and molting processes under chronic methoprene exposure in the mud fiddler crab Uca pugnax. Comp Biochem Physiol C 147:366–377
Suggett DJ, Hall-Spencer JM, Rodolfo-Metalpa R, Boatman TG, Payton R, Tye Pettay D, Johnson VR, Warner ME, Lawson T (2012) Sea anemones may thrive in a high CO2 world. Glob Change Biol 18:3015–3025
Sweet M, Kirkham N, Bendall M, Currey L, Bythell J, Heupel M (2012) Evidence of melanoma in wild marine fish populations. PLoS One 7(8):e41989. doi:10.1371/journal.pone.0041989
Thain JE (1984) Effects of mercury on the prosobranch mollusk Crepidula fornicata: acute lethal toxicity and effects on growth and reproduction of chronic exposure. Mar Environ Res 12:285–309
Thetmeyer H, Waller U, Black KD, Inselmann S, Rosenthal H (2001) Growth of European sea bass (Dicentrarchus labrax L.) under hypoxic and oscillating oxygen conditions. Aquaculture 174:355–367
Thresher RE, Tilbrook B, Fallon S, Wilson NC, Adkins J (2011) Effects of chronic low carbonate saturation levels on the distribution, growth and skeletal chemistry of deep-sea corals and other seamount megabenthos. Mar Ecol Prog Ser 442:87–99
Touart L, Rao KR (1987) Influence of diflubenzuron on survival, molting, and limb regeneration in the grass shrimp Palaemonetes pugio. In: Vernberg WB, Calabrese A, Thurberg FP, Vernberg FJ (eds) Pollution physiology of estuarine organisms. University of South Carolina Press, Columbia, pp 333–349
Vogelbein WK, Fournie JW, van Veld PA, Huggett RJ (1990) Hepatic neoplasms in the mummichog Fundulus heteroclitus from a creosote-contaminated site. Cancer Res 50:5978–5986
Waldock MJ, Thain JE (1983) Shell thickening in Crassostrea gigas: organotin antifouling or sediment induced. Mar Pollut Bull 14:411–415
Walsh GE, McLaughlin L, Louie MK, Deans CH, Lores EM (1986) Inhibition of arm regeneration by Ophioderma brevispina (Echinodermata, Ophiuroidea) by tributyltin oxide and triphenyltin oxide. Ecotoxicol Environ Saf 12:95–100
Wang SY, Stickle WB (1987) Bioenergetics, growth and molting of the blue crab, Callinectes sapidus, exposed to the water-soluble fraction of Couth Louisiana crude oil. In: Vernberg WB, Calabrese A, Thurberg FP, Vernberg FJ (eds) Pollution physiology of estuarine organisms. University of South Carolina Press, Columbia, pp 107–126
Watson SA, Peck LS, Tyler PA, Southgate PC, Tan KS, Day RW, Morley SA (2012) Marine invertebrate skeleton size varies with latitude, temperature, and carbonate saturation: implications for global change and ocean acidification. Glob Change Biol 18:3026–3038
Weis JS, Mantel LH (1976) DDT as an accelerator of limb regeneration and molting in fiddler crabs. Estuar Coast Mar Sci 4:461–466
Weis JS, Weis P (1975) Retardation of fin regeneration in Fundulus by several insecticides. Trans Am Fish Soc 104:135–137
Weis P, Weis JS (1976) Effects of heavy metals on fin regeneration in the killifish, Fundulus heteroclitus. Bull Environ Contam Toxicol 16:197–202
Weis P, Weis JS (1978) Methylmercury inhibition of fin regeneration in fishes and its interaction with salinity and cadmium. Estuar Coast Mar Sci 6:327–334
Weis P, Weis JS (1980) Effect of zinc on fin regeneration in the mummichog, Fundulus heteroclitus, and its interaction with methylmercury. Fish Bull 78:163–166
Weis P, Weis JS (1986) Cadmium acclimation and hormesis in Fundulus heteroclitus during fin regeneration. Environ Res 39:356–363
Weis JS, Cohen R, Kwiatkowski J (1987a) Effects of diflubenzuron on limb regeneration and molting in the fiddler crab, Uca pugilator. Aquat Toxicol 10:279–290
Weis JS, Gottlieb J, Kwiatkowski J (1987b) Tributyltin retards regeneration and produces deformities of limbs in the fiddler crab Uca pugilator. Arch Environ Contam Toxicol 16:321–326
Weis JS, Cristini A, Rao KR (1992) Effects of pollutants on molting and regeneration in Crustacea. Am Zool 32:495–500
Wirgin I, Grunwald C, Courtenay S, Kreamer G-L, Reichert WL, Stein JE (1994) A biomarker approach to assessing xenobiotic exposure in Atlantic tomcod from the North American Atlantic Coast. Environ Health Perspect 102:764–770
Wisshak M, Schönberg CH, Form A, Freiwald A (2012) Ocean acidification accelerates reef bioerosion. PLoS One 7(9):e45124. doi:10.1371/journal.pone.0045124
Wolfe K, Smith AM, Trimby P, Byrne M (2012) Vulnerability of the paper nautilus (Argonauta nodosa) shell to a climate change ocean: potential for extinction by dissolution. Biol Bull 223:236–244
Wolowicz M, Smolarz K, Sokołowski A (2005) Neoplasia in estuarine bivalves: effect of feeding behaviour and pollution in the Gulf of Gdansk (Baltic Sea, Poland). In: The comparative roles of suspension-feeders in ecosystems. NATO Sci Ser IV Earth Environ Sci 47:165–182
Woltering D (1984) The growth response in fish chronic and early life stage toxicity tests: a critical review. Aquat Toxicol 5:1–21
Wood HL, Spicer JL, Widdicombe S (2008) Ocean acidification may increase calcification rates, but at a cost. Proc R Soc B 275:1767–1773
Wood HL, Spicer JI, Kendall MA, Lowe DM, Widdicombe S (2011) Ocean warming and acidification; implications for the Arctic brittlestar Ophiocten sericeum. Polar Biol 34:1033–1044
Yevich P, Barszcz CA (1977) Neoplasia in soft-shell clams (Mya arenaria) collected from oil-impacted sites. Ann N Y Acad Sci 298:409–426
Young JS, Pearce JB (1975) Shell disease in crabs and lobsters from New York Bight. Mar Pollut Bull 6:101–105
Ziskowski J, Mercaldo-Allen R, Pereira JJ, Kuropat C, Goldberg R (2008) The effects of fin rot disease and sampling method on blood chemistry and hematocrit measurements of winter flounder, Pseudopleuronectes americanus from New Haven Harbor (1987–1990). Mar Pollut Bull 56:740–750
Zuykov M, Pelletier E, Belzile C, Demers S (2011) Alteration of shell nacre micromorphology in blue mussel Mytilus edulis after exposure to free-ionic silver and silver nanoparticles. Chemosphere 84:701–706
Author information
Authors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer Science+Business Media Dordrecht
About this chapter
Cite this chapter
Weis, J.S. (2014). Developmental Processes Later in Life. In: Physiological, Developmental and Behavioral Effects of Marine Pollution. Springer, Dordrecht. https://doi.org/10.1007/978-94-007-6949-6_8
Download citation
DOI: https://doi.org/10.1007/978-94-007-6949-6_8
Published:
Publisher Name: Springer, Dordrecht
Print ISBN: 978-94-007-6948-9
Online ISBN: 978-94-007-6949-6
eBook Packages: Earth and Environmental ScienceEarth and Environmental Science (R0)