Abstract
Tabun Cave, located at Mount Carmel (Israel), is a key site in our understanding of human evolution, as it held alternate occupations of Anatomically Modern Humans (AMH) and archaic species during MIS 7-MIS 5. Unlike the well-studied paleoanthropological and lithic assemblages, faunal remains have received less attention due to a clear excavation bias. Nonetheless, the application of modern archaeozoological and taphonomical techniques has overcome the existing difficulties, providing new insights on the economic behavior of these human species. Consequently, evidence of a higher logistic mobility amongst AMH, probably favored by their particular anatomy, has been identified at Tabun and other contemporaneous sites, which could be regarded as a reasonable cause of an evolving hunting specialization. These differences, however, were not enough to guarantee their survival in a region that went through radical climatic changes, a fact that might be related to a lack of a more efficient technology to maximize the carrying capacity of the ecosystem.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
Introduction
In terms of human evolution, the Near East is an exceptional region. It was the only area where Neanderthals and Anatomically Modern Humans (AMH) were known to have inhabited the same geographical territory during the Middle Paleolithic (~250–40 ka), sharing also the same lithic technology, known as the “Levantine Mousterian.” This interesting scenario took place during Marine Isotope Stages (MIS) 7-3, a period involving several abrupt climatic changes that threatened the survival of both human species, thus provoking two subsequent replacements, AMH by Neanderthals and these later in turn by Homo sapiens (Bar‐Yosef 2000; Shea 2003b) although probably with no direct confrontation. This makes the Levant a unique place to assess the adaptive skills of Neanderthals and AMH independently, without the disturbing effect of distinctive culture, as is the case during the Middle to Upper Paleolithic transition. In this sense, the paleoeconomic differences found between them can reasonably be related both to biological or cognitive dissimilarities or environmental conditions, the latter being known by means of paleoclimatic reconstructions.
Thanks to the large number of Mousterian assemblages found in the region, the archaeozoological record of the period is well known. However, many of the existing sites were excavated in the first half of the twentieth century (Turville‐Petre 1932; Garrod and Bate 1937; McCown and Keith 1939; Neuville 1951) with old‐fashioned analytical techniques, the aim of which was principally to recover human fossils, to document the succession of lithic industries and to reconstruct the paleoenvironment as a way to understand the available food resources. However, little attention was paid to characterizing human subsistence strategies or to understanding the accumulative agents of the bone assemblages (de la Torre and Domínguez-Rodrigo 2000). Nonetheless, this limitation was progressively overcome with new projects starting in the 1970–1980s (including the sites of Tabun, Kebara, Hayonim, Qafzeh and Amud), where not only excavation procedures and dating techniques were radically improved, but also where a wide range of multidisciplinary approaches were applied in order to better understand the way each site was formed and preserved and to accurately reconstruct the behavior of the human groups who lived there (see for example Rabinovich and Tchernov 1995; Speth and Tchernov 2001 or Stiner 2005).
Based on this new information, there is now a wide consensus on the importance that large-game hunting had during the Levantine Middle Paleolithic (Shea 2003a), the residual significance of scavenging as represented in preferences for certain anatomical parts and taphonomic alterations (Speth and Tchernov 1998) and the influence of climate change on human dispersal and replacement in the region (Frumkin et al. 2011; Shea 2008). However, data regarding comparative hunting skills between AMH and archaic populations are still scarce, probably due to the fact that both human species inhabited similar topographic areas but very different ecological scenarios (Bar-Yosef 2000) although the lack of well preserved faunal assemblages associated with anatomically modern humans in the region, limited only to Qafzeh and Skhul, is also a constraint. The resulting variations in the type and abundance of available ungulate species due to climatic oscillations (Tchernov 1998) might have then concealed any particular pattern in human behavior.
Given this background, detailed new studies are clearly needed to boost our understanding of human economic evolution, as relates to both environmental and biological changes, leading thus to a better evaluation of the reasons behind the final success of Homo sapiens during the Middle to Upper Paleolithic transition. Until new sites are discovered and analyzed, an interesting option to expand our paleoeconomic knowledge is to pay new attention to classic sites, but with a modern methodological approach that can overcome the existing biases of old excavation procedures. One of these classic sites is Tabun Cave, located in the foothills of Mount Carmel (Israel), in a cliff-line overlooking the nearby coastal plain. The importance of Tabun in the cultural sequencing of the Near East, through the characterisation of its industrial phases, is well-established (Bar-Yosef 1998; Copeland 1975; Jelinek 1982b) as is its key place in paleoanthropological studies due to the fact that it holds human remains of both AMH (Quam and Smith 1998; Rak 1998) and Neanderthals (Coppa et al. 2005; McCown and Keith 1939) dated to the Middle Paleolithic. In addition, Tabun Level D is one of the few known Early Middle Paleolithic sites of the region, thus providing a unique chronological and evolutionary sequence throughout MIS 7-MIS 5 that can reveal relevant information about economic differences between AMH and archaic populations.
However, the long-available faunal assemblage has only been studied from a paleontological point of view (Bate 1937; Garrard 1981, 1982), usually without any taphonomic considerations. Not surprisingly, the lack of adequate excavation procedures during the 1930s, with a marked preference for the more diagnostic elements (i.e. epiphyses and teeth) and the almost total neglect of the shafts of long bones and axial remains, has discouraged a deeper analysis. Despite this, with the necessary precautions, the deposit might still provide new insights into subsistence strategies, if exhaustive taphonomic techniques to maximize the information that can be extracted are used. In this sense, the differential collection of faunal remains that occurred during the excavations may make the study more difficult, but does not prevent it. On one hand, it is possible that the biases were not as significant as previously thought, as the results of the skeletal profile study will show later. On the other hand, being aware of this problem, there are still useful comparative techniques that can be applied.
Consequently, the objective of the study presented here is to reappraise the faunal material of Garrod’s excavation at Tabun Cave from a new paleoeconomic perspective, and to contextualize the obtained data with other well-known Levantine Middle Paleolithic sites. This has allowed the identification of behavioral differences between the two human species that occupied the site. As a result, a new hypothesis of human mobility and hunting preferences has been formulated, which is in agreement with biological and cognitive changes. Bearing this information in mind, the reasons behind the replacement of AMH at the end of MIS 5 by Neanderthals have been evaluated.
Materials and Methods
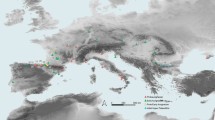
Tabun Cave is located on Mount Carmel (northern Israel) 45 m above present sea level in the Wadi el-Mughara valley (see Fig. 5.1). The entrance faces northwest, overlooking the Mediterranean coastal plain. The site contained a very long stratigraphic sequence from Lower Paleolithic to historical times. Dorothy Garrod first excavated Tabun between 1929 and 1934 (Garrod and Bate 1937), with rather expedited procedures, although this did not prevent her from recovering one of the richest Middle Paleolithic assemblages in the Levant. Garrod designated the Levantine Mousterian levels as B, C and D based mainly on differences in lithic industries. Between 1967 and 1972, Jelinek carried out a new excavation of the site, but unfortunately neither human nor faunal remains were recovered (Jelinek et al. 1973; Jelinek 1982a). There have been different estimates of the age of the various levels at Tabun over time. At present, thermoluminescence, electron spin resonance and U‐series absolute dates are available for Tabun (Grün and Stringer 2000: 602; Mercier and Valladas 2003), placing Level D, attributed to archaic human species, at the end of MIS 8 or second half of MIS 7, Level C, with AMH fossils, in the first part of MIS 5, and Level B at the end of MIS 5, with Neanderthal remains present (Shea 2003a, b, 2008). I have followed Bar-Yosef and Callander’s (1999) assumption that the Neanderthal skeleton C1 is better placed within Level B and Rak’s (1998) study when he attributes specimen C2 to an AMH jaw.
The current analysis was conducted in the Department of Paleontology at the Natural History Museum in London (NHM), where the material is currently curated. Only macromammalian remains were considered. Starting with the taxonomic and anatomical identification made by Garrard (1981), newly found specimens that were previously ignored, together with remains a priori unidentified but now successfully recognized using the large comparative collection of the NHM, were added to the study. The following indices were applied in terms of quantification: Number of Identified Specimens (NISP) (Payne 1975), Minimum Number of Individuals (MNI; Klein and Cruz-Uribe 1984), Minimum Number of Skeletal Elements (MNE; Marín-Arroyo 2009a) and Minimum Animal Units (MAU; Binford 1978). Biomass calculation was made by multiplying values of useable meat given by Garrard (1998: 63) by the MNI. This might be representative of human diet regardless of the differential transport that each prey might have undergone, because most edible parts would have been consumed should primary access to carcasses have occurred. In order to favor comparative analysis, different taxa were grouped together according to their size into large (Rhinoceros/Hippopotamus/Bos/Equus/Camelids), medium (Cervus/Dama/Capra/Sus) and small (Gazella/Capreolus) ungulate groups. Mountain gazelle and ibex are considered mountain species whereas the remaining ones are better suited to plains environments. The latter classification will allow the identification of trends in hunting preferences as a function of topographic relief, as this variable is not altered by climate change, unlike vegetation cover, which, on the other hand, can not be utilized due to a lack of adequate paleoenvironmental reconstructions in space and time.
Every element was examined under a 40× binocular microscope in order to detect any anthropogenic bone surface modifications, such as cut marks [identified as evisceration, skinning, dismembering and defleshing marks following Binford (1981) and Pérez Ripoll (1992)], hammerstone percussion marks [including conchoidal notches (Bunn 1981; Capaldo and Blumenschine 1994; Pickering and Egeland 2006), type and angle of fracturing (fresh‐green versus old‐dry), thermal alterations and trampling (Behrensmeyer et al. 1986). Carnivore and rodent gnawing marks and traces of digestion were also identified (Binford 1981; Stiner 1994) as well as other biological and physico-chemical alterations, such as weathering (Behrensmeyer 1978), root etching, carbonate deposits (Shipman 1981; Fisher 1995), dissolution or mineral coatings (mainly manganese, see Marín-Arroyo et al. 2008).
The ungulate mortality patterns (i.e. juvenile, prime or old adult) was assessed by both dental eruption and wear stage of dp4, P4 and M3, following Stiner (1991, 2005), and fusion of the bone epiphyses. For gazelle, the work of Munro et al. (2009) was used. The diet breadth and the degree of anthropogenic use of the environment has been evaluated with the Inverse of Simpson’s Index (1949), whose maximum value equals the number of consumed species only when they are in the same proportion.
Differential transport of carcasses and the related functionality of the site was investigated using bivariable correlations of %MAU with utility (in terms of %MGUI following Binford 1978) and bone survivorship (represented by bone density following Lam et al. 1999). Pielou’s evenness index (1966) was used to assess skeletal completeness for each size-class.
Finally, the relationship between the consumed fauna and the environment was investigated by estimating the catchment areas associated with several contemporaneous sites (Tabun, Misliya, Hayonim, Amud, Kebara, Skhul and Qafzeh) and characterizing the local relief, following the methodology described by Marín-Arroyo (2009b). This approach is based on the hypothesis that more carcass processing at the kill-site would arise when animals are hunted far from the base camp, implying thus non-desirable larger transport costs of low-yield anatomical parts. The Central Place Prey Foraging Model (Cannon 2003) predicts that small animals will likely be transported complete but that there is also a threshold of distance from the base camp beyond which the energetic yield of hunting this size of animals will diminish, favoring the exploitation of large game. Encounter rates will also affect these decisions, so that the shortage of a type of prey should prevent their usual consumption, above all when longer travel times are needed. Biotope abundance has been used as a proxy for encounter rates and so, the higher the biotope surface at a certain distance from the base camp, the larger the likelihood of hunting animals related to that kind of environment compared with other animals located at the same distance. From a practical point of view, a digital model of the terrain around the sites has been produced including the continental shelf (ASTER GLOBAL DEM and ETOPO1 data). An average sea level height of −80 and −40 m has been estimated for glacial and interglacial periods respectively (Siddall et al. 2006). Traveling times across the territory have been estimated with empirical formulae depending on distance, slope angles, movement direction (uphill or downhill) and the existence of insurmountable barriers (Marín-Arroyo 2008). In addition, to define the preferred biotopes or habitats for plains and mountain species within the boundaries determined by the catchment area, a threshold value of 10 % slope has been fixed to differentiate areas related to one or the other group of taxa. Beyond that value, grazing suitability is depleted (Holechek et al. 1998).
Results
Quantification of the Faunal Assemblage
The faunal assemblage is mainly dominated by complete bones. As previously mentioned, shaft fragments are rare and usually associated with epiphyses. A total of 3,402 specimens were accounted for, with roughly two-thirds belonging to Tabun B. 46 new bone remains were added to Garrard’s total by means of a thorough evaluation of previously unidentified fragments. As a result of the recovery bias during Garrod’s excavation, almost 99 % of the assemblage was identifiable taxonomically and anatomically. Table 5.1 shows the final figures for NISP, MNE and MNI together with biomass estimations. As can be seen, the NISP/MNE ratio (1.4 in Tabun D, 1.1 in Tabun C and 1.07 in Tabun B) is extremely low due to the same recovery problem.
Species Representation
A total of 25 mammal species were identified through the Tabun sequence; the carnivore frequency is low and ungulates clearly dominate the assemblage. In terms of NISP, Dama mesopotamica and Gazella gazella stand out, followed by a relatively large number of Bos primigenius (see Table 5.2). Taxonomic representation based on MNI values do not differ significantly. However, there are variations between levels in NISP. Thus, Tabun B shows a high presence of fallow deer (78.5 % NISP) in comparison with Tabun C (8.9 %) and Tabun D (31.3 %) while the percentage of gazelle decreases through time between 41.5 % in Tabun D to 15.1 % in Tabun B.
The significant difference in faunal composition of Tabun B was explained by Bate (1937) as being a result of climatic change, assuming then that the animal remains were introduced into the cave by humans, although she also considered that some of the Layer B fauna had been washed into the cave (1931: 150). However, as will be explained later, the origin of the accumulation of Tabun B was in fact mainly natural, and therefore the data from this level must not be interpreted economically. Other ungulates, such as Cervus elaphus, Capreolus capreolus, Sus scrofa, Capra sp. and Equus sp., are also represented in the deposit, although in small quantities, while large mammals such as Dicerorhinus mercki and Hippopotamus amphibius appear only in Levels D and C.
The percentage of large game, represented by rhinoceros, hippopotamus, horse, aurochs and camel, is highest in Tabun C (see Fig. 5.2). This is also the case with other Middle Paleolithic sites with known AMH accumulations, such as Skhul B (Marín-Arroyo 2011) and Qafzeh (Rabinovich and Tchernov 1995). In contrast, occupations by archaic hominins during the Early and Late Middle Paleolithic, such as Misliya (Yeshurun et al. 2007), Hayonim E (Stiner 2005), Amud B (Rabinovich and Hovers 2004) and Kebara VI-XIII (Speth and Tchernov 1998; Speth and Clark 2006), show a higher preference for the consumption of small ungulates, in these cases mountain gazelle. A similar comparison regarding the percentage of bone remains (NISP) belonging to taxa related to plains or mountain environments shows evidence of a greater use of rough terrain by archaic populations (see Fig. 5.2), which is not clearly related to the topographic relief around each settlement (see the heterogeneity of relative abundance of plains against mountains in Table 5.6). The diet breadth represented by the inverse of Simpson’s Index notably reflects a more diverse use of the available resources during AMH occupations, with the inclusion of a larger proportion of high-ranked prey species typical of plains environments.
When biomass is calculated, the role of large fauna in all levels is more emphasized, as it comprises the most important caloric input in the diet, leaving gazelle as of secondary importance. This clearly shows the need to reconsider the role of each taxon in the subsistence strategies, based on the quantification of NISP (Marín-Arroyo and González Morales 2009), and opens new interpretative possibilities, as it would imply a more selective hunting strategy based on high‐ranked species during the Middle Paleolithic, especially with regards to AMH. It is important to point out, however, that biomass calculations will establish the upper contribution of a species in the diet while NISP will define the lower. The real contribution will depend on both transport strategies and the role of scavenging in the paleoeconomy.
Bone Surface Modifications
Table 5.3 shows the abundance of different types of bone surface alterations in the assemblage. Due to the almost complete absence of bone shafts, cut marks were found mostly on epiphyses and near-epiphyses. In Tabun D (3.7 %) and especially Tabun C (9.2 %), the frequency of tool-marked bones is high enough to confirm a human origin of the faunal deposit, whereas in Tabun B (1.1 %) the accumulating agent is unclear. The abundance of burnt bones and anthropogenic fractures confirms this assumption, with values of 20.7 and 10.6 % in Tabun C respectively. This is undoubtedly the level where more carcass processing occurred, perhaps due to a more permanent occupation (see some examples in Fig. 5.3). This is coherent with the abundant thin layers (red, white and black) resulting from repeated fires found at this level (Jelinek et al. 1973: 158; Albert et al. 1999). In addition, out of the 29 cut marks identified on the main ungulate remains from Level C, one corresponds to skinning, 24 to dismembering and five to defleshing (Marín-Arroyo 2011). They are located on meat-bearing skeletal elements such as the pelvis, ribs, radius and vertebrae, suggesting primary access to carcasses.
Examples of tool-marked bones identified in Tabun Cave. a Lumbar vertebra of aurochs from Tabun C. b Metapodial of fallow deer from Tabun C with cut marks in the caudal proximal meta-diaphysis to cut tendons. c Shaft of gazelle femur from Tabun C with defleshing marks. d Tibia of fallow deer from Tabun B with dismembering marks. e Close-up of D. f Metacarpal of fallow deer from Tabun B
Carnivore activity was very scarce. Only 0.4 % of the assemblage shows gnawing marks, indicating that, at least in Tabun D and C, humans were the primary accumulating agent with carnivores having a secondary and less important role (see results from actualistic studies in Marean 1998: 129; Marean and Spencer 1991; Marean et al. 1992).
The case with Tabun B is rather different. In addition to a low, although not negligible, level of human activity, the overall appearance of the bone assemblage is different throughout. On the one hand, bones show a white colouring with some manganese dendrites, related to a likely period of ponding in the cave, contrasting clearly with the general brown colouring of the bones in Tabun C and D (high presence of organic material in the sediment as a result of intense human occupation, see Marín-Arroyo et al. 2008). The action of water is also evidenced by the larger amount of dissolution and subaerial weathering. On the other hand, some bones appear in anatomical articulation and predominantly complete. The breakage pattern is typical of mechanical destruction produced by pressure applied by stones/bones within the sediment (Fisher 1995). Bearing this in mind, a natural accumulation of carcasses caused by animals falling through the hole in the cave roof can be confirmed (Bate 1937: 150). However, this does not imply that the Tabun B assemblage is completely paleontological. The presence of cut marks on some fallow deer remains, as well as thermally alterated bones (identified on 1.6 % of the remains), seems to suggest an occasional human consumption of ungulates, possibly those accumulated naturally and found later by humans who knew the potential of the cave as a natural trap.
Apart from the cut marks found on ungulate remains, there are also some tool-marked carnivore bones, such as a metatarsal of Panthera pardus in Tabun B, probably related to skinning, and a radius of Ursus sp. in Tabun C that shows fresh breakage, burn traces and several dismembering marks, which could mean meat and marrow exploitation as well as skin use.
Skeletal Element Profiles
The MNE values for each skeletal element and class-size are presented in Table 5.4. Tabun B, which was not quantified anatomically by Garrard (1981), is also shown. The vast majority of skeletal elements in the assemblage are teeth (61 %), followed by feet (metapodial + phalanx) (24 %). The representation of long bones is only 6 % and it is mainly characterized by epiphyses of small-medium sized taxa, mostly with shafts attached. The axial skeleton is only represented by a proportion of 1 %.
The study of skeletal profiles at Tabun is problematic due to the excavation bias. There is now a wide consensus on the importance of recovering and quantifying long bone shafts in order not to hinder the economic interpretation (Marean and Frey 1997). However, taking into account the limitations of the available information, there is still room for drawing relevant conclusions if comparative analyses are applied, hence playing down individual absolute results. In this way, the effect of the bias in the collection can be side-stepped to a certain extent, as its influence would have been similar in the whole assemblage.
Figure 5.4 shows the skeletal profiles in terms of MAU. In general, small ungulates are more evenly represented than larger game, whereas long bones are less abundant in medium-size game. These results are consistent with a mostly whole transport of mountain gazelle and roe deer carcasses, whose smaller weight does not require the processing of the prey at the kill site (Cannon 2003), and with an intense defleshing of large fauna before transport, in order to maximize the energetic contribution to the base camp, particularly if the animal was hunted a long distance away (O’Connell et al. 1988, 1989). Taking into account that there are 24 different anatomical elements, Pileou’s evenness index has been also calculated for each body-size class and level using MAU values. The higher values correspond to small ungulates in each level (average value of 0.76) followed by medium and large prey (average values of 0.74 and 0.53 respectively). Spearman’s correlations between large, medium and small ungulates have been calculated for each level in order to better evaluate dissimilarities. As a result, significant positive correlations have been found between species in Tabun B (\(\uprho\) s = 0.80, p = 0 between medium and small ungulates, and \(\uprho\) s = 0.62, p = 0.002 between large and medium ungulates), thus corroborating the probable natural origin of this assemblage. In Tabun D, there is also a significant positive correlation between medium and small ungulates (\(\uprho\) s = 0.46, p = 0.05) and higher similarities between body-size skeletal profiles than in Tabun C, which can be interpreted as a greater importance of prey weight in transport decisions within AMH groups, probably due to longer travel times.
To better assess this assumption, correlations between %MAU and %MGUI, as well as between %MAU and maximum bone density, have been calculated (see Table 5.5). Correlations with bone survivorship are always positive (in three cases significantly), which could be related to the existence of moderate attrition. Not surprisingly, correlations with utility are low or negative (even significantly in two cases) as a result of bone attrition and the known bias in the assemblage recovery methods against long bone shafts and axial elements, the most meat-bearing parts of the skeleton. However, as regards medium and small ungulates, Tabun D shows more negative values compared with Tabun C, which could indicate less processing at the kill-site as a consequence of smaller catchment areas or a less residential use of the settlement.
In addition, to estimate how much the non-collection of shafts might have biased the economic interpretation of the assemblage, the MNE of the long bones (humerus, radio-ulna, femur, tibia and metapodials) was multiplied by two and by four, and the correlations with %MGUI and maximum bone density were repeated. The aim of this analysis was not to reconstruct the original assemblage, but rather to evaluate the possible loss of information that might have occurred. The results (see Table 5.5) point towards similar interpretations regarding the differential transport (the correlations are only a little less negative, but with no appreciable changes), which would support the use of the present information as representative of the subsistence behavior of the human groups. However, the presence of more long bones would have resulted in a more clearly-defined influence of attritional processes.
Seasonality and Ungulate Mortality Profiles
The mortality profiles of Tabun were assessed mainly by tooth eruption and wear stages and in some cases by bone fusion, for some species such as gazelle. Figure 5.5 shows the Tabun results together with those from other nearby sites; Qafzeh was not included due to the fact that only bone fusion was used to determine age, which neglects the possibility of identifying old individuals. The mortality profiles reflect a general predominance of prime-age individuals, with a percentage in agreement with their typical abundance in natural populations. However, within this general trend a decrease can also be seen in the number of juvenile individuals in favor of elderly ones in the levels associated with AMH occupations (Tabun C and Skhul) which might imply a greater preference for more productive animals. Although not shown in the graphic, the age profile of Tabun B lies right in the centre of the living structure area, agreeing thus with the proposed natural origin of the deposit.
Mortality profiles of ungulates at Tabun C and D, Skhul B (Marín-Arroyo 2011), Misliya (Yeshurun et al. 2007), Hayonim E (Stiner 2005), Amud B (Rabinovich and Hovers 2004) and Kebara VI-XIII (Speth and Tchernov 1998; Speth and Clark 2006). AMH occupation (open circles) and archaic ones (filled circles)
Concerning seasonality, apart from the winter occupations estimated by Lieberman (1993) for these levels, the identification of three mandibles of juvenile individuals with teeth in the first stage of eruption also points towards a late spring/early summer use of the cave in Levels C and D.
Human Mobility
Figure 5.6 shows the catchment areas corresponding to 1, 1.5 and 2 h of travel time around Tabun and other Middle Paleolithic sites. The surface area of plains and mountain biotopes, as identified with a 10 % slope threshold, is presented in Table 5.6, together with the so-called topographic factor (TF), obtained as a quotient between plain and mountain extensions (in km2).
As can be seen, the higher proportion of plains in the surroundings of AMH occupations corresponds to a travel time of 1.5 h, whilst the lower proportion in the surroundings of archaic occupations is established in 1 h travel time. Given the comparative preference for plains ungulates among AMH (see Fig. 5.2), this would mean that they usually travelled longer distances to hunt their prey. Moreover, taking into account only Tabun levels C and D, the slightly higher dependence on plains ungulates seen in Tabun C (AMH occupation) is at odds with the reduction in plains biotopes in the surroundings of the cave due to the rise in sea level during MIS 5. A probable explanation for this incongruence would again be that AMH relied more heavily on logistic mobility than their predecessors, leading to greater travel distances during their expeditions which, in turn, would mean less residential mobility. A greater degree of carcass processing should then be expected then in Tabun C, which is consistent with the taphonomy of the analyzed assemblage.
To objectively test this hypothesis, bivariate correlations have been made between the topographic factor and the faunal preference for plains ungulates, represented by the quotient between plains and mountain NISP values at each site. Nine possible combinations of the three different travel times proposed for AMH and archaic populations (1, 1.5 or 2 h) were examined. The most probable combination in terms of Spearman’s test (\(\uprho\) s = 0.58, p = 0.10) points towards travel times for AMH and archaic populations of 1.5 and 1 h respectively (see Fig. 5.7) thus reinforcing the idea of longer logistic movements amongst AMH.
Discussion
Subsistence Patterns in the Levantine Middle Paleolithic
The re-analysis of the faunal assemblage from Tabun Cave presented here, and its proper contextualisation with other Levantine Middle Paleolithic sites, allows the drawing of relevant conclusions on the subsistence patterns adopted by both AMH and archaic hominin populations in this key region for human evolutionary studies.
First, the origin of the assemblage has been clarified. Thus, Tabun B seems to have acted as a natural trap for ungulates (but also some carnivores) that would have fallen through the chimney (Bate 1937: 150; but see Kaufman 2002). Nontheless, there is evidence of human exploitation of some carcasses, probably scavenging the animals that perished there. On the other hand, the anthropogenic origin of Tabun C and D is clear, due to the presence of tool-marked bones and traces of burning, above all in level C, and the almost complete absence of carnivore marks. The location and abundance of butchering marks, including filleting, also corroborates the assumption that the human groups that inhabited the cave were capable of regular hunting episodes. In fact, the frequency of anthropogenic evidence in the assemblage is higher than that from other well known human settlements, such as Qafzeh, where 2.3 % of bone remains show cut-marks or burn traces (Rabinovich and Hovers 2004) or Misliya, where 3–4 % of bone specimens bear cut-marks and 4–6 % impact notches (Yeshurun et al. 2007). The anthropogenic alteration of Tabun C is even more intense than in Kebara, which is seen as a long-term seasonal basecamp (Bar-Yosef et al. 1992). Scavenging as a usual subsistence strategy can then be ruled out in Tabun C and D, even more so taking into account the presence of large and relatively dangerous taxa in the assemblage, such as aurochs, rhinoceros, red deer or wild boar (Bar-Yosef et al. 1992; Speth and Clark 2006).
Secondly, there is a certain distinction in hunting strategies between AMH and archaic populations. The former would have depended more on high-ranked prey during MIS 5, dismissing small mountain gazelle to some extent. This trend could be seen as an incipient specialisation strategy (Rabinovich and Tchernov 1995; Rabinovich 1990) above all taking into account that it would mean longer expeditions to more distant plains areas, as ungulates related to plains habitats were bigger in the area of study. This trend towards more productive strategies is also slightly reflected in the mortality profiles, where juvenile individuals were partially substituted by prime-age or old ones (Stiner 2005; Stiner and Tchernov 1998). In terms of diet-breadth, the emphasis on large game typical of grassy plains or woodlands resulted in a significant broadening that could reflect the even exploitation of several different environments. The recognition of more efficient strategies and a higher territorial control is usually related to optimal foraging behavior, implying more advanced cognitive capacities. This would imply that the cognitive changes that were common during the Upper Paleolithic might have already been taking place (Klein 2009).
And thirdly, the more specialized economy recognized before is usually related to lower residential mobility. In the case of the Levantine Middle Paleolithic, this fact has also been tested by means of comparing the topographic relief in the surroundings of each site with the faunal preference. As a result, a greater reliance on logistic mobility has been suggested among AMH (with travel times usually 0.5 h longer), a fact that was also proposed by Wallace and Shea (2006) by analyzing differences in the production of lithic tools, and by Lieberman (1998) based on the age of death of gazelles. In addition, body size of carcasses seems to have played a greater role in transport decisions, probably involving more intense butchering at the kill-site (with the subsequent contribution of less-useful parts to the base camp) as prey-weight increases (Marín-Arroyo 2009c). Rabinovich and Tchernov (1995) also identified in Qafzeh a selective transport strategy of hunted prey for rhinoceros and red deer. In contrast, Stiner (2005) suggests an almost complete transport of carcasses during the archaic occupation of MIS 6 at Hayonim.
This pattern of mobility was probably favored among AMH by their particular anatomy; hence, their longer lower limbs and smaller body mass (Weaver and Steudel-Numbers 2005; MacDonald et al. 2009) would have reduced their energetic expenditure while travelling (from/to base camp) in their hunting expeditions, allowing them efficient exploitation of certain distant taxa. It is still unknown whether this more mobile pattern of AMH behavior and the subsequent depletion of the resources located close to the base camp led them to focus more on larger game in order to maintain the necessary energetic income, or perhaps instead it was changes in cognitive skills permitted them to better discern between different productive choices, allowing them to maintain more permanent occupations.
Implications for AMH Replacement in the Region
Interestingly enough, the economic behavior just noted amongst AMH did not provide them with a clear evolutionary advantage in terms of survivorship or adaptive skills, as they ceased to occupy the region at the end of MIS 5, being replaced by Neanderthals coming from northern latitudes (Tchernov 1998; Shea and Bar-Yosef 2005). Two possible explanations are currently proposed for this failure. The first involves climatic deterioration that would have significantly decreased terrestrial productivity during the MIS 5/4 transition ca. 75 ka (Shea 2008). This episode would have affected the number of available taxa (Davies et al. 2011; Dynesius and Jansson 2000) which, together with the great dependency on large ungulates seen in the archaeozoological record, would have reduced their numbers and even provoked their extinction due to overhunting (Haynes 2002; Miller et al. 2005; Marín-Arroyo 2009c; Speth 2004). In this scenario, AMH would have had to cope with dietary stress that, in other conditions, would have led to an expansion in diet-breadth, including previously considered low-ranked prey (Stiner et al. 1999). In a similar way, droughts and rapid shifts to colder conditions would have seriously limited Homo sapiens’ ability to disperse during the Upper Paleolithic (Finlayson and Carrión 2007). However, either small or difficult-to-hunt animals such as lagomorphs, birds, etc. were not sufficient to maintain the existing human population due to their low calorific yield/low abundance, or the available technology of AMH was not sufficient to effectively exploit these kind of resources. For example, Shea and Sisk (2010) argue that it was not until projectile weapons of the kind used by ethnographic hunter-gatherers, such as the bow and arrow, and spearthrower and dart, were developed in Africa between 50 and 100 ka that ecological niche broadening strategies were enabled (Brooks et al. 2006; d’Errico and Henshilwood 2007), which then allowed Homo sapiens to successfully colonize Eurasia at the Middle to Upper Paleolithic transition.
An alternative hypothesis would dismiss the effect that climatic worsening would have had on biomass production and consequently on resource availability (Frumkin et al. 2011), due to the existence of reliable and spatially dispersed seasonal food resources (Bar-Yosef et al. 1992; Hovers 2006). If that were the case, AMH would have not abandoned the region when Neanderthal populations arrived from northern latitudes, and thus, some kind of competition would have occurred between both human species (Shea and Bar-Yosef 2005) in which Neanderthals might possibly have prevailed, due to a better physical adaptation to the increasing glacial conditions of MIS 4.
There are several climate studies in the Levant that can shed light on this dilemma, above all isotope and growth-rate studies of speleothems, such as those from Soreq and Peqi’in Caves (Bar-Matthews et al. 1997, 2003a), Negev Caves (Vaks et al. 2006) and Jerusalem West Caves (Frumkin et al. 1999). Thus, according to Vaks et al. (2007) the arrival of AMH in the Levant was favored by a humid period in the Negev desert region between 140 and 110 ka, coinciding with an intensification of monsoonal effects in the Arabian Peninsula (Fleitmann et al. 2003) and the Sahara (Osmond and Dabous 2004), which would have provided a suitable land bridge between sub-Saharan latitudes and the Levant. This bridge seems to have disappeared afterwards, which would have isolated AMH populations during MIS 5, a period that became drier as seen both in the growth-rate of calcite (Vaks et al. 2006; Lisker et al. 2010) and in water level reconstruction of Lake Samra, the precursor of the Holocene Dead Sea (Waldmann et al. 2009). AMH populations probably dealt with this situation by migrating to the more humid central and northern regions to the West of the central mountain ridge of Israel (in caves such as Tabun, Skhul and Qafzeh). This isolated situation might have increased survival risks at times of rapid environmental change, as it constrained migration possibilities as well as reduced genetic flow. Why AMH did not expand farther north through Taurus-Zagros Mountains is still unknown. As a possible explanation, Frumkin et al. (2011) argued that the heterogeneity of the Levant ecosystems together with a colder climate in the mountain ridges, quite different from the African one, kept AMH within the region.
Based on the characterization of spelothem fluid inclusions of Soreq Cave, McGarry et al. (2004) determined a drop of around 4 °C in temperature between 80–86 and 73–74 ka. In addition, based on: (1) present measures of \(\updelta\) 18O abundance of rain and drip-water at the Soreq Cave site; (2) the similar isotopic depletion between the Eastern Mediterranean Sea and cave locations observed during the last 250 ka as evidenced in the \(\updelta\) 18O record of planktonic foraminifera species Globigerinoides ruber of a maritime core (Fontugne and Calvert 1992); (3) the similar climate of interglacial periods; and (4) the same \(\updelta\) 18O composition of the sea in Sapropel S3 event in comparison with present times (Kallel et al. 2000). Bar-Matthews et al. (2003b) were able to estimate an annual decrease in rainfall between 82 and 77 ka of 300 mm/year (from 500 to 200 mm/year). This trend has been confirmed by Almogi-Labin et al. (2009) when they eliminated the first-order “source effect” from the \(\updelta\) 18O record of Soreq Cave with the help of two eastern Mediterranean maritime cores. These authors found a significant drop in second-order \(\updelta\) 18O during the MIS 5/4 transition that was related to the so-called “amount effect” (Dansgaard 1964) and that also indicates a pronounced decrease in rainfall. This meteorological shift was also hydrologically recorded in the water level reconstruction of Lake Samra, which presents a 10 m drop between 80.2 and 71.5 ka (Waldmann et al. 2009) and in the \(\updelta\) 13C record throughout the region that reflects an increase in C4 vegetation.
The impact of this change in terrestrial productivity can be assessed by means of estimation of real evapotranspiration (ETr), i.e. the amount of water used by the vegetation cover, which has a direct relationship with existing biomass. There are two major forces that affect evapotranspiration: the availability of water (i.e. rainfall) and the availability of solar radiation and heat. The annual ETr can be roughly estimated as a function of mean annual temperature following Turc’s equation (1954), later modified by Pike (1964). Therefore ETr at the onset of the MIS 5/4 transition would have been 450 mm/year whilst 10 ka later it would have decreased to 195 mm/year. According to Budyko (1974), this would have changed the environment from sub-humid to subarid conditions, implying a major modification of resource availability.
Given this, the possibility of an AMH extinction before the arrival of Neanderthals is considered more plausible, above all as they did not possess the necessary technology to effectively broaden their diet (Shea 2009), which would have improved their survival chances (Betinger 1991), especially if they had intensively depended on large ungulates as has been argued before. There is, however, no direct proof for this assumption, as there is currently a lack of archaeozoological assemblages from this period that could show evidence of dietary stress, such as a progressive widening of diet-breadth or a greater consumption of old and juvenile individuals (Marín-Arroyo 2009c).
References
Albert, R., Lavi, O., Estroff, L., Weiner, S., Tsatskin, A., Ronen, A., et al. (1999). Mode of occupation of Tabun Cave, Mt Carmel, Israel during the Mousterian Period: A study of the sediments and phytoliths. Journal of Archaeological Science, 26, 1249–1260.
Almogi-Labin, A., Bar-Matthews, M., Shriki, D., Kolosovsky, E., Paterne, M., Schilman, B., et al. (2009). Climatic variability during the last ~90 ka of the southern and northern Levantine Basin as evident from marine records and speleothems. Quaternary Science Reviews, 28, 2882–2896.
Bar-Matthews, M., Ayalon, A., & Kaufman, A. (1997). Late Quaternary paleoclimate in the eastern Mediterranean region from stable isotope analysis of speleothems at Soreq Cave, Israel. Quaternary Research, 47, 155–168.
Bar-Matthews, M. A., Ayalon, M., Gilmour, A., Matthews, A., & Hawkesworth, C. J. (2003a). Soreq and Peqiin Caves, Israel Speleothem Stable Isotope Data, IGBP PAGES/World Data Center for Paleoclimatology Data Contribution Series #2003-061. NOAA/NGDC Paleoclimatology Program, Boulder, CO, USA.
Bar-Matthews, M., Ayalon, A., Gilmour, M., Matthews, A., & Hawkesworth, C. J. (2003b). Seaeland oxygen isotopic relationships from planktonic foraminifera and speleothems in the Eastern Mediterranean region and their implication for paleorainfall during interglacial intervals. Geochimica et Cosmochimica Acta, 67(17), 3181–3199.
Bar-Yosef, O. (1998). The chronology of the Middle Paleolithic of the Levant. In T. Akazawas, K. Aoki, & O. Bar-Yosef (Eds.), Neanderthals and modern humans in Western Asia (pp. 39–56). New York: Plenum.
Bar-Yosef, O. (2000). The Middle and early Upper Paleolithic in Southwest Asia and neighbouring regions. In O. Bar-Yosef & D. Pilbeam (Eds.), The geography of Neanderthals and modern humans in Europe and the Greater Mediterranean (pp. 107–156). Cambridge, MA: Peabody Museum of Archaeology and Ethnology, Bulletin No. 8.
Bar-Yosef, O., & Callander, J. (1999). The woman from Tabun: Garrod’s doubts in historical perspective. Journal of Human Evolution, 37, 879–885.
Bar-Yosef, O., Vandermeersch, B., Arensburg, B., Belfer-Cohen, A., Goldberg, P., Laville, H., et al. (1992). The excavations in Kebara Cave, Mt. Carmel. Current Anthropology, 33, 497–550.
Bate, D. M. A. (1937). Paleontology: The fossil fauna of Wady el-Mughara caves. In D. A. E. Garrod & D. M. A. Bate (Eds.), The Stone Age of Mount Carmel, Part 2 (pp. 137–240). Oxford, UK: Clarendon Press.
Behrensmeyer, A. K. (1978). Taphonomic and ecological information from bone weathering. Paleobiology, 4, 150–162.
Behrensmeyer, A. K., Gordon, K. D., & Yanagi, G. T. (1986). Trampling as a cause of bone surface damage and pseudo-cutmarks. Nature, 319, 768–771.
Betinger, R. L. (1991). Hunter-gatherers: Archaeological and evolutionary theory. New York: Plenum Press.
Binford, L. R. (1978). Nunamiut Ethnoarchaeology. New York: Academic Press.
Binford, L. R. (1981). Bones: Ancient men and modern myths. New York: Academic Press.
Brooks, A. S., Yellen, J. E., Nevell, L., & Hartman, G. (2006). Projectile technologies of the African MSA: Implications for Modern Human Origins. In E. Hovers & S. L. Kuhn (Eds.), Transitions before the transition (pp. 233–256). New York: Plenum/Kluwer.
Budyko, M. I. (1974). Climate and life. Orlando, FL: Academic Press.
Bunn, H. T. (1981). Archaeological evidence for meat-eating by Plio-Pleistocene hominids from Koobi Fora and Olduvai Gorge. Nature, 291, 574–577.
Cannon, M. D. (2003). A model of central place forager prey choice and an application to faunal remains from the Mimbres Valley, New Mexico. Journal of Anthropological Archaeology, 22, 1–25.
Capaldo, S. D., & Blumenschine, R. J. (1994). A quantitative diagnosis of notches made by hammerstone percussion and carnivore gnawing on bovid long bones. American Antiquity, 59, 724–748.
Copeland, L. (1975). The Middle and Upper Paleolithic of Lebanon and Syria in the light of recent research. In F. Wendorf & A. E. Marks (Eds.), Problems in prehistory: North Africa and the Levant (pp. 317–350). Dallas: SMU Press.
Coppa, A., Grün, R., Stringer, C., Eggins, S., & Vargiu, T. (2005). Newly recognized Pleistocene human teeth from Tabun Cave, Israel. Journal of Human Evolution, 49, 301–315.
d’Errico, F., & Henshilwood, C. S. (2007). Additional evidence for bone technology in the southern African Middle Stone Age. Journal of Human Evolution, 52, 142–163.
Dansgaard, W. (1964). Stable isotopes in precipitation. Tellus, 16, 436–468.
Davies, J., Buckley, L. B., Grenyer, R., & Gittleman, J. L. (2011). The influence of past and present climate on the biogeography of modern mammal diversity. Philosophical Transactions of the Royal Society of Biological Sciences, 366(1577), 2526–2535.
De la Torre, I., & Domínguez-Rodrigo, M. (2000). El Paleolítico Medio en el Próximo Oriente. Una síntesis regional. Complutum, 11, 9–27.
Dynesius, M., & Jansson, R. (2000). Evolutionary consequences of changes in species’ geographical distributions driven by Milankovitch climate oscillations. Proceedings of the National Academy of Sciences of the United States of America, 97(16), 9115–9120.
Finlayson, C., & Carrión, J. S. (2007). Rapid ecological turnover and its impact on Neanderthal and other human populations. Trends in Ecology & Evolution, 22(4), 213–222.
Fisher, J. W. (1995). Bone surface modifications in zooarchaeology. Journal of Archaeology of Method and Theory, 2, 7–68.
Fleitmann, D., Burns, S. J., Neff, U., Mangini, A., & Matter, A. (2003). Changing moisture sources over the last 330000 years in northern Oman from fluid-inclusion evidence in speleothems. Quaternary Research, 60, 223–232.
Fontugne, M., & Calvert, S. E. (1992). Late Pleistocene variability of the carbon isotopic composition of organic matter in the Eastern Mediterranean: Monitor of changes in carbon sources and atmosphere CO2 concentrations. Paleoceanography, 7, 1–20.
Frumkin, A., Ford, D. C., & Schwarcz, H. P. (1999). Continental oxygen isotopic record of the last 170,000 years in Jerusalem. Quaternary Research, 51, 317–327.
Frumkin, A., Bar-Yosef, O., & Schwarcz, H. P. (2011). Possible paleohydrologic and paleoclimatic effects on hominin migration and occupation of the Levantine Middle Paleolithic. Journal of Human Evolution, 60, 437–451.
Garrard, A. (1981). Man-animal-plant relationship during the Upper Pleistocene and early Holocene of the Levant. PhD dissertation, University of Cambridge, Cambridge.
Garrard, A. (1982). The environmental implications of the re-analysis of the large mammal fauna from the Wadi-el-Mughara caves, Palestine. In J. L. Bintliff & W. Van Zeist (Eds.), Paleoclimates, paleoenvironments and human communities in the Eastern Mediterranean region in later prehistory (pp. 165–198). Oxford: British Archaeological Reports.
Garrard, A. (1998). Food procurement by Middle Paleolithic hominids as Ras el- Kelb Cave in Lebanon. In L. Copeland & N. Moloney (Eds.), The Mousterian Site of Ras el-Kelb, Lebanon (pp. 45–65). Oxford: British Archaeological Reports.
Garrod, D. A. E., & Bate, D. M. A. (1937). The Stone Age of Mount Carmel. Excavations at the Wadi Mughara (Vol. I). Oxford: Clarendon Press.
Grün, R., & Stringer, C. (2000). Tabun revisited: Revised ESR chronology and new ESR and U-series analyses of dental material from Tabun C1. Journal of Human Evolution, 39, 601–612.
Haynes, G. (2002). The catastrophic extinction of North American mammoths and mastodonts. World Archaeology, 33(3), 391–416.
Holechek, J. L., Pieper, R. D., & Herbel, C. H. (1998). Range management principles and practices (3rd ed.). Upper Saddle River, NJ: Prentice-Hall, Inc.
Hovers, E. (2006). Neanderthals and modern humans in the Middle Paleolithic of the Levant: What kind of interaction? In N. J. Conard (Ed.), When Neanderthals and moderns met (pp. 65–85). Tübingen: Kerns Verlag.
Jelinek, A. J. (1982a). The Tabun cave and Paleolithic man in the Levant. Science, 216, 1369–1375.
Jelinek, A. J. (1982b). The Middle Paleolithic in the Southern Levant with comments on the appearance of modern Homo sapiens. In A. Ronen (Ed.), The transition from Lower to Middle Paleolithic and the origins of modern man (pp. 57–104). Oxford: British Archaeological Reports.
Jelinek, A., Farrard, W. R., Haas, G., Horowitz, A., & Golberg, P. (1973). New excavations at the Tabun cave, Mount Carmel, Israel, 1967–1972: A preliminary report. Paléorient, 1(2), 151–183.
Kallel, N., Duplessy, J.-C., Labeyrie, L., Fontugne, M., Paterne, M., & Montacer, M. (2000). Mediterranean pluvial periods and sapropel formation during the last 200,000 years. Paleogeography, Paleoclimatology, Paleoecology, 157, 45–58.
Kaufman, D. (2002). Re-evaluating subsistence skills of Levantine Middle and Upper Paleolithic hunters: A comparison of the faunal assemblages. Oxford Journal of Archaeology, 21, 217–229.
Klein, R. (2009). The human career. Chicago: University of Chicago Press.
Klein, R. G., & Cruz-Uribe, K. (1984). The analysis of animal bones from archaeological sites. Chicago: The University of Chicago Press.
Lam, Y. M., Chen, X., & Pearson, O. M. (1999). Intertaxonomic variability in patterns of bone density and the differential representation of bovid, cervid and equid elements in the archaeological record. American Antiquity, 64, 343–362.
Lieberman, D. E. (1993). The rise and fall of seasonal mobility among hunter-gatherers. Current Anthropology, 34, 599–631.
Lieberman, D. E. (1998). Neanderthal and early modern human mobility patterns: Comparing archaeological and anatomical evidence. In T. Akazawa, K. Aoki, & O. Bar-Yosef (Eds.), Neanderthals and modern humans in Western Asia (pp. 263–276). New York: Plenum.
Lisker, S., Vaks, A., Bar-Matthews, M., Porat, R., & Frumkin, A. (2010). Late Pleistocene paleoclimatic and paleoenvironmental reconstruction of the Dead Sea area (Israel) based on speleothems and cave stromatolites. Quaternary Science Reviews, 29, 1201–1211.
MacDonald, K., Roebroeks, W., & Verpoorte, A. (2009). An energetic perspective on the Neanderthal Record. In J. J. Hublin & M. P. Richards (Eds.), The evolution of hominin diets: Integrating approaches to the study of Paleolithic Subsistence (pp. 211–220). Vertebrate Paleobiology and Paleoanthropology Series. Dordrecht: Springer.
Marean, C. W. (1998). A critique of the evidence for scavenging by Neanderthals and early modern humans: New data from Kobeh Cave (Zagros Mountains, Iran) and Die Kelders Cave 1 layer 10 (South Africa). Journal of Human Evolution, 35, 111–136.
Marean, C. W., & Frey, C. (1997). Animal bones from caves to cities: Reverse utility curves as methodological artefacts. American Antiquity, 62, 698–716.
Marean, C. W., & Spencer, L. M. (1991). Impact of carnivore ravaging on zooarchaeological measures of element abundance. American Antiquity, 56, 645–658.
Marean, C. W., Spencer, L. M., Blumenschine, R. J., & Capaldo, S. (1992). Captive hyena bone choice and destruction, the Schlepp Effect, and Olduvai archaeofaunas. Journal of Archaeological Science, 19, 101–121.
Marín-Arroyo, A. B., & González Morales, M. R. (2009). Comportamiento económico de los últimos cazadores-recolectores y primeras evidencias de domesticación en el occidente de Asturias. La Cueva de Mazaculos II. Trabajos de Prehistoria, 66(1), 47–74.
Marín-Arroyo, A. B. (2008). Patrones de movilidad y control del territorio en el Cantábrico Oriental durante el Tardiglacial. Trabajos de Prehistoria, 65(1), 29–45.
Marín-Arroyo, A. B. (2009a). A comparative study of analytic techniques for skeletal part profile interpretation at El Mirón Cave (Cantabria, Spain). Archaeofauna, 18, 79–98.
Marín-Arroyo, A. B. (2009b). The use of Optimal Foraging Theory to estimate Late Glacial site catchment areas from a central place. The case of eastern Cantabria, Spain. Journal of Anthropological Archaeology, 28, 27–36.
Marín-Arroyo, A. B. (2009c). Economic adaptations during the Late Glacial in northern Spain. A simulation approach. Before Farming, 2, 1–18.
Marín-Arroyo, A. B. (2011). Paleolithic human subsistence in Mount Carmel (Israel). A taphonomic assessment of Middle and Early Upper Paleolithic faunal remains from Tabun, Skhul and el-Wad. International Journal of Osteoarcaheology. doi:10.1002/oa.1241.
Marín-Arroyo, A. B., Landete, D., Vidal, G., Seva, R., González Morales, M., & Straus, L. G. (2008). Archaeological implications of human-derived manganese coatings: A study of blackened bones in El Mirón Cave, Cantabrian Spain. Journal of Archaeological Science, 35(3), 801–813.
McCown, T. D., & Keith, A. (1939). The Stone Age of Mt. Carmel, Vol. 2: The fossil human remains from the Levallois-Mousterian. Oxford: Clarendon Press.
McGarry, S., Bar-Matthews, M., Matthews, A., Vaks, A., Schilman, B., & Ayalon, A. (2004). Constraints on hydrological and paleotemperature variations in the Eastern Mediterranean region in the last 140 ka given by the δD values of speleothem fluid inclusions. Quaternary Science Reviews, 23, 919–934.
Mercier, N., & Valladas, H. (2003). Reassessment of TL age estimates of burnt flints from the Paleolithic site of Tabun Cave, Israel. Journal of Human Evolution, 45, 401–409.
Miller, G. H., Fogel, M. L., Magee, J. W., Gagan, M. K., Clarke, S. J., & Johnson, B. J. (2005). Ecosystem collapse in Pleistocene Australia and a human role in megafaunal extinction. Science, 309(5732), 287–290.
Munro, N. D., Bar-Oz, G., & Stutz, A. J. (2009). Aging mountain gazelle (Gazella gazella): Refining methods of tooth eruption and wear and bone fusion. Journal of Archaeological Science, 36, 752–763.
Neuville, R. (1951). Le Paléolithique et le Mésolithique de Désert de Judée. Paris: Masson et Cie Editeurs.
O’Connell, J. F., Hawkes, K., & Blurton Jones, N. (1988). Hadza hunting, butchering, and bone transport and their archaeological implications. Journal of Anthropological Research, 44, 113–161.
O’Connell, J. F., Hawkes, K., & Blurton-Jones, N. (1989). Reanalysis of large mammal body part transport among the Hadza. Current Anthropology, 29, 356–363.
Osmond, J. K., & Dabous, A. A. (2004). Timing and intensity of groundwater movement during Egyptian Sahara pluvial periods by U-series analysis of secondary U in ores and carbonates. Quaternary Research, 61, 85–94.
Payne, S. (1975). Partial recovery and sample bias. In A. Classon (Ed.), Archaeozoological studies (pp. 7–17). Ámsterdam: North Holland.
Pérez Ripoll, M. (1992). Marcas de carnicería, fracturas intencionadas y mordeduras de carnívoros en huesos prehistóricos del Mediterráneo español. Alicante: Instituto de Cultura Juan Gil-Albert.
Pickering, T. R., & Egeland, C. P. (2006). Experimental patterns of hammerstone percussion damage on bones: Implications for inferences of carcass processing by humans. Journal of Archaeological Science, 33, 459–469.
Pielou, E. C. (1966). The measurement of diversity in different types of biological collections. Journal of Theoretical Biology, 13, 131–144.
Pike, J. G. (1964). The estimation of annual runoff from meteorological data in a tropical climate. Journal of Hydrology, 2, 116–123.
Quam, R. M., & Smith, F. H. (1998). A reassessment of the Tabun C2 Mandible. In T. Akazawa, K. Aoki, & O. Bar-Yosef (Eds.), Neanderthals and modern humans in Western Asia (pp. 405–422). New York: Plenum.
Rabinovich, R. (1990). Taphonomic research on the faunal assemblage from the Quneitra site. In N. Goren-Inbar (Ed.), Quneitra: A Mousterian site on the Golan heights (pp. 189–219). Jerusalem: The Hebrew University.
Rabinovich, R., & Hovers, E. (2004). Faunal analysis from Amud Cave: Preliminary results and interpretations. International Journal of Osteoarchaeology, 14, 287–306.
Rabinovich, R., & Tchernov, E. (1995). Chronological, paleoecological and taphonomical aspects of the Middle Paleolithic site of Qafzeh, Israel. In H. Buitenhuis & H. P. Uerpmann (Eds.), Archaeozoology of the Near East II (pp. 5–44). Leiden: H. Backhuys.
Rak, Y. (1998). Does any Mousterian Cave present evidence of two species? In T. Akazawa, K. Aoki, & O. Bar-Yosef (Eds.), Neanderthals and modern humans in Western Asia (pp. 353–366). New York: Plenum.
Shea, J. J. (2003a). The Middle Paleolithic of the East Mediterranean Levant. Journal of World Prehistory, 17(4), 313–394.
Shea, J. J. (2003b). Neanderthals, competition, and the origin of modern human behavior in the Levant. Evolutionary Anthropology, 12, 173–187.
Shea, J. J. (2008). Transitions or turnovers? Climatically-forced extinctions of Homo sapiens and Neanderthals in the east Mediterranean Levant. Quaternary Science Reviews, 27, 2253–2270.
Shea, J. (2009). The impact of projectile weaponry on Late Pleistocene hominin evolution. In J.-J. Hublin & M. P. Richards. (Eds.), The evolution of hominin diets: Integrating approaches to the study of Paleolithic subsistence (pp. 189–199). Vertebrate Paleobiology and Paleoanthropology Series. Dordrecht: Springer.
Shea, J. J., & Bar-Yosef, O. (2005). Who were the Skhul/Qafzeh people? An archaeological perspective on Eurasia’s earliest modern humans. Journal of the Israel Prehistoric Society, 35, 449–466.
Shea, J. J., & Sisk, M. L. (2010). Complex projectile technology and Homo sapiens dispersal into Western Eurasia. PaleoAnthropology, 2010, 100–122.
Shipman, P. (1981). Life history of a fossil: An introduction to taphonomy and paleoecology. Cambridge: Harvard University Press.
Siddall, M., et al. (2006). Red Sea sea Level Reconstruction. IGBP PAGES/World Data Center for Paleoclimatology. Data Contribution Series # 2006-063. NOAA/NCDC Paleoclimatology Program, Boulder, CO, USA.
Simpson, E. H. (1949). Measurement of diversity. Nature, 163, 688.
Speth, J. D. (2004). Hunting pressure, subsistence intensification and demographic change in the Levantine late Middle Paleolithic. In N. Goren-Inbar & J. D. Speth (Eds.), Human paleoecology in the Levantine Corridor (pp. 149–166). Oxford: Oxbow Press.
Speth, J. D., & Clark, J. (2006). Hunting and overhunting in the Levantine late Middle Paleolithic. Before Farming, 3, 1–42.
Speth, J. D., & Tchernov, E. (1998). The role of hunting and scavenging in Neanderthal procurement strategies: New evidence from Kebara Cave. In T. Akazawa, K. Aoki, & O. Bar-Yosef (Eds.), Neanderthals and modern humans in Western Asia (pp. 223–240). New York: Plenum.
Speth, J. D., & Tchernov, E. (2001). Neanderthal hunting and meat processing in the Near East: Evidence from Kebara Cave (Israel). In C. B. Stanford & H. T. Bunn (Eds.), Meat eating and human evolution (pp. 52–72). New York: Oxford University Press.
Stiner, M. C. (1991). Food procurement and transport by human and no-human predators. Journal of Archaeological Science, 18, 455–482.
Stiner, M. C. (1994). Honour among thieves: A zooarchaeological study of Neanderthal ecology. Princeton: Princeton University Press.
Stiner, M. C. (2005). The Faunas of Hayonim Cave (Israel): A 200,000-year record of Paleolithic diet, demography and society. Cambridge: Peabody Museum of Archaeology and Ethnology.
Stiner, M. C., & Tchernov, E. (1998). Pleistocene species trends at Hayonim Cave: Changes in climate versus human behavior. In T. Akazawa, K. Aoki, & O. Bar-Yosef (Eds.), Neanderthals and modern humans in Western Asia (pp. 241–262). New York: Plenum.
Stiner, M. C., Munro, N., Surovell, T. A., Tchernov, E., & Bar-Yosef, O. (1999). Paleolithic growth pulses evidenced by small animal exploitation. Science, 283, 190–194.
Tchernov, E. (1998). The faunal sequences of the southwest Asian Middle Paleolithic in relation to hominid dispersal events. In T. Akazawa, K. Aoki, & O. Bar-Yosef (Eds.), Neanderthals and modern humans in Western Asia (pp. 77–90). New York: Plenum.
Turc, L. (1954). Le bilan d’eau des sols. Relation entre la précipitation, l’évaporation et l’écoulement. Annales agronomiques, 5, 491–569.
Turville-Petre, F. (1932). Excavations in the Mugharet el-Kebarah. Journal of the Royal Anthropology Institute, 62, 271–276.
Vaks, A., Bar-Matthews, M., Ayalon, A., Matthews, A., Frumkin, A., Dayan, U., et al. (2006). Paleoclimate and location of the border between Mediterranean climate region and the Saharo-Arabian Desert as revealed by speleothems from the northern Negev Desert, Israel. Earth Planetary Science Letters, 249, 384–399.
Vaks, A., Bar-Matthews, M., Ayalon, A., Halicz, L., & Frumkin, A. (2007). Desert speleothems reveal climatic window for African exodus of anatomically modern humans. Geology, 35, 831–834.
Waldmann, N., Stein, M., Ariztegui, D., & Starinsky, A. (2009). Stratigraphy, depositional environments and level reconstruction of the last interglacial Lake Samra in the Dead Sea basin. Quaternary Research, 72, 1–15.
Wallace, I., & Shea, J. (2006). Mobility patterns and core technologies in the Middle Paleolithic of the Levant. Journal of Archaeological Science, 33, 1293–1309.
Weaver, T., & Steudel-Numbers, K. (2005). Does climate or mobility explain the differences in body proportions between Neanderthals and their Upper Paleolithic successors? Evolutionary Anthropology, 14, 218–223.
Yeshurun, R., Bar-Oz, G., & Weinstein-Evron, M. (2007). Modern hunting behavior in the early Middle Paleolithic: Faunal remains from Misliya Cave, Mount Carmel, Israel. Journal of Human Evolution, 53, 656–677.
Acknowledgments
I would like to especially thank John Speth and Jamie Clark for inviting me to participate in this fascinating ICAZ session. This work could not have been done without the kind help of NHM (London) staff, above all from Mark Lewis, Adrian Lister, Chris Stringer and Andy Currant. Mark Lewis also edited the English for this manuscript. The research was funded with a British Academy Postdoctoral Fellowship in LCHES (University of Cambridge), where Robert Foley and Marta Lahr-Mirazón gave me all their support and encouragement. Finally, my sincere thanks to David Ocio for all his constant support.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2013 Springer Science+Business Media Dordrecht
About this chapter
Cite this chapter
Marín-Arroyo, A.B. (2013). New Opportunities for Previously Excavated Sites: Paleoeconomy as a Human Evolutionary Indicator at Tabun Cave (Israel). In: Clark, J., Speth, J. (eds) Zooarchaeology and Modern Human Origins. Vertebrate Paleobiology and Paleoanthropology. Springer, Dordrecht. https://doi.org/10.1007/978-94-007-6766-9_5
Download citation
DOI: https://doi.org/10.1007/978-94-007-6766-9_5
Published:
Publisher Name: Springer, Dordrecht
Print ISBN: 978-94-007-6765-2
Online ISBN: 978-94-007-6766-9
eBook Packages: Earth and Environmental ScienceEarth and Environmental Science (R0)