Abstract
A wealth of data points to the existence of a subset of tumor-initiating cells that have properties similar to stem cells, termedcancer stem cells (CSCs). CSCs are thought to be at the apex of a cellular hierarchy, where they are capable of differentiating into the other cells found within a tumor. They may also be responsible for both patient relapse due to their relative resistance to chemotherapy as well as metastasis. In recent years, much research has focused on these cells, their properties and potential targets within these cells for cancer treatment. This chapter will introduce the CSC theory, discuss important properties of these cells, and highlight the need to target them for improved patient outcome.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Cancer Stem Cell
- Acute Myelogenous Leukemia
- Side Population
- Side Population Cell
- Oral Squamous Cell Carcinoma Cell
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
1.1 The Etiology of Cancer
Many hypotheses have been put forth through the years that attempt to explain the etiology of cancer. They have come from divergent fields of study; pathology, molecular biology and genetics but they all attempt to explain how a normal tissue can go from homeostatic equilibrium to something that grows without the checks and balances that govern normal biology. These theories include, but are not limited to, a viral basis of disease, clonal expansion, and the cancer stem cell hypothesis. While each of these theories attempts to explain the etiology of cancer, it is most likely that some of these independent theories work together to give rise to not only tumors, but tumors that are able to evade treatment strategies.
1.2 The Cancer Stem Cell Hypothesis
Since the early pathologists could look at tissues microscopically the origins and progression of cancer has been pondered. It was noted that advanced tumors often presented with diverse areas of differentiation, proliferation, invasion and vascularity. It was during this time of early microscopic inspection that led Cohnheim, a student of Virchow, to propose his embryonal rest hypothesis ([1] and references therein). Cohnheim had noted that cancer tissues, teratomas in particular, had many of the same properties as embryonic tissue. This lead him to speculate that carcinogenesis occurs from dormant remnants of the embryo that are later reactivated. However, this theory was largely ignored. The spirit of this hypothesis reappeard in the 1970s when Barry Pierce et al. examined differentiation in teratomas (reviewed in [1])and determined that a stem cell was responsible for initiation of the teratomas. They then furthered these observations into a theory that all epithelial cancers arise as a result of differentiation-paused adult tissue stem cells [2].
On the heels of this hypothesis, came some of the first evidence that leukemias may have a CSC origin. John Dick and colleagues showed that a sub-population of acute myelogenous leukemia (AML) cells, which shared a phenotype with normal hematopoietic stem cells, could confer cancer when transplanted into immunocompromised mice. Furthermore, the cells that did not have the stem cell-phenotype could not transfer AML to recipient mice [3, 4]. Several years later, CSCs were identified in breast cancer [5], followed by a myriad of solid malignancies (discussed in more detail below in “1.4 Identification of CSCs”).
1.3 Properties and Cell of Origin of CSCs
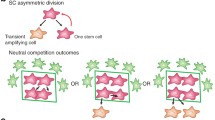
Cancer stem cells are so named because they share many of the same properties as normal stem cells. They are capable of tumorigenesis, self-renewal and can differentiate to form the heterogeneous cell types present in tumors (Fig. 1.1a). These functional properties led to the CSC moniker; however, many argue that “tumor-initiating cells” would be a better description of these cells. For the purposes of this book, we will refer to these cells as CSCs, where a .CSC has been defined as “a cell within a tumor that possess the capacity to self-renew and to cause the heterogeneous lineages of cancer cells that comprise the tumor” [6]. It has also been observed that CSCs are relatively resistant to chemotherapy and therefore may be responsible for patient relapse following treatment (Fig. 1.1b, discussed in detail in Chap. 3). Thus, the CSC hypothesis attempts to explain several observations of tumors, including the frequency at which tumor cells can give rise to new tumors, the generation of cells with multiple genetic alterations, and the heterogeneity of cell types present within tumors.
Schematic representation of CSC-driven tumor formation. (a) CSCs can either divide symmetrically (self-renewal) to give rise to two CSCs or they can divide asymmetrically (differentiation) giving rise to one CSC and one differentiated progeny. The ability to give rise to a tumor, to self-renewal and differentiate into the heterogeneous cell populations found in the tumor are the defining characteristic of a CSC. The differentiated progeny of a CSC are often more proliferative than the CSC itself, but they have a finite replicative capacity. (b) CSCs are resistant to conventional therapies, including chemotherapy and radiation. These treatments can eliminate the more rapidly dividing differentiated cells but leave behind the CSC (discussed in more detail in Chap. 3). Once treatment is stopped, the remaining CSCs can begin to divide again and form a new tumor resulting in patient relapse
In theory, cancer arises from a single cell that has somehow subverted normal growth restrictions. However, experimental evidence has shown a requirement of many cells in order to seed a tumor. For example the growth of tumor cells in immunocompromised mice, typically requires that 1–10 million cells are implanted in order to see tumor formation. This observation has many possible explanations, including injury to cells upon injection, the requirement for the right microenvironment (and the difference of this between human and mouse), as well as the requirement to have a variety of cell types present in order to efficiently induce tumor formation. However, even in experiments where human cancer patients were reinjected with their own tumor cells at different sites within their bodies, it took large numbers of cells in order for a new tumor to establish. Again, this could be consistent with the role of the tumor microenvironment, this time from one site to another; it is also consistent with the notion that only a small proportion of cells are capable of giving rise to a tumor. The idea that only a small proportion of the cells found within a tumor are capable of giving rise to a tumor is consistent with the CSC hypothesis. The frequency of CSCs in most cases has been reported to be low (typically less than 5 %) except in the case of melanoma where a variety of cells were shown to have equally high tumorigenicity [7, 8].
The origin of the cancer stem cell is still under debate. The requirement for multiple genetic insults in order to drive tumor formation has been recognized for a long time [9]. It is suggested that in order for a cell to live long enough to sustain the genetic insults required to drive tumor formation, it is likely that a normal adult stem cell is the originator of a CSC phenotype. Mathematical models support this theory [10]. Furthermore, the leukemic stem cells (LSCs) of CML patients express BCR-ABL, the common translocation that drives cellular transformation [11]. This provides experimental evidence that CSCs sustain the genetic insults that are seen to drive carcinogenesis. The identification in CSCs of oncogenic driver mutations is also beginning to emerge for solid tumors. For example, the TMPRSS:ERG fusion has been found in prostate CSCs [12].
However, it is still possible that a more differentiated cell undergoes these genomic rearrangements and additionally picks up further mutations that impart the ability to self-renew as well as differentiate. Indeed fibroblasts can acquire the properties of pluripotent stem cells with the activation of just a few genes [13–15]. Moreover, it was recently shown that a catastrophic event in the cell can lead to many genetic alterations at a single time [16]. It may also be that environmental cues can trigger CSC properties even independent of DNA rearrangements. For example, it has been noted that cells undergoing epithelial-to-mesenchymal transition (EMT ), a normal developmental process that promotes cancer invasion and metastasis , can acquire characteristics of CSCs [17]. These ilines of evidence suggest that a cell does not need to be long-lived in order to sustain many genetic alterations and that CSC properties can be bestowed by biological processes, and would therefore argue that any cell may be the fodder for CSCs.
1.4 Isolation of CSCs
Currently, there are several commonly used approaches for the isolation of cancer stem cells, including: (1) sorting of a side population (SP) by flow cytometry based on Hoechst dye efflux, (2) sorting of CSCs by flow cytometry based on cell surface marker expression, (3) enriching of CSCs by non-adherent sphere culture and (4) sorting of CSCs by flow cytometry based on aldehyde dehydrogenase (ALDH) activity (Fig. 1.2). All of these approaches enrich for CSCs to varying degrees, and each of them has its own advantages and limitations, which will be discussed below.
1.4.1 Side Populations
Goodell and colleagues, while analyzing murine bone marrow cells, discovered a small and distinct subset of whole bone marrow cells that were unstained by Hoechst 33342, a vital dye [18]. This Hoechst 33342 low population is termed SP. They found that the SP had the phenotypic markers of multipotential hematopoietic stem cells and were able to repopulate the bone marrow. Following their work, the SP has been extended to a variety of cancer types, including leukemia [19, 20], ovarian cancer [21], hepatocellular carcinoma [22], brain tumors [23–25], lung cancer [26, 27], thyroid cancer [28], nasopharyngeal carcinoma [29], mesenchymal tumors [30], colon cancer [31], prostate cancer [32], breast cancer [33–35], head and neck cancer [36] and other cancers [37–40]. The SP cells purified from these tumor types harbor cancer stem cell-like cells with properties such as a “stemness” gene signature [23, 26, 28, 29, 35], self-renewal capacity [25, 26, 28, 29, 33, 35] and tumorigenicity [19, 21, 24–26, 28, 29, 35].
The ATP dependent transporter, ATP-binding cassette sub-family G member 2 (ABCG2, BRCP1), is generally believed to be responsible for Hoechst 33342 efflux by the SP. Several lines of evidence support this hypothesis. First, ABCG2 knockout mice show significantly decreased numbers of SP cells in both the bone marrow as well as in skeletal muscle [41]. Second, overexpression of ABCG2 dramatically increases the SP percentage of bone-marrow cells and reduces maturing progeny both in vitro and in vivo [42]. Third, ABCG2 is highly expressed in a wide variety of stem cells including the SP cells of neuroblastoma patients [23, 42]. Fourth, ABCG2 may be responsible for conferring drug resistance to the SP and CSCs. ABCG2 is a multidrug resistant pump expressed at variable levels in cancer cells, which can bind and expel cytotoxic drugs [43]. Thus, ABCG2 may lower intracellular levels of anticancer agents below the threshold for cell death in tumors, leaving resistant cells to repopulate the tumor. Indeed, inhibition of the ABC transporters sensitized SP cells of various cancer types to chemotherapeutic agents [44, 45].
However, there is not always a correlation between ABCG2 expression and the SP phenotype. For example, erythroblasts highly express the ABC transporter ABCG2 but do not have an SP phenotype [46]. Furthermore, the expression of ABCG2, and the presence of the SP in general, do not always define the CSC population. For instance, in prostate cancer both purified ABCG2+ and ABCG2− cancer cells have similar tumorigenicity to the SP cells in vivo, although the SP cells express higher level of ABCG2 mRNA than the non-SP cells [32]. Additionally, ABCG2 is not the only multi-drug resistance gene identified in SP stem-like cells. The expression of P-glycoprotein (ABCB1, MDR1) is also significantly up-regulated in SP cells of an oral squamous cell carcinoma cell line [47], although Feuring-Buske et al. found that there is no correlation between the expression of ABCB1 and the SP in acute myeloid leukemia [19]. Instead, Zhou et al. demonstrated that Hoechst 33342 efflux activity is compensated by ABCG2 in Abcb1 null mice [42], indicating that SP cells may utilize different drug transportation machineries in different environments. In addition, the presence of the SP may instead be the result of inefficient dye uptake as a reflection of the presence of largely quiescent cells, another characteristic of stem cells [26, 48].
Compared to other methods, isolation of the side population has two advantages. First, it carries additional information about the functional status of the cells, since this assay is based on an active metabolic process. Second, it is highly sensitive, with even rare SP events (< 0.5 % of the total cell population) detected within heterogeneous samples [49].
But this method also has many disadvantages. Some of the limitations of this method have to do with the Hoechst 33342 dye itself. Owing to the fact that Hoechst 33342 is a DNA binding dye, it is toxic to cells. Shen and colleagues found that Hoechst 33342 staining for a prolonged periods of time increases apoptosis in C6 cells [25]. Furthermore, it is highly sensitive to slight variations in staining conditions. Hoechst concentration, the staining time, and the staining temperatures all are critical for the success of this approach.
Other disadvantages of SP isolation are due to the ability of this method to accurately define and purify CSCs. Importantly, the SP is not always necessary or sufficient for a CSC phenotype. In glioblastoma multiforme (GBM), the SP from the GBM lines did not enrich for stem-like activity in vitro, and tumorigenicity was lower in sorted SP compared with non-SP and parental cells [50]. Equally important, is that SP cells represent a heterogeneous cell population. Wan et al. demonstrated that SP cells purified from a laryngeal cancer cell line does harbor cancer stem cell-like properties, but they are heterogeneous indicating that SP cells are not identical to stem cells [51]. Combining SP detection with cell surface marker selection, may lead to a more efficient and reliable isolation of CSCs.
Despite the limitations existing in SP isolation, the presence of the SP population has some clinical relevance in certain disease indications. For example, the SP population can be identified in gastric cancer tissue and correlates with patient survival [37]. A limited clinical study in ovarian cancer also revealed a higher SP frequency in recurrent or metastatic tumors compared with primary tumors, suggesting a good correlation between the presence of SP and recurrence in ovarian cancer [52].
Besides the use in prognosis of cancer, the SP may also serve as a potential therapeutic target for cancer. Several studies have pioneered the possibility of specifically targeting SP cells by exploiting pathways involved in drug resistance and differentiation [53]. For instance, Praveen et al. have isolated SP cells from multiple cancer cell lines and found that these SP cells are resistant to cytochrome C release and apoptosis. Based on this finding, they developed a high-throughput imaging assay, in which the cytochrome C-EGFP translocation is monitored in the sorted SP cells. Through this assay, the heat shock protein 90 inhibitors have been identified to sensitize the SP cells to some antitumor agents, such as cisplatin [54]. Moreover, an autologous vaccine for B-cell chronic lymphocytic leukemia (B-CLL) was made using a patient’s SP cells. Following vaccination, the study showed an increase in (B-CLL)-reactive T-cells followed by a corresponding decline in circulating B-CLL SP cells [55]. This indicates that the SP may be a valid ground for cancer therapy.
1.4.2 Surface Markers
Initially used to identify and isolate normal stem cells, surface markers are now extensively used for the identification and isolation of CSCs in many malignances. Lapidot and colleagues were the first to isolate leukemia-initiating cells based on cell-surface marker expression and found CD34+ CD38- cells, but not the CD34+ CD38+ and CD34- cells, harbored serial leukemic transplantation potential [3]. Following this initial prospective isolation of leukemia stem cells, breast CSCs were identified as CD44 + CD24 -/low by Al-Hajj and colleagues [5]. Later, based on their surface marker expression, CSCs have been isolated from various tumors (Table 1.1). Among all the surface markers used for CSCs isolation, CD133 and CD44 are the most commonly used in a variety of tumor types.
CD133 (Prominin 1), a five transmembrane glycoprotein, was originally identified both in the neuroepithelium and in various other epithelia of the mouse embryo [77]. Later, a novel monoclonal antibody recognizing the AC133 antigen, a glycosylation-dependent epitope of CD133, detected that CD133 is restricted in CD34+ progenitor populations from adult blood, bone marrow and fetal liver cells [78]. In addition, CD133 expression is rapidly down regulated upon cell differentiation [79]. These characteristics of CD133 make it a unique cell surface marker for the identification and isolation of various CSCs (Table 1.1). The biological function of CD133 is still largely unknown. A single nucleotide deletion, which caused the truncation of CD133, is linked to an inherited form of human retinal degeneration [80]. A recent report has linked CD133 with endocytosis. In this study, CD133 knockdown improved Alexa488-transferrin (Tf) uptake in Caco-2 cells, while cell treatment with the AC133 antibody resulted in down regulated Tf uptake, [81]. Despite its utility as a marker of CSCs, CD133 does not appear to play a significant role in the maintenance of at least some CSCs. In colon cancer cells isolated from patients, CD133 knockdown did not affect their tumorigenicity in vitro and in vivo [82]. Instead, CD44 knockdown prevented tumor formation of the same cells.
CD44 , a single transmembrane glycoprotein, is a major component of the extracellular matrix [83]. Besides acting as an adhesion molecule, it also functions as a principle receptor for hyaluronan (HA) [84]. HA is enriched in the pericellular matrices of many malignant human tumors and plays an important role in tumor progression via regulation of receptor tyrosine kinases (RTKs), such as ERBB2 and EGFR [85]. Thus, as a critical receptor for HA, CD44 plays an important role in cell proliferation and survival via activation of the MAPK and PI3K/AKT pathways, respectively [85]. Furthermore, CD44 also plays an important role in the invasion of a variety of cancer cells, including breast [86], prostate [87], hepatoma [88], and mesotheliomas [89], and has been significantly correlated with the circulating prostate tumor cells [90]. Therefore, CD44 stands out as a surface marker for CSCs, as first shown by Al-hajj and colleagues in breast cancer [5]. Following their work, CD44 has been utilized as a surface marker to isolate CSCs from a variety of different tumors (Table 1.1).
CD44 was also explored as a potential diagnostic target for cancer detection as well as a drug target for cancer therapy [91–93]. For instance, in 2003, the humanized anti-CD44 antibody (bivatuzumab) labeled with rhenium-186 was used in phase I studies in patients with head and neck squamous cell carcinoma (HNSCC) [94, 95]. In this trial, these radiolabeled CD44 antibodies showed promising anti-tumor effects with low toxicity. Further, a different trial with the non-radiolabeled CD44 antibody (bivatuzumab mertansine) also had good patient response rates, although the development of this drug was terminated due to the death of a patient [96]. Recently, CD44 was also shown to target CSCs. In 2006, Jin et al. found that interruption of CSCs interaction with their microenvironment by monoclonal antibody directed against CD44 markedly decreased the number of the AML LSCs in vivo, indicating a key regulatory role of CD44 in AML LSCs [97].
While CD44 and CD133 appear to have broad tumor applicability, the choice of the cell surface markers tends to be tissue specific and is often based on previous knowledge of the development of that tissue. For instance, CD34 and CD38, the markers used for isolation of AML CSCs, are also the markers used to identify normal early hematopoietic progenitor cells [3, 4]. Another good example is CD138. CD138 is a marker for terminally-differentiated B cells (plasma cells). It has been shown that CSCs from multiple myeloma, a plasma cell malignancy, are CD138-.
Compared to other methods of isolation, cell surface marker isolation has a major advantage of obtaining a precise population. However, the selection of which surface markers to use is one of the greatest pitfalls of this approach. As discussed earlier, many times markers selected are based on previous knowledge of the development of the tissue. For this reason, it may not be easy to find the right markers. For example, until recently there were no markers identified for CSCs of human primary gastric tumors [98]. Furthermore, the choice of markers made by researchers has not been unified, even within the same tumor type. As shown in Table 1.1, different markers have been used for the same tumor type. To further, complicate the picture many times there have not been careful comparisons done within the same study of all the proposed CSC markers in order to definitively test which marker combination is the best at identifying CSCs. A further complication is that the surface markers may be heterogeneous between patients even within the same tumor type. A recent study from 16 AML patients shows that the majority of LSCs are in the minor CD34+ CD38- fraction in 50 % of the subjects, and in the CD34+ CD38+ fraction in the other 50 % [99]. Similar findings were also obtained from breast cancer patients. When Park et al. [92] evaluated the expression of stem cell-related markers at the cellular level in human breast tumors of different subtypes and histologic stages, they found that the cancer stem cell markers vary according to tumor subtype and histologic stage [100]. Ali et al. also demonstrated that breast CSC markers, such as CD44/CD24, ALDH and ITGA6, do not identify identical subpopulations in primary tumors [101].
Other limitations of choosing surface markers for the identification of CSCs are methodological. First, a large number of cells is required to sort and the number of CSCs identified by this approach is usually low (< 1 - 10 %). Furthermore, when using tumor samples, the cells must first be dissociated typically with collagenase and/or other proteolytic enzymes. This dissociation step may damage the presentation of the cell surface antigens [102, 103].
1.4.3 Nonadherent Sphere Culture
Initially, nonadherent sphere culture was used to culture neural stem cells [104]. The cells isolated from the stratum of adult mouse by this method could generate both neurons and astrocytes. Later, it was shown that purified stem cells were capable of growing as spheres. For instance, CD133+ cells isolated from normal human fetal brain formed spheres in vitro [105]. These observations were then extended to cancerous tissues where further studies demonstrated that CD133+ cells from the human brain tumors are also capable of forming neurospheres [57]. Since then, the ability of CSCs to form spheres in culture has been shown for most solid malignancies, including breast [106], melanoma [107], pancreatic [108], prostate [109], ovarian [75] and colon CSCs [110].
Most importantly, researchers found that the nonadherent sphere culture condition can enrich cells with CSC phenotypes [106]. They demonstrated that, in breast cancer, the CSC population with CD44+ CD24- phenotype increased more than two-fold after culturing under nonadherent sphere culture conditions. In addition, the spheres were more tumorigenic. Since then, researchers have used sphereto formation to enrich CSCs from brain [56, 57, 111, 112], colon [110], pancreas [108], bone sarcomas [113] melanomas [107] and prostate [109]. The enrichment of CSCs by growing cells under sphere culture conditions has been confirmed by surface marker expression in all mentioned cases, except bone sarcoma, in which the surface marker remain to be determined.
Compared to the other methods of isolating CSCs, this method is easy to perform and allows researchers to obtain a larger number of CSCs. However, like all methods of isolation, this method also suffers from several disadvantages. The spheres are a heterogeneous population of cells containing both CSCs and non-CSCs. For example it has been shown that only a portion of the spheres are capable of self-renewal [106, 114]. Immunostaining of spheres from prostate cancer cell lines indicated that the spheres are heterogeneous for CSC markers [115]. Also, the conditions of sphere formation are critical to the overall success of CSC enrichment. In neurosphere cultures, it has been shown that the composition of spheres can be different due to the differences in sphere size, passage, culture medium, and technique [116].
1.4.4 ALDH Activity
In addition to the above three isolation methods, isolation of CSCs based on their aldehyde dehydrogenase (ALDH) activity is also commonly used. ALDH is a detoxifying enzyme responsible for the oxidation of intracellular aldehydes [117]. It functions in drug resistance, cell differentiation and oxidative stress response [118]. ALDH activity may be easily assessed in living cells using the ALDEFLUOR kit (Stem Cell Technologies). This kit utilizes an ALDH substrate BODIPY aminoacetaldehyde (BAAA), which is converted in the cytoplasm into a florescent molecule by ALDH enzymatic activity [119]. Recent studies have demonstrated a positive correlation between ALDH expression and overall survival of patients with different cancers [120]. To date, ALDH activity has been used to identify and isolate CSCs from AML [121], breast [122], melanoma [123], prostate [124], liver [71], ovarian [125], lung [126] and osteosarcoma [127].
Like other isolation methods, ALDH is also not a universal marker for cancer stem cells in any tumor type. In a CSC marker profiling study, the breast cancer cells selected by ALDH activity did not always identify the most tumorigenic cells [128]. Furthermore, in prostate cancer, Yu et al. [14] found that ALDHlo CD44- cells were also able to develop tumors with similar frequency as the ALDHhiCD44+ cells, although with longer latency periods [129].
To date, there is not a universal isolation method in the CSC field. Researchers have to select their isolation method depending on the purpose of their studies, and sample source. Recently, Zhou et al. demonstrated that SP rather than CD133(+) cells indicate enriched tumorigenicity in hTERT-immortalized primary prostate cancer cells [130]. Sometimes, two different methods may need to be combined to isolate CSCs. For instance, combining SP determination with cell-surface marker phenotyping leads to efficient, reliable characterization of the HSC subset [131]. Further, Eppert and colleagues found that AML LSCs from different patient samples have diverse surface marker profiles and frequency [99]. However, these LSCs shared a core gene signature, indicating that the stemness gene signature might be a more accurate way to identify CSCs.
1.5 Implications of the CSC Hypothesis for Cancer Treatment
The discovery of cancer stem cells has important implications for oncology, and many groups are now working to understand and exploit CSC biology to improve cancer treatment. Despite tremendous progress in the identification of new cancer targets and drug development, most cancers relapse after treatment and the goal of increasing overall survival of cancer patients has been largely unmet by most new drugs [132]. One explanation for this lack of sustained effect is that most current therapies do not effectively inhibit CSCs.
Indeed, CSCs are resistant to chemo- and radiation therapies that effectively kill other cells that comprise the tumor (discussed in detail in Chap. 3). It is for this reason that CSCs are thought to regenerate cancer when therapy is discontinued. This hypothesis holds that, because CSCs are a small population within the tumor mass, drugs developed to kill the cells comprising the bulk of the tumor are initially effective in shrinking tumor size but do not eradicate the cellular source and tumors with higher CSC content and perhaps harboring additional drug-resistance mutations regrow. This has been termed the “dandelion hypothesis,” analogous to the regrowth of a weed if the root is not destroyed [133]. Although this hypothesis has yet to be fully confirmed using a CSC-directed therapy in cancer patients, numerous studies have correlated CSCs with poor prognosis in both leukemia and solid malignancies. For example, glioblastoma multiforme patients whose tumors bear a relatively high proportion of CD133 + cells, which have been found to possess characteristics of CSCs, suffer from decreased response to therapy, higher malignancy, and significantly lower survival time [134]. Additionally, gene signatures derived from CSCs are themselves independent predictors of patient survival [99, 135] indicating the relevance of targeting these cells.
1.6 Concluding Remarks
A consensus is emerging that the CSC population represents a distinct and clinically important population. However, there is still a further need to determine the precise definition and cellular source of CSCs, to elucidate the appropriate cell markers to isolate them, and to understand the pathways contributing their biological behavior. In the end there is growing hope that this understanding will translate into clinically useful treatments against cancer.
References
Pierce GB, Johnson LD (1971) Differentiation and cancer. In Vitro 7:140–145
Sell S, Pierce GB (1994) Maturation arrest of stem cell differentiation is a common pathway for the cellular origin of teratocarcinomas and epithelial cancers. Lab Invest 70:6–22
Lapidot T, Sirard C, Vormoor J et al (1994) A cell initiating human acute myeloid leukaemia after transplantation into SCID mice. Nature 367:645–648
Bonnet D, Dick JE (1997) Human acute myeloid leukemia is organized as a hierarchy that originates from a primitive hematopoietic cell. Nat Med 3:730–737
Al-Hajj M, Wicha MS, Benito-Hernandez A, Morrison SJ, Clarke MF (2003) Prospective identification of tumorigenic breast cancer cells. Proc Natl Acad Sci USA 100:3983–3988
Clarke MF, Dick JE, Dirks PB et al (2006) Cancer stem cells—perspectives on current status and future directions: AACR Workshop on cancer stem cells. Cancer Res 66:9339–9344
Ishizawa K, Rasheed ZA, Karisch R et al (2010) Tumor-initiating cells are rare in many human tumors. Cell Stem Cell 7:279–282
Shackleton MJ, Quintana E, Fullen DR, Sabel MS, Johnson TM (2009) Melanoma: do we need a hatchet or a scalpel? Arch Dermatol 145:307–308
Nordling CO (1953) A new theory on cancer-inducing mechanism. Br J Cancer 7:68–72
Ashkenazi R, Gentry SN, Jackson TL (2008) Pathways to tumorigenesis—modeling mutation acquisition in stem cells and their progeny. Neoplasia 10:1170–1182
Maguer-Satta V, Petzer AL, Eaves AC, Eaves CJ (1996) BCR-ABL expression in different subpopulations of functionally characterized Ph+ CD34+ cells from patients with chronic myeloid leukemia. Blood 88:1796–1804
Birnie R, Bryce SD, Roome C et al (2008) Gene expression profiling of human prostate cancer stem cells reveals a pro-inflammatory phenotype and the importance of extracellular matrix interactions. Genome Biol 9:R83
Takahashi K, Yamanaka S (2006) Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 126:663–676
Yu J, Vodyanik MA, Smuga-Otto K et al (2007) Induced pluripotent stem cell lines derived from human somatic cells. Science 318:1917–1920
Takahashi K, Tanabe K, Ohnuki M et al (2007) Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell 131:861–872
Stephens PJ, Greenman CD, Fu B et al (2011)Massive genomic rearrangement acquired in a single catastrophic event during cancer development. Cell 144:27–40
Mani SA, Guo W, Liao MJ et al (2008) The epithelial-mesenchymal transition generates cells with properties of stem cells. Cell 133:704–715
Goodell MA, Brose K, Paradis G, Conner AS, Mulligan RC (1996) Isolation and functional properties of murine hematopoietic stem cells that are replicating in vivo. J Exp Med 183:1797–1806
Feuring-Buske M, Hogge DE (2001) Hoechst 33342 efflux identifies a subpopulation of cytogenetically normal CD34(+)CD38(-) progenitor cells from patients with acute myeloid leukemia. Blood 97:3882–3889
Wulf GG, Wang RY, Kuehnle I et al. (2001) A leukemic stem cell with intrinsic drug efflux capacity in acute myeloid leukemia. Blood 98:1166–1173
Szotek PP, Pieretti-Vanmarcke R, Masiakos PT et al (2006) Ovarian cancer side population defines cells with stem cell-like characteristics and Mullerian Inhibiting Substance responsiveness. Proc Natl Acad Sci USA 103:11154–11159
Chiba T, Kita K, Zheng YW et al (2006) Side population purified from hepatocellular carcinoma cells harbors cancer stem cell-like properties. Hepatology 44:240–251
Hirschmann-Jax C, Foster AE, Wulf GG et al (2004) A distinct “side population” of cells with high drug efflux capacity in human tumor cells. Proc Natl Acad Sci USA 101:14228–14233
Setoguchi T, Taga T, Kondo T (2004) Cancer stem cells persist in many cancer cell lines. Cell Cycle 3:414–415
Shen G, Shen F, Shi Z et al (2008) Identification of cancer stem-like cells in the C6 glioma cell line and the limitation of current identification methods. In Vitro Cell Dev Biol Anim 44:280–289
Ho MM, Ng AV, Lam S, Hung JY (2007) Side population in human lung cancer cell lines and tumors is enriched with stem-like cancer cells. Cancer Res 67:4827–4833
Salcido CD, Larochelle A, Taylor BJ, Dunbar CE, Varticovski L (2010) Molecular characterisation of side population cells with cancer stem cell-like characteristics in small-cell lung cancer. Br J Cancer 102:1636–1644
Mitsutake N, Iwao A, Nagai K et al (2007) Characterization of side population in thyroid cancer cell lines: cancer stem-like cells are enriched partly but not exclusively. Endocrinology 148:1797–1803
Wang J, Guo LP, Chen LZ, Zeng YX, Lu SH (2007) Identification of cancer stem cell-like side population cells in human nasopharyngeal carcinoma cell line. Cancer Res 67:3716–3724
Wu C, Wei Q, Utomo V et al (2007) Side population cells isolated from mesenchymal neoplasms have tumor initiating potential. Cancer Res 67:8216–8222
Sussman RT, Ricci MS, Hart LS, Sun SY, El-Deiry WS (2007) Chemotherapy-resistant side-population of colon cancer cells has a higher sensitivity to TRAIL than the non-SP, a higher expression of c-Myc and TRAIL-receptor DR4. Cancer Biol Ther 6:1490–1495
Patrawala L, Calhoun T, Schneider-Broussard R, Zhou J, Claypool K, Tang DG (2005) Side population is enriched in tumorigenic, stem-like cancer cells, whereas ABCG2+ and ABCG2- cancer cells are similarly tumorigenic. Cancer Res 65:6207–6219
Christgen M, Ballmaier M, Bruchhardt H, von Wasielewski R, Kreipe H, Lehmann U (2007) Identification of a distinct side population of cancer cells in the Cal-51 human breast carcinoma cell line. Mol Cell Biochem 306:201–212
Nakanishi T, Chumsri S, Khakpour N et al (2010) Side-population cells in luminal-type breast cancer have tumour-initiating cell properties, and are regulated by HER2 expression and signalling. Br J Cancer 102:815–826
Engelmann K, Shen H, Finn OJ (2008) MCF7 side population cells with characteristics of cancer stem/progenitor cells express the tumor antigen MUC1. Cancer Res 68:2419–2426
Tabor MH, Clay MR, Owen JH et al (2011) Head and neck cancer stem cells: the side population. Laryngoscope 121:527–533
Schmuck R, Warneke V, Behrens HM, Simon E, Weichert W, Rocken C (2011) Genotypic and phenotypic characterization of side population of gastric cancer cell lines. Am J Pathol 178:1792–1804
Yanamoto S, Kawasaki G, Yamada S et al (2011) Isolation and characterization of cancer stem-like side population cells in human oral cancer cells. Oral Oncol 47:855–860
Yang M, Yan M, Zhang R, Li J, Luo Z (2011) Side population cells isolated from human osteosarcoma are enriched with tumor-initiating cells. Cancer Sci 102:1774–1781
Zhang SN, Huang FT, Huang YJ, Zhong W, Yu Z (2011) Characterization of a cancer stem cell-like side population derived from human pancreatic adenocarcinoma cells. Tumori 96:985–992
Zhou S, Morris JJ, Barnes Y, Lan L, Schuetz JD, Sorrentino BP (2002) Bcrp1 gene expression is required for normal numbers of side population stem cells in mice, and confers relative protection to mitoxantrone in hematopoietic cells in vivo. Proc Natl Acad Sci USA 99:12339–12344
Zhou S, Schuetz JD, Bunting KD et al (2001) The ABC transporter Bcrp1/ABCG2 is expressed in a wide variety of stem cells and is a molecular determinant of the side-population phenotype. Nat Med 7:1028–1034
Mao Q, Unadkat JD (2005)Role of the breast cancer resistance protein (ABCG2) in drug transport. AAPS J 7:E118–133
Loebinger MR, Giangreco A, Groot KR et al (2008) Squamous cell cancers contain a side population of stem-like cells that are made chemosensitive by ABC transporter blockade. Br J Cancer 98:380–387
Katayama R, Koike S, Sato S, Sugimoto Y, Tsuruo T, Fujita N (2009) Dofequidar fumarate sensitizes cancer stem-like side population cells to chemotherapeutic drugs by inhibiting ABCG2/BCRP-mediated drug export. Cancer Sci 100:2060–2068
Yamamoto K, Suzu S, Yoshidomi Y, Hiyoshi M, Harada H, Okada S (2007) Erythroblasts highly express the ABC transporter Bcrp1/ABCG2 but do not show the side population (SP) phenotype. Immunol Lett 114:52–58
Yajima T, Ochiai H, Uchiyama T, Takano N, Shibahara T, Azuma T (2009) Resistance to cytotoxic chemotherapy-induced apoptosis in side population cells of human oral squamous cell carcinoma cell line Ho-1-N-1. Int J Oncol 35:273–80
Bhatt RI, Brown MD, Hart CA et al (2003) Novel method for the isolation and characterisation of the putative prostatic stem cell. Cytometry A 54:89–99
Golebiewska A, Brons NH, Bjerkvig R, Niclou SP (2011) Critical appraisal of the side population assay in stem cell and cancer stem cell research. Cell Stem Cell 8:136–147
Broadley KW, Hunn MK, Farrand KJ et al (2011) Side population is not necessary or sufficient for a cancer stem cell phenotype in glioblastoma multiforme. Stem Cells 29:452–461
Wan G, Zhou L, Xie M, Chen H, Tian J (2010) Characterization of side population cells from laryngeal cancer cell lines. Head Neck 32:1302–1309
Hosonuma S, Kobayashi Y, Kojo S et al (2011) Clinical significance of side population in ovarian cancer cells. Hum Cell 24:9–12
Moserle L, Ghisi M, Amadori A, Indraccolo S (2009) Side population and cancer stem cells: therapeutic implications. Cancer Lett 288:1–9
Sobhan PK, Seervi M, Joseph J et al (2011) Identification of heat shock protein 90 inhibitors to sensitize drug resistant side population tumor cells using a cell based assay platform. Cancer Lett 317:78–88
Foster AE, Okur FV, Biagi E et al (2010) Selective elimination of a chemoresistant side population of B-CLL cells by cytotoxic T lymphocytes in subjects receiving an autologous hCD40L/IL-2 tumor vaccine. Leukemia 24:563–572
Singh SK, Clarke ID, Terasaki M et al (2003) Identification of a cancer stem cell in human brain tumors. Cancer Res 63:5821–5828
Singh SK, Hawkins C, Clarke ID et al (2004) Identification of human brain tumour initiating cells. Nature 432:396–401
Son MJ, Woolard K, Nam DH, Lee J, Fine HA (2009) SSEA-1 is an enrichment marker for tumor-initiating cells in human glioblastoma. Cell Stem Cell 4:440–452
Collins AT, Berry PA, Hyde C, Stower MJ, Maitland NJ (2005) Prospective identification of tumorigenic prostate cancer stem cells. Cancer Res 65:10946–10951
Hurt EM, Kawasaki BT, Klarmann GJ, Thomas SB, Farrar WL (2008) CD44+ CD24(-) prostate cells are early cancer progenitor/stem cells that provide a model for patients with poor prognosis. Br J Cancer 98:756–765
Prince ME, Sivanandan R, Kaczorowski A et al (2007) Identification of a subpopulation of cells with cancer stem cell properties in head and neck squamous cell carcinoma. Proc Natl Acad Sci USA 104:973–978
Ma S, Chan KW, Hu L et al (2007) Identification and characterization of tumorigenic liver cancer stem/progenitor cells. Gastroenterology 132:2542–2556
Yang ZF, Ho DW, Ng MN et al (2008) Significance of CD90+ cancer stem cells in human liver cancer. Cancer Cell 13:153–166
Ricci-Vitiani L, Lombardi DG, Pilozzi E et al (2007) Identification and expansion of human colon-cancer-initiating cells. Nature 445:111–115
O’Brien CA, Pollett A, Gallinger S, Dick JE (2007) A human colon cancer cell capable of initiating tumour growth in immunodeficient mice. Nature 445:106–110
Dalerba P, Dylla SJ, Park IK et al (2007) Phenotypic characterization of human colorectal cancer stem cells. Proc Natl Acad Sci USA 104:10158–10163
Botchkina IL, Rowehl RA, Rivadeneira DE et al (2009) Phenotypic subpopulations of metastatic colon cancer stem cells: genomic analysis. Cancer Genomics Proteomics 6:19–29
Li C, Heidt DG, Dalerba P et al (2007) Identification of pancreatic cancer stem cells. Cancer Res 67:1030–1037
Hermann PC, Huber SL, Herrler T et al (2007) Distinct populations of cancer stem cells determine tumor growth and metastatic activity in human pancreatic cancer. Cell Stem Cell 1:313–323
Atsumi N, Ishii G, Kojima M, Sanada M, Fujii S, Ochiai A (2008) Podoplanin, a novel marker of tumor-initiating cells in human squamous cell carcinoma A431. Biochem Biophys Res Commun 373:36–41
Eramo A, Lotti F, Sette G et al (2008) Identification and expansion of the tumorigenic lung cancer stem cell population. Cell Death Differ 15:504–514
Schatton T, Murphy GF, Frank NY et al (2008) Identification of cells initiating human melanomas. Nature 451:345–349
Takaishi S, Okumura T, Tu S et al (2009) Identification of gastric cancer stem cells using the cell surface marker CD44. Stem Cells 27:1006–1020
Curley MD, Therrien VA, Cummings CL et al (2009) CD133 expression defines a tumor initiating cell population in primary human ovarian cancer. Stem Cells 27:2875–2883
Zhang S, Balch C, Chan MW et al (2008) Identification and characterization of ovarian cancer-initiating cells from primary human tumors. Cancer Res 68:4311–4320
Alvero AB, Chen R, Fu HH et al (2009) Molecular phenotyping of human ovarian cancer stem cells unravels the mechanisms for repair and chemoresistance. Cell Cycle 8:158–166
Weigmann A, Corbeil D, Hellwig A, Huttner WB (1997) Prominin, a novel microvilli-specific polytopic membrane protein of the apical surface of epithelial cells, is targeted to plasmalemmal protrusions of non-epithelial cells. Proc Natl Acad Sci USA 94:12425–12430
Yin AH, Miraglia S, Zanjani ED et al (1997) AC133, a novel marker for human hematopoietic stem and progenitor cells. Blood 90:5002–5012
Corbeil D, Roper K, Hellwig A et al (2000) The human AC133 hematopoietic stem cell antigen is also expressed in epithelial cells and targeted to plasma membrane protrusions. J Biol Chem 275:5512–5520
Maw MA, Corbeil D, Koch J et al (2000) A frameshift mutation in prominin (mouse)-like 1 causes human retinal degeneration. Hum Mol Genet 9:27–34
Bourseau-Guilmain E, Griveau A, Benoit JP, Garcion E (2011) The importance of the stem cell marker prominin-1/CD133 in the uptake of transferrin and in iron metabolism in human colon cancer Caco-2 cells. PLoS One 6:e25515
Du L, Wang H, He L et al (2008) CD44 is of functional importance for colorectal cancer stem cells. Clin Cancer Res 14:6751–6760
Goodison S, Urquidi V, Tarin D (1999) CD44 cell adhesion molecules. Mol Pathol 52:189–196
Ahrens T, Assmann V, Fieber C et al (2001) CD44 is the principal mediator of hyaluronic-acid-induced melanoma cell proliferation. J Invest Dermatol 116:93–101
Misra S, Toole BP, Ghatak S (2006) Hyaluronan constitutively regulates activation of multiple receptor tyrosine kinases in epithelial and carcinoma cells. J Biol Chem 281:34936–34941
Sheridan C, Kishimoto H, Fuchs RK et al (2006) CD44+/CD24- breast cancer cells exhibit enhanced invasive properties: an early step necessary for metastasis. Breast Cancer Res 8:R59
Omara-Opyene AL, Qiu J, Shah GV, Iczkowski KA (2004) Prostate cancer invasion is influenced more by expression of a CD44 isoform including variant 9 than by Muc18. Lab Invest 84:894–907
Zhang T, Huang XH, Dong L et al (2010) PCBP-1 regulates alternative splicing of the CD44 gene and inhibits invasion in human hepatoma cell line HepG2 cells. Mol Cancer 9:72
Li Y, Heldin P (2001) Hyaluronan production increases the malignant properties of mesothelioma cells. Br J Cancer 85:600–607
Paradis V, Eschwege P, Loric S et al (1998) De novo expression of CD44 in prostate carcinoma is correlated with systemic dissemination of prostate cancer. J Clin Pathol 51:798–802
Orian-Rousseau V (2010) CD44, a therapeutic target for metastasising tumours. Eur J Cancer 46:1271–1277
Park HY, Lee KJ, Lee SJ, Yoon MY Screening of Peptides Bound to Breast Cancer Stem Cell Specific Surface Marker CD44 by Phage Display. Mol Biotechnol 51:212–220
Joshua B, Kaplan MJ, Doweck I et al Frequency of cells expressing CD44, a Head and Neck cancer stem cell marker: correlation with tumor aggressiveness. Head Neck 34:42–49
Colnot DR, Roos JC, de Bree R et al (2003) Safety, biodistribution, pharmacokinetics, and immunogenicity of 99mTc-labeled humanized monoclonal antibody BIWA 4 (bivatuzumab) in patients with squamous cell carcinoma of the head and neck. Cancer Immunol Immunother 52:576–582
Borjesson PK, Postema EJ, Roos JC et al (2003) Phase I therapy study with (186)Re-labeled humanized monoclonal antibody BIWA 4 (bivatuzumab) in patients with head and neck squamous cell carcinoma. Clin Cancer Res 9(39):61 S–72 S
Tijink BM, Buter J, de Bree R et al (2006) A phase I dose escalation study with anti-CD44v6 bivatuzumab mertansine in patients with incurable squamous cell carcinoma of the head and neck or esophagus. Clin Cancer Res 12:6064–6072
Jin L, Hope KJ, Zhai Q, Smadja-Joffe F, Dick JE (2006) Targeting of CD44 eradicates human acute myeloid leukemic stem cells. Nat Med 12:1167–1174
Rocco A, Liguori E, Pirozzi G et al (2011) CD133 and CD44 cell surface markers do not identify cancer stem cells in primary human gastric tumours. J Cell Physiol 227:2686–2693
Eppert K, Takenaka K, Lechman ER et al (2011) Stem cell gene expression programs influence clinical outcome in human leukemia. Nat Med 17:1086–1093
Park SY, Lee HE, Li H, Shipitsin M, Gelman R, Polyak K (2010) Heterogeneity for stem cell-related markers according to tumor subtype and histologic stage in breast cancer. Clin Cancer Res 16:876–887
Ali HR, Dawson SJ, Blows FM, Provenzano E, Pharoah PD, Caldas C (2011) Cancer stem cell markers in breast cancer: pathological, clinical and prognostic significance. Breast Cancer Res 13:R118
Allalunis-Turner MJ, Siemann DW (1986) Recovery of cell subpopulations from human tumour xenografts following dissociation with different enzymes. Br J Cancer 54:615–622
Abuzakouk M, Feighery C, O’Farrelly C (1996) Collagenase and Dispase enzymes disrupt lymphocyte surface molecules. J Immunol Methods 194:211–216
Reynolds BA, Weiss S (1992) Generation of neurons and astrocytes from isolated cells of the adult mammalian central nervous system. Science 255:1707–1710
Yu S, Zhang JZ, Zhao CL, Zhang HY, Xu Q (2004) Isolation and characterization of the CD133+ precursors from the ventricular zone of human fetal brain by magnetic affinity cell sorting. Biotechnol Lett 26:1131–1136
Ponti D, Costa A, Zaffaroni N et al (2005) Isolation and in vitro propagation of tumorigenic breast cancer cells with stem/progenitor cell properties. Cancer Res 65:5506–5511
Fang D, Nguyen TK, Leishear K et al (2005) A tumorigenic subpopulation with stem cell properties in melanomas. Cancer Res 65:9328–9337
Gou S, Liu T, Wang C et al (2007) Establishment of clonal colony-forming assay for propagation of pancreatic cancer cells with stem cell properties. Pancreas 34:429–35
Duhagon MA, Hurt EM, Sotelo-Silveira JR, Zhang X, Farrar WL (2010)Genomic profiling of tumor initiating prostatospheres. BMC Genomics 11:324
Todaro M, Alea MP, Di Stefano AB et al (2007) Colon cancer stem cells dictate tumor growth and resist cell death by production of interleukin-4. Cell Stem Cell 1:389–402
Yuan X, Curtin J, Xiong Y et al (2004) Isolation of cancer stem cells from adult glioblastoma multiforme. Oncogene 23:9392–9400
Galli R, Binda E, Orfanelli U et al (2004) Isolation and characterization of tumorigenic, stem-like neural precursors from human glioblastoma. Cancer Res 64:7011–7021
Gibbs CP, Kukekov VG, Reith JD et al (2005) Stem-like cells in bone sarcomas: implications for tumorigenesis. Neoplasia 7:967–976
Suslov ON, Kukekov VG, Ignatova TN, Steindler DA (2002) Neural stem cell heterogeneity demonstrated by molecular phenotyping of clonal neurospheres. Proc Natl Acad Sci USA 99:14506–14511
Patrawala L, Calhoun T, Schneider-Broussard R et al (2006) Highly purified CD44+ prostate cancer cells from xenograft human tumors are enriched in tumorigenic and metastatic progenitor cells. Oncogene 25:1696–1708
Jensen JB, Parmar M (2006) Strengths and limitations of the neurosphere culture system. Mol Neurobiol 34:153–161
Sladek NE (2003) Human aldehyde dehydrogenases: potential pathological, pharmacological, and toxicological impact. J Biochem Mol Toxicol 17:7–23
Moreb JS (2008) Aldehyde dehydrogenase as a marker for stem cells. Curr Stem Cell Res Ther 3:237–246
Storms RW, Trujillo AP, Springer JB et al (1999) Isolation of primitive human hematopoietic progenitors on the basis of aldehyde dehydrogenase activity. Proc Natl Acad Sci USA 96:9118–9123
Alison MR, Guppy NJ, Lim SM, Nicholson LJ (2010)Finding cancer stem cells: are aldehyde dehydrogenases fit for purpose? J Pathol 222:335–344
Pearce DJ, Taussig D, Simpson C et al (2005) Characterization of cells with a high aldehyde dehydrogenase activity from cord blood and acute myeloid leukemia samples. Stem Cells 23:752–760
Ginestier C, Hur MH, Charafe-Jauffret E et al (2007) ALDH1 is a marker of normal and malignant human mammary stem cells and a predictor of poor clinical outcome. Cell Stem Cell 1:555–567
Boonyaratanakornkit JB, Yue L, Strachan LR et al (2010) Selection of tumorigenic melanoma cells using ALDH. J Invest Dermatol 130:2799–2808
van den Hoogen C, van der Horst G, Cheung H et al (2010) High aldehyde dehydrogenase activity identifies tumor-initiating and metastasis-initiating cells in human prostate cancer. Cancer Res 70:5163–5173
Silva IA, Bai S, McLean K et al (2011) Aldehyde dehydrogenase in combination with CD133 defines angiogenic ovarian cancer stem cells that portend poor patient survival. Cancer Res 71:3991–4001
Liang D, Shi Y (2012) Aldehyde dehydrogenase-1 is a specific marker for stem cells in human lung adenocarcinoma. Med Oncol 29:633–639
Wang L, Park P, Zhang H, La Marca F, Lin CY (2011) Prospective identification of tumorigenic osteosarcoma cancer stem cells in OS99-1 cells based on high aldehyde dehydrogenase activity. Int J Cancer 128:294–303
Hwang-Verslues WW, Kuo WH, Chang PH et al (2009) Multiple lineages of human breast cancer stem/progenitor cells identified by profiling with stem cell markers. PLoS One 4:e8377
Yu C, Yao Z, Dai J et al (2011) ALDH activity indicates increased tumorigenic cells, but not cancer stem cells, in prostate cancer cell lines. In Vivo 25:69–76
Zhou J, Wang H, Cannon V, Wolcott KM, Song H, Yates C (2011) Side population rather than CD133(+) cells distinguishes enriched tumorigenicity in hTERT-immortalized primary prostate cancer cells. Mol Cancer 10:112
Camargo FD, Chambers SM, Drew E, McNagny KM, Goodell MA (2006) Hematopoietic stem cells do not engraft with absolute efficiencies. Blood 107:501–507
Grotenhuis BA, Wijnhoven BP, van Lanschot JJ (2012)Cancer stem cells and their potential implications for the treatment of solid tumors. J Surg Oncol doi: 10.1002/jso.23069
Jones RJ (2009) Cancer stem cells-clinical relevance. J Mol Med (Berl) 87:1105–1110
Zeppernick F, Ahmadi R, Campos B et al (2008) Stem Cell Marker CD133 affects clinical outcome in glioma patients. Clin Cancer Res 14:123–129
Liu R, Wang X, Chen GY et al (2007) The prognostic role of a gene signature from tumorigenic breast-cancer cells. N Engl J Med 356:217–226
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2013 Springer Science+Business Media Dordrecht
About this chapter
Cite this chapter
Gao, C., Hollingsworth, R., Hurt, E. (2013). Introduction to Cancer Stem Cells. In: Mathews, L., Cabarcas, S., Hurt, E. (eds) DNA Repair of Cancer Stem Cells. Springer, Dordrecht. https://doi.org/10.1007/978-94-007-4590-2_1
Download citation
DOI: https://doi.org/10.1007/978-94-007-4590-2_1
Published:
Publisher Name: Springer, Dordrecht
Print ISBN: 978-94-007-4589-6
Online ISBN: 978-94-007-4590-2
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)