Abstract
Abstract
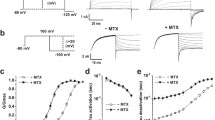
In the presence of oxygen (O2), carbon monoxide (CO) is synthesised from heme by endogenous hemeoxygenases, and is a powerful activator of BKCa channels. This transduction pathway has been proposed to contribute to cellular O2 sensing in rat carotid body. In the present study we have explored the role that four cysteine residues (C820, C911, C995 and C1028), located in the vicinity of the “calcium bowl” of C-terminal of human BKCa-αsubunit, have on channel CO sensitivity. Mutant BKCa-αsubunits were generated by site-directed mutagenesis (single, double and triple cysteine residue substitutions with glycine residues) and were transiently transfected into HEK 293 cells before subsequent analysis in inside-out membrane patches. Potassium cyanide (KCN) completely abolished activation of wild type BKCa channels by the CO donor, tricarbonyldichlororuthenium (II) dimer, at 100μM. In the absence of KCN the CO donor increased wild-type channel activity in a concentration-dependent manner, with an EC50 of ca. 50μM. Single cysteine point mutations of residues C820, C995 and C1028 affected neither channel characteristics nor CO EC50 values. In contrast, the CO sensitivity of the C911G mutation was significantly decreased (EC50 ca. 100 M). Furthermore, all double and triple mutants which contained the C911G substitution exhibited reduced CO sensitivity, whilst those which did not contain this mutation displayed essentially unaltered CO EC50 values. These data highlight that a single cysteine residue is crucial to the activation of BKCa by CO. We suggest that CO may bind to this channel subunit in a manner similar to the transition metal-dependent co-ordination which is characteristic of several enzymes, such as CO dehydrogenase.
Access provided by Autonomous University of Puebla. Download to read the full chapter text
Chapter PDF
Similar content being viewed by others
References
Bennett B, Lemon BJ, & Peters JW (2000). Reversible carbon monoxide binding and inhibition at the active site of the Fe-only hydrogenase. Biochemistry 39, 7455–7460.
Drennan CL, Heo J, Sintchak MD, Schreiter E, & Ludden PW (2001). Life on carbon monoxide: X-ray structure of Rhodospirillum rubrum Ni-Fe-S carbon monoxide dehydrogenase. Proc Natl Acad Sci USA 98, 11973–11978.
Ha SW, Korbas M, Klepsch M, Meyer-Klaucke W, Meyer O, & Svetlitchnyi V (2007). Interaction of potassium cyanide with the Ni-4Fe-5S] active site cluster of CO dehydrogenase from Carboxydothermus hydrogenoformans. J Biol Chem 282, 10639–10646.
Hou S, Xu R, Heinemann SH, & Hoshi T (2008). The RCK1 high-affinity Ca2+ sensor confers carbon monoxide sensitivity to Slo1 BK channels. Proc Natl Acad Sci USA 105, 4039–4043.
Jaggar JH, Li A, Parfenova H, Liu J, Umstot ES, Dopico AM, & Leffler CW (2005). Heme is a carbon monoxide receptor for large-conductance Ca2+-activated K+ channels. Circ Res 97, 805–812.
Jeoung JH & Dobbek H (2007). Carbon dioxide activation at the Ni,Fe-cluster of anaerobic carbon monoxide dehydrogenase. Science 318, 1461–1464.
Kemp PJ & Peers C (2007). Oxygen sensing by ion channels. Essays Biochem 43, 77–90.
Lemon BJ & Peters JW (1999). Binding of exogenously added carbon monoxide at the active site of the iron-only hydrogenase (CpI) from Clostridium pasteurianum. Biochemistry 38, 12969–12973.
Maines MD, Trakshel GM, & Kutty RK (1986). Characterization of two constitutive forms of rat liver microsomal heme oxygenase. Only one molecular species of the enzyme is inducible. J Biol Chem 261, 411–419.
Mann BE & Motterlini R (2007). CO and NO in medicine. Chem Commun 41, 4197–4208.
McCoubrey WK, Jr., Huang TJ, & Maines MD (1997). Isolation and characterization of a cDNA from the rat brain that encodes hemoprotein heme oxygenase-3. Eur J Biochem 247, 725–732.
Moss BL & Magleby KL (2001). Gating and conductance properties of BK channels are modulated by the S9-S10 tail domain of the alpha subunit. A study of mSlo1 and mSlo3 wild-type and chimeric channels. J Gen Physiol 118, 711–734.
Riesco-Fagundo AM, Perez-Garcia MT, Gonzalez C, & Lopez-Lopez JR (2001). O2 modulates large-conductance Ca2+-dependent K+ channels of rat chemoreceptor cells by a membrane-restricted and CO-sensitive mechanism. Circ Res 89, 430–436.
Schreiber M, Wei A, Yuan A, Gaut J, Saito M, & Salkoff L (1998). Slo3, a novel pH-sensitive K+ channel from mammalian spermatocytes. J Biol Chem 273, 3509–3516.
Tenhunen R, Marver HS, & Schmid R (1968). The enzymatic conversion of heme to bilirubin by microsomal heme oxygenase. Proc Natl Acad Sci USA 61, 748–755.
Wang R & Wu L (1997). The chemical modification of KCa channels by carbon monoxide in vascular smooth muscle cells. J Biol Chem 272, 8222–8226.
Wang R, Wu L, & Wang Z (1997). The direct effect of carbon monoxide on KCa channels in vascular smooth muscle cells. Pflugers Arch 434, 285–291.
Williams SE, Brazier SP, Baban N, Telezhkin V, Muller CT, Riccardi D, & Kemp PJ (2008). A structural motif in the C-terminal tail of slo1 confers carbon monoxide sensitivity to human BKCa channels. Pflugers Arch 456, 561–572.
Williams SE, Wootton P, Mason HS, Bould J, Iles DE, Riccardi D, Peers C, & Kemp PJ (2004). Hemoxygenase-2 is an oxygen sensor for a calcium-sensitive potassium channel. Science 306, 2093–2097.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2009 Springer Science+Business Media B.V.
About this chapter
Cite this chapter
Brazier, S., Telezhkin, V., Mears, R., Müller, C., Riccardi, D., Kemp, P. (2009). Cysteine Residues in the C-terminal Tail of the Human BKCaα Subunit Are Important for Channel Sensitivity to Carbon Monoxide. In: Gonzalez, C., Nurse, C.A., Peers, C. (eds) Arterial Chemoreceptors. Advances in Experimental Medicine and Biology, vol 648. Springer, Dordrecht. https://doi.org/10.1007/978-90-481-2259-2_5
Download citation
DOI: https://doi.org/10.1007/978-90-481-2259-2_5
Publisher Name: Springer, Dordrecht
Print ISBN: 978-90-481-2258-5
Online ISBN: 978-90-481-2259-2
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)