Abstract
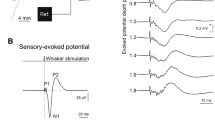
In the present study, electrophysiological mapping methods were used to estimate the size of the tail representation area of the primary somatosensory cortex (SI) of the rat. Using a half-maximal evoked potential method and multiunit recording method, we estimated that the SI tail area was 0.51 and 0.78 mm2, respectively. A dissector method was used to estimate the neuronal densities. There was, on average, 84 829 neurons/mm3 and 117 750 neurons under 1 mm2 of cortical area in the tail area of the SI. Therefore, there are about 94 000 neurons in the estimated 0.8 mm2 of the SI that are involved in processing sensory signals from the tail. Anteroposteriorly oriented, evenly spaced 16-channel microwires were chronically implanted in the frontoparietooccipital cortex that was centered on the SI. Thereafter, evoked field potentials were used to estimate the change in the size of the tail area with two modalities—pain and touch—under two states: anesthetized and conscious. No significant difference was found between the size of the tail area when tactile and noxious stimulations were used. However, the number of tail responsive channels showed a significant increase when the rat was awake and behaving.
Access provided by Autonomous University of Puebla. Download to read the full chapter text
Chapter PDF
Similar content being viewed by others
References
D’Amour FE, Smith DL (1941) A method for determining loss of pain sensation. J Pharmacol Exp Ther 72:74–79.
Hebel R, Stromberg MW (1986) Anatomy and embryology of the laboratory rat. In: Nervous system. Biomed Verlag, Worthsee, Germany, p 125.
Jaw FS, Yen CT, Tsao HW, et al (1991) A modified “triangular pulse” stimulator for C fiber stimulation. J Neurosci Methods 37:169–172
Chapin JK, Lin CS (1984) Mapping the body representation with SI cortex of anesthetized and awake rats. J Comp Neurol 229:199–213.
Mitchell D, Hellon RF (1977) Neuronal and behavioral responses in rats during noxious stimulation of the tail. Proc R Soc Lond B 197:169–194.
Welker C (1971) Microelectrode delineation of fine grain somatotopic organization of SmI cerebral neocortex in albino rat. Brain Res 26:259–275.
Woolsey CN (1958) Organization of somatic sensory and motor areas of the cerebral cortex. In: Harlow HF, Woolsey CN (eds) Biological and biochemical bases of behavior. University of Wisconsin Press, Madison, WI, pp 63–82.
Coggeshall RE (1992) A consideration of neural counting methods. Trends Neurosci 15: 9–13.
Swanson LW (1992) Brain maps: structure of the rat brain. Elsevier, Amsterdam.
Paxinos G, Watson C (2007) The rat brain in stereotaxic coordinates (6th ed). Academic, London.
Harding AJ, Halliday GM, Cullen K (1994) Practical considerations for the use of the optical dissector in estimating neuronal. J Neurosci Methods 51:83–89.
Pakkenberg B, Gundersen HJG (1989) New stereological method for obtaining unbiased efficient estimates of total nerve cell number in human brain areas. APMIS 97:677–681.
Tsai ML, Yen CT (2003) A simple method for fabricating horizontal and vertical microwire arrays. J Neurosci Methods 131:107–110.
Yen CT, Huang CH, Fu SE (1994) Surface temperature change, cortical evoked potential and pain behavior elicited by CO2 lasers. Chin J Physiol 37:193–199.
Katz LC, Shatz CJ (1996) Synaptic activity and the construction of cortical circuits. Science 274:1133–1138.
Kaas JH (1997) Topographic maps are fundamental to sensory processing. Brain Res Bull 44:107–112.
Willshaw DJ, von der Malsburg C (1976) How patterned neural connections can be set up by self-organization. Proc R Soc Lond B 194:431–445.
Van Essen DC (2002) Surface-based atlases of cerebellar cortex in the human, macaque, and mouse. Ann N Y Acad Sci 978:468–479.
Oakley B, Schafer R (1978) Experimental neurobiology, a laboratory manual. University of Michigan Press, Ann Arbor, p 121.
Beaulieu C, Colonnier M (1983) The number of neurons in the different laminae of the binocular and monocular regions of area 17 in the cat. J Comp Neurol 217:337–344.
Beaulieu C, Colonnier M (1989) Number of neurons in individual laminae of areas 3B, 4γ, and 6aα of the cat cerebral cortex: a comparison with major visual areas. J Comp Neurol 279:228–234
Beaulieu C (1993) Numerical date on neocortical neurons in adult rat, with special reference to the GABA population. Brain Res 609:284–292.
Ren JQ, Alika Y, Heizmann CW, et al (1992) Quantitative analysis of neurons and glial cells in the rat somatosensory cortex, with special reference to GABAergic neurons and parvalbumin-containing neurons. Exp Brain Res 92:1–14.
Schuz A, Palm G (1989) Density of neurons and synapses in the cerebral cortex of the mouse. J Comp Neurol 286:442–455.
Shouenborg J, Kalliomake J, Gustavsson P, et al (1986) Field potentials evoked in the rat somatosensory cortex by impulses in cutaneous Aβ-and C-fibers. Brain Res 397:86–92.
Shaw FZ, Chen RF, Tsao HW, et al (1999) Comparison of touch-and laser heat-evoked cortical field potentials in conscious rats. Brain Res 824:183–196.
Sun JJ, Yang JW, Shyu BC (2006) Current source density analysis of laser heat-evoked intra-cortical field potentials in the primary somatosensory cortex of rats. Neuroscience 140:1321–1136.
Merzenich MM, Nelson RJ, Stryker MP, et al (1984) Somatosensory cortical map changes following digit amputation in adult monkeys. J Comp Neurol 224:591–605.
Faggin BM, Nguyen KT, Nicolelis MAL (1997) Immediate and simultaneous sensory reorganization at cortical and subcortical levels of the somatosensory system. Proc Natl Acad Sci U S A 94:9428–9433.
Shaw FZ, Chen RF, Yen CT (2001) Dynamic changes of touch-and laser heat-evoked field potentials of primary somatosensory cortex in awake and pentobarbital-anesthetized rats. Brain Res 911:105–115.
Tsai ML, Kuo CC, Sun WZ, et al (2004) Differential morphine effects on short-and longlatency laser-evoked cortical responses in the rat. Pain 110:665–674.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2008 Springer
About this chapter
Cite this chapter
Yen, CT., Chen, RS. (2008). Tail Region of the Primary Somatosensory Cortex and Its Relation to Pain Function. In: Onozuka, M., Yen, CT. (eds) Novel Trends in Brain Science. Springer, Tokyo. https://doi.org/10.1007/978-4-431-73242-6_14
Download citation
DOI: https://doi.org/10.1007/978-4-431-73242-6_14
Publisher Name: Springer, Tokyo
Print ISBN: 978-4-431-73241-9
Online ISBN: 978-4-431-73242-6
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)