Abstract
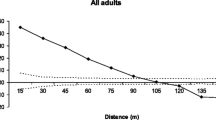
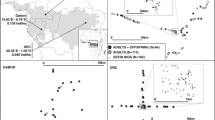
In order to evaluate the consequences of forest fragmentation on populations of Magnolia obovata, we compared genetic diversity and reproductive characteristics at two nearby sites, one conserved and one fragmented. The genetic diversity between adults trees of the different sites was not significantly different. However, saplings in the conserved site showed a significantly higher genetic diversity than both adult trees in the conserved site and saplings in the fragmented sites; this was found to be the result of the larger gene flow into the conserved site. The density of the adult trees was significantly related to all of the reproductive traits analyzed (fertilization of ovules, insect attack to seeds, ovules that developed into seeds and outcrossing at the stage of seeds) at both sites. At both sites, fertilization of ovules and insect attack on seeds were positively correlated to adult tree density while outcrossing rate was negatively correlated to adult tree density. The fertilization of ovules and outcrossing were more dependent on adult tree density in the fragmented site than in the conserved site. The probability of ovules developing into outcrossed seeds showed a negative correlation with adult tree density at both sites, indicating the advantage of low density for this species and possibly implying a resilience to habitat fragmentation. A two-generation-analysis did not identify significant differences between sites in terms of the structure of the pollen pool and the number of pollen donors. Although fragmentation affected reproductive characteristics, the effect on seedling establishment and subsequent survival remains to be determined. Proposals for future studies that will assist in the development of management strategies for forests suffering fragmentation are made.
Access provided by Autonomous University of Puebla. Download to read the full chapter text
Chapter PDF
Similar content being viewed by others
References
Aizen MA, Feinsinger P (1994a) Forest fragmentation, pollination, and plant reproduction in a Chaco dry forest, Argentina. Ecology 75:330–351
Aizen MA, Feinsinger P (1994b) Habitat fragmentation, native insect pollinators, and feral honey bees in Argentine “Chaco Serrano”. Ecol Appl 4:378–39
Aldrich PR, Hamrick JL (1998) Reproductive dominance of pasture trees in a fragmented tropical forest mosaic. Science 281:103–105
Aldrich PR, Hamrick JL, Chavarriaga P, Kochert G (1998) Microsatellite analysis of demographic genetic structure in fragmented populations of the tropical tree Symphonia globulifera. Mol Ecol 7:933–944
Austerlitz F, Smouse PE (2001) Two-generation analysis of pollen flow across a landscape. II. Relation between ϕft, pollen dispersal, and inter-female distance. Genetics 157:851–857
Benitez-Malvido J (1998) Impact of forest fragmentation on seedling abundance in a tropical rain forest. Conserv Biol 12:380–389
Benitez-Malvido J, Martínez-Ramos M (2003) Impact of forest fragmentation on understorey plant species richness in Amazonia. Conserv Biol 17:389–400
Cascante A, Quesada M, Lobo JA, Fuchs EJ (2002) Effects of dry tropical forest fragmentation on the reproductive success and genetic structure of the tree Samanea saman. Conserv Biol 16:137–147
Collevatti RG, Grattapaglia D, Hay JD (2001) Population genetic structure of the endangered tropical tree species Caryocar brasiliense, based on variability at microsatellite loci. Mol Ecol 10:349–356
Cunningham SA (2000) Depressed pollination in habitat fragments causes low fruit set. Proc R Soc Lond B Biol 267:1149–1152
Dayanandan S, Dole J, Bawa K, Kesseli R (1999) Population structure delineated with microsatellite markers in fragmented populations of a tropical tree, Carapa guianensis (Meliaceae). Mol Ecol 8:1585–1592
Dick CW (2001) Genetic rescue of remnant tropical trees by an alien pollinator. Proc R Soc Lond B Biol 268:2391–2396
Dick CW, Etchelecu G, Austerlitz F (2003) Pollen dispersal of tropical trees (Dinizia excelsa: Fabaceae) by native insects and African honeybees in pristine and fragmented Amazonian rain forest. Mol Ecol 12:753–764
Duncan DH, Nicotra AB, Wood JT, Cunningham SA (2004) Plant isolation reduces outcross pollen receipt in a partially self-compatible herb. J Ecol 92:977–985
Ellstrand NC, Marshall DL (1985) Interpopulation gene flow by pollen in wild radish, Raphanus sativus. Am Nat 126:606–616
Ellstrand NC, Devlin B, Marshall DL (1989) Gene flow by pollen into small populations: data from experimental and natural stands of wild radish. Proc Nat Acad Sci USA 86:9044–9047
Fenster CB (1991) Gene flow in Chamaecrista fasciculata (Leguminosae) I. Gene dispersal. Evolution 45:398–409
Fuchs EJ, Lobo JA, Quesada M (2003) Effects of forest fragmentation and flowering phenology on the reproductive success and mating patterns of the tropical dry forest tree Pachira quinata. Conserv Biol 17:149–157
Godt MJW, Hamrick JL (1993) Patterns and levels of pollen mediated gene flow in Lathyrus latifolius. Evolution 47:98–110
Goudet J (1995) FSTAT version 1.2: a computer program to calculate F statistics. J Hered 86:485–486
Ghazoul J, Liston KA, Boyle TJB (1998) Disturbance-induced density-dependent seed set in Shorea siamensis (Dipterocarpaceae), a tropical forest tree. J Ecol 86:462–473
Handel SN (1983) Pollination ecology, plant population structure, and gene flow. In: Real L (ed) Pollination biology. Academic, New York, pp 163–211
Hedrick PW (2005) Genetics of populations, 3rd edn. Jones and Bartlett, Boston
Hobbs RJ, Yates CJ (2003) Impacts of ecosystem fragmentation on plant populations: generalising the idiosyncratic. Aust J Bot 51:471–488
Isagi Y, T Kanazashi W Suzuki H Tanaka T Abe (1999) Polymorphic DNA markers for Magnolia obovata Thunb. and their utility in related species. Mol Ecol 8:698–700
Isagi Y, Kanazashi T, Suzuki W, Tanaka H, Abe T (2000) Microsatellite analysis on regeneration process of Magnolia obovata. Heredity 84:143–151
Isagi Y, Kanazashi T, Suzuki W, Tanaka H, Abe T (2004) Highly variable pollination patterns in Magnolia obovata revealed by microsatellite paternity analysis. Int J Plant Sci 165:1047–1053
Ishida K, Yoshimaru H, Ito H (2003) Effects of geitonogamy on the seed set of Magnolia obovata Thunb. (Magnoliaceae). Int J Plant Sci 164:729–735
Judd WS, Campbell CS, Kellogg EA, Stevens PF, Donoghue MJ (2002) Plant systematics: a phylogenetic approach, 2nd edn. Sinauer Assoc, Sunderland
Kameyama Y, Isagi Y, Naito K, Nakagoshi N (2000) Microsatellite analysis of pollen flow in Rhododendron metternichii var. hondoense. Ecol Res 15:263–269
Kameyama Y, Isagi Y, Nakagoshi N (2001) Patterns and levels of gene flow in Rhododendron metternichii var. hondoense revealed by microsatellite analysis. Mol Ecol 10:205–216
Kearns CA, Inouye DW, Waser NM (1998) Endangered mutualisms: the conservation of plant-pollinator interactions. Annu Rev Ecol Syst 29:83–112
Kikuzawa K, Mizui N (1990) Flowering and fruiting phenology of Magnolia hypoleuca. Plant Species Biol 5:255–261
Lowe AJ, Boshier D, Ward M, Bacles CFE, Navarro C (2005) Genetic resource impacts of habitat loss and degradation; reconciling empirical evidence and predicted theory for neotropical trees. Heredity 95:255–273
Masaki T, Kominami Y, Nakashizuka T (1994) Spatial and seasonal patterns of seed dissemination of Cornus controversa in a temperate forest. Ecology 75:1903–1910
Moriguchi Y, Morishita T, Ohtani Y (2002) Climate in Ogawa Forest Reserve. In: Makashizuka T, Matsumoto Y (eds) Diversity and interaction in a temperate forest community. Springer, Tokyo, pp 11–18
Nakashizuka T, Iida S, Tanaka H, Shibata M, Abe S, Masaki T, Niiyama K (1992) Community dynamics of Ogawa Forest Reserve, a species-rich deciduous forest, central Japan. Vegetatio 103:105–112
Obayashi K, Tsumura Y, Ihara-Ujino T, Niiyama K, Tanouchi H, Suyama Y, Washitani I, Lee C, Lee SL, Muhammad N (2002) Genetic diversity and outcrossing rate between undisturbed and selectively logged forests of Shorea curtisii (Dipterocarpaceae) using microsatellite data analysis. Int J Plant Sci 163:151–158
Oostermeijer JGB, Luijten SH, Den Nijs JCM (2003) Integrating demographic and genetic approaches in plant conservation. Biol Conserv 113:389–398
Quesada M, Stoner KE, Rosas-Guerrero V, Palacios-Guevara C, Lobo JA (2003) Effects of habitat disruption on the activity of nectarivorous bats in a dry forest: implications for the reproductive success of the Neotropical tree Ceiba grandiflora. Oecologia 135:400–406
Quesada M, Stoner KE, Lobo JA, Herrerías-Diego Y, Palacios-Guevara C, Munguía-Rosas MA, O-Salazar KA, Rosas-Guerrero V (2004) Effects of forest fragmentation on pollinator activity and consequences for plant reproductive success and mating patterns in bat-pollinated bombacaceoos trees. Biotropica 36:131–13
R Development Core Team (2005) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria
Schnabel A, Hamrick JL (1995) Understanding the population genetic structure of Gleditsia triacanthos L. the scale and pattern of pollen gene flow. Evolution 49:921–931
Shibata M, Nakashizuka T (1995) Seed and seedling demography of four co-occurring Carpinus species in a temperate deciduous forest. Ecology 76:1099–1108
Slatkin M (1985) Gene flow in natural populations. Annu Rev of Ecol Syst 16:393–430
Smouse PE, Dyer RJ, Westfall RD, Sork VL (2001) Two-generation analysis of pollen flow across a landscape. I. Male gamete heterogeneity among females. Evolution 55:260–271
Sork VL, Smouse PE, Apsit VJ, Dyer RJ, Westfall RD (2005) A two-generation analysis of pollen pool genetic structure in flowering dogwood, Cornis florida (Cornaceae), in the Missouri Ozarks. Am J Bot 92:262–271
Streiff R, Ducousso A, Lexer C, Steinkellner H, Gloessl J, Kremer A (1999) Pollen dispersal inferred from paternity analysis in a mixed oak stand of Quercus robur L. and Q. petraea (Matt.) Liebl. Mol Ecol 8:831–841
Suzuki W (2002) Forest vegetation in and around Ogawa Forest Reserve in relation to human impact. In: Makashizuka T, Matsumoto Y (eds) Diversity and interaction in a temperate forest community. Springer, Tokyo, pp 25–41
Tanaka H (1995) Seed demography of three co-occurring Acer species in a Japanese temperate deciduous forest. J Veg Sci 6:887–896
Tanaka H, Yahara T (1988) The pollination of Magnolia obovata. In: Kawano S (eds) The world of plants, vol. 2. Kenkyusya, Tokyo, p 37
Thien LB (1974) Floral biology of Magnolia. Am J Bot 61:1037–1045
Tomimatsu H (2005) How do plant populations respond to habitat fragmentation (in Japanese)? Jpn J Conserv Ecol 10:163–171
White GM, Boshier DH, Powell W (1999) Genetic variation within a fragmented population of Swietenia humilis Zucc. Mol Ecol 8:1899–1909
White GM, Boshier DH, Powell W (2002) Increased pollen flow counteracts fragmentation in a tropical dry forest: an example from Swietenia humilis Zuccarini. Proc Nat Acad Sci USA 99:2038–2042
Wright S (1951) The genetical structure of populations. Ann Eugen 15:323–354
Young A, Boyle T, Brown AHD (1996) The population genetic consequences of habitat fragmentation for plants. Trends Ecol Evol 11:413–418
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2007 The Ecological Society of Japan
About this chapter
Cite this chapter
Isagi, Y., Tateno, R., Matsuki, Y., Hirao, A., Watanabe, S., Shibata, M. (2007). Genetic and reproductive consequences of forest fragmentation for populations of Magnolia obovata . In: Nakashizuka, T. (eds) Sustainability and Diversity of Forest Ecosystems. Springer, Tokyo. https://doi.org/10.1007/978-4-431-73238-9_3
Download citation
DOI: https://doi.org/10.1007/978-4-431-73238-9_3
Received:
Accepted:
Published:
Publisher Name: Springer, Tokyo
Print ISBN: 978-4-431-73237-2
Online ISBN: 978-4-431-73238-9
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)