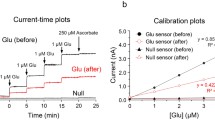
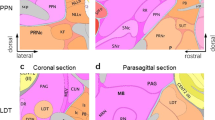
Abstract
Acetylcholine (ACh) is one of the predominant neurotransmitters in the brain. The majority of cholinergic cells are found in the medial septal nucleus, the basal forebrain, the striatum, and the brainstem (Struble et al. 1986). The major projection areas include cortex, hippocampus, striatum, substantia nigra, and medial habenula (Butcher 1995; Woolf et al. 1984). ACh is involved in the regulation of cortical arousal (Semba 1991), attention (Murphy and Sillito 1991), and sleep-wake cycles (Hobson 1990). Thus, the actions of ACh are manifold. The actions of ACh are mediated by two different types of receptors: the ionotropic nicotinic type and the metabotropic muscarinic type. Each of these classes of ACh receptor (AChR) has multiple subtypes with unique structural and functional characteristics, and thus ACh released from a nerve terminal may contribute to a wide variety of brain functions by activating different intracellular pathways depending on the distribution of the receptor types.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Abdulla FA, Calaminici M-R, Stephenson JD, et al (1993) Chronic treatment with cholinoceptor drugs influences spatial learning in rats. Psychopharmacology 111:508–511
Albuquerque EX, Pereira EFR, Castro NG, et al (1995) Neuronal nicotinic receptors: function, modulation and structure. Sem Neurosci 7:91–101
Alkondon M, Pereira EFR, Albuquerque EX (1996a) Mapping the location of functional nicotinic and γ-aminobutyric acid A receptors on hippocampal neurons. J Pharmacol Exp Ther 279:1491–1506
Alkondon M, Pereira EFR, Albuquerque EX (1996b) Characterization of nicotinic acetylcholine receptors in CA1 neurons of rat hippocampal slices. Soc Neurosci Abstr 22:1267
Amarai DG, Kurz J (1985) An analysis of the origins of the cholinergic and noncholinergic septal projections to the hippocampal formation of the rat. J Comp Neurol 240:37–59
Arai A, Guidotti A, Costa E, et al (1996) Effect of the AMPA receptor modulator IDRA 21 on LTP in hippocampal slices. Neuroreport 7:2211–2215
Bartus RT, Dean RD, Beer B, et al (1982) The cholinergic hypothesis of geriatric memory dysfunction. Science 217:408–414
Beatty WW, Carbone CP (1980) Septal lesions, intramaze cues and spatial behavior in rats. Physiol Behav 24:675–678
Benardo LS, Prince DA (1982) Cholinergic excitation of mammalian hippocampal pyramidal cells. Brain Res 249:315–331
Benowitz NL, Porchet H, Jacob P (1989) Nicotine dependence and tolerance in man: pharmacokinetic and pharmacodynamic investigations. Prog Brain Res 79:279–287
Bliss TVP, Lomo T (1973) Long-lasting potentiation of synaptic transmission in dentate area of the anesthetized rabbit following stimulation of perforant path. J Physiol 232:331–356
Bliss TVP, Gardner-Medwin AR (1973) Long-lasting potentiation of synaptic transmission in the dentate area of the unanesthetized rabbit following stimulation of perforant path. J Physiol 232:357–374
Brenner DE, Kuku UWA, Vanbelle G, et al (1993) Relationship between cigarette smoking and Alzheimer’s disease in a population-based case-control study. Neurology 43:293–300
Butcher LL (1995) Cholinergic neurons and networks. The rat nervous system. Academic Press, New York, pp 1003–1015
Chavez NL, Crona JH, Washburn MS, et al (1997) Pharmacological characterization of recombinant human neuronal nicotinic acetylcholine receptors (α2β2, α2β4, α3β2, α3β4, α4β2, α4β4 and α7 expressed in Xenopus oocytes. J Pharmacol Exp Ther 280:346–356
Clarke PB (1993) Nicotinic receptors in mammalian brain: localization and relation to cholinergic innervation. Prog Brain Res 98:77–83
Clarke PB, Reuben M (1996) Release of [3H]-noradrenaline from rat hippocampal synaptosomes by nicotine: mediation by different nicotinic receptor subtypes from striatal [3H]-dopamine release. Br J Pharmacol 117:595–606
Clarke PB, Schwartz RD, Paul SM, et al (1985) Nicotinic binding in rat brain: autoradiographic comparison of [3H] acetylcholine, [3H]nicotine, and [125I]-α-bungarotoxin. J Neurosci 5:1307–1315
Cole AE, Nicoli RA (1984) The pharmacology of cholinergic excitatory responses in hippocampal pyramidal cells. Brain Res 305:283–290
Court JA, Perry EK (1994) CNS nicotinic receptors. Possible therapeutic targets in neurodegenerative disorders. CNS Drugs 2:216–233
Couturier S, Bertrand D, Matter J-M, et al (1990) A neuronal nicotinic acetylcholine receptor subunit (α7) is developmentally regulated and forms a homooligomeric channel blocked by α-BTX. Neuron 5:847–856
Dani JA, Heinemann S (1996) Molecular and cellular aspects of nicotine abuse. Neuron 16:905–908
Decker MW, Curzon P, Brioni JD (1995) Influence of separate and combined septal and amygdala lesions on memory, acoustic startle, anxiety, and locomotor activity in rats. Neurobiol Learn Mem 64:156–168
Delbono O, Gopalakrishnan M, Renganathan M, et al (1997) Activation of the recombinant humanα7 nicotinic acetylcholine receptor significantly raises intracellular free calcium. J Pharmacol Exp Ther 280:428–438
Donnelly RD, Xue IC, Americ SP, et al (1996) In vitro neuroprotective properties of the novel cholinergic channel activator (ChCA), ABT-418. Brain Res 719:36–44
Dunnett SB, Low WC, Iverson SD, et al (1982) Septal transplants restore maze learning in rats with fornix-fimbria lesions. Brain Res 251:348–355
Freedman R, Wetmore C, Stromberg I, et al (1993) a-Bungaro toxin binding to hippocampal interneurons: immunocytochemical characterization and effects on growth factor expression. J Neurosci 13:1965–1975
Freund TF, Antal M (1988) GABA-containing neurons in the septum control inhibitory interneurons in the hippocampus. Nature (Lond) 336:170–173
Freund RK, Jungschaffer DA, Collins AC, et al (1988) Evidence for modulation of GABAergic neurotransmission by nicotine. Brain Res 453:215–220
Fu WM, Liu JJ (1997) Regulation of acetylcholine release by presynaptic nicotinic receptors at developing neuromuscular synapses. Mol Pharmacol 51:390–398
Gähwiler BH (1988) Organotypic cultures of neural tissue. Trends Neurosci 11:484–489
Gähwiler BH, Brown DA (1985) Functional innervation of cultured hippocampal neurons by cholinergic afferents from co-cultured septal explants. Nature (Lond) 313:577–579
Gray R, Rajan AS, Radcliffe KA, et al (1996) Hippocampal synaptic transmission enhanced by low concentrations of nicotine. Nature (Lond) 383:713–716
Heinemann S, Boulter J, Connolly J, et al (1991) The nicotinic receptor genes. Clin Neuropharmacol 14:S45-S61
Hepler DJ, Wenk GL, Cribbs BL, et al (1985) Memory impairments following basal forebrain lesions. Brain Res 346:8–14
Hobson JA (1990) Sleeping and dreaming. J Neurosci 10:371–382
Jones GMM, Sahakian BJ, Levy R, et al (1992) Effects of acute subcutaneous nicotine on attention, information processing and short-term memory in Alzheimer’s disease. Psychopharmacology 108:485–494
Leanza G, Muir J, Nilsson OG, et al (1996) Selective immunolesioning of the basal forebrain cholinergic system disrupts short-term memory in rats. Eur J Neurosci 8:1535–1544
Lee PN (1994) Smoking and Alzheimer’s disease: a review of the epidemiological evidence. Neuroepidemiology 13:131–144
Leranth C, Frotscher M (1989) Organization of the septal region in the rat brain: cholinergic-GAB Aergic interconnections and the termination of hippocamposeptal fibers. J Comp Neurol 289:304–314
Levin ED (1992) Nicotine systems and cognitive function. Psychopharmacology 108:417–431
Levin ED (1993) Nicotinic involvement in cognitive function: possible therapeutic applications. Med Chem Res 2:612–627
Lewis PR, Shute CCD (1967) The cholinergic limbic system: projections to hippocampal formation, medial cortex, nuclei of the ascending cholinergic reticular system and subfornical organ and supraoptic crest. Brain 90:521–540
Lindstrom J, Schoepfer R, Conroy W, et al (1991) The nicotinic acetylcholine receptor gene family. Adv Exp Med Biol 287:255–278
Madison DV, Lancaster B, Nicoli RA (1987) Voltage clamp analysis of cholinergic action in the hippocampus. J Neurosci 7:733–741
McGehee DS, Heath MJ, Gelber S, et al (1995) Nicotine enhancement of fast excitatory synaptic transmission in CNS by presynaptic receptors. Science 269:1692–1696
McGehee DS, Role LW (1995) Physiological diversity of nicotinic acetylcholine receptors expressed by vertebrate neurons. Annu Rev Physiol 57:521–546
McGehee DS, Role LW (1996) Presynaptic ionotropic receptors. Curr Opin Neurobiol 6:342–349
Morris RGM, Anderson E, Lynch GS, et al (1986) Selective impairment of learning and blockade of long-term potentiation by an N-methyl-D-aspartate receptor antagonist, AP5. Nature (Lond) 319:774–776
Muller D, Buchs PA, Stoppini L (1993) Time course of synaptic development in hippocampal organotypic cultures. Dev Brain Res 71:93–100
Murphy PC, Sillito AM (1991) Cholinergic enhancement of direction selectivity in the visual cortex of the cat. Neuroscience 40:13–20
Newhouse PA, Potter A, Lenox RH (1993) The effects of nicotinic agents on human cognition: possible therapeutic applications in Alzheimer’s and Parkinson’s diseases. Med Chem Res 2:628–642
Nordberg A, Romanelli L, Sundwall A, et al (1989) Effect of acute and subchronic nicotine treatment on cortical acetylcholine release and on nicotinic receptors in rats and guinea-pigs. Br J Pharmacol 98:71–78
Picciotto MR, Zoli M, Rimondini R, et al (1998) Acetylcholine receptors containing the beta 2 subunit are involved in the reinforcing properties of nicotine. Nature (Lond) 391:173–177
Poincheval-Fuhrman S, Sara SJ (1993) Chronic nicotine ingestion improves radial arm mazeperformance in rats. Behav Pharmacol 4:535–539
Price DL, Koliatsos VE, Clatterbuck RC (1993) Cholinergic systems: human diseases, animal models, and prospects for therapy. Prog Brain Res 98:51–60
Reece LJ, Schwartzkroin PA (1991) Nicotine exerts the differential effects on different CAl hippocampal cell types. Brain Res 540:287–290
Ridley RM, Thomley HD, Baker HF, et al (1991) Cholinergic neural transplants into hippocampus restore learning ability in monkeys with fornix lesions. Exp Brain Res 83:533–538
Role LW, Berg DK (1996) Nicotinic receptors in the development and modulation of CNS synapses. Neuron 16:1077–1085
Rosecrans JA, Karan LD (1993) Neurobehavioral mechanisms of nicotine action: role in the initiation and maintenance of tobacco dependence. J Subst Abuse Treat 10:161–170
Sahakian B J, Coull JT (1994) Nicotine and tetrahydroaminoacridine: evidence for improved attention in patients with dementia of the Alzheimer type. Drug Dev Res 31:80–88
Sargent PB (1993) The diversity of neuronal nicotinic acetylcholine receptors. Annu Rev Neurosci 16:403–443
Seguela P, Wadiche J, Dineley MK, et al (1993) Molecular cloning, functional properties, and distribution of rat brain α7: a nicotinic cation channel highly permeable to calcium. J. Neurosci 13:596–604
Semba K (1991) The cholinergic basal forebrain: a critical role in cortical arousal. In: Napier TC (ed) The basal forebrain. Plenum Press, New York, pp 197–218
Sparks DL, Hunsaker JD, Slevin JT, et al (1992) Monoaminergic and cholinergic synaptic markers in the nucleus basalis of Meynert (nbM): normal age-related changes and the effect of heart disease and Alzheimer’s disease. Ann Neurol 31:611–620
Struble RG, Lehmann J, Mitchell SJ, et al (1986) Basal forebrain neurons provide major cholinergic innervation of primate neocortex. Neurosci Lett 66:215–220
Summers KL, Giacobini E (1995) Effects of local and repeated systemic administration of (-)-nicotine on extracellular levels of acetylcholine, norepinephrine, dopamine, and serotonin in rat cortex. Neurochem Res 20:753–759
Whiting PJ, Schoepfer R, Conroy, WG, et al (1991) Expression of nicotinic acetylcholine receptor subtypes in brain and retina. Mol Brain Res 10:61–70
Wilkie GI, Hutson P, Sullivan JP, et al (1996) Pharmacological characterization of a nicotinic autoreceptor in rat hippocampal synaptosomes. Neurochem Res 21:1141–1148
Woolf NJ, Eckenstein F, Butcher LL (1984) Cholinergic systems in the rat brain. I. Projections to the limbic telencephalon. Brain Res Bull 13:751–784
Zamani MR, Allen YS, Owen GP, et al (1997) Nicotine modulates the neurotoxic effect of β-amyloid protein (25-35) in hippocampal cultures. Neuroreport 8:513–517
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 1999 Springer Japan
About this chapter
Cite this chapter
Fujii, S., Walcott, E.C., Sumikawa, K. (1999). The Action of Nicotine in the Mammalian Brain. In: Yamamoto, I., Casida, J.E. (eds) Nicotinoid Insecticides and the Nicotinic Acetylcholine Receptor. Springer, Tokyo. https://doi.org/10.1007/978-4-431-67933-2_10
Download citation
DOI: https://doi.org/10.1007/978-4-431-67933-2_10
Publisher Name: Springer, Tokyo
Print ISBN: 978-4-431-68011-6
Online ISBN: 978-4-431-67933-2
eBook Packages: Springer Book Archive