Abstract
Alzheimer’s disease (AD) is a neurodegenerative disorder accompanied by severe progressive memory and cognitive impairment. The brain of AD patients has an abundance of two abnormal structures, amyloid plaques (senile plaques) and neurofibrillary tangles. In addition, drebrin loss is another hallmark of AD brains, which is a common feature in the brain of both AD patients and AD mouse models. Strong evidence from human genetics and transgenic mouse models has indicated that amyloid β (Aβ) is part of the etiology and pathogenesis of AD. Recently, it has become clear that synaptic dysfunction, including reduced synaptic transmission and loss of dendritic spines, occurs prior to the formation of amyloid plaques and neuronal cell loss. Furthermore, immunohistochemistry using postmortem human brains and AD mouse models has shown that drebrin loss in postsynaptic sites occurs earlier than the presynaptic change in AD brains. In addition, dysregulation of glutamate receptor trafficking and the p21-activated kinase/LIM kinase pathway has been observed in AD brains. It is now believed that soluble Aβ oligomers, namely, Aβ-derived diffusible ligands (ADDLs), but not insoluble Aβ aggregation mediates Aβ toxicity. ADDLs bind to the postsynaptic site and induce the aberrant morphology and density of dendritic spines. Consistent with the AD mouse models, the surface expression of glutamate receptors decreases after ADDL exposure. Importantly, the ADDL-induced drebrin loss in dendritic spines occurs prior to aberrations in dendritic spine morphology and density. These observations indicate that drebrin loss in dendritic spines occurs at the prodromal stage of AD, before the density and morphology of dendritic spines change. Quantitation of drebrin may be a possible tool for diagnosing the prodromal stage of AD, before dementia development in AD.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
- Drebrin
- Cofilin
- α-Amino-3-hydroxy-5-methyl-4-isoxaxole propionic acid receptor
- N-Methyl-D-aspartate receptor
- Alzheimer’s disease
- Aβ-derived diffusible ligands
- Synaptic dysfunction
1 Introduction
Alzheimer’s disease (AD) is a neurodegenerative disorder affecting numerous senior citizens worldwide (Prince et al. 2016). Its main clinical symptoms include mental changes such as progressive memory and cognitive impairment, personality changes, and some behavioral disorders, which may substantially lower the quality of life of elderly individuals (Sosa-Ortiz et al. 2012). The pathology of AD is caused by excessive production of amyloid β (Aβ). Amyloid plaques in the extracellular space, which are generated with Aβ, were originally thought to contribute to neuronal cell death, resulting in memory and cognitive defects in AD (Hardy and Higgins 1992). However, the loss of synapses correlates more closely with the severity of dementia in AD (DeKosky and Scheff 1990; Terry et al. 1991).
Drebrin is an actin-binding protein and is classified into two major isoforms in mammals: drebrin E (ubiquitous isoform) and drebrin A (neuron-specific isoform). Drebrin A accumulates in the mature dendritic spines of excitatory synapses (Sekino et al. 2007). In the past decades, drebrin has been drawing intense research interest, because of the robust decrease in drebrin level in several neurodegenerative and neuropsychiatric disorders that show cognitive defects. In this chapter, we review the drebrin loss underlying the pathology of AD both in vitro and in vivo and propose using drebrin as a marker for estimating synaptic dysfunction in AD.
2 Pathological Changes in Drebrin Level in Alzheimer’s Disease and Mild Cognitive Impairment
The cognitive impairment observed in AD was considered to correlate with the degree of brain atrophy including neuronal cell loss. However, the loss of synapses, which correlates more robustly with the degree of cognitive impairment than the number of amyloid plaques, tangles, and neuronal loss does (Terry et al. 1991), was observed in AD brains. In addition, several AD mouse models showed abnormal long-term potentiation (LTP) and learning defects well before amyloid plaque formation (Chapman et al. 1999; Hsia et al. 1999; Jacobsen et al. 2006). These observations indicate that synaptic failure is likely to occur prior to neuronal cell loss in the pathogenesis of AD. The synapse consists of presynaptic terminals containing neurotransmitters and postsynaptic sites containing neurotransmitter receptors, postsynaptic density, and actin cytoskeleton. Alterations in the synaptic protein stoichiometry in each synapse may induce synaptic dysfunction and synapse loss, because several studies have shown that the expression levels of various pre- and postsynaptic proteins change in various neurological disorders. Drebrin is an actin-binding protein that accumulates in dendritic spines, which are typically postsynaptic receptive regions of excitatory synapses (Sekino et al. 2007). Several studies have revealed that the protein level of drebrin is decreased in the brain of AD patients and the decreased drebrin level correlates with cognitive dysfunction.
2.1 Drebrin in Alzheimer’s Disease Patients
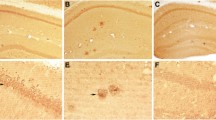
Immunohistochemical analysis revealed that drebrin expression is decreased in the hippocampus of AD patients (Harigaya et al. 1996). Western blot analysis also showed an approximately 70–80% decrease in the levels of drebrin protein. Furthermore, the decrease in drebrin was not restricted to the hippocampus, as it was also observed throughout the cerebral cortex (Hatanpää et al. 1999). In contrast to the marked postsynaptic changes in drebrin level in AD, the content and distribution of synaptophysin, a presynaptic protein, were barely changed (Harigaya et al. 1996). These asymmetric changes in pre- and postsynaptic proteins may result in synaptic dysfunction and cognitive impairment in AD. However, Counts et al. have shown that synaptophysin levels were decreased by ~35% even in severe AD brains (Counts et al. 2006). Therefore, drebrin quantification is more sensitive and consistent than the classical markers for synaptopathy evaluation in AD. Drebrin is also decreased in the brain of Down syndrome (Shim and Lubec 2002) and bipolar disorder (Kim et al. 2010) patients, both of which are associated with cognitive defects (Cotrena et al. 2016; Head et al. 2012). It is known that because of the extra copy of chromosome 21 harboring the amyloid precursor protein (APP) gene, many Down syndrome patients develop AD-like neuropathology by the age of 40 years (Head et al. 2012). Overproduction of Aβ may be involved in the decrease in drebrin in Down syndrome.
2.2 Drebrin in Mild Cognitive Impairment Patients
Mild cognitive impairment (MCI) is a condition in which an individual has mild but measurable changes in thinking abilities that are noticeable to the affected person, family members, and friends. However, MCI patients have the ability to carry out everyday activities (Petersen and Morris 2005). MCI is thought to be a putative prodromal stage of AD, because people with MCI, especially those with memory problems, are more likely to develop AD than people without MCI (Fischer et al. 2007; Busse et al. 2006). In MCI, drebrin is significantly decreased in the superior temporal cortex, although it remains unchanged in most cortical regions (Counts et al. 2006). However, in AD, the drebrin protein level further declines, as it has been observed that in the superior temporal cortex, the decrease in drebrin protein level from no cognitive impairment to AD was greater (an approximate decrease of 57%) than that from no cognitive impairment to MCI (an approximate decrease of 35%). The cognitive function of subjects was also measured by Mini-Mental State Examination (MMSE), which is widely used to measure general cognitive function (Folstein et al. 1975). This study, combining pre- and postmortem analyses, demonstrated that the drebrin level is drastically decreased in AD patients who show an MMSE score lower than 25. Intriguingly, in MCI patients, drebrin is decreased although the MMSE score is almost normal. These findings suggest that drebrin levels begin to decline before the development of the severe cognitive decline in AD. Drebrin may be a useful marker for estimating cognitive functions. Interestingly, a significant decrease in the drebrin level also occurs during normal aging (Hatanpää et al. 1999). The cognitive function declines with aging; thus, it is reasonable to speculate that drebrin loss underlies synaptic dysfunction accompanied by cognitive defects in AD.
3 Drebrin in Mouse Models of Alzheimer’s Disease
As previously stated, the AD brain is characterized by massive neuronal cell and synapse loss in specific regions of the brain (Selkoe 2002), as well as amyloid plaques and neurofibrillary lesions. The major protein component of amyloid plaques is the Aβ polypeptide, which is derived from APP. Many genetically modified mice that mimic the clinical and pathological phenotype of AD have been generated (Guo et al. 2012), because a detailed analysis of the molecular mechanisms underlying synaptic dysfunction in AD is difficult to perform using human postmortem brains. Using these mouse models revealed several molecular mechanisms underlying the pathology of AD. Drebrin loss may cause synaptic dysfunction in AD, because the decrease in drebrin is a common feature among several mouse models exhibiting memory and cognitive impairment.
3.1 Disruption of the Balance Between Drebrin and Cofilin Induces Actin Disassembly in Dendritic Spines, Resulting in Cognitive Defects in Tg2576 Mice
The Tg2576 transgenic mouse is one of the most well-characterized and widely used AD mouse models (Hsiao et al. 1996). It overexpresses a mutant form of human APP (isoform 695) containing the double mutation, K670N and M671L (Lys670 → Asn, Met671 → Leu, Swedish mutation), which is observed in familial AD (FAD). Tg2576 mice develop normally, but the loss of dendritic spine is observed after 4 months of age in the CA1 region and dentate gyrus of the hippocampus (Lanz et al. 2003; Jacobsen et al. 2006). However, neuronal cell loss and neurofibrillary tangles are not found in these mice at 16 months of age (Irizarry et al. 1997). Tg2576 mice develop amyloid plaques by 12 months of age and exhibit impaired learning and memory at 10 months of age (Hsiao et al. 1996). Additionally, the basal synaptic transmission is normal at both 2–8 months of age (no amyloid plaques) and 15–17 months of age (many amyloid plaques). However, the hippocampal LTP becomes severely impaired at the latter age (Chapman et al. 1999). A separate study has demonstrated that contextual memory deficits are observed in Tg2576 mice from 4–5 months of age (Jacobsen et al. 2006).
Drebrin is decreased in the cortex of Tg2576 mice without a significant decrease in the synaptophysin level at 22 months of age (Calon et al. 2004). This asymmetric synaptic marker loss is consistent with human AD brains. In contrast to drebrin, the cofilin level is increased around amyloid plaques in Tg2576 mice at 22 months of age (Zhao et al. 2006). Cofilin is an actin filament (F-actin)-severing protein. The cofilin activity is regulated by phosphorylation of Ser3 through the p21-activated kinase (PAK)/LIM-kinase (LIMK) pathway (Arber et al. 1998; Yang et al. 1998b). Additionally, PAK inhibition causes drebrin loss and memory deficits in normal mice at 11 months of age (Zhao et al. 2006). Thus, actin depolymerization may be upregulated in AD brains, because the PAK/LIMK activity decreases both in the brain of aged Tg2576 mice and AD patients (Zhao et al. 2006; Bamburg and Wiggan 2002). The details of the molecular mechanism of the drebrin-cofilin interaction in Aβ-derived diffusible ligands (ADDL)-exposed neurons are mentioned in Sect. 12.4 of this chapter.
Fractin (fragment of actin) also increases in the aged Tg2576 mouse brain (Calon et al. 2004) and in AD brains (Yang et al. 1998a; Rossiter et al. 2000). Thus, it is probable that an increased fractin level indicates the disruption of actin assembly induced by cofilin hyperactivation in AD. The reciprocal relationship between drebrin and cofilin may be a result of the competitive binding to F-actin. This hypothesis is supported by a study that revealed that drebrin inhibits severing of F-actin induced by cofilin (Grintsevich and Reisler 2014). Drebrin-decorated F-actin is resistant to the depolymerization activity of cofilin. Therefore, imbalanced actin dynamics regulated by drebrin and cofilin may cause the morphological change of dendritic spines and synaptic dysfunction in AD.
3.2 Drebrin Loss Is Involved in Cognitive Deficit in DHA-Depleted Tg2576 Mice
In addition to genetically induced vulnerability to AD, a number of environmental risk factors are thought to be involved in the pathogenesis and progression of AD. One risk factor candidate is docosahexaenoic acid (DHA). DHA is an essential omega-3 polyunsaturated fatty acid (PUFA), which comprises approximately 15% of the total fatty acids in the brain and is enriched in synapses (Salem et al. 2001). Epidemiological studies have shown that DHA levels are significantly lower in AD (Conquer et al. 2000) and correlate with the severity of dementia (Tully et al. 2003). Lipid peroxidation, which is enhanced in the AD brain, accelerates the degradation of PUFAs, including DHA (Montine and Morrow 2005). These findings indicate that the DHA level is a significant risk factor for the development of AD. DHA-depleted Tg2576 mice show decreased postsynaptic drebrin and postsynaptic density-95 (PSD-95) and increased fractin without altering the synaptophysin level (Calon et al. 2004). Additionally, dietary depletion of n-3 PUFA evokes impaired spatial memory in Tg2576 mice. Note that drebrin and PSD-95 levels and memory impairment are restored by supplementing the mice’s diet with DHA. Overexpression of PSD-95 promotes synaptic maturation and enhances synaptic strength (El-Husseini et al. 2000; Schnell et al. 2002), whereas its knockdown causes synaptic dysfunctions, implying a role in synaptic plasticity and dendritic spine formation (Beique et al. 2006; Ehrlich et al. 2007; Migaud et al. 1998). Importantly, decreased PSD-95 level in DHA-depleted Tg2576 may be secondary to drebrin loss, because drebrin regulates the synaptic targeting of PSD-95 (Takahashi et al. 2003). In cultured neurons, antisense suppression of drebrin prevents the synaptic clustering of PSD-95. In addition, drebrin A-specific knockout mice, in which isoform conversion from drebrin E to drebrin A is disrupted (Kojima et al. 2010), exhibit abnormal dendritic spine morphology, impaired LTP, and hippocampus-dependent memory impairment (Kojima et al. 2016). Taking these findings into account, drebrin loss is a defect that can exert a fatal influence on synaptic dysfunction and cognitive defects in AD. Furthermore, we would like to emphasize that the maintenance of drebrin content in the brain may be a crucial factor for cognition.
3.3 Impaired AMPAR Activity Reduces the Drebrin Level in Dendritic Spines in APP and Presenilin-1-Double-Knockin Mice
Another useful AD mouse model is the APPNLh/NLh/PS1P264L/P264L-double-knockin (DKI) mouse, which harbors both mutant APP and mutant presenilin (PS)-1 (Flood et al. 2002). APPNLh/NLh/PS1P264L/P264L-DKI mice were generated by crossing APPNLh/NLh-KI mice with PS1P264L/P264L-KI mice. The APPNLh/NLh KI mouse is a FAD-KI line, as it has human Aβ in the endogenous mouse APP gene (Reaume et al. 1996). Compared with transitional transgenic models such as Tg2576, these KI mice have several advantages. The expression level of APP remains at the physiological level, because the KI allele is controlled by the native APP promoter. In addition, APPNLh/NLh-KI mice bear the Swedish mutation (K679/M671L) with the human sequence, resulting in amyloidogenic β-secretase cleavage of this mutant APP, which is accurate and enhanced, but does not show Aβ deposition. PS1P264L/P264L-KI mice were generated by insertion of the P264L mutation into the mouse PS1 gene, because this mutation (Pro264 → Leu) causes an onset of FAD in humans (Mann et al. 2001). However, neuropathology is not observed in PS1P264L/P264L-KI mice, and primary cortical neurons derived from these mice exhibit basal levels of neurodegeneration comparable to wild-type mice (Siman et al. 2000). In contrast to these single KI mice, APPNLh/NLh/PS1P264L/P264L-DKI mice exhibit Aβ deposition beginning at 6 months of age and reaching robust levels by 15 months of age and show the age-related impairment of memory flexibility (Chang et al. 2006). Electrophysiological analysis revealed that APPNLh/NLh/PS1P264L/P264L-DKI mice show decreased α-amino-3-hydroxy-5-methyl-4-isoxaxole propionic acid receptor (AMPAR) activity in the CA1 region of the hippocampus at 9–12 months of age with low amyloid plaques and age-related impaired long-lasting synaptic plasticity, such as LTP and long-term depression (LTD) (Chang et al. 2006). These findings are supported by anatomical data obtained from quantitative immunoelectron microscopy analysis showing a decrease in the number of synaptic AMPARs in CA1 pyramidal cells. Thus, these results suggest that the regulation of AMPAR trafficking on the postsynaptic membrane is impaired in APPNLh/NLh/PS1P264L/P264L-DKI mice. Deficit in endocytotic and lysosomal trafficking of the AMPAR pathway is known to significantly contribute to AD pathogenesis (Pimplikar et al. 2010). Consistent with this notion, several in vitro studies using organotypic hippocampal slice cultures and dissociated neuronal cultures have demonstrated that APP overexpression and exposure to Aβ induce endocytosis of surface and synaptic AMPAR, leading to synaptic depression and LTP inhibition (Hsieh et al. 2006; Minano-Molina et al. 2011).
A decrease in the drebrin level is also observed in this model. Quantitative immunoelectron microscopy revealed that these mice have fewer drebrin-positive spines than wild-type mice by 6 months of age (Mahadomrongkul et al. 2005; Aoki et al. 2007). It is possible that this drebrin loss is due to the impairment of AMPAR activity, because drebrin accumulation and stability within dendritic spines depend on AMPAR activity (Takahashi et al. 2009). In addition, chronic AMPAR blockade induces abnormal morphology of dendritic spines. Importantly, several studies have demonstrated that Aβ promotes AMPAR endocytosis and hence synaptic depression (Hsieh et al. 2006; Zhao et al. 2010; Liu et al. 2010; Minano-Molina et al. 2011). As mentioned above, drebrin loss induces impairment of PSD-95 clustering in dendritic spines. Taken together, it is beyond doubt that the impairment of AMPAR activity induces drebrin loss, resulting in morphological changes of dendritic spines in AD.
3.4 Drebrin Is Involved in the Dysregulation of NMDAR Distribution Induced by Presenilin Loss
PS1 and PS2 have been identified as pathogenic loci involved in the majority of early onset, autosomal dominant AD. Many mutations are located in genes encoding PS1 and PS2 in FAD patients (Sherrington et al. 1995; Rogaev et al. 1995). PS1 mutations have been demonstrated to increase the production of the pathogenic Aβ (Moehlmann et al. 2002). PS conditional double knockout (cDKO) mice lack both PS1 and PS2. PS-cDKO mice were generated by crossing floxed PS1, αCaMKII-Cre transgenic mice (Yu et al. 2001), and PS2−/− mice (Steiner et al. 1999). PS-cDKO mice have PS1 conditional deletion in excitatory neurons of the postnatal forebrain from about 1 month of age along with PS2 germline deletion (Saura et al. 2004). At 2 months of age, PS-cDKO mice exhibit normal brain morphology, including a normal neuron number, volume, and spine density. In addition, open-field and rotarod tests revealed no significant alterations in behavior, motor coordination, and exploratory anxiety. However, at 2 months of age, mice exhibit mild impairments in hippocampal learning and memory, contextual fear learning, LTP, and N-methyl-D-aspartate receptor (NMDAR) response (Saura et al. 2004). By 6 months of age, PS-cDKO mice exhibit severer symptoms. These mice fail to learn the water maze and contextual fear conditioning tasks and exhibit deficits in the open-field and rotarod tests. By 9 months of age, the loss of dendritic spines is observed in CA1 pyramidal neurons.
At 2 months of age, drebrin A expression decreases both near the PSD and far from the PSD in the CA1 hippocampus of PS-cDKO mice (Lee and Aoki 2012). In contrast to drebrin, the GluN2A subunit of NMDARs is abnormally increased at pre- and postsynaptic sites (Aoki et al. 2009b). Importantly, these alterations are observed at 2 months of age, even though other severe symptoms such as spine loss and neurodegeneration are not observed until 6 months of age. Drebrin loss from dendritic spines may lead to abnormal trafficking of NMDAR to postsynapses at the earlier stage of AD, because drebrin is essential for the homeostatic synaptic scaling of NMDARs (Takahashi et al. 2006; Aoki et al. 2009a). Taken together, the loss of both PS1 and PS2 may result in impaired regulation of NMDARs through a mechanism involving drebrin A. Additionally, it is possible that these early events eventually lead to severer phenomena such as decreased spine density, neuronal cell loss, and cognitive defects in AD.
4 Molecular Mechanisms of ADDL-Induced Drebrin Loss in Dendritic Spines in Alzheimer’s Disease
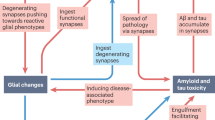
As mentioned above, synapse loss shows the strongest correlation with AD-associated cognitive and memory impairments. Accordingly, advances in the past decades have highlighted the molecular mechanisms of synapse loss in AD. Conventionally, it has been thought that synapse loss depends on the toxicity of insoluble amyloid fibrils, because amyloid plaques are found in AD postmortem brains and in many mouse models of AD exhibiting AD-like cognitive deficits. Although many transgenic AD mouse models generally develop amyloid plaques, some AD mouse models show synaptic dysfunction, such as abnormal LTP and cognitive deficits, well in advance of plaque formation. Recently, it has been revealed that soluble Aβ oligomers, known as ADDLs, are potent central nervous system neurotoxins that accumulate in the AD brain (Gong et al. 2003; Lambert et al. 1998). Based on these observations, it is considered that Aβ toxicity is mediated not only by the insoluble amyloid fibrils but also by ADDLs, and synaptic failure is likely to be one of the earliest events that occur in the pathogenesis of AD prior to amyloid plaque formation and neuronal cell loss. Moreover, several studies performed on human subjects have revealed deficits in LTP-like cortical plasticity in mild-to-moderate AD patients (Inghilleri et al. 2006; Battaglia et al. 2007; Koch et al. 2012). Application of ADDLs induces a decrease in the drebrin level accompanied by aberrant dendritic spine morphology and a reduction in spine density (Lacor et al. 2007; Ishizuka et al. 2014). Additionally, ADDLs facilitate the internalization of AMPAR and NMDAR (Minano-Molina et al. 2011; Snyder et al. 2005), dendritic spine shrinkage (Lacor et al. 2007), and eventual synaptic loss (Shankar et al. 2007). As mentioned in the previous section, drebrin loss may cause dysregulation of NMDAR distribution, resulting in synaptic dysfunction and hence cognitive defects. This section focuses on the molecular mechanisms of ADDL-induced aberrations in the dendritic spine architecture such as drebrin loss, cofilin hyperactivation, and impaired glutamate receptor trafficking.
4.1 ADDLs Induce NMDAR-Mediated Drebrin Loss in Dendritic Spines
The effects of ADDLs on dendritic spines may be mediated, at least in part, by selective binding of ADDLs to certain receptors, such as α7 nicotinic acetylcholine receptor (α7 nAChR) (Wang et al. 2000), leukocyte immunoglobulin-like receptor B2 (Kim et al. 2013), and cellular prion protein (PrPc) (Lauren et al. 2009). ADDLs bind to PrPc, inducing Fyn kinase activation and subsequent phosphorylation of the GluN2B subunit of NMDARs at Tyr1472 (Freir et al. 2011; Um et al. 2012) (Fig. 12.1). This phosphorylation induces an initial increase in NMDAR activity at the postsynaptic site, which induces an increase in Ca2+ influx (De Felice et al. 2007). Transient activation of NMDARs induced by the ADDLs-PrPc interaction is thought to be a trigger of AD pathology, because inhibition of NMDAR or PrPc prevents the toxicity of ADDLs (De Felice et al. 2007; Decker et al. 2010; Lauren et al. 2009; Shankar et al. 2007). ADDLs reduce the amount of drebrin as well as the dendritic spine density in cultured neurons (Ishizuka et al. 2014; Lacor et al. 2007; Zhao et al. 2006). As previously described, decreased drebrin is consistent with the observation in AD patients and AD mouse models. Collectively, there is little doubt that ADDLs induce drebrin loss and hence synaptic failure in the progression of AD. We have previously shown that NMDAR activation induces the disappearance of drebrin from dendritic spines and its appearance in dendritic shafts (Sekino et al. 2006). We have also revealed that activation of myosin II ATPase by Ca2+ influx through NMDARs induces the translocation of drebrin A from dendritic spines to the shaft, so-called drebrin exodus (Mizui et al. 2014) (Fig. 12.1). Based on the above observations, it is clear that ADDL-induced drebrin loss is mediated by excessive Ca2+ influx through abnormal activation of NMDARs, because drebrin loss induced by ADDLs can be prevented by memantine, an NMDAR antagonist (Lacor et al. 2007). It should be noted that low exposure to ADDLs induces drebrin loss in dendritic spines without affecting the morphology and density of dendritic spines, or the protein level of drebrin (Ishizuka et al. 2014). Therefore, we are certain that drebrin loss in dendritic spines occurs prior to the changes in the dendritic spine morphology during the progression of AD.
PrPc-mediated excessive Ca2+ influx induces drebrin exodus and AMPAR internalization. ADDL-PrPc interaction initially activates GluN2B-containing NMDAR by phosphorylation of GluN2B at Tyr1472. Ca2+ influx through NMDAR induces an interaction between myosin II and F-actin, leading to drebrin exodus by myosin II ATPase activation. This Ca2+ influx also induces calcineurin (CaN)/PP1 activation, leading to dephosphorylation of the GluA1 subunit of AMPARs at Ser845. Dephosphorylated AMPARs are internalized by endocytosis. The internalization of AMPAR suppresses the localization of drebrin in dendritic spines
4.2 Possible Involvement of Drebrin in ADDL-Induced Impairment of NMDAR Trafficking to the Postsynaptic Site
Following ADDL-induced initial activation of NMDARs, the surface expression of NMDARs decreases (Lacor et al. 2007; Snyder et al. 2005). ADDLs also bind to α7 nAChR, leading to Ca2+ influx and calcineurin activation (also known as protein phosphatase 2B), which in turn leads to protein phosphatase 1 (PP1) activation (Fig. 12.2). Activated PP1 dephosphorylates and activates striatal-enriched protein tyrosine phosphatase 61 (STEP61). Activated STEP61 promotes the dephosphorylation of GluN2B at Tyr1472, leading to NMDAR internalization from the postsynaptic membranes (Snyder et al. 2005; Zhang et al. 2011). Consistent with these findings, STEP61 is upregulated in several AD mouse models and in the brain of AD patients (Snyder et al. 2005; Kurup et al. 2010). ADDL-induced drebrin dysfunction may impair the reinsertion of NMDARs into the postsynaptic membrane, resulting in impaired synaptic transmission, because drebrin is essential for the synaptic targeting of NMDARs (Aoki et al. 2009a).
α7 nAChR-mediated AMPAR internalization inhibits the accumulation of drebrin in dendritic spines, resulting in impairment of NMDAR trafficking to postsynaptic sites. ADDL-induced Ca2+ influx through α7 nAChR activates CaN/PP1, leading to STEP61 activation. STEP61 dephosphorylates both AMPARs (GluA2 subunit at Tyr876) and NMDARs (GluN2B at Try1472). The dephosphorylation of these receptors induces their internalization by endocytosis. The internalization of AMPAR suppresses the localization of drebrin in dendritic spines. This drebrin loss in dendritic spines induces the impairment of NMDAR reinsertion into postsynaptic membranes
Under physiological conditions, drebrin loss in dendritic spines is a temporary phenomenon. Drebrin reenters into dendritic spines after its exodus, which is induced by NMDAR activation. This bidirectional translocation of drebrin is thought to be essential for synaptic plasticity (Mizui et al. 2014). LTP is associated with drebrin influx into dendritic spines in vivo (Fukazawa et al. 2003; Bosch et al. 2014). However, ADDLs eventually reduce the expression level of drebrin in cultured neurons (Lacor et al. 2007; Zhao et al. 2006; Ishizuka et al. 2014). This discrepancy raised the following question: what are the molecular mechanisms involved in the ADDL-induced irreversible drebrin loss observed in AD?
4.3 AMPAR Internalization Induces Drebrin Loss in Dendritic Spines
In cultured neurons and acute brain slices, ADDLs induce excessive Ca2+ influx through NMDARs, which subsequently activates the calcineurin/PP1 pathway (Shankar et al. 2007; De Felice et al. 2007). Calcineurin/PP1 activation mediates the ADDL-induced internalization of AMPARs by dephosphorylating the GluA1 subunit of AMPARs at Ser845 (Hsieh et al. 2006; Minano-Molina et al. 2011) (Fig. 12.1). The GluA1 subunit at Ser845 is known to be phosphorylated by protein kinase A (PKA) and is crucial for maintaining the stability of AMPARs at synaptic sites (He et al. 2009; Oh et al. 2006; Man et al. 2007). Thus, ADDLs induce AMPAR internalization and impair the synaptic scaling of AMPARs by activating calcineurin/PP1. STEP61 dephosphorylates the GluA2 subunit at Tyr876, leading to internalization of AMPARs (Moult et al. 2006; Zhang et al. 2008) (Fig. 12.2). STEP61 activity is required for ADDL-induced internalization of AMPARs. Genetic and pharmacological inhibition of STEP61 restores the number of AMPARs on the postsynaptic membrane, enhances LTP, and improves cognitive function in AD mice (Zhang et al. 2011, 2010; Xu et al. 2014). This STEP61-mediated AMPAR internalization is consistent with several AD mouse models. As described above, APPNLh/NLh/PS1P264L/P264L-DKI mice show decreased AMPAR activity accompanied by a decrease in synaptic AMPAR number in the CA1 region of the hippocampus (Chang et al. 2006). Additionally, APPswe, Ind transgenic mice, which harbor both Swedish (K670N/M670L) and Indiana (V717F, valine717 → phenylalanine) mutations in human APP (Chishti et al. 2001), display lower levels of Ser845 phosphorylation, a decrease in surface AMPARs expression and impaired learning and memory (Minano-Molina et al. 2011). Thus, we presume that drebrin loss is partly due to the impairment of AMPAR activity induced by ADDLs, because drebrin accumulation and stability within dendritic spines require AMPAR activity (Takahashi et al. 2009).
4.4 ADDL-Induced Cofilin Activation Causes Actin Disassembly in Dendritic Spines
The ADDL-induced initial increase in NMDAR activity induces calcineurin-mediated dephosphorylation and activation of cofilin (Wang et al. 2005; Wu et al. 2010) (Fig. 12.3). This activation of cofilin may also induce actin disassembly in dendritic spines, resulting in synaptic dysfunction. Consistent with this model, both calcineurin and cofilin are required for the ADDL-induced loss of dendritic spines (Shankar et al. 2007). As discussed above, the cofilin level is elevated in the brain of AD patients and Tg2576 mice. ADDL exposure induces a rapid and persistent reduction in the PAK activity and an abnormal distribution of phosphorylated PAK in cultured hippocampal neurons (Zhao et al. 2006). These effects of ADDLs are protected by pretreatment with the oligomer-specific A11 antibody, which does not recognize contaminating fibrils or monomers (Kayed et al. 2003). Exogenously overexpressed PAK prevents ADDL-induced drebrin loss in cultured neurons, although ADDL exposure induces drebrin loss in control neurons. On the contrary, dominant-negative PAK does not prevent ADDL toxicity. These in vitro experiments’ results support the hypotheses that ADDLs cause PAK signaling defects and drebrin loss. Therefore, disruption of the balance between drebrin and cofilin may induce actin disassembly in dendritic spines, resulting in synaptic dysfunction in AD.
ADDL-induced calpain and cofilin activation induce drebrin degradation. Ca2+ influx through NMDAR activates both the calpain and CaN/PP1 pathways. Activated calpain decomposes drebrin and F-actin and induces the conversion of p35 to p25 for the activation of Cdk5. Activated Cdk5 phosphorylates drebrin at Ser142. Cofilin is also activated by CaN/PP1-induced dephosphorylation at Ser3. The activation of calpain and cofilin induces actin disassembly in dendritic spines
4.5 ADDLs Induce Proteolysis of Drebrin
Disturbance of intracellular Ca2+ promotes the activation of calcium-dependent papain-like enzyme (calpain) (Murachi et al. 1980). A recent study has reported that NMDAR-mediated calpain activation elicits the degradation of drebrin and F-actin in cultured neurons (Chimura et al. 2015) (Fig. 12.3). Thus, ADDL exposure may induce calpain-mediated drebrin degradation in AD, because ADDLs induce both NMDAR hyperactivation and a decrease in drebrin. In agreement with this finding, calpain activity is elevated in AD (Saito et al. 1993). Similarly, calpastatin, an endogenous calpain inhibitor, is decreased in AD (Rao et al. 2008). Therefore, this calpain-induced degradation of drebrin and F-actin may also contribute to the loss of dendritic spines in AD. Additionally, calpain activation induces the proteolytic conversion of p35, which is a regulatory subunit of cyclin-dependent kinase 5 (Cdk5), to p25. p25 is a neuron-specific Cdk5 activator and a constitutively active form that enhances Cdk5 activity in AD (Patrick et al. 1999; Lew et al. 1994). Intriguingly, Cdk5 phosphorylates drebrin at Ser142, possibly resulting in a conformational change of drebrin (Worth et al. 2013). If the phosphorylated form of drebrin is more vulnerable to proteolysis than the non-phosphorylated form, the CDK5-mediated phosphorylation of this site may be involved in the ADDL-induced drebrin degradation.
4.6 HDAC Mediates ADDL-Induced Drebrin Loss in Dendritic Spines
Recently, we have revealed that histone deacetylase (HDAC) mediates drebrin loss in dendritic spines induced by ADDL exposure (Ishizuka et al. 2014). HDACs are a class of enzymes that remove acetyl groups from lysine amino acids on histones, allowing the histones to wrap the DNA more tightly and downregulate gene expression (Gräff and Tsai 2013). It is clear that increased HDAC activity correlates with the pathology and the etiology of AD, because HDAC activity is elevated in the AD mouse models (Gräff et al. 2012) and some HDAC inhibitors can improve memory and cognitive function, as well as reduced dendritic spine density in Tg2576 (Ricobaraza et al. 2009, 2012) and APP/PS1 (Kilgore et al. 2010; Francis et al. 2009) mouse models of AD. Intriguingly, drebrin loss induced by ADDL exposure is ameliorated by the HDAC inhibitor, suberoylanilide hydroxamic acid (SAHA). Thus, epigenetic modification may be involved in the regulation of drebrin localization. Although the precise molecular mechanism of this process is not known, yet, the inhibition of HDAC may induce the expression of a protein that inhibits Ca2+ signal transduction, such as a Ca2+ sensor that triggers the disinhibition of myosin II ATPase involved in the drebrin exodus. Alternatively, HDAC inhibition may induce the expression of a protein that promotes drebrin accumulation in dendritic spines. On the contrary, HDAC activation may suppress the expression of a protein involved in drebrin accumulation in dendritic spines in AD brains. These findings suggest that HDAC is involved in ADDL-induced synaptic defects and that the regulation of histone acetylation/deacetylation is crucial for synaptic functions under cellular stress conditions, such as ADDL exposure. Taken together, drebrin disappearance from dendritic spines followed by ADDL-induced excessive NMDAR activation underlies the synaptic dysfunction in AD, and HDAC may be a potential therapeutic target for AD patients.
5 Conclusions and Outlook
In this chapter, we discussed the aberrant behaviors of drebrin in AD by focusing on three main topics: (1) human AD brain, (2) mouse models of AD, and (3) the molecular mechanism of ADDL-induced drebrin loss. Drebrin loss is a common feature in the brain of AD patients and AD mouse models. The study performed by combining ante- and postmortem analyses of human subjects revealed that the drebrin level closely correlates with the severity of cognitive impairment. Additionally, the AD mouse models showed a decreased drebrin level and impaired glutamate receptor distribution. It is also clear that ADDLs induce an excess influx of Ca2+, leading to glutamate receptor internalization and drebrin loss. It should be noted that acute exposure to a low dose of ADDLs induces drebrin loss in dendritic spines without reducing both the density of dendritic spines and the expression level of drebrin. This observation strongly suggests that drebrin loss in dendritic spines occurs at the prodromal stage of AD, in which the patients begin to display slight cognitive impairments prior to the development of MCI, because the amount of drebrin is decreased in MCI patients. HDAC also contributes to drebrin loss in AD, because HDAC inhibitor attenuates ADDL-induced drebrin loss. These findings provide insight into the mechanisms of ADDL-toxicity and suggest that HDAC inhibitors may hold therapeutic potential against AD.
In conclusion, by focusing on drebrin, our understanding of the molecular mechanisms underlying synaptic dysfunction accompanied by cognitive defects in AD has been greatly improved. Increasing evidence has uncovered the molecular mechanisms of synaptic dysfunction in AD, such as drebrin loss in dendritic spines, impaired glutamate receptor trafficking, and decreased PAK/LIMK signal. These signals act cooperatively to mediate the toxic effects of ADDLs; there is no doubt that the aberration in drebrin behavior in dendritic spines is one of the direct causes of dendritic spine abnormality. Therefore, further study of drebrin and its associated signaling mechanisms would be of value to develop more effective therapeutic strategies for AD treatment.
References
Aoki C, Mahadomrongkul V, Fujisawa S, Habersat R, Shirao T (2007) Chemical and morphological alterations of spines within the hippocampus and entorhinal cortex precede the onset of Alzheimer's disease pathology in double knock-in mice. J Comp Neurol 505:352–362
Aoki C, Kojima N, Sabaliauskas N et al (2009a) Drebrin a knockout eliminates the rapid form of homeostatic synaptic plasticity at excitatory synapses of intact adult cerebral cortex. J Comp Neurol 517:105–121
Aoki C, Lee J, Nedelescu H, Ahmed T, Ho A, Shen J (2009b) Increased levels of NMDA receptor NR2A subunits at pre- and postsynaptic sites of the hippocampal CA1: an early response to conditional double knockout of presenilin 1 and 2. J Comp Neurol 517:512–523
Arber S, Barbayannis FA, Hanser H, Schneider C, Stanyon CA, Bernard O, Caroni P (1998) Regulation of actin dynamics through phosphorylation of cofilin by LIM-kinase. Nature 393:805–809
Bamburg JR, Wiggan OP (2002) ADF/cofilin and actin dynamics in disease. Trends Cell Biol 12:598–605
Battaglia F, Wang HY, Ghilardi MF, Gashi E, Quartarone A, Friedman E, Nixon RA (2007) Cortical plasticity in Alzheimer's disease in humans and rodents. Biol Psychiatry 62:1405–1412
Beique JC, Lin DT, Kang MG, Aizawa H, Takamiya K, Huganir RL (2006) Synapse-specific regulation of AMPA receptor function by PSD-95. Proc Natl Acad Sci U S A 103:19535–19540
Bosch M, Castro J, Saneyoshi T, Matsuno H, Sur M, Hayashi Y (2014) Structural and molecular remodeling of dendritic spine substructures during long-term potentiation. Neuron 82:444–459
Busse A, Hensel A, Guhne U, Angermeyer MC, Riedel-Heller SG (2006) Mild cognitive impairment: long-term course of four clinical subtypes. Neurology 67:2176–2185
Calon F, Lim GP, Yang F et al (2004) Docosahexaenoic acid protects from dendritic pathology in an Alzheimer's disease mouse model. Neuron 43:633–645
Chang EH, Savage MJ, Flood DG, Thomas JM, Levy RB, Mahadomrongkul V, Shirao T, Aoki C, Huerta PT (2006) AMPA receptor downscaling at the onset of Alzheimer's disease pathology in double knockin mice. Proc Natl Acad Sci U S A 103:3410–3415
Chapman PF, White GL, Jones MW et al (1999) Impaired synaptic plasticity and learning in aged amyloid precursor protein transgenic mice. Nat Neurosci 2:271–276
Chimura T, Launey T, Yoshida N (2015) Calpain-mediated degradation of Drebrin by Excitotoxicity in vitro and in vivo. PLoS One 10:e0125119
Chishti MA, Yang DS, Janus C et al (2001) Early-onset amyloid deposition and cognitive deficits in transgenic mice expressing a double mutant form of amyloid precursor protein 695. J Biol Chem 276:21562–21570
Conquer JA, Tierney MC, Zecevic J, Bettger WJ, Fisher RH (2000) Fatty acid analysis of blood plasma of patients with Alzheimer's disease, other types of dementia, and cognitive impairment. Lipids 35:1305–1312
Cotrena C, Branco LD, Shansis FM, Fonseca RP (2016) Executive function impairments in depression and bipolar disorder: association with functional impairment and quality of life. J Affect Disord 190:744–753
Counts SE, Nadeem M, Lad SP, Wuu J, Mufson EJ (2006) Differential expression of synaptic proteins in the frontal and temporal cortex of elderly subjects with mild cognitive impairment. J Neuropathol Exp Neurol 65:592–601
De Felice FG, Velasco PT, Lambert MP, Viola K, Fernandez SJ, Ferreira ST, Klein WL (2007) Abeta oligomers induce neuronal oxidative stress through an N-methyl-D-aspartate receptor-dependent mechanism that is blocked by the Alzheimer drug memantine. J Biol Chem 282:11590–11601
Decker H, Jurgensen S, Adrover MF et al (2010) N-methyl-D-aspartate receptors are required for synaptic targeting of Alzheimer’s toxic amyloid-beta peptide oligomers. J Neurochem 115:1520–1529
DeKosky ST, Scheff SW (1990) Synapse loss in frontal cortex biopsies in Alzheimer's disease: correlation with cognitive severity. Ann Neurol 27:457–464
Ehrlich I, Klein M, Rumpel S, Malinow R (2007) PSD-95 is required for activity-driven synapse stabilization. Proc Natl Acad Sci U S A 104:4176–4181
El-Husseini AE, Schnell E, Chetkovich DM, Nicoll RA, Bredt DS (2000) PSD-95 involvement in maturation of excitatory synapses. Science 290:1364–1368
Fischer P, Jungwirth S, Zehetmayer S, Weissgram S, Hoenigschnabl S, Gelpi E, Krampla W, Tragl KH (2007) Conversion from subtypes of mild cognitive impairment to Alzheimer dementia. Neurology 68:288–291
Flood DG, Reaume AG, Dorfman KS et al (2002) FAD mutant PS-1 gene-targeted mice: increased a beta 42 and a beta deposition without APP overproduction. Neurobiol Aging 23:335–348
Folstein MF, Folstein SE, McHugh PR (1975) Mini-mental state. A practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res 12:189–198
Francis YI, Fà M, Ashraf H, Zhang H, Staniszewski A, Latchman DS, Arancio O (2009) Dysregulation of histone acetylation in the APP/PS1 mouse model of Alzheimer's disease. J Alzheimers Dis 18:131–139
Freir DB, Nicoll AJ, Klyubin I et al (2011) Interaction between prion protein and toxic amyloid beta assemblies can be therapeutically targeted at multiple sites. Nat Commun 2:336
Fukazawa Y, Saitoh Y, Ozawa F, Ohta Y, Mizuno K, Inokuchi K (2003) Hippocampal LTP is accompanied by enhanced F-actin content within the dendritic spine that is essential for late LTP maintenance in vivo. Neuron 38:447–460
Gong Y, Chang L, Viola KL, Lacor PN, Lambert MP, Finch CE, Krafft GA, Klein WL (2003) Alzheimer's disease-affected brain: presence of oligomeric a beta ligands (ADDLs) suggests a molecular basis for reversible memory loss. Proc Natl Acad Sci U S A 100:10417–10422
Gräff J, Tsai LH (2013) Histone acetylation: molecular mnemonics on the chromatin. Nat Rev Neurosci 14:97–111
Gräff J, Rei D, Guan JS et al (2012) An epigenetic blockade of cognitive functions in the neurodegenerating brain. Nature 483:222–226
Grintsevich EE, Reisler E (2014) Drebrin inhibits cofilin-induced severing of F-actin. Cytoskeleton 71:472–483
Guo Q, Wang Z, Li H, Wiese M, Zheng H (2012) APP physiological and pathophysiological functions: insights from animal models. Cell Res 22:78–89
Hardy JA, Higgins GA (1992) Alzheimer's disease: the amyloid cascade hypothesis. Science 256:184–185
Harigaya Y, Shoji M, Shirao T, Hirai S (1996) Disappearance of actin-binding protein, drebrin, from hippocampal synapses in Alzheimer's disease. J Neurosci Res 43:87–92
Hatanpää K, Isaacs KR, Shirao T, Brady DR, Rapoport SI (1999) Loss of proteins regulating synaptic plasticity in normal aging of the human brain and in Alzheimer disease. J Neuropathol Exp Neurol 58:637–643
He K, Song L, Cummings LW, Goldman J, Huganir RL, Lee HK (2009) Stabilization of Ca2+−permeable AMPA receptors at perisynaptic sites by GluR1-S845 phosphorylation. Proc Natl Acad Sci U S A 106:20033–20038
Head E, Silverman W, Patterson D, Lott IT (2012) Aging and down syndrome. Curr Gerontol Geriatr Res 2012:412536
Hsia AY, Masliah E, McConlogue L et al (1999) Plaque-independent disruption of neural circuits in Alzheimer's disease mouse models. Proc Natl Acad Sci U S A 96:3228–3233
Hsiao K, Chapman P, Nilsen S, Eckman C, Harigaya Y, Younkin S, Yang F, Cole G (1996) Correlative memory deficits, Abeta elevation, and amyloid plaques in transgenic mice. Science 274:99–102
Hsieh H, Boehm J, Sato C, Iwatsubo T, Tomita T, Sisodia S, Malinow R (2006) AMPAR removal underlies Abeta-induced synaptic depression and dendritic spine loss. Neuron 52:831–843
Inghilleri M, Conte A, Frasca V, Scaldaferri N, Gilio F, Santini M, Fabbrini G, Prencipe M, Berardelli A (2006) Altered response to rTMS in patients with Alzheimer's disease. Clin Neurophysiol 117:103–109
Irizarry MC, McNamara M, Fedorchak K, Hsiao K, Hyman BT (1997) APPSw transgenic mice develop age-related a beta deposits and neuropil abnormalities, but no neuronal loss in CA1. J Neuropathol Exp Neurol 56:965–973
Ishizuka Y, Shimizu H, Takagi E, Kato M, Yamagata H, Mikuni M, Shirao T (2014) Histone deacetylase mediates the decrease in drebrin cluster density induced by amyloid beta oligomers. Neurochem Int 76:114–121
Jacobsen JS, Wu CC, Redwine JM et al (2006) Early-onset behavioral and synaptic deficits in a mouse model of Alzheimer's disease. Proc Natl Acad Sci U S A 103:5161–5166
Kayed R, Head E, Thompson JL, McIntire TM, Milton SC, Cotman CW, Glabe CG (2003) Common structure of soluble amyloid oligomers implies common mechanism of pathogenesis. Science 300:486–489
Kilgore M, Miller CA, Fass DM, Hennig KM, Haggarty SJ, Sweatt JD, Rumbaugh G (2010) Inhibitors of class 1 histone deacetylases reverse contextual memory deficits in a mouse model of Alzheimer's disease. Neuropsychopharmacology 35:870–880
Kim HW, Rapoport SI, Rao JS (2010) Altered expression of apoptotic factors and synaptic markers in postmortem brain from bipolar disorder patients. Neurobiol Dis 37:596–603
Kim T, Vidal GS, Djurisic M, William CM, Birnbaum ME, Garcia KC, Hyman BT, Shatz CJ (2013) Human LilrB2 is a beta-amyloid receptor and its murine homolog PirB regulates synaptic plasticity in an Alzheimer's model. Science 341:1399–1404
Koch G, Di Lorenzo F, Bonni S, Ponzo V, Caltagirone C, Martorana A (2012) Impaired LTP- but not LTD-like cortical plasticity in Alzheimer's disease patients. J Alzheimers Dis 31:593–599
Kojima N, Hanamura K, Yamazaki H, Ikeda T, Itohara S, Shirao T (2010) Genetic disruption of the alternative splicing of drebrin gene impairs context-dependent fear learning in adulthood. Neuroscience 165:138–150
Kojima N, Yasuda H, Hanamura K, Ishizuka Y, Sekino Y, Shirao T (2016) Drebrin a regulates hippocampal LTP and hippocampus-dependent fear learning in adult mice. Neuroscience 324:218–226
Kurup P, Zhang Y, Xu J, Venkitaramani DV, Haroutunian V, Greengard P, Nairn AC, Lombroso PJ (2010) Abeta-mediated NMDA receptor endocytosis in Alzheimer's disease involves ubiquitination of the tyrosine phosphatase STEP61. J Neurosci 30:5948–5957
Lacor PN, Buniel MC, Furlow PW, Clemente AS, Velasco PT, Wood M, Viola KL, Klein WL (2007) Abeta oligomer-induced aberrations in synapse composition, shape, and density provide a molecular basis for loss of connectivity in Alzheimer's disease. J Neurosci 27:796–807
Lambert MP, Barlow AK, Chromy BA et al (1998) Diffusible, nonfibrillar ligands derived from Abeta1-42 are potent central nervous system neurotoxins. Proc Natl Acad Sci U S A 95:6448–6453
Lanz TA, Carter DB, Merchant KM (2003) Dendritic spine loss in the hippocampus of young PDAPP and Tg2576 mice and its prevention by the ApoE2 genotype. Neurobiol Dis 13:246–253
Lauren J, Gimbel DA, Nygaard HB, Gilbert JW, Strittmatter SM (2009) Cellular prion protein mediates impairment of synaptic plasticity by amyloid-beta oligomers. Nature 457:1128–1132
Lee D, Aoki C (2012) Presenilin conditional double knockout mice exhibit decreases in drebrin a at hippocampal CA1 synapses. Synapse 66:870–879
Lew J, Huang QQ, Qi Z, Winkfein RJ, Aebersold R, Hunt T, Wang JH (1994) A brain-specific activator of cyclin-dependent kinase 5. Nature 371:423–426
Liu SJ, Gasperini R, Foa L, Small DH (2010) Amyloid-beta decreases cell-surface AMPA receptors by increasing intracellular calcium and phosphorylation of GluR2. J Alzheimers Dis 21:655–666
Mahadomrongkul V, Huerta PT, Shirao T, Aoki C (2005) Stability of the distribution of spines containing drebrin a in the sensory cortex layer I of mice expressing mutated APP and PS1 genes. Brain Res 1064:66–74
Man HY, Sekine-Aizawa Y, Huganir RL (2007) Regulation of {alpha}-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptor trafficking through PKA phosphorylation of the Glu receptor 1 subunit. Proc Natl Acad Sci U S A 104:3579–3584
Mann DM, Pickering-Brown SM, Takeuchi A, Iwatsubo T, Members of the Familial Alzheimer's Disease Pathology Study Group (2001) Amyloid angiopathy and variability in amyloid beta deposition is determined by mutation position in presenilin-1-linked Alzheimer's disease. Am J Pathol 158:2165–2175
Migaud M, Charlesworth P, Dempster M et al (1998) Enhanced long-term potentiation and impaired learning in mice with mutant postsynaptic density-95 protein. Nature 396:433–439
Minano-Molina AJ, Espana J, Martin E, Barneda-Zahonero B, Fado R, Sole M, Trullas R, Saura CA, Rodriguez-Alvarez J (2011) Soluble oligomers of amyloid-beta peptide disrupt membrane trafficking of alpha-amino-3-hydroxy-5-methylisoxazole-4-propionic acid receptor contributing to early synapse dysfunction. J Biol Chem 286:27311–27321
Mizui T, Sekino Y, Yamazaki H, Ishizuka Y, Takahashi H, Kojima N, Kojima M, Shirao T (2014) Myosin II ATPase activity mediates the long-term potentiation-induced exodus of stable F-actin bound by drebrin a from dendritic spines. PLoS One 9:e85367
Moehlmann T, Winkler E, Xia X et al (2002) Presenilin-1 mutations of leucine 166 equally affect the generation of the notch and APP intracellular domains independent of their effect on Abeta 42 production. Proc Natl Acad Sci U S A 99:8025–8030
Montine TJ, Morrow JD (2005) Fatty acid oxidation in the pathogenesis of Alzheimer's disease. Am J Pathol 166:1283–1289
Moult PR, Gladding CM, Sanderson TM, Fitzjohn SM, Bashir ZI, Molnar E, Collingridge GL (2006) Tyrosine phosphatases regulate AMPA receptor trafficking during metabotropic glutamate receptor-mediated long-term depression. J Neurosci 26:2544–2554
Murachi T, Tanaka K, Hatanaka M, Murakami T (1980) Intracellular Ca2+−dependent protease (calpain) and its high-molecular-weight endogenous inhibitor (calpastatin). Adv Enzym Regul 19:407–424
Oh MC, Derkach VA, Guire ES, Soderling TR (2006) Extrasynaptic membrane trafficking regulated by GluR1 serine 845 phosphorylation primes AMPA receptors for long-term potentiation. J Biol Chem 281:752–758
Patrick GN, Zukerberg L, Nikolic M, de la Monte S, Dikkes P, Tsai LH (1999) Conversion of p35 to p25 deregulates Cdk5 activity and promotes neurodegeneration. Nature 402:615–622
Petersen RC, Morris JC (2005) Mild cognitive impairment as a clinical entity and treatment target. Arch Neurol 62:1160–1163. discussion 1167
Pimplikar SW, Nixon RA, Robakis NK, Shen J, Tsai LH (2010) Amyloid-independent mechanisms in Alzheimer's disease pathogenesis. J Neurosci 30:14946–14954
Prince M, Comas-Herrera A, Knapp M, Guerchet M, Karagiannidou M (2016) Improving healthcare for people living with dementia: coverage, quality and costs now and in the future. In: World Alzheimer Report 2016
Rao MV, Mohan PS, Peterhoff CM et al (2008) Marked calpastatin (CAST) depletion in Alzheimer's disease accelerates cytoskeleton disruption and neurodegeneration: neuroprotection by CAST overexpression. J Neurosci 28:12241–12254
Reaume AG, Howland DS, Trusko SP, Savage MJ, Lang DM, Greenberg BD, Siman R, Scott RW (1996) Enhanced amyloidogenic processing of the beta-amyloid precursor protein in gene-targeted mice bearing the Swedish familial Alzheimer's disease mutations and a humanized Abeta sequence. J Biol Chem 271:23380–23388
Ricobaraza A, Cuadrado-Tejedor M, Perez-Mediavilla A, Frechilla D, Del Rio J, Garcia-Osta A (2009) Phenylbutyrate ameliorates cognitive deficit and reduces tau pathology in an Alzheimer's disease mouse model. Neuropsychopharmacology 34:1721–1732
Ricobaraza A, Cuadrado-Tejedor M, Marco S, Perez-Otano I, Garcia-Osta A (2012) Phenylbutyrate rescues dendritic spine loss associated with memory deficits in a mouse model of Alzheimer disease. Hippocampus 22:1040–1050
Rogaev EI, Sherrington R, Rogaeva EA et al (1995) Familial Alzheimer's disease in kindreds with missense mutations in a gene on chromosome 1 related to the Alzheimer's disease type 3 gene. Nature 376:775–778
Rossiter JP, Anderson LL, Yang F, Cole GM (2000) Caspase-cleaved actin (fractin) immunolabelling of Hirano bodies. Neuropathol Appl Neurobiol 26:342–346
Saito K, Elce JS, Hamos JE, Nixon RA (1993) Widespread activation of calcium-activated neutral proteinase (calpain) in the brain in Alzheimer disease: a potential molecular basis for neuronal degeneration. Proc Natl Acad Sci U S A 90:2628–2632
Salem N Jr, Litman B, Kim HY, Gawrisch K (2001) Mechanisms of action of docosahexaenoic acid in the nervous system. Lipids 36:945–959
Saura CA, Choi SY, Beglopoulos V et al (2004) Loss of presenilin function causes impairments of memory and synaptic plasticity followed by age-dependent neurodegeneration. Neuron 42:23–36
Schnell E, Sizemore M, Karimzadegan S, Chen L, Bredt DS, Nicoll RA (2002) Direct interactions between PSD-95 and stargazin control synaptic AMPA receptor number. Proc Natl Acad Sci U S A 99:13902–13907
Sekino Y, Tanaka S, Hanamura K, Yamazaki H, Sasagawa Y, Xue Y, Hayashi K, Shirao T (2006) Activation of N-methyl-D-aspartate receptor induces a shift of drebrin distribution: disappearance from dendritic spines and appearance in dendritic shafts. Mol Cell Neurosci 31:493–504
Sekino Y, Kojima N, Shirao T (2007) Role of actin cytoskeleton in dendritic spine morphogenesis. Neurochem Int 51:92–104
Selkoe DJ (2002) Alzheimer's disease is a synaptic failure. Science 298:789–791
Shankar GM, Bloodgood BL, Townsend M, Walsh DM, Selkoe DJ, Sabatini BL (2007) Natural oligomers of the Alzheimer amyloid-beta protein induce reversible synapse loss by modulating an NMDA-type glutamate receptor-dependent signaling pathway. J Neurosci 27:2866–2875
Sherrington R, Rogaev EI, Liang Y et al (1995) Cloning of a gene bearing missense mutations in early-onset familial Alzheimer's disease. Nature 375:754–760
Shim KS, Lubec G (2002) Drebrin, a dendritic spine protein, is manifold decreased in brains of patients with Alzheimer's disease and down syndrome. Neurosci Lett 324:209–212
Siman R, Reaume AG, Savage MJ, Trusko S, Lin YG, Scott RW, Flood DG (2000) Presenilin-1 P264L knock-in mutation: differential effects on abeta production, amyloid deposition, and neuronal vulnerability. J Neurosci 20:8717–8726
Snyder EM, Nong Y, Almeida CG et al (2005) Regulation of NMDA receptor trafficking by amyloid-beta. Nat Neurosci 8:1051–1058
Sosa-Ortiz AL, Acosta-Castillo I, Prince MJ (2012) Epidemiology of dementias and Alzheimer’s disease. Arch Med Res 43:600–608
Steiner H, Duff K, Capell A et al (1999) A loss of function mutation of presenilin-2 interferes with amyloid beta-peptide production and notch signaling. J Biol Chem 274:28669–28673
Takahashi H, Sekino Y, Tanaka S, Mizui T, Kishi S, Shirao T (2003) Drebrin-dependent actin clustering in dendritic filopodia governs synaptic targeting of postsynaptic density-95 and dendritic spine morphogenesis. J Neurosci 23:6586–6595
Takahashi H, Mizui T, Shirao T (2006) Down-regulation of drebrin a expression suppresses synaptic targeting of NMDA receptors in developing hippocampal neurones. J Neurochem 97(Suppl 1):110–115
Takahashi H, Yamazaki H, Hanamura K, Sekino Y, Shirao T (2009) Activity of the AMPA receptor regulates drebrin stabilization in dendritic spine morphogenesis. J Cell Sci 122:1211–1219
Terry RD, Masliah E, Salmon DP, Butters N, DeTeresa R, Hill R, Hansen LA, Katzman R (1991) Physical basis of cognitive alterations in Alzheimer's disease: synapse loss is the major correlate of cognitive impairment. Ann Neurol 30:572–580
Tully AM, Roche HM, Doyle R, Fallon C, Bruce I, Lawlor B, Coakley D, Gibney MJ (2003) Low serum cholesteryl ester-docosahexaenoic acid levels in Alzheimer's disease: a case-control study. Br J Nutr 89:483–489
Um JW, Nygaard HB, Heiss JK, Kostylev MA, Stagi M, Vortmeyer A, Wisniewski T, Gunther EC, Strittmatter SM (2012) Alzheimer amyloid-beta oligomer bound to postsynaptic prion protein activates Fyn to impair neurons. Nat Neurosci 15:1227–1235
Wang HY, Lee DH, Davis CB, Shank RP (2000) Amyloid peptide Abeta(1–42) binds selectively and with picomolar affinity to alpha7 nicotinic acetylcholine receptors. J Neurochem 75:1155–1161
Wang Y, Shibasaki F, Mizuno K (2005) Calcium signal-induced cofilin dephosphorylation is mediated by slingshot via calcineurin. J Biol Chem 280:12683–12689
Worth DC, Daly CN, Geraldo S, Oozeer F, Gordon-Weeks PR (2013) Drebrin contains a cryptic F-actin-bundling activity regulated by Cdk5 phosphorylation. J Cell Biol 202:793–806
Wu HY, Hudry E, Hashimoto T et al (2010) Amyloid beta induces the morphological neurodegenerative triad of spine loss, dendritic simplification, and neuritic dystrophies through calcineurin activation. J Neurosci 30:2636–2649
Xu J, Chatterjee M, Baguley TD et al (2014) Inhibitor of the tyrosine phosphatase STEP reverses cognitive deficits in a mouse model of Alzheimer's disease. PLoS Biol 12:e1001923
Yang F, Sun X, Beech W, Teter B, Wu S, Sigel J, Vinters HV, Frautschy SA, Cole GM (1998a) Antibody to caspase-cleaved actin detects apoptosis in differentiated neuroblastoma and plaque-associated neurons and microglia in Alzheimer's disease. Am J Pathol 152:379–389
Yang N, Higuchi O, Ohashi K, Nagata K, Wada A, Kangawa K, Nishida E, Mizuno K (1998b) Cofilin phosphorylation by LIM-kinase 1 and its role in Rac-mediated actin reorganization. Nature 393:809–812
Yu H, Saura CA, Choi SY et al (2001) APP processing and synaptic plasticity in presenilin-1 conditional knockout mice. Neuron 31:713–726
Zhang Y, Venkitaramani DV, Gladding CM, Zhang Y, Kurup P, Molnar E, Collingridge GL, Lombroso PJ (2008) The tyrosine phosphatase STEP mediates AMPA receptor endocytosis after metabotropic glutamate receptor stimulation. J Neurosci 28:10561–10566
Zhang Y, Kurup P, Xu J et al (2010) Genetic reduction of striatal-enriched tyrosine phosphatase (STEP) reverses cognitive and cellular deficits in an Alzheimer's disease mouse model. Proc Natl Acad Sci U S A 107:19014–19019
Zhang Y, Kurup P, Xu J, Anderson GM, Greengard P, Nairn AC, Lombroso PJ (2011) Reduced levels of the tyrosine phosphatase STEP block beta amyloid-mediated GluA1/GluA2 receptor internalization. J Neurochem 119:664–672
Zhao L, Ma QL, Calon F et al (2006) Role of p21-activated kinase pathway defects in the cognitive deficits of Alzheimer disease. Nat Neurosci 9:234–242
Zhao WQ, Santini F, Breese R et al (2010) Inhibition of calcineurin-mediated endocytosis and alpha-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA) receptors prevents amyloid beta oligomer-induced synaptic disruption. J Biol Chem 285:7619–7632
Acknowledgment
We would like to thank Dr. Masaki Ikeda for his comments and useful discussion of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer Japan KK
About this chapter
Cite this chapter
Ishizuka, Y., Hanamura, K. (2017). Drebrin in Alzheimer’s Disease. In: Shirao, T., Sekino, Y. (eds) Drebrin. Advances in Experimental Medicine and Biology, vol 1006. Springer, Tokyo. https://doi.org/10.1007/978-4-431-56550-5_12
Download citation
DOI: https://doi.org/10.1007/978-4-431-56550-5_12
Published:
Publisher Name: Springer, Tokyo
Print ISBN: 978-4-431-56548-2
Online ISBN: 978-4-431-56550-5
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)