Abstract
Certain fish exhibit lunar and semilunar reproductive activities that are repeated at regular intervals of 1 month and 2 weeks, respectively. Fish with the lunar cycle exhibit spawning around a selective moon phase, whereas those with the semilunar cycle repeat spawnings twice within a month. Environmental changes in moonlight or tides may be involved in the synchronization of fish reproductive activity. Spawning of the goldlined spinefoot Siganus guttatus, a lunar spawner around the first quarter moon, is disrupted under constant moonlight conditions. Exposing fish to moonlight around the full moon resulted in suppression of melatonin, suggesting the perception and utilization of changes in moonlight for synchrony. Of the clock genes examined, Period (Per2) in the pineal organ was higher at midnight around the full moon than the new moon, whereas Cryptochrome (Cry1 and Cry3) in the brain peaked around the first quarter moon. Although circalunar-specific genes have not yet been identified in any organism, some elements of the circadian system are likely involved in the exertion of lunar-related reproductive activities.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
Many organisms on the Earth possess endogenous clocks that respond to rhythmic changes in light and temperature (Pittendrigh 1993; Panda et al. 2002). Endogenous clocks help organisms to anticipate upcoming daily and annual environmental changes in their habitat and to adjust biochemical, physiological, and behavioral processes accordingly (Pittendrigh 1993). The acquisition of these clocks is closely related to long-term evolutionary and adaptation processes under constant repetition of the Earth’s rotation and revolution. In addition, the moon is also considered to affect the entrainment of endogenous clocks in organisms. Although information on lunar-related clocks is limited, rhythmic changes in a habitat arising from the movement of the moon around the Earth undoubtedly are utilized for synchronization of various biological activities, including locomotion, migration, and reproduction. Some lunar-related activities even oscillate under constant conditions (Hsiao and Meier 1989; Motohashi et al. 2010), implying the involvement of endogenous clocks (e.g., circalunar, circasemilunar, or circatidal clocks).
Several lunar-related cycles exist: the lunar cycle, the semilunar cycle, and the tidal cycle. The lunar cycle involves a periodic change repeated at 1-month intervals, and biological activities of organisms peak around the species-selective lunar phase. The semilunar cycle is periodic activity that seems to appear at a 14.7-day interval. In many examples of this cycle, peaks of biological activity occur twice within a month. The tidal cycle also involves moon-related activity, which is well correlated with a daily tidal cycle and caused by the combined effects of gravitational forces exerted by the moon and the sun and the rotation of the Earth (Leatherland et al. 1992).
Marine organisms inhabiting shallow waters in tropical and subtropical zones often exhibit various types of lunar-related rhythmicity (Harrison et al. 1984; Thresher 1984). One possible reason why such patterns are typical of these regions is that the lower variation in water temperature and photoperiod in these zones is correlated with a relative increase in the importance and reliability of cues from the moon (Takemura et al. 2010). In this chapter, we focus on the lunar and semilunar cycles occurring in the reproductive events of teleost fishes and review physiological processes of these lunar-related reproductive rhythms. Based on recent information regarding the molecular mechanisms of circadian clocks, we propose a possible role of lunar-related clocks in the exertion of reproductive activities in fish.
2 Moon-Related Cycles in Fish Reproduction
2.1 Lunar Spawning Cycle
This lunar-related periodicity is characterized by a relationship between a trigger point of a reproductive event and a lunar phase and can be observed in various reproductive events of teleost fishes. For example, the downstream migration of the European eel Anguilla anguilla toward their spawning area, the Sargasso Sea, is initiated between the last quarter and new moon period (Miyai et al. 2004). In the North Equatorial Current to the west of the Mariana Islands, searches for hatched larvae of the Japanese eel Anguilla japonica by research cruises have suggested the possibility of synchronous spawning around the new moon period (Tsukamoto 2006). Cues peaking around the new moon period appear to trigger reproductive activities in eels. Lunar-related spawning migration has also been reported in certain groupers inhabiting coral reefs. Groupers aggregate at species-selective spawning sites and spawn around a peak of the lunar period; the honeycomb grouper Epinephelus merra spawns around the full moon period (Lee et al. 2002), whereas the coral trout Plectropomus leopardus releases its gametes around the new moon period (Samoilys and Squire 1994). Following a synchronous spawning event, matured groupers return from the spawning site to the inner reef that they most commonly inhabit. This coming-and-going behavior around a specific lunar phase is repeated during the spawning season (Samoilys and Squire 1994), which occurs annually at a fixed period of a year. Therefore, cues peaking toward a specific lunar phase may be transduced as internal stimuli and may consequently trigger lunar-related reproductive activities.
Comparative studies on lunar-related spawning rhythmicity have been conducted in four spinefoot species (formerly known as rabbitfish) inhabiting Okinawan waters, Japan, in the subtropical zone (Takemura et al. 2010). The white-spotted spinefoot Siganus canaliculatus spawns around the new moon from April to June, the little spinefoot Siganus spinus spawns around the new moon from May to July, the goldlined spinefoot Siganus guttatus spawns around the first quarter moon from June to July, and the streamlined spinefoot Siganus argenteus spawns around the last quarter moon from May to July (Takemura et al. 2010). The spawning rhythmicity of the same four species distributed in the tropical zone has also been examined. Results of these studies have indicated that the goldlined spinefoot spawns around the first quarter moon both from March to May and from September to November in waters around the Karimunjawa Islands, Indonesia (Susilo et al. 2009), and the little spinefoot spawns around the new moon both from January to March and from July to September in Chuuk Lagoon, Micronesia (Park et al. 2006). Although the spawning season of each species differs among habitats with different backgrounds of seasonal cues, exactly the same lunar phase is utilized for the same species in all regions of the Indo-Pacific Ocean. Notably, differences in the spawning lunar phase among these spinefoot species partially suggest the evolutionary acquisition of chronologically based reproductive isolation (Takemura et al. 2010).
Lunar cyclic ovarian development has been histologically examined in certain spinefoot species. In all cases reported so far, a clutch of oocytes in an ovary synchronously develops from the immature to mature stage toward a species-selective lunar phase. In the case of the goldlined spinefoot, for example, oocytes at vitellogenic stages appear among immature oocytes around the full moon period and develop fully around the new moon period. Following final oocyte maturation, ovulation and release of gametes from the maternal body occur around the first quarter moon (Fig. 9.1a) (Rahman et al. 2000). When weekly changes in plasma steroid hormone levels were measured, we found lunar patterns in estradiol-17β (E2) and 17α,20β-dihydroxy-4-pregnen-3-one (DHP), which are responsible for induction of vitellogenesis and final oocyte maturation, respectively (Nagahama 1994). Both steroid hormones increased toward the first quarter moon period. More precise experiments using an in vitro culture technique showed that under the presence of human chorionic gonadotropin (hCG) used as a stimulator of steroidogenesis, synthesis of E2 and DHP in ovarian segments increased around the new moon period (1 week before spawning) and the first quarter moon period (just before spawning), respectively (Fig. 9.2) (Rahman et al. 2002). Therefore, profiles of these steroid hormones are clearly in accordance with sequenced events in the ovary based on lunar phases. Biological characterizations of fish with lunar reproductive cycles have shown that an entire cycle in relationship to ovarian development and spawning proceeds to a species-selective lunar phase and is completed within 1 lunar month.
Schema of ovarian development according to lunar spawning cycle in the goldlined spinefoot (a) and semilunar spawning cycle in the brackish damsel (b). The lunar spawning cycle completes the whole processes of ovarian development within a month and spawning at 1-month intervals. In the semilunar spawning cycle, a clutch of oocytes develops from vitellogenic oocytes and spawning is repeated at intervals of 2 weeks. FM full moon, FQM first quarter moon, LQM last quarter moon, NM new moon, NT neap tide, ST spring tide. (Redrawn from Takemura et al. 2010)
In vitro production of steroid hormones by intact follicles of oocytes of the goldlined spinefoot in response to different concentrations of human chorionic gonadotropin (hCG). Estradiol-17β (E2) production at (a) 1 week before spawning (NM new moon) and (b) just before spawning (FQM first quarter moon). 17α,20β-Dihydroxy-4-pregnen-3-one (DHP) production at (c) 1 week before spawning (NM new moon) and (d) just before spawning (FQM first quarter moon). Asterisks indicate significant difference (P < 0.05) in E2 and DHP productions between the new moon and the first quarter moon. (Modified from Rahman et al. 2002)
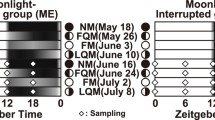
How do fish recognize a species-selective moon phase for their spawning rhythmicity? Because the goldlined spinefoot repeats spawning at the selective moon phase (the first quarter moon), even in tanks without tidal changes for several years (Rahman et al. 2000), this species likely does not require tidal stimuli for the continuity of the lunar-related spawning cycle. The fish may be able to perceive and utilize dynamic changes such as moonlight illumination (Horning and Trillmich 1999) and geomagnetic fields (Stolov 1965; Bell and Defouw 1966), which peak around the full moon and the last quarter moon, respectively, at 1-month intervals (Fig. 9.3). When the goldlined spinefoot is reared in tanks under artificial constant full moon and new moon conditions, the expected spawning during the spawning season is disrupted or delayed (Takemura et al. 2004). This observation indicates that cyclic changes in moonlight intensity play at least a partial role in the synchronization of the timing of spawning in this species. In contrast, a 4-year tagging study on the migration behavior of smolts of the chinook salmon Oncorhynchus tshawytscha demonstrated that the timing of saltwater entry from a river was closely related to the date of lunar apogee (the farthest distance from the earth to the moon), followed by the date of quarter moons (Fig. 9.3) (DeVries et al. 2004). Salmonid fish may thus perceive changes in lunar gravitation for the start of migration.
Lunar effects on the Earth. Moonlight illumination and geomagnetic field fluctuate at 1-month intervals and peak around the full moon and the last quarter moon, respectively. Gravitational pull and tidal amplitude fluctuate at a 14.7-day interval and peak around the full moon and the new moon. FM full moon, FQM first quarter moon, LQM last quarter moon, NM new moon
2.2 Semilunar Spawning Cycle
Semilunar reproductive events are explained as apparent spawning cycles at a 14.7-day interval at a selective lunar phase; these cycles have been observed in many fish species that, in most cases, inhabit coastal areas. For example, the grass puffer Takifugu niphobles, a common coastal puffer species in Japan, aggregates at certain seashore locations and spawns several hours before the morning or evening high tide, only during the spring tide (Yamahira 1994, 2004). The group spawning of the grass puffer occurs at the same location every year and every 2 weeks during a spawning season (Motohashi et al. 2010). Similar spawning behavior at spring tide has been reported in the California grunion Leuresthes tenuis (Clark 1925) and the killifishes (Fundulus heteroclitus or F. grandis) (Hsiao and Meier 1989). In these cases, the fertilized eggs or hatched larvae are washed out from the coast to the open sea until the high tide of the next spring tide. One advantage of this reproductive strategy is that it appears to reduce predation risk by aquatic predators for offspring (Leatherland et al. 1992).
Details on ovarian development in terms of semilunar spawning cycles have been reported in two species, the brackish damsel Pomacentrus taeniometopon and the amboina cardinalfish Apogon amboinensis. Both are brackish species exposed to vigorous changes in tidal stimuli. Females of the brackish damsel spawn around the first quarter moon and the last quarter moon (Fig. 9.1b). Because territorial males protect fertilized eggs for 1 week, hatching occurs around the new moon and full moon periods (Pisingan et al. 2006). In the brackish damsel, vitellogenic oocytes can always be found in an ovary during the spawning season. A clutch of vitellogenic oocytes starts to mature and is spawned around both quarter moons. Females of the amboina cardinalfish spawn around the first quarter moon and the last quarter moon period. Because this species is a mouth-brooder, the fertilized eggs are incubated in the mouth of males for 2 weeks until hatching. The release of hatched larvae from the male mouth occurs around the first quarter moon and the last quarter moon period (Pisingan and Takemura 2007). In the amboina cardinalfish, a clutch of yolk-laden oocytes appears among immature oocytes after spawning and then develop synchronously toward the first or last quarter moon. When ovarian segments of the amboina cardinalfish were cultured in medium with hCG, the synthesis of E2 increased around the new and full moon periods, whereas that of DHP increased around the first and last quarter moon (Fig. 9.4a, b). These studies indicated at least two types of semilunar spawners, which may be categorized as the true or apparent spawner. The former (e.g., brackish damsel) repeats oocyte development and spawning at an interval of 2 weeks; thus, an individual experiences two spawnings within a month during the spawning season (true semilunar spawner). On the other hand, the latter (e.g., amboina cardinalfish) undergoes one-time oocyte development and spawning within a month. In this case, therefore, there may be two populations spawning around the first quarter moon or last quarter moon period (Takemura et al. 2010).
Comparisons of (a) estradiol-17β (E2) and (b) 17α,20β-dihydroxy-4-pregnen-3-one (DHP) from the cultured ovarian segments of the amboina cardinalfish in response to human chorionic gonadotropin (hCG) between the new and full moon (NM and FM) and the first and last quarter moon (FQM and LQM). E2 and DHP were measured using ELISA. Asterisks indicate significant difference (P < 0.05) between the new and full moon (NM and FM, open columns) and the first and last quarter moon (FQM and LQM, filled columns). (Modified from Pisingan and Takemura 2007)
Because the timing of larval dispersal from the male’s territory or mouth is correlated with the peak of spring tides (Fig. 9.3), tidal stimuli appear to be perceived by fish species to synchronize gonadal development and spawning. Notably, females of the red-clawed crab Chiromantes haematocheir repeat the release of zoea larvae to the sea during high tide around the spring tide. This semilunar spawning rhythm has been suggested to be induced not only by cyclic changes in tides but also by changes in moonlight illumination (Saigusa 1988). Therefore, the semilunar rhythmicity of organisms is likely affected by a conjugated system of cues from the moon. Robertson et al. (1990) hypothesized that the release of hatched larvae with the spring tide is likely related to the rapid dispersal of the offspring from the coast to the open sea, consequently reducing predation pressure and enhancing the survival of the offspring (Robertson et al. 1990).
Hsiao and Meier (1989) collected the gulf killifish F. grandis and the mummichog F. heteroclitus from the Gulf Coast and the Atlantic Coast, respectively, and reared them under standard laboratory conditions. They found that several spawning cycles were maintained at a mean duration of 13.7 days for the gulf killifish and 14.8 days for the mummichog, and the spawning cycle of these two species was synchronized with the moonlight and tidal cycle in their respective habitats (Hsiao and Meier 1989). Motohashi et al. (2010) observed the aggregating and spawning behaviors of the grass puffer in an aquarium lacking tidal changes. Although spawning was not confirmed in the aquarium mimicking field conditions, the small group of mature fish only aggregated repeatedly in the rising tidal phase during and after the spring tides (Motohashi et al. 2010). Furthermore, in the mudskipper Boleophthalmus pectinirostris, the synthesis of plasma steroid hormones (i.e., testosterone, E2, and 17α-hydroxyprogesterone) was regulated by a biological clock; these steroids peaked twice on the third day after the first quarter moon and on the fourth day after the last quarter moon, although steroid hormone profiles and gonadal development were not positively correlated (Wang et al. 2008). These findings suggest that an endogenous clock controls spawning periodicity and that this clock can oscillate temporarily without external moon-related stimuli. Interestingly, mummichogs (the Arasaki strain) reared for generations under laboratory conditions became daily spawners during the spawning season (Shimizu 1997). Exposing the fish to moon-related stimuli at specific stages of their life cycle may be necessary to exert the semilunar spawning rhythmicity.
3 Melatonin as a Possible Transducer in Lunar Spawners
Melatonin is an indoleamine hormone synthesized mainly in the pineal organ and the retina (Zachmann et al. 1992). This hormone is believed to be a transmitter of photoperiodic information to central and peripheral organs, as its level in the blood increases during the nighttime and decreases during the daytime (Bromage et al. 2001). The pineal organ of teleost fishes serves a dual biological function as a neuroendocrine organ and photoreceptive organ. With the exception of salmonid species, the pineal organ of most fish species can release melatonin in a circadian manner under constant dark conditions of in vivo and in vitro experiments, implying that the entire system of the circadian clock functions in the pineal organ (Iigo et al. 1991, 2004; Molina-Borja et al. 1996).
A daytime-low and nighttime-high fluctuation persisted in plasma melatonin levels under light–dark (LD) conditions in the goldlined spinefoot (Takemura et al. 2004), suggesting that melatonin also serves a common role in transducing daily variation in environmental photoperiodic information into the central and peripheral organs in this lunar spawner. Moreover, nocturnal plasma melatonin levels during the new moon period were higher than those during the full moon period (Fig. 9.5a) (Takemura et al. 2004). This result was supported by in vitro experiments; when the pineal organ of the goldlined spinefoot was cultured under natural illumination during the full and new moon periods, secretion of melatonin into the culture medium was higher during the new moon period than during the full moon period (Takemura et al. 2006). In addition, exposing spinefoots to “brightness at the full moon or new moon night” resulted in a rapid suppression of plasma melatonin levels (Rahman et al. 2004; Takemura et al. 2004). Therefore, the melatonin profile in a lunar spawner would likely show a composite pattern at 1-month intervals: the amplitude in melatonin levels between daytime and nighttime during the new moon period would be large, whereas that during the full moon period would be small.
Comparisons of melatonin concentration in the plasma (a) and clock gene expression in the pineal organ (b) of the goldlined spinefoot between the new moon (NM) and the full moon (FM). The plasma sample and the pineal organ were taken from the fish at midnight during the new moon (filled columns) and full moon (open columns). Melatonin levels in the plasma were measured by time-resolved immunofluoroassay. Expression of Period2 (SgPer2) mRNA in the pineal organ was determined by quantitative real-time polymerase chain reaction (PCR) and normalized by the amount of Sgβ-actin mRNA. Each bar represents mean ± SE. Asterisk indicates significant difference (P < 0.05) between the new moon and the full moon. (Modified from Takemura et al. 2004 and Sugama et al. 2008)
The actions of melatonin are mediated via melatonin receptors belonging to the G protein-coupled receptor superfamily. Melatonin receptors are also expected to be involved in the exertion of lunar rhythmicity in fish. Because three subtypes of melatonin receptors are known in lower vertebrates, the MT1 (formerly known as Mel1a), MT2 (formerly known as Mel1b), and Mel1c (Ikegami et al. 2009a, b), the cDNAs of MT1 and Mel1c have been cloned in the goldlined spinefoot and their expression patterns in the pineal organ have been examined using quantitative real-time polymerase chain reaction (qPCR) (Park et al. 2007a, b). The mRNA abundance of MT1 and Mel1c exhibited diurnal variation, with an increase during nighttime and a decrease during daytime under light and dark conditions (Park et al. 2007a, b). An in vitro experiment using the goldlined spinefoot pineal organ revealed that melatonin production is strongly correlated with melatonin receptor mRNA expression (Park et al. 2007a), suggesting that melatonin regulates the expression of these melatonin receptors. These results raise the possibility that melatonin receptors fluctuate according to changes in the intensity of moonlight illumination under regulation of melatonin. In fact, MT1 and Mel1c mRNA abundance was higher at midnight of the new moon than midnight of the full moon (Park et al. 2014). As demonstrated with plasma melatonin levels, exposing fish to “brightness at the full moon night” resulted in downregulation of Mel1c mRNA in the pineal organ (Park et al. 2014). Therefore, the melatonin–melatonin receptor system is potentially involved in generating 1-month periodicity in certain lunar synchronized spawners.
Melatonin likely becomes a useful messenger of various lunar-related environmental changes (Takemura et al. 2010). In fact, a magnetic field generated by Helmholtz coils (maximum flux density 40 mT, frequency 1 Hz, 200 ms on, 800 ms off) resulted in increases in nocturnal pineal and serum melatonin levels of brook trout Salvelinus fontinalis (Lerchl et al. 1998). This stimulatory effect of a magnetic field on melatonin production may be attributable to an increased Ca2+ influx into pineal photoreceptors (direct effect; Gasser and Gern 1997) or to secondary stress responses (indirect effect; Bullock 1977). Wagner et al. (2007) cultured the pineal organ of two abyssal fish species, Coryphaenoides armatus and Synaphobranchus kaupii, whose habitats in deep seas experience rhythmic changes in current velocity and direction at intervals of 12.4 h. Melatonin release tended to increase at the beginning of the lunar day and night and was lower during the second half of the lunar day and night and during moonrise and moonset. Thus, tidal currents may act as a zeitgeber at the bottom of the deep sea, and melatonin metabolism with tidal periodicity may be a possible sign of the transduction of tidal periodicity in deep-sea fishes (Wagner et al. 2007).
4 Clock Genes in Lunar Rhythmicity
As noted, circumstantial evidence exists that endogenous clocks are involved in the semilunar reproductive cycle in certain teleost fishes. However, how circasemilunar and circalunar cycles are regulated endogenously and what types of biological clocks are involved in oscillating such cycles remain unclear. A pioneer study on the involvement of clock genes in lunar spawning synchrony was performed in the reef-building coral Acropora millepora, which spawns around the full moon period (Levy et al. 2007). The mRNA abundance of light-responsible genes, Cryptochrome (Cry1 and Cry2), exhibited daily variation with a peak during daytime under light–dark cycles, and Cry2 mRNA abundance at night was higher during the full moon period than during the new moon. These results suggest that an element of the circadian system is involved in the lunar-related mass spawning in corals.
Initially, Period and Cryptochrome received the most attention in terms of the genetics of lunar clocks in fish because the family of these elements is believed to include photo-responsible genes in lower vertebrates (Cahill 2002; Yasuo et al. 2004). In the goldlined spinefoot, following the cloning of cDNA, the expression patterns of Period (SgPer2) and Cryptochrome (SgCry1 and SgCry3) mRNAs were examined in the pineal organ and the medial part of the brain (mesencephalon and diencephalon), respectively (Fukushiro et al. 2011; Sugama et al. 2008). The abundance of SgPer2 mRNA displayed a daily expression pattern with increases during photophase. The abundance of this gene was higher at the culmination of the full moon period than during the new moon period (Fig. 9.5b). Additionally, exposing the fish to moonlight irradiation around the full moon period resulted in upregulation of SgPer2 mRNA abundance, suggesting that the expression of SgPer2 mRNA in the pineal organ was affected not only by daylight, but also by moonlight (Sugama et al. 2008). On the other hand, the mRNA abundance of SgCry1, but not SgCry3, exhibited daily fluctuations, with a peak at dawn and gradual decreases during daytime. Notably, the expression of both genes showed lunar-dependent variation; their abundance increased around the first quarter moon and decreased around the full moon and the last quarter moon period (Fig. 9.6) (Fukushiro et al. 2011). Thereby, an increase in SgCry mRNA abundance occurred around the spawning lunar phase of the goldlined spinefoot.
Lunar-dependent variations in the expression of genes encoding cryptochrome, SgCry1 (a), and SgCry3 (b) in the brain of the goldlined spinefoot. Each Cry mRNA was calculated as a value relative to that of Sgβ-actin gene. Each bar represents mean ± SD. Lunar phases are indicated above the graph by schematic moon images. Probability values (P) of ANOVA for statistically significant difference among dates are indicated. Second peak of mRNA expressions of SgCry1 and SgCry3 coincided with spawning day (June 29) whereas the first peak of those existed before spawning season. (Modified from Fukushiro et al. 2011)
Despite the lack of information for characterizing the fish lunar clock system in detail, further research should address hypotheses that test whether certain elements of the circadian system are involved in oscillations of lunar-dependent rhythmicity and if periodic changes in moonlight irradiation act as a possible zeitgeber to synchronize this rhythm. Differences in moonlight irradiation between the new moon and full moon period may simply affect the expression patterns of clock genes. Alternatively, lunar phase-dependent gene expression is presumably controlled by moonlight signals around a putative “photo-responsible phase,” which is expected to appear at a certain point of the night (ZT18‒ZT21). This hypothesis was derived from comparisons between a transient pattern of moonlight irradiation at night (Fig. 9.7) and weekly changes in SgCry mRNA expression.
Schematic pattern of solar light phase, moonlight phase, and moonlight intensity in Okinawa, Japan (26°38′N, 127°51′E) during the spawning season (June) of the goldlined spinefoot. A putative photo-responsible phase is indicated by the white outline. Shading indicates differences in light intensity. (Modified from Fukushiro et al. 2011)
5 Closing Remarks
The purpose of this chapter was to provide an outline of the physiological and molecular mechanisms influencing lunar and semilunar rhythmicity in fish reproduction. To date, whether lunar- or semilunar-specific genes are expressed in the central nervous system and are involved in regulating lunar-related rhythms in fish remains unknown. However, the findings detailed in this chapter reveal that the main players, melatonin and clock genes, of the circadian system are associated with the entrainment of fish reproduction to a peak point of cues from the moon. Lunar and semilunar rhythmicity in fish has presumably evolved as an applicable form of the circadian system, particularly if fish share a common transduction system for sensing photic information such as daylight as well as moonlight. Alternatively, the lunar/semilunar rhythmicity is a potentially innate characteristic in most fish but one that oscillates robustly in only certain fish species after adaptation to aquatic environments. If so, it is not necessary for fish to evolve a novel system for lunar/semilunar rhythmicity.
The genome project of the tiger puffer Takifugu rubripes was completed in 2002 (Aparicio et al. 2002). Because Takifugu species share very high similarity in their genome sequences (Yamanoue et al. 2009), the grass puffer offers a particular advantage for elucidating the molecular mechanisms of the semilunar cycle using the genome resources of the tiger puffer. Despite vigorous accumulation of ecological evidence, physiological and molecular studies on lunar and semilunar cycles in fish have been limited. Advances in genome-wide search might allow us to identify novel clock genes in relationship to the circalunar/circasemilunar cycle or to find new functions of known molecules in oscillating lunar/semilunar cycles. By capitalizing upon new approaches, further studies can help to achieve an overall understanding of lunar and semilunar cycles in fish.
References
Aparicio S, Chapman J, Stupka E et al (2002) Whole-genome shotgun assembly and analysis of the genome of Fugu rubripes. Science 297:1301–1310
Bell B, Defouw RJ (1966) Dependence of the lunar modulation of geomagnetic activity on the celestial latitude of the moon. J Geophys Res 71:951–957
Bromage N, Porter M, Randall C (2001) The environmental regulation of maturation in farmed finfish with special reference to the role of photoperiod and melatonin. Aquaculture 197:63–98
Bullock TH (1977) Electromagnetic sensing in fish. Neurosci Res Prog Bull 15:17–22
Cahill GM (2002) Clock mechanisms in zebrafish. Cell Tissue Res 309:27–34
Clark FN (1925) The life history of Leuresthes tenuis, an atherine fish with tide controlled spawning habits. Fish Bull 10:1–51
DeVries P, Goetz F, Fresh K, Seiler D (2004) Evidence of a lunar gravitation cue on timing of estuarine entry by Pacific salmon smolts. Trans Am Fish Soc 133:1379–1395
Fukushiro M, Takeuchi T, Takeuchi Y, Hur S-P, Sugama N, Takemura A, Kubo Y, Okano K, Okano T (2011) Lunar phase-dependent expression of cryptochrome and a photoperiodic mechanism for lunar phase-recognition in a reef fish, goldlined spinefoot. PLoS One 6:e28643
Gasser PJ, Gern WA (1997) Regulation of melatonin synthesis in rainbow trout (Oncorhyncus mykiss) pineal organs: effects of calcium depletion and calcium channel drugs. Gen Comp Endocrinol 105:210–217
Harrison PL, Babcock RC, Bull GD, Oliver JK, Wallace CC, Willis BL (1984) Mass spawning in tropical reef corals. Science 223:1186–1189
Horning M, Trillmich F (1999) Lunar cycles in diel prey migrations exert a stronger effect on the diving of juveniles than adult Galápagos fur seals. Proc Biol Soc Wash 266:1127–1132
Hsiao S-M, Meier AH (1989) Comparison of semilunar cycles of spawning activity in Fundulus grandis and F. heteroclitus held under constant laboratory conditions. J Exp Zool 252:213–218
Iigo M, Kezuka H, Aida K, Hanyu I (1991) Circadian rhythms of melatonin secretion from superfused goldfish (Carassius auratus) pineal glands in vitro. Gen Comp Endocrinol 83:152–158
Iigo M, Fujimoto Y, Gunji-Suzuki M, Yokosuka M, Hara M, Ohtani-Kaneko R, Tabata M (2004) Circadian rhythm of melatonin release from the photoreceptive pineal organ of a teleost, ayu (Plecoglossus altivelis) in flow-through culture. J Neuroendocrinol 16:45–51
Ikegami T, Azuma K, Nakamura M, Suzuki N, Hattori A, Ando H (2009a) Diurnal expressions of four subtypes of melatonin receptor genes in the optic tectum and retina of goldfish. Comp Biochem Physiol A 152:219–224
Ikegami T, Motohashi E, Doi H, Hattori A, Ando H (2009b) Synchronized diurnal and circadian expressions of four subtypes of melatonin receptor genes in the diencephalon of a puffer fish with lunar-related spawning cycles. Neurosci Lett 462:58–63
Leatherland JF, Farbridge KJ, Boujard T (1992) Lunar and semi-lunar rhythms in fishes. In: Ali MA (ed) Rhythms in fishes. Plenum Press, New York
Lee YD, Park SH, Takemura A, Takano K (2002) Histological observations of seasonal reproductive and lunar-related spawning cycles in the female honeycomb grouper Epinephelus merra in Okinawan waters. Fish Sci 68:872–877
Lerchl A, Zachmann A, Ali MA, Reiter RJ (1998) The effects of pulsing magnetic fields on pineal melatonin synthesis in a teleost fish (brook trout, Salvelinus fontinalis). Neurosci Lett 256:171–173
Levy O, Appelbaum L, Leggat W, Gothlif Y, Hayward DC, Miller DJ, Hoegh-Guldberg O (2007) Light-responsive cryptochromes from a simple multicellular animal, the coral Acropora millepora. Science 318:467–470
Miyai T, Aoyama J, Sasai S, Inoue JG, Miller MJ, Tsukamoto K (2004) Ecological aspects of the downstream migration of introduced European eels in the Uono River, Japan. Environ Biol Fish 71:105–114
Molina-Borja M, Falcón J, Urquinola E, Ravault JP (1996) Production of melatonin by the gilthead sea bream pineal: an in vivo and in vitro study. Fish Physiol Biochem 15:413–419
Motohashi E, Yoshihara T, Doi H, Ando H (2010) Aggregating behavior of the grass puffer, Takifugu niphobles, observed in aquarium during the spawning period. Zool Sci 27:559–564
Nagahama Y (1994) Endocrine regulation of gametogenesis in fish. Int J Dev Biol 38:217–229
Panda S, Hogenesch JB, Kay SA (2002) Circadian rhythms from flies to human. Nature (Lond) 417:329–335
Park YJ, Takemura A, Lee YD (2006) Annual and lunar-synchronized ovarian activity in two rabbitfish species in the Chuuk lagoon, Micronesia. Fish Sci 72:166–172
Park YJ, Park JG, Hiyakawa N, Lee YD, Kim SJ, Takemura A (2007a) Diurnal and circadian regulation of a melatonin receptor, MT1, in the golden rabbitfish, Siganus guttatus. Gen Comp Endocrinol 150:253–262
Park YJ, Park JG, Jeong HB, Takeuchi Y, Kim SJ, Lee YD, Takemura A (2007b) Expression of the melatonin receptor Mel1c in neural tissues of the reef fish Siganus guttatus. Comp Biochem Physiol A 147:103–111
Park YJ, Park JG, Takeuchi Y, Hur SP, Lee YD, Takemura A (2014) Influence of moonlight on mRNA expression patterns of melatonin receptor subtypes in the pineal organ of a tropical fish. Mar Genomics 14:67–70
Pisingan RS, Takemura A (2007) Apparent semi-lunar spawning rhythmicity in a brackish cardinalfish. J Fish Biol 70:1512–1522
Pisingan RS, Harnadi L, Takemura A (2006) Semilunar spawning periodicity in brackish damsel Pomacentrus taeniometopon. Fish Sci 72:1256–1260
Pittendrigh CS (1993) Temporal organization: reflections of a Darwinian clock-watcher. Annu Rev Physiol 55:17–54
Rahman MD, Takemura A, Takano K (2000) Correlation between plasma steroid hormones and vitellogenin profiles and lunar periodicity in the female golden rabbitfish, Siganus guttatus (Bloch). Comp Biochem Physiol B 127:113–122
Rahman MS, Takemura A, Takano K (2002) Lunar synchronization of in vitro steroidogenesis in ovaries of the golden rabbitfish, Siganus guttatus (Bloch). Gen Comp Endocrinol 125:1–8
Rahman MS, Kim BH, Takemura A, Park CB, Lee YD (2004) Effects of moonlight exposure on plasma melatonin rhythms in the seagrass rabbitfish, Siganus canaliculatus. J Biol Rhythm 19:325–334
Robertson DR, Petersen CW, Brawn JD (1990) Lunar reproductive cycles of benthic-brooding reef fishes: reflections of larval biology or adult biology? Ecol Monogr 60:311–329
Saigusa M (1988) Entrainment of tidal and semilunar rhythms by artificial moonlight cycles. Biol Bull 174:126–138
Samoilys MA, Squire LC (1994) Preliminary observations on the spawning behavior of coral trout, Plectropomus Leopardus (Pisces: Serranidae), on the Great Barrier Reef. Bull Mar Sci 54:332–342
Shimizu A (1997) Reproductive cycles in a reared strain of the mummichog, a daily spawner. J Fish Biol 51:724–737
Stolov HL (1965) Further investigations of a variation of geomagnetic activity with lunar phase. J Geophys Res 70:4921–4926
Sugama N, Park JG, Park YJ, Takeuchi Y, Kim SJ, Takemura A (2008) Moonlight affects nocturnal Period2 transcript levels in the pineal gland of the reef fish Siganus guttatus. J Pineal Res 45:133–141
Susilo ES, Harnadi L, Takemura A (2009) Tropical monsoon environments and the reproductive cycle of the orange-spotted spinefoot Siganus guttatus. Mar Biol Res 5:179–185
Takemura A, Susilo ES, Rahman MDS, Morita M (2004) Perception and possible utilization of moonlight intensity for reproductive activities in a lunar-synchronized spawner, the golden rabbitfish. J Exp Zool 301A:844–851
Takemura A, Ueda S, Hiyakawa N, Nikaido Y (2006) A direct influence of moonlight intensity on changes in melatonin production by cultured pineal glands of the golden rabbitfish, Siganus guttatus. J Pineal Res 40:236–241
Takemura A, Rahman MS, Park YJ (2010) External and internal controls of lunar-related reproductive rhythms in fishes. J Fish Biol 76:7–26
Thresher RE (1984) Coral reef fishes: reproduction. T.F.H Publications, Hong Kong/Neptune City
Tsukamoto K (2006) Oceanic biology: spawning of eels near a seamount. Nature (Lond) 439:929
Wagner HJ, Kemp K, Mattheus U, Priede IG (2007) Rhythms at the bottom of the deep sea: cyclic current flow changes and melatonin patterns in two species of demersal fish. Deep Sea Res Pt I 54:1944–1956
Wang Q, Hong W, Chen S, Zhang Q (2008) Variation with semilunar periodicity of plasma steroid hormone production in the mudskipper Boleophthalmus pectinirostris. Gen Comp Endocrinol 155:821–826
Yamahira K (1994) Combined effects of tidal and diurnal cycles on spawning of the puffer, Takifugu niphobles (Tetraodontidae). Environ Biol Fish 40:255–261
Yamahira K (2004) How do multiple environmental cycles in combination determine reproductive timing in marine organisms? A model and test. Funct Ecol 18:4–15
Yamanoue Y, Miya M, Matsuura K, Miyazawa S, Tsukamoto N, Doi H, Takahashi H, Mabuchi K, Nishida M, Sakai H (2009) Explosive speciation of Takifugu: another use of fugu as a model system for evolutionary biology. Mol Biol Evol 26:623–629
Yasuo S, Watanabe M, Tsukada A, Takagi T, Iigo M, Shimada K, Ebihara S, Yoshimura T (2004) Photoinducible phase-specific light induction of Cry1 gene in the pars tuberalis of Japanese quail. Endocrinology 145:1612–1616
Zachmann A, Ali MA, Falcón J (1992) Melatonin and its effects in fishes. In: Ali MA (ed) Rhythms in fishes. Plenum Press, New York
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer Japan
About this chapter
Cite this chapter
Ikegami, T., Takeuchi, Y., Takemura, A. (2014). Lunar Clock in Fish Reproduction. In: Numata, H., Helm, B. (eds) Annual, Lunar, and Tidal Clocks. Springer, Tokyo. https://doi.org/10.1007/978-4-431-55261-1_9
Download citation
DOI: https://doi.org/10.1007/978-4-431-55261-1_9
Published:
Publisher Name: Springer, Tokyo
Print ISBN: 978-4-431-55260-4
Online ISBN: 978-4-431-55261-1
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)