Abstract
Photoreceptor cells are sensory neurons and does not possess dendrites, whereas photoreceptor axonal terminals develop a specialized synaptic structure called ribbon synapses that contains electro-dense horseshoe-like ribbons. Photoreceptor ribbon synapses have connections with dendritic terminals of bipolar cells and tips of horizontal cell processes, and therefore are critical for visual transduction. In both humans and genetically modified mice, loss of function in photoreceptor synaptic components and its regulators often causes visual impairment and retinal diseases including retinitis pigmentosa and night blindness. In this chapter, we review and discuss the development of photoreceptor ribbon synapses and the functions of ribbon synapse key components.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Bipolar cell dendrite
- Cabp4
- CASK
- Crx
- Dystroglycan
- Dystrophin
- ERG
- Horizontal cell process
- L-type Ca2+ channel
- Night blindness
- Nrl
- Photoreceptor axonal terminal
- Pikachurin
- Retinitis pigmentosa
- Ribbon synapses
8.1 Synaptic Connection of Photoreceptor Cells with Other Neurons in the Retina
In the vertebrate retina, synaptic connections are formed between five different types of neurons, including photoreceptor, bipolar, horizontal, amacrine, and ganglion cells. These synapses are organized into distinct laminae: the outer plexiform layer (OPL) and the inner plexiform layer (IPL) (Masland 2001). Photoreceptor axonal terminals contact both horizontal cell processes and bipolar cell dendrites in the OPL. Synaptic connections of bipolar cell axonal terminals and ganglion cell dendrites to amacrine cell processes are localized in the IPL. Rod photoreceptor cells connect with rod ON-bipolar cells and horizontal cells, whereas cone photoreceptor cells have connections with cone ON- and cone OFF bipolar cells as well as horizontal cells (Fig. 8.1).
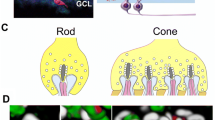
Rod and cone photoreceptor synapses in the outer plexiform layer (OPL). Rod photoreceptor axonal terminals connect with rod ON-bipolar cell dendrites and horizontal cell processes. Cone photoreceptor terminals connect with cone ON- and cone OFF-bipolar cell dendrites as well as horizontal cell processes. These synapses are located in the outer plexiform layer (OPL)
In the OPL, photoreceptors and bipolar cells transfer information at specialized synapses, the ribbon synapses, which transmit signals tonically and in a graded fashion (tom Dieck and Brandstatter 2006). In vertebrates, several types of neurons in sensory organs form ribbon synapses including retinal bipolar cells, photoreceptor-like neurons in the pineal grand, and auditory and vestibular hair cells (Sterling and Matthews 2005). Graded synaptic output in photoreceptors requires that a large number of synaptic vesicles be released. To accomplish this, photoreceptors possess large pools of regularly aligned, fast-release vesicles tethered to the synaptic ribbons (Sterling and Matthews 2005).
A synaptic ribbon is an electron-dense organelle in the photoreceptor presynaptic cytoplasm, observable by electron microscopy. A photoreceptor synaptic ribbon has a plate-like structure that anchors it to the presynaptic membrane of an axon terminal. The photoreceptor synaptic ribbons are associated with invaginations present along the base of the photoreceptor terminals (Fig. 8.2). Horizontal cell processes and bipolar cell dendrites are inserted into photoreceptor invaginations and terminate near the ridge in which the synaptic ribbon lies. The synaptic structures of rod and cone photoreceptors are different. The rod photoreceptor terminals contain a single ribbon that has a horseshoe-shaped structure (Rao-Mirotznik et al. 1995). Rod photoreceptor synapses are called spherules. The cone photoreceptor terminals are larger than those of rods and contain multiple smaller synaptic ribbons in a single cone photoreceptor axonal terminal. The cone photoreceptor synapses are called pedicles. In the vertebrate rod synaptic invaginations, the rod ON-bipolar dendrites terminate as the central element. In the cone synaptic invaginations, the cone ON-bipolar dendrites end as the central element. The dendrites of cone OFF-bipolar cells contact the basal surfaces of cone terminals. In the invaginations of both rod and cone terminals, horizontal cell processes end laterally and deeper, and synaptic ribbons are located at the apex of invaginations.
Structure of rod and cone photoreceptor synaptic terminals. The photoreceptor ribbons (green bars) and synaptic vesicles (yellow circles) are associated with invaginations present at the base of the rod (left panel) and cone (right panel) photoreceptor synapses. In both rod and cone photoreceptors, horizontal cell processes (blue) and ON-bipolar cell dendrites (red) penetrate into photoreceptor invaginations. OFF-bipolar dendrites (purple) contact cone photoreceptor terminals on the surface of the synaptic terminals. The rod photoreceptor terminals contain a single ribbon. The cone photoreceptor terminals contain multiple ribbons and more horizontal and bipolar connections than those of rod photoreceptor cells
8.2 Development of Photoreceptor Synapses
In vertebrates, retinal circuit development occurs in a sequential manner that is common across species (Olney 1968; Blanks et al. 1974; McLaughlin 1976; Fisher 1979; Schmitt and Dowling 1999). In the developing mouse retina, synaptic connections are mainly formed after birth (Blanks et al. 1974). Photoreceptor synaptic maturation occurs in three different stages of configuration termed apposition, dyad, and triad configurations (Fig. 8.3).
Developmental stages of photoreceptor synapses. Photoreceptor synaptic formation in the mouse retina occurs postnatally (Blanks et al. 1974). Mouse photoreceptor synaptic formation occurs in three different stages referred to as apposition, dyad, and triad configurations. Left panel: After P4, the synaptic processes from horizontal cells are juxtaposed with the membrane of the photoreceptor synaptic terminal, but have not yet penetrated into the photoreceptor synaptic terminal. Middle panel: At around P7, the dyad configuration in the photoreceptor synaptic terminal is formed with two lateral horizontal processes and a synaptic ribbon within the photoreceptor synaptic terminal. Right panel: Around P14, a bipolar central process enters the photoreceptor synaptic invagination and forms the triad configuration
8.2.1 Apposition Configuration (P4–P5)
Soon after birth in the mouse, retinal progenitor cells continue proliferating. The synaptic layer in the OPL is difficult to recognize at postnatal day 3 (P3). The first sign of synaptic formation in the OPL is observable at P4 by electron microscopy analysis. Immature photoreceptor axonal synaptic terminals contain clusters of developing ribbons. The synaptic processes from horizontal cells are juxtaposed with the membrane of the photoreceptor synaptic terminal in the OPL but have not yet penetrated into the photoreceptor synaptic terminals. The synaptic contacts of horizontal cell processes are located on the base of the photoreceptor terminal, usually near developing synaptic ribbons. This stage of photoreceptor synapse formation is referred to as apposition (Blanks et al. 1974). The photoreceptor dystrophin complex containing pikachurin and dystroglycan is a useful marker for synaptic connections between photoreceptor presynapses and bipolar dendritic postsynapses (Schmitz and Drenckhahn 1997; Sato et al. 2008; Omori et al. 2012). At this stage, weak pikachurin signals are observed in the OPL adjacent to immature CtBP2-positive ribbons. Although dystroglycan and dystrophin signals are less intense than the pikachurin signal, these signals are weakly concentrated at the OPL. This finding suggests that the photoreceptor presynaptic complex including dystrophin, dystroglycan, and pikachurin begins forming on the surface of synaptic terminals before the photoreceptor terminus is connected with bipolar dendrites.
8.2.2 Dyad Configuration (P6–P9)
After the formation of synaptic contacts with horizontal cell processes on the surface of photoreceptor axonal terminals, horizontal cell processes penetrate the invagination of the photoreceptor synaptic terminal lateral to a synaptic ribbon at around P7 (Blanks et al. 1974). The dyad configuration is two lateral horizontal cell processes aligned with a synaptic ribbon within the photoreceptor synaptic terminal. At this stage, bipolar processes have not yet penetrated; however, strong dystroglycan and pikachurin signals are observed adjacent to the synaptic ribbon, suggesting that pikachurin, dystroglycan, and dystrophin form a complex in the photoreceptor synaptic terminals before the insertion of bipolar dendrites. At this stage, the complexity of the OPL increases, and a row of photoreceptor terminals with several synaptic dyads as well as a layer of neuropil with horizontal cell processes and bipolar cell dendrites is formed.
8.2.3 Triad Configuration (After P10)
Around P10, a bipolar central process enters the photoreceptor synaptic invagination to yield a triad configuration, which is a synaptic ribbon with two lateral processes from horizontal cells and with at least one central dendrite from bipolar cells in a rod photoreceptor synaptic terminal. The formation of synaptic triads at photoreceptor synaptic terminals is complete by P14. At this stage, photoreceptor synapses with dystrophin, dystroglycan, and pikachurin puncta are formed in the vicinity of a horseshoe-like ribbon in the OPL of adult mice.
8.3 Components of Synaptic Ribbons and Ribbon-Associated Proteins
In the presynaptic region of conventional synapses, neurotransmitter release is restricted to an electron-dense cytoskeletal meshwork, the cytomatrix at the active zone (CAZ), usually observable by electron microscopy. The photoreceptor synaptic ribbon represents a specialization of the CAZ of conventional synapses (tom Dieck et al. 2005) (Fig. 8.4). Presynaptic CAZ is situated opposite the postsynaptic neurotransmitter reception regions, an electron-dense cytoskeletal matrix called the postsynaptic density (Ziff 1997; Dresbach et al. 2001). At photoreceptor ribbons, CAZ proteins segregate into two compartments. The first compartment associated with the ribbon contains Piccolo, Ctbp1, Ctbp2, RIM1, and KIF3A. The plasma membrane-associated second compartment contains RIM2, Munc13-1, a Ca2+ channel α1-subunit, and CAST/ERC2. Ctbp2 and Bassoon interact directly and link these two compartments. These proteins are important for signal transmission between photoreceptor and bipolar cells.
Schematic diagram of photoreceptor ribbon compartments. Localization of CAZ proteins defines two compartments of the ribbon synaptic complex in photoreceptor synaptic terminals. The ribbon-associated compartment includes Ctbp2, Ctbp1, KIF3A, Piccolo, and RIM1. The plasma membrane-associated compartment includes CAST and an L-type Ca2+ channel α1-subunit. Bassoon localizes at the border and connects these two compartments. (Diagram modified from tom Dieck et al. 2005)
8.3.1 Bassoon
Bassoon is a presynaptic protein and a major component of the photoreceptor ribbon. Bassoon is an approximately 4,000-amino-acid protein containing a zinc finger motif similar to another ribbon component, Piccolo (tom Dieck et al. 1998). In Bassoon-deficient photoreceptor cells, synaptic ribbons are not anchored to the presynaptic active zones, resulting in impaired photoreceptor synaptic transmission (Dick et al. 2003). Most of the ribbons in Bassoon-deficient mice are not docked at the synaptic site but float freely in the cytoplasm (free-floating ribbons) (Fig. 8.5). More than half of the rod photoreceptor terminals are empty, without presynaptic ribbons and postsynaptic invaginating elements, in Bassoon mutant mice. In a normal cone synaptic terminal, multiple ribbons with the horizontal cell processes and bipolar dendrites are formed (Boycott and Wassle 1999); however, in Bassoon-deficient cone terminals, free-floating ribbons without invaginations dominate. The free-floating presynaptic ribbons in Bassoon mutants aggregate and form “ribbon fields.” Fewer postsynaptic processes penetrate into the cone terminals of Bassoon mutants. In addition, abnormal dendritic branching of horizontal and bipolar cells to photoreceptors and ectopic synapses form in the ONL of Bassoon mutant mice. These results suggest that Bassoon is essential for the proper positioning of cone and rod photoreceptor synaptic ribbons and proper connections between photoreceptors and horizontal or bipolar cells (Dick et al. 2003).
Formation of aberrant ribbons and invaginations in Bassoon-deficient photoreceptor terminals. Ribbons in the Bassoon-deficient mice are not docked at the synaptic site and float freely in the cytoplasm. Empty rod photoreceptor terminals without presynaptic ribbons and postsynaptic invaginating elements are often observed in the Bassoon-deficient retina
8.3.2 CAST
CAST/ERC2 is a component of the photoreceptor CAZ, a site of synaptic vesicle fusion (Ohtsuka et al. 2002). CAST contains multiple coiled-coil domains and a C-terminal PDZ-binding motif that interacts with RIM1, a Rab3a effector protein (Ohtsuka et al. 2002; Takao-Rikitsu et al. 2004). In the CAST-null retina, the size of the rod presynaptic active zones and the extension of the OPL are diminished (Fig. 8.6) (tom Dieck et al. 2012). Previous studies reported that sproutings of postsynaptic elements into the ONL with ectopic synapse formation were found in the retinas of mutant mice with impaired photoreceptor synaptic transmission (Mansergh et al. 2005; Haeseleer et al. 2004; Specht et al. 2009). Similar to these mutants, CAST-null mouse horizontal cells extended processes into the ONL. These horizontal cell sproutings in CAST-deficient mice contain a ribbon marker, CtBP2, in the ONL. Electron micrographs of CAST-deficient retinas also showed the presence of ectopic neurites and ectopic synapses with ribbons in the ONL. In addition, CAST-deficient mice show diminished b-wave amplitudes in scotopic ERGs, indicating that synaptic transmissions from rod photoreceptors to rod bipolar cells are impaired. Thus, in rod photoreceptor ribbon synapses, CAST regulates proper CAZ formation and effective synaptic transmission between photoreceptor terminals and bipolar dendrites.
CAST is required for proper photoreceptor CAZ formation. The size of the active zone is reduced in CAST-deficient photoreceptor synaptic terminals. CAST-deficient mice show ectopic synaptic formation in the ONL and diminished ERG b-waves. CAST is essential for proper photoreceptor CAZ formation in the OPL
8.3.3 Endocytotic Regulators
At photoreceptor ribbon synapses, continuous exocytosis of vesicles needs to be balanced by compensatory endocytosis to replenish the vesicle pools. The rate of vesicle recycling is essential for signaling at photoreceptor ribbon synapses (Jackman et al. 2009; Babai et al. 2010). Synaptic ribbons are required for the resupply of release-ready synaptic vesicles (Frank et al. 2010; Schnee et al. 2011; Snellman et al. 2011; Tian et al. 2012). Recently, by super-resolution structured illumination microscopy and electron microscopy analysis, the precise localizations of major components of the endocytotic membrane retrieval machinery in photoreceptor synapses were determined (Wahl et al. 2013). Local endocytic machinery is enriched at the periactive zone in the vicinity of the synaptic ribbon. This periactive zone endocytic machinery is ideally placed to replenish the exocytotic machinery of the continuously active photoreceptor ribbon synapse. Dynamin, syndapin, amphiphysin, and calcineurin are enriched around the active zone and the synaptic ribbon in photoreceptor terminals. Clathrin heavy chain variant 1 (CHC-V1) is enriched in the periactive zone of photoreceptor synapses, and clathrin heavy chain variant 2 (CHC-V2) is located in an endosomal compartment proximal to the presynaptic terminal. Concentration of endocytic proteins around the ribbon is consistent with the focal uptake of endocytic markers at that site. The photoreceptor periactive zone surrounding the ribbons seems to be a hotspot of endocytosis.
8.3.4 L-Type Ca2+ Channel and Its Regulator Cabp4
At ribbon synapses, Ca2+ influx through L-type Ca2+ channels triggers neurotransmitter release (Schmitz et al. 2000). The L-type Cav1.4 channel α1-subunit (Cacna1f) is expressed in photoreceptor cells and is localized in photoreceptor synaptic terminals (Barnes and Kelly 2002). Null mutations in the Cav1.4 gene are responsible for an X-linked disorder, congenital stationary night blindness type 2A (CSNB2A) (Strom et al. 1998; Bech-Hansen et al. 2000). The lack of an ERG observed in these patients suggests that Cav1.4 is essential for synaptic signal transmission between rod photoreceptor and bipolar cells. In the animal model, mutation in the Cacna1f gene causes a similar phenotype to that observed in human patients, including abnormal ERG and diminished photoreceptor calcium signals (Mansergh et al. 2005; Specht et al. 2009).
Cabp4, a Ca2+-binding protein similar to calmodulin, directly interacts with Cav1.4 α1-subunit and modulates Cav1.4 activity in cells. Cabp4 is also specifically expressed in retinal photoreceptors. Cabp4 is localized to both rod and cone photoreceptor synaptic terminals in the OPL. Cabp4-deficient mice showed a thinner OPL than in control mice (Haeseleer et al. 2004). In the Cabp4-deficient retina, ectopic photoreceptor synapses are formed in the ONL. In addition, processes of rod bipolar and horizontal cells aberrantly extend into the ONL in Cabp4-deficient mice. Rod bipolar cell responses are remarkably reduced in sensitivity, and the ERG indicated a reduction in both cone and rod synaptic function in Cabp4-deficient mice. Thus, Cabp4 regulates Ca2+ influx and neurotransmitter release in photoreceptor synaptic terminals. In humans, mutations in the CABP4 gene were reported to cause congenital stationary night blindness type 2B (CSNB2B) (Zeitz et al. 2006). These findings show that the Cav1.4 channel and its regulator Cabp4 are essential factors for ribbon synapse function in photoreceptor cells.
8.4 Dystrophin Complex in Photoreceptor Synapses
Dystrophin, one of the genes responsible for muscular dystrophy, plays an essential role in the formation of synapses including neuromuscular junctions (NMJ) and retinal photoreceptor synapses. Dystroglycan, a transmembrane proteoglycan, forms a dystrophin–glycoprotein complex and connects the extracellular matrix (ECM) with the actin cytoskeleton in the NMJ and in non-muscle tissues such as the brain and retina (Henry and Campbell 1996). Dystroglycan consists of an extracellular α-subunit and a transmembrane β-subunit. In both rod and cone photoreceptor synapses, dystrophin forms a complex with the α-dystroglycan subunit and ECM protein pikachurin (Fig. 8.7) (Kanagawa et al. 2010; Howard et al. 1998; Sato et al. 2008; Omori et al. 2012). The dystrophin-dystroglycan-pikachurin (DGP) complex is required for the formation of proper synaptic connections between photoreceptor axon terminals and dendritic tips of bipolar cells.
Dystrophin-dystroglycan-pikachurin (DGP) complex in photoreceptor synaptic terminals. In photoreceptor synapses, dystrophin, a protein responsible for muscular dystrophy, forms a complex with dystroglycan, a transmembrane proteoglycan, and pikachurin, an extracellular matrix (ECM) protein. These components are essential for the connection between photoreceptor axon terminals and bipolar dendrites. Defects of either of these genes cause abnormal electroretinograms (ERGs) in mammals
8.4.1 Dystrophin
In humans, mutations in the dystrophin gene cause Duchenne–Becker muscular dystrophy (DMD/BMD), an X-linked recessive disease (Burghes et al. 1987; Kunkel et al. 1986; Monaco et al. 1986; Hoffman et al. 1987). Loss of the dystrophin protein in the NMJ causes progressive abnormalities of muscular functions. Aberrant ERGs are observed in both human patients (Pillers et al. 1993; Fitzgerald et al. 1994) and mice with mutations in dystrophin (Pillers et al. 1995; Kameya et al. 1997). Analysis of ERGs from allelic variants of muscular dystrophic (mdx) mutant mice and DMD/BMD patients with mutations in different regions of the dystrophin gene indicates a correlation between the position of the mutation and the severity of the ERG abnormality (Schmitz and Drenckhahn 1997).
In rod and cone photoreceptor cells, dystrophin localizes to the microdomain of the photoreceptor synaptic plasma membrane that forms the lateral wall of the synaptic cavity and projects with finger-like extensions into the postsynaptic dendritic complex. The DGP complex of the cavital plasma membrane appears to stabilize the elaborate synaptic morphology.
8.4.2 Pikachurin
Pikachurin, an ECM protein, is essential for proper synaptic connection between photoreceptor cells and bipolar cells (Sato et al. 2008). Pikachurin was first identified as a photoreceptor-specific gene. Pikachurin contains fibronectin-3 repeats and EGF-like and Laminin-G domains with N-terminal signal sequences. Pikachurin is localized to the photoreceptor synaptic cleft in the vicinity of the synaptic ribbon. Ultrastructural analysis revealed that pikachurin-deficient mice exhibited improper apposition of the bipolar dendritic tip to the photoreceptor synaptic invagination (Fig. 8.8). Consistent with these observations, pikachurin-deficient mice showed an increase in implicit times of both scotopic and photopic b-waves compared with those of control mice. In addition, pikachurin-deficient mice exhibited impaired optokinetic responses (OKR) (Sato et al. 2008; Omori et al. 2012). Pikachurin directly binds to the extracellular domain of dystroglycan, indicating that pikachurin is a ligand for dystroglycan in the retina (Kanagawa et al. 2010).
Photoreceptor synaptic terminals in control and pikachurin-deficient mice. Pikachurin-deficient mice show aberrant ERGs as well as mislocalization of dystroglycan in the OPL. Pikachurin interacts with dystroglycan on the surface of the photoreceptor synaptic membrane and is anchored by dystroglycan. Glycosylation of dystroglycan is important for the interaction between pikachurin and dystroglycan
8.4.3 Dystroglycan
In contrast to the predominantly retinal expression of pikachurin, dystroglycan is broadly expressed in various tissues, including the central nervous system (CNS) (Ibraghimov-Beskrovnaya et al. 1992; Ibraghimov-Beskrovnaya et al. 1993), and dystroglycan-deficient embryos exhibit gross developmental abnormalities beginning at around 6.5 days of gestation with early embryonic lethality (Williamson et al. 1997). This lethality prevents analyses of dystroglycan roles in organ development or maintenance at postnatal stages. To investigate the function of dystroglycan in photoreceptor cells, retinal photoreceptor-specific dystroglycan conditional knockout mice (CKO) were analyzed (Sato et al. 2008; Omori et al. 2012). Dystroglycan CKO mice show a reduced ERG amplitude and a prolonged ERG b-wave implicit time (Fig. 8.9). Electron microscopic analysis shows that bipolar cell dendritic penetration into the photoreceptor terminus is perturbed in the dystroglycan-deficient retina. In the dystroglycan-deficient retina, the pikachurin signal is markedly decreased at photoreceptor synapses. Conversely, in the pikachurin-deficient retina, the dystroglycan signals at the ribbon synaptic terminus are severely reduced. These findings suggest that pikachurin is required for the presynaptic accumulation of dystroglycan at the photoreceptor synaptic terminus and vice versa. Thus, the presynaptic interaction of pikachurin with dystroglycan at photoreceptor terminals is essential for both the formation of proper photoreceptor ribbon synaptic structures and normal retinal electrophysiology.
Electroretinograms recorded from dystroglycan-null and pikachurin-null retina: scotopic ERGs of a control mouse (left), a dystroglycan CKO mouse (middle), and pikachurin-deficient mouse (right). Implicit times of scotopic ERG b-waves are elongated in the dystroglycan CKO and pikachurin-deficient mice, and the amplitude of scotopic ERG b-waves is severely decreased in dystroglycan-CKO mice and mildly reduced in pikachurin-deficient mice
8.5 Synaptic Formation Is Regulated by Photoreceptor-Specific Transcription Factors
Photoreceptor synaptic abnormalities are reported in mice with defects in transcription factors Crx and Nrl, which control photoreceptor differentiation (Morrow et al. 2005; Strettoi et al. 2004). These transcription factors control synaptogenesis through regulating the expression of genes involved in photoreceptor synapse formation.
8.5.1 Crx
The Crx gene encodes an Otx-family homeodomain transcription factor that is expressed predominantly in cone and rod photoreceptors and is essential for photoreceptor development (Furukawa et al. 1997; Furukawa et al. 1999). Mutations in the human CRX gene are associated with retinal diseases including retinitis pigmentosa, LCA, and cone-rod dystrophy 2 (CRD2) (Freund et al. 1997; Swain et al. 1997; Sohocki et al. 1998; Swaroop et al. 1999). An ultrastructural study of Crx-deficient mice demonstrated that both rod and cone photoreceptor terminals are highly disorganized at P21 in the Crx-deficient OPL (Morrow et al. 2005). In Crx-deficient mice, processes with synaptic vesicles and ribbon-like structures are observed, showing that synapse components are at least generated in the mutant mice. A normal rod photoreceptor contains a single ribbon in the wild-type retina, however, many of photoreceptor synaptic terminals contain multiple ribbons in the Crx-null retina. In addition, aberrant ribbons are not tethered to the plasma membrane and are occasionally observed in perinuclear positions in the Crx-deficient mice. These finding suggest that Crx regulates expression of an essential factor for photoreceptor synaptogenesis. Microarray study of the Crx-deficient retina revealed that the expressions of many photoreceptor-specific genes including Cabp4 are markedly decreased in the Crx-deficient retina compared to the wild type (Livesey et al. 2000; Hsiau et al. 2007).
8.5.2 Nrl
Nrl, a basic leucine zipper transcription factor, is expressed in rods, but not cone photoreceptors, and is necessary for rod photoreceptor differentiation (Swaroop et al. 1992; Rehemtulla et al. 1996; Bessant et al. 1999; Swain et al. 2001; Zhu et al. 2003). In the Nrl-null retina, photoreceptor precursors do not develop into rods but generate an excess number of cone-like photoreceptors (Strettoi et al. 2004). Electron microscopy analysis revealed that the Nrl-deficient synaptic terminals are smaller than cone synaptic pedicles, and larger than typical rod synaptic spherules. Normal cone synaptic terminals form a single layer in the OPL, however, Nrl-deficient synaptic terminals occupy at least two layers. In Nrl-deficient mice, horizontal and bipolar processes form invaginations in photoreceptor terminals as they do in wild-type mice. Microarray study of the Nrl-deficient retina showed that the expressions of many rod photoreceptor-specific genes are markedly decreased (Yoshida et al. 2004); however, it is unknown which gene is responsible for the rod photoreceptor synaptic phenotypes observed in the Nrl-deficient retina.
8.6 Open Questions/Perspectives
Recently, marked progress has been achieved in numerous studies of the molecular components of the photoreceptor synaptic ribbon and associated proteins; however, the molecular mechanisms that create the differences between rod and cone photoreceptor synapses are still poorly understood. How are the numbers of ribbons in rod and cone regulated? How are the specific connections between the special types of bipolar cells and proper invaginations in multiple types of photoreceptor cells formed? Which molecules regulate the complex structures of photoreceptor ribbon synapses? These questions will be addressed using new techniques involving genetically manipulated animals including tissue-specific and/or developmental stage-specific gene targeting, as well as an increasing bounty of animal resources, including thousands of systematically generated floxed mouse lines (https://www.komp.org/). Microarray and proteomic analysis of retinal components using genetically modified animal tissues will provide valuable information in finding new factors. Research answering these questions will provide new knowledge not only for basic visual science but also for better understanding of the pathogenesis underlying retinal diseases such as retinitis pigmentosa, cone-rod dystrophy, and Leber’s congenital amaurosis.
References
Babai N, Bartoletti TM, Thoreson WB (2010) Calcium regulates vesicle replenishment at the cone ribbon synapse. J Neurosci 30:15866–15877
Barnes S, Kelly ME (2002) Calcium channels at the photoreceptor synapse. Adv Exp Med Biol 514:465–476
Bech-Hansen NT, Naylor MJ, Maybaum TA, Sparkes RL, Koop B, Birch DG, Bergen AA, Prinsen CF, Polomeno RC, Gal A, Drack AV, Musarella MA, Jacobson SG, Young RS, Weleber RG (2000) Mutations in NYX, encoding the leucine-rich proteoglycan nyctalopin, cause X-linked complete congenital stationary night blindness. Nat Genet 26:319–323
Bessant DA, Payne AM, Mitton KP, Wang QL, Swain PK, Plant C, Bird AC, Zack DJ, Swaroop A, Bhattacharya SS (1999) A mutation in NRL is associated with autosomal dominant retinitis pigmentosa. Nat Genet 21:355–356
Blanks JC, Adinolfi AM, Lolley RN (1974) Synaptogenesis in the photoreceptor terminal of the mouse retina. J Comp Neurol 156:81–93
Boycott B, Wassle H (1999) Parallel processing in the mammalian retina: the Proctor Lecture. Invest Ophthalmol Vis Sci 40:1313–1327
Burghes AH, Logan C, Hu X, Belfall B, Worton RG, Ray PN (1987) A cDNA clone from the Duchenne/Becker muscular dystrophy gene. Nature 328:434–437
Dick O, tom Dieck S, Altrock WD, Ammermuller J, Weiler R, Garner CC, Gundelfinger ED, Brandstatter JH (2003) The presynaptic active zone protein bassoon is essential for photoreceptor ribbon synapse formation in the retina. Neuron 37:775–786
Dresbach T, Qualmann B, Kessels MM, Garner CC, Gundelfinger ED (2001) The presynaptic cytomatrix of brain synapses. Cell Mol Life Sci 58:94–116
Fisher LJ (1979) Development of synaptic arrays in the inner plexiform layer of neonatal mouse retina. J Comp Neurol 187:359–372
Fitzgerald KM, Cibis GW, Giambrone SA, Harris DJ (1994) Retinal signal transmission in Duchenne muscular dystrophy: evidence for dysfunction in the photoreceptor/depolarizing bipolar cell pathway. J Clin Invest 93:2425–2430
Frank T, Rutherford MA, Strenzke N, Neef A, Pangrsic T, Khimich D, Fejtova A, Gundelfinger ED, Liberman MC, Harke B, Bryan KE, Lee A, Egner A, Riedel D, Moser T (2010) Bassoon and the synaptic ribbon organize Ca2+ channels and vesicles to add release sites and promote refilling. Neuron 68:724–738
Freund CL, Gregory-Evans CY, Furukawa T, Papaioannou M, Looser J, Ploder L, Bellingham J, Ng D, Herbrick JA, Duncan A, Scherer SW, Tsui LC, Loutradis-Anagnostou A, Jacobson SG, Cepko CL, Bhattacharya SS, McInnes RR (1997) Cone-rod dystrophy due to mutations in a novel photoreceptor-specific homeobox gene (CRX) essential for maintenance of the photoreceptor. Cell 91:543–553
Furukawa T, Morrow EM, Cepko CL (1997) Crx, a novel otx-like homeobox gene, shows photoreceptor-specific expression and regulates photoreceptor differentiation. Cell 91:531–541
Furukawa T, Morrow EM, Li T, Davis FC, Cepko CL (1999) Retinopathy and attenuated circadian entrainment in Crx-deficient mice. Nat Genet 23:466–470
Haeseleer F, Imanishi Y, Maeda T, Possin DE, Maeda A, Lee A, Rieke F, Palczewski K (2004) Essential role of Ca2+-binding protein 4, a Cav1.4 channel regulator, in photoreceptor synaptic function. Nat Neurosci 7:1079–1087
Henry MD, Campbell KP (1996) Dystroglycan: an extracellular matrix receptor linked to the cytoskeleton. Curr Opin Cell Biol 8:625–631
Hoffman EP, Brown RH Jr, Kunkel LM (1987) Dystrophin: the protein product of the Duchenne muscular dystrophy locus. Cell 51:919–928
Howard PL, Dally GY, Wong MH, Ho A, Weleber RG, Pillers DA, Ray PN (1998) Localization of dystrophin isoform Dp71 to the inner limiting membrane of the retina suggests a unique functional contribution of Dp71 in the retina. Hum Mol Genet 7:1385–1391
Hsiau TH, Diaconu C, Myers CA, Lee J, Cepko CL, Corbo JC (2007) The cis-regulatory logic of the mammalian photoreceptor transcriptional network. PLoS One 2:e643
Ibraghimov-Beskrovnaya O, Ervasti JM, Leveille CJ, Slaughter CA, Sernett SW, Campbell KP (1992) Primary structure of dystrophin-associated glycoproteins linking dystrophin to the extracellular matrix. Nature 355:696–702
Ibraghimov-Beskrovnaya O, Milatovich A, Ozcelik T, Yang B, Koepnick K, Francke U, Campbell KP (1993) Human dystroglycan: skeletal muscle cDNA, genomic structure, origin of tissue specific isoforms and chromosomal localization. Hum Mol Genet 2:1651–1657
Jackman SL, Choi SY, Thoreson WB, Rabl K, Bartoletti TM, Kramer RH (2009) Role of the synaptic ribbon in transmitting the cone light response. Nat Neurosci 12:303–310
Kameya S, Araki E, Katsuki M, Mizota A, Adachi E, Nakahara K, Nonaka I, Sakuragi S, Takeda S, Nabeshima Y (1997) Dp260 disrupted mice revealed prolonged implicit time of the b-wave in ERG and loss of accumulation of beta-dystroglycan in the outer plexiform layer of the retina. Hum Mol Genet 6:2195–2203
Kanagawa M, Omori Y, Sato S, Kobayashi K, Miyagoe-Suzuki Y, Takeda S, Endo T, Furukawa T, Toda T (2010) Post-translational maturation of dystroglycan is necessary for Pikachurin binding and ribbon synaptic localization. J Biol Chem 285:31208–31216
Kunkel LM et al (1986) Analysis of deletions in DNA from patients with Becker and Duchenne muscular dystrophy. Nature 322:73–77
Livesey FJ, Furukawa T, Steffen MA, Church GM, Cepko CL (2000) Microarray analysis of the transcriptional network controlled by the photoreceptor homeobox gene Crx. Curr Biol 10:301–310
Mansergh F, Orton NC, Vessey JP, Lalonde MR, Stell WK, Tremblay F, Barnes S, Rancourt DE, Bech-Hansen NT (2005) Mutation of the calcium channel gene Cacna1f disrupts calcium signaling, synaptic transmission and cellular organization in mouse retina. Hum Mol Genet 14:3035–3046
Masland RH (2001) The fundamental plan of the retina. Nat Neurosci 4:877–886
McLaughlin BJ (1976) A fine structural and E-PTA study of photoreceptor synaptogenesis in the chick retina. J Comp Neurol 170:347–364
Monaco AP, Neve RL, Colletti-Feener C, Bertelson CJ, Kurnit DM, Kunkel LM (1986) Isolation of candidate cDNAs for portions of the Duchenne muscular dystrophy gene. Nature 323:646–650
Morrow EM, Furukawa T, Raviola E, Cepko CL (2005) Synaptogenesis and outer segment formation are perturbed in the neural retina of Crx mutant mice. BMC Neurosci 6:5
Ohtsuka T, Takao-Rikitsu E, Inoue E, Inoue M, Takeuchi M, Matsubara K, Deguchi-Tawarada M, Satoh K, Morimoto K, Nakanishi H, Takai Y (2002) Cast: a novel protein of the cytomatrix at the active zone of synapses that forms a ternary complex with RIM1 and munc13-1. J Cell Biol 158:577–590
Olney JW (1968) An electron microscopic study of synapse formation, receptor outer segment development, and other aspects of developing mouse retina. Invest Ophthalmol 7:250–268
Omori Y, Araki F, Chaya T, Kajimura N, Irie S, Terada K, Muranishi Y, Tsujii T, Ueno S, Koyasu T, Tamaki Y, Kondo M, Amano S, Furukawa T (2012) Presynaptic dystroglycan-pikachurin complex regulates the proper synaptic connection between retinal photoreceptor and bipolar cells. J Neurosci 32:6126–6137
Pillers DA, Weleber RG, Woodward WR, Green DG, Chapman VM, Ray PN (1995) mdxCv3 mouse is a model for electroretinography of Duchenne/Becker muscular dystrophy. Invest Ophthalmol Vis Sci 36:462–466
Pillers DA, Bulman DE, Weleber RG, Sigesmund DA, Musarella MA, Powell BR, Murphey WH, Westall C, Panton C, Becker LE et al (1993) Dystrophin expression in the human retina is required for normal function as defined by electroretinography. Nat Genet 4:82–86
Rao-Mirotznik R, Harkins AB, Buchsbaum G, Sterling P (1995) Mammalian rod terminal: architecture of a binary synapse. Neuron 14:561–569
Rehemtulla A, Warwar R, Kumar R, Ji X, Zack DJ, Swaroop A (1996) The basic motif-leucine zipper transcription factor Nrl can positively regulate rhodopsin gene expression. Proc Natl Acad Sci USA 93:191–195
Sato S, Omori Y, Katoh K, Kondo M, Kanagawa M, Miyata K, Funabiki K, Koyasu T, Kajimura N, Miyoshi T, Sawai H, Kobayashi K, Tani A, Toda T, Usukura J, Tano Y, Fujikado T, Furukawa T (2008) Pikachurin, a dystroglycan ligand, is essential for photoreceptor ribbon synapse formation. Nat Neurosci 11:923–931
Schmitt EA, Dowling JE (1999) Early retinal development in the zebrafish, Danio rerio: light and electron microscopic analyses. J Comp Neurol 404:515–536
Schmitz F, Drenckhahn D (1997) Localization of dystrophin and beta-dystroglycan in bovine retinal photoreceptor processes extending into the postsynaptic dendritic complex. Histochem Cell Biol 108:249–255
Schmitz F, Konigstorfer A, Sudhof TC (2000) RIBEYE, a component of synaptic ribbons: a protein's journey through evolution provides insight into synaptic ribbon function. Neuron 28:857–872
Schnee ME, Santos-Sacchi J, Castellano-Munoz M, Kong JH, Ricci AJ (2011) Calcium-dependent synaptic vesicle trafficking underlies indefatigable release at the hair cell afferent fiber synapse. Neuron 70:326–338
Snellman J, Mehta B, Babai N, Bartoletti TM, Akmentin W, Francis A, Matthews G, Thoreson W, Zenisek D (2011) Acute destruction of the synaptic ribbon reveals a role for the ribbon in vesicle priming. Nat Neurosci 14:1135–1141
Sohocki MM, Sullivan LS, Mintz-Hittner HA, Birch D, Heckenlively JR, Freund CL, McInnes RR, Daiger SP (1998) A range of clinical phenotypes associated with mutations in CRX, a photoreceptor transcription-factor gene. Am J Hum Genet 63:1307–1315
Specht D, Wu SB, Turner P, Dearden P, Koentgen F, Wolfrum U, Maw M, Brandstatter JH, tom Dieck S (2009) Effects of presynaptic mutations on a postsynaptic Cacna1s calcium channel colocalized with mGluR6 at mouse photoreceptor ribbon synapses. Invest Ophthalmol Vis Sci 50:505–515
Sterling P, Matthews G (2005) Structure and function of ribbon synapses. Trends Neurosci 28:20–29
Strettoi E, Mears AJ, Swaroop A (2004) Recruitment of the rod pathway by cones in the absence of rods. J Neurosci 24:7576–7582
Strom TM, Nyakatura G, Apfelstedt-Sylla E, Hellebrand H, Lorenz B, Weber BH, Wutz K, Gutwillinger N, Ruther K, Drescher B, Sauer C, Zrenner E, Meitinger T, Rosenthal A, Meindl A (1998) An L-type calcium-channel gene mutated in incomplete X-linked congenital stationary night blindness. Nat Genet 19:260–263
Swain PK, Chen S, Wang QL, Affatigato LM, Coats CL, Brady KD, Fishman GA, Jacobson SG, Swaroop A, Stone E, Sieving PA, Zack DJ (1997) Mutations in the cone-rod homeobox gene are associated with the cone-rod dystrophy photoreceptor degeneration. Neuron 19:1329–1336
Swain PK, Hicks D, Mears AJ, Apel IJ, Smith JE, John SK, Hendrickson A, Milam AH, Swaroop A (2001) Multiple phosphorylated isoforms of NRL are expressed in rod photoreceptors. J Biol Chem 276:36824–36830
Swaroop A, Wang QL, Wu W, Cook J, Coats C, Xu S, Chen S, Zack DJ, Sieving PA (1999) Leber congenital amaurosis caused by a homozygous mutation (R90W) in the homeodomain of the retinal transcription factor CRX: direct evidence for the involvement of CRX in the development of photoreceptor function. Hum Mol Genet 8:299–305
Swaroop A, Xu JZ, Pawar H, Jackson A, Skolnick C, Agarwal N (1992) A conserved retina-specific gene encodes a basic motif/leucine zipper domain. Proc Natl Acad Sci USA 89:266–270
Takao-Rikitsu E, Mochida S, Inoue E, Deguchi-Tawarada M, Inoue M, Ohtsuka T, Takai Y (2004) Physical and functional interaction of the active zone proteins, CAST, RIM1, and Bassoon, in neurotransmitter release. J Cell Biol 164:301–311
Tian M, Xu CS, Montpetit R, Kramer RH (2012) Rab3A mediates vesicle delivery at photoreceptor ribbon synapses. J Neurosci 32:6931–6936
tom Dieck S, Brandstatter JH (2006) Ribbon synapses of the retina. Cell Tissue Res 326:339–346
tom Dieck S, Altrock WD, Kessels MM, Qualmann B, Regus H, Brauner D, Fejtova A, Bracko O, Gundelfinger ED, Brandstatter JH (2005) Molecular dissection of the photoreceptor ribbon synapse: physical interaction of Bassoon and RIBEYE is essential for the assembly of the ribbon complex. J Cell Biol 168:825–836
tom Dieck S, Sanmarti-Vila L, Langnaese K, Richter K, Kindler S, Soyke A, Wex H, Smalla KH, Kampf U, Franzer JT, Stumm M, Garner CC, Gundelfinger ED (1998) Bassoon, a novel zinc-finger CAG/glutamine-repeat protein selectively localized at the active zone of presynaptic nerve terminals. J Cell Biol 142:499–509
tom Dieck S, Specht D, Strenzke N, Hida Y, Krishnamoorthy V, Schmidt KF, Inoue E, Ishizaki H, Tanaka-Okamoto M, Miyoshi J, Hagiwara A, Brandstatter JH, Lowel S, Gollisch T, Ohtsuka T, Moser T (2012) Deletion of the presynaptic scaffold CAST reduces active zone size in rod photoreceptors and impairs visual processing. J Neurosci 32:12192–12203
Wahl S, Katiyar R, Schmitz F (2013) A local, periactive zone endocytic machinery at photoreceptor synapses in close vicinity to synaptic ribbons. J Neurosci 33:10278–10300
Williamson RA, Henry MD, Daniels KJ, Hrstka RF, Lee JC, Sunada Y, Ibraghimov-Beskrovnaya O, Campbell KP (1997) Dystroglycan is essential for early embryonic development: disruption of Reichert’s membrane in Dag1-null mice. Hum Mol Genet 6:831–841
Yoshida S, Mears AJ, Friedman JS, Carter T, He S, Oh E, Jing Y, Farjo R, Fleury G, Barlow C, Hero AO, Swaroop A (2004) Expression profiling of the developing and mature Nrl−/− mouse retina: identification of retinal disease candidates and transcriptional regulatory targets of Nrl. Hum Mol Genet 13:1487–1503
Zeitz C, Kloeckener-Gruissem B, Forster U, Kohl S, Magyar I, Wissinger B, Matyas G, Borruat FX, Schorderet DF, Zrenner E, Munier FL, Berger W (2006) Mutations in CABP4, the gene encoding the Ca2+-binding protein 4, cause autosomal recessive night blindness. Am J Hum Genet 79:657–667
Zhu X, Brown B, Li A, Mears AJ, Swaroop A, Craft CM (2003) GRK1-dependent phosphorylation of S and M opsins and their binding to cone arrestin during cone phototransduction in the mouse retina. J Neurosci 23:6152–6160
Ziff EB (1997) Enlightening the postsynaptic density. Neuron 19:1163–1174
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer Japan
About this chapter
Cite this chapter
Omori, Y., Furukawa, T. (2014). Structure and Development of the Photoreceptor Ribbon Synapse. In: Furukawa, T., Hurley, J., Kawamura, S. (eds) Vertebrate Photoreceptors. Springer, Tokyo. https://doi.org/10.1007/978-4-431-54880-5_8
Download citation
DOI: https://doi.org/10.1007/978-4-431-54880-5_8
Published:
Publisher Name: Springer, Tokyo
Print ISBN: 978-4-431-54879-9
Online ISBN: 978-4-431-54880-5
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)