Abstract
The Fragile X syndrome (FXS) is the most frequent form of inherited mental retardation and also considered a monogenic cause of Autism Spectrum Disorder. FXS symptoms include neurodevelopmental delay, anxiety, hyperactivity, and autistic-like behavior. The disease is due to mutations or loss of the Fragile X Mental Retardation Protein (FMRP), an RNA-binding protein abundant in the brain and gonads, the two organs mainly affected in FXS patients. FMRP has multiple functions in RNA metabolism, including mRNA decay, dendritic targeting of mRNAs, and protein synthesis. In neurons lacking FMRP, a wide array of mRNAs encoding proteins involved in synaptic structure and function are altered. As a result of this complex dysregulation, in the absence of FMRP, spine morphology and functioning is impaired. Consistently, model organisms for the study of the syndrome recapitulate the phenotype observed in FXS patients, such as dendritic spine anomalies and defects in learning.
Here, we review the fundamentals of genetic and clinical aspects of FXS, devoting a specific attention to ASD comorbidity and FXS-related diseases. We also review the current knowledge on FMRP functions through structural, molecular, and cellular findings. Finally, we discuss the neuroanatomical, electrophysiological, and behavioral defects caused by FMRP loss, as well as the current treatments able to partially revert some of the FXS abnormalities.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Genetics of Fragile X (FXS) and Fragile X Tremor Ataxia (FXTAS) Syndromes
1.1 Fragile Mental Retardation 1: A Gene Associated to Two Neurological Diseases
The Fragile X syndrome (FXS) is the most frequent form of inherited intellectual disability (Jacquemont et al. 2007). Patients with FXS show physical features, such as large ears, elongated face, and high-arched palate, which have been reported in 60% of prepubertal FXS boys. Other symptoms include connective tissue anomalies, which can lead to mitral valve prolapse, scoliosis, flat feet, and joint laxity. Recurrent otitis media and strabismus are also common. Macroorchidism due to a hypothalamic dysfunction affects about 90% of boys with FXS by the age of 14 (Jacquemont et al. 2007).
The neurological involvement displays a broad spectrum of cognitive and behavioral deficits. The developmental delay is the most consistent feature, with a mean IQ of 42 in boys and severe mental retardation in about 25% of cases. Since the disorder is X-linked and the penetrance is variable, females are usually in a low–normal range, with an IQ ranging from 70 to 90 (Jacquemont et al. 2007). Moreover, epilepsy has been described in 13–18% of boys and 4% in girls, but normally, the seizures and EEG alterations tend to resolve during childhood or early adulthood (Berry-Kravis 2002; Musumeci et al. 1999). Despite the severe neurobehavioral symptoms, the anatomical studies revealed minor abnormalities in postmortem brains from FXS patients (Hallahan et al. 2010; Reiss et al. 1995). The most prominent neuroanatomical feature is the dysgenesis of the dendritic spines, which appear longer and thinner than normal, likely due to a developmental delay in spine dynamics and transition from immature to mature spines (Cruz-Martin et al. 2010; Irwin et al. 2001). FXS is also the most common monogenic cause of Autism Spectrum Disorder, ASD (Hatton et al. 2006), a heterogenous group of neurodevelopmental pathologies affecting approximately 37 individuals in 10,000 (Fombonne 2005) and present in more than 40% of patients with intellectual disability (Moss and Howlin 2009). About 25% of FXS boys and 6% of girls meet criteria for ASD, while 1–2% of patients affected by ASD have FXS (Abrahams and Geschwind 2008; Hatton et al. 2006). In particular, recent reports estimated that about 30% of FXS subjects meet criteria for Autistic Disorder and 30% for Pervasive Developmental Disorder Not Otherwise Specified (Harris et al. 2008). However, up to 90% of children with Fragile X display behavioral alterations which resemble ASD, such as social anxiety, gaze avoidance, delayed speech development, echolalia, sensory hypersensitivity, tactile defensiveness, stereotypic movements, and poor motor coordination (Belmonte and Bourgeron 2006; Hernandez et al. 2009). The cognitive delay is more severe in FXS children with ASD, and additional neurological disorders, genetic problems, or seizures may increase the risk of autism (Garcia-Nonell et al. 2008).
FXS is due to triplet repeat expansion or point mutations in the Fragile X mental retardation 1 (FMR1) gene, located on chromosome Xq27-3 (Fig. 23.1). A severe FXS form has also been documented in a patient with a mutation in the coding region of the gene, leading to the substitution of isoleucine 304 for asparagine (Ile304Asn, see below) (De Boulle et al. 1993). Few cases with deletions in the coding regions have also been identified (Gedeon et al. 1992; Meijer et al. 1994; Mila et al. 2000; Wohrle et al. 1992). In over 90% of patients, a CGG triplet in the 5′ UTR of the gene is expanded over 200 copies, leading to hypermethylation of the CGG, transcriptional silencing, and abolished production of the Fragile X Mental Retardation Protein (FMRP) (Jacquemont et al. 2007). The CGG triplet region is highly polymorphic in the population. Normal alleles (5–44 CGG copies) are stably transmitted to the offspring; “gray-zone” alleles (45–54 copies) and “premutation” alleles (55–200 copies) are rather unstable and can evolve into a “full mutation” (>200 repeats) during the maternal transmission (Fig. 23.1). While the gray-zone alleles require at least two generations before expanding to a full mutation (Fernandez-Carvajal et al. 2009), the premutation is highly unstable, and the risk of transmitting an allele with the full mutation is function of the repeat length (Hagerman and Hagerman 2002). The carriers of premutation alleles were considered clinically unaffected since the discovery of a dominant late-onset neurodegenerative disorder: the Fragile X Tremor Ataxia Syndrome (FXTAS). In 2001, the Hagerman laboratory described for the first time action tremor associated with executive function impairments and brain atrophy in five elderly men with the premutation (Hagerman et al. 2001). The frequency of premutation carriers has been estimated in 1:800 in men and 1:300 in women, although FXTAS displays reduced penetrance (~33% in men, not yet determined in women). Major behavioral diagnostic criteria for FXTAS are gait ataxia and intention tremor, eventually associated with parkinsonism and cognitive decline, often progressing to dementia (Hagerman and Hagerman 2007). Psychiatric disturbances often observed in FXTAS patients include anxiety, depression, and hostility (Bacalman et al. 2006). In some premutation patients, a psychiatric phenotype with features resembling attention deficit hyperactivity disorder (ADHD) and ASD can also appear in childhood or adolescence (Farzin et al. 2006). Further studies on postmortem brains from premutation carriers revealed a generalized brain atrophy, white matter disease, and middle cerebellar peduncle lesions (Hagerman and Hagerman 2007). One of the key cytological hallmarks of FXTAS is the presence of intranuclear ubiquitin-positive inclusions in neurons and astrocytes throughout the brain (Greco et al. 2006). The intranuclear foci were consistently observed in model organisms for FXTAS, both mouse (Berman and Willemsen 2009) and Drosophila (Jin et al. 2007; Sofola et al. 2007). Remarkably, the number of inclusions correlates with the size of the CGG expansion (Greco et al. 2002, 2006).
FXS and FXTAS are both due to triplet expansions. While FXS is a loss-of-function disease, FXTAS is thought to be a consequence of RNA toxic gain-of-function mechanism. First, a consistent molecular feature in both FXTAS patients and mouse models is the elevation of aberrant CGG expanded FMR1 mRNA levels (Allen et al. 2004; Kenneson et al. 2001; Tassone et al. 2000), due to increased transcription (Tassone et al. 2007). Nevertheless, carriers of premutation alleles show decreased levels of FMRP (Brouwer et al. 2008; Entezam et al. 2007) caused by the reduced translational efficiency of the FMR1 mRNA carrying the CGG expansion (Primerano et al. 2002), as well as by a differential use of the FMR1 mRNA 3′ UTR (Tassone et al. 2011). It has been proposed that the CGG expansion in the 5′ UTR would form a secondary structure inhibiting the ribosome scanning and thus leading to a scarce translational efficiency (Feng et al. 1995). The intranuclear foci contain in addition to the aberrant FMR1 mRNA (Greco et al. 2002; Tassone et al. 2004) a variety of RNA-binding proteins (RBPs) interacting with the rCGG tract, such as PURalpha, hnRNP A2/B1, and CUG-BP1 (Iwahashi et al. 2006).
The FMR1 gene encodes for FMRP (Bassell and Warren 2008; De Rubeis and Bagni 2010), an RNA-binding protein that contributes to the posttranscriptional control of gene expression (see below). In neurons, FMRP is part of messenger ribonucleoparticles (mRNPs) and regulates dendritic transport of associated mRNAs, their stability and local translation (Bassell and Warren 2008; De Rubeis and Bagni 2010). The roles of FMRP have been mainly addressed by using animal models that mimic FXS.
1.2 Model Organisms for the Study of the Fragile X Syndrome
The FMR1 gene is conserved along evolution, and this allowed researchers to develop murine (Bakker 1994; Mientjes et al. 2006), Drosophila (Zhang et al. 2001), and zebrafish (den Broeder et al. 2009; Tucker et al. 2006) animal models to study the molecular, cellular, and behavioral phenotypes of the syndrome.
Mouse models. The first model available, the Fmr1 knockout (KO) mouse, was generated by interrupting exon 5 of the Fmr1 gene with a neomycin cassette (Bakker 1994). Although the insertional mutation does not mimic FXS in humans, it leads to the functional ablation of Fmr1 gene since the interrupted Fmr1 mRNA prevents the translation of a functional FMRP (Bakker 1994). This mouse model presents an array of anatomical, behavioral, and neurological similarities to FXS patients (see below). Recently, a conditional KO (Fmr1 CKO) and a second generation Fmr1 KO null for Fmr1 mRNA (Fmr1 KO 2) have been generated by flanking the murine promoter and the first exon with loxP sites (Mientjes et al. 2006). In Fmr1 CKO, Fmr1 expression can be suppressed at specific developmental stages or in specific cell types, as showed by crossing these mice with a line carrying the Cre recombinase driven by a Purkinje cell–specific promoter (Mientjes et al. 2006). Moreover, a mouse model mimicking the mutation Ile304Asn that leads to a severe FXS manifestation (De Boulle et al. 1993) has been recently generated; of interest, this model phenocopies the behavioral and electrophysiological defects observed in Fmr1 KO mice (Zang et al. 2009) (see below).
Fruit fly Models. In Drosophila melanogaster, several loss-of-function mutations, ranging from hypomorphs to nulls, have been generated (Dockendorff et al. 2002; Inoue et al. 2002; Morales et al. 2002; Zhang et al. 2001). Such models display a variety of behavioral and developmental defects (Zarnescu et al. 2005).
Zebrafish Models. The first attempt to produce a model for FXS in Danio rerio was performed in 2006 using a knockdown approach by microinjecting morpholinos in early embryos (Tucker et al. 2006). Although the authors described defects in craniofacial development and neuronal branching in embryos, further studies failed in reproducing this phenotype in two Fmr1 KO lines (den Broeder et al. 2009).
2 An Insight into the Structure of the Fragile X Mental Retardation Protein
The FMR1 gene is composed of 17 exons and subjected to alternative splicing, occurring preferentially at the level of exons 12, 14, 15, and 17. This generates up to 12 different protein isoforms, with a molecular weight ranging between 70 and 80 kDa, with the longest isoform containing 632 amino acidic residues (Bassell and Warren 2008). The role of each isoform still needs to be clarified (Bassell and Warren 2008). Whereas FMRP isoforms are similarly expressed in many tissues and organs, the relative abundance of each isoform seems to be tissue specific (Kaufmann et al. 2002; Xie et al. 2009).
FMRP is a multidomain RNA-binding protein able to recognize several coding and noncoding RNAs, including the brain cytoplasmic RNA BC1/BC200 (Ashley et al. 1993; Johnson et al. 2006; Napoli et al. 2008; O’Donnell and Warren 2002; Siomi et al. 1993; Zalfa et al. 2005) and microRNAs (Edbauer et al. 2010). Moreover, FMRP homodimerizes and interacts with several cytoplasmic and nuclear proteins, including the two paralogs Fragile-X-related proteins 1 and 2 (FXRP1 and FXRP2) (O’Donnell and Warren 2002; Tamanini et al. 1999; Zhang et al. 1995).
The protein can be structurally divided into three main regions: N-terminal region, central region, and C-terminal region (Fig. 23.2a). The N-terminal region is characterized by the presence of two Tudor domains (TD), a putative Helix-Loop-Helix domain (HLH), and a Nuclear Localization Signal (NLS) (Sjekloca et al. 2009) (Fig. 23.2a). The central region contains two K Homology domains (KH) that share a high degree of homology with the hnRNP K domain and a Nuclear Export Signal (NES) (Valverde et al. 2008) (Fig. 23.2a). The C-terminal region, which is the less conserved region among the different species, is characterized by the presence of an RGG box containing a conserved Arg-Gly-Gly triplet (Darnell et al. 2001; Menon et al. 2004; Sjekloca et al. 2009) (Fig. 23.2a).
Structural modules of FMRP. (a) The different domains that compose the protein are represented following the color code: yellow = Tudor domains, blue = nuclear localization signal, green = helix-loop-helix motif, red = K homology domains, purple = nuclear export signal, gray = RGG box. The division in N-terminal, central, and C-terminal regions is also represented. (b) Ribbon representation of the structure of the two Tudor domains represented in yellow and (c) of the KH2 domain represented in red. In both panels, the succession of the secondary structure elements is underlined. The position of residue Ile304 is highlighted in gray
A region modulated by phosphorylation is localized between the FMRP central portion and the RGG box, specifically located between the amino acids 483 and 521 and conserved along different species. Ceman and colleagues showed that the phosphorylation of serine 499 triggers hierarchical phosphorylation events of nearby serines (Ceman et al. 2003). This phosphorylation modulates the association of FMRP with Dicer involving FMRP in the miRNA pathway (Cheever and Ceman 2009).
The N-terminal region of FMRP contains two Tudor domains able to bind single-strand (ss) nucleic acids. The same activity is shared by the two KH domains in the central region (Darnell et al. 2001; Musco et al. 1996; Ramos et al. 2006; Valverde et al. 2007) and the RGG box in the C-terminal region. The structure of the entire protein has not been resolved so far, but NMR and X-ray structures of single domains are available (Ramos et al. 2006; Valverde et al. 2007). The structure of the first 134 residues of the N-terminal domain, resolved by NMR, reveals the three-dimensional organization of the two Tudor domains, each one formed by a barrel-like fold made of four-stranded antiparallel β sheet (Fig. 23.2b). The two Tudor domains are linked by an unstructured fragment (linker) (Ramos et al. 2006) (Fig. 23.2b). The structure of the first 134 residues reveals three structural motifs, the two Tudor domains repeats and one α helix (Ramos et al. 2006) (Fig. 23.2b). Extensive interactions are observed between these elements, strongly suggesting that all the elements are necessary for the stability of the overall N-terminal domain (Ramos et al. 2006). The structure also reveals hydrophobic pockets on the surface of the two Tudor domains, in analogy with other Tudor domains, such as the Survival of Motor Neuron (SMN) Tudor domain and the heterochromatin-associated protein 1 (HP1) chromo domains (Nielsen et al. 2002b; Sprangers et al. 2003). These hydrophobic pockets can bind methylated amino acids (Ramos et al. 2006).
The three-dimensional structure of the two KH domains located in the central region of FMRP has been solved by X-ray (Valverde et al. 2007). The KH domains, usually present in multiple copies in a protein, contain the consensus (ILV)-I-G-X 2-G-X 2-I sequence and are responsible for the interaction with ssDNA, mRNA, and rRNA. The KH domains consist of a β1α1α2β2β′α′ fold, three-dimensionally oriented as a three-stranded β-sheet domain opposed to a three α-helices domain (Valverde et al. 2008), as evidenced by the X-ray structure (Valverde et al. 2007) (Fig. 23.2c). The KH domains are classified as Type I or Type II folds. Both contain the minimal KH motif but with different C- or N-terminal extensions giving β1α1α2β1β′α′ and α′β′β1α1α2β2 for Type I and Type II, respectively (Valverde et al. 2007) (Fig. 23.2c). KH domains in eukaryotic proteins are exclusively Type I, whereas in prokaryotic proteins are exclusively Type II (Grishin 2001; Siomi et al. 1993). β-Sheets β2 and β′ are connected by a variable loop, while α-helices α2 and α′ are connected by the so-called GXXG loop (Fig. 23.2c). This HLH (Helix-Loop-Helix) region of the domain forms the nucleic acid binding site (Valverde et al. 2008).
The best characterized missense mutation for FMRP is Ile304Asn, located on the KH2 domain (De Boulle et al. 1993). As previously mentioned, this mutation has been reported in an individual with a severe manifestation of FXS, including very low IQ, macroorchidism, and severe social and behavioral impairment (De Boulle et al. 1993). This underlines the importance of the KH2 domain for the neuronal functions of FMRP. The structure of the two KH domains revealed that the Ile304 residue is located on helix α2 of the KH2 domain (Fig. 23.2c). This residue is part of the hydrophobic core that stabilizes the three-dimensional folding of the domain. Indeed, the hydrophobic residues present at the interface of the α-helices and β-sheets domain establish a hydrophobic network of interactions that maintains the domain structure. This network of hydrophobic packing and van der Waals interactions is hypothesized to be conserved among all the Fragile-X-related proteins, such as FXRP1 and FXRP2, since all the hydrophobic residues abovementioned are conserved (Valverde et al. 2007). The substitution of residue Ile304 by an Asn, located in this hydrophobic core may disrupt this network, affecting the structure of the domain (Fig. 23.2c). Furthermore, being Ile304 buried in the domain core and not solvent accessible, it seems that the residue is not directly involved in the binding with nucleic acids, but the structural rearrangements caused by the mutation could affect the nucleic acid binding (Valverde et al. 2007).
FMRP belongs to a multiprotein complex and not only interacts with nucleic acids but also with a series of other proteins. Among all, the best characterized are the Cytoplasmic Fragile X Mental-Retardation-Interacting Protein 1 and 2 (CYFIP1 and 2), the Eukaryotic Translation Initiation Factor 4E (eIF4E), the Insulin-like Growth Factor 2 mRNA-Binding Protein (IGF2BP1), the Survival of Motor Neuron (SMN), the Tudor-Domain-Containing Protein 3 (TDRD3), the FXR1 and 2, the 58-kDa Microspherule Protein (MSP58), and the Nuclear Fragile X Mental-Retardation-Interacting Protein 1 and 2 (NUFIP1 and 2). FMRP also interacts with a series of other proteins involved in several diseases (Table 23.1). The extensive network of interactions explains the presence of FMRP in several multiprotein complexes.
Most of the protein–protein and protein–RNA interactions occur at the N-terminal and central regions of FMRP. These two regions display a high degree of conservation among different family members (FMRP-FXR1P–FXR2P), while the C-terminal region, where the RGG box is located, is found to be the less conserved (Menon et al. 2004).
Among the best characterized interactions, the region encoded by exon 7 (residues 173–218) of FMRP is responsible for the interaction with CYFIP1 and 2. This interaction leads to the formation of the eIF4E-CYFIP1-FMRP complex that blocks mRNAs translation process (Napoli et al. 2008) (see below). Interaction of FMRP with NUFIP (Nuclear FMRP Interacting Protein) occurs through the N-terminal region (residues 1–217) (Bardoni et al. 2003). Residues 470–485 of the N terminus are also essential for the interaction with SMN, while residues 430–486 as well as the second KH domain are crucial for the binding with TDRD3 (Linder et al. 2008). Interestingly, the Ile304Asn mutation affects the FMRP–TDRD3 interaction (Linder et al. 2008). The C terminus is involved in the interaction with RanBPM, and this interaction also modulates the FMRP RNA binding activity (Menon et al. 2004).
3 Cellular and Molecular Functions of FMRP
FMRP is mainly expressed in the brain and gonads (Khandjian et al. 1995; Verheij et al. 1993) where it is mostly confined to the cytoplasm. FMRP has also been localized in nucleus (Willemsen et al. 1996). Despite existing a clear shuttling process of the protein from both compartments (Eberhart et al. 1996; Sittler et al. 1996), the neuronal cytoplasmic function of FMRP has been mainly addressed. FMRP forms large cytoplasmic ribonucleoparticles (RNPs) containing several other proteins and RNAs (Johnson et al. 2006; Zalfa et al. 2005; Zalfa and Bagni 2005; Zalfa et al. 2003). FMRP-RNPs have also been found to cosediment with both polyribosomes and mRNPs (Zalfa et al. 2006) consequently being involved not only in the traffic and stability of the transported mRNAs but also in their translation. FMRP has also been detected in P bodies (PB) and stress granules (SG) containing translationally silent preinitiation complexes (Anderson and Kedersha 2006). Several studies have shown that FMRP plays a critical role in regulating mRNA translation, transport, and stability (Bagni and Greenough 2005; Bassell and Warren 2008; De Rubeis and Bagni 2010) (see Table 23.2 for a list of validated mRNA targets). In addition, the expression of FMRP in dendrites increases after synaptic stimulation suggesting a direct link between FMRP function and synaptic plasticity activation (Antar et al. 2004, 2006; Ferrari et al. 2007).
3.1 Regulation of Protein Synthesis
The translational dysregulation of a subset of FMRP target mRNAs is probably the major contribution to FXS (Table 23.2) (Bassell and Warren 2008). In neurons, protein synthesis occurs not only in the soma but also along axons (Holt and Bullock 2009), dendrites, and postsynaptic sites (Steward and Schuman 2003). Local protein synthesis is required for long-lasting forms of synaptic plasticity that underlie consolidation of long-term memories (Flavell and Greenberg 2008).
In the brain, protein synthesis is a mechanism that follows different states of synaptic plasticity activation, and it is orchestrated by the action of glutamate receptors. The ionotropic receptors N-methyl-D-aspartate (NMDAR), alpha-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPAR), and the metabotrobic glutamate receptor (mGluR) play a key role in basic synaptic plasticity as well as in the activation of different synaptic plasticity states (Massey and Bashir 2007; Shi et al. 1999). In vitro models to study synaptic plasticity have been developed by depolarization of the postsynaptic membrane. Specifically, activation of mGluR by applying dihydroxyphenylglycine (DHPG) induces a synaptic plasticity state called long-term depression (LTD) (DHPG-induced LTD) (Massey and Bashir 2007) which involves several synaptic events including mRNA targeting and local protein synthesis and degradation (Gladding et al. 2009). However, in the Fmr1 KO mice, DHPG-induced LTD plasticity is strongly increased (Huber et al. 2002), and it is also protein synthesis independent. This effect on LTD is likely due to deregulated local protein synthesis (Lu et al. 2004; Muddashetty et al. 2007; Zalfa et al. 2007; Huber et al. 2002; Nosyreva and Huber 2006; Ronesi and Huber 2008) and has settled the bases to describe the “mGluR theory” (Bear et al. 2004) (see below).
FMRP is involved in both basal and activity-dependent local protein synthesis by repressing in vivo and in vitro translation (Laggerbauer et al. 2001; Li et al. 2001; Lu et al. 2004; Muddashetty et al. 2007; Napoli et al. 2008; Zalfa et al. 2003). This has been proved in lymphoblastoid cells from individuals affected by FXS, in which 251 FMRP mRNA targets showed an abnormal polysomal distribution, explaining an increased translation (Brown et al. 2001). Moreover, protein synthesis of FMRP target mRNAs is increased in Fmr1 KO mice, especially in purified synaptosomes, extending to synapses the function of FMRP as a translation repressor (Muddashetty et al. 2007; Schütt et al. 2009; Zalfa et al. 2003). Regarding proteins localized near the postsynaptic membrane, a single study performed by Schütt and colleagues detected an increase in the expression levels of the postsynaptic proteins SAPAP1, SAPAP2, SAPAP3, Shank1, Shank3, IRSp53 as well as the NMDA receptor subunits NR1 and NR2B and GluR1 (Schütt et al. 2009). These differences were either cortical or hippocampus specific. However, while FMRP was shown to bind the mRNAs encoding SAPAP1, SAPAP2, SAPAP3, Shank1, and the NMDA receptor subunits NR1 and NR2B, the loss of FMRP did not affect their total and synaptic mRNA levels indicating the role of FMRP in their translation control and not in mRNA stability (Schütt et al. 2009). In addition to this, Bassell and collaborators have recently found that FMRP binds and represses the translation of the mRNA encoding phosphatidylinositol-4,5-bisphosphate 3-kinase 110 kDa catalytic subunit beta (p110β), the catalytic subunit of PI3K, a signaling molecule downstream activation of mGluRs (Gross et al. 2010). Further studies extending the proteomic analysis to the entire synaptosome are required to extend the role of FMRP and its mRNA targets at synapses.
FMRP expression and consequently its function can be regulated by posttranslational modifications such as ubiquitination (Hou et al. 2006) and/or phosphorylation. FMRP is rapidly translated at synapses in response to chemically induced LTD (Antar et al. 2004; Ferrari et al. 2007; Kao et al. 2010) and followed by a quick degradation (5 min) through the ubiquitin–proteasome system after LTD induction (Hou et al. 2006). In parallel, FMRP has also been found to be highly phosphorylated when it cosediments with polyribosomes whereas its dephosphorylation releases FMRP from polysomes allowing protein synthesis (Ceman et al. 2003). The activation of mTOR pathway, through protein phosphatase 2A (PP2A) and ribosomal protein S6 kinase (S6K) activation, seems to be involved in the dephosphorylation and phosphorylation of FMRP at different time points during LTD stimulation (Narayanan et al. 2007). These investigations highlight the fine-tuned mechanism that regulates translation and ultimately gene expression. However, whether FMRP represses translation during initiation and/or elongation step is still a controversy. While some laboratories have found FMRP mainly cosedimenting with polyribosomes (Ceman et al. 2003; Khandjian et al. 2004; Stefani et al. 2004), others have found FMRP cosedimenting with mRNPs (Ishizuka et al. 2002; Monzo et al. 2006; Napoli et al. 2008; Papoulas et al. 2010; Siomi et al. 1996; Zalfa et al. 2003), and one laboratory found FMRP equally distributed among the two fractions (Brown et al. 2001). The different distribution could be explained through the association of FMRP to a variety of mRNPs that may aggregate and form different neuronal granules such as P bodies, stress, and transport granules (Anderson and Kedersha 2006; Kanai et al. 2004; Zalfa et al. 2006). The shuttle of FMRP from polysomes to mRNPs might be influenced also by posttranslational modifications (Ceman et al. 2003). However, recent findings suggested that FMRP is involved in the repression of translation initiation through CYFIP1, early identified as a partner of FMRP in neurons (Napoli et al. 2008; Schenck et al. 2003). In fact, CYFIP1 can act as a binding protein for the eukaryotic initiation factor 4E (eIF-4E), sequestering and repressing the assembly of the translation machinery. Specific mRNAs are tethered on CYFIP1-eIF4E by FMRP, and thus, only a subclass of mRNAs is repressed in a CYFIP1-dependent manner. Upon stimuli, CYFIP1-FMRP is released from eIF4E, and translation is activated (Napoli et al. 2008).
3.2 Regulation of mRNA Transport
It is worthwhile to mention that the levels of all FMRP-bound mRNAs are not necessarily translationally dysregulated in the absence of FMRP. Indeed, some experimental evidence indicate that FMRP is also involved in mRNA transport by delivering mRNAs which are thought to be in a dormant state from cell body, through dendrites, to spines where protein synthesis occurs (Fig. 23.3) (Bramham and Wells 2007). Upon synaptic LTD stimulation with DHPG, Fmr1 mRNA is transported to dendrites and newly synthesized in proximity to metabotrobic receptor mGluR5 (Antar et al. 2004; Ferrari et al. 2007; Kao et al. 2010) where is further recruited along microtubules. Following DHPG stimulation, FMRP also interacts with motor proteins on microtubules, promoting the activity-dependent localization of bound mRNAs into synaptic spines (Antar et al. 2004, 2005; Davidovic et al. 2007; Dictenberg et al. 2008; Ferrari et al. 2007; Kanai et al. 2004). Therefore, certain FMRP targets such as Map1b and Sapap4 have been found to be mislocalized in neurons of Fmr1 KO mice (Table 23.2) (Dictenberg et al. 2008; Kao et al. 2010).
FMRP forms part of a protein complex together with translationally arrested mRNAs. FMRP travels within an RNA–protein complex from the cell body to the synapses transporting dendritically localized mRNAs. After synaptic stimulation, FMRP liberates its mRNA targets allowing their local translation. The reversible translational repression and activation of the mRNA targets are regulated by a signaling pathway described in the text (see Sect. 23.3). Transported mRNAs are then locally translated in dendrites contributing to local protein synthesis and synaptic rearrangement that occurs after synaptic stimulation. FMRP can bind its mRNA targets through direct interaction or through noncoding RNAs such as BC1 RNA and microRNAs
3.3 Regulation of mRNA Stability
For some FMRP-bound mRNAs, FMRP is a direct modulator of mRNA stability either by sustaining or preventing mRNA decay (De Rubeis and Bagni 2010). This has been revealed by the difference on the abundance of several mRNAs in Fmr1 KO mice in comparison with wild type (WT) in three independent upscale screenings (Brown et al. 2001; Gantois et al. 2006; Miyashiro et al. 2003). In the first analysis, Brown et al. identified 144 mRNAs dysregulated in lymphoblastoid cells from FXS patients (Brown et al. 2001). Some of these mRNAs were also found to be dysregulated in a second analysis carried out in hippocampus of Fmr1 KO mice (Miyashiro et al. 2003). Further analysis on the shared mRNAs showed that the dendritic localization of the mRNAs coding for the ribosomal component p40/LRP and the G-protein-coupled receptor kinase 4 (GRK4) was unaffected, while for the dystroglycan-associated glycoprotein 1 (DAG1) mRNA, both localization and abundance were reduced (Miyashiro et al. 2003). The latter is an example of how FMRP may act on two different regulation mechanisms on the same mRNA.
Another example which shows how FMRP can regulate an mRNA at different levels is described by its action on PSD-95 mRNA. Specifically, FMRP protects PSD-95 mRNA from decay (Zalfa et al. 2007), and its stabilization is activity dependent. Downregulation of PSD-95 mRNA occurs only in hippocampus and not in cortex, leading to decreased protein levels in hippocampus of Fmr1 KO mice (Zalfa et al. 2007). However, PSD-95 protein levels are also downregulated in cortex of Fmr1 KO mice indicating a defect on the synaptic translation of this mRNA in the cortex (Muddashetty et al. 2007). A different effect of FMRP on stability has been reported for the Nxf1 mRNA. In this context, FMRP together with the nuclear export factor NXF2 facilitates the decay of Nxf1 mRNA in a neuroblastoma cell line (Zhang et al. 2007). Following NXF2 overexpression, Nxf1 is rapidly degraded. However, the degradation is impaired in absence of FMRP, suggesting that FMRP mediates Nxf1 mRNA decay induced by NXF2 (Zhang et al. 2007).
In a genome-wide expression profiling study performed in hippocampus of Fmr1 KO mice (Gantois et al. 2006), 224 mRNAs showed differences in the expression levels between fmr1 KO and WT, being 143 mRNA underexpressed and 81 overexpressed. However, further analyses using microarrays and real-time PCR confirmed that only eight mRNAs were underexpressed in Fmr1 KO mice. Among those eight mRNAs, it is worth to highlight that the GABAA receptor subunit δ shows almost two times of underexpression (Gantois et al. 2006), decrease that was already reported by El Idrissi et al. (2005). These results together with the finding that GABAA receptor subunit δ mRNA was found to be a FMRP target (Dictenberg et al. 2008; Miyashiro et al. 2003) further suggest the role of FMRP as a modulator of this mRNA stability. Noteworthy, eight GABA receptor subunits(α1, α3, α4, β1, β2, γ1, γ2, and δ) were significantly reduced in cortex but not in cerebellum of Fmr1 KO mice (D’Hulst et al. 2006).
All these results lead to the hypothesis that different FMRP protein complexes might play different roles in cortex and hippocampus and that FMRP regulates mRNAs through different mechanisms accordingly to cell type and subcellular localization.
3.4 RNA Sequence–Structure Recognition
Up to now, several different mechanisms through which FMRP binds to its mRNA targets have been described. One mechanism is mediated through the direct binding of FMRP to the mRNAs, as described for PSD-95 mRNA (Zalfa et al. 2007). In this case, FMRP binds to G-rich sequences that can, in some cases (i.e., Map1B mRNA), also be folded as G-quartets (Darnell et al. 2001) Another mechanism is through the binding of FMRP to noncoding RNAs. The first example is via BC1, a small noncoding RNA that acts as a bridge between FMRP and its mRNA targets (Zalfa et al. 2003). A third mechanism of action involves the interaction of FMRP with its targets through microRNAs. Edbauer and colleagues have recently reported that FMRP is associated with at least 12 different miRNAs, and few of them have indeed a relevant effect on spine morphology (Edbauer et al. 2010). NMDA receptor subunit 2A (NR2A) mRNA is an FMRP target that indeed partially depends on miR-125b binding and whose expression has a direct impact on synaptic plasticity (Edbauer et al. 2010). Further studies are required to address which are the other molecular mechanisms mediated by FMRP which affect NR2A translation in neurons.
Considering the involvement of FMRP in the regulation of several genes, both in the cell body and at synapses, as a consequence, its absence causes several cellular phenotypic abnormalities including dendritic spine dysmorphogenesis and behavioral deficits that summarize the impaired molecular synaptic plasticity events. Figure 23.3 summarizes the current model on the multiple FMRP functions in neurons.
4 Learning, Memory, and Behavioral Phenotypes: Learning from the Mouse Model
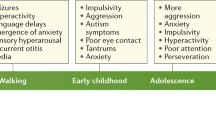
Behavior impairment is one of the most compelling evidence in FXS. Since the animal model for FXS (Fmr1 KO) has been developed (Bakker 1994), several behavioral analysis have been performed on this mouse model. Fmr1 KO mice showed behavioral similarities to the syndrome affecting human individuals (see Table 23.3). This includes hyperactivity, abnormal anxiety-related responses, hyperreactivity to auditory stimuli, abnormal sensorimotor gating, and impaired motor coordination (Bakker 1994; Peier et al. 2000). Learning and memory tests performed with Fmr1 KO mice have shown minor differences compared to their control WT. Morris water maze and radial arm maze tests showed only mild cognitive impairment in Fmr1 KO mice (Bakker 1994; D’Hooge et al. 1997; Dobkin et al. 2000; Kooy et al. 1996; Mineur et al. 2002; Paradee et al. 1999; Peier et al. 2000; Van Dam et al. 2000; Yan et al. 2004), indicating that Fmr1 KO mice have only a slight impairment in spatial learning. Only recently, Fmr1 KO mice generated in the C57 albino genetic background showed a consistent impairment in spatial learning (Baker et al. 2010). Moreover, no impairment in associative aversive learning or memory has been reported for the Fmr1 KO, since they successfully expressed conditioned taste aversion (Nielsen et al. 2009). Transfer of learning, or reversal learning, based on measures of learning rate is not impaired either (Moon et al. 2008). The major impairment revealing a learning and memory deficiency is reported for tests such as object recognition, eyeblink conditioning, and lever press avoidance (Brennan et al. 2006; Koekkoek et al. 2005; Ventura et al. 2004; Yan et al. 2004). In the learning paradigm of conditioned and contextual fear tests, which involve hippocampal and amygdaloid tasks, in two separate studies, Fmr1 KO mice show no significant differences compared to their WT (Peier et al. 2000; Van Dam et al. 2000).
Genetic background has also a major impact on behavioral phenotypes (Bucan and Abel 2002; Wolfer and Lipp 2000). For example, the recently generated Fmr1 KO C57-albino mice showed impaired contextual fear but unaltered conditioned fear (Baker et al. 2010). However, these characteristics were not fully detected in other Fmr1 KO mice generated in other genetic background (FVB-129 mice) (Zhao et al. 2005) (Table 23.3). In prepulse inhibition (PPI) test, measuring sensory gating, Fmr1 KO mice perform even better, and these findings contrast the human phenotype in which there is a decrease in PPI (Frankland et al. 2004; Van Dam et al. 2000). Nonetheless, in the PPI test, Fmr1 KO mice are more reactive than their controls (Chen and Toth 2001; Nielsen et al. 2002a) but only at a near-threshold level of the startle stimulus (Chen and Toth 2001; Nielsen et al. 2002a), indicating a general impairment in sensorimotor gating. This is somehow in agreement with the overall decrease in functioning of the neuronal network of Fragile X patients and also with the decreased ratio of excitatory to inhibitory amino acids observed in the brain of Fmr1 KO mice (Gruss and Braun 2001; Kooy 2003). Importantly, impaired attention and inhibitory control, two features clearly impaired in humans with FXS (Cornish et al. 2008; Garber et al. 2008), have not been studied in the mouse model for FXS, although a heightened emotional reactivity has been described in Fmr1 KO by Moon and colleagues (Moon et al. 2008).
4.1 Social Behavior Phenotypes
Fmr1 KO mice are impaired in social dominance with unfamiliar mice and, even though they show interest in social interaction, they spend a longer period of time than WT mice before approaching an unfamiliar mouse (Spencer et al. 2005). Anxiety is a psychological and physiological state characterized by somatic, emotional, cognitive, and behavioral components observed in patients with FXS (Cornish et al. 2008; Garber et al. 2008). When the Fmr1 KO were studied for this specific state, Mines and colleagues reported that they displayed more anxiety-related behaviors during social interaction (grooming, rearing, and digging) than WT mice (Mines et al. 2010) and reduced social approach and response to social novelty (Liu and Smith 2009). Fmr1 KO males have also a reduced interest in social interaction with novel females (Mineur et al. 2006). Moreover, KO mice seem to prefer social involvement as their WT controls, even though they show social anxiety in approaching the novel conspecific (McNaughton et al. 2008). Fmr1 KO mice also show impairment in social discrimination between positive and negative social interactions (McNaughton et al. 2008). In the acoustic startle reflex test, a response of mind and body to a sudden unexpected stimulus, and a measure of anxiety, Fmr1 KO mice react less that their WT controls (Nielsen et al. 2002a) in contrast with the human behavior, in which FXS patients show an excessive reaction to external stimuli (Jacquemont et al. 2007). Finally, Fmr1 KO mice show hyperactivity, decreased spatial and environmental anxiety-related responses, and altered motor coordination (Bakker 1994; Liu and Smith 2009; Peier et al. 2000). Since sleep problems are common in children with FXS, it was interesting that Zhang and colleagues reported an altered expression of clock genes in the FXS mouse model (Zhang et al. 2008).
Finally, the recently generated mouse model carrying the Ile304Asn point mutation in the C57BL/6J background seems to recapitulate some of the above reported behavioral characteristics (Zang et al. 2009).
Recently, Spencer and colleague also demonstrate that almost all the abovementioned behavioral characteristics depend on the genetic background of the FXS mouse model; therefore, modifier genes may play a role in phenotype expression (Spencer et al. 2011). This implicates a strong correlation with the variable phenotype in patients and also the difficulty to have a reliable animal model for the study of this pathology.
4.2 Rescue of the FXS Behavioral Phenotypes
Genetic, behavioral, and pharmacological rescues have been developed over the last few years. Overexpression of FMRP in the Fmr1 KO mouse model using a YAC vector reduces to some extent the described behavioral abnormalities (Paylor et al. 2008; Spencer et al. 2005, 2008). Improvement in social behavior, such as in chamber test of social interaction, direct social interaction test, and resident–intruder task, has been demonstrated after enhanced neuroligin1 expression in Fmr1 KO, even though no rescue is seen in learning and memory tasks (novel object recognition and plus shaped water maze tests) (Dahlhaus and El-Husseini 2010). One of the FMRP-dysregulated mRNAs encodes for the RGS4 (regulator of G-protein signaling) (Tervonen et al. 2005). Fmr1 × RGS4 double KO shows a rescue of some of the behavioral phenotypes observed in the Fmr1 KO such as tube test for social dominance, conditioned place preference, and reduced susceptibility to audiogenic seizures (Pacey et al. 2009, 2011). Moreover, treatment with group I mGluRs antagonist (MPEP) and lithium, through intervention on glycogen synthase kinase-3 (GSK3), ameliorates several behavioral aspects (Choi et al. 2010; Dolen et al. 2010; Liu et al. 2010; Min et al. 2009; Mines et al. 2010; Yuskaitis et al. 2010). Finally, it has been proposed that the absence of FMRP leads to higher levels of matrix metalloproteinase-9 activity (MMP-9) in the brain. In agreement, minocycline inhibits MMP-9 activity and alleviates behavioral measurement of the Aberrant Behavior Checklist – Community Edition (ABC-C) (irritability subscale, clinical global improvement scale (CGI), and the visual analog scale for behavior (VAS)) and synapse abnormalities in Fmr1 knockout mice (Bilousova et al. 2009). Recently, minocycline administration was shown to provide significant functional benefits to FXS patients. These findings are consistent with the Fmr1 knockout mouse model results, suggesting that minocycline modifies underlying neural defects that account for behavioral abnormalities. As the authors correctly report, a placebo-controlled trial of minocycline in FXS is warranted (Bilousova et al. 2009; Paribello et al. 2010).
As described below, absence of FMRP leads to an impaired GABA pathway due to its control on the mRNAs encoding different GABAA receptor subunits; therefore, a GABAergic approach to treat FXS treatment has been considered through the use of taurine, a GABAA agonist (El Idrissi et al. 2009). The authors show that taurine supplementation to Fragile X mice resulted in a significant improvement in acquisition of a passive avoidance task. Since taurine is an agonist for GABAA receptor, they suggest that chronic activation of GABAA receptors may have beneficial effects in ameliorating the learning deficits characteristic of the Fragile X syndrome (El Idrissi et al. 2009). Additionally, environmental enrichment has been proved of some efficacy in ameliorating the outcome of the pathology (Restivo et al. 2005). In this study, the authors showed that some behavior phenotypes such as habituation to object and motor activity as well as spine morphology were rescued. In addition, an increase of glutamate receptor subunit 1 (GluR1) levels in both genotypes was observed suggesting that FMRP-independent pathways activating glutamatergic signaling are preserved in Fmr1 KO mice and that they can be elicited by environmental stimulation (Restivo et al. 2005). These findings indicate that the environment is of extreme importance for the patients. A follow-up study showed that in Fmr1 KO mice that were raised in enriched environments, LTP was restored to WT levels, indicating that mechanisms for synaptic plasticity are in place in the Fmr1 KO mouse but require stronger neuronal activity to be triggered (Meredith et al. 2007). Patients with FXS show in about 20% of the cases epileptic seizures (Garber et al. 2008). Even if the mouse model for FXS does not display spontaneous seizures, it is susceptible to audiogenic-induced seizures (Bakker 1994; Chen and Toth 2001; Kooy 2003; Kooy et al. 1996; Musumeci et al. 2000). Although seizure age dependency is still debated, a trend in the impaired response to acoustic seizures at P17 and P21 (postnatal days 17 and 21) has been reported (Chen and Toth 2001; Kooy 2003; Musumeci et al. 2000). This effect may be related to an increased cortical excitability or due to a deficit in long-term plasticity (Kooy 2003). Chemically induced seizures, by kainic acid, bicuculline, and pentylenetatrazole injection, do not show difference between Fmr1 KO and their controls, suggesting that Fmr1 KO mice have specific susceptibility to audiogenic stimuli (Chen and Toth 2001; Todd and Mack 2000) This is in agreement with a possible developmental impairment of the auditory system in Fragile X mice (Brown et al. 2010) proposed by Chen and Toth (Chen and Toth 2001). Only recently, a study on amygdala showed that Fmr1 KO mice have a more accelerated kindling development and longer electrographic seizure duration. Both NMDA antagonist, MK-801, and mGluR5 antagonist, MPEP, were able to repress accelerated rate of kindling development (Qiu et al. 2009).
4.3 Electrophysiological Phenotypes
Due to the learning and memory alterations, characteristics of FXS, synaptic plasticity events monitored through ex vivo LTP and LTD paradigms have been extensively investigated in Fmr1 KO mice.
Effects on LTP. In the Fmr1 KO mice, LTP has been found altered in hippocampus only after theta burst stimulation (Lauterborn et al. 2007), a stimulation able to induce LTP in close resemblance to physiological hippocampal frequency of theta rhythm (5–10 Hz) (Capocchi et al. 1992). Only recently, a reduction of LTP in dentate gyrus has been observed in Fmr1 KO mice possibly due to a reduction of NMDA excitatory postsynaptic currents (EPSCs), which is a consequence of a reduced ratio of NMDA/AMPA receptors (Eadie et al. 2010; Yun and Trommer 2011). On the other hand, high-frequency stimulation (100–400 Hz) does not affect LTP in hippocampus of Fmr1 KO mice (Godfraind et al. 1996; Li et al. 2002; Paradee et al. 1999; Zhang et al. 2009). At the level of cortex, the LTP responses are different and indeed appear impaired in the Fmr1 KO mice (Hayashi et al. 2007; Larson et al. 2005; Li et al. 2002; Meredith et al. 2007; Zhao et al. 2005). Additionally, induction of LTP has also been shown to be altered in lateral amygdala (Suvrathan et al. 2010; Zhao et al. 2005). FMRP is required for glycine-induced LTP (Gly-LTP) in the CA1 of hippocampus. This form of LTP requires activation of postsynaptic NMDA receptors and metabotropic glutamatergic receptors, as well as the subsequent activation of extracellular signal-regulated kinase (ERK) 1/2. Genetic deletion of FMRP interrupts the phosphorylation of ERK1/2, suggesting the possible role of FMRP in the regulation of the activity of ERK1/2 (Shang et al. 2009).
Effects on LTD. LTD is altered in hippocampus of Fmr1 KO mice either applying low-frequency-paired-pulse stimulation (PP-LFS), which is rescued by FMRP replacement through viral strategy (Zeier et al. 2009), or with application of the group I mGluR agonist DHPG (Huber et al. 2000). mGluR5-LTD and FMRP are also connected: DHPG induces FMRP expression in synaptosomes and cultured neurons (Antar et al. 2004; Ferrari et al. 2007; Kao et al. 2010; Weiler et al. 1997), and DHPG-induced LTD is increased in Fmr1 KO mice (Hou et al. 2006; Huber et al. 2002). Therefore, mGluR5-induced LTD in the context of Fragile X has received a large attention. CA1 LTD is reported to be translation dependent and transcription independent when explored in WT mice (Hou and Klann 2004; Huber et al. 2000, 2002), in line with the signaling pathways described above and activated by mGluR5 which involve protein synthesis (Dolen et al. 2010). mGluR LTD occurs in a protein-synthesis-dependent manner and correlates glutamatergic receptors (NMDA and AMPA) regulation to morphologic changes in spine number, shape, and size (Dolen et al. 2010).
mGluR-induced LTD (via DHPG) is increased in Fmr1 KO mice (Huber et al. 2002) and is protein synthesis independent, possibly due to deregulated local protein synthesis (Lu et al. 2004; Muddashetty et al. 2007; Zalfa et al. 2007), which in turn enhances a constant AMPAR internalization (Huber et al. 2002; Nosyreva and Huber 2006; Ronesi and Huber 2008). This mechanism has been described as the base of the so-called mGluR theory (Bear et al. 2004) which hypothesize that an excessive group 1 mGluRs activation, upstream to the lack of FMRP, increases the protein synthesis and induces excessive AMPAR internalization, responsible for the consequent increase in LTD. The theory has been validated by a genetic rescue of the Fmr1 KO mouse model crossing it with the heterozygous mouse for mGluR5. The crossed Fmr1 KO/mGluR5 (+/−) mice demonstrated amelioration of several mouse phenotypes. Specifically, 50% reduction of mGluR5 level of expression in the Fmr1 KO background restored the altered protein expression, which in turn reduced the abnormal response to mGluR5-induced LTD, rescued the abnormal spine morphology, and ameliorated some behavioral phenotypes including a reduced incidence of the audiogenic seizures (AGS) (Dolen et al. 2007).
Nevertheless, the excessive activity of mGluR5 seems not to be the only possible cause of seizures in the Fmr1 KO mouse model. As previously mentioned, another possible cause to consider is the deficit of GABA neurotransmission. Indeed, several lines of research have proposed that absence of FMRP leads to a dysfunction in the GABAergic system (Olmos-Serrano et al. 2010). Recent studies demonstrate that GABAergic inhibition is impaired at cellular (Selby et al. 2007), physiological (Centonze et al. 2008; Curia et al. 2009), and molecular level (Curia et al. 2009; D’Hulst et al. 2006; El Idrissi et al. 2005). Therefore, decrease of interneuron number, altered GABAergic transmission, and/or altered GABAA subunit expression may be the cause for epileptic seizures and/or EEG abnormalities associated to FXS. Moreover, an involvement of G-protein-coupled GABAB receptors demonstrated to be effective in attenuating the AGS phenotype in Fmr1 KO mice (Pacey et al. 2009). The mechanism, possibly involved in efficacy for GABAB receptors, seems to be the coupling of GABAB receptors to regulator of RGS4 (Fowler et al. 2007) which in turn results as an inhibitor of Gp1 mGluRs (Saugstad et al. 1998). In agreement with this hypothesis, a double KO for Fmr1 and RGS4 mouse model has reduced susceptibility to seizures (Pacey et al. 2009).
4.4 Spine Dysgenesis
Alterations of dendritic spines represent a common hallmark of mental retardation diseases and other synaptopathies (Purpura 1974). Although FXS is not characterized by gross brain defects, a consistent microanatomical phenotype is an increased spine density and an altered ratio of mature and immature spines (Comery et al. 1997; Irwin et al. 2001). During development, spines are stabilized and change their dynamic properties and morphology. Motility and turnover decrease, and thin, filopodia-like protrusions mature in stubby and mushroom-like shapes (Portera-Cailliau et al. 2003). A variety of intermediate shapes can exist, giving rise to a filopodia–spine continuum (Irwin et al. 2001).
The first evidence of spine dysgenesis in individuals with FXS was obtained by Golgi impregnation of autopsy material from a 62-year-old FXS patient (Rudelli et al. 1985). This analysis revealed long, tortuous, and thin dendritic spines on apical dendrites of pyramidal neurons from layers III and V of the parieto-occipital cortex (Rudelli et al. 1985). Further studies of three FXS adult individuals estimated higher spine density and length along the entire dendritic tree of cortical layer V pyramidal neurons (Irwin et al. 2001).
Mouse models of FXS (Fmr1 KO) recapitulate the spine dysgenesis observed in patients. Mutant mice present increase density of long, immature spines in visual cortex, somatosensory cortex, and hippocampal dentate gyrus (Comery et al. 1997; Dolen et al. 2007; Galvez and Greenough 2005; Grossman et al. 2010; Irwin et al. 2002; Nimchinsky et al. 2001; Restivo et al. 2005). Spine defects in cortical neurons were detected during early postnatal development (1–3 weeks) and adulthood but not in 4-week-old mice (Galvez and Greenough 2005; Nimchinsky et al. 2001). However, in contrast with neocortex, spine abnormalities in the dentate gyrus remain constant during development (Grossman et al. 2010). Data obtained on ex vivo and in vitro systems do not consistently corroborate in vivo observations. Spine defects were not detected in neocortical and hippocampal organotypic cultures (Nimchinsky et al. 2001; Pfeiffer et al. 2010). Increased spine length and excessive filopodia protrusions were reported in primary Fmr1 KO hippocampal neurons (Antar et al. 2006; Bilousova et al. 2009), although earlier reports produced divergent results (Braun and Segal 2000).
Excess of spines with immature morphology may be due to defects in spine dynamics and maturation, especially altered pruning (Galvez et al. 2003). Two recent studies examined spine plasticity in living Fmr1 KO animals using transcranial two-photon imaging of somatosensory cortex. Both reports highlighted that the major abnormality is the augmented spine turnover, a process that includes formation of new spines and elimination of existing spines (Cruz-Martin et al. 2010; Pan et al. 2010). In Fmr1 KO mice, spine turnover fails to rapidly decrease during the first 2 postnatal weeks, leading to a delay in spine stabilization and transition from immature to mature spine types (Cruz-Martin et al. 2010). Increased spine turnover is maintained in late development (4 postnatal weeks) and in adulthood and may be due to a larger population of short-lived spines observed in KO mice (Pan et al. 2010). Since these transient spines display smaller head and longer neck, they could contribute to the immature spine morphology in KO animals (Pan et al. 2010).
Spine dynamics are known to change in response to experience-dependent modulation of specific circuits in the somatosensory and visual cortex (Holtmaat and Svoboda 2009). Although Fmr1 KO animals display hypersensitivity to sensory stimuli (Chen and Toth 2001), this effect does not cause the enhanced spine dynamics, which in fact was not hampered by somatosensory deprivation (whisker trimming) (Pan et al. 2010). Furthermore, KO mice lack spine plasticity to somatosensory modulation (Pan et al. 2010). However, other circuits of experience-dependent neuronal plasticity seem to be preserved, since environmental enrichment rescues the spine abnormalities in visual cortex (Restivo et al. 2005).
Evidence of defects in spine maturation are compatible with the overall decrease of functional synapses, measured as dendritic protrusions juxtaposed with presynaptic markers in cultured hippocampal neurons (Antar et al. 2006; Braun and Segal 2000). Of interest, loss of synapses in Fmr1 KO mice is also corroborated by electrophysiological data (Pfeiffer and Huber 2007; Pfeiffer et al. 2010).
The mechanisms leading to spine dysgenesis are not fully understood. Despite recent reports suggesting the involvement of the transcription regulator MEF2 (Pfeiffer et al. 2010) and neuronal microRNAs (Edbauer et al. 2010), the spine phenotype is likely multifactorial. As mentioned above, FMRP regulates the synthesis of a variety of proteins crucial for proper synaptic morphology and functionality (Bassell and Warren 2008). However, interfering with some of the signaling pathways altered in Fmr1 KO mice – group I mGluRs, PI3K, and PAK1 RGS4 – is effective to partially rescue the spine anomalies.
As for the mGluR cascade, DHPG administration in cultured hippocampal neurons increases spine length in a protein-synthesis-dependent manner (Vanderklish and Edelman 2002). Hampering the excessive mGluR signaling by administration of the antagonist MPEP (de Vrij et al. 2008; Su et al. 2010) or genetic reduction of mGluR5 (Dolen et al. 2007) ameliorates the spine phenotype in Fmr1 KO neurons. Furthermore, inhibition of PI3K, a signaling molecule excessively translated in the absence of FMRP, reduces spine density in cultured neurons (Gross et al. 2010). Similarly, inhibition of PAK1, a signaling cascade controlling actin cytoskeleton, partially rescues the defects in spine density and length (Hayashi et al. 2007). Finally, pharmacological treatments with lithium and minocycline, whose mechanisms are not fully elucidated, may also alleviate the spine phenotype in FXS mice (Bilousova et al. 2009; Liu et al. 2010).
References
Abrahams, B. S., & Geschwind, D. H. (2008). Advances in autism genetics: On the threshold of a new neurobiology. Nature Reviews Genetics, 9, 341–355.
Allen, E. G., He, W., Yadav-Shah, M., & Sherman, S. L. (2004). A study of the distributional characteristics of FMR1 transcript levels in 238 individuals. Human Genetics, 114, 439–447.
Anderson, P., & Kedersha, N. (2006). RNA granules. The Journal of Cell Biology, 172, 803–808.
Antar, L. N., Afroz, R., Dictenberg, J. B., Carroll, R. C., & Bassell, G. J. (2004). Metabotropic glutamate receptor activation regulates fragile X mental retardation protein and FMR1 mRNA localization differentially in dendrites and at synapses. Journal of Neuroscience, 24, 2648–2655.
Antar, L. N., Dictenberg, J. B., Plociniak, M., Afroz, R., & Bassell, G. J. (2005). Localization of FMRP-associated mRNA granules and requirement of microtubules for activity-dependent trafficking in hippocampal neurons. Genes, Brain, and Behavior, 4, 350–359.
Antar, L. N., Li, C., Zhang, H., Carroll, R. C., & Bassell, G. J. (2006). Local functions for FMRP in axon growth cone motility and activity-dependent regulation of filopodia and spine synapses. Molecular and Cellular Neurosciences, 32, 37–48.
Ashley, C. T., Jr., Wilkinson, K. D., Reines, D., & Warren, S. T. (1993). FMR1 protein: Conserved RNP family domains and selective RNA binding. Science, 262, 563–566.
Bacalman, S., Farzin, F., Bourgeois, J. A., Cogswell, J., Goodlin-Jones, B. L., Gane, L. W., Grigsby, J., Leehey, M. A., Tassone, F., & Hagerman, R. J. (2006). Psychiatric phenotype of the fragile X-associated tremor/ataxia syndrome (FXTAS) in males: Newly described fronto-subcortical dementia. The Journal of Clinical Psychiatry, 67, 87–94.
Bagni, C., & Greenough, W. T. (2005). From mRNP trafficking to spine dysmorphogenesis: The roots of fragile X syndrome. Nature Reviews Neuroscience, 6, 376–387.
Baker, K. B., Wray, S. P., Ritter, R., Mason, S., Lanthorn, T. H., & Savelieva, K. V. (2010). Male and female Fmr1 knockout mice on C57 albino background exhibit spatial learning and memory impairments. Genes, Brain, and Behavior, 9, 562–574.
Bardoni, B., Willemsen, R., Weiler, I. J., Schenck, A., Severijnen, L. A., Hindelang, C., Lalli, E., & Mandel, J. L. (2003). NUFIP1 (nuclear FMRP interacting protein 1) is a nucleocytoplasmic shuttling protein associated with active synaptoneurosomes. Experimental Cell Research, 289, 95–107.
Bassell, G. J., & Warren, S. T. (2008). Fragile X syndrome: Loss of local mRNA regulation alters synaptic development and function. Neuron, 60, 201–214.
Bear, M. F., Huber, K. M., & Warren, S. T. (2004). The mGluR theory of fragile X mental retardation. Trends in Neurosciences, 27, 370–377.
Belmonte, M. K., & Bourgeron, T. (2006). Fragile X syndrome and autism at the intersection of genetic and neural networks. Nature Neuroscience, 9, 1221–1225.
Berman, R. F., & Willemsen, R. (2009). Mouse models of fragile x-associated tremor ataxia. Journal of Investigative Medicine, 57, 837–841.
Berry-Kravis, E. (2002). Epilepsy in fragile X syndrome. Developmental Medicine and Child Neurology, 44, 724–728.
Bilousova, T. V., Dansie, L., Ngo, M., Aye, J., Charles, J. R., Ethell, D. W., & Ethell, I. M. (2009). Minocycline promotes dendritic spine maturation and improves behavioural performance in the fragile X mouse model. Journal of Medical Genetics, 46, 94–102.
Bramham, C. R., & Wells, D. G. (2007). Dendritic mRNA: Transport, translation and function. Nature Reviews Neuroscience, 8, 776–789.
Braun, K., & Segal, M. (2000). FMRP involvement in formation of synapses among cultured hippocampal neurons. Cerebral Cortex, 10, 1045–1052.
Brennan, F. X., Albeck, D. S., & Paylor, R. (2006). Fmr1 knockout mice are impaired in a leverpress escape/avoidance task. Genes, Brain, and Behavior, 5, 467–471.
Brouwer, J. R., Huizer, K., Severijnen, L. A., Hukema, R. K., Berman, R. F., Oostra, B. A., & Willemsen, R. (2008). CGG-repeat length and neuropathological and molecular correlates in a mouse model for fragile X-associated tremor/ataxia syndrome. Journal of Neurochemistry, 107, 1671–1682.
Brown, V., Jin, P., Ceman, S., Darnell, J. C., O’Donnell, W. T., Tenenbaum, S. A., Jin, X., Feng, Y., Wilkinson, K. D., Keene, J. D., Darnell, R. B., & Warren, S. T. (2001). Microarray identification of FMRP-associated brain mRNAs and altered mRNA translational profiles in fragile X syndrome. Cell, 107, 477–487.
Bucan, M., & Abel, T. (2002). The mouse: Genetics meets behaviour. Nature Reviews Genetics, 3, 114–123.
Brown, M. R., Kronengold, J., Gazula, V.-R., Chen, Y., Strumbos, J. G., Sigworth, F. J., Navaratnam, D., & Kaczmarek. L. K., (2010). Fragile X mental retardation protein controls gating of the sodium-activated potassium channel Slack. Nature Neuroscience Vol 13 (7) Jul 2010.
Bakker, C. (1994). Fmr1 knockout mice: A model to study fragile X mental retardation. The Dutch-Belgian fragile X consortium. Cell, 78, 23–33.
Capocchi, G., Zampolini, M., & Larson, J. (1992). Theta burst stimulation is optimal for induction of LTP at both apical and basal dendritic synapses on hippocampal CA1 neurons. Brain Research, 591, 332–336.
Ceman, S., O’Donnell, W. T., Reed, M., Patton, S., Pohl, J., & Warren, S. T. (2003). Phosphorylation influences the translation state of FMRP-associated polyribosomes. Human Molecular Genetics, 12, 3295–3305.
Centonze, D., Rossi, S., Mercaldo, V., Napoli, I., Ciotti, M. T., De Chiara, V., Musella, A., Prosperetti, C., Calabresi, P., Bernardi, G., & Bagni, C. (2008). Abnormal striatal GABA transmission in the mouse model for the fragile X syndrome. Biological Psychiatry, 63, 963–973.
Chen, L., & Toth, M. (2001). Fragile X mice develop sensory hyperreactivity to auditory stimuli. Neuroscience, 103, 1043–1050.
Cheever, A., Ceman, S., (2009). Phosphorylation of FMRP inhibits association with Dicer. RNA. 15(3):362–6.
Choi, C. H., Schoenfeld, B. P., Bell, A. J., Hinchey, P., Kollaros, M., Gertner, M. J., Woo, N. H., Tranfaglia, M. R., Bear, M. F., Zukin, R. S., McDonald, T. V., Jongens, T. A., & McBride, S. M. (2010). Pharmacological reversal of synaptic plasticity deficits in the mouse model of fragile X syndrome by group II mGluR antagonist or lithium treatment. Brain Research, 1380, 106–119.
Comery, T. A., Harris, J. B., Willems, P. J., Oostra, B. A., Irwin, S. A., Weiler, I. J., & Greenough, W. T. (1997). Abnormal dendritic spines in fragile X knockout mice: Maturation and pruning deficits. Proceedings of the National Academy of Sciences of the United States of America, 94, 5401–5404.
Cornish, K. M., Li, L., Kogan, C. S., Jacquemont, S., Turk, J., Dalton, A., Hagerman, R. J., & Hagerman, P. J. (2008). Age-dependent cognitive changes in carriers of the fragile X syndrome. Cortex, 44, 628–636.
Cruz-Martin, A., Crespo, M., & Portera-Cailliau, C. (2010). Delayed stabilization of dendritic spines in fragile X mice. Journal of Neuroscience, 30, 7793–7803.
Curia, G., Papouin, T., Seguela, P., & Avoli, M. (2009). Downregulation of tonic GABAergic inhibition in a mouse model of fragile X syndrome. Cerebral Cortex, 19, 1515–1520.
D’Hooge, R., Nagels, G., Franck, F., Bakker, C. E., Reyniers, E., Storm, K., Kooy, R. F., Oostra, B. A., Willems, P. J., & De Deyn, P. P. (1997). Mildly impaired water maze performance in male Fmr1 knockout mice. Neuroscience, 76, 367–376.
D’Hulst, C., De Geest, N., Reeve, S. P., Van Dam, D., De Deyn, P. P., Hassan, B. A., & Kooy, R. F. (2006). Decreased expression of the GABAA receptor in fragile X syndrome. Brain Research, 1121, 238–245.
Dahlhaus, R., & El-Husseini, A. (2010). Altered neuroligin expression is involved in social deficits in a mouse model of the fragile X syndrome. Behavioural Brain Research, 208, 96–105.
Darnell, J. C., Jensen, K. B., Jin, P., Brown, V., Warren, S. T., & Darnell, R. B. (2001). Fragile X mental retardation protein targets G quartet mRNAs important for neuronal function. Cell, 107, 489–499.
Davidovic, L., Jaglin, X. H., Lepagnol-Bestel, A. M., Tremblay, S., Simonneau, M., Bardoni, B., & Khandjian, E. W. (2007). The fragile X mental retardation protein is a molecular adaptor between the neurospecific KIF3C kinesin and dendritic RNA granules. Human Molecular Genetics, 16, 3047–3058.
De Boulle, K., Verkerk, A. J., Reyniers, E., Vits, L., Hendrickx, J., Van Roy, B., Van den Bos, F., de Graaff, E., Oostra, B. A., & Willems, P. J. (1993). A point mutation in the FMR-1 gene associated with fragile X mental retardation. Nature Genetics, 3, 31–35.
De Rubeis, S., & Bagni, C. (2010). Fragile X mental retardation protein control of neuronal mRNA metabolism: Insights into mRNA stability. Molecular and Cellular Neurosciences, 43, 43–50.
de Vrij, F. M., Levenga, J., van der Linde, H. C., Koekkoek, S. K., De Zeeuw, C. I., Nelson, D. L., Oostra, B. A., & Willemsen, R. (2008). Rescue of behavioral phenotype and neuronal protrusion morphology in Fmr1 KO mice. Neurobiology of Disease, 31, 127–132.
den Broeder, M. J., van der Linde, H., Brouwer, J. R., Oostra, B. A., Willemsen, R., & Ketting, R. F. (2009). Generation and characterization of FMR1 knockout zebrafish. PLoS One, 4, e7910.
Dictenberg, J. B., Swanger, S. A., Antar, L. N., Singer, R. H., & Bassell, G. J. (2008). A direct role for FMRP in activity-dependent dendritic mRNA transport links filopodial-spine morphogenesis to fragile X syndrome. Developmental Cell, 14, 926–939.
Dobkin, C., Rabe, A., Dumas, R., El Idrissi, A., Haubenstock, H., & Brown, W. T. (2000). Fmr1 knockout mouse has a distinctive strain-specific learning impairment. Neuroscience, 100, 423–429.
Dockendorff, T. C., Su, H. S., McBride, S. M., Yang, Z., Choi, C. H., Siwicki, K. K., Sehgal, A., & Jongens, T. A. (2002). Drosophila lacking dfmr1 activity show defects in circadian output and fail to maintain courtship interest. Neuron, 34, 973–984.
Dolen, G., Carpenter, R. L., Ocain, T. D., & Bear, M. F. (2010). Mechanism-based approaches to treating fragile X. Pharmacology and Therapeutics, 127, 78–93.
Dolen, G., Osterweil, E., Rao, B. S., Smith, G. B., Auerbach, B. D., Chattarji, S., & Bear, M. F. (2007). Correction of fragile X syndrome in mice. Neuron, 56, 955–962.
Eadie, B. D., Cushman, J., Kannangara, T. S., Fanselow, M. S., & Christie, B. R. (2010). NMDA receptor hypofunction in the dentate gyrus and impaired context discrimination in adult Fmr1 knockout mice. Hippocampus. 3 NOV 2010, DOI: 10.1002/hipo.20890.
Eberhart, D. E., Malter, H. E., Feng, Y., & Warren, S. T. (1996). The fragile X mental retardation protein is a ribonucleoprotein containing both nuclear localization and nuclear export signals. Human Molecular Genetics, 5, 1083–1091.
Edbauer, D., Neilson, J. R., Foster, K. A., Wang, C. F., Seeburg, D. P., Batterton, M. N., Tada, T., Dolan, B. M., Sharp, P. A., & Sheng, M. (2010). Regulation of synaptic structure and function by FMRP-associated microRNAs miR-125b and miR-132. Neuron, 65, 373–384.
El Idrissi, A., Boukarrou, L., Dokin, C., & Brown, W. T. (2009). Taurine improves congestive functions in a mouse model of fragile X syndrome. Advances in Experimental Medicine and Biology, 643, 191–198.
El Idrissi, A., Ding, X. H., Scalia, J., Trenkner, E., Brown, W. T., & Dobkin, C. (2005). Decreased GABA(A) receptor expression in the seizure-prone fragile X mouse. Neuroscience Letters, 377, 141–146.
Entezam, A., Biacsi, R., Orrison, B., Saha, T., Hoffman, G. E., Grabczyk, E., Nussbaum, R. L., & Usdin, K. (2007). Regional FMRP deficits and large repeat expansions into the full mutation range in a new fragile X premutation mouse model. Gene, 395, 125–134.
Farzin, F., Perry, H., Hessl, D., Loesch, D., Cohen, J., Bacalman, S., Gane, L., Tassone, F., Hagerman, P., & Hagerman, R. (2006). Autism spectrum disorders and attention-deficit/hyperactivity disorder in boys with the fragile X premutation. Journal of Developmental and Behavioral Pediatrics, 27, S137–144.
Feng, Y., Zhang, F., Lokey, L. K., Chastain, J. L., Lakkis, L., Eberhart, D., & Warren, S. T. (1995). Translational suppression by trinucleotide repeat expansion at FMR1. Science, 268, 731–734.
Fernandez-Carvajal, I., Lopez Posadas, B., Pan, R., Raske, C., Hagerman, P. J., & Tassone, F. (2009). Expansion of an FMR1 grey-zone allele to a full mutation in two generations. Journal of Molecular Diagnostics, 11, 306–310.
Ferrari, F., Mercaldo, V., Piccoli, G., Sala, C., Cannata, S., Achsel, T., & Bagni, C. (2007). The fragile X mental retardation protein-RNP granules show an mGluR-dependent localization in the post-synaptic spines. Molecular and Cellular Neurosciences, 34, 343–354.
Flavell, S. W., & Greenberg, M. E. (2008). Signaling mechanisms linking neuronal activity to gene expression and plasticity of the nervous system. Annual Review of Neuroscience, 31, 563–590.
Fombonne, E. (2005). Epidemiology of autistic disorder and other pervasive developmental disorders. The Journal of Clinical Psychiatry, 66(Suppl 10), 3–8.
Fowler, C. E., Aryal, P., Suen, K. F., & Slesinger, P. A. (2007). Evidence for association of GABA(B) receptors with Kir3 channels and regulators of G protein signalling (RGS4) proteins. The Journal of Physiology, 580, 51–65.
Frankland, P. W., Wang, Y., Rosner, B., Shimizu, T., Balleine, B. W., Dykens, E. M., Ornitz, E. M., & Silva, A. J. (2004). Sensorimotor gating abnormalities in young males with fragile X syndrome and Fmr1-knockout mice. Molecular Psychiatry, 9, 417–425.
Galvez, R., Gopal, A. R., & Greenough, W. T. (2003). Somatosensory cortical barrel dendritic abnormalities in a mouse model of the fragile X mental retardation syndrome. Brain Research, 971, 83–89.
Galvez, R., & Greenough, W. T. (2005). Sequence of abnormal dendritic spine development in primary somatosensory cortex of a mouse model of the fragile X mental retardation syndrome. American Journal of Medical Genetics A, 135, 155–160.
Gantois, I., Vandesompele, J., Speleman, F., Reyniers, E., D’Hooge, R., Severijnen, L. A., Willemsen, R., Tassone, F., & Kooy, R. F. (2006). Expression profiling suggests underexpression of the GABA(A) receptor subunit delta in the fragile X knockout mouse model. Neurobiology of Disease, 21, 346–357.
Garber, K. B., Visootsak, J., & Warren, S. T. (2008). Fragile X syndrome. European Journal of Human Genetics, 16, 666–672.
Garcia-Nonell, C., Ratera, E. R., Harris, S., Hessl, D., Ono, M. Y., Tartaglia, N., Marvin, E., Tassone, F., & Hagerman, R. J. (2008). Secondary medical diagnosis in fragile X syndrome with and without autism spectrum disorder. American Journal of Medical Genetics A, 146A, 1911–1916.
Gedeon, A. K., Baker, E., Robinson, H., Partington, M. W., Gross, B., Manca, A., Korn, B., Poustka, A., Yu, S., Sutherland, G. R., et al. (1992). Fragile X syndrome without CCG amplification has an FMR1 deletion. Nature Genetics, 1, 341–344.
Gladding, C. M., Fitzjohn, S. M., & Molnar, E. (2009). Metabotropic glutamate receptor-mediated long-term depression: Molecular mechanisms. Pharmacological Reviews, 61, 395–412.
Godfraind, J. M., Reyniers, E., De Boulle, K., D’Hooge, R., De Deyn, P. P., Bakker, C. E., Oostra, B. A., Kooy, R. F., & Willems, P. J. (1996). Long-term potentiation in the hippocampus of fragile X knockout mice. American Journal of Medical Genetics, 64, 246–251.
Greco, C. M., Berman, R. F., Martin, R. M., Tassone, F., Schwartz, P. H., Chang, A., Trapp, B. D., Iwahashi, C., Brunberg, J., Grigsby, J., Hessl, D., Becker, E. J., Papazian, J., Leehey, M. A., Hagerman, R. J., & Hagerman, P. J. (2006). Neuropathology of fragile X-associated tremor/ataxia syndrome (FXTAS). Brain, 129, 243–255.
Greco, C. M., Hagerman, R. J., Tassone, F., Chudley, A. E., Del Bigio, M. R., Jacquemont, S., Leehey, M., & Hagerman, P. J. (2002). Neuronal intranuclear inclusions in a new cerebellar tremor/ataxia syndrome among fragile X carriers. Brain, 125, 1760–1771.
Grishin, N. V. (2001). KH domain: One motif, two folds. Nucleic Acids Research, 29, 638–643.
Gross, C., Nakamoto, M., Yao, X., Chan, C. B., Yim, S. Y., Ye, K., Warren, S. T., & Bassell, G. J. (2010). Excess phosphoinositide 3-kinase subunit synthesis and activity as a novel therapeutic target in fragile X syndrome. Journal of Neuroscience, 30, 10624–10638.
Grossman, A. W., Aldridge, G. M., Lee, K. J., Zeman, M. K., Jun, C. S., Azam, H. S., Arii, T., Imoto, K., Greenough, W. T., & Rhyu, I. J. (2010). Developmental characteristics of dendritic spines in the dentate gyrus of Fmr1 knockout mice. Brain Research, 1355, 221–227.
Gruss, M., & Braun, K. (2001). Alterations of amino acids and monoamine metabolism in male Fmr1 knockout mice: A putative animal model of the human fragile X mental retardation syndrome. Neural Plasticity, 8, 285–298.
Hagerman, R. J., & Hagerman, P. J. (2002). The fragile X premutation: Into the phenotypic fold. Current Opinion in Genetics and Development, 12, 278–283.
Hagerman, P. J., & Hagerman, R. J. (2007). Fragile X-associated tremor/ataxia syndrome – An older face of the fragile X gene. Nature Clinical Practice Neurology, 3, 107–112.
Hagerman, R. J., Leehey, M., Heinrichs, W., Tassone, F., Wilson, R., Hills, J., Grigsby, J., Gage, B., & Hagerman, P. J. (2001). Intention tremor, parkinsonism, and generalized brain atrophy in male carriers of fragile X. Neurology, 57, 127–130.
Hallahan, B. P., Craig, M. C., Toal, F., Daly, E. M., Moore, C. J., Ambikapathy, A., Robertson, D., Murphy, K. C., & Murphy, D. G. (2010). In vivo brain anatomy of adult males with fragile X syndrome: an MRI study. NeuroImage, 54(1):16–24.
Harris, S. W., Hessl, D., Goodlin-Jones, B., Ferranti, J., Bacalman, S., Barbato, I., Tassone, F., Hagerman, P. J., Herman, H., & Hagerman, R. J. (2008). Autism profiles of males with fragile X syndrome. American Journal on Mental Retardation, 113, 427–438.
Hatton, D. D., Sideris, J., Skinner, M., Mankowski, J., Bailey, D. B., Jr., Roberts, J., & Mirrett, P. (2006). Autistic behavior in children with fragile X syndrome: Prevalence, stability, and the impact of FMRP. American Journal of Medical Genetics A, 140A, 1804–1813.
Hayashi, M. L., Rao, B. S., Seo, J. S., Choi, H. S., Dolan, B. M., Choi, S. Y., Chattarji, S., & Tonegawa, S. (2007). Inhibition of p21-activated kinase rescues symptoms of fragile X syndrome in mice. Proceedings of the National Academy of Sciences of the United States of America, 104, 11489–11494.
Hernandez, R. N., Feinberg, R. L., Vaurio, R., Passanante, N. M., Thompson, R. E., & Kaufmann, W. E. (2009). Autism spectrum disorder in fragile X syndrome: A longitudinal evaluation. American Journal of Medical Genetics A, 149A, 1125–1137.
Holt, C. E., & Bullock, S. L. (2009). Subcellular mRNA localization in animal cells and why it matters. Science, 326, 1212–1216.
Holtmaat, A., & Svoboda, K. (2009). Experience-dependent structural synaptic plasticity in the mammalian brain. Nature Reviews Neuroscience, 10, 647–658.
Hou, L., Antion, M. D., Hu, D., Spencer, C. M., Paylor, R., & Klann, E. (2006). Dynamic translational and proteasomal regulation of fragile X mental retardation protein controls mGluR-dependent long-term depression. Neuron, 51, 441–454.
Hou, L., & Klann, E. (2004). Activation of the phosphoinositide 3-kinase-Akt-mammalian target of rapamycin signaling pathway is required for metabotropic glutamate receptor-dependent long-term depression. Journal of Neuroscience, 24, 6352–6361.
Huber, K. M., Gallagher, S. M., Warren, S. T., & Bear, M. F. (2002). Altered synaptic plasticity in a mouse model of fragile X mental retardation. Proceedings of the National Academy of Sciences of the United States of America, 99, 7746–7750.
Huber, K. M., Kayser, M. S., & Bear, M. F. (2000). Role for rapid dendritic protein synthesis in hippocampal mGluR-dependent long-term depression. Science, 288, 1254–1257.
Inoue, S., Shimoda, M., Nishinokubi, I., Siomi, M. C., Okamura, M., Nakamura, A., Kobayashi, S., Ishida, N., & Siomi, H. (2002). A role for the Drosophila fragile X-related gene in circadian output. Current Biology, 12, 1331–1335.
Irwin, S. A., Idupulapati, M., Gilbert, M. E., Harris, J. B., Chakravarti, A. B., Rogers, E. J., Crisostomo, R. A., Larsen, B. P., Mehta, A., Alcantara, C. J., Patel, B., Swain, R. A., Weiler, I. J., Oostra, B. A., & Greenough, W. T. (2002). Dendritic spine and dendritic field characteristics of layer V pyramidal neurons in the visual cortex of fragile-X knockout mice. American Journal of Medical Genetics, 111, 140–146.
Irwin, S. A., Patel, B., Idupulapati, M., Harris, J. B., Crisostomo, R. A., Larsen, B. P., Kooy, F., Willems, P. J., Cras, P., Kozlowski, P. B., Swain, R. A., Weiler, I. J., & Greenough, W. T. (2001). Abnormal dendritic spine characteristics in the temporal and visual cortices of patients with fragile-X syndrome: A quantitative examination. American Journal of Medical Genetics, 98, 161–167.
Ishizuka, A., Siomi, M. C., & Siomi, H. (2002). A Drosophila fragile X protein interacts with components of RNAi and ribosomal proteins. Genes & Development, 16, 2497–2508.
Iwahashi, C. K., Yasui, D. H., An, H. J., Greco, C. M., Tassone, F., Nannen, K., Babineau, B., Lebrilla, C. B., Hagerman, R. J., & Hagerman, P. J. (2006). Protein composition of the intranuclear inclusions of FXTAS. Brain, 129, 256–271.
Jacquemont, S., Hagerman, R. J., Hagerman, P. J., & Leehey, M. A. (2007). Fragile-X syndrome and fragile X-associated tremor/ataxia syndrome: Two faces of FMR1. Lancet Neurology, 6, 45–55.
Jin, P., Duan, R., Qurashi, A., Qin, Y., Tian, D., Rosser, T. C., Liu, H., Feng, Y., & Warren, S. T. (2007). Pur alpha binds to rCGG repeats and modulates repeat-mediated neurodegeneration in a Drosophila model of fragile X tremor/ataxia syndrome. Neuron, 55, 556–564.
Johnson, E. M., Kinoshita, Y., Weinreb, D. B., Wortman, M. J., Simon, R., Khalili, K., Winckler, B., & Gordon, J. (2006). Role of Pur alpha in targeting mRNA to sites of translation in hippocampal neuronal dendrites. Journal of Neuroscience Research, 83, 929–943.
Kanai, Y., Dohmae, N., & Hirokawa, N. (2004). Kinesin transports RNA: Isolation and characterization of an RNA-transporting granule. Neuron, 43, 513–525.
Kao, D. I., Aldridge, G. M., Weiler, I. J., & Greenough, W. T. (2010). Altered mRNA transport, docking, and protein translation in neurons lacking fragile X mental retardation protein. Proceedings of the National Academy of Sciences of the United States of America, 107, 15601–15606.
Kaufmann, W. E., Cohen, S., Sun, H. T., & Ho, G. (2002). Molecular phenotype of fragile X syndrome: FMRP, FXRPs, and protein targets. Microscopy Research and Technique, 57, 135–144.
Kenneson, A., Zhang, F., Hagedorn, C. H., & Warren, S. T. (2001). Reduced FMRP and increased FMR1 transcription is proportionally associated with CGG repeat number in intermediate-length and premutation carriers. Human Molecular Genetics, 10, 1449–1454.
Khandjian, E. W., Fortin, A., Thibodeau, A., Tremblay, S., Cote, F., Devys, D., Mandel, J. L., & Rousseau, F. (1995). A heterogeneous set of FMR1 proteins is widely distributed in mouse tissues and is modulated in cell culture. Human Molecular Genetics, 4, 783–789.
Khandjian, E. W., Huot, M. E., Tremblay, S., Davidovic, L., Mazroui, R., & Bardoni, B. (2004). Biochemical evidence for the association of fragile X mental retardation protein with brain polyribosomal ribonucleoparticles. Proceedings of the National Academy of Sciences of the United States of America, 101, 13357–13362.
Kindler, S., Rehbein, M., Classen, B., Richter, D., Böckers, TM., (2004). Distinct spatiotemporal expression of SAPAP transcripts in the developing rat brain: a novel dendritically localized mRNA. Brain Res Mol Brain Res. 126(1):14–21.
Koekkoek, S. K., Yamaguchi, K., Milojkovic, B. A., Dortland, B. R., Ruigrok, T. J., Maex, R., De Graaf, W., Smit, A. E., VanderWerf, F., Bakker, C. E., Willemsen, R., Ikeda, T., Kakizawa, S., Onodera, K., Nelson, D. L., Mientjes, E., Joosten, M., De Schutter, E., Oostra, B. A., Ito, M., & De Zeeuw, C. I. (2005). Deletion of FMR1 in Purkinje cells enhances parallel fiber LTD, enlarges spines, and attenuates cerebellar eyelid conditioning in fragile X syndrome. Neuron, 47, 339–352.
Kooy, R. F. (2003). Of mice and the fragile X syndrome. Trends in Genetics, 19, 148–154.
Kooy, R. F., D’Hooge, R., Reyniers, E., Bakker, C. E., Nagels, G., De Boulle, K., Storm, K., Clincke, G., De Deyn, P. P., Oostra, B. A., & Willems, P. J. (1996). Transgenic mouse model for the fragile X syndrome. American Journal of Medical Genetics, 64, 241–245.
Laggerbauer, B., Ostareck, D., Keidel, E. M., Ostareck-Lederer, A., & Fischer, U. (2001). Evidence that fragile X mental retardation protein is a negative regulator of translation. Human Molecular Genetics, 10, 329–338.
Larson, J., Jessen, R. E., Kim, D., Fine, A. K., & du Hoffmann, J. (2005). Age-dependent and selective impairment of long-term potentiation in the anterior piriform cortex of mice lacking the fragile X mental retardation protein. Journal of Neuroscience, 25, 9460–9469.
Lauterborn, J. C., Rex, C. S., Kramar, E., Chen, L. Y., Pandyarajan, V., Lynch, G., & Gall, C. M. (2007). Brain-derived neurotrophic factor rescues synaptic plasticity in a mouse model of fragile X syndrome. Journal of Neuroscience, 27, 10685–10694.
Li, J., Pelletier, M. R., Perez Velazquez, J. L., & Carlen, P. L. (2002). Reduced cortical synaptic plasticity and GluR1 expression associated with fragile X mental retardation protein deficiency. Molecular and Cellular Neurosciences, 19, 138–151.
Li, Z., Zhang, Y., Ku, L., Wilkinson, K. D., Warren, S. T., & Feng, Y. (2001). The fragile X mental retardation protein inhibits translation via interacting with mRNA. Nucleic Acids Research, 29, 2276–2283.
Linder, B., Plottner, O., Kroiss, M., Hartmann, E., Laggerbauer, B., Meister, G., Keidel, E., & Fischer, U. (2008). Tdrd3 is a novel stress granule-associated protein interacting with the fragile-X syndrome protein FMRP. Human Molecular Genetics, 17, 3236–3246.
Liu, Z. H., Chuang, D. M., Smith, C. B., Lithium ameliorates phenotypic deficits in a mouse model of fragile X syndrome. Int J Neuropsychopharmacol. 2011 Jun;14(5):618-30. Epub 2010 May 25.
Liu, Z. H., & Smith, C. B. (2009). Dissociation of social and nonsocial anxiety in a mouse model of fragile X syndrome. Neuroscience Letters, 454, 62–66.
Lu, R., Wang, H., Liang, Z., Ku, L., O’Donnell, W. T., Li, W., Warren, S. T., & Feng, Y. (2004). The fragile X protein controls microtubule-associated protein 1B translation and microtubule stability in brain neuron development. Proceedings of the National Academy of Sciences of the United States of America, 101, 15201–15206.
Massey, P. V., & Bashir, Z. I. (2007). Long-term depression: Multiple forms and implications for brain function. Trends in Neurosciences, 30, 176–184.
McNaughton, C. H., Moon, J., Strawderman, M. S., Maclean, K. N., Evans, J., & Strupp, B. J. (2008). Evidence for social anxiety and impaired social cognition in a mouse model of fragile X syndrome. Behavioral Neuroscience, 122, 293–300.
Meijer, H., de Graaff, E., Merckx, D. M., Jongbloed, R. J., de Die-Smulders, C. E., Engelen, J. J., Fryns, J. P., Curfs, P. M., & Oostra, B. A. (1994). A deletion of 1.6 kb proximal to the CGG repeat of the FMR1 gene causes the clinical phenotype of the fragile X syndrome. Human Molecular Genetics, 3, 615–620.
Menon, R. P., Gibson, T. J., & Pastore, A. (2004). The C terminus of fragile X mental retardation protein interacts with the multi-domain Ran-binding protein in the microtubule-organising centre. Journal of Molecular Biology, 343, 43–53.
Meredith, R. M., Holmgren, C. D., Weidum, M., Burnashev, N., & Mansvelder, H. D. (2007). Increased threshold for spike-timing-dependent plasticity is caused by unreliable calcium signaling in mice lacking fragile X gene FMR1. Neuron, 54, 627–638.
Mientjes, E. J., Nieuwenhuizen, I., Kirkpatrick, L., Zu, T., Hoogeveen-Westerveld, M., Severijnen, L., Rife, M., Willemsen, R., Nelson, D. L., & Oostra, B. A. (2006). The generation of a conditional Fmr1 knock out mouse model to study Fmrp function in vivo. Neurobiology of Disease, 21, 549–555.
Mila, M., Castellvi-Bel, S., Sanchez, A., Barcelo, A., Badenas, C., Mallolas, J., & Estivill, X. (2000). Rare variants in the promoter of the fragile X syndrome gene (FMR1). Molecular and Cellular Probes, 14, 115–119.
Min, W. W., Yuskaitis, C. J., Yan, Q., Sikorski, C., Chen, S., Jope, R. S., & Bauchwitz, R. P. (2009). Elevated glycogen synthase kinase-3 activity in fragile X mice: Key metabolic regulator with evidence for treatment potential. Neuropharmacology, 56, 463–472.
Mines, M. A., Yuskaitis, C. J., King, M. K., Beurel, E., & Jope, R. S. (2010). GSK3 influences social preference and anxiety-related behaviors during social interaction in a mouse model of fragile X syndrome and autism. PLoS One, 5, e9706.
Mineur, Y. S., Huynh, L. X., & Crusio, W. E. (2006). Social behavior deficits in the Fmr1 mutant mouse. Behavioural Brain Research, 168, 172–175.
Mineur, Y. S., Sluyter, F., de Wit, S., Oostra, B. A., & Crusio, W. E. (2002). Behavioral and neuroanatomical characterization of the Fmr1 knockout mouse. Hippocampus, 12, 39–46.
Miyashiro, K. Y., Beckel-Mitchener, A., Purk, T. P., Becker, K. G., Barret, T., Liu, L., Carbonetto, S., Weiler, I. J., Greenough, W. T., & Eberwine, J. (2003). RNA cargoes associating with FMRP reveal deficits in cellular functioning in Fmr1 null mice. Neuron, 37, 417–431.
Monzo, K., Papoulas, O., Cantin, G. T., Wang, Y., Yates, J. R., 3rd, & Sisson, J. C. (2006). Fragile X mental retardation protein controls trailer hitch expression and cleavage furrow formation in Drosophila embryos. Proceedings of the National Academy of Sciences of the United States of America, 103, 18160–18165.
Moon, J., Ota, K. T., Driscoll, L. L., Levitsky, D. A., & Strupp, B. J. (2008). A mouse model of fragile X syndrome exhibits heightened arousal and/or emotion following errors or reversal of contingencies. Developmental Psychobiology, 50, 473–485.
Morales, J., Hiesinger, P. R., Schroeder, A. J., Kume, K., Verstreken, P., Jackson, F. R., Nelson, D. L., & Hassan, B. A. (2002). Drosophila fragile X protein, DFXR, regulates neuronal morphology and function in the brain. Neuron, 34, 961–972.
Moss, J., & Howlin, P. (2009). Autism spectrum disorders in genetic syndromes: Implications for diagnosis, intervention and understanding the wider autism spectrum disorder population. Journal of Intellectual Disability Research, 53, 852–873.
Muddashetty, R. S., Kelic, S., Gross, C., Xu, M., & Bassell, G. J. (2007). Dysregulated metabotropic glutamate receptor-dependent translation of AMPA receptor and postsynaptic density-95 mRNAs at synapses in a mouse model of fragile X syndrome. Journal of Neuroscience, 27, 5338–5348.
Musco, G., Stier, G., Joseph, C., Castiglione Morelli, M. A., Nilges, M., Gibson, T. J., & Pastore, A. (1996). Three-dimensional structure and stability of the KH domain: Molecular insights into the fragile X syndrome. Cell, 85, 237–245.
Musumeci, S. A., Bosco, P., Calabrese, G., Bakker, C., De Sarro, G. B., Elia, M., Ferri, R., & Oostra, B. A. (2000). Audiogenic seizures susceptibility in transgenic mice with fragile X syndrome. Epilepsia, 41, 19–23.
Musumeci, S. A., Hagerman, R. J., Ferri, R., Bosco, P., Dalla Bernardina, B., Tassinari, C. A., De Sarro, G. B., & Elia, M. (1999). Epilepsy and EEG findings in males with fragile X syndrome. Epilepsia, 40, 1092–1099.
Napoli, I., Mercaldo, V., Boyl, P. P., Eleuteri, B., Zalfa, F., De Rubeis, S., Di Marino, D., Mohr, E., Massimi, M., Falconi, M., Witke, W., Costa-Mattioli, M., Sonenberg, N., Achsel, T., & Bagni, C. (2008). The fragile X syndrome protein represses activity-dependent translation through CYFIP1, a new 4E-BP. Cell, 134, 1042–1054.
Narayanan, U., Nalavadi, V., Nakamoto, M., Pallas, D. C., Ceman, S., Bassell, G. J., & Warren, S. T. (2007). FMRP phosphorylation reveals an immediate-early signaling pathway triggered by group I mGluR and mediated by PP2A. Journal of Neuroscience, 27, 14349–14357.
Nielsen, D. M., Derber, W. J., McClellan, D. A., & Crnic, L. S. (2002a). Alterations in the auditory startle response in Fmr1 targeted mutant mouse models of fragile X syndrome. Brain Research, 927, 8–17.
Nielsen, D. M., Evans, J. J., Derber, W. J., Johnston, K. A., Laudenslager, M. L., Crnic, L. S., & Maclean, K. N. (2009). Mouse model of fragile X syndrome: Behavioral and hormonal response to stressors. Behavioral Neuroscience, 123, 677–686.
Nielsen, P. R., Nietlispach, D., Mott, H. R., Callaghan, J., Bannister, A., Kouzarides, T., Murzin, A. G., Murzina, N. V., & Laue, E. D. (2002b). Structure of the HP1 chromodomain bound to histone H3 methylated at lysine 9. Nature, 416, 103–107.
Nimchinsky, E. A., Oberlander, A. M., & Svoboda, K. (2001). Abnormal development of dendritic spines in FMR1 knock-out mice. Journal of Neuroscience, 21, 5139–5146.
Nosyreva, E. D., & Huber, K. M. (2006). Metabotropic receptor-dependent long-term depression persists in the absence of protein synthesis in the mouse model of fragile X syndrome. Journal of Neurophysiology, 95, 3291–3295.
O’Donnell, W. T., & Warren, S. T. (2002). A decade of molecular studies of fragile X syndrome. Annual Review of Neuroscience, 25, 315–338.
Olmos-Serrano, J. L., Paluszkiewicz, S. M., Martin, B. S., Kaufmann, W. E., Corbin, J. G., & Huntsman, M. M. (2010). Defective GABAergic neurotransmission and pharmacological rescue of neuronal hyperexcitability in the amygdala in a mouse model of fragile X syndrome. Journal of Neuroscience, 30, 9929–9938.
Pacey, L. K., Doss, L., Cifelli, C., der Kooy, D. V., Heximer, S. P., & Hampson, D. R. (2011). Genetic deletion of regulator of G-protein signaling 4 (RGS4) rescues a subset of fragile X related phenotypes in the FMR1 knockout mouse. Molecular and Cellular Neurosciences, 46(3), 563–72.
Pacey, L. K., Heximer, S. P., & Hampson, D. R. (2009). Increased GABA(B) receptor-mediated signaling reduces the susceptibility of fragile X knockout mice to audiogenic seizures. Molecular Pharmacology, 76, 18–24.
Pan, F., Aldridge, G. M., Greenough, W. T., & Gan, W. B. (2010). Dendritic spine instability and insensitivity to modulation by sensory experience in a mouse model of fragile X syndrome. Proceedings of the National Academy of Sciences of the United States of America, 107, 17768–17773.
Papoulas, O., Monzo, K. F., Cantin, G. T., Ruse, C., Yates, J. R., 3rd, Ryu, Y. H., & Sisson, J. C. (2010). dFMRP and Caprin, translational regulators of synaptic plasticity, control the cell cycle at the Drosophila mid-blastula transition. Development, 137, 4201–4209.
Paradee, W., Melikian, H. E., Rasmussen, D. L., Kenneson, A., Conn, P. J., & Warren, S. T. (1999). Fragile X mouse: Strain effects of knockout phenotype and evidence suggesting deficient amygdala function. Neuroscience, 94, 185–192.
Paribello, C., Tao, L., Folino, A., Berry-Kravis, E., Tranfaglia, M., Ethell, I. M., & Ethell, D. W. (2010). Open-label add-on treatment trial of minocycline in fragile X syndrome. BMC Neurology, 10, 91.
Park, S., Park, J. M., Kim, S., Kim, J. A., Shepherd, J. D., Smith-Hicks, C. L., Chowdhury, S., Kaufmann, W., Kuhl, D., Ryazanov, A. G., Huganir, R. L., Linden, D. J., Worley, P. F., (2008). Elongation factor 2 and fragile X mental retardation protein control the dynamic translation of Arc/Arg3.1 essential for mGluR-LTD. Neuron. 59(1):70–83.
Paylor, R., Yuva-Paylor, L. A., Nelson, D. L., & Spencer, C. M. (2008). Reversal of sensorimotor gating abnormalities in Fmr1 knockout mice carrying a human Fmr1 transgene. Behavioral Neuroscience, 122, 1371–1377.
Peier, A. M., McIlwain, K. L., Kenneson, A., Warren, S. T., Paylor, R., & Nelson, D. L. (2000). (Over)correction of FMR1 deficiency with YAC transgenics: Behavioral and physical features. Human Molecular Genetics, 9, 1145–1159.
Pfeiffer, B. E., & Huber, K. M. (2007). Fragile X mental retardation protein induces synapse loss through acute postsynaptic translational regulation. Journal of Neuroscience, 27, 3120–3130.
Pfeiffer, B. E., Zang, T., Wilkerson, J. R., Taniguchi, M., Maksimova, M. A., Smith, L. N., Cowan, C. W., & Huber, K. M. (2010). Fragile X mental retardation protein is required for synapse elimination by the activity-dependent transcription factor MEF2. Neuron, 66, 191–197.
Portera-Cailliau, C., Pan, D. T., & Yuste, R. (2003). Activity-regulated dynamic behavior of early dendritic protrusions: Evidence for different types of dendritic filopodia. Journal of Neuroscience, 23, 7129–7142.
Primerano, B., Tassone, F., Hagerman, R. J., Hagerman, P., Amaldi, F., & Bagni, C. (2002). Reduced FMR1 mRNA translation efficiency in fragile X patients with premutations. RNA, 8, 1482–1488.
Purpura, D. P. (1974). Dendritic spine “dysgenesis” and mental retardation. Science, 186, 1126–1128.
Qiu, L. F., Lu, T. J., Hu, X. L., Yi, Y. H., Liao, W. P., & Xiong, Z. Q. (2009). Limbic epileptogenesis in a mouse model of fragile X syndrome. Cerebral Cortex, 19, 1504–1514.
Ramos, A., Hollingworth, D., Adinolfi, S., Castets, M., Kelly, G., Frenkiel, T. A., Bardoni, B., & Pastore, A. (2006). The structure of the N-terminal domain of the fragile X mental retardation protein: A platform for protein–protein interaction. Structure, 14, 21–31.
Reiss, A. L., Abrams, M. T., Greenlaw, R., Freund, L., & Denckla, M. B. (1995). Neurodevelopmental effects of the FMR-1 full mutation in humans. Nature Medicine, 1, 159–167.
Restivo, L., Ferrari, F., Passino, E., Sgobio, C., Bock, J., Oostra, B. A., Bagni, C., & Ammassari-Teule, M. (2005). Enriched environment promotes behavioral and morphological recovery in a mouse model for the fragile X syndrome. Proceedings of the National Academy of Sciences of the United States of America, 102, 11557–11562.
Ronesi, J. A., & Huber, K. M. (2008). Metabotropic glutamate receptors and fragile X mental retardation protein: Partners in translational regulation at the synapse. Science Signaling, 1, pe6.
Rudelli, R. D., Brown, W. T., Wisniewski, K., Jenkins, E. C., Laure-Kamionowska, M., Connell, F., & Wisniewski, H. M. (1985). Adult fragile X syndrome. Clinico-neuropathologic findings. Acta Neuropathologica, 67, 289–295.
Saugstad, J. A., Marino, M. J., Folk, J. A., Hepler, J. R., & Conn, P. J. (1998). RGS4 inhibits signaling by group I metabotropic glutamate receptors. Journal of Neuroscience, 18, 905–913.
Schenck, A., Bardoni, B., Langmann, C., Harden, N., Mandel, J. L., & Giangrande, A. (2003). CYFIP/Sra-1 controls neuronal connectivity in Drosophila and links the Rac1 GTPase pathway to the fragile X protein. Neuron, 38, 887–898.
Schütt, J., Falley, K., Richter, D., Kreienkamp, H. J., & Kindler, S. (2009). Fragile X mental retardation protein regulates the levels of scaffold proteins and glutamate receptors in postsynaptic densities. Journal of Biological Chemistry, 284, 25479–25487.
Selby, L., Zhang, C., & Sun, Q. Q. (2007). Major defects in neocortical GABAergic inhibitory circuits in mice lacking the fragile X mental retardation protein. Neuroscience Letters, 412, 227–232.
Schaeffer, C., Bardoni, B., Mandel, J. L., Ehresmann, B., Ehresmann, C., Moine, H,. (2001). The fragile X mental retardation protein binds specifically to its mRNA via a purine quartet motif. EMBO J. 20(17):4803–13.
Shang, Y., Wang, H., Mercaldo, V., Li, X., Chen, T., & Zhuo, M. (2009). Fragile X mental retardation protein is required for chemically-induced long-term potentiation of the hippocampus in adult mice. Journal of Neurochemistry, 111, 635–646.
Shi, S. H., Hayashi, Y., Petralia, R. S., Zaman, S. H., Wenthold, R. J., Svoboda, K., & Malinow, R. (1999). Rapid spine delivery and redistribution of AMPA receptors after synaptic NMDA receptor activation. Science, 284, 1811–1816.
Siomi, H., Siomi, M. C., Nussbaum, R. L., & Dreyfuss, G. (1993). The protein product of the fragile X gene, FMR1, has characteristics of an RNA-binding protein. Cell, 74, 291–298.
Siomi, M. C., Zhang, Y., Siomi, H., & Dreyfuss, G. (1996). Specific sequences in the fragile X syndrome protein FMR1 and the FXR proteins mediate their binding to 60S ribosomal subunits and the interactions among them. Molecular and Cellular Biology, 16, 3825–3832.
Sittler, A., Devys, D., Weber, C., & Mandel, J. L. (1996). Alternative splicing of exon 14 determines nuclear or cytoplasmic localisation of fmr1 protein isoforms. Human Molecular Genetics, 5, 95–102.
Sjekloca, L., Konarev, P. V., Eccleston, J., Taylor, I. A., Svergun, D. I., & Pastore, A. (2009). A study of the ultrastructure of fragile-X-related proteins. Biochemical Journal, 419, 347–357.
Sofola, O. A., Jin, P., Qin, Y., Duan, R., Liu, H., de Haro, M., Nelson, D. L., & Botas, J. (2007). RNA-binding proteins hnRNP A2/B1 and CUGBP1 suppress fragile X CGG premutation repeat-induced neurodegeneration in a Drosophila model of FXTAS. Neuron, 55, 565–571.
Spencer, C. M., Alekseyenko, O., Hamilton, S. M., Thomas, A. M., Serysheva, E., Yuva-Paylor, L. A., & Paylor, R. (2011). Modifying behavioral phenotypes in Fmr1KO mice: Genetic background differences reveal autistic-like responses. Autism Research, 4, 40–56.
Spencer, C. M., Alekseyenko, O., Serysheva, E., Yuva-Paylor, L. A., & Paylor, R. (2005). Altered anxiety-related and social behaviors in the Fmr1 knockout mouse model of fragile X syndrome. Genes, Brain, and Behavior, 4, 420–430.
Spencer, C. M., Graham, D. F., Yuva-Paylor, L. A., Nelson, D. L., & Paylor, R. (2008). Social behavior in Fmr1 knockout mice carrying a human FMR1 transgene. Behavioral Neuroscience, 122, 710–715.
Sprangers, R., Groves, M. R., Sinning, I., & Sattler, M. (2003). High-resolution X-ray and NMR structures of the SMN Tudor domain: Conformational variation in the binding site for symmetrically dimethylated arginine residues. Journal of Molecular Biology, 327, 507–520.
Stefani, G., Fraser, C. E., Darnell, J. C., & Darnell, R. B. (2004). Fragile X mental retardation protein is associated with translating polyribosomes in neuronal cells. Journal of Neuroscience, 24, 7272–7276.
Steward, O., & Schuman, E. M. (2003). Compartmentalized synthesis and degradation of proteins in neurons. Neuron, 40, 347–359.
Su, T., Fan, H. X., Jiang, T., Sun, W. W., Den, W. Y., Gao, M. M., Chen, S. Q., Zhao, Q. H., & Yi, Y. H. (2010). Early continuous inhibition of group 1 mGlu signaling partially rescues dendritic spine abnormalities in the Fmr1 knockout mouse model for fragile X syndrome. Psychopharmacology (Berlin), 215(2):291–300.
Sung, Y. J., Dolzhanskaya, N., Nolin, S. L., Brown, T., Currie, J. R., Denman, R. B,. (2003). The fragile X mental retardation protein FMRP binds elongation factor 1A mRNA and negatively regulates its translation in vivo. J Biol Chem. 278(18):15669–78. Epub 2003 Feb 19.
Suvrathan, A., Hoeffer, C. A., Wong, H., Klann, E., & Chattarji, S. (2010). Characterization and reversal of synaptic defects in the amygdala in a mouse model of fragile X syndrome. Proceedings of the National Academy of Sciences of the United States of America, 107, 11591–11596.
Tamanini, F., Van Unen, L., Bakker, C., Sacchi, N., Galjaard, H., Oostra, B. A., & Hoogeveen, A. T. (1999). Oligomerization properties of fragile-X mental-retardation protein (FMRP) and the fragile-X-related proteins FXR1P and FXR2P. Biochemical Journal, 343(Pt 3), 517–523.
Tassone, F., Beilina, A., Carosi, C., Albertosi, S., Bagni, C., Li, L., Glover, K., Bentley, D., & Hagerman, P. J. (2007). Elevated FMR1 mRNA in premutation carriers is due to increased transcription. RNA, 13, 555–562.
Tassone, F., De Rubeis, S., Carosi, C., La Fata, G., Serpa, G., Raske, C., Willemsen, R., Hagerman, P. J., Bagni, C., (2011). Differential usage of transcriptional start sites and polyadenylation sites in FMR1 premutation alleles. Nucleic Acids Res, 2011. Aug: 39(14):6172–85. Epub 2011 Apr 7.
Tassone, F., Hagerman, R. J., Taylor, A. K., Gane, L. W., Godfrey, T. E., & Hagerman, P. J. (2000). Elevated levels of FMR1 mRNA in carrier males: A new mechanism of involvement in the fragile-X syndrome. American Journal of Human Genetics, 66, 6–15.
Tassone, F., Iwahashi, C., & Hagerman, P. J. (2004). FMR1 RNA within the intranuclear inclusions of fragile X-associated tremor/ataxia syndrome (FXTAS). RNA Biology, 1, 103–105.
Tervonen, T., Akerman, K., Oostra, B. A., & Castren, M. (2005). Rgs4 mRNA expression is decreased in the brain of Fmr1 knockout mouse. Brain Research. Molecular Brain Research, 133, 162–165.
Todd, P. K., & Mack, K. J. (2000). Sensory stimulation increases cortical expression of the fragile X mental retardation protein in vivo. Brain Research: Molecular Brain Research, 80, 17–25.
Tucker, B., Richards, R. I., & Lardelli, M. (2006). Contribution of mGluR and Fmr1 functional pathways to neurite morphogenesis, craniofacial development and fragile X syndrome. Human Molecular Genetics, 15, 3446–3458.
Valverde, R., Edwards, L., & Regan, L. (2008). Structure and function of KH domains. FEBS Journal, 275, 2712–2726.
Valverde, R., Pozdnyakova, I., Kajander, T., Venkatraman, J., & Regan, L. (2007). Fragile X mental retardation syndrome: Structure of the KH1-KH2 domains of fragile X mental retardation protein. Structure, 15, 1090–1098.
Van Dam, D., D’Hooge, R., Hauben, E., Reyniers, E., Gantois, I., Bakker, C. E., Oostra, B. A., Kooy, R. F., & De Deyn, P. P. (2000). Spatial learning, contextual fear conditioning and conditioned emotional response in Fmr1 knockout mice. Behavioural Brain Research, 117, 127–136.
Vanderklish, P. W., & Edelman, G. M. (2002). Dendritic spines elongate after stimulation of group 1 metabotropic glutamate receptors in cultured hippocampal neurons. Proceedings of the National Academy of Sciences of the United States of America, 99, 1639–1644.
Ventura, R., Pascucci, T., Catania, M. V., Musumeci, S. A., & Puglisi-Allegra, S. (2004). Object recognition impairment in Fmr1 knockout mice is reversed by amphetamine: Involvement of dopamine in the medial prefrontal cortex. Behavioural Pharmacology, 15, 433–442.
Verheij, C., Bakker, C. E., de Graaff, E., Keulemans, J., Willemsen, R., Verkerk, A. J., Galjaard, H., Reuser, A. J., Hoogeveen, A. T., & Oostra, B. A. (1993). Characterization and localization of the FMR-1 gene product associated with fragile X syndrome. Nature, 363, 722–724.
Weiler, I. J., Irwin, S. A., Klintsova, A. Y., Spencer, C. M., Brazelton, A. D., Miyashiro, K., Comery, T. A., Patel, B., Eberwine, J., & Greenough, W. T. (1997). Fragile X mental retardation protein is translated near synapses in response to neurotransmitter activation. Proceedings of the National Academy of Sciences of the United States of America, 94, 5395–5400.
Willemsen, R., Bontekoe, C., Tamanini, F., Galjaard, H., Hoogeveen, A., & Oostra, B. (1996). Association of FMRP with ribosomal precursor particles in the nucleolus. Biochemical and Biophysical Research Communications, 225, 27–33.
Westmark, C. J., Malter, J. S., (2007). FMRP mediates mGluR5-dependent translation of amyloid precursor protein. PLoS Biol. 5(3):e52.
Wohrle, D., Kotzot, D., Hirst, M. C., Manca, A., Korn, B., Schmidt, A., Barbi, G., Rott, H. D., Poustka, A., Davies, K. E., et al. (1992). A microdeletion of less than 250 kb, including the proximal part of the FMR-I gene and the fragile-X site, in a male with the clinical phenotype of fragile-X syndrome. American Journal of Human Genetics, 51, 299–306.
Wolfer, D. P., & Lipp, H. P. (2000). Dissecting the behaviour of transgenic mice: Is it the mutation, the genetic background, or the environment? Experimental Physiology, 85, 627–634.
Xie, W., Dolzhanskaya, N., LaFauci, G., Dobkin, C., & Denman, R. B. (2009). Tissue and developmental regulation of fragile X mental retardation 1 exon 12 and 15 isoforms. Neurobiology of Disease, 35, 52–62.
Yan, Q. J., Asafo-Adjei, P. K., Arnold, H. M., Brown, R. E., & Bauchwitz, R. P. (2004). A phenotypic and molecular characterization of the fmr1-tm1Cgr fragile X mouse. Genes, Brain, and Behavior, 3, 337–359.
Yun, S. H., & Trommer, B. L. (2011). Fragile X mice: Reduced long-term potentiation and N-methyl-D-aspartate receptor-mediated neurotransmission in dentate gyrus. Journal of Neuroscience Research, 89, 176–182.
Yuskaitis, C. J., Mines, M. A., King, M. K., Sweatt, J. D., Miller, C. A., & Jope, R. S. (2010). Lithium ameliorates altered glycogen synthase kinase-3 and behavior in a mouse model of fragile X syndrome. Biochemical Pharmacology, 79, 632–646.
Zalfa, F., Achsel, T., & Bagni, C. (2006). mRNPs, polysomes or granules: FMRP in neuronal protein synthesis. Current Opinion in Neurobiology, 16, 265–269.
Zalfa, F., Adinolfi, S., Napoli, I., Kuhn-Holsken, E., Urlaub, H., Achsel, T., Pastore, A., & Bagni, C. (2005). Fragile X mental retardation protein (FMRP) binds specifically to the brain cytoplasmic RNAs BC1/BC200 via a novel RNA-binding motif. Journal of Biological Chemistry, 280, 33403–33410.
Zalfa, F., & Bagni, C. (2005). Another view of the role of FMRP in translational regulation. Cellular and Molecular Life Sciences, 62, 251–252.
Zalfa, F., Eleuteri, B., Dickson, K. S., Mercaldo, V., De Rubeis, S., di Penta, A., Tabolacci, E., Chiurazzi, P., Neri, G., Grant, S. G., & Bagni, C. (2007). A new function for the fragile X mental retardation protein in regulation of PSD-95 mRNA stability. Nature Neuroscience, 10, 578–587.
Zalfa, F., Giorgi, M., Primerano, B., Moro, A., Di Penta, A., Reis, S., Oostra, B., & Bagni, C. (2003). The fragile X syndrome protein FMRP associates with BC1 RNA and regulates the translation of specific mRNAs at synapses. Cell, 112, 317–327.
Zang, J. B., Nosyreva, E. D., Spencer, C. M., Volk, L. J., Musunuru, K., Zhong, R., Stone, E. F., Yuva-Paylor, L. A., Huber, K. M., Paylor, R., Darnell, J. C., & Darnell, R. B. (2009). A mouse model of the human fragile X syndrome I304N mutation. PLoS Genetics, 5, e1000758.
Zarnescu, D. C., Shan, G., Warren, S. T., & Jin, P. (2005). Come FLY with us: Toward understanding fragile X syndrome. Genes, Brain, and Behavior, 4, 385–392.
Zeier, Z., Kumar, A., Bodhinathan, K., Feller, J. A., Foster, T. C., & Bloom, D. C. (2009). Fragile X mental retardation protein replacement restores hippocampal synaptic function in a mouse model of fragile X syndrome. Gene Therapy, 16, 1122–1129.
Zhang, Y. Q., Bailey, A. M., Matthies, H. J., Renden, R. B., Smith, M. A., Speese, S. D., Rubin, G. M., & Broadie, K. (2001). Drosophila fragile X-related gene regulates the MAP1B homolog Futsch to control synaptic structure and function. Cell, 107, 591–603.
Zhang, J., Fang, Z., Jud, C., Vansteensel, M. J., Kaasik, K., Lee, C. C., Albrecht, U., Tamanini, F., Meijer, J. H., Oostra, B. A., & Nelson, D. L. (2008). Fragile X-related proteins regulate mammalian circadian behavioral rhythms. American Journal of Human Genetics, 83, 43–52.
Zhang, J., Hou, L., Klann, E., & Nelson, D. L. (2009). Altered hippocampal synaptic plasticity in the FMR1 gene family knockout mouse models. Journal of Neurophysiology, 101, 2572–2580.
Zhang, Y., O’Connor, J. P., Siomi, M. C., Srinivasan, S., Dutra, A., Nussbaum, R. L., & Dreyfuss, G. (1995). The fragile X mental retardation syndrome protein interacts with novel homologs FXR1 and FXR2. EMBO Journal, 14, 5358–5366.
Zhang, M., Wang, Q., & Huang, Y. (2007). Fragile X mental retardation protein FMRP and the RNA export factor NXF2 associate with and destabilize Nxf1 mRNA in neuronal cells. Proceedings of the National Academy of Sciences of the United States of America, 104, 10057–10062.
Zhao, M. G., Toyoda, H., Ko, S. W., Ding, H. K., Wu, L. J., & Zhuo, M. (2005). Deficits in trace fear memory and long-term potentiation in a mouse model for fragile X syndrome. Journal of Neuroscience, 25, 7385–7392.
Acknowledgments
This study was supported by Telethon, Compagnia di San Paolo, COFIN, FWO, and FP7 (SynSys). We are thankful to the Associazione Italiana Sindrome X Fragile (http://www.xfragile.net/).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2012 Springer-Verlag/WIen
About this chapter
Cite this chapter
De Rubeis, S., Fernández, E., Buzzi, A., Di Marino, D., Bagni, C. (2012). Molecular and Cellular Aspects of Mental Retardation in the Fragile X Syndrome: From Gene Mutation/s to Spine Dysmorphogenesis. In: Kreutz, M., Sala, C. (eds) Synaptic Plasticity. Advances in Experimental Medicine and Biology, vol 970. Springer, Vienna. https://doi.org/10.1007/978-3-7091-0932-8_23
Download citation
DOI: https://doi.org/10.1007/978-3-7091-0932-8_23
Published:
Publisher Name: Springer, Vienna
Print ISBN: 978-3-7091-0931-1
Online ISBN: 978-3-7091-0932-8
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)