Abstract
Radionuclide pediatric bone scintigraphy is a useful modality to assess benign diseases of the skeleton. Bone scintigraphy has high sensitivity in assessing bone metabolism in children. Bone scintigraphy is most commonly performed with 99mTc-labelled bisphosphonate SPECT agents and less commonly with 18F-NaFluoride (NaF), a bone-seeking PET tracer with similar biodistribution. The lower protein binding in the blood of NaF results in its more rapid first-pass extraction and faster soft tissue clearance, allowing early image acquisition. Accurate interpretation of bone images is often challenging and requires adequate training and knowledge of pathophysiology to recognize normal variants, patterns, and pitfalls. In general, bone scintigraphy is best interpreted in conjunction with available clinical, laboratory, and radiologic data. In addition, there have been significant advances in hybrid imaging technology such as SPECT/CT, PET/CT, and PET/MRI over the years which have improved accurate localization, and characterization of benign and malignant bone lesions in children.
You have full access to this open access chapter, Download chapter PDF
Similar content being viewed by others
Keywords
10.1 Clinical Indications
-
Assessment of alterations in bone metabolism due to trauma, infection, inflammation.
-
Assessment for conditions with impaired blood flow (e.g., avascular necrosis).
-
Assessment of soft tissue (ST).
10.2 Pre-exam Information
-
Are symptoms focal or generalized? Duration of symptoms
-
History of trauma, systemic metabolic or oncologic conditions
-
History of previous interventional procedures (i.e., joint aspiration or drainage, surgery)
-
History of treatment such as antibiotics or anti-inflammatory drugs, radiation therapy (including involved fields)
-
Serum inflammatory markers available for correlation
-
Relevant X-rays, ultrasound (US), CT, or MRI available for correlation?
10.3 Bone Scintigraphy
Study Protocol for 99mTc-MDP Bone Scintigraphy [1, 2]
Patient preparation
-
Good hydration is important, instruct to drink at least 2 cups during the uptake time prior to delayed imaging.
-
Infants should be fed prior to or immediately after tracer injection.
-
Children should be encouraged to urinate frequently to reduce the exposure of the bladder.
-
Sedation is usually not required, except for uncooperative neurodevelopmentally delayed children.
Radiopharmaceutical, administered activity, mode of delivery
Radiopharmaceutical:
-
[99mTc]methylene diphosphonate (MDP) or similar diphosphonates.
Activity:
-
9.3 MBq/kg (0.25 mCi/Kg), minimum dose 40 MBq (1.1 mCi).
Refer to the EANM pediatric dosage card and to the North American consensus guidelines on radiopharmaceutical administration in children in the respective EANM and SNMMI and image gently web sites.
Reference to national regulation guidelines, if available, should be considered.
Delivery:
-
Select intravenous (IV) injection site to avoid possible sites of pathology. For example, upper extremity lesions will require a foot injection.
Acquisition protocol: Two- or three-phase bone scan is recommended, four-phase protocol can be performed.
-
Early imaging (blood flow and blood pool) to the area of concern and the adjacent region which may cause referred pain (e.g., in the presence of symptoms in a lower extremity include images from thoracic spine down).
-
Dynamic blood flow study.
-
Blood pool (regional or whole body), immediately after the completion of the dynamic study.
-
-
Late skeletal phase scan at 2–4 h post-injection.
-
Image total body vertex to toes in anterior and posterior projection
-
SPECT should be included in the third step in selected scenarios:
-
To areas of localized symptoms.
-
If an abnormality is detected on planar imaging.
SPECT/CT, if available should replace SPECT
-
Limit the field-of-view (FOV) of the CT to the abnormality seen on SPECT to reduce radiation exposure.
-
CT can be acquired as a pediatric low-dose CT for localization and attenuation correction or as diagnostic CT. Many skeletal lesions are adequately evaluated even with low-dose CT parameters.
-
-
A delayed 24-h scan can be performed:
-
In cases of uncertain findings on routine 3-h scintigraphy.
-
When residual bladder activity overlies the pelvis and the child refuses to urinate or when bladder emptying is incomplete.
Acquisition parameters
-
-
Position: The child should lie supine, comfortably secured to the bed.
-
Collimators.
-
High or ultrahigh low-energy collimator.
-
Pinhole collimator, if available, can improve detection of lesions in the hip joint of distal extremities.
-
-
Blood flow images.
-
2–5 s/frame for a total of 60 s, matrix 128 × 128, size-appropriate zoom
-
-
Early blood pool images.
-
Recommended counts: Torso 300 Kcounts, extremities 150–200 Kcounts.
-
For lower extremity lesions the entire lower limbs pelvis and lumbar spine should be in the FOV.
-
-
Late (skeletal) phase images.
-
Whole body sweeps with bed speed adjusted to the child’s age. The EANM guidelines recommend [3]:
-
8 cm/min for ages 4–8 years
-
10 cm/min for ages 8–12 years
-
12 cm/min for ages 12–16 years
-
15 cm/min above 16 years of age
-
-
Alternative: Multiple spot views covering the entire skeleton, anterior and posterior projections, start with the pelvis when the bladder is empty.
-
Matrix 256 × 256.
-
Recommended counts: Torso 500 Kcounts, skull 300 Kcounts, knees 100–200 Kcounts, hands and feet 50–100 Kcounts.
-
Alternatively: The time to obtain 500 Kcounts for the torso should be recorded and used to time the acquisition of the other body parts.
-
-
SPECT: 120 projections, 15–30 s/view, matrix 128 × 128.
-
SPECT/CT: For the CT component use pediatric settings with dose modulation.
-
Reduce the CT field to include only the findings on the SPECT component.
-
Tube settings depend on manufacturer and the applied protocol: low-dose or fully diagnostic CT (accordingly the settings can range between 80 and110 kVp).
-
CT slice thickness 2–2.5 mm with overlapping cuts.
-
10.4 Study Interpretation [3, 4]
-
Review the SPECT or SPECT/CT in orthogonal planes, MIP, and, when available, fused co-registered study.
-
Review CT study on bone and soft tissue windows in addition to co-registered study.
-
Patterns of tracer localization:
Normal bone growth patterns in infants and children [5]
-
Reduced or absent activity due to physiologic delayed ossification of certain bones (e.g., navicular bone of the foot, femoral head) must be differentiated from pathologic photopenic lesion.
-
Increased activity in the orbits on the anterior view of the skull is normal in the first months of life.
-
Focal activity in the ischio-pubic synchondrosis is normal in most cases and reflects asymmetric closure of bone centers.
-
Focally increased tracer activity:
-
Osteomyelitis: Increased focal activity in the metaphysis of long bone or in the metaphyseal equivalents. There is also often increased tracer uptake in early blood flow and blood pool images.
-
Arthritis: Increased blood flow and blood pool in the involved joint(s). Tracer uptake in the skeletal phase can be normal or increased in a diffuse manner.
-
Multifocal tracer uptake: Multi-trauma, metastatic disease, multifocal osteomyelitis. Clinical context, labs, and correlative imaging can distinguish between the options.
-
Non-accidental injury (NAI, child abuse): Bilateral, symmetrical foci in costovertebral junctions (typical for the shaken baby syndrome), multiple foci in ribs in various locations, metaphyseal fractures in the shafts of lower extremity bones in young infants who are not walking or bearing weight on their lower extremities, diffuse uptake in the calvarium that may suggest a skull fracture.
Focal reduced/photopenic tracer activity:
-
In suspected osteomyelitis, photopenic lesions are suggestive of an aggressive form known as “cold osteomyelitis.”
-
In a child with an unexplained limp, photopenia in the femoral head can suggest Legg-Calve-Perthes disease.
-
In children treated with high-dose steroids, multiple photopenic lesions can be due to multifocal avascular necrosis.
-
Multiple photopenic lesions occur early on in cases with known hemoglobinopathies.
-
Sometimes photopenic lesions, especially in the spine are due to metastatic disease (e.g., neuroblastoma, leukemia, or lymphoma)
Diffuse decreased tracer activity:
-
Can be caused by prolonged limb immobilization or decreased use.
-
Associated with severe pain, reduced skin temperature, and faint peripheral pulses could represent a complex regional pain syndrome (CRPS, reflex sympathetic dystrophy).
Diffuse increased tracer uptake:
-
Along the cortex of the tibia and/or fibula in children and adolescents undergoing strenuous physical activities can be due to shin splints, while focal uptake can be due to a stress fracture.
-
In the feet and ankles often observed in the unaffected side of limping children due to increased weight bearing on the healthy side.
Bone Scintigraphy Protocol Adjustments in Suspected Non-Accidental Injury [6, 7]
-
Add routinely to standard views: lateral and oblique views of the ribs, anterior, posterior, and lateral views of the skull.
-
Make sure the hands are flat, so all digits are visualized.
-
Make sure the urinary bladder does not obscure pelvic structures.
10.5 Correlative Imaging [8]
-
Correlation with radiographs is required when assessing skeleton.
-
MRI is a sensitive modality for the evaluation of bone marrow and ST pathologies. In some centers, it is a first-line investigation for the MSK system. The examination is typically focused on the symptomatic region and can therefore miss sites of disease in cases of referred pain as well as additional sites in multifocal disease. MRI availability and cost can be limiting factors.
-
Check if the child has not performed a recent CT or MRI or if one is already planned before deciding on SPECT/CT.
10.6 Red Flags [4, 6]
-
When positioning patients for planar imaging, the feet should be secured with a medial (inward) tilt of the feet, thus allowing adequate visualization of the fibulae.
-
Early blood pool static images should start immediately after the completion of the dynamic blood flow study as skeletal uptake begins quickly after tracer injection.
-
In young children, due to high bone to ST contrast, delayed static images can be acquired as early as 2 h post-injection.
-
If bladder emptying is difficult and there is no other alternative, bladder catheterization may be considered.
-
All children should have delayed imaging of the entire body, performed as a whole body sweep or multiple planar static images.
-
In young children recording, sets of overlapping planar static images allow for repeated selected images in case of motion with improved spatial resolution.
-
Additional spot views are often necessary to improve detectability of lesions. For example, lateral, medial plantar, and dorsal views for foot and ankle pathology, lateral views for the legs, palmar, and dorsal views for the hands, oblique and lateral views for the ribs, lateral views for the skull and cervical spine.
-
In older children, SPECT/CT can replace multiple spot views.
-
If SPECT/CT is used attention must be paid to the additional radiation dose of the CT component.
-
In general CT acquisition should be strictly limited to the body segment of interest.
-
Sensitivity of bone scans is low in the detection of linear skull fractures.
-
Sensitivity of bone scans in the detection of arthritis is limited.
-
Suspected fractures or ischemia in weight-bearing bones should be promptly reported to the referring physician to initiate preventive measures to avoid complications.
10.7 Take Home Message
-
Whole-body blood pool images are useful in suspected multifocal osteomyelitis (MFO), rheumatologic conditions, generalized pain, and NAI.
-
In most cases, ST pathologies are best identified on the blood pool images and blood flow images do not provide additional information.
-
When vascular compromise is suspected (e.g., limb ischemia reflex sympathetic dystrophy, avascular necrosis) blood flow images should be obtained.
-
All children should have a whole-body delayed bone scintigraphy assessment. Regional bone scans should not be performed.
-
While adequate count density and optimal spatial resolution are desired, some flexibility is advised keeping in mind that long acquisition times increase the likelihood of motion artifacts that degrade image quality.
-
Review correlative radiologic imaging, when possible, for findings worrisome for malignancy.
-
Findings raising the possibility of NAIs should be immediately reported to the referring physician before the patient is discharged.
10.8 Representative Case Examples
Case 10.1 Osteomyelitis (Fig. 10.1)
History: An 11-year-old boy presented with a painful left hip 2 weeks after playing soccer. X-rays and US were normal. Study report: In the early blood pool study (a) there is increased tracer uptake in the left anterior inferior iliac spine (arrow). The late planar anterior (b) and posterior (c) scans show focal increased osteoblastic activity in the same area (arrow). SPECT/CT, axial, coronal, and sagittal low-dose CT slices (d) show no bone erosion around marked focal osteoblastic reaction seen in the left iliac bone on fused images (e). Impression: The pathological uptake on bone scintigraphy, in association with the lack of abnormalities on the CT part of the SPECT/CT most likely exclude tumor. SPECT/CT cannot exclude subtle lesions such as stress fractures or enthesopathy. Acute osteomyelitis was the suggested diagnosis despite the lack of systemic fever, further confirmed by blood cultures positive for S. aureus
Case 10.2 Non-Accidental Bone Injuries (Fig. 10.2)
History: A 4-month-old girl presented with a swollen left arm. X-ray showed a left humeral fracture. Study report: On early blood pool images (not shown), there is an increased blood pool in the distal left humerus and mid-left tibia. Late planar scans show foci of increased tracer uptake in the mid-to-distal left humerus and mid-left tibia. Impression: The pattern of focal increased tracer uptake in the left humerus and left tibia are highly suggestive of non-accidental injury despite normal X-ray of the tibia
Case 10.3 Spondylolysis (Fig. 10.3)
History: An 11-year-old boy presented with a month-long low back pain after playing football. X-rays showed mild scoliosis, convex to the left, but was otherwise normal. Study report: Bone scintigraphy shows a normal blood pool (a) but mildly increased tracer uptake on the right side of the lower lumbar spine in the late planar scans (b). SPECT, transaxial (c) and coronal (d) slices, shows focal increased uptake in the right pars interarticularis of L4 vertebra. SPECT/CT (e), transaxial, coronal and sagittal CT (left) and fused (right) images, demonstrate bilateral sclerosis, more pronounced at the site of the focal increased tracer uptake in the right pars interarticularis of L4 vertebra. Impression: The findings are consistent with an L4 spondylolytic fracture
Case 10.4. Legg-Calve-Perthes Disease (Fig. 10.4)
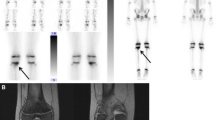
History: An 8-year-old boy presented with complaints of right hip pain and right-sided limping for 2 weeks, without a history of fever or trauma. Study report: The early phase images (not shown) were unremarkable. Anterior and posterior late whole-body images (a, b) show reduced tracer uptake in the right femoral head (arrows). SPECT anterior projection MIP (c) and coronal, sagittal, and transaxial slices (d–f) show a photopenic area in the right proximal femoral epiphysis (arrows) confirming the findings on the whole body sweeps. Impression: The findings suggest avascular necrosis of the right femoral head (Legg-Calve-Perthes disease)
10.9 18F-Sodium Fluoride (NAF) Bone PET/CT
Study Protocol for NaF Bone PET/CT [1, 9,10,11]
Patient preparation
-
Patient should be well hydrated.
-
Change diaper right before imaging.
-
Remove metal objects from around the patient before imaging.
Radiopharmaceutical, administered activity and mode of delivery
Radiopharmaceutical:
-
[18F] Sodium Fluoride (NaF)
Activity:
-
2.22 MBq/kg (0.06 mCi/kg), range 18.5–185 MBq (0.5–5 mCi).
Refer to the EANM pediatric dosage card and to the North American consensus guidelines on radiopharmaceutical administration in children in the respective EANM and SNMMI and image gently web sites.
Reference to national regulation guidelines, if available, should be considered.
Delivery:
-
Select the injection site to avoid possible sites of pathology.
Acquisition protocol
-
Uptake period: 45 min after tracer administration.
-
PET: 2–5 min/bed position (depending on equipment).
-
FOV: varies by patient size.
-
CT: Either low-dose CT (70–80 mA, 80–100 kVp) or diagnostic CT
10.10 Study Interpretation
-
Review the PET MIP image, as well as fused orthogonal planes.
-
Review CT with bone and soft tissue windows.
-
Correlative imaging such as radiographs or other cross-sectional imaging should be reviewed at the time of reporting.
10.11 Correlative Imaging
-
Correlation with radiographs is required when assessing skeleton.
10.13 Take Home Messages [14, 15]
-
NaF imaging has the advantage of shorter total study duration, higher image quality, and quantification.
-
Effective dose from NaF is comparable to effective dose from Tc-MDP.
-
The main current indications for NaF bone PET studies are in cancer patients for evaluation of primary and metastatic bone lesions and children with chronic recurrent multifocal osteomyelitis.
10.14 Representative Case Examples
Case 10.5 Normal NaF PET Study (Fig. 10.5)
History: A 7-year-old boy was referred to NaF PET/CT for further evaluation of low back pain. Study report: Whole-body PET, anterior (left) and lateral (right) MIP, show increased symmetric tracer uptake in the epiphyseal plates of the long bones in the upper and lower limbs and the anterior end of the ribs, bilaterally, consistent with areas of physiologic activity in growing plates. Impression: No evidence of abnormal osteoblastic lesions in the spine
Case 10.6 Fibrous Cortical Defect (Fig. 10.6)
History: A 9-year-old boy was referred to NaF PET/CT following 1-month long complaints of pain in the left leg. Study report: Whole-body MIP (left) shows a focal area of increased tracer uptake at the medial aspect of the upper shaft of left tibia. Coronal and sagittal (right) and transaxial (center) PET, CT, and fused PET/CT slices at the level of the proximal tibia show an eccentric, well-defined lytic lesion with a rim of sclerosis, with corresponding increased tracer uptake at the posteromedial part of the left upper tibia. Impression: The findings are consistent with a fibrous cortical defect
Case 10.7 Bilateral Ischio-Pubic Synchondrosis (Fig. 10.7)
History: A 7-year-old boy with right orbital rhabdomyosarcoma was evaluated as part of a metastatic survey. Study report: Anterior (a) and lateral MIP (b), as well as transaxial PET, CT, and fused NaF PET/CT slices at the level of the symphysis pubis (c) show physiological symmetric increase uptake at the growing ends of the bone, including costochondral junctions, and the epiphyseal plates of the upper and lower limbs. There is increased tracer uptake in both distal rami of the pubis (arrows). Impression: The findings demonstrate focal increased tracer uptake in sites of bilateral ischio-pubic synchondrosis. This is a normal variant and should not be interpreted as fractures. No evidence of bone metastases
Case 10.8 Patellar Tendon Insertion Enthesopathy (Fig. 10.8)
History: A 13-year-old boy is complaining of right knee pain for 3 months. Plain X-rays were unremarkable. Study report: NaF PET/CT, anterior (a) and oblique MIP (b), as well as sagittal (c) and transaxial (d) PET, CT, and fused slices at the level of the proximal tibiae show focal increased tracer uptake at the cortical surface of right tibial tuberosity (arrow). Impression: The findings are consistent with patellar tendon insertion enthesopathy
Case 10.9 Sickle Cell Anemia (Fig. 10.9)
History: A 16-year-old boy with known sickle cell anemia presented with subacute bone pain. Study report: NaF PET MIP (a), transaxial (b) and coronal (c) PET, CT and fused images show multiple areas of increased tracer uptake at right clavicle, bilateral ribs, the left humeral shaft, and the right trochanteric region. Impression: The osteoblastic lesions represent active bone remodeling at multiple bone infarcts
References
Beheshti M, et al. (18)F-NaF PET/CT: EANM procedure guidelines for bone imaging. Eur J Nucl Med Mol Imaging. 2015;42(11):1767–77.
Van den Wyngaert T, et al. The EANM practice guidelines for bone scintigraphy. Eur J Nucl Med Mol Imaging. 2016;43(9):1723–38.
Stauss J, et al. Guidelines for paediatric bone scanning with 99mTc-labelled radiopharmaceuticals and 18F-fluoride. Eur J Nucl Med Mol Imaging. 2010;37(8):1621–8.
Nadel HR. SPECT/CT in pediatric patient management. Eur J Nucl Med Mol Imaging. 2014;41(Suppl 1):S104–14.
Atlas of bone scintigraphy in the developing paediatric skeleton: the normal skeleton variants and pitfalls. Vienna: International Atomic Energy Agency; 2011.
Nadel HR. Pediatric bone scintigraphy update. Semin Nucl Med. 2010;40(1):31–40.
Treves ST. Skeletal scintigraphy: general considerations. In: Treves ST, editor. Pediatric nuclear medicine and molecular imaging. New York: Springer; 2014. p. 365–84.
Shammas A. Nuclear medicine imaging of the pediatric musculoskeletal system. Semin Musculoskelet Radiol. 2009;13(3):159–80.
Segall G, et al. SNM practice guideline for sodium 18F-fluoride PET/CT bone scans 1.0. J Nucl Med. 2010;51(11):1813–20.
Drubach LA. Pediatric bone scanning: clinical indication of (18)F NaF PET/CT. PET Clin. 2012;7(3):293–301.
Fischer DR. Musculoskeletal imaging using fluoride PET. Semin Nucl Med. 2013;43(6):427–33.
Drubach LA, Connolly SA, Palmer EL 3rd. Skeletal scintigraphy with 18F-NaF PET for the evaluation of bone pain in children. AJR Am J Roentgenol. 2011;197(3):713–9.
Panagiotidis E, et al. Skeletal metastases and benign mimics on NaF PET/CT: a pictorial review. AJR Am J Roentgenol. 2018;211(1):W64–w74.
Jadvar H, Desai B, Conti PS. Sodium 18F-fluoride PET/CT of bone, joint, and other disorders. Semin Nucl Med. 2015;45(1):58–65.
Usmani S, et al. Technical feasibility, radiation dosimetry and clinical use of (18)F-sodium fluoride (NaF) in evaluation of metastatic bone disease in pediatric population. Ann Nucl Med. 2018;32(9):594–601.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
The opinions expressed in this chapter are those of the author(s) and do not necessarily reflect the views of the IAEA: International Atomic Energy Agency, its Board of Directors, or the countries they represent
Open Access This chapter is licensed under the terms of the Creative Commons Attribution 3.0 IGO license (http://creativecommons.org/licenses/by/3.0/igo/), which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the IAEA: International Atomic Energy Agency, provide a link to the Creative Commons license and indicate if changes were made.
Any dispute related to the use of the works of the IAEA: International Atomic Energy Agency that cannot be settled amicably shall be submitted to arbitration pursuant to the UNCITRAL rules. The use of the IAEA: International Atomic Energy Agency's name for any purpose other than for attribution, and the use of the IAEA: International Atomic Energy Agency's logo, shall be subject to a separate written license agreement between the IAEA: International Atomic Energy Agency and the user and is not authorized as part of this CC-IGO license. Note that the link provided above includes additional terms and conditions of the license.
The images or other third party material in this chapter are included in the chapter's Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the chapter's Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.
Copyright information
© 2023 The Author(s)
About this chapter
Cite this chapter
Gnanasegaran, G., Usmani, S., Nadel, H. (2023). Musculoskeletal System (Non-Oncologic Indications). In: Bar-Sever, Z., Giammarile, F., Israel, O., Nadel, H. (eds) A Practical Guide for Pediatric Nuclear Medicine. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-662-67631-8_10
Download citation
DOI: https://doi.org/10.1007/978-3-662-67631-8_10
Published:
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-662-67630-1
Online ISBN: 978-3-662-67631-8
eBook Packages: MedicineMedicine (R0)