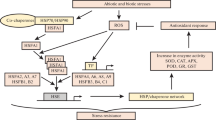
Abstract
Antibodies prepared against two major polypeptides of the 15- to 18-kD class of soybean heat shock proteins (HSPs) individually reacted with its antigen and cross-reacted with 12 other 15- to 18-Kd HSPs. We also found that this antibody preparation cross-reacted with the same class low molecular weight (LMW) HSPs of mung bean, rice and other seven plant species tested based on western blot analysis. The 70 to 100% ammonium sulfate (AS) fraction from heat shocked seedlings of mung bean and rice, as in soybean, contained a high percentage of all the HSPs. The proteins in this fraction were resistant to heat denaturation, as judged by their unpelletability after heat treatment. Moreover, this fraction showed a significant ability to protect the soluble proteins from heat denaturation. The HSPsenriched fractions prepared from mung bean and rice heat shocked seedlings were able to thermostabilize the homologous soluble proteins. Additionally, the HSPs-enriched fractions were exchangeable among these three plant species for thermostabilization.
In soybean, when the HSPs-enriched fraction depleted of the 15- to 18-kD class HSPs, the effectiveness in prevention of heat denaturation was lost. However, when the 15- to 18-kD HSPs recovered in the AS fraction, the thermostabilization was restored again. This suggests that the HSPs of 15- to 18-kD class are important for providing the protection from heat denaturation.
Supported by National Science Council of Taiwan under grants NSC 78-0211-B002-02, NSC 79-0211-B002-02 and NSC 80-0211-B002-02.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Barnett. T, Altssschuler M, Mcdaniel CN, Mascarenhas JP (1980). Heat shock induced proteins in plant cells. Dev Genet 1: 331–340.
Chou, M., Chen, Y.M. and Lin C.Y. (1989). Thermotolerance of isolated mitochondria associated with heat shock proteins. Plant Physiol. 89: 617–621.
Hsieh, M.H., Chen, J.T., Jinn, T.L., Chen, Y.M., and Lin, C.Y. (1992). A class of soybean low molecular weight heat shock proteins: immunological study and quantitation. Plant Physiol. 99: 1279–1284.
Jinn, T.L., Yeh, Y.C., Chen, Y.M., and Lin, C.Y. (1989). Stabilization of soluble proteins in vitro by heat shock proteins-enriched ammonium sulfate fraction from soybean seedlings. Plant Cell Physiol. 30: 463–469.
Key, J.L., Lin, C.Y., and Chen, Y.M. (1981). Heat shock proteins of higher plants. Proc. Natl Acad. Sci. USA 78: 3526–3530.
Kimpel, J.A., and Key, J.L. (1985). Heat shock in plants. Trends Biochem Sci 10: 353–357.
Laemmli, U.K. (1970). Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227: 680–685.
Li, G.C. and Werb, Z. (1982). Correlation between synthesis of heat shock proteins and development of thermotolerance in Chinese hamster fibroblasts. Proc. Natl. Acad. Sci. USA 98: 3218–3222.
Lin, C.Y., Roberts, J.K. and Key, J.L. (1984). Acquisition of thermotolerance in soybean seedlings: synthesis and accumulation of heat shock proteins and their cellular localization. Plant Physiol. 74: 152–160.
Lindquist, S. (1986). The heat-shock response. Annu. Rev. Biochem. 55: 1151–1191.
Lowry, O.H., Rosebrough, N.J., Farr, A.L. and Randoll R.J. (1951). Protein measurement with the Folin phenol reagent. J. Biol. Chem. 193: 265–275.
Mans, R.J. and Noveli, G.D. (1961). Measurement of the incorporation of radioactive amino acids into protein by a filter paper disk method. Arch. Biochem. Biophys. 4: 48–53.
Mansfield, M.A., and Key, J.L. (1987). Synthesis of the low molecular weight heat shock proteins in plants. Plant Physiol. 84: 1007–1017.
McAlister, L., and Finkelstein, D.B. (1980). Heat shock proteins and thermal resistance in yeast. Biochem. Biophys. Res. Commun. 93: 819–824.
Minton, K.W., Karmin, P., Hahn, G.H. and Minton, A.P. (1982). Nonspecific stabilization of stresssusceptible proteins by stress-resistant proteins: a model for the biological roles of heat shock proteins. Proc. Natl. Acad. Sci. USA 79: 7107–7111.
O’Farrell, P.H. (1975). High resolution two-dimensional electrophoresis of proteins. J. Biol. Chem. 250: 4007–4021.
Peterson, N.S. and Mitchell, H.K. (1981). Recovery of protein synthesis after heat shock: prior heattreatment affects the ability of cells to translate mRNA. Proc. Natl. Acad. Sci. USA 78: 1078–1711.
Schlesinger, M.J., Ashburner, M. and Tissieres, A. (1982). Heat shock from bacteria to man. New York: Cold Spring Harbor Laboratory Press. 440 pp.
Schlesinger, M., Alliterate, G. and Kelley, P.M. (1982). The response of cells to heat shock. Trends Biochem. Sci. 1: 222–225.
Towbin, H., Staehelin, T. and Gordon, J. (1979). Electrophoretic transfer of protein from polyacrylamide gels to nitrocellulose sheet: procedure and some application. Proc. Natl. Acad. Sci. USA 76: 4350–4354.
Tseng, T.S., Yeh, K.W., Yeh, C.H., Chang, F.C., Chen, Y.M. and Lin, C.Y. (1992). Two-rice (Oryza sativa) full-length cDNA clones encoding low-molecular-weight heat-shock proteins. Plant Mol. Biol. 18: 963–965.
Vierling, E. (1991). The roles of heat shock proteins in plants. Annu. Rev. Plant Physiol. Plant Mol. Biol. 42: 579–620.
Wu, S.H. (1992). The biological function of low molecular weight heat shock proteins in plants. Master thesis. Department of Botany, National Taiwan University, Taipei, Taiwan, Republic of China
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 1994 Springer-Verlag Berlin Heidelberg
About this paper
Cite this paper
Lin, CY., Jinn, TL., Hsieh, MH., Chen, YM. (1994). Class I low molecular weight heat shock proteins in plants: immunological study and thermoprotection against heat denaturation of soluble proteins. In: Cherry, J.H. (eds) Biochemical and Cellular Mechanisms of Stress Tolerance in Plants. NATO ASI Series, vol 86. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-642-79133-8_7
Download citation
DOI: https://doi.org/10.1007/978-3-642-79133-8_7
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-642-79135-2
Online ISBN: 978-3-642-79133-8
eBook Packages: Springer Book Archive