Abstract
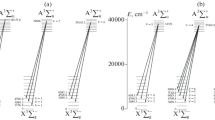
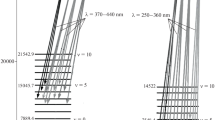
Of my papers on molecules published in 1928, I was especially proud of two, one being that of the above-mentioned assignment of electron configurations to molecules like BO and CN (done also by Hund). The other was an interpretation of the absorption bands of the oxygen molecule (O2) which are seen in the red region of the sun’s spectrum for sunlight which has passed through the earth’s atmosphere. I identified this spectrum as a “forbidden” one, that is, one disobeying certain rules that are obeyed in most molecular spectra. Forbidden spectra, if seen at all, are intrinsically extremely weak, but we can see these “atmospheric oxygen bands” because the sunlight has gone through many miles of atmosphere before reaching us. One important by-product of the analysis was that it showed that the normal state of the oxygen molecule, from which the absorption arises, is a 3Σ −g state. This is a radical-like triplet state, which makes oxygen magnetic. I saw the latter property demonstrated by the professor in my physics course at MIT. He poured liquid oxygen over a magnet, to which it clung, defying gravity.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 1989 Springer-Verlag Berlin Heidelberg
About this chapter
Cite this chapter
Mulliken, R.S., Ransil, B.J. (1989). Interpretation of Atmospheric Oxygen Bands, 1928. In: Ransil, B.J. (eds) Life of a Scientist. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-642-61320-3_11
Download citation
DOI: https://doi.org/10.1007/978-3-642-61320-3_11
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-540-50375-0
Online ISBN: 978-3-642-61320-3
eBook Packages: Springer Book Archive