Abstract
Seasonal fluctuations in mood, behaviour, energy level and appetite are common in humans living in temperate and polar zones. These changes are not necessarily associated with clinical symptoms; however, some people regularly experience severe changes in mood and drive during the dark season. Seasonal affective disorder (SAD) is regarded as an extreme reaction to changes in environmental light. The underlying mechanism of these seasonal changes and the pathobiology of SAD still remain unclear. However, several lines of evidence suggest a key role of monoamines in modulating seasonal fluctuations in animals and humans. Here, we review the literature on neuroimaging including MRI, SPECT and PET in SAD. Furthermore, the effects of season on the monoamine neurotransmitter systems serotonin and dopamine are discussed.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Positron Emission Tomography
- Single Photon Emission Compute Tomography
- Light Therapy
- Single Photon Emission Compute Tomography Study
- Seasonal Affective Disorder
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
1 Introduction
Seasonal fluctuations in metabolism and behaviour are common in organisms living in temperate and polar zones. These fluctuations are evolutionary coping strategies, necessary for adapting to dramatic changes in temperature, light and food availability (Levitan et al. 2006, 2010; Praschak-Rieder and Willeit 2012). The degree of this adaptation in humans is known as “seasonality”. Seasonal changes in mood, behaviour, energy level and appetite are not necessarily associated with psychopathological symptoms, as they are normally distributed in the general population (Hardin et al. 1991; Kasper et al. 1989; Praschak-Rieder and Willeit 2012; Winkler et al. 2002). Extreme seasonal variations in mood and drive were first described in a psychiatric context by Rosenthal et al. (1984). “Seasonal affective disorder” (SAD) is considered a clinical subtype of major depression. A milder form of SAD, termed “winter blues” or “subsyndromal SAD (s-SAD)”, was described by Kasper and colleagues (1988). Altogether, prevalence rates of SAD and s-SAD have been reported between 1.5 and 17.8 % in the Northern Hemisphere (Kasper 1994). Based on the hypothesis that SAD is triggered by photoperiod variations and the fact that these variations are larger in higher latitudes closer to the poles, increased prevalence rates of SAD have been assumed in these regions (Mersch et al. 1999). Although, some studies found a significant positive correlation between latitude and prevalence of SAD (Potkin et al. 1986; Rosen et al. 1990), climate, social as well as cultural factors seem to have a more considerable impact on its prevalence (Mersch et al. 1999). Gender disparity is substantially greater in SAD than in other forms of depression with a female-to-male sex ratio of up to 9:1 according to some studies (Boyce and Parker 1988; Thompson and Isaacs 1988; Winkler et al. 2002; Wirz-Justice et al. 1986).
The “winter seasonal pattern” constitutes the most common form of SAD. According to DSM-IV, this form of SAD is characterised by a recurrent pattern of major depressive episodes during fall and winter (in the absence of seasonal psychosocial stressors) and remission of depressive symptoms during spring and summer (Rosenthal et al. 1984; Lam and Levitan 2000). In contrast to the winter form of SAD, Wehr described a less prevalent form of SAD with depressive symptoms during summer and hypomania during winter (Wehr et al. 1987). Furthermore, seasonal depressive symptoms are reported with a higher frequency during summer and attributed to intense heat and humidity in some parts of the world (Avasthi et al. 2001; Morrissey et al. 1996). On a symptom level, winter SAD is frequently characterised by atypical depressive symptoms such as increased sleep duration, hyperphagia and subsequent weight gain (Praschak-Rieder and Willeit 2003; Rosenthal et al. 1984). In parallel to non-seasonal depression, the neurotransmitters serotonin, norepinephrine and dopamine have been suggested to play a crucial role in the aetiology and pathophysiology of SAD (Levitan 2007). A transient decline in brain serotonin due to depletion of tryptophan, the amino acid precursor of serotonin, has been reported to result in lower mood and increased irritability or aggressive responding in several studies (for review, see Young and Leyton 2002). Tryptophan depletion caused a relapse of depressive symptoms in remitted SAD patients (Neumeister et al. 1998a) and reversed the therapeutic effect of bright light treatment (Lam et al. 1996; Neumeister et al. 1998b). Alterations in norepinephrine and dopamine neurotransmission were hypothesised to be essential for the occurrence of fatigue and reduced levels of subjective arousal in SAD patients (for review, see Levitan 2007). In addition, dopamine has been reported to act as a chemical messenger for light adaptation (Witkovsky 2004). Patients with SAD show reduced light sensitivity (Hebert et al. 2004), supporting the hypothesis of an involvement of dopamine in the pathogenesis of SAD.
Based on evidence derived from several randomised, placebo-controlled studies using dim light or deactivated ion generators as comparator, light therapy is recognised as an effective therapy and is recommended as first-line treatment for SAD (Lewy et al. 1998; Terman 2006; Terman et al. 1989). The pathophysiology of SAD is still not sufficiently understood (Magnusson and Partonen 2005), though theories on its pathogenesis are intimately tied to the biological mechanisms of light therapy (Lam and Levitan 2000).
Although SAD and its subsyndromal form show a high prevalence, imaging studies investigating patients with SAD and the effects of seasonality on the brain are scarce. The following synopsis will give an overview about neuroimaging in SAD and seasonal effects on brain monoamine pathways.
2 Structural and Functional Magnetic Resonance Imaging
2.1 Structural MRI Studies
Volumetric studies in non-seasonal depressed patients showed a nonspecific brain atrophy including an increase in ventricular-brain ratio, increased cerebrospinal fluid volume and sulcal atrophy (Steffens and Krishnan 1998). Additionally, a lateralisation of atrophy was found to the left medial temporal lobe in patients with a late-onset depression (Greenwald et al. 1997), whereas other studies were not able to demonstrate grey matter differences in this region (Coffey et al. 1993; Pantel et al. 1997). In addition to the findings of Greenwald, Drevets et al. (1997) were able to show a left-lateralised reduction in grey matter volume in the subgenual prefrontal cortex (Brodmann area 24) in patients with familial forms of major depressive disorder (MDD) and bipolar disorder (BD). The subgenual prefrontal cortex is a region mediating emotional and autonomic responses to socially significant or provocative stimuli. Given the suggested critical role of the hippocampus in the pathophysiology of depression (Campbell and Macqueen 2004), several studies revealed smaller sizes of this region in patients with a depressive episode (Caetano et al. 2004; Frodl et al. 2002b; Shah et al. 1998; Sheline 1996). In addition, contradictory results of structural abnormalities in the amygdala in depression have been shown. An enlargement of the amygdala was found in patients with a first episode of major depression (Frodl et al. 2002a), whereas this enlargement was not found in recurrent depression (Frodl et al. 2003). A smaller amygdala volume in depressed patients was reported by Sheline et al. (1998). The structural changes in grey matter mentioned above were only found in patients suffering from non-seasonal depression, and there is still a lack of volumetric MRI investigations in the field of SAD.
Only two MRI studies focusing on structural abnormalities in SAD patients were available in the literature. In line with a hyperactivity of the hypothalamic-pituitary adrenal (HPA) axis in depression, larger pituitary volumes have been found in patients with major depression (Krishnan et al. 1991). In contrast to these findings, a study in 19 patients with SAD did not show any significant pituitary volume changes (Schwartz et al. 1997). Since the participants of this study underwent MRI scans of the pituitary gland both in summer and winter, this study was further able to demonstrate that pituitary volumes did not change between seasons, supporting the notion that the aetiology of SAD is associated with factors other than HPA dysregulation (Sheline et al. 1998). Recently, the findings of Schwartz et al. have been replicated in a Brazilian investigation (Miranda-Scippa et al. 2008). Miranda-Scippa and her colleagues compared pituitary gland volumes of 12 patients suffering from SAD and 12 healthy controls matched for age, gender and menstrual cycle. No significant differences in pituitary gland volume between patients and controls were found. Light therapy was shown to significantly reduce depressive symptoms, but it did not alter pituitary gland volumes. Although no significant changes of pituitary volumes have been found in this study, pituitary volumes in winter correlated positively with the severity of depression in patients. While some studies have suggested adrenal gland enlargement in non-seasonal depression (Kessing et al. 2011), to our knowledge, there are no investigations focusing on adrenal gland volumes in SAD.
In sum, recent data on structural changes in SAD provide no clear evidence of structural brain alteration in SAD.
2.2 Functional MRI (fMRI) Studies
Electroretinographical studies have shown a reduced retinal light sensitivity in SAD patients (Hebert et al. 2004) with seasonal variations in rod and cone function. Furthermore, a normalisation of rod and cone function was found after 4 weeks of bright light therapy (Lavoie et al. 2009). Based on these findings, Vandewalle et al. (2011) conducted an fMRI study investigating the impact of light on emotional processing in untreated SAD patients (n = 14). Patients showed an increased response to auditory emotional stimuli in the posterior hypothalamus under blue light (480 nm) exposure, whereas green light (550 nm) decreased hypothalamic response. Furthermore, increased responsiveness to vocal stimuli was found in thalamus and brainstem areas in patients. The authors suggested that altered emotional processing during coloured light exposure, as shown by the abnormal light responsiveness of the hypothalamus, may constitute a neurobiological substrate of SAD.
3 Single Photon Emission Computed Tomography (SPECT)
The monoamine neurotransmitter serotonin (5-hydroxytryptamine, 5-HT) has been implicated in various physiological functions, including regulation of circadian rhythms as well as in the pathophysiology of numerous neuropsychiatric disorders. Based on several findings of a seasonal rhythm in brain and peripheral serotonin (5-HT) activity in humans, this monoamine neurotransmitter was suggested to play a major role in the pathomechanisms of SAD (Kasper et al. 1996; Lam and Levitan 2000; Willeit et al. 2000, 2008). In a human post-mortem study, Carlsson and colleagues were able to show a seasonal variation in hypothalamic 5-HT concentrations with lowest levels of 5-HT occurring in winter (Carlsson et al. 1980). Confirming these findings in vivo, an Australian study by Lambert et al. reported reduced serotonin turnover in the Australian winter months between June and August (Lambert et al. 2002).
One of the key molecules in serotonergic neurotransmission is the serotonin transporter (SERT or 5-HTT). After release of 5-HT into the synaptic cleft, SERT mediates reuptake into the presynaptic neuron. Thereby, SERT activity is able to control spatial and temporal spread of the serotonergic signal. Selective serotonin reuptake inhibitors (SSRIs) exert their antidepressive effect by blocking SERT and show comparable efficacy to light therapy in the treatment of SAD (Lam et al. 2006; Praschak-Rieder and Willeit 2003).
The availability of brain SERT binding sites can be assessed in vivo via the nonspecific monoamine transporter ligand [123I]-2-beta-carbomethoxy-3-beta-(4-iodophenyl)-tropane ([123I]ß-CIT) and single photon emission computed tomography (SPECT; Brucke et al. 1993). By means of this technique, Neumeister et al. first demonstrated seasonal effects on brain SERT binding by investigating a small sample of healthy females (n = 11; Neumeister et al. 2000). Variations in the availability of thalamus/hypothalamus SERT binding sites were found between summer and winter with higher SERT availability in summer. A recent study of SERT availability in a larger sample of non-seasonal depressed patients (n = 49) demonstrated opposite findings with significantly higher [123I]ß-CIT binding in winter (Ruhe et al. 2009). However, a significant reduction in SERT availability was only shown in male depressed patients.
Only a limited number of SPECT studies have been conducted in patients with SAD. In a study by Willeit et al. (2000), drug-free SAD patients (n = 11) showed decreased [123I]ß-CIT binding in the midbrain thalamus-hypothalamus area compared to controls matched for age, gender, menstrual cycle and time of scanning. Based on animal (Laruelle et al. 1993) and post-mortem displacement studies (Staley et al. 1994), [123I]ß-CIT is known to bind predominantly to SERT in the midbrain. Therefore, the finding of Willeit et al. may reflect a reduced SERT availability in untreated depressed patients with SAD in winter, a result that is partly in line with findings in non-seasonal depression (Ruhe et al. 2009). A study on SERT binding in platelets of patients with SAD and healthy controls failed to show differences between the two groups (Willeit et al. 2008). However, this study showed increased efficiency in SERT-mediated 5-HT uptake during winter depression. After successful light therapy and during natural remission in summer, SERT function returned to control levels.
The dopaminergic system was also suggested to be involved in the pathophysiology of SAD by a [123I]ß-CIT study of the Vienna group (Neumeister et al. 2001). In contrast to the midbrain, [123I]ß-CIT binds—after achieving equilibrium binding (later than in the midbrain)—predominantly to dopamine transporters (DAT) in the striatum. According to that, Neumeister et al. were able to show a reduced availability of striatal DAT in untreated SAD patients in winter time.
A recent SPECT study from Taiwan investigated striatal dopamine D2/3 availability in 68 healthy subjects with respect to their exposure to sunshine 30 days prior to their individual SPECT scan (Tsai et al. 2011). Since there is little seasonal variation in day length and daily sunlight in Taiwan, only 35 subjects in the lowest (n = 18) and highest (n = 17) quartile of average sunshine duration were analysed. Higher [123I]iodobenzamide ([123I]IBZM) binding was revealed in subjects exposed to higher amounts of sunshine than in those with lower sunshine exposure prior to SPECT scans. Results have to be interpreted with caution: [123I]IBZM is sensitive towards changes in extracellular dopamine levels (Laruelle 2000), and the findings of higher [123I]IBZM binding could either be due to a higher amount of dopamine D2/3 receptors or reduced levels in extracellular dopamine. Moreover, rates of tobacco use differed significantly between groups.
Apart from transporter and receptor studies, an investigation on regional cerebral blood flow (rCBF) in a small sample of untreated patients with SAD and healthy controls using [99mTc]hexamethylpropyleneamine oxime ([99mTc]HMPAO), and SPECT suggested an increased left frontal rCBF in patients with SAD (Praschak-Rieder et al. 1998). Following successful bright light treatment, normalisation in left frontal rCBF was found.
The mentioned SPECT studies revealed reduced availabilities of SERT and DAT as well as alterations in regional cerebral blood flow in depressed patients with SAD. Moreover, possible seasonal effects on D2/3 receptors have been demonstrated in healthy subjects. Findings of these preliminary studies were partly strengthened by results obtained in studies using more selective radioligands and positron emission tomography (PET—see Sect. 8.4). However, independent replications in larger samples of patients with SAD are still warranted.
4 Positron Emission Tomography (PET)
During the last decades, only two PET studies specifically investigating patients with SAD were conducted. Both studies on cerebral metabolism used [11F]deoxy-glucose ([11F]FDG) to investigate if patients with SAD showed abnormalities in cerebral metabolic rates. A study by Cohen et al. (1992) compared brain metabolic rates between a small sample (n = 7) of patients with winter SAD and healthy controls. All patients were drug-free for at least 3 months and were investigated in an untreated condition (off-lights). Furthermore, six patients were also PET-scanned after at least 10 days of light treatment (on-lights). To avoid possible order effects, three patients were investigated in the off-lights condition first and after 10 days of light treatment. The other three patients were studied during the on-lights condition first and after 10 days of discontinuation of light treatment. Light treatment consisted of 2.5 h of 2,500-lux full-spectrum light twice a day (morning between 6 and 9 AM, evening between 6 and 9 PM). Patients with SAD showed lower global metabolic rates under on- and off-lights condition, suggesting that a lowered metabolic state might be a trait marker of SAD. As suggested by the authors, an alternative explanation for the failure to detect differences in patients during on- and off-light condition may have been the insufficient length of light therapy. However, light therapy was sufficient to reverse depressive symptoms in these patients.
The second PET study on cerebral glucose metabolism was conducted in patients suffering from summer SAD (Goyer et al. 1992). Nine patients were investigated showing significantly different regional glucose metabolic rates in orbital frontal cortex and in left inferior parietal lobule compared to healthy controls.
In contrast to the small number of studies investigating SAD patients (see Table 8.1), several neuroimaging studies analysing seasonal effects on monoaminergic neurotransmitter systems have been conducted.
As mentioned before, SPECT studies using the non-selective radioligand [123I]ß-CIT revealed contradictory results with respect to seasonal effects on SERT binding (Neumeister et al. 2000; Ruhe et al. 2009). A PET study by Praschak-Rieder et al. conducted in a larger group of healthy drug-naïve subjects (n = 88) (Praschak-Rieder et al. 2008) was able to demonstrate a considerable effect of season on SERT by using the specific SERT radioligand [11C]3-amino-4-(2-dimethylaminomethyl-phenylsulfanyl)benzonitrile ([11C]DASB) and PET. This study revealed high [11C]DASB binding potential (BPND) values in autumn and winter in six different predefined regions of interest (ROI). A uniform decrease of regional BPND values was found in spring and summer (Fig. 8.1). Peak differences in [11C]DASB BPND values between months with highest and lowest binding potential values as large as 40 %. Furthermore, [11C]DASB BPND values showed a negative correlation with the duration of daily sunshine and day length. In accordance with this study, Kalbitzer et al. (2010) reported a negative correlation of [11C]DASB BPND values and daylight minutes. In the latter study, 54 healthy subjects were investigated using [11C]DASB PET and genotyped for a polymorphism in the promoter region of the SERT gene (5-HTTLPR). Only carriers of 5-HTTLPR s-allele showed significant effects of season in [11C]DASB binding. In contrast, 5-HTTLPR l-allele homozygous subjects did not exhibit seasonal variation of SERT availability. The methodology used in both studies does not allow for differentiation between the influence of daily sunshine and the astronomical photoperiod (daylight minutes) on [11C]DASB binding because both parameters are highly intercorrelated. Although the negative correlation between [11C]DASB binding and duration of daylight was found in both studies, a study by Murthy et al. (2010) did not replicate these findings.
Additionally, effects of season on SERT binding were demonstrated by Buchert and colleagues (2006) using PET and the radioligand trans-1,2,3,5,6,10b-hexahydro-6-[4-(methylthio)-phenyl]pyrrolo-[2,1-a]-isoquinoline ([11C]-(+)McN5652). This study investigated age-related effects on SERT binding in 29 healthy subjects. In line with the results obtained by [11C]DASB PET, SERT binding measured with [11C]-(+)McN5652 PET was higher in winter, while age did not show any effect on SERT availability in this sample.
Reciprocal peaks and troughs of serotonin transporter (SERT) binding and duration of sunshine in 88 healthy subjects. Shaded area: duration of sunshine in Toronto, Ontario (range, between 2.4 and 9.2 h a day. SERT BPND measured by the selective SERT radioligand [11C]DASB and positron emission tomography. Circles represent bimonthly moving averages of mean binding potential values in six predefined regions of interest (prefrontal cortex, anterior cingulate, caudate, putamen, thalamus and midbrain). X-axis: calendar months (Modified according to Praschak-Rieder et al. (2012))
In summary, variations in SERT, with higher serotonin transporter availability in times of less light, as shown by the aforementioned studies, may facilitate extracellular serotonin loss during winter, potentially leading to hyposerotonergic symptoms and lower mood. To our knowledge, there are no studies on seasonal variations in SERT binding in patients with SAD. However, the data provided by Ruhe et al. (2009) suggest that there is a similar increase in SERT binding in patients with major depressive disorder in winter.
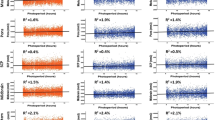
Recently, a study by Spindelegger et al. (2012) revealed light-dependent alterations of brain serotonin 1A (5-HT1A) receptor binding. Among the different subtypes of serotonin receptors, the inhibitory 5-HT1A receptor has a particular role. Located on GABAergic and glutamatergic neurons in limbic and cortical brain regions, the receptor mediates the inhibition of postsynaptic firing (Varnas et al. 2004). In contrast, 5-HT1A receptors located on serotonergic neuronal somatodendrites inhibit serotonergic cell firing and modulate 5-HT transmitter release into the synaptic cleft. Consequently, these 5-HT1A autoreceptors constitute the decisive factor in a negative auto-regulatory loop of serotonin release (Bundgaard et al. 2006). Alterations in 5-HT1A receptor binding have been reported in several neuropsychiatric disorders such as anxiety (Akimova et al. 2009) and depression (Drevets et al. 2007). One recent animal study provided evidence for seasonal alterations in 5-HT1A receptor expression (Naumenko et al. 2008). The study by Spindelegger et al. investigated 36 healthy drug-naïve subjects by quantifying 5-HT1A BPND using PET and the highly specific 11C-labelled tracer [N-(2-(1-(4-(2-methoxyphenyl)-1-piperazinyl)ethyl))-N-(2-pyridyl)-cyclohexane-carboxamide] (carbonyl-[11C]WAY-100635). Individual exposure to external factors such as global radiation (defined as total of direct solar radiation and diffuse sky radiation received by a unit horizontal surface) correlated with regional 5-HT1A BPND, demonstrating a positive correlation between the accumulated (5 days prior to PET scan) amount of global radiation and 5-HT1A receptor binding. Moreover, this investigation showed a significant difference between the groups of subjects exposed to low versus high amounts of global radiation (see Fig. 8.2). Up to 30 % differences in regional 5-HT1A BPND were found between the different exposure groups.
Mean serotonin-1A receptor binding potential (5-HT1A BPND) in subjects exposed to low amounts of global radiation (left) versus subjects exposed to high amounts of global radiation (right) showing 5-HT1A BPND values in the group exposed to a low amount of global radiation, especially in limbic brain regions. Subjects exposed to low amounts of global radiation (n = 22): 5-day accumulation of global radiation was lower than 8,946 J/cm2; subjects exposed to high amounts of global radiation (n = 14): 5-day accumulation of global radiation was higher than 8,946 J/cm2 (Modified according to Spindelegger et al. (2012))
In regard to the effects of season on serotonergic neurotransmission, higher SERT availability in times of less light was revealed in four different studies using SPECT and PET. Furthermore, serotonin receptor binding has been shown to be influenced by external factors such as global radiation. However, these results warrant independent replication. Altogether, these findings underline the importance of research in the field of the effects of season on the serotonergic system, as they provide additional insights into regulatory processes in 5-HT neurotransmission. Consequently, future studies investigating serotonergic target structures such as SERT or serotonin receptors should consider seasonal effects.
Dopamine neurotransmission has been suggested to be regulated in part by photoperiodic and light-dependent rhythms. Dopamine is strongly involved in physiological functions such as motor control, cognition, reward, emotion and memory processes (Dalley and Everitt 2009). Limited evidence for seasonal effects on dopamine neurotransmission is provided by SPECT studies mentioned before (Neumeister et al. 2001; Tsai et al. 2011) and a PET study by Eisenberg et al. (2010) reporting higher striatal fluorine-18-L-dihydroxyphenylalanine ([18F]DOPA) uptake in autumn and winter as compared to spring and summer. Eisenberg and colleagues investigated a large sample of healthy subjects (n = 86) showing higher striatal Ki values in subjects scanned during the fall and winter season. The increased Ki values in the posterior putamen were interpreted as greater presynaptic dopamine synthesis and storage capacity in this region. Based on the resulting higher levels of dopamine in times of less light, these results would be in line with recent findings of lower striatal [123I]IBZM binding in times of less light exposure (Tsai et al. 2011) due to greater competition at postsynaptic D2/3 receptors. Since there is only limited evidence supporting this hypothesis, further investigations are needed to clarify the underlying mechanisms.
5 Summary
Seasonal affective disorder and its subsyndromal form constitute a prevalent neuropsychiatric disorder characterised by severe seasonal changes in mood and behaviour. Atypical or reverse vegetative symptoms such as increased sleep duration, hyperphagia and subsequent weight gain are frequent in SAD, and severity of symptoms tends to correlate positively with latitude. During the last decades, only a limited number of studies specifically investigating SAD have been conducted. Apart from brain metabolic changes, monoamine systems in the human brain have been revealed to have a key role in seasonal modulation of behavioural and psychological domains. One of the most consistent findings is the seasonal variation of serotonin transporters with higher availability in winter as shown by four neuroimaging studies using different imaging technologies (Buchert et al. 2006; Kalbitzer et al. 2010; Praschak-Rieder et al. 2008; Ruhe et al. 2009). Other intriguing findings, such as seasonal changes in dopamine neurotransmission (Eisenberg et al. 2010; Tsai et al. 2011) or light-induced alterations in serotonin receptors (Spindelegger et al. 2012), are still awaiting replication. Given the lack of neuroimaging studies in SAD, further research (e.g. seasonal variations in monoamine oxidase activity) is needed to enhance the progress in understanding the molecular background of SAD and seasonal changes in the human brain. Furthermore, knowledge of seasonal effects on brain monoamine function might lead to additional treatment strategies in SAD.
References
Akimova E, Lanzenberger R, Kasper S (2009) The serotonin-1A receptor in anxiety disorders. Biol Psychiatry 66:627–635
Avasthi A, Sharma A, Gupta N et al (2001) Seasonality and affective disorders: a report from North India. J Affect Disord 64:145–154
Boyce P, Parker G (1988) Seasonal affective disorder in the southern hemisphere. Am J Psychiatry 145:96–99
Brucke T, Kornhuber J, Angelberger P et al (1993) SPECT imaging of dopamine and serotonin transporters with [123I]beta-CIT. Binding kinetics in the human brain. J Neural Transm Gen Sect 94:137–146
Buchert R, Schulze O, Wilke F et al (2006) Is correction for age necessary in SPECT or PET of the central serotonin transporter in young, healthy adults? J Nucl Med 47:38–42
Bundgaard C, Larsen F, Jorgensen M et al (2006) Mechanistic model of acute autoinhibitory feedback action after administration of SSRIs in rats: application to escitalopram-induced effects on brain serotonin levels. Eur J Pharm Sci 29(5):394–404
Caetano SC, Hatch JP, Brambilla P et al (2004) Anatomical MRI study of hippocampus and amygdala in patients with current and remitted major depression. Psychiatry Res 132:141–147
Campbell S, Macqueen G (2004) The role of the hippocampus in the pathophysiology of major depression. J Psychiatry Neurosci 29:417–426
Carlsson A, Svennerholm L, Winblad B (1980) Seasonal and circadian monoamine variations in human brains examined post mortem. Acta Psychiatr Scand Suppl 280:75–85
Coffey CE, Wilkinson WE, Weiner RD et al (1993) Quantitative cerebral anatomy in depression. A controlled magnetic resonance imaging study. Arch Gen Psychiatry 50:7–16
Cohen RM, Gross M, Nordahl TE et al (1992) Preliminary data on the metabolic brain pattern of patients with winter seasonal affective disorder. Arch Gen Psychiatry 49:545–552
Dalley JW, Everitt BJ (2009) Dopamine receptors in the learning, memory and drug reward circuitry. Semin Cell Dev Biol 20:403–410
Drevets WC, Price JL, Simpson JR Jr et al (1997) Subgenual prefrontal cortex abnormalities in mood disorders. Nature 386:824–827
Drevets WC, Thase ME, Moses-Kolko EL et al (2007) Serotonin-1A receptor imaging in recurrent depression: replication and literature review. Nucl Med Biol 34:865–877
Eisenberg DP, Kohn PD, Baller EB et al (2010) Seasonal effects on human striatal presynaptic dopamine synthesis. J Neurosci 30:14691–14694
Frodl T, Meisenzahl E, Zetzsche T et al (2002a) Enlargement of the amygdala in patients with a first episode of major depression. Biol Psychiatry 51:708–714
Frodl T, Meisenzahl EM, Zetzsche T et al (2002b) Hippocampal changes in patients with a first episode of major depression. Am J Psychiatry 159:1112–1118
Frodl T, Meisenzahl EM, Zetzsche T et al (2003) Larger amygdala volumes in first depressive episode as compared to recurrent major depression and healthy control subjects. Biol Psychiatry 53:338–344
Goyer PF, Schulz PM, Semple WE et al (1992) Cerebral glucose metabolism in patients with summer seasonal affective disorder. Neuropsychopharmacology 7:233–240
Greenwald BS, Kramer-Ginsberg E, Bogerts B et al (1997) Qualitative magnetic resonance imaging findings in geriatric depression. Possible link between later-onset depression and Alzheimer’s disease? Psychol Med 27:421–431
Hardin TA, Wehr TA, Brewerton T et al (1991) Evaluation of seasonality in six clinical populations and two normal populations. J Psychiatr Res 25:75–87
Hebert M, Beattie CW, Tam EM et al (2004) Electroretinography in patients with winter seasonal affective disorder. Psychiatry Res 127:27–34
Kalbitzer J, Erritzoe D, Holst KK et al (2010) Seasonal changes in brain serotonin transporter binding in short serotonin transporter linked polymorphic region-allele carriers but not in long-allele Homozygotes. Biol Psychiatry 67:1033–1039
Kasper S (1994) Diagnosis, epidemiology and therapy of seasonal depression. Nervenarzt 65:69–72
Kasper S, Rogers SL, Yancey AL et al (1988) Phototherapy in subsyndromal seasonal affective disorder (S-SAD) and “diagnosed” controls. Pharmacopsychiatry 21:428–429
Kasper S, Wehr TA, Bartko JJ et al (1989) Epidemiological findings of seasonal changes in mood and behavior. A telephone survey of Montgomery County, Maryland. Arch Gen Psychiatry 46:823–833
Kasper S, Neumeister A, Praschak-Rieder N et al (1996) Serotonergic mechanisms in the pathophysiology and treatment of seasonal affective disorder. de Gruyter, Atlanta/Berlin
Kessing LV, Willer IS, Knorr U (2011) Volume of the adrenal and pituitary glands in depression. Psychoneuroendocrinology 36:19–27
Krishnan KR, Doraiswamy PM, Lurie SN et al (1991) Pituitary size in depression. J Clin Endocrinol Metab 72:256–259
Lam RW, Levitan RD (2000) Pathophysiology of seasonal affective disorder: a review. J Psychiatry Neurosci 25:469–480
Lam RW, Zis AP, Grewal A et al (1996) Effects of rapid tryptophan depletion in patients with seasonal affective disorder in remission after light therapy. Arch Gen Psychiatry 53:41–44
Lam RW, Levitt AJ, Levitan RD et al (2006) The Can-SAD study: a randomized controlled trial of the effectiveness of light therapy and fluoxetine in patients with winter seasonal affective disorder. Am J Psychiatry 163:805–812
Lambert GW, Reid C, Kaye DM et al (2002) Effect of sunlight and season on serotonin turnover in the brain. Lancet 360:1840–1842
Laruelle M (2000) Imaging synaptic neurotransmission with in vivo binding competition techniques: a critical review. J Cereb Blood Flow Metab 20:423–451
Laruelle M, Baldwin RM, Malison RT et al (1993) SPECT imaging of dopamine and serotonin transporters with [123I]beta-CIT: pharmacological characterization of brain uptake in nonhuman primates. Synapse 13:295–309
Lavoie MP, Lam RW, Bouchard G et al (2009) Evidence of a biological effect of light therapy on the retina of patients with seasonal affective disorder. Biol Psychiatry 66:253–258
Levitan RD (2007) The chronobiology and neurobiology of winter seasonal affective disorder. Dialogues Clin Neurosci 9:315–324
Levitan RD, Masellis M, Lam RW et al (2006) A birth-season/DRD4 gene interaction predicts weight gain and obesity in women with seasonal affective disorder: a seasonal thrifty phenotype hypothesis. Neuropsychopharmacology 31:2498–2503
Levitan RD, Kaplan AS, Davis C et al (2010) A season-of-birth/DRD4 interaction predicts maximal body mass index in women with bulimia nervosa. Neuropsychopharmacology 35:1729–1733
Lewy AJ, Bauer VK, Cutler NL et al (1998) Morning vs evening light treatment of patients with winter depression. Arch Gen Psychiatry 55:890–896
Magnusson A, Partonen T (2005) The diagnosis, symptomatology, and epidemiology of seasonal affective disorder. CNS Spectr 10:625–634, quiz 621–614
Mersch PP, Middendorp HM, Bouhuys AL et al (1999) Seasonal affective disorder and latitude: a review of the literature. J Affect Disord 53:35–48
Miranda-Scippa AM, Pires ML, Handfas BW et al (2008) Pituitary volume and the effects of phototherapy in patients with seasonal winter depression: a controlled study. Rev Bras Psiquiatr 30:50–54
Morrissey SA, Raggatt PT, James B et al (1996) Seasonal affective disorder: some epidemiological findings from a tropical climate. Aust N Z J Psychiatry 30:579–586
Murthy NV, Selvaraj S, Cowen PJ et al (2010) Serotonin transporter polymorphisms (SLC6A4 insertion/deletion and rs25531) do not affect the availability of 5-HTT to [11C] DASB binding in the living human brain. Neuroimage 52:50–54
Naumenko VS, Tkachev SE, Kulikov AV et al (2008) The brain 5-HT1A receptor gene expression in hibernation. Genes Brain Behav 7:300–305
Neumeister A, Praschak-Rieder N, Hesselmann B et al (1998a) Effects of tryptophan depletion in fully remitted patients with seasonal affective disorder during summer. Psychol Med 28:257–264
Neumeister A, Turner EH, Matthews JR et al (1998b) Effects of tryptophan depletion vs catecholamine depletion in patients with seasonal affective disorder in remission with light therapy. Arch Gen Psychiatry 55:524–530
Neumeister A, Pirker W, Willeit M et al (2000) Seasonal variation of availability of serotonin transporter binding sites in healthy female subjects as measured by [123I]-2 beta-carbomethoxy-3 beta-(4-iodophenyl)tropane and single photon emission computed tomography. Biol Psychiatry 47:158–160
Neumeister A, Willeit M, Praschak-Rieder N et al (2001) Dopamine transporter availability in symptomatic depressed patients with seasonal affective disorder and healthy controls. Psychol Med 31:1467–1473
Pantel J, Schroder J, Essig M et al (1997) Quantitative magnetic resonance imaging in geriatric depression and primary degenerative dementia. J Affect Disord 42:69–83
Potkin SG, Zetin M, Stamenkovic V et al (1986) Seasonal affective disorder: prevalence varies with latitude and climate. Clin Neuropharmacol 9(Suppl 4):181–183
Praschak-Rieder N, Willeit M (2003) Treatment of seasonal affective disorders. Dialogues Clin Neurosci 5:389–398
Praschak-Rieder N, Willeit M (2012) Imaging of seasonal affective disorder and seasonality effects on serotonin and dopamine function in the human brain. Curr Top Behav Neurosci 11:149–167
Praschak-Rieder N, Neumeister A, Willeit M et al (1998) HMPAO-SPECT in SAD patients before and after light therapy. Biol Psychiatry 43:17
Praschak-Rieder N, Willeit M, Wilson AA et al (2008) Seasonal variation in human brain serotonin transporter binding. Arch Gen Psychiatry 65:1072–1078
Rosen LN, Targum SD, Terman M et al (1990) Prevalence of seasonal affective disorder at four latitudes. Psychiatry Res 31:131–144
Rosenthal NE, Sack DA, Gillin JC et al (1984) Seasonal affective disorder. A description of the syndrome and preliminary findings with light therapy. Arch Gen Psychiatry 41:72–80
Ruhe HG, Booij J, Reitsma JB et al (2009) Serotonin transporter binding with [123I]beta-CIT SPECT in major depressive disorder versus controls: effect of season and gender. Eur J Nucl Med Mol Imaging 36:841–849
Schwartz PJ, Loe JA, Bash CN et al (1997) Seasonality and pituitary volume. Psychiatry Res 74:151–157
Shah PJ, Ebmeier KP, Glabus MF et al (1998) Cortical grey matter reductions associated with treatment-resistant chronic unipolar depression. Controlled magnetic resonance imaging study. Br J Psychiatry 172:527–532
Sheline YI (1996) Hippocampal atrophy in major depression: a result of depression-induced neurotoxicity? Mol Psychiatry 1:298–299
Sheline YI, Gado MH, Price JL (1998) Amygdala core nuclei volumes are decreased in recurrent major depression. Neuroreport 9:2023–2028
Spindelegger C, Stein P, Wadsak W et al (2012) Light-dependent alteration of serotonin-1A receptor binding in cortical and subcortical limbic regions in the human brain. World J Biol Psychiatry 13(6):413–422
Staley JK, Basile M, Flynn DD et al (1994) Visualizing dopamine and serotonin transporters in the human brain with the potent cocaine analogue [125I]RTI-55: in vitro binding and autoradiographic characterization. J Neurochem 62:549–556
Steffens DC, Krishnan KR (1998) Structural neuroimaging and mood disorders: recent findings, implications for classification, and future directions. Biol Psychiatry 43:705–712
Terman M (2006) Review: light therapy is an effective treatment for seasonal affective disorder. Evid Based Ment Health 9:21
Terman M, Terman JS, Quitkin FM et al (1989) Light therapy for seasonal affective disorder. A review of efficacy. Neuropsychopharmacology 2:1–22
Thompson C, Isaacs G (1988) Seasonal affective disorder–a British sample. Symptomatology in relation to mode of referral and diagnostic subtype. J Affect Disord 14:1–11
Tsai HY, Chen KC, Yang YK et al (2011) Sunshine-exposure variation of human striatal dopamine D(2)/D(3) receptor availability in healthy volunteers. Prog Neuropsychopharmacol Biol Psychiatry 35:107–110
Vandewalle G, Hebert M, Beaulieu C et al (2011) Abnormal hypothalamic response to light in seasonal affective disorder. Biol Psychiatry 70:954–961
Varnas K, Halldin C, Hall H (2004) Autoradiographic distribution of serotonin transporters and receptor subtypes in human brain. Hum Brain Mapp 22:246–260
Wehr TA, Sack DA, Rosenthal NE (1987) Seasonal affective disorder with summer depression and winter hypomania. Am J Psychiatry 144:1602–1603
Willeit M, Praschak-Rieder N, Neumeister A et al (2000) [123I]-beta-CIT SPECT imaging shows reduced brain serotonin transporter availability in drug-free depressed patients with seasonal affective disorder. Biol Psychiatry 47:482–489
Willeit M, Sitte HH, Thierry N et al (2008) Enhanced serotonin transporter function during depression in seasonal affective disorder. Neuropsychopharmacology 33:1503–1513
Winkler D, Willeit M, Praschak-Rieder N et al (2002) Changes of clinical pattern in seasonal affective disorder (SAD) over time in a German-speaking sample. Eur Arch Psychiatry Clin Neurosci 252:54–62
Wirz-Justice A, Bucheli C, Graw P et al (1986) Light treatment of seasonal affective disorder in Switzerland. Acta Psychiatr Scand 74:193–204
Witkovsky P (2004) Dopamine and retinal function. Doc Ophthalmol 108:17–40
Young SN, Leyton M (2002) The role of serotonin in human mood and social interaction. Insight from altered tryptophan levels. Pharmacol Biochem Behav 71:857–865
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer-Verlag Berlin Heidelberg
About this chapter
Cite this chapter
Spindelegger, C., Willeit, M., Praschak-Rieder, N., Lanzenberger, R., Kasper, S. (2014). Neuroimaging in Seasons and Winter Depression. In: Dierckx, R., Otte, A., de Vries, E., van Waarde, A., den Boer, J. (eds) PET and SPECT in Psychiatry. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-642-40384-2_8
Download citation
DOI: https://doi.org/10.1007/978-3-642-40384-2_8
Published:
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-642-40383-5
Online ISBN: 978-3-642-40384-2
eBook Packages: MedicineMedicine (R0)