Abstract
Most moulds propagate and disseminate via release of airborne spores, but none surpass the capacity of Aspergillus fumigatus to cause human disease. A. fumigatus is a filamentous fungus that is capable of saprophytic growth in the natural environment and parasitic growth in susceptible human and animal hosts. How does the transition from saprophytic soil-dweller to life-threatening pathogen come about? As the quest for A. fumigatus virulence factors continues apace, our understanding of the pathogenic qualities expressed by this “accidental pathogen” has reached a new, higher order appreciation of the host–pathogen interaction and how this fragile interrelationship might be tipped towards or against initiation or resolution of disease. A. fumigatus has the ability, more than any other airborne fungal pathogen, to radically alter the trade-off between parasitic growth rate and host response in favour of fungal dominance. In this chapter, we will examine the pivotal processes occurring after spore inhalation and incorporate them into a three-step cause-and-effect framework, comprising a comprehensive overview of the host and pathogen factors that drive disease.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Cystic Fibrosis Transmembrane Conductance Regulator
- Invasive Aspergillosis
- Pathogen Interaction
- Invasive Pulmonary Aspergillosis
- Conidial Germination
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
I. Introduction
Most moulds propagate and disseminate via release of airborne spores, but none surpass the capacity of Aspergillus fumigatus to cause human disease. A. fumigatus is a filamentous fungus that is capable of saprophytic growth in the natural environment and parasitic growth in susceptible human and animal hosts. How does the transition from saprophytic soil-dweller to life-threatening pathogen come about? As the quest for A. fumigatus virulence factors continues apace, our understanding of the pathogenic qualities expressed by this “accidental pathogen” has reached a new, higher order appreciation of the host–pathogen interaction and how this fragile interrelationship might be tipped towards or against initiation or resolution of disease.
A. fumigatus has the ability, more than any other airborne fungal pathogen, to radically alter the trade-off between parasitic growth rate and host response in favour of fungal dominance. In this chapter, we will examine the pivotal processes occurring after spore (or conidia) inhalation and incorporate them into a three-step cause-and-effect framework, comprising a comprehensive overview of the host and pathogen factors that drive disease.
The genus Aspergillus includes several hundred species of filamentous, ascomycetous mould fungi, which disseminate via spore release (Bennet 2010; Baker and Bennet 2008). Ubiquitous on Earth, and occurring with a worldwide distribution, it is likely that all of these species contribute to recycling of planetary carbon and nitrogen via degradation of organic matter. Several species have even greater significance to mankind, serving as conventional workhorses of food production or genetic study, or agents of devastating disease in plants and animals (Bennet 2010; Baker and Bennet 2008). A. fumigatus exerts, via a range of infectious and inflammatory diseases, a huge toll upon public health (Brown et al. 2012) and is the most significant mould pathogen of human beings (Latge 1999). Efforts to define the molecular basis of A. fumigatus pathogenicity have been ongoing for several decades and are continuing in the post-genomic era. Multiple excellent reviews have recently catalogued studies addressing A. fumigatus attributes required for pathogenicity and the host factors predisposing to disease (Dagenais and Keller 2009; Hohl and Feldmesser 2007; Askew 2008; Abad et al. 2010). These works, combined with an ever-increasing understanding of immune responses to infectious microbes, have revealed the intricacy of the A. fumigatus host–pathogen interaction and motivated the scientific community to reach a consensus on the huge significance of host factors in driving disease (Romani 2011). Although it is clear that this pathogen can utilise efficiently the nutrients provided by the mammalian niche and will do so in the absence of an adequate immune challenge, it is also clear that A. fumigatus draws upon multiple physiological traits, operative at distinct stages of the host–pathogen interaction, to exploit the weakened host and accomplish longevity in the host environment. Recent research has identified highly significant events that occur during the establishment of disease and contribute to the outcome of the host–pathogen interaction at the level of the whole animal.
II. Aspergillus fumigatus: A Three-Step Transition from Saprotroph to Pathogen
There are many host and pathogen factors that can drive initiation and establishment of human Aspergillus-related disease. To fully understand the pathogenic potential of A. fumigatus one must integrate a plethora of experimental and clinical data to compile a working model of the host–pathogen interaction. Currently, it remains difficult to assign relative measures of significance to many of these factors, and a more quantitative understanding will be required if immunomodulatory interventions are to be effectively implemented. Progress towards this aim will also permit a more standardised approach to the study of the host–pathogen interaction and place our understanding of it within a context having relevance to all types of Aspergillus-related disease.
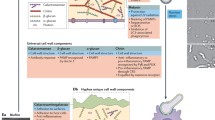
To construct a fully panoramic view of the pathogenic status of A. fumigatus the progression from saprotroph to pathogen is here considered as a series of three temporally distinct stages of the host–pathogen interaction: (1) primary encounter, (2) persistence and (3) permanence (Fig. 2.1). Crucially, for the transition from saprotroph to pathogen to be realised all three stages must be completed thereby leading to a permanent alteration of host homeostasis. Within this framework, the activities of both host and pathogen can be readily incorporated as causes of disease (Fig. 2.1) and events that impact the outcome of infection are readily identifiable. In this chapter, the extent to which each of these activities impacts upon disease will be assessed within the context of the available experimental evidence.
Saprotroph to pathogen: a three-step cause-and-effect framework. Diseases caused by A. fumigatus arise as a result of both pathogen and host activities, and the transition from saprotroph to pathogen can be considered as occurring in three stages: primary encounter, persistence and permanence. A quantitative understanding of the contributions made by each factor is currently lacking; however, factors that can alter the outcome of the host–pathogen interaction, at the level of the whole animal host, are indicated in italicised, underlined text
III. Primary Encounter
A. Spore Size, Abundance and Human Exposure
A. fumigatus spores are a common component of the airborne microflora and are usually more abundant in outdoor, rather than indoor, air samples (Torpy et al. 2012). Despite seasonal variations in spore abundance, the estimated number of viable A. fumigatus colony forming units per square metre (CFU/m 2 ) rarely exceeds 100, so A. fumigatus spores are certainly not the most abundant of those we normally breathe. Aspergillus spp. are, however, the fungi most often cultured from the lungs of chronically ill humans (Lass-Florl et al. 1999) and since Cladosporium spores greatly outnumber those of A. fumigatus in outdoor air (O’Gorman and Fuller 2008) the spectrum of disease-causing fungi is not reflected by the results of air surveys. Clearly therefore, some mould spores enjoy a positive advantage in the host.
A. fumigatus sporulates abundantly, producing green pigmented, uninucleate, mitotic spores of approximately 2 μm diameter extruded as chains from aerial hyphae positioned high above the vegetative mycelial mass (Fig. 2.2a). This organisation of newly generated spores ensures efficient dispersal of spores in air currents, wherein A. fumigatus spores remain buoyant and viable for long periods of time. Spore dispersal is influenced by air mass movement, turbulence and thermal convection, with physical characteristics such as size, shape, density and surface texture exacting comparatively minor effects. Terminal velocity (or fall speed) is the dominant physical property, and actinomycete spores (approximating 2 μm diameter) are estimated to fall very slowly at approximately 40 μm/s (Lacey and West 2007). According to the scheme proposed by Lacey and West (2007), their extremely small size promotes the deepest possible penetration of the respiratory tree (Fig. 2.2b).
Organisation of A. fumigatus colonies, conidial structure and airway penetration by particle size. (a) A. fumigatus is a prolific spore producer, generating thousands of uninucleate mitotic spores (conidia) per aerial hypha. Mitotic spores, approximately 2 μm in size, are extruded from aerial hyphae and become airborne. (b) A. fumigatus spores are small enough to achieve deepest possible penetration of the human airway
Stored products, including hay, straw, grain, wood chips and composts, represent sources of A. fumigatus spores that can be aerosolised by human activities. Incredibly, the immediate vicinity of combine harvesters has been found to contain 200 million fungal spores per cubic metre, a concentration capable of eliciting an immediate hypersensitivity reaction (O’Gorman 2011). The potential for exposure to airborne A. fumigatus spores is heightened in the immediate vicinity of refuse dumps and landfill sites, and associated hazards to respiratory health have been formally assessed by Swan et al. (2013). In a recent review of the risk posed by A. fumigatus spores O’Gorman (2011) cites guidelines issued by the UK Environment Agency in 2009 that state that total emissions of bacterial and fungal spores from compost facilities not exceeding 103 CFU/m3 are within “acceptable levels”. Potential environmental reservoirs governing everyday exposures include dust, bedding, water supplies, air handling systems, fresh and dried flowers, the soil of ornamental plants, certain foods including tea, biscuits, fruits and spices, particularly pepper, and the smoking of tobacco and marijuana. Activities such as grass cutting, digging, potting plants, building works and household cleaning also aerosolise A. fumigatus conidia (O’Gorman 2011).
Although initiation of hypersensitivity reactions can be elicited by massive exposures to A. fumigatus spores, infections of immunocompromised hosts are probably initiated by single or repeated exposures to far fewer fungal spores. The true relationship between Aspergillus concentration in the air and the probability of invasive aspergillosis is not quantitatively known, and the circumstances surrounding initiation of infection pose important unanswered questions. It is not certain whether prior carriage of, or repeated exposure to, A. fumigatus spores primes the host for subsequent infection. Some have surmised that large inocula of fungal spores may overwhelm an otherwise adequately functioning immune system (Hope et al. 2005) whereas others have sought correlations between fungal colonisation and subsequent invasive aspergilloses (IA) (Einsele et al. 1998).
Leleu and co-workers (Leleu et al. 2013) used a combination of Bayesian modelling and in vivo experiments to determine the relationship between spore exposure and occurrence of IA. This study determined that in immunosuppressed (neutropenic) mice, exposed by aerosol to A. fumigatus spores, the median exposure dose required to achieve fatal infection of 50% of mice is 1.8–3.2 × 104 A. fumigatus spores. Animals exposed to lower doses were unlikely to contract infections causing death within 6 days of spore exposure. The authors acknowledge that a single exposure to ~ 104 spores is unlikely to occur in humans, where a cumulative dose over time is more likely. However, for patients with prolonged neutropenia or undergoing other immunosuppressive therapies, a significant cumulative infective exposure could be reached within a few hours or days. This study established, for the first time, a direct correlation between aerosol spore burdens and the probability of mammalian infection; however, the relevance of these data to human disease remains speculative because of the marked differences in the exposure conditions and in the respiratory system of mice and humans.
B. Physiological Traits That Favour Pathogenicity
The primary route of infection with Aspergillus is via the inhalation of airborne conidia, which are deposited in the bronchioles or alveolar spaces (Fig. 2.2b). The average size of A. fumigatus conidia (2–3 μm) is ideal for infiltrating deep into the alveoli, whereas larger particles, including the conidia of human pathogens such as A. flavus and A. niger, could be removed more easily by mucociliary clearance of the upper respiratory tract. Evaluation of parameters influencing the germination of Aspergillus conidia revealed significant differences in germination between differing pathogenic species, whereby A. fumigatus germinates much more efficiently and in a shorter time frame than Aspergillus flavus or Aspergillus niger (Araujo and Rodrigues 2004). Following 12 h of incubation in a nutritive medium, the average germination rate of A. niger strains was about 36.5%. A similar rate was obtained more quickly in A. flavus (8 h) and A. fumigatus (5.5 h). At ambient temperatures of up to 30°C, germination rates were similar for all three species; however, at higher temperatures germination of A. flavus decreased by 45%, and in the case of A. niger no germination was possible for 24 h. Thus, among pathogenic Aspergilli, A. fumigatus appears better equipped for initiating growth in the mammalian host.
The importance of thermotolerance for colonisation and infection of mammals is supported by the attenuation of virulence in a mutant lacking the cgrA gene product, which is involved in ribosome biogenesis. ΔcgrA mutants grow normally at 25°C and are fully virulent in a Drosophila insect model (25°C) of infection, but grow slower than the wild-type progenitor isolate at 37°C and are attenuated for virulence in mice (Bhabhra et al. 2004).
C. Host Susceptibility: How Does Immunity Fail the Host?
The potency of A. fumigatus as a pathogen and source of antigens is evidenced by the spectrum of diseases it causes in humans (Fig. 2.3). For example, in severely immunocompromised hosts inhaled spores remain largely unchallenged by innate immune defences and tissue-invasive disease predominates. However, in atopic individuals an over-exuberant response to spore and/or hyphal antigens can prompt life-threatening allergy or chronic pulmonary inflammation (Zmeili and Soubani 2007). Thus, the problem posed by A. fumigatus extends beyond the mere ability to proliferate within the host environment. The disparate outcomes resulting from spore inhalation in immunologically distinct host settings demonstrate the crucial contribution made by the host environment to progression of disease. In humans, Aspergillosis can be broadly grouped into three major categories: invasive aspergillosis (IA; with or without angioinvasion), chronic pulmonary aspergillosis (CPA; including aspergilloma) and allergic bronchopulmonary aspergillosis (ABPA; Fig. 2.3).
Outcomes of A. fumigatus spore inhalation in humans. (a) Allergic bronchopulmonary aspergillosis (ABPA). Patient has longstanding ABPA with central bronchiectasis (white arrow). Total IgE is 500 times higher than normal at 5,000 versus 100 IU/ml. Patient has undergone itraconazole therapy for 7 years with stability and no exacerbations (b) Invasive pulmonary aspergillosis (IPA). Heart transplant recipient with a recent episode of rejection requiring pulse corticosteroids in high dose, presenting with chest complaints and headache. A cavitary lesion is apparent (white arrow), and patient also had multiple abscesses in the brain (c) Chronic pulmonary aspergillosis (CPA). A 65-year-old patient with rheumatoid arthritis and rheumatoid lung disease who gradually deteriorated, with weight loss and severe breathlessness over severalmonths. A cavity is evident (white arrow) that shows early signs of aspergilloma (fungus ball)
D. Invasive Aspergillosis
In an authoritative review of the clinical presentations, risk factors and diagnosis of invasive aspergillosis, Hope and Denning (Hope et al. 2005) described an entire spectrum of distinct IA syndromes having differing pathophysiological profiles. For simplicity we here consider IA to include aspergillosis of both the angioinvasive and non-angioinvasive types, among which invasive pulmonary aspergillosis (IPA) is the commonest manifestation. Zmeili and Soubani (2007) defined seven major risk factors for IA: (i) prolonged neutropenia or neutrophil dysfunction (e.g., as seen in chronic granulomatous disease, CGD), (ii) organ or stem cell transplantation (highest risk is with lung and haematopoietic stem cell transplantation, HSCT), (iii) prolonged (>3 weeks) and high-dose corticosteroid therapy, (iv) haematological malignancy (risk is higher with leukaemia), (v) chemotherapy and (vi) advanced AIDS.
In humans, profound neutropenia (< 100 neutrophils/μL) lasting more than 10–15 days is the major risk factor for invasive mould infections (Portugal et al. 2009) and this provides the major basis for assuming that neutrophils provide the most important defence to the human host. Hope and Denning (Hope et al. 2005) distinguish between angioinvasive and non-angioinvasive aspergilloses on the basis of both clinical presentation, and predisposing risk factors whereby angioinvasive disease occurs exclusively in the neutropenic setting and is characterised by vascular invasion by hyphal elements, coagulative necrosis and haemorrhagic infarction. In contrast, non-angioinvasive disease lacks any evidence of vascular invasion but is characterised by any, all or some of: pyogranulomatous inflammatory infiltrate, inflammatory necrosis or cavitation and occurs in multiple non-neutropenic clinical settings, most significantly following corticosteroid therapy, non-neutropenic HSCT, graft-versus-host disease (GVHD), HIV/AIDS, CGD and solid organ transplantation. Murine models approximating the progression of angioinvasive, non-angioinvasive, CGD bone marrow and solid organ transplantation have provided a useful proxy for the study of the host–pathogen interaction as in all instances the major features of disease progression can be replicated (Clemons and Stevens 2005; Lewis and Wiederhold 2005; Balloy et al. 2005; Pollock et al. 1995). The major drawback of murine experimentation is the large infectious inocula that are routinely employed to achieve measurable read-outs of disease progression. Future advances in bioimaging and mathematical modelling will permit the detailed study of diseases resulting from lower inocula.
E. Chronic Pulmonary Aspergillosis
Chronic forms of pulmonary aspergillosis are distinguishable from IA by the timescale of disease progression (Hope et al. 2005). In contrast to IPA, CPAs run a slowly progressive course over weeks to months and include several syndromes, variously characterised by slowly progressive cavitary lung disease, chronic respiratory symptoms and the presence of precipitating antibodies to Aspergillus spp. Hope and Denning (Hope et al. 2005) noted that chronic necrotising pulmonary aspergillosis (CPNA) may present with direct invasion of pulmonary parenchyma by hyphal elements but for the most part no clear evidence of parenchymal invasion can be secured, despite progressive tissue damage. Prior structural lung disease appears to be a crucial factor and defects in systemic immunity may also be important. The precise mechanism of new cavity formation remains unclear, and where cavities are present they may or may not contain fungus balls. The presence, or not, of an aspergilloma provides the basis upon which the distinction between aspergilloma (fungus ball) and CPA (cavitating disease) is made. Aspergillus tracheobronchitis is a further variant of CPA, occurring frequently in solid organ transplantations that incorporate calcineurin inhibition regimens. Tracheobronchitis occurs higher in the respiratory tree and involves profuse, but superficial inflammation with a mucus exudate. The depth of infection ranges from no, to extensive, involvement of the bronchial wall (Herbst et al. 2013; Zmeili and Soubani 2007).
F. Allergic Bronchopulmonary Aspergillosis
Allergic bronchopulmonary aspergillosis (ABPA) is an immunological disorder caused by hypersensitivity to Aspergillus fumigatus and occurs predominantly in people with asthma or cystic fibrosis (Patterson and Strek 2010; Zmeili and Soubani 2007). Sensitisation to Aspergillus prompts activation of T helper 2 lymphocytes, which play a key role in recruiting eosinophils and other inflammatory mediators. Clinical presentations therefore include granulomatous inflammation consisting of histiocytes, lymphocytes and eosinophils. Local inflammation results in mucus production, airway hyperreactivity and, ultimately, bronchiectasis in which fungal hyphae may be seen, but without evidence of tissue invasion. Early diagnosis and treatment is likely to prevent disease progression, parenchymal damage and loss of lung function. To this end, ABPA is defined by a combination of clinical and laboratory criteria that include asthma, serum eosinophilia, elevated total IgE, pulmonary opacity, bronchiectasis and sensitisation to A. fumigatus antigens by skin testing. Treatment with corticosteroids can be effective but may be required indefinitely. The incidence of ABPA in patients with asthma is approximately 2%, and 1–15% of cystic fibrosis sufferers develop ABPA (Sorci et al. 2011; Agarwal 2009; Eaton et al. 2000).
G. Aspergillosis in Animals
Aspergillosis is relatively uncommon in other mammals, but dogs, horses, cows and dolphins are susceptible and birds are particularly susceptible to infection. Avian aspergillosis affects both immunocompetent and immunosuppressed birds, in particular turkeys, penguins, raptors and waterfowl. Immunosuppression caused by forced production or physical exertion can predispose to infection. As in humans, A. fumigatus is the major pathogen (Tell 2005).
H. Spore Composition
A. fumigatus spores are the particles with highest relevance to the primary encounter between host and pathogen. Dependent upon the immunosuppressive regimen adopted, germination of A. fumigatus spores in murine airways can take as little as 6–8 h (severely neutropenic hosts) or more than 24 h (corticosteroid-treated hosts) (Balloy et al. 2005). Spores can therefore enter into extended interactions with host cells prior to germination or elimination. This is a key consideration in the study of the Aspergillus host–pathogen interaction because (a) the antigenic identity of spores, swollen conidia and hyphae differs considerably and (b) in contrast to Candida albicans, which conditionally undergoes filamentation in response to relevant stimuli, A. fumigatus is obliged to undergo the morphogenetic transition from uninucleate single cells to multinucleate hyphae during vegetative growth. During IA, this transition might be rapid or slow, but it will always occur.
Concordant with a role for adhesion to host cells during initiation of mammalian infection, histological analysis of murine lung tissue at early time-points of experimental murine aspergillosis reveals the apparent adherence of spores to alveolar and bronchial epithelial cells. The molecular basis for such adhesion has been probed via in vitro experimentation, revealing that spores of A. fumigatus specifically bind to extracellular matrix (ECM) components of A549 epithelial cells. They also bind specifically to fibrinogen, fibronectin, laminin, type I collagen, and type IV collagen. Pre-incubation of spores with an Arg-Gly-Asp tripeptide inhibits binding to fibronectin and type I collagen by 50% (Tronchin et al. 1997; Bouchara et al. 1997; Penalver et al. 1996; Gil et al. 1996). Both carbohydrate and protein molecules on the conidial surface are involved in binding to host proteins (Sheppard 2011).
The outermost cell-wall layer of the A. fumigatus conidium is adorned with a mesh of interwoven rodlet proteins (Fig. 2.2a), including an immunoprotective rodlet protein RodA, which confers conidial hydrophobicity. A rodlet-lacking A. fumigatus mutant, ΔrodA, (Thau et al. 1994) is defective in adherence to collagen and albumin, but retains the capacity to bind host cells (both in vivo and in vitro) and is fully virulent. In a modified murine model of pulmonary aspergillosis, using milder immunosuppressive drug regimens, Shibuya and colleagues (Shibuya et al. 1999) studied the inflammatory response to infections with A. fumigatus wild-type and ΔrodA conidia. In comparison to mice infected with the wild-type isolate, pulmonary lesions induced by the rodletless mutant were limited and inflammatory responses were weak. The mechanistic basis of this pathogenic phenotype was proposed as being due to RodA-mediated depletion of neutrophils and macrophages during the early stages of infection. However, although this hypothesis has not formally been disproved in whole animal studies, it is now clear that the RodA hydrophobin acts by exerting an immunoprotective barrier to recognition by host cells (Aimanianda et al. 2009).
Early observations of the outermost conidial surfaces, conducted via electron microscopy, determined that spore swelling and germination lead to alterations in surface characteristics and adherence of A. fumigatus conidia. Furthermore, the cell wall was noted to undergo reorganisation during swelling and germination (Tronchin et al. 1995). The ultrastructural appearance of the conidial cell wall differs significantly from that of hyphal cells (Bernard and Latge 2001) whereby the conidial cell wall is composed of a dense pigmented outer layer (containing melanin) and a translucent inner layer (Fig. 2.2a). Wasylnka and colleagues (Wasylnka et al. 2001) showed that A. fumigatus conidia bind significantly better than those of other Aspergillus species to intact lung cell basal lamina and, seeking the mechanism of this binding, found that neither desialylation nor complete deglycosylation of fibronectin decreased the binding of A. fumigatus conidia. This suggested that fibronectin binding was not oligosaccharide-mediated, a finding that was further supported by binding of A. fumigatus spores to a nonglycosylated 40 kDa fragment of fibronectin containing the glycosaminoglycan (GAG)-binding domain. Because binding could be inhibited by negatively charged carbohydrates, the authors proposed a novel mechanism of conidial attachment whereby adherence to fibronectin (and other basal lamina proteins) might be mediated via negatively charged carbohydrates on the conidial surface. In another study, the same authors demonstrated that surface-localised sialic acids, at an estimated density of 7 × 105 sialic acid residues per conidium, mediated adhesion of conidia to the positively charged polymer poly(l-lysine) (Wasylnka and Moore 2000). Thus, sialic acids on the conidial wall may be involved in adhesion to fibronectin as such binding is strongly inhibited in the presence of a sialylated glycoprotein. Sialic acids are terminal components of many glycoproteins and glycolipids, contributing to the structural properties of these molecules and regulating cellular and molecular interactions (Kelm and Schauer 1997). Sialic acid residues can act as, or to mask, recognition sites such as subterminal carbohydrate structures or proteins. Although the role of such residues in mediating the binding of pathogens to host cells has been documented over many years, the identity of the A. fumigatus sialic acid-presenting moiety is unknown.
Levdansky and colleagues (Sharon et al. 2011) performed a molecular genetic analysis of conidial cell wall integrity, initially focusing upon the cell wall-associated protein CspA, originally identified as one of four putatively GPI-anchored cell wall proteins whose gene sequences contained different numbers of DNA repeats in different patient isolates. The predicted cpaA translation product includes a long serine-threonine-proline-rich N-terminal region, followed by a variably (18–47 repeats) large six-amino-acid serine-proline [P-G-Q-P-S-(A/V)]-rich tandem repeat region having significant homology to the repeat domains found in mammalian type XXI collagen. A myc-tagged cspA-encoded protein was detectable in hydrofluoric acid extracts of conidial cell walls, suggesting covalent linkage of CspA to cell wall glucan via a GPI anchor. Further, discrepancy between predicted and actual sizes of the protein suggests heavy glycosylation. Immunofluorescence microscopy revealed a hyphal-specific expression profile (6–12 h post-germination). However, western blotting revealed an abundance of the protein in dormant and swollen conidia. Taken together, these observations suggest that CspA is located below the cell wall surface and becomes unmasked as germination proceeds. In support of this theory, sonication of conidia, intended to remove the outer hydrophobin layer, significantly heightened surface exposure of CspA. In combination with deletions of genes encoding the GPI-anchored cell wall proteins ecm33 or gel2, cpsA deletion additively affects conidal adhesion to culture-derived ECM, and leads to morphological abnormality of the conidial cell wall, which exhibits defective layering of melanin-rich and carbohydrate-rich domains, and high levels of exposed chitin, mannose and glucan. Although these structural reorganisations of the cell wall could be correlated with increased internalisation of the double mutants by human monocyte-derived macrophages, and heightened susceptibility to hyphal damage mediated by polymorphonuclear monocytes, virulence was unaltered in neutropenic mice.
I. Immunological Tolerance of Inhaled Aspergillus fumigatus Spores
Hyperinflammatory responses to inhaled spores occur in patients and mice suffering from CGD (Song et al. 2011; Patterson and Strek 2010) or ABPA (Patterson and Strek 2010). Clearly then, A. fumigatus spores are potently capable of evoking immune responses in the mammalian host and, given the frequency with which A. fumigatus spores enter the human pulmonary tract, mammalian immunity has evolved to cope with this omnipotent challenge. In 2009, Aimanianda and co-workers (Aimanianda et al. 2009), in attempting to dissect the reasons for the immunologically inert nature of the dormant conidia, made some findings that could explain this phenomenon. Analysis of a hydrofluoric acid extract derived from A. fumigatus conidia identified three protein species, the molecular weights of which (32, 16 and 14.5 kDa) corresponded, respectively, to the dimeric form of the native RodA protein, native RodA and partially degraded or processed RodA. Crucially, RodA was undetectable in fungal culture supernatant following conidial germination, indicating that the RodA rodlet layer, covalently bound to the cell wall of dormant conidia (Figs. 2.2 and 2.4), was shed upon spore germination. RodA extracted directly from A. fumigatus condia was found to be immunologically inert. This is in contrast to germinating conidia, which prompt, in dendritic cells, significant expression of co-stimulatory molecules (CD80, CD86, CD40 and CD83) and the antigen-presenting molecule human leukocyte antigen DR (HLA-DR), and secretion of pro- and inflammatory and anti-inflammatory cytokines. The removal of RodA, either chemically (using hydrofluoric acid), genetically (ΔrodA mutant) or biologically (germination) resulted in immune activation. Thus, the hydrophobic rodlet layer on the A. fumigatus conidial cell surface acts as an “immunological silencer”. A similar effect was documented in murine alveolar macrophages where inflammatory cytokines, chemokines and reactive oxygen intermediates (produced in response to hydrofluoric acid-treated conidia, ΔrodA dormant conidia and germinated conidia) were undetectable in macrophages exposed to dormant conidia or RodA protein. The authors concluded that the surface rodlet layer of the conidial cell wall makes A. fumigatus conidia inert to both innate and adaptive immunity. Evidence first documented by Hohl et al. (2005) revealed a further layer of immunomodulatory control, manifested by the gradual exposure of cell surface β-glucan during swelling and germination of A. fumigatus spores (see subsequent discussions).
First encounter, immune evasion and fungal persistence in the host. A. fumigatus spores, both resting and swollen, can prompt inflammatory responses in epithelial cells (ECs). ECs internalise both resting and swollen spores, eliciting differential responses to each morphotype. A. fumigatus spores are adorned with a layer of proteinaceous rodlets, which are shed upon swelling to reveal cell-wall-associated polysaccharides, some of which act as pathogen-associated molecular patterns. Orange pentagons represent β-glucan moieties
IV. Persistence
A. Spore Germination
In environments conducive to germination, A. fumigatus spores become metabolically active and increase in size, eventually germinating to produce tip-extending elongated cells called hyphae, a hallmark characteristic of filamentous fungi. In most fungi, conidial germination is governed by moisture, oxygen and nutrients and, when dormancy is broken, A. fumigatus conidia begin nuclear division and morphological development (Momany and Taylor 2000). This morphological transition multiply impacts upon A. fumigatus pathogenicity (Fig. 2.4), from shedding of the RodA hydrophobic layer (Aimanianda et al. 2009) to gradual exposure of pathogen-associated molecular pattern molecules (PAMPs) and activation of immune responses (Hohl et al. 2005) and invasion of the host epithelium (Kamai et al. 2009).
Recognising that morphological landmarks coincide with specific events in the S. cerevisiae cell cycle, Momany and Taylor (2000) sought similar landmarks in the transition of A. fumigatus and Aspergillus nidulans from conidia to hyphae. To score A. fumigatus cells for mitotic and morphological events, Hoechst 33258 and calcofluor white, respectively, were used to stain fungal cells. Following inoculation into rich medium, the first nuclear division in A. fumigatus occurs after a lag of just over 4 h, with subsequent nuclear divisions every 45 min. This represents a significantly faster rate of nuclear duplication than that observed in A. nidulans, where nuclei divide once per hour. Initially spherical, conidia of both A. fumigatus and A. nidulans assume a pear-type shape that indicates that an axis of polarity has been established. This event often (22%) happened prior to the first nuclear division in A. fumigatus cells, which also differs significantly from the less pathogenic A. nidulans where only 12% were polarised after the first mitosis. A further important difference between the morphogenetic transitions of these two species included a reduction in second germ tube emergence in A. fumigatus (19% versus 98%) after the fifth mitosis. The authors proposed that by directing growth primarily in one direction, the lower percentage of second germ tubes allows A. fumigatus to scavenge nutrients more efficiently in the host.
Dague and colleagues (2008) used real-time atomic force microscopy with a temperature-controlled 37°C stage to probe the structural and Physiochemical dynamics of single A. fumigatus conidia during germination. By capturing images of the same cell after 20, 60 and 120 min, significant swelling could be quantified. Ultrastructural alterations were imaged via high-resolution images of the spore surface (Fig. 2.2a). At early germination times, the spore surface was observed to be covered with an array of 10 nm diameter rodlets. Notably, dramatic changes in cell surface structure were observed after 2 h of germination, corresponding to the swelling of the conidium. At this point the rodlet layer was found to alter into a layer of amorphous material, thereby buttressing the hypothesis that significant remodelling of the cell surface, including shedding of RodA (Aimanianda et al. 2009), represents a significant restructuring event during spore germination.
From a molecular perspective, the regulatory mechanisms that govern conidial germination in A. fumigatus have not been fully described, and much is extrapolated from studies in the model ascomycete A. nidulans. However, the number of gene products that have been demonstrated as important for conidial germination is rapidly growing, and a plethora of functional categorisations have been implicated, from calcineurin-mediated signalling (da Silva Ferreira et al. 2007; Steinbach et al. 2006) to MAPK signalling (Liebmann et al. 2004; Xue et al. 2004; Zhao et al. 2006) and glucose metabolism (Fleck and Brock 2010). An integrated and hierarchical understanding of the process will gradually emerge once functional genomics approaches reveal the entire cohort of gene products involved.
Fortwendel et al. (2006) recently established the mechanism by which a membrane-associated Ras GTPase switch, RasA, governs morphological transitions in A. fumigatus. Ras-GTPases regulate a multiplicity of cellular processes in a highly precise spatial and temporal manner, commanding control over differential pathway activation, which is governed in part by post-translational modification and consequent modulation of subcellular localisation. Deletion of RasA in A. fumigatus causes delayed germination and subsequent formation of wide, blunted hyphae that continually switch polarised growth axes.
A functional RasA-GFP translational fusion was used to interrogate the correlation between RasA localisation and morphogenesis. RasA was found to localise at the cell periphery of emerging germ tubes and the outer edge of hyphae, consistent with a plasma membrane association of the protein during polarised hyphal growth. Incubation of fungal cultures with a nonreversible inhibitor of protein palmitoylation inhibited hyphal growth in a dose-dependent manner and prompted aberrant localisation of GFP-RasA to internal foci. Taken together, these observations suggested that palmitoylation might be important for proper RasA localisation and polarised hyphal growth. The targeted double mutation (RasAC206/207S) of a conserved RasA palmitoylation motif prevented full complementation of the RasA hyphal growth blockade and resulted in a dramatic reduction of GFP signal at the plasma membrane. GFP-RasAC206/207S was mislocalised from the hyphal periphery and septa to the cytosol and internal patches, presumably to compartments of the endomembrane system. Both a RasA null mutant and the GFP-RasAC206/207S mutant displayed low germination rates and formed stunted, highly branched germlings during early phases of hyphal growth and displayed heightened sensitivity to cell wall damaging agents and aberrant deposition of β-glucan. To determine the contribution of plasma membrane localisation of RasA to A. fumigatus virulence, the ΔrasA and GFP-RasAC206/207S mutants were compared to the wild-type strain in a murine model of invasive aspergillosis. Virulence of both the ΔrasA and GFP-RasAC206/207S mutants was significantly attenuated (P < 0.0001), with infected mice displaying 25% and 20% mortality at 15 days postinoculation, respectively. Appropriate RasA localisation to the plasma membrane is therefore essential for full virulence in A. fumigatus.
Several groups have applied functional genomics techniques to the study of the conidial to hyphal transition in A. fumigatus. Lamarre and colleagues (2008) adopted a transcriptomic approach to analyse the exit of A. fumigatus spores from dormancy, identifying a shift from fermentative to respiratory metabolism and immediate protein synthesis upon breaking dormancy. In the first 30 minutes of incubation in YPD medium at 37°C, modulation of expression of 787 genes (of a possible 3,000) was observed in freshly harvested conidia. Downregulated genes encoded functions involved in fermentative metabolism and oxidoreductase activity whereas upregulated genes indicated a heightened emphasis upon RNA and phosphorus metabolism, amino acid and protein biosynthesis and protein complex assembly. Concordant with the predicted timing of mitosis in germinating spores, the authors found no evidence of upregulation of DNA processing/replication or cell cycle regulation. Additionally, expression of multiple genes involved in amino acid biosynthesis were upregulated during the first 30 minutes of the study, perhaps reflecting the rapid depletion of a pre-existing pool of free amino acids. This trend was supported by observed upregulation of genes in other pathways leading to amino acid biosynthesis, including glutamate dehydrogenase and glutamate synthase, NADPH isocitrate and succinate dehydrogenase. Downregulation of 28 mitochondrial genes, along with genes encoding alcohol dehydrogenases, lactate dehydrogenase, pyruvate decarboxylase and phosphoenol pyruvate synthase suggested a shift from a fermentative metabolism when the conidium is in a dormant stage to a respirative metabolism as soon as the germination process has started. Combining microarray and quantitative proteomic data, Cagas et al. (2011) were also able to identify a trend towards protein biosynthesis in time-series analyses of germinating conidia, during 0–16 hours of growth. A further quantitative proteomic analysis performed by Suh et al. (2012) identified an abundance of small, lineage-specific proteins amongst the conidial proteome. Teutschbein and colleagues (2010) applied two-dimensional polyacrylamide gel electrophoresis to establish a reference map of conidial proteins, identifying 449 different proteins, 57 of which were more abundant in conidia relative to mycelia. These included enzymes involved in detoxification of reactive oxygen intermediates, pigment biosynthesis and conidial rodlet layer formation. In agreement with the transcriptional analyses of Lamarre et al. (2008), pyruvate decarboxylase and alcohol dehydrogenase were detectable in dormant conidia, further supporting the notion that alcoholic fermentation plays a role during dormancy or early germination.
These studies set the scene for analysing differences between germination undergone in vitro and germination occurring during mammalian infection. Although the technologies to analyse the infecting fungal proteome thus far elude us, the transcriptional data from murine infections are already under analysis as part of our research programme.
B. Thermotolerance
The ability to thrive at 37°C is characteristic of all human pathogens and distinguishes A. fumigatus from most other environmental moulds. Relative to other Aspergillus spp., A. fumigatus has a natural propensity for thermotolerance, and in being capable of growth at temperatures that approach the upper limit for all eukaryotes, approximates an extremophilic mode of growth in the human host (Bhabhra and Askew 2005; Bhabhra et al. 2004). An intuitive hypothesis is that mutants that fail to grow at 37°C will be attenuated for virulence. This holds true in at least once case as A. fumigatus mutants lacking the cgrA gene product are attenuated for virulence. CgrA is required for the synthesis of ribosomes during conidial germination and has a distinct nucleolar localisation in A. fumigatus.
Mutants deficient in trehalose biosynthesis also demonstrate thermosensitive phenotypes in A. fumigatus (Al-Bader et al. 2010). Contingent with a role in stress tolerance, the trehalose content of A. fumigatus hyphae becomes elevated in response to heat shock and two genes, tpsA and tpsB, whose expression can be correlated with heightened trehalose content, serve partially redundant roles in trehalose accumulation during development and heat shock. A ΔtpsAB double mutant is devoid of trehalose and exhibits delayed (by 2–3 h) germination at 37°C and is nonviable at 50°C. However, virulence of the double mutant was unabated in a murine model of aspergillosis. Trehalose is regarded as an important source of energy during fungal development and acts to heighten stress tolerance by preventing the aggregation of denatured proteins. Being absent from mammalian cells it is of obvious therapeutic interest. It is intriguing that, despite the prevailing belief that rapidity of spore germination at 37°C provides a competitive advantage for A. fumigatus in the face of the host environment (Araujo and Rodrigues 2004), a mutant such as the ΔtpsAB double mutant does not suffer any deficit in the diseased mammalian host. A plausible explanation put forward for this involves aberrancy of cell wall biosynthesis in the double mutant, which might exacerbate the immune response sufficiently to worsen disease outcome.
C. Adhesion
Adhesion is postulated, but not yet formally proven, to be crucial for A. fumigatus virulence; however, significant correlations between the capacity to adhere to host proteins in vitro and the ability to cause mammalian disease have been documented (Sheppard 2011). The polysaccharide galactosaminogalactan (GAG), discovered in both cell wall extracts and culture media (Fontaine et al. 2000), is a polymer of galactopyranose linked to N-acetylgalactosamine. GAG is implicated as an important component of the A. fumigatus extracellular matrix, as evidenced by analyses of human biopsy material (Muller et al. 2011). In independent studies seeking genes that govern biofilm formation and adherence Gravelat et al. ( 2010 ) identified a regulatory gene, medA , that acts to modulate GAG synthesis. Transcriptomic analyses identified the genetic locus that directs GAG biosynthesis as a cluster of genes on chromosome 3. MedA was found to mediate biofilm formation and adherence to several substrates including fibronectin and pulmonary epithelial cells, and a ΔmedA mutant was attenuated for virulence in a murine model of disease. GAG-deficient strains also exhibited a reduction in their ability to injure and stimulate pulmonary epithelial cells in vitro.
D. Host-Mediated Fungal Clearance and Immune Evasion by Aspergillus fumigatus
In the immune-competent host, the respiratory epithelium and alveolar macrophages present the first host interface encountered by the fungal pathogen (Fig. 2.4). Alveolar macrophages are potent inactivators of A. fumigatus spores in vitro and, following spore recognition and internalisation, they efficiently kill ungerminated spores.
In human and murine pulmonary macrophages, acidification of A. fumigatus phagolysosomes and phagocyte oxidase-derived reactive oxygen intermediates (ROIs) are requisite for killing of A. fumigatus spores (Ibrahim-Granet et al. 2003; Philippe et al. 2003). Obligatory morphological transitions undertaken by the invading pathogen ensure that invasive, but not dormant, fungal elements act as immunostimulants for macrophages, a phenomenon that is governed by the stage-specific cell surface exposure of cell wall β-glucans (Fig. 2.4).
Macrophages secrete tumour necrosis factor-α (TNF-α) and macrophage inflammatory protein-2 (MIP-2) in response to metabolically active, but not heat-killed spores (Hohl et al. 2005). Swollen spores, live or dead, recruit macrophages and neutrophils to the murine lung following intratracheal infection (Hohl et al. 2005).
Unlike macrophages, neutrophils kill both conidia and hyphae, the latter probably being simply too large to be phagocytosed prior to killing. Human (McCormick et al. 2010) and murine (Bonnett et al. 2006) neutrophils inhibit germination of A. fumigatus conidia, and delayed polymorphonuclear neutrophil (PMN) recruitment leads to conidial germination in murine lungs (Bonnett et al. 2006). Phagocytic uptake of conidia by human neutrophils is crucial for the observed inhibition of germination (McCormick et al. 2010). Neutrophil extracellular traps (NETs) limit hyphal growth in vitro but have minimal, if any, direct capacity for killing hyphae (McCormick et al. 2010); however, the soluble pattern recognition receptor (PRR) PTX3, which is nonredundant in whole animals for defence against A. fumigatus infection, is localised in specific PMN granules and is secreted in response to exposure to A. fumigatus spores (Jaillon et al. 2007). Neutrophils release ROIs onto A. fumigatus hyphae in a NADPH oxidase-dependent manner, requiring the class IA phosphatidylinositol 3-kinases PI3Kβ and PI3Kδ, which have also been found to regulate the spreading of neutrophils over the hyphal surface. β2-Integrins play a major role in the activation of the NADPH oxidase in response to hyphae, whereas dectin-1 has only a minor, redundant role. Neutrophil recruitment in immunocompetent mice requires CXCR2, neutralisation of which leads to marked impairment of neutrophil influx and severe IA with nearly 100% mortality. Similarly, CXCR2-deficient animals challenged with intrapulmonary conidia suffer impaired recruitment of neutrophils to the lungs, permitting conidial germination. Pulmonary dendritic cells phagocytose conidia and hyphae via distinct receptors and orchestrate differential cytokine responses to each fungal morphology to instruct local and peripheral Th1-type inflammatory responses (Bozza et al. 2002, 2003). Release of cytokines by fungus-stimulated dendritic cell (DC) subsets recruits and activates other immune cells, thereby boosting innate responses and helping to initiate adaptive immunity. Pulmonary DCs transport Aspergillus conidia or hyphae to the draining lymph nodes and spleens and mount differential responses to the two distinct morphological forms of the fungus. Whereas TNF-α is produced in response to either form, IL-12p70 is produced upon exposure to conidia, but not to hyphae, and IL-4 and IL-10 production is prompted by phagocytosis of hyphae, but not conidia.
Plasmacytoid DCs (pDCs), comprising 0.2–0.8% of the total peripheral blood mononuclear cells (PBMCs) link innate to adaptive immunity by secreting cytokines such as IFN-α and TNF-α and by differentiating into mature pDCs with upregulated major histocompatibility complexes (MHC) and co-stimulatory molecules capable of priming naive T cells. Pulmonary DCs are required for effective antifungal defences in vivo, as mice depleted of pDCs are hypersusceptible to invasive aspergillosis (Ramirez-Ortiz et al. 2011).
Natural killer (NK) cells are directly cytotoxic to A. fumigatus spores and hyphae and germinated A. fumigatus morphologies are highly immunogenic, able to induce a Th1-like response and capable of upregulating cytokines such as IFN-γ and TNF-α. NK cells do not mediate anti-Aspergillus cytotoxicity through degranulation of their cytotoxic proteins (perforin, granzymes, granulysine), but via IFN-γ, which directly damages A. fumigatus, attributing new properties to both human NK cells and IFN-γ and suggesting them as possible therapeutic tools against IA.
In addition to the RodA hydrophobin, which prevents inhaled fungal spores from immediately initiating inflammatory responses in the host (Aimanianda et al. 2009), a further layer of immunomodulatory control, documented simultaneously by Hohl et al. (2005) and Steele et al. (2005), was revealed. This phenomenon is manifested by the gradual exposure of cell surface β-glucan during swelling and germination of A. fumigatus spores (Fig. 2.4). In contrast to dormant spores, germinating conidia induce neutrophil recruitment to the airways and TNF-α/MIP-2 secretion by alveolar macrophages, with fungal β-glucans acting as the inflammatory trigger via their stage-specific exposure on the surface of germinating conidia. Dectin-1, the innate immune receptor that mediates this immune response is recruited in vivo to alveolar macrophage phagosomes that have internalised conidia with exposed β-glucans.
E. Interactions of Spores with Epithelial Cells: Reservoirs for Fungal Persistence?
Airway epithelial cells (ECs) are central participants in innate and adaptive immune responses as well as in mucosal inflammation and allergy. At present, the contribution of epithelial cell activities to defence against A. fumigatus is unknown, but this is a highly active area of current research (Osherov 2012) and present theories implicate both immunomodulatory and directly antimicrobial roles of epithelial cells in anti-Aspergillus defence (Osherov 2012). Additionally, not conversely. The ability of ECs to internalise and kill A. fumigatus spores has prompted the suggestion that epithelial cells might provide a reservoir in which dormant A. fumigatus spores escape clearance by innate immune cells.
Using electron microscopy, Paris et al. (1997) were the first to show that epithelial cells (of rabbit tracheal, rat alveolar and human umbilical cord origins) internalise A. fumigatus conidia. These observations were replicated in several subsequent studies (Wasylnka and Moore 2002, 2003). Germinating, but not resting, conidia were found to induce production of interleukin (IL)-8 (Balloy et al. 2008) in human bronchial epithelial cells (HBECs). IL-8 production was found to be governed by phosphatidylinositol 3-kinase, p38 MAPK and ERK1/2 but was independent of the TLR-MyD88 pathway. However, integrity of the MyD88 pathway was found to be required for A. fumigatus-mediated NF-κB activation, suggesting that two independent signalling pathways become activated in respiratory epithelial cells by A. fumigatus.
Resting conidia are also recognised by, and stimulate, differentiated HBECs in an internalisation-dependent manner, resulting in the activation of the interferon (IFN)-β signalling pathway (Beisswenger et al. 2012). Conida-derived double-stranded RNA was found to be responsible for IFN-β and IP-10 expression when it was transfected into HBECs, but not when RNA was added directly to the culture medium. This supports the theory that internalisation and degradation of A. fumigatus spores leads to RNA release and IFN-β signalling. The observation that these responses to A. fumigatus challenge require internalisation of spores was substantiated by inhibition of endocytosis using cytochalasin D, which prompted the dose-dependent reduction of IFN-β and IP-10 expression.
In the epithelial cell line A549, and primary respiratory epithelial cells, internalised conidia are directed into the endosomal–lysosomal compartment where they are acidified and some are killed (Wasylnka and Moore 2003). Internalisation of conidia by type II pneumocytes such as A549 cells may be important in the development of aspergillosis in vivo because sequestration by these cells could allow conidia to evade the immune response of the host.
F. Melanin
A. fumigatus produces at least two types of melanin, pyomelanin and dihydroxynaphthalene (DHN)-melanin (Heinekamp et al. 2012), the latter being a crucial contributor to fungal resilience in the host environment. Detailed molecular genetic studies, (reviewed by Heinekamp et al. 2012), employing a variety of mutants in DHN biosynthesis have documented the contribution of DHN-melanin to intracellular trafficking of A. fumigatus in phagocytic cells and also to mammalian virulence.
The phagocytosis and intracellular degradation of conidia by phagocytic cells contributes to fungal and proinflammatory responses, thus making a dual contribution to essential host defences. Following phagocytosis, the intracellular fusion of conidia-containing phagosomes with lysosomes is a crucial step in microbial killing because the resulting phagolysosome provides an acidic environment conducive to antimicrobial activities, including degrading enzymes. Conidia of the A. fumigatus pksP mutant, which lack DHN-melanin, produce smooth white conidia and have a much higher propensity than wild-type spores to localise in phagolysosomes (Thywissen et al. 2011). DHN-melanin probably provides a further morphotype-specific example of the host-modulating potential of A. fumigatus particles because resting or fixed conidia showed stronger inhibition of phagolysosome acidification than swollen conidia.
V. Permanence
A. Fungal Stress Tolerance and Nutrient Acquisition
To identify fungal attributes preferentially employed during adaptation to the host niche, McDonagh et al. (2008) compared the transcriptomes of developmentally matched A. fumigatus isolates following laboratory culture or initiation of infection in the neutropenic murine lung. This analysis unveiled a remarkable level of co-ordinated gene expression during mammalian pathogenesis, including hallmarks of growth in alkaline, iron-limited and nutrient-limiting environments. A minimum of 11 siderophore biosynthesis/transport genes were identified as having heightened relevance during growth in the murine lung, as well as 13 amino acid permease genes and the general amino acid permease, Gap1. The functional categories of ergosterol biosynthesis, heme biosynthesis and aerobic respiration were significant among genes underrepresented during infection, relative to laboratory culture, as well as multiple functional categories representing ribosome biogenesis and assembly, and protein biosynthesis and processing. This may reflect the poor nutritional value of murine lung relative to YPD medium. A further intriguing aspect of the host adapting transcriptome was the co-ordinated expression of multiple groups of physically clustered genes, including genes known to direct biosynthesis of siderophores, and two known secondary metabolites, pseurotin and gliotoxin. These insights have been borne out by a plethora of subsequent studies addressing the role of stress tolerance and nutrient acquisition, a selection of which are discussed below.
B. Iron
Iron is an essential nutrient, the acquisition of which is exquisitely regulated in A. fumigatus to guard against the toxic effects of an excess of this element [recently reviewed by Haas (2012)]. This requires the co-ordinated activities of two transcription factors, SreA and HapX. When iron is in sufficient supply, SreA represses iron uptake to exclude toxic effects. When iron is limiting for growth, HapX acts to repress iron-consuming pathways such as heme biosynthesis. With respect to mammalian virulence, iron acquisition is a greater concern for the infecting fungus because deficiency in HapX, but not in SreA, attenuates virulence of A. fumigatus in a murine model of aspergillosis (Schrettl et al. 2008, 2010). In a series of eloquent molecular genetic analyses, Haas and colleagues have, to date, identified and characterised 24 genes involved in iron homeostasis in A. fumigatus and/or A. nidulans. In 10 out of 19 described A. fumigatus genes, knockout mutants proved to be attenuated for murine virulence. When it comes to iron acquisition, A. fumigatus differs somewhat from A. nidulans in possessing an additional capacity for high-affinity iron uptake by reductive iron assimilation (RIA); however, both organisms produce low molecular weight chaperones called siderophores to assist with high-affinity iron uptake. The first committed step in the biosynthesis of siderophores, and in A. fumigatus for RIA, is the hydroxylation of ornithine catalysed by the ornithine monooxygenase SidA. Subsequently, the pathways for biosynthesis of extra and intracellular siderophores split but, crucially, in the absence of sidA both RIA and siderophore biosynthesis are abolished. This has catastrophic consequences for A. fumigatus survival in the host, and a SidA null mutant is completely attenuated for virulence in neutropenic mice (Schrettl et al. 2004).
C. Hypoxia
Hypoxia ranks high amongst abiotic stimuli and varies greatly between the natural and other environments colonised by A. fumigatus. Variations in ambient oxygen concentrations from 21% (atmospheric) through 14% (alveolar) and 4% (human tissues) to 1.5% (compost) are predicted as being well within the repertoire of manageable stresses imposed upon the activities of this ubiquitous organism. In A. fumigatus , the sterol-regulatory element binding protein (SREBP) transcription factor governs growth in hypoxic conditions and mammalian virulence (Willger et al. 2008) but is not required for oxidative stress resistance and resistance to macrophage killing. Seeking to verify the direct involvement of hypoxic microenvironments upon observed attenuation of SREB functionality, Grahl et al. (2011) used 1H-NMR metabolomics to forage for ethanol production in the lungs of mice infected with A. fumigatus, thus confirming, via a surrogate marker of fungal anaerobic respiration, that oxygen-depleted microenvironments occur during murine infection. The additional use of a chemical hypoxia reporter, pimonidazole hydrochloride, confirmed via histopathological investigations that hypoxia could be observed during A. fumigatus infection of neutropenic, X-CGD and corticosteroid-treated mice. Relative to mice infected with a wild-type isolate, animals infected with an alcohol dehydrogenase-deficient mutant suffered heightened inflammation and a reduction in fungal burden. In conclusion, therefore, environmental conditions encountered by A. fumigatus in the mammalian host probably alter fungal metabolism in a manner conducive to effecting local modulation of the host immune response.
D. Invasive Growth and Tissue Damage
It has long been assumed that fungal proteases support the degradation of host lung tissue during invasive aspergillosis. However, due to the assumed redundancy of multiple such functions in A. fumigatus, no mutant lacking a single protease-encoding gene has ever demonstrated attenuation of virulence in murine aspergillosis. The assimilation of proteinaceous substrates is an essential facet of the fungal lifestyle and multiple studies here, and elsewhere (Lamarre et al. 2008; Latge 1999; Abad et al. 2010; Dagenais and Keller 2009; Krappmann et al. 2004; Oliver et al. 2012; McDonagh et al. 2008) documented have identified a clear requirement for amino acid acquisition/biosynthesis during germinative growth in vitro and during murine virulence. Two research groups (Sharon et al. 2009; Krappmann et al. 2004) simultaneously assessed the importance of extracellular proteases during in vitro growth and virulence by assessing the impact of PrtT (a transcriptional regulator that acts to modulate the expression of secreted proteases) on utilisation of alternative nitrogen sources and in murine models of invasive aspergillosis. The ΔprtT strain proved unable to grow on bovine serum albumin as the sole nitrogen source, and in a number of other assays for proteinaceous activity the mutant fared less well than the wild-type progenitor. Concordant with the phenotypic assays, transcriptomic analyses identified that the mutant was defective in expressing six extracellular proteases; however, both groups, using three independent approaches to virulence analysis, concluded that the mutant is fully virulent. Although it is tempting to conclude that the ability to degrade extracellular protein is dispensable for virulence, Sharon et al. (2009) duly noted that still other secreted proteases might be crucial during A. fumigatus lung infection. Indeed, of the proteases upregulated in the transcriptome study of McDonagh et al. (2008), an abundance of transcripts for the secreted proteases MEP, Dpp4, Dpp5, AFUA_6G10250 and AFUA_3G07850 was uncovered. Of these, only MEP and Dpp4 exhibited reduced transcription in the ΔprtT mutant.
Study of PrtT gathered further relevance when the effects of the gene deletion on infection of epithelial cells was investigated. Previous studies had shown that A. fumigatus proteases disrupt A549 cell actin fibres and focal adhesions, leading to cell detachment and death. In a further study, culture filtrates derived from the ΔprtT mutant were shown to cause significantly less A549 cell detachment and cell death (Sharon et al. 2011). Comparing the A549 transcriptional and cell signalling responses activated in the presence of A. fumigatus wild-type and ΔprtT conidia and culture filtrates, differential phosphorylation, and therefore activation, of JNK and ERK1/2 was observed in response to challenge with the two isolates. Inhibition of JNK or ERK1/2 kinase activity substantially decreased CF-induced cell damage, including cell peeling, actin-cytoskeleton damage, reduced metabolic activity and necrotic death. Because deletion of PrtT results primarily in the loss of secreted protease activity, it is likely that protease activity may be responsible for the subsequent MAPK activation in the treated A549 cells. Inhibition of this effect might provide a route to protecting the host against fungus-mediated damage and resultant inflammation.
E. Mycotoxins
Like other filamentous fungi, A. fumigatus produces multiple small molecule compounds biosynthesised by secondary metabolism. The demonstration that a wide domain regulator of secondary metabolism gene expression, LaeA, is required for fungal virulence affirms the potential importance of secondary metabolite “cocktails” produced in the lung of the infected host. Furthermore, a LaeA regulatory signature, characterised by transcriptomic analysis of a LaeA null mutant (Perrin et al. 2007) was found to be significantly similar to that derived from analyses of fungal infection, performed by McDonagh et al. (2008), and indicates that the selective expression of a subset of secondary metabolite loci might facilitate initiation of mammalian infection. In the post-genomic era, interest in secondary metabolism has become refuelled as bioinformatic scrutiny of the genome sequence has revealed interspecies variation in secondary metabolism genes, offering a plausible explanation for differential virulence amongst Aspergillus spp. (Khaldi et al. 2010; Nierman et al. 2005; Machida et al. 2005; Galagan et al. 2005).
At the time of its discovery, the DHN-melanin gene cluster was the largest cluster of fungal biosynthetic genes to be reported. Gene deletion mutants lacking any of six open reading frames, including those encoding a polyketide synthase, scytalone dehydratase and HN reductase, lacked the typical blue-green spore pigmentation characteristic of A. fumigatus and regulation of gene expression was observed to be developmentally linked to sporulation (Tsai et al. 1999). As discussed earlier in this chapter, conidial pigment biosynthesis in A. fumigatus appears to be an important virulence factor in the establishment of infection.
The right subtelomere of A. fumigatus chromosome 3 contains two predicted (Perrin et al. 2007) secondary metabolism gene clusters, Afu3g15200-Afu3g15340 and Afu3g14560-Afu3g14760, whose biosynthetic products are unknown. The former of the two gene clusters contains a polyketide synthase encoding the gene designated pes3, which has recently been shown to affect murine virulence and insect virulence (O’Hanlon et al. 2011). A Pes3 null mutant was found to be increased for fungal burden in corticosteroid-treated mice at 5 days post-infection, a phenotype that was accompanied by a more rapid germination of spores within murine lung tissues and a reduction in proinflammatory cytokine release, relative to wild type, from macrophages exposed to Δpes3 mutants in vitro. Subtelomeres 4 (left and right arms), 5 (left) and 7 (left) collectively accommodate five predicted (Perrin et al. 2007) secondary metabolism gene clusters, the biosynthetic products of which are currently unknown.
Subtelomere 8 (left) is exceptional because it encodes genes for predominantly secondary metabolism (n = 61), including multiple polyketide synthase-encoding genes (Afu8g00370 and Afu8g00890), a hybrid polyketide synthase/NRPS-encoding gene (Afu8g00540) and a further NRPS (Afu8g00170). Sheppard and colleagues noted the presence of genes in this region that are known to be required for sterigmatocystin/aflatoxin biosynthesis, as well as genes required for ergot alkaloid synthesis (Sheppard et al. 2005). Intriguingly, this combination of secondary metabolism genes is not found in other Aspergillus species. Previous gene deletion studies by Turner and colleagues have defined the biosynthetic products of some subclusters within this supercluster on chromosome 8, including the pseurotin A gene cluster (Maiya et al. 2007) and the fumitremorgin gene cluster (Maiya et al. 2006). Work in my laboratory is currently addressing the role of such metabolites in murine virulence. Indications to date are that loss of at least one of the metabolites whose biosynthesis is directed by genes resident in the chromosome 8 supercluster can impact virulence at whole animal level (Bignell, unpublished observations). However, our recent analyses indicate only a partial requirement for these metabolites during infection, as strain-dependent variability is observed when similar mutants are constructed in different genetic backgrounds. Thus, further characterisation is required before firm conclusions on the role of the metabolites encoded by genes in this region can be conclusively ascertained. A further crucial point is that the combinatorial activity of A. fumigatus secondary metabolites is a possibility thus far unexplored. It is highly feasible that certain secondary metabolites act in concert to disable residual host immune defences. The construction of mutants lacking multiple biosynthetic properties is therefore required to address this important question.
The chemical structures of a large number of metabolites have been well characterised, e.g. gliotoxin, which impacts virulence of A. fumigatus in corticosteroid, but not neutropenic mice (Sugui et al. 2007; Cramer et al. 2006; Bok et al. 2006; Kupfahl et al. 2006; Spikes et al. 2008) Thus far, gliotoxin, which has multiple toxic and inhibitory effects upon host cells is the only toxic secondary metabolite to have been isolated from the sera of rodents and patients suffering from invasive aspergillosis.
Until recently, few data on the identity of other toxic metabolites have been forthcoming and no attempt had been made to evaluate the toxicity of these metabolites on the respiratory tract. To address this, Gauthier and co-workers (Gauthier et al. 2012) extracted metabolites from the spores of A. fumigatus and evaluated their cytotoxic activity towards A549 epithelial cells using an MTT assay. The composition of each fraction regarding other secondary metabolites produced by Aspergillus fumigatus was determined by LC-MS analysis. The most toxic fractions were those containing all of the metabolite compounds. Sequential analyses of toxic fractions, by a deductive process, identified toxicity in several fractions that could be attributed to trypacidin and other fractions containing trypacidin precursor compounds. Of all fractions tested, viability of A549 cells exposed to spore extracts ranged from 92.5% (vehicle) to 0.2%. The use of cell lysis assays demonstrated that 50 μm trypacidin reduced cell viability by nearly 100% and triggered 85% cell lysis whereas the other metabolites showed only slight or no toxicity at this concentration. The question remains whether trypacidin could be toxic to epithelial and other cells in vivo.
F. Inflammation
Our advancing understanding of host–fungus interactions has revealed that fungal elements pose a potent antigenic stimulus (Romani 2011). For A. fumigatus, resting and germinating conidia and hyphae are recognised by host PRRs on epithelial cells and, in various cell types, induce the production of cytokines and chemokines such as IL-6, TNF-α, IL-8 and IFN-β. Importantly, amelioration of this inflammatory response can be achieved via corticosteroid administration, providing support for the importance of epithelial cells in directing host responses and resolution of infection. Macrophages activate at least two receptor-mediated signalling pathways in response to A. fumigatus. Proinflammatory cytokine production following challenge of peritoneal macrophages from TLR2 and TLR4 knockout mice revealed that TLR4 plays an important role in TNF-α, IL-1α and IL1-β production in response to conidia but that the stimulus for such production during spore-to-hyphal conversions is lost. TLR2, on the other hand, is required for responses of both conidia and hyphae (Gersuk et al. 2006; Netea et al. 2003).
Dectin-1, an innate immune receptor that recognises fungal β-glucans, associates with inhaled A. fumigatus conidia in a β-glucan-dependent manner (Hohl et al. 2005) and the production of TNF-α, IL-1α, IL-1β, IL-6, CXCL2/MIP-2, CCL3/ MIP-1α, granulocyte-colony stimulating factor (G-CSF), and granulocyte monocyte-CSF (GM-CSF), in response to live A. fumigatus is dectin-1 dependent (Steele et al. 2005). This complex inflammatory milieu requires precise regulation in order to ensure protection against host damage. Most likely, for the A. fumigatus host–pathogen interaction, deregulation of immune responses contribute as much to host damage as do pathogen-associated pathogenicity factors.
In seeking a unifying framework within which to study diseases caused by A. fumigatus, this working model can be applied to all types of disease caused by this mould. Allergic bronchopulmonary aspergillosis (ABPA) is a good case in point; in this disease host activities are probably more important than pathogen activities in driving host damage since allergic inflammation leads to infiltration of inflammatory cells and injury and detachment of the epithelium. ABPA occurs predominantly in asthmatic or cystic fibrosis (CF) patients and up to 15% of CF patients mount an allergic response, which is associated with exaggerated Th2 responses to the organism (Patterson and Strek 2010; Eaton et al. 2000). In a study assessing ABPA in CF patients, the binding and internalisation of A. fumigatus spores by epithelial cells was monitored in normal and mutated epithelial cells derived from both mice and cell lines (Chaudhary et al. 2012). Cell lines and murine tracheal epithelial cells (MTECS) that harbour defects in the cystic fibrosis transmembrane conductance regulator (CFTR) were found to bind, internalise and kill conidia less well than control cells. Histological analysis of murine infections (wild-type and CFTR null mice) clearly showed invasion of the lung parenchyma at 24 h in the absence of the CFTR channel. Proinflammatory mediators were measured after MTEC exposure to inactivated conidia and hyphal cells. Compared to wild-type MTEC, unstimulated CFTR null cells demonstrated an overall decreased production of MIP-2, IL-6 and IP-10. These analyses demonstrated a defective uptake of A. fumigatus conidia as well as aberrant inflammatory and apoptotic responses to different forms of A. fumigatus, thus contributing to altered pulmonary inflammation in the setting of CF, at least in part associated with ineffective epithelial cell clearance of conidia and subsequent aberrant inflammatory responses to germinated A. fumigatus morphotypes.
G. Perspectives
A unified model of the A. fumigatus host–pathogen interaction is required to understand the basis of diseases caused by this pathogen. The outcome of disease can be equally impacted by host and pathogen activities, and this must be accommodated into the design of novel therapeutic strategies. Although the administration of harsh antifungal agents is the mainstay of the current antifungal arsenal, it is likely that moderation of the host immune response might be equally effective and potentially less damaging to the host. A quantitative understanding of the host–pathogen interaction and the impact of antifungal drugs upon host homeostasis will be a necessary prerequisite to confidently apply the next generation of interventative strategies.
References
Abad A, Fernandez-Molina JV, Bikandi J, Ramirez A, Margareto J, Sendino J, Hernando FL, Ponton J, Garaizar J, Rementeria A (2010) What makes Aspergillus fumigatus a successful pathogen? Genes and molecules involved in invasive aspergillosis. Rev Iberoam Micol 27:155–182
Agarwal R (2009) Allergic bronchopulmonary aspergillosis. Chest 135:805–826
Aimanianda V, Bayry J, Bozza S, Kniemeyer O, Perruccio K, Elluru SR, Clavaud C, Paris S, Brakhage AA, Kaveri SV, Romani L, Latge JP (2009) Surface hydrophobin prevents immune recognition of airborne fungal spores. Nature 460:1117–1121
Al-Bader N, Vanier G, Liu H, Gravelat FN, Urb M, Hoareau CM, Campoli P, Chabot J, Filler SG, Sheppard DC (2010) Role of trehalose biosynthesis in Aspergillus fumigatus development, stress response, and virulence. Infect Immun 78:3007–3018
Araujo R, Rodrigues AG (2004) Variability of germinative potential among pathogenic species of Aspergillus. J Clin Microbiol 42:4335–4337
Askew DS (2008) Aspergillus fumigatus: virulence genes in a street-smart mold. Curr Opin Microbiol 11:331–337
Baker SE, Bennet JW (2008) An overview of the genus Aspergillus. In: Goldman GH, Osmani SA (eds) The Aspergilli: genomics, medical aspects and research methods. Taylor & Francis, Boca Raton, pp 3–13
Balloy V, Huerre M, Latge JP, Chignard M (2005) Differences in patterns of infection and inflammation for corticosteroid treatment and chemotherapy in experimental invasive pulmonary aspergillosis. Infect Immun 73:494–503
Balloy V, Sallenave JM, Wu Y, Touqui L, Latge JP, Si-Tahar M, Chignard M (2008) Aspergillus fumigatus-induced interleukin-8 synthesis by respiratory epithelial cells is controlled by the phosphatidylinositol 3-kinase, p38 MAPK, and ERK1/2 pathways and not by the toll-like receptor-MyD88 pathway. J Biol Chem 283:30513–30521
Beisswenger C, Hess C, Bals R (2012) Aspergillus fumigatus conidia induce interferon-beta signalling in respiratory epithelial cells. Eur Respir J 39:411–418
Bennet JW (2010) An overview of the genus Aspergillus. In: Masayuki M, Katsuya G (eds) Aspergillus: molecular biology and genomics. Caister Academic, Norfolk, pp 1–18
Bernard M, Latge JP (2001) Aspergillus fumigatus cell wall: composition and biosynthesis. Med Mycol 39(Suppl 1):9–17
Bhabhra R, Askew DS (2005) Thermotolerance and virulence of Aspergillus fumigatus: role of the fungal nucleolus. Med Mycol 43(Suppl 1):S87–S93
Bhabhra R, Miley MD, Mylonakis E, Boettner D, Fortwendel J, Panepinto JC, Postow M, Rhodes JC, Askew DS (2004) Disruption of the Aspergillus fumigatus gene encoding nucleolar protein CgrA impairs thermotolerant growth and reduces virulence. Infect Immun 72:4731–4740
Bok JW, Chung D, Balajee SA, Marr KA, Andes D, Nielsen KF, Frisvad JC, Kirby KA, Keller NP (2006) GliZ, a transcriptional regulator of gliotoxin biosynthesis, contributes to Aspergillus fumigatus virulence. Infect Immun 74:6761–6768
Bonnett CR, Cornish EJ, Harmsen AG, Burritt JB (2006) Early neutrophil recruitment and aggregation in the murine lung inhibit germination of Aspergillus fumigatus conidia. Infect Immun 74:6528–6539
Bouchara JP, Sanchez M, Chevailler A, Marot-Leblond A, Lissitzky JC, Tronchin G, Chabasse D (1997) Sialic acid-dependent recognition of laminin and fibrinogen by Aspergillus fumigatus conidia. Infect Immun 65:2717–2724
Bozza S, Gaziano R, Spreca A, Bacci A, Montagnoli C, Di FP, Romani L (2002) Dendritic cells transport conidia and hyphae of Aspergillus fumigatus from the airways to the draining lymph nodes and initiate disparate Th responses to the fungus. J Immunol 168:1362–1371
Bozza S, Perruccio K, Montagnoli C, Gaziano R, Bellocchio S, Burchielli E, Nkwanyuo G, Pitzurra L, Velardi A, Romani L (2003) A dendritic cell vaccine against invasive aspergillosis in allogeneic hematopoietic transplantation. Blood 102:3807–3814
Brown GD, Denning DW, Gow NA, Levitz SM, Netea MG, White TC (2012) Hidden killers: human fungal infections. Sci Transl Med 4:165rv13
Cagas SE, Jain MR, Li H, Perlin DS (2011) The proteomic signature of Aspergillus fumigatus during early development. Mol Cell Proteomics 10:M111
Chaudhary N, Datta K, Askin FB, Staab JF, Marr KA (2012) CFTR regulates epithelial cell response to Aspergillus and resultant pulmonary inflammation. Am J Respir Crit Care Med 185(3):301–310
Clemons KV, Stevens DA (2005) The contribution of animal models of aspergillosis to understanding pathogenesis, therapy and virulence. Med Mycol 43(Suppl 1):S101–S110
Cramer RA Jr, Gamcsik MP, Brooking RM, Najvar LK, Kirkpatrick WR, Patterson TF, Balibar CJ, Graybill JR, Perfect JR, Abraham SN, Steinbach WJ (2006) Disruption of a nonribosomal peptide synthetase in Aspergillus fumigatus eliminates gliotoxin production. Eukaryot Cell 5:972–980
da Silva Ferreira ME, Heinekamp T, Hartl A, Brakhage AA, Semighini CP, Harris SD, Savoldi M, de Gouvea PF, de Souza Goldman MH, Goldman GH (2007) Functional characterization of the Aspergillus fumigatus calcineurin. Fungal Genet Biol 44:219–230
Dagenais TR, Keller NP (2009) Pathogenesis of Aspergillus fumigatus in invasive aspergillosis. Clin Microbiol Rev 22:447–465
Dague E, Alsteens D, Latge JP, Dufrene YF (2008) High-resolution cell surface dynamics of germinating Aspergillus fumigatus conidia. Biophys J 94:656–660
Eaton T, Garrett J, Milne D, Frankel A, Wells AU (2000) Allergic bronchopulmonary aspergillosis in the asthma clinic. A prospective evaluation of CT in the diagnostic algorithm. Chest 118:66–72
Einsele H, Quabeck K, Muller KD, Hebart H, Rothenhofer I, Loffler J, Schaefer UW (1998) Prediction of invasive pulmonary aspergillosis from colonisation of lower respiratory tract before marrow transplantation. Lancet 352:1443
Fleck CB, Brock M (2010) Aspergillus fumigatus catalytic glucokinase and hexokinase: expression analysis and importance for germination, growth, and conidiation. Eukaryot Cell 9:1120–1135
Fontaine T, Simenel C, Dubreucq G, Adam O, Delepierre M, Lemoine J, Vorgias CE, Diaquin M, Latge JP (2000) Molecular organization of the alkali-insoluble fraction of Aspergillus fumigatus cell wall. J Biol Chem 275:41528
Fortwendel JR, Juvvadi PR, Rogg LE, Asfaw YG, Burns KA, Randell SH, Steinbach WJ (2006) Plasma membrane localization is required for RasA-mediated polarized morphogenesis and virulence of Aspergillus fumigatus. Eukaryot Cell 11(8):966–977. doi:10.1128/EC.00091-12 (Epub 4 May 2012)
Galagan JE, Calvo SE, Cuomo C, Ma LJ, Wortman JR, Batzoglou S, Lee SI, Basturkmen M, Spevak CC, Clutterbuck J, Kapitonov V, Jurka J, Scazzocchio C, Farman M, Butler J, Purcell S, Harris S, Braus GH, Draht O, Busch S, D’Enfert C, Bouchier C, Goldman GH, Bell-Pedersen D, Griffiths-Jones S, Doonan JH, Yu J, Vienken K, Pain A, Freitag M, Selker EU, Archer DB, Penalva MA, Oakley BR, Momany M, Tanaka T, Kumagai T, Asai K, Machida M, Nierman WC, Denning DW, Caddick M, Hynes M, Paoletti M, Fischer R, Miller B, Dyer P, Sachs MS, Osmani SA, Birren BW (2005) Sequencing of Aspergillus nidulans and comparative analysis with A. fumigatus and A. oryzae. Nature 438:1105–1115
Gauthier T, Wang X, Dos Sifuentes SJ, Fysikopoulos A, Tadrist S, Canlet C, Artigot MP, Loiseau N, Oswald IP, Puel O (2012) Trypacidin, a spore-borne toxin from Aspergillus fumigatus, is cytotoxic to lung cells. PLoS One 7:e29906
Gersuk GM, Underhill DM, Zhu L, Marr KA (2006) Dectin-1 and TLRs permit macrophages to distinguish between different Aspergillus fumigatus cellular states. J Immunol 176:3717–3724
Gil ML, Penalver MC, Lopez-Ribot JL, O’Connor JE, Martinez JP (1996) Binding of extracellular matrix proteins to Aspergillus fumigatus conidia. Infect Immun 64:5239–5247
Grahl N, Puttikamonkul S, Macdonald JM, Gamcsik MP, Ngo LY et al (2011) In vivo hypoxia and a fungal alcohol dehydrogenase influence the pathogenesis of invasive pulmonary aspergillosis. PLoS Pathog 7(7):e1002145
Gravelat FN, Ejzykowicz DE, Chiang LY, Chabot JC, Urb M, Macdonald KD, Al-Bader N, Filler SG, Sheppard DC (2010) Aspergillus fumigatus MedA governs adherence, host cell interactions and virulence. Cell Microbiol 12:473–488
Haas H (2012) Iron – a key nexus in the virulence of Aspergillus fumigatus. Front Microbiol 3:28
Heinekamp T, Thywissen A, Macheleidt J, Keller S, Valiante V, Brakhage AA (2012) Aspergillus fumigatus melanins: interference with the host endocytosis pathway and impact on virulence. Front Microbiol 3:440
Herbst S, Shah A, Carby M, Kikkeri N, Dorling A, Bignell E, Shaunak S, Armstrong-James D (2013) A new and clinically relevant model of solid-organ transplant aspergillosis. Dis Model Mech 6(3):643–651
Hohl TM, Feldmesser M (2007) Aspergillus fumigatus: principles of pathogenesis and host defense. Eukaryot Cell 6:1953–1963
Hohl TM, Van Epps HL, Rivera A, Morgan LA, Chen PL, Feldmesser M, Pamer EG (2005) Aspergillus fumigatus triggers inflammatory responses by stage-specific beta-glucan display. PLoS Pathog 1:e30
Hope WW, Walsh TJ, Denning DW (2005) The invasive and saprophytic syndromes due to Aspergillus spp. Med Mycol 43(Suppl 1):S207–S238
Ibrahim-Granet O, Philippe B, Boleti H, Boisvieux-Ulrich E, Grenet D, Stern M, Latge JP (2003) Phagocytosis and intracellular fate of Aspergillus fumigatus conidia in alveolar macrophages. Infect Immun 71:891–903
Jaillon S, Peri G, Delneste Y, Fremaux I, Doni A, Moalli F, Garlanda C, Romani L, Gascan H, Bellocchio S, Bozza S, Cassatella MA, Jeannin P, Mantovani A (2007) The humoral pattern recognition receptor PTX3 is stored in neutrophil granules and localizes in extracellular traps. J Exp Med 204:793–804
Kamai Y, Lossinsky AS, Liu H, Sheppard DC, Filler SG (2009) Polarized response of endothelial cells to invasion by Aspergillus fumigatus. Cell Microbiol 11:170–182
Kelm S, Schauer R (1997) Sialic acids in molecular and cellular interactions. Int Rev Cytol 175:137–240
Khaldi N, Seifuddin FT, Turner G, Haft D, Nierman WC, Wolfe KH, Fedorova ND (2010) SMURF: genomic mapping of fungal secondary metabolite clusters. Fungal Genet Biol 47:736–741
Krappmann S, Bignell EM, Reichard U, Rogers T, Haynes K, Braus GH (2004) The Aspergillus fumigatus transcriptional activator CpcA contributes significantly to the virulence of this fungal pathogen. Mol Microbiol 52:785–799
Kupfahl C, Heinekamp T, Geginat G, Ruppert T, Hartl A, Hof H, Brakhage AA (2006) Deletion of the gliP gene of Aspergillus fumigatus results in loss of gliotoxin production but has no effect on virulence of the fungus in a low-dose mouse infection model. Mol Microbiol 62:292–302
Lacey ME, West JS (2007) The aerobiology pathway. In: The air spora: a manual for catching and identifying airborne biological particles. Springer, Berlin, p 156
Lamarre C, Sokol S, Debeaupuis JP, Henry C, Lacroix C, Glaser P, Coppee JY, Francois JM, Latge JP (2008) Transcriptomic analysis of the exit from dormancy of Aspergillus fumigatus conidia. BMC Genomics 9:417
Lass-Florl C, Salzer GM, Schmid T, Rabl W, Ulmer H, Dierichi MP (1999) Pulmonary Aspergillus colonization in humans and its impact on management of critically ill patients. Br J Haematol 104:745–747
Latge JP (1999) Aspergillus fumigatus and aspergillosis. Clin Microbiol Rev 12:310–350
Leleu C, Menotti J, Meneceur P, Choukri F, Sulahian A, Garin Y-J, Denis J-B, Derouin F (2013) Bayesian development of a dose-response model for Aspergillus fumigatus and invasive aspergillosis. Risk Anal 33(4):1–13. doi:10.1111/risa.12007
Lewis RE, Wiederhold NP (2005) Murine model of invasive aspergillosis. Methods Mol Med 118:129–142
Liebmann B, Muller M, Braun A, Brakhage AA (2004) The cyclic AMP-dependent protein kinase a network regulates development and virulence in Aspergillus fumigatus. Infect Immun 72:5193–5203
Machida M, Asai K, Sano M, Tanaka T, Kumagai T, Terai G, Kusumoto K, Arima T, Akita O, Kashiwagi Y, Abe K, Gomi K, Horiuchi H, Kitamoto K, Kobayashi T, Takeuchi M, Denning DW, Galagan JE, Nierman WC, Yu J, Archer DB, Bennett JW, Bhatnagar D, Cleveland TE, Fedorova ND, Gotoh O, Horikawa H, Hosoyama A, Ichinomiya M, Igarashi R, Iwashita K, Juvvadi PR, Kato M, Kato Y, Kin T, Kokubun A, Maeda H, Maeyama N, Maruyama J, Nagasaki H, Nakajima T, Oda K, Okada K, Paulsen I, Sakamoto K, Sawano T, Takahashi M, Takase K, Terabayashi Y, Wortman JR, Yamada O, Yamagata Y, Anazawa H, Hata Y, Koide Y, Komori T, Koyama Y, Minetoki T, Suharnan S, Tanaka A, Isono K, Kuhara S, Ogasawara N, Kikuchi H (2005) Genome sequencing and analysis of Aspergillus oryzae. Nature 438:1157–1161
Maiya S, Grundmann A, Li SM, Turner G (2006) The fumitremorgin gene cluster of Aspergillus fumigatus: identification of a gene encoding brevianamide F synthetase. Chembiochem 7:1062–1069
Maiya S, Grundmann A, Li X, Li SM, Turner G (2007) Identification of a hybrid PKS/NRPS required for pseurotin A biosynthesis in the human pathogen Aspergillus fumigatus. Chembiochem 8:1736–1743
McCormick A, Heesemann L, Wagener J, Marcos V, Hartl D, Loeffler J, Heesemann J, Ebel F (2010) NETs formed by human neutrophils inhibit growth of the pathogenic mold Aspergillus fumigatus. Microbes Infect 12:928–936
McDonagh A, Fedorova ND, Crabtree J, Yu Y, Kim S, Chen D, Loss O, Cairns T, Goldman G, Armstrong-James D, Haynes K, Haas H, Schrettl M, May G, Nierman WC, Bignell E (2008) Sub-telomere directed gene expression during initiation of invasive aspergillosis. PLoS Pathog 4:e1000154
Momany M, Taylor I (2000) Landmarks in the early duplication cycles of Aspergillus fumigatus and Aspergillus nidulans: polarity, germ tube emergence and septation. Microbiology 146(Pt 12):3279–3284
Muller FM, Seidler M, Beauvais A (2011) Aspergillus fumigatus biofilms in the clinical setting. Med Mycol 49(Suppl 1):S96–S100
Netea MG, Warris A, Van der Meer JW, Fenton MJ, Verver-Janssen TJ, Jacobs LE, Andresen T, Verweij PE, Kullberg BJ (2003) Aspergillus fumigatus evades immune recognition during germination through loss of toll-like receptor-4-mediated signal transduction. J Infect Dis 188:320–326
Nierman WC, Pain A, Anderson MJ, Wortman JR, Kim HS, Arroyo J, Berriman M, Abe K, Archer DB, Bermejo C, Bennett J, Bowyer P, Chen D, Collins M, Coulsen R, Davies R, Dyer PS, Farman M, Fedorova N, Fedorova N, Feldblyum TV, Fischer R, Fosker N, Fraser A, Garcia JL, Garcia MJ, Goble A, Goldman GH, Gomi K, Griffith-Jones S, Gwilliam R, Haas B, Haas H, Harris D, Horiuchi H, Huang J, Humphray S, Jimenez J, Keller N, Khouri H, Kitamoto K, Kobayashi T, Konzack S, Kulkarni R, Kumagai T, Lafon A, Latge JP, Li W, Lord A, Lu C, Majoros WH, May GS, Miller BL, Mohamoud Y, Molina M, Monod M, Mouyna I, Mulligan S, Murphy L, O’Neil S, Paulsen I, Penalva MA, Pertea M, Price C, Pritchard BL, Quail MA, Rabbinowitsch E, Rawlins N, Rajandream MA, Reichard U, Renauld H, Robson GD, de Rodriguez CS, Rodriguez-Pena JM, Ronning CM, Rutter S, Salzberg SL, Sanchez M, Sanchez-Ferrero JC, Saunders D, Seeger K, Squares R, Squares S, Takeuchi M, Tekaia F, Turner G, Vazquez de Aldana CR, Weidman J, White O, Woodward J, Yu JH, Fraser C, Galagan JE, Asai K, Machida M, Hall N, Barrell B, Denning DW (2005) Genomic sequence of the pathogenic and allergenic filamentous fungus Aspergillus fumigatus. Nature 438:1151–1156
O’Gorman C (2011) Airborne Aspergillus fumigatus conidia: a risk factor for aspergillosis. Fungal Biol Rev 25(3):151–157
O’Gorman C, Fuller HT (2008) Prevalence of culturable airborne spores of selected allergenic and pathogenic fungi in outdoor air. Atmos Environ 42(18):4355–4368
O’Hanlon KA, Cairns T, Stack D, Schrettl M, Bignell EM, Kavanagh K, Miggin SM, O’Keeffe G, Larsen TO, Doyle S (2011) Targeted disruption of nonribosomal peptide synthetase pes3 augments the virulence of Aspergillus fumigatus. Infect Immun 79:3978–3992
Oliver JD, Kaye SJ, Tuckwell D, Johns AE, Macdonald DA, Livermore J, Warn PA, Birch M, Bromley MJ (2012) The Aspergillus fumigatus dihydroxyacid dehydratase Ilv3A/IlvC is required for full virulence. PLoS One 7:e43559
Osherov N (2012) Interaction of the pathogenic mold Aspergillus fumigatus with lung epithelial cells. Front Microbiol 3:346
Paris S, Boisvieux-Ulrich E, Crestani B, Houcine O, Taramelli D, Lombardi L, Latge JP (1997) Internalization of Aspergillus fumigatus conidia by epithelial and endothelial cells. Infect Immun 65:1510–1514
Patterson K, Strek ME (2010) Allergic bronchopulmonary aspergillosis. Proc Am Thorac Soc 7:237–244
Penalver MC, O’Connor JE, Martinez JP, Gil ML (1996) Binding of human fibronectin to Aspergillus fumigatus conidia. Infect Immun 64:1146–1153
Perrin RM, Fedorova ND, Bok JW, Cramer RA, Wortman JR, Kim HS, Nierman WC, Keller NP (2007) Transcriptional regulation of chemical diversity in Aspergillus fumigatus by LaeA. PLoS Pathog 3:e50
Philippe B, Ibrahim-Granet O, Prevost MC, Gougerot-Pocidalo MA, Sanchez PM, Van der Meeren A, Latge JP (2003) Killing of Aspergillus fumigatus by alveolar macrophages is mediated by reactive oxidant intermediates. Infect Immun 71:3034–3042
Pollock JD, Williams DA, Gifford MA, Li LL, Du X, Fisherman J, Orkin SH, Doerschuk CM, Dinauer MC (1995) Mouse model of X-linked chronic granulomatous disease, an inherited defect in phagocyte superoxide production. Nat Genet 9:202–209
Portugal RD, Garnica M, Nucci M (2009) Index to predict invasive mold infection in high-risk neutropenic patients based on the area over the neutrophil curve. J Clin Oncol 27:3849–3854
Ramirez-Ortiz ZG, Lee CK, Wang JP, Boon L, Specht CA, Levitz SM (2011) A nonredundant role for plasmacytoid dendritic cells in host defense against the human fungal pathogen Aspergillus fumigatus. Cell Host Microbe 9:415–424
Romani L (2011) Immunity to fungal infections. Nat Rev Immunol 11:275–288
Schrettl M, Bignell E, Kragl C, Joechl C, Rogers T, Arst HN Jr, Haynes K, Haas H (2004) Siderophore biosynthesis but not reductive iron assimilation is essential for Aspergillus fumigatus virulence. J Exp Med 200:1213–1219
Schrettl M, Kim HS, Eisendle M, Kragl C, Nierman WC, Heinekamp T, Werner ER, Jacobsen I, Illmer P, Yi H, Brakhage AA, Haas H (2008) SreA-mediated iron regulation in Aspergillus fumigatus. Mol Microbiol 70:27–43
Schrettl M, Beckmann N, Varga J, Heinekamp T, Jacobsen ID, Jochl C, Moussa TA, Wang S, Gsaller F, Blatzer M, Werner ER, Niermann WC, Brakhage AA, Haas H (2010) HapX-mediated adaption to iron starvation is crucial for virulence of Aspergillus fumigatus. PLoS Pathog 6:e1001124
Sharon H, Hagag S, Osherov N (2009) Transcription factor PrtT controls expression of multiple secreted proteases in the human pathogenic mold Aspergillus fumigatus. Infect Immun 77:4051–4060
Sharon H, Amar D, Levdansky E, Mircus G, Shadkchan Y, Shamir R, Osherov N (2011) PrtT-regulated proteins secreted by Aspergillus fumigatus activate MAPK signaling in exposed A549 lung cells leading to necrotic cell death. PLoS One 6:e17509
Sheppard DC (2011) Molecular mechanism of Aspergillus fumigatus adherence to host constituents. Curr Opin Microbiol 14:375–379
Sheppard DC, Doedt T, Chiang LY, Kim HS, Chen D, Nierman WC, Filler SG (2005) The Aspergillus fumigatus StuA protein governs the up-regulation of a discrete transcriptional program during the acquisition of developmental competence. Mol Biol Cell 16:5866–5879
Shibuya K, Takaoka M, Uchida K, Wakayama M, Yamaguchi H, Takahashi K, Paris S, Latge JP, Naoe S (1999) Histopathology of experimental invasive pulmonary aspergillosis in rats: pathological comparison of pulmonary lesions induced by specific virulent factor deficient mutants. Microb Pathog 27:123–131
Song E, Jaishankar GB, Saleh H, Jithpratuck W, Sahni R, Krishnaswamy G (2011) Chronic granulomatous disease: a review of the infectious and inflammatory complications. Clin Mol Allergy 9:10
Sorci G, Giovannini G, Riuzzi F, Bonifazi P, Zelante T, Zagarella S, Bistoni F, Donato R, Romani L (2011) The danger signal S100B integrates pathogen- and danger-sensing pathways to restrain inflammation. PLoS Pathog 7:e1001315
Spikes S, Xu R, Nguyen CK, Chamilos G, Kontoyiannis DP, Jacobson RH, Ejzykowicz DE, Chiang LY, Filler SG, May GS (2008) Gliotoxin production in Aspergillus fumigatus contributes to host-specific differences in virulence. J Infect Dis 197:479–486
Steele C, Rapaka RR, Metz A, Pop SM, Williams DL, Gordon S, Kolls JK, Brown GD (2005) The beta-glucan receptor dectin-1 recognizes specific morphologies of Aspergillus fumigatus. PLoS Pathog 1:e42
Steinbach WJ, Cramer RA Jr, Perfect BZ, Asfaw YG, Sauer TC, Najvar LK, Kirkpatrick WR, Patterson TF, Benjamin DK Jr, Heitman J, Perfect JR (2006) Calcineurin controls growth, morphology, and pathogenicity in Aspergillus fumigatus. Eukaryot Cell 5:1091–1103
Sugui JA, Pardo J, Chang YC, Zarember KA, Nardone G, Galvez EM, Mullbacher A, Gallin JI, Simon MM, Kwon-Chung KJ (2007) Gliotoxin is a virulence factor of Aspergillus fumigatus: gliP deletion attenuates virulence in mice immunosuppressed with hydrocortisone. Eukaryot Cell 6:1562–1569
Suh MJ, Fedorova ND, Cagas SE, Hastings S, Fleischmann RD, Peterson SN, Perlin DS, Nierman WC, Pieper R, Momany M (2012) Development stage-specific proteomic profiling uncovers small, lineage specific proteins most abundant in the Aspergillus fumigatus conidial proteome. Proteome Sci 10:30
Swan J, Gilbert E, Kelsey A, Crook B (2013) Occupational and environmental exposure to bioaerosols from composts and potential health effects - a critical review of published data. HSE Books, Sudbury
Tell LA (2005) Aspergillosis in mammals and birds: impact on veterinary medicine. Med Mycol 43(Suppl 1):S71–S73
Teutschbein J, Albrecht D, Potsch M, Guthke R, Aimanianda V, Clavaud C, Latge JP, Brakhage AA, Kniemeyer O (2010) Proteome profiling and functional classification of intracellular proteins from conidia of the human-pathogenic mold Aspergillus fumigatus. J Proteome Res 9:3427–3442
Thau N, Monod M, Crestani B, Rolland C, Tronchin G, Latge JP, Paris S (1994) Rodletless mutants of Aspergillus fumigatus. Infect Immun 62:4380–4388
Thywissen A, Heinekamp T, Dahse HM, Schmaler-Ripcke J, Nietzsche S, Zipfel PF, Brakhage AA (2011) Conidial dihydroxynaphthalene melanin of the human pathogenic fungus Aspergillus fumigatus interferes with the host endocytosis pathway. Front Microbiol 2:96
Torpy FR, Irga PJ, Brennan J, Burchett MD (2012) Do indoor plants contribute to the aeromycota in city buildings? Aerobiologia (in press) doi: 10.1007/s10453-012-9282-y
Tronchin G, Bouchara JP, Ferron M, Larcher G, Chabasse D (1995) Cell surface properties of Aspergillus fumigatus conidia: correlation between adherence, agglutination, and rearrangements of the cell wall. Can J Microbiol 41:714–721
Tronchin G, Esnault K, Renier G, Filmon R, Chabasse D, Bouchara JP (1997) Expression and identification of a laminin-binding protein in Aspergillus fumigatus conidia. Infect Immun 65:9–15
Tsai HF, Wheeler MH, Chang YC, Kwon-Chung KJ (1999) A developmentally regulated gene cluster involved in conidial pigment biosynthesis in Aspergillus fumigatus. J Bacteriol 181:6469–6477
Wasylnka JA, Moore MM (2000) Adhesion of Aspergillus species to extracellular matrix proteins: evidence for involvement of negatively charged carbohydrates on the conidial surface. Infect Immun 68:3377–3384
Wasylnka JA, Moore MM (2002) Uptake of Aspergillus fumigatus conidia by phagocytic and nonphagocytic cells in vitro: quantitation using strains expressing green fluorescent protein. Infect Immun 70:3156–3163
Wasylnka JA, Moore MM (2003) Aspergillus fumigatus conidia survive and germinate in acidic organelles of A549 epithelial cells. J Cell Sci 116:1579–1587
Wasylnka JA, Simmer MI, Moore MM (2001) Differences in sialic acid density in pathogenic and non-pathogenic Aspergillus species. Microbiology 147:869–877
Willger SD, Puttikamonkul S, Kim KH, Burritt JB, Grahl N, Metzler LJ, Barbuch R, Bard M, Lawrence CB, Cramer RA Jr (2008) A sterol-regulatory element binding protein is required for cell polarity, hypoxia adaptation, azole drug resistance, and virulence in Aspergillus fumigatus. PLoS Pathog 4:e1000200
Xue T, Nguyen CK, Romans A, May GS (2004) A mitogen-activated protein kinase that senses nitrogen regulates conidial germination and growth in Aspergillus fumigatus. Eukaryot Cell 3:557–560
Zhao W, Panepinto JC, Fortwendel JR, Fox L, Oliver BG, Askew DS, Rhodes JC (2006) Deletion of the regulatory subunit of protein kinase A in Aspergillus fumigatus alters morphology, sensitivity to oxidative damage, and virulence. Infect Immun 74:4865–4874
Zmeili OS, Soubani AO (2007) Pulmonary aspergillosis: a clinical update. QJM 100:317–334
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer-Verlag Berlin Heidelberg
About this chapter
Cite this chapter
Bignell, E. (2014). 2 Aspergillus fumigatus: Saprotroph to Pathogen. In: Kurzai, O. (eds) Human Fungal Pathogens. The Mycota, vol 12. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-642-39432-4_2
Download citation
DOI: https://doi.org/10.1007/978-3-642-39432-4_2
Published:
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-642-39431-7
Online ISBN: 978-3-642-39432-4
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)