Abstract
Temperature is one of the most important factors controlling the biogeography of seaweeds. To identify worldwide prospective distributional shifts of major biogeographical regions under a global change scenario, we applied a macroecological modeling approach. We compared the borders of biogeographical regions between present and end of the century sea surface temperatures (SST) taken from global climate model simulations and drew conclusions for distributional changes. All regions will extend towards the poles. As a consequence, the tropical region will widen considerably. However, there will be almost no change in the northern extent of the Antarctic region. According to the model data, the annual SST gradient will change along extensive coastlines creating broad transitional regions, some of which contain high seaweed genus diversity. As a consequence, the structure of the seaweed assemblages in these biogeographical regions will probably be reorganized.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Biogeographical Region
- Seaweed Species
- Western Antarctic Peninsula
- Kelp Species
- Eastern Equatorial Pacific
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
1 Introduction
Climate changes and concurrent changes in temperatures, atmospheric CO2 concentration, and other greenhouse gases have often occurred during the earth’s history (Zachos et al. 2008) and it is well known that biogeographical distribution patterns of species are directly controlled by climate (Pearson and Dawson 2003 and references therein). Considering only the last 740,000 years, we know of about eight glaciations associated with strong variations in temperature, precipitation, and thermohaline circulation worldwide (Augustin et al. 2004; Knutti et al. 2004). In the past century, overall global warming was around 0.8°C (Hansen et al. 2006). Changes are particularly strong in polar and cold-temperate regions of the northern hemisphere (Levitus et al. 2000; Hansen et al. 2006). At the western Antarctic Peninsula a recent rapid regional warming with a sea surface temperature (SST) increase of 3.7°C century−1 has been recorded (Vaughan et al. 2003). Massive retreat of icefronts and glaciers and an increase of melt water production has been documented since 1956 (Braun and Gossmann 2002). For the Arctic, a similar warming trend has been observed over recent decades. Monthly temperature anomalies can be as large as 3–4°C (Turner et al. 2007). In cold- and warm-temperate localities, the reported mean annual temperature increase was as high as >2°C depending on location and duration of time series within the last decades (e.g., California: Sagarin et al. 1999, Portugal: Lima et al. 2007, North Sea: Wiltshire et al. 2008, Baltic: Andersen et al. 2011, Australia: Poloczanska et al. 2011). Current warming is larger over landmasses than over the open ocean and is larger in higher latitudes than in the tropical region. In the Equatorial Tropical Atlantic and the Western Equatorial Pacific (WEP), the SST increase between 2001 and 2005 relative to 1870–1900 was 0.5–1°C, while the Eastern Equatorial Pacific (EEP) did not show any temperature increase (Hansen et al. 2006). Whether the increased temperature difference between the near-equatorial WEP and EEP may be responsible for dampening or enhancing the frequency of El Niño-Southern Oscillation (ENSO) activity is still in debate (e.g., Hansen et al. 2006; Collins et al. 2010). The two most pronounced ENSO events in the last 100 years took place in 1983 and 1998 accompanied by an unprecedented warming in the EEP (Hansen et al. 2006).
Within the last two decades, there have been many attempts to develop predictive modeling approaches to project present-day biogeographical distribution patterns into the future. Principally, two different directions have been followed: (1) the “bioclimate envelope” models correlate species distributions with climate variables including the knowledge about the physiological responses of species to climate change (Pearson and Dawson 2003 and references therein). A special form of these models is niche modeling which correlates the macroecological preferences of a species at sample locations (e.g., demands for temperature, substrate, light, etc.) with their distributional records. Niche models predict potentially suitable habitats and the fundamental biogeographical niche (Guisan and Thuiller 2005; Graham et al. 2007; Verbruggen et al. 2009). (2) On the other hand, marine ecologists stressed the importance to also consider the variety of biotic interactions between species which are mostly responsible for shaping the realized niche. A recent review extensively summarizes possible consequences of both the abiotic and biotic environment and their interactions in coastal marine environments with respect to climate change (Harley et al. 2006). Although biotic interactions locally shape communities, they do not explain global biogeographical patterns (Santelices et al. 2009). Recently, Müller et al. (2009, 2011) presented a new bioclimate envelope model comparing observed winter and summer SSTs of 1980–1999 to model SSTs of 2080–2099 based on a moderate greenhouse gas emission scenario in order to predict future distributional range shifts of selected polar and cold-temperate seaweed species of both hemispheres. Here, we use the same approach providing a macroecological view on seaweed distribution based on a worldwide model of present and future oceanic isotherms, carving out the resulting changes in spatial extent of major biogeographical coastal regions (after Briggs 1995) due to temperature change and discussing expected changes of seaweed floras on a worldwide scale for the end of the century.
2 Coastal Marine Biogeographical Regions and Their Relation to Oceanic Isotherms and Temperature Responses of Seaweeds
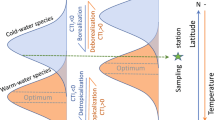
Especially temperature changes had and will have strong effects in the geographical distribution of seaweeds as temperature is the main abiotic factor directly controlling geographic boundaries of seaweed species (van den Hoek 1982a; Lüning 1990; Chap. 3 by Eggert) and also has recently been identified in shaping global marine biodiversity pattern (Tittensor et al. 2010). Principally, the presence of seaweeds in these regions is determined by the physiological tolerance of their life cycle stages to temperature. There are two fundamentally different types of temperature boundaries in seaweeds, lethal boundaries and growth or reproduction boundaries (Breeman 1988; van den Hoek and Breeman 1989). Lethal boundaries are determined by the capacity of the species to survive during the unfavorable season, e.g., a cold-water alga in the summer season. Growth and/or reproduction boundaries are determined by the ability of the species to grow and reproduce during the favorable season, e.g., a cold-water alga in the winter/spring season. Moreover, seaweed distribution is affected by temperature–daylength interactions (Dring 1984; Molenaar 1996) affecting the coordinated timing between life cycle stages.
Seven broad biogeographical regions have been recognized in coastal marine zoogeography and phytogeography (Briggs 1974, 1995; Lüning 1990): the tropical region of the Atlantic and Indo-Pacific, the cold- and warm-temperate regions of both hemispheres, and the two Polar regions (for details see Chap. 3 by Eggert). Although Spalding et al. (2007) recently proposed a new and more sophisticated global system of marine ecoregions than Briggs (1995), the major biogeographical boundaries along the continents are similar in both approaches. We therefore chose the Briggs (1995) system as it is better suited for demonstrating the broad global pattern of modeled future change. The boundaries between biogeographical regions after Briggs (1995) are determined by fundamental changes in the composition of coastal biota and have been defined by their degree of endemism (>10%; Spalding et al. 2007). The transition between regions may be characterized by SSTs as temperature is a major factor in shaping marine phytogeographical regions (e.g., van den Hoek 1982a, b; Lüning 1990). Lüning (1990) described the boundaries of phytogeographical regions by intermediate mean summer and winter isotherms to show the general broad pattern. The specific mean summer and winter isotherms prevailing at the eastern and western coasts of the Oceans and in the northern and southern hemisphere at the phytogeographic boundaries deviate, however, considerably from these averages and are presented in Table 18.1 based on isotherm data from 1980 to 1999 provided by Müller et al. (2009).
The current tropical coastal biogeographical regions are broad and characterized by summer isotherms between 24 and 29°C and winter isotherms between 18 and 23°C (Table 18.1, Figs. 18.1a and 18.2a). The adjacent warm-temperate regions are much smaller and generally slightly compressed on the western sides of the Pacific and Atlantic compared to the eastern sides of the oceans. The boundary between the warm- and cold-temperate regions is characterized by 16–29°C summer SSTs and by 10–20°C winter SSTs. These extreme differences are attributed to the pronounced compression of isotherms along the western part of the Atlantic and Pacific in contrast to the eastern part of the oceans (see Figs 12.2, 12.3 in Müller et al. 2011). When comparing both hemispheres pronounced differences become apparent as well. The southern boundary of the Arctic region, for example, is described by the 9–10°C summer isotherm, whereas the northern boundary of the Antarctic region is the 4°C summer isotherm (Table 18.1, Figs. 18.1a and 18.2b). The situation in the southern hemisphere might be partially attributed to missing continuous land masses in the circumpolar Southern Ocean. Thus, the limit between the polar and cold-temperate coastal regions in the southern hemisphere is not clearly defined. The largest differences between northern and southern hemispherical biogeographic regions become apparent at the boundary of the warm- and cold-temperate regions at the western coasts of both the Atlantic and the Pacific. In the N-Atlantic, this boundary is situated at the 27°C summer isotherm. In contrast, in the S-Atlantic it is located at the 19°C summer isotherm (Table 18.1, Figs. 18.1a and 18.2a). At the west coasts of the Pacific the differences are smaller (26°C August isotherm in the north to 20°C February isotherm in the south).
Changes in the extent of biogeographical regions after Briggs (1995) due to global warming at the end of the twentieth century inferred from current (1980–1999) and future (2080–2099) mean February sea surface isotherms. The mean February sea-surface isotherms delimiting current biogeographical regions are depicted in figure a. The future extension of the biogeographical regions (b) is based on the modeled change of the mean February sea surface isotherms at the present boundaries of the respective regions. Projected changes are indicated by bold coastal lines. Color code of biogeographical regions: turquoise: Polar regions, blue-green: cold-temperate regions, yellow: warm-temperate regions, red: tropical regions. Color code of isotherms: blue: winter isotherms, red: summer isotherms, gray: isotherms characterizing the central tropics
Changes in the extent of biogeographical regions after Briggs (1995) due to global warming at the end of the twentieth century inferred from current (1980–1999) and future (2080–2099) mean August sea surface isotherms. The mean August sea-surface isotherms delimiting current biogeographical regions are depicted in figure a. The future extension of the biogeographical regions (b) is based on the modeled change of the mean August sea surface isotherms at the present boundaries of the respective regions. Projected changes are indicated by bold coastal lines. Color code of biogeographical regions: turquoise: Polar regions, blue-green: cold-temperate regions, yellow: warm-temperate regions, red: tropical regions. Color code of isotherms: blue: winter isotherms, red: summer isotherms, gray: isotherms characterizing the central tropics
3 Responses of Seaweeds to Temperature Changes
3.1 General Responses
Seaweeds can principally respond to environmental changes in four ways which all may result in distributional and diversity changes: on short timescales, they can acclimate. On medium to long timescales they either adapt to new conditions or are able to slowly migrate keeping pace with the changing environmental pressure. Species unable to acclimate, adapt, or disperse may be captured in isolated refugia or become extinct. Acclimation to temperature stress in seaweeds has been mostly studied on the level of photosynthesis or growth (e.g., Davison et al. 1991; Kübler and Davison 1995; Eggert et al. 2006; see Chap. 3 by Eggert). Locally, the acclimation potential of thermal traits can shape the vertical or seasonal distribution pattern of species (Davison and Pearson 1996; Ateweberhan et al. 2005; Zardi et al. 2011) and on a broader scale eurythermal species have a broader distribution range than stenothermal species (see also Chap. 3 by Eggert). Hitherto, it is unknown whether species with a broad acclimation potential also had or will have a faster genetic adaptation potential—a fact which would help to explain historical or future biogeographical processes.
Adaptive processes on the physiological and molecular level are explained in detail in Chap. 3 by Eggert. On the organism level, the adaptation processes to temperature changes have been identified by comparing the temperature requirements of tropical and cold-water seaweeds from both hemispheres in relation to the climatic history of the various regions (Wiencke et al. 1994). Since Mesozoic times [251–65.5 million years before present (My)] there was a continuous warm water girdle around the earth so that cold-water seaweed species probably only evolved after the glaciation events in the Tertiary (65 My) (Lüning 1990). An upper survival temperature of 33–35°C may still be found in representatives of temperate and tropical seaweed species, indicating that this thermal trait is rather deeply entrenched and not subject to fast adaptation (Lüning 1990). While strictly tropical seaweed species are stenothermal and may survive 30–37°C and grow best between 25 and 30°C, they do not have the ability to live below temperatures of 7–12°C (Pakker et al. 1995; Bischoff-Bäsmann et al. 1997). The first steps in the adaptation of seaweeds to lower temperatures are an increase in cold tolerance and an increase of growth and reproduction rates at lower temperatures leading to eurythermal species. This temperature trait is apparent in tropical to warm-temperate species which acquired a better lower temperature tolerance with survival temperatures between 6 and ≤1°C without losing the upper temperature tolerance (Yarish et al. 1984; Pakker et al. 1995; Bischoff-Bäsmann et al. 1997). Later, the ability to survive temperatures ≥20°C and to grow and reproduce at ≥15–20°C was lost. This type of temperature response is typical for endemic Arctic and Arctic to cold-temperate distributed seaweeds exposed to low temperatures for about 3 My (Briggs 1995). The last steps in the adaptation to low temperatures are the loss of the ability to grow and reproduce at ≥5–10°C and to survive temperatures ≥6–13°C (Wiencke et al. 1994; Bischoff-Bäsmann and Wiencke 1996). This type of temperature response is exemplified in endemic Antarctic species exposed to cold water for at least 14 My (Crame 1993; Briggs 1995). So, the climatic history during species evolution determines the temperature requirements of seaweeds in all biogeographical regions. All these cases give moreover an insight into the time periods needed for adaptation to changing temperatures. Physical barriers and differential environmental gradients along coastlines also may produce ecotypic adaptation. There has been a wealth of studies tackling this question. It became evident that upper tolerance limits seemingly are quite stable within several seaweed species (Lüning 1990 and references therein). True temperature ecotypes have only been found in a few species yet (e.g., Ectocarpus siliculosus: Bolton 1983, Saccharina latissima: Gerard and Du Bois 1988).
3.2 Changes of Seaweed Distribution and Oceanic Temperature in the Geological Past
Interesting examples for migration in the geological past are present-day amphiequatorial species such as Acrosiphonia arcta (Chlorophyta) or the species pair Desmarestia confervoides /D. viridis (Phaeophyceae), which are absent in the tropics. The fast evolving internal transcribed spacer region of the rDNA of northern and southern hemisphere populations of both entities only exhibit minor variation, suggesting that a migration across the equator took place at the maximum of the Würm/Wisconsin glaciation 18,000 years ago, the so-called last glacial maximum (LGM; van Oppen et al. 1993). The hypothesized migrationist transit of the tropics is supported by the temperature tolerance of both species which is high enough (25°C) to survive a passage through the tropics at the LGM (Peters and Breeman 1992; van Oppen et al. 1994; Bischoff and Wiencke 1995). Similarly it is assumed that a common ancestor of the warm-temperate NE-Atlantic kelp species Laminaria ochroleuca and its S-Atlantic sister species L. abyssalis and L. pallida was able to survive the passage through the tropics during glacial lowering of seawater temperatures by its gametophytic microstages being able to survive at least 25°C (tom Dieck 1992; tom Dieck and de Oliveira 1993).
During the last glaciations, the Arctic ice cap extended south to ~45–55°N and there was a considerable southward shift of the Gulf Stream (CLIMAP Project members 1981; Bradley 1985) inducing major latitudinal dislocations of marine biota in the N-Atlantic between glacial and interglacial periods (van den Hoek and Breeman 1989; Breeman 1990). Some seaweed species were faced with an extreme reduction in their distribution area. The conditions were particularly severe in the NW-Atlantic, where distributions became excessively reduced. A pertinent example is the cold-temperate green alga Cladophora sericea. By comparing temperature demands of the species with modeled glacial sea surface isotherms (van den Hoek and Breeman 1989; Cambridge et al. 1990), it became evident that the distribution area of the species was probably very strongly reduced during the LGM in the NW-Atlantic. In the NE-Atlantic, the distribution was shifted from the coasts of Scandinavia, Great Britain, and France to the Iberian Peninsula, Northwest Africa, and even the Mediterranean became hospitable. These proposed migrational shifts during the LGM within the N-Atlantic have been corroborated by recent phylogeographic studies of diverse seaweed species, indicating the English Channel Region as a primary refugium (e.g., Provan et al. 2005) from which species redispersed to their current distributional range.
An example for a probable extinction during the LGM is the current restriction of the kelp species Laminaria hyperborea to the NE-Atlantic. The much greater compression of the distribution areas on the NW-Atlantic during the LGM—if compared to the NE-Atlantic coasts—suggests that L. hyperborea was not able to survive the inhospitable conditions in the NW-Atlantic during the LGM and became locally extinct (van den Hoek and Breeman 1989). Other kelp species possibly became isolated in cold-water pockets after the LGM. Examples are the deep-water species L. rodriguezii (Huvé 1955; Žuljević et al. 2011) or the isolated population of the southern European kelp L. ochroleuca in southern Italy (Giaccone 1972). Nowadays both species are only found below the thermocline in selected Mediterranean habitats.
3.3 Changes of Oceanic Temperature and Seaweed Distribution in the Future
Following Müller et al. 2009, we analyzed the observed modern (1980–1999; Rayner et al. 2003) and projected end of century (2080–2099) SST distribution for February and August as representatives of summer and winter extreme temperatures. The SST changes are based on the multimodel ensemble mean of coupled climate model simulations prepared for phase 3 of the Coupled Model Intercomparison Project (CMIP3; http://www-pcmdi.llnl.gov) using an emission scenario which assumes a moderate increase of greenhouse gases (SRESA1B) (for details, see Müller et al. 2009). Both the global mean February and August SSTs are projected to warm by 1.9°C until the end of the century (Fig. 18.3). The maximum warming of around 4°C is predicted for high latitudes of the northern hemisphere in summer. The larger land area in the northern hemisphere leads to a stronger response to the radiative forcing than in the southern hemisphere. Additionally, the decrease in summer sea-ice cover amplifies the summer warming. In contrast, minimum warming is predicted in the Southern ocean due to strong ocean heat uptake in this region. In addition to this global scale pattern, some regional scale patterns in the temperature changes become visible. As most climate models predict a reduction in the thermohaline circulation as a response to the increasing greenhouse gas concentration, the heat transport from the tropics to the Polar regions will be affected in the northern hemisphere (Schmittner et al. 2005). This will result in a local cooling in the Labrador Sea in winter and a reduced warming in summer according to the model simulations (Fig. 18.3).
The simulated future changes in sea surface temperatures shown in Fig. 18.3 will result in a general poleward movement of biogeographical regions (see Sect. 18.2) until the end of the twentieth century. The extent of the present-day biogeographical regions and the resulting future changes are shown in Figs. 18.1 and 18.2. This projection is based on the location of modeled mean winter and summer sea surface isotherms for the end of the twentieth century (Müller et al. 2009, Appendix Figs. 1 and 2) characteristic for the current boundaries of the respective regions (see Table 18.1, Figs. 18.1a and 18.2a). Despite the proven recent regional warming in the western Antarctic Peninsula (Vaughan et al. 2003) there will be almost no changes in the northern delimitation of the Antarctic region until the end of this century based on our model. The Arctic region, in contrast, will shrink considerably at its southern border, especially due to warmer winter temperatures while new ice-free coastal Arctic habitats will become available in the north following the retreating pack-ice border (1°C August isotherm) in summer (Fig. 18.2b; Müller et al. 2009). The current cold-temperate regions of both hemispheres will become compressed as the warm-temperate regions are shifting polewards. The cold-temperate regions will gain much new area at the expense of the Polar region only in the N-Atlantic while in the S-Atlantic only the small sub-Antarctic islands will become cold-temperate. The poleward expansion of the warm-temperate regions at the expense of the cold-temperate regions is especially evident in western and eastern S-America, in southeastern Australia and New Zealand, western N-America, and along the European coastline. The warm-temperate regions themselves will lose habitats at the expense of the projected widening of the tropical regions (Figs. 18.1 and 18.2). Recently, it was shown that this process has already started and five different types of climatological measurements revealed a widening of the tropical region of several degrees latitude since 1979 (Seidel et al. 2008). According to our results, this future widening will be especially pronounced along the west and east coast of S-America, along the northern and eastern coastlines of S-Africa and Australia, in SE-Asia, W-Africa, and the Gulf of Mexico (Figs. 18.1 and 18.2).
It becomes apparent that the location of the boundary isotherms characterizing present-day biogeographical regions (Table 18.1) will not move polewards at the same pace according to the model simulations (Figs. 18.1 and 18.2). Thereby, the annual mean temperature minima and maxima and the resulting temperature gradient will change along vast coastlines compared to present day. As a consequence, some biogeographical regions will not be extended or reduced as a whole (Figs. 18.1 and 18.2). Two examples shall demonstrate resulting consequences. Along the coast of China and Japan tropical winter temperatures will not change in the future so that the 22°C February isotherm will stay at approx. 23°N (Fig. 18.1b). In contrast, the corresponding 29°C summer isotherm characterizing the warm-temperate/tropical boundary nowadays will move from approx. 23°N up to the Yellow Sea and Strait of Korea to approx. 36°N (Fig. 18.2b). Hence, this whole area will become a transitional region where biota will experience a reorganization according to the individual growth, reproduction, and lethal limits of the species present (van den Hoek 1982a, b; see Chap. 3 by Eggert). These temperature changes will probably also have consequences for the intensive seaweed aquaculture industry along the Chinese and Japanese coastlines (see Chap. 22 by Buchholz et al.). Another prominent example of a future transitional region is the west coast of S-America. The current austral 21°C winter and 26°C summer SST delimit the biogeographical boundary between the warm-temperate and tropical regions approx. at a latitude of 5°S (northern Peru). According to our simulations, both isotherms will move southwards to approx. 10°S in future austral winter or to approx. 20°S (northern Chile) in future austral summer. The difference between the current and future boundary region thereby spans either 5° or 15° latitude (Figs. 18.1 and 18.2). Thus, a coastline of more than 1,000 km length will become a biogeographical transition region and composition of biota will probably change. It should be noted, however, that due to the influence of the cold Humboldt current and coastal upwelling regions in general, biogeographical regions may not always correspond to the system developed by Briggs (1995) and the model assumptions. Recent evidence, for example, suggests that the northern boundary of the cold-temperate region along the western coastline of S-America set to approx. 40°S by Briggs (1995) actually extends further north to approx. 30°S (Camus 2001; see also Chap. 14 by Huovinen and Gómez).
In addition, the model data identify areas where winter and summer isotherms and thereby possibly the whole biogeographical region will shift homogenously. Examples are the warm-temperate region in eastern and western S-America or the tropical region along eastern and western southern Africa (Figs. 18.1b and 18.2b). Although it is not yet predictable whether minimal and maximal or mean annual SST or altogether will be most responsible for shaping future phytogeographical regions, our data obviously show that new correlations of biogeographical regions with SSTs will establish.
3.4 Specific Effects of Oceanic Warming on Seaweed Distribution and Ecology
3.4.1 Polar Regions
Since the late 1970s the glacial ice sheets have retreated by up to 2% per decade (Serreze et al. 2007) and the Arctic will probably be ice free by the end of this century (Johannessen et al. 2004). In the Antarctic, 87% of the glaciers of the West Antarctic Peninsula are retreating (Cook et al. 2005), the ice season has shortened by about 90 days, and perennial ice does not occur any more at this location (Martinson et al. 2008; Stammerjohn et al. 2008). In the Arctic, oceanic warming leads to a retreat of the pack-ice border coinciding with the 1°C summer isotherm. This will provide new habitats for algal colonization in the Arctic and Antarctic along rocky coastlines (Fig. 18.1; Müller et al. 2009, 2011). As ice-related pressures on shallow water biota of the Arctic and Antarctic will be reduced, perennial macroalgae, which are so far restricted mainly to the sublittoral, will be able to colonize the eulittoral, resulting in an increase in biomass and diversity (Weslawski et al. 2010, 2011). On the other hand, prolonged inflow of glacial melt water will reduce salinity and increase turbidity of the water due to a higher sediment impact (Campana et al. 2011). The concomitant reduction of the euphotic region will change production rates (Pivovarov et al. 2003; Deregibus et al. personal communication; Spurkland and Iken 2011) and probably will cause an upward shift of the depth limit of seaweeds. Biomass and seaweed cover already increased between 1988 and 2008 in the rocky littoral of Sorkappland (Svalbard; Weslawski et al. 2010) in the Arctic accompanied by an increase in air temperature and SST and a marked decrease in the duration and extent of sea-ice cover. However, no “new” species were detected but are expected in future (Müller et al. 2009). The described upward shift of seaweeds might though be counteracted by high levels of ultraviolet-B radiation (UVBR) due to stratospheric ozone depletion (Weatherhead and Andersen 2006; Zacher et al. 2011) which still prevails in the Arctic and Antarctica. UVBR is one of the most important factors controlling the upper depth distribution of seaweeds. Effects have been demonstrated from the cellular to the ecosystem level, affecting community structure and diversity in the Arctic and Antarctic (Bischof et al. 2006; Zacher et al. 2007; Campana et al. 2011; Karsten et al. 2011; Fricke et al. 2011; see Chap. 20 by Bischof and Steinhoff). UVBR, turbidity, water temperature, and sea-ice conditions are interdependent factors but multifactorial interactive effects on polar biota have scarcely been investigated (Müller et al. 2008; Fredersdorf et al. 2009). Bifactorial experiments on Arctic kelp species indicated that negative effects of UVBR can be mitigated by the interaction with increased temperature. For example, germination of zoospores of the kelp Laminaria digitata was inhibited almost completely by UVBR at 2°C, but not at 7°C (Müller et al. 2008).
Compared to changes in the Arctic, the distributional changes of seaweeds in the Antarctic will probably be minor (Müller et al. 2009, 2011) as the model data predict an SST increase of only 1°C throughout the year in the Antarctic region. Moreover, only few cold-temperate species will be able to colonize present-day Antarctic coasts. One example might be the brown alga Chordaria linearis, which has been found already on two locations in West Antarctica (Müller et al. 2009, 2011). The estimation of minor changes for seaweed richness along the coastal West Antarctica Peninsula under climate change conditions contrasts to demonstrated changes in the respective pelagic ecosystem in response to rapid climate changes, which include a shift in phytoplankton biomass, in zooplankton community structure, and expected effects on higher trophic levels (Schofield et al. 2010).
3.4.2 Temperate Regions
Within the last decades, an increase in SSTs of >2°C has been documented in many cold- and warm-temperate regions (see Sect. 18.1). At many temperate European coastlines, migrational shifts of benthic and pelagial species have already taken place (e.g., benthos overview: Mieszkowska et al. 2006; Hawkins et al. 2008; plankton: Beaugrand and Reid 2003; fish: Rijnsdorp et al. 2009). Climate driven biomass changes or loss of kelp vegetation has recently been reported from many local sites worldwide (e.g., Japan: Kirihara et al. 2006, Tasmania: Johnson et al. 2011, Norway: Andersen et al. 2011, Spain: Díez et al. 2012). In contrast, no evidence for broadscale latitudinal shifts of kelps since 1850 was found in the transition region between the boreal and sub-arctic region in the NW-Atlantic (Merzouk and Johnson 2011). A recent investigation into the decline of the sugar kelp Saccharina latissima along southern Norwegian shorelines (Andersen et al. 2011) provides a good example for the complex interactions in the field. After transplantation of S. latissima from healthy to impacted sites, normal growth and maturation took place in winter and spring, but heavy fouling of epiphytes occurred over summer followed by mortality. Although duration of periods with summer temperatures >20°C increased in recent years, a temperature which is sublethal for S. latissima (Bolton and Lüning 1982), mortality could not unequivocally be correlated to high summer temperatures alone. Instead, Andersen et al. (2011) assumed a cascade of reduced growth at sublethal temperatures, followed by heavy epiphytism at locations with low wave activity leading to shading, thereby causing a negative carbon balance and brittleness of thalli and finally mortality—all factors together possibly preventing recruitment and recovery of the species at the impacted sites. Successful recruitment is crucial for the continuous recovery of boundary populations which becomes impacted if the environmental pressure surpasses critical limits. Within a few years of unfavorable abiotic conditions, the reproductive capacity was dramatically reduced in southern European marginal populations of the intertidal brown alga Fucus serratus (Viejo et al. 2011). Similarly, along the SST gradient in western Australia density of kelp recruits was inversely related to increasing mean ocean temperature, suggesting an effect of temperatures on either reproduction or recruits (Wernberg et al. 2010).
There are a few long-term case studies from temperate coastal regions investigating possible ecological consequences of environmental warming for rocky shore communities. Three examples are given here. A 10-year thermal outfall of a power station which induced a long-lasting SST increase by up to 3.5°C resulted in a complete change of the intertidal rocky shore community structure at Diablo Potter Cove in California. The initial dense cover of foliose algae was replaced by bare rocks, algal crusts, or turf algae and there was a major replacement of species and decrease of algal richness (Schiel et al. 2004). A mean SST increase of 0.79°C in the intertidal of Monterey Bay California between 1931 and 1996 led to a significant increase in southern species and a decrease of northern species (Sagarin et al. 1999). In the northern Baltic, another monitoring study of long-lasting warming in the sea (up to 10°C) diminished ice cover and thereby increased light availability in winter. The situation also caused major changes in the quantitative composition of species over the seasonal cycle. While growth of cyanobacteria was promoted and red and brown algae decreased in abundance or disappeared over summer, the latter had a prolonged growth season in autumn and winter due to “better” winter temperatures. Generally, a species-specific response was evident (Snoeijs and Prentice 1989). In future, we expect similar transitional changes in rocky shore communities along all warm- and cold-temperate shorelines possibly subjected to change according to our model results (Figs. 18.1 and 18.2).
3.4.3 Tropical Regions
Many coastal hard-bottom tropical and subtropical regions are characterized by coral reefs which also inherit a high seaweed species richness (Diaz-Pulido et al. 2007). The abundance of macroalgae in reefs has been thought to be generally low and controlled by grazing pressure of herbivorous fish (e.g., Wanders 1977; Carpenter 1986; Hay 1997, see Chap. 16 by Mejia et al.). Only in recent years, it was realized that tropical reefs are also algal reefs and a high coverage of macroalgae among corals and natural variability of seaweed abundance on coral reefs is not necessarily indicative of environmental degradation (Vroom et al. 2006, 2010; Vroom and Timmers 2009). Coral–algal interactions are manifold and it is known that algae may inhibit or kill corals (e.g., Titlyanov et al. 2007; Rasher et al. 2011) and vice versa dead corals may negatively influence macroalgal growth (Liu et al. 2009).
As tropical corals and seaweeds are currently living near to their lethal limit, a slight temperature increase of 1–2°C above the mean summer temperatures as predicted for the end of the twentieth century (Fig. 18.3) may already lead to catastrophic events. Coral reefs worldwide have faced severe damage by periodic heat waves especially through extreme ENSO activities since the 1980s inducing so-called coral bleaching events which involve the loss of the symbiotic zooxanthellae after thermal stress (Jokiel and Coles 1990). Baker et al. (2008) describe in their extensive review all facets of this phenomenon. There is a correlation between coral bleaching with maximum monthly SSTs (Manzello et al. 2007). Temperature thresholds for coral bleaching are not uniform but site specific and range from 27.5 to 32°C (Baker et al. 2008). Thereby, they are generally above current mean tropical summer SST of 27–29°C (Fig. 18.2a; Müller et al. 2009, Appendix Figs. 1 and 2), but this will change in future when this region will experience an unprecedented warming (Solomon et al. 2007) with annual mean SSTs of 30–31°C over wide areas (Fig. 18.2b). A possible acclimation of corals to increased temperatures has been observed in the Great Barrier Reef as threshold temperatures increased over time (Berkelmans 2009) and up to now no coral species became extinct. Currently, reefs are still able to recuperate with highly variable rates, indicating a differential recovery capacity. But full recovery of reefs after thermal stress probably needs decades to centuries (Baker et al. 2008). As growth of macroalgae is much faster and their temperature tolerance is several degree Celsius higher than those of corals (Pakker et al. 1995; Bischoff-Bäsmann et al. 1997), a shift from coral to seaweed dominated coastal ecosystems has been proposed for future tropical areas (Hoegh-Guldberg et al. 2007) and has already been observed during recent decades (Rasher et al. 2011 and references therein).
Current warm-temperate coastlines which will become tropical in future (Figs. 18.1 and 18.2) will not be able to compensate for the prospected loss of reefs as rates needed to establish coral reefs are slow (Baker et al. 2008). Similarly as corals, subtidal reef macroalgae potentially face local extinction if temperature exceeds algal tolerance limits which are firmly set to 30–33°C (Pakker et al. 1995). In contrast, eulittoral tropical macroalgae with their higher lethal temperature limits of up 32–37°C (Bischoff-Bäsmann et al. 1997) will be better able to withstand future temperature increase in the central tropics. First local extinctions after warming events have been recorded: In the Galapagos Archipelago six tropical macroalgal species disappeared after the ENSO warming event in 1982/1983. Here a transition of the macroalgal and coral habitats to heavily grazed reefs dominated by crustose coralline “urchin barrens” was observed (Edgar et al. 2010). Additionally to ocean warming, ocean acidification may decrease coralline abundance in future which will enhance the cascade effect of decreasing coral recruitment, opening space for turf algal species, and further inhibition of coral recruitment, coral fecundity, and coral growth (Hoegh-Guldberg et al. 2007). Unfortunately, the functional ecology and thermal traits of coralline red algae which are important contributors to reef structure and facilitate settlement of corals are virtually unknown (Nelson 2009).
As the tropical region will considerably extend polewards at the expense of the current warm-temperate region (Figs. 18.1 and 18.2), substantial new transitional areas will develop along rocky shore coastlines characterized, for example, by assemblages dominated by tropical to subtropical members of the brown algal order Dictyotales and not corals such as described for tropical to warm-temperate transitional areas of the Canary Islands (Sangil et al. 2011). Locally, other factors such as wave exposure, local currents, and physical barriers may be more important than temperature for biogeographic distribution as has been observed along a tropical to warm-temperate coastal transition region in E-Australia where species distribution is still stable despite recent rapid warming (Poloczanska et al. 2011).
4 Assumptions for Global Seaweed Biodiversity
Global patterns of marine diversity differ from terrestrial habitats where species numbers decrease moving away from the equator in both hemispheres (Pianka 1966; Willig et al. 2003). Marine coastal taxa also show clear latitudinal trends, but overall seaweed genus diversity peaks in mid latitudes and not the tropical region (Kerswell 2006; Santelices et al. 2009; Tittensor et al. 2010). SST was identified as one significant predictor of overall coastal species richness (Tittensor et al. 2010) and high interannual temperature variability was proposed as explanation for the depauperate brown algal flora along the southwest African and north-central Chilean coastline (Bolton 1996). Thus, the predicted changes in SST for the end of the twentieth century will probably have a substantial effect not only on distribution of seaweeds and marine communities in general, but also on global marine diversity. Kerswell (2006) analyzed the latitudinal pattern of global seaweed genus diversity and seaweed endemism in detail. As in other coastal taxonomic groups (Tittensor et al. 2010) there is a band of high diversity in the northwestern Pacific surrounding the Japanese archipelago and in the southwestern Pacific along the southern Australian coastline (Kerswell 2006). In the Atlantic, there is a major hotspot of diversity along the European coastline (Kerswell 2006). All these regions inherit a high seaweed genus diversity and have been identified by our model data to be impacted by future warming. Thus, major changes in seaweed species richness, and the functionality of assemblages through species extinctions, species invasions, and changes in trophic relationships (Sala and Knowlton 2006) are expected especially in these regions. Within the last decade, a wealth of cryptic seaweed species has been described with the help of modern molecular biological tools (e.g., van der Strate et al. 2002; Brodie et al. 2007; Verbruggen et al. 2009). Thus, species richness of marine algae and genetic diversity of their populations probably is strongly underestimated (Zuccarello et al. 2011) and thereby possibly also their adaptive potential to change. But it is not yet clear whether genetic differentiation always coincides with ecological differentiation (Tronholm et al. 2010).
5 Synopsis
Clearly, the prospected worldwide changes in SSTs will exert a differential pressure on seaweed species and assemblages along biogeographical regions. As the ice-free Arctic coastlines and all other regions will expand polewards, the tropical biogeographical region will widen considerably. In contrast, there will be almost no change in the northern limit of the Antarctic region. Most effects on biota will be expected in biogeographical transition regions which have been identified here, for example along the warm- to cold-temperate European coastline or along the warm-temperate to tropical coasts of SW and SE-America, Japan, or China. In these areas, summer and winter SST will move polewards with a differential magnitude so that annual temperature gradients will become more pronounced in some areas. Hence, severe biotic changes are expected as the assemblages characterizing the biogeographic regions will not be able to shift as a whole. Rather, we predict differential species-specific shifts depending on the respective temperature-dependent life cycle characteristics of species which additionally will be shaped by other abiotic and biotic factors. Comparison of coastal areas comprising present-day highest seaweed genus diversity with future biogeographical transition areas revealed a correlation in some cases, for example along western European, Japanese, or southern Australian shorelines. In the future, it will be particularly interesting to investigate the changing structure and function in these transitional biogeographical regions to gain a better understanding of fast acclimation and adaptation rates on an ecosystem level.
References
Andersen GS, Stehen H, Christie H, Fredriksen S, Moy FE (2011) Seasonal patterns of sporophyte growth, fertility, fouling, and mortality of Saccharina latissima in Skagerrak, Norway: Implications for forest recovery. J Mar Biol 2011:Article ID 690375
Ateweberhan M, Bruggemann JH, Breeman AM (2005) Seasonal patterns of biomass, growth and reproduction in Dictyota cervicornis and Stoechospermum polypodioides (Dictyotales, Phaeophyta) on a shallow reef flat in the southern Red Sea (Eritrea). Bot Mar 48:8–17
Augustin L, Barbante C, Barnes PRF, Barnola JM, Bigler M, Castellano E, Cattani O, Chappellaz J, Dahl-Jensen D, Delmonte B, Dreyfus G, Durand G, Falourd S, Fischer H, Flückiger J, Hansson ME, Huybrechts P, Jugie G, Johnsen SJ, Jouzel J, Kaufmann P, Kipfstuhl J, Lambert F, Lipenkov VY, Littot GC, Longinelli A, Lorrain R, Maggi V, Masson-Delmotte V, Miller H, Mulvaney R, Oerlemans J, Oerter H, Orombelli G, Parrenin F, Peel DA, Petit J-R, Raynaud D, Ritz C, Ruth U, Schwander J, Siegenthaler U, Souchez R, Stauffer B, Steffensen JP, Stenni B, Stocker TF, Tabacco IE, Udisti R, van de Wal RSW, van den Broeke M, Weiss J, Wilhelms F, Winther JG, Wolff EW, Zucchelli M (2004) Eight glacial cycles from an Antarctic ice core. Nature 429:623–628
Baker AC, Glynn PW, Riegl B (2008) Climate change and coral reef bleaching: An ecological assessment of long-term impacts, recovery trends and future outlook. Estuar Coastal Shelf Sci 80:435–471
Beaugrand G, Reid PC (2003) Long-term changes in phytoplankton, zooplankton and salmon related to climate. Global Change Biol 9:801–817
Berkelmans R (2009) Bleaching and mortality thresholds: How much is too much? In: van Oppen MJH, Lough JM (eds) Coral bleaching: patterns and processes, causes and consequences, Ecol Studies, vol 205. Springer, New York, pp 103–120
Bischof K, Gómez I, Molis M, Hanelt D, Karsten U, Lüder U, Roleda MY, Zacher K, Wiencke C (2006) Ultraviolet radiation shapes seaweed communities. Rev Environ Sci Biotechnol 5:141–166
Bischoff B, Wiencke C (1995) Temperature ecotypes and biogeography of Acrosiphoniales (Chlorophyta) with Arctic-Antarctic disjunct and Arctic/cold-temperate distributions. Eur J Phycol 30:19–27
Bischoff-Bäsmann B, Wiencke C (1996) Temperature requirements for growth and survival of Antarctic Rhodophyta. J Phycol 32:525–535
Bischoff-Bäsmann B, Bartsch I, Xia B, Wiencke C (1997) Temperature responses of macroalgae from the tropical island Hainan (P.R. China). Phycol Res 45:91–104
Bolton JJ (1983) Ecoclinal variation in Ectocarpus siliculosus (Phaeophyceae) with respect to temperature growth optima and survival limits. Mar Biol 73:131–138
Bolton JJ (1996) Patterns of species diversity and endemism in comparable temperate brown algal floras. Hydrobiologia 327:173–178
Bolton JJ, Lüning K (1982) Optimal growth and maximal survival temperatures of Atlantic Laminaria species (Phaeophyta) in culture. Mar Biol 66:89–94
Bradley RS (1985) Quaternary paleoclimatology. Allan and Unwin, London
Braun M, Gossmann H (2002) Glacial changes in the areas of Admiralty Bay and Potter Cove, King George Island, Maritime Antarctica. In: Beyer L, Bölter M (eds) Geoecology of Antarctic ice-free coastal landscapes, Ecol Studies, vol 154. Springer, Heidelberg, pp 75–90
Breeman AM (1988) Relative importance of temperature and other factors in determining geographic boundaries of seaweeds: experimental and phenological evidence. Helgoländer Meeresunters 42:199–241
Breeman AM (1990) Expected effects of changing seawater temperatures on the geographic distribution of seaweed species. In: Beukema JJ et al (eds) Expected effects of climatic change on marine coastal ecosystems. Kluwer Academic, Netherlands, pp 69–76
Briggs JC (1974) Marine zoogeography. McGraw-Hill, New York
Briggs JC (1995) Global biogeography. Elsevier, Amsterdam
Brodie J, Bartsch I, Neefus C, Orfanidis S, Bray T, Mathieson AC (2007) New insights into the cryptic diversity of the North Atlantic–Mediterranean ‘Porphyra leucosticta’ complex: P. olivii sp. nov. and P. rosengurttii (Bangiales, Rhodophyta). Eur J Phycol 42:3–28
Cambridge ML, Breeman AM, van den Hoek C (1990) Temperature responses limiting the geographical distribution of two temperate species of Cladophora (Cladophorales; Chlorophyta) in the North Atlantic. Phycologia 29:74–85
Campana G, Zacher K, Fricke A, Molis M, Wulff A, Wiencke C (2011) Drivers of colonization and succession in polar benthic macro- and microalgal communities. In: Wiencke C (ed) Biology of polar benthic algae. de Gruyter, Berlin, pp 299–320
Camus PA (2001) Biogeografía marina de Chile continental. Rev Chi Hist Nat 74:587–617
Carpenter RC (1986) Partitioning herbivory and its effects on coral reefs algal communities. Ecol Monogr 56:345–363
CLIMAP Project Members (1981) Seasonal reconstructions of the earth’s surface at the last glacial maximum. The Geological Society of America, Map and Chart Service MC-36, Washington DC
Collins M, An S-I, Cai W, Ganachaud A, Guilyardi JF-F, Jochum M, Lengaigne M, Power S, Timmermann A, Vecchi G, Wittenberg A (2010) The impact of global warming on the tropical Pacific ocean and El Niño. Nat Geosci 3:391–397
Cook AJ, Fox AJ, Vaughan DG, Ferrigno JG (2005) Retreating glacier fronts on the Antarctic Peninsula over the past half-century. Science 308:541–544
Crame JA (1993) Latitudinal range fluctuations in the marine realm through geological time. Trends Ecol Evol 10:1096–1111
Davison IR, Pearson GA (1996) Stress tolerance in intertidal seaweeds. J Phycol 32:197–211
Davison IR, Greene RM, Podolak EJ (1991) Temperature acclimation of respiration and photosynthesis in the brown alga Laminaria saccharina. Mar Biol 110:449–454
Diaz-Pulido G, McCook LJ, Larkum AWD, Lotze HK, Raven JA, Schaffelke B, Smith JE, Steneck RS S (2007) Vulnerability of macroalgae of the Great Barrier Reef to climate change. In: Johnson JE, Marshall PA (eds) Climate change and the Great Barrier Reef. Great Barrier Reef Marine Park Authority and Australian Greenhouse Office, Townsville, Queensland, pp 153–192
Díez I, Muguerza N, Santolaria A, Ganzedo U, Gorostiaga JM (2012) Seaweed assemblage changes in the Eastern Cantabrian Sea and their potential relationship to climate change. Estuar, Coastal Shelf Sci 99:108–120
Dring MJ (1984) Photoperiodism and phycology. In: Round F, Chapman DJ (eds) Progress in phycological research, vol 3. Biopress, Bristol, pp 159–192
Edgar GJ, Banks SA, Brandt M, Bustamante RH, Chiriboga A, Earle SA, Garske LE, Glynn PW, Grove JS, Henderson S, Hickmam CP, Miller KA, Rivera F, Wellington GM (2010) El Niño, grazers and fisheries interact to greatly elevate extinction risk for Galapagos marine species. Global Change Biol 10:2876–2890
Eggert A, Visser RJW, van Hasselt PR, Breeman AM (2006) Differences in acclimation potential of photosynthesis in seven isolates of the tropical to warm temperate macrophyte Valonia utricularis (Chlorophyta). Phycologia 45:546–556
Fredersdorf J, Müller R, Becker S, Wiencke W, Bischof K (2009) Interactive effects of radiation, temperature and salinity on different life history stages of the Arctic kelp Alaria esculenta (Phaeophyceae). Oecologia 160:483–492
Fricke A, Molis M, Wiencke C, Valdivia N, Chapman AS (2011) Effects of UV radiation on the structure of Arctic macrobenthic communities. Polar Biol 34:995–1009
Gerard VA, Du Bois KR (1988) Temperature ecotypes near the southern boundary of the kelp Laminaria saccharina. Mar Biol 97:575–580
Giaccone G (1972) Struttura, ecologia e corologia dei popolamenti a Laminarie dell stretto di Messina e del mare di Alboran. Mem Biol Mar Ocean NS 2:37–49
Graham MH, Kinlan BP, Druehl LD, Garske LE, Banks S (2007) Deep-water kelp refugia as potential hotspots of tropical marine diversity and productivity. Proc Nat Acad Sci USA 104:16576–16580
Guisan A, Thuiller W (2005) Predicting species distribution: offering more than simple habitat models. Ecol Lett 8:993–1009
Hansen J, Sato M, Ruedy R, Lo K, Lea DW (2006) Global temperature change. Proc Natl Acad Sci 103:14288–14293
Harley CDG, Hughes AR, Hultgren KM, Miner BG, Sorte CJB, Thornber CS, Rodriguez LF, Tomanek L, Williams SL (2006) The impacts of climate change in coastal marine systems. Ecol Lett 9:228–241
Hawkins SJ, Moore PJ, Burrows MT, Poloczanska E, Mieszkowska N, Herbert RJH, Jenkins SR, Thompson RC, Genner MJ, Southward AJ (2008) Complex interactions in a changing world: Responses of rocky shore communities to recent climate change. Climate Res 37:123–133
Hay ME (1997) The ecology and evolution of seaweed-herbivore interactions on coral reefs. Coral Reefs 16:S67–S76
Hoegh-Guldberg O, Mumby PJ, Hooten AJ, Steneck RS, Greenfield P, Gomez E, Harvell CD, Sale PF, Edwards AJ, Caldeira K, Knowlton N, Eakin CM, Iglesias-Prieto R, Muthiga N, Bradbury RH, Dubi A, Hatziolos ME (2007) Coral reefs under rapid climate change and ocean acidification. Science 318:1737–1742
Huvé H (1955) Présence de Laminaria rodriguezii Bornet sur les côtes de Mediterraneée. Rec Trav Stat Mar Endoume 15:73, 89 + 11 plates
Johannessen O, Bengtsson L, Miles M, Kuzmina S, Semenov V, Alekseev G, Nagurnyi A, Zakharov V, Bobylev L, Pettersson L (2004) Arctic climate change: observed and modeled temperature and sea-ice variability. Tellus 56(A):328–341
Johnson CR, Banks SC, Barrett NS, Cazassus F, Dunstan PK, Edgar GJ, Frusher SD, Gardner C, Haddon M, Helidoniotis F, Hill KL, Holbrook NJ, Hosie GW, Last PR, Ling SD, Melbourne-Thomas J, Miller K, Pecl GT, Richardson AJ, Ridgway KR, Rintoul SR, Ritz DA, Ross DJ, Sanderson JC, Shepherd SA, Slotwinski A, Swadling KM, Taw N (2011) Climate change cascades: Shifts in oceanography, species’ ranges and subtidal marine community dynamics in eastern Tasmania. J Exp Mar Biol Ecol 400:17–32
Jokiel PL, Coles SL (1990) Response of Hawaiian and other Indo-Pacific reef corals to elevated temperature. Coral Reefs 8:155–162
Karsten U, Wulff A, Roleda M, Müller R, Steinhoff FS, Fredersdorf J, Wiencke C (2011) Physiological responses of polar benthic algae to ultraviolet radiation. In: Wiencke C (ed) Biology of polar benthic algae. De Gruyter, Berlin, pp 271–298
Kerswell AP (2006) Global biodiversity patterns of benthic marine algae. Ecology 87:2479–2488
Kirihara S, Nakamura T, Kon N, Fujita D, Notoya M (2006) Recent fluctuations in distribution and biomass of cold and warm temperature species of Laminarialean algae at Cape Ohma, northern Honshu, Japan. J Appl Phycol 18:521–527
Knutti R, Flückinger J, Stocker TF, Timmermann A (2004) Strong hemispheric coupling of glacial climate through freshwater discharge and ocean circulation. Nature 430:851–856
Kübler JE, Davison IR (1995) Thermal acclimation of light-use characteristics of Chondrus crispus (Rhodophyta). Eur J Phycol 30:189–195
Levitus S, Antonov JI, Boyer TP, Stephens C (2000) Warming of the world ocean. Science 287:2225–2229
Lima FP, Ribeiro PA, Queiroz N, Hawkins SJ, Santos AM (2007) Do distributional shifts of northern and southern species of algae match the warming pattern? Glob Change Biol 13:2592–2604
Liu PJ, Lin SM, Fan TY, Meng PJ, Shao KT, Lin HJ (2009) Rates of overgrowth by macroalgae and attack by sea anemones are greater for live coral than dead coral under conditions of nutrient enrichment. Limnol Oceanogr 54:1167–1175
Lüning K (1990) Seaweeds: Their environment, biogeography and ecophysiology. Wiley, New York
Manzello DP, Berkelmans R, Hendee JC (2007) Coral bleaching indices and thresholds for the Florida reef tract, Bahamas, and St. Croix, US Virgin Islands. Mar Pollut Bull 54:1923–1931
Martinson DG, Stammerjohn SE, Iannuzzi RA, Smith RC, Vernet M (2008) Western Antarctic Peninsula physical oceanography and spatio-temporal variability. Deep Sea Res II 55:1964–1987
Merzouk A, Johnson LE (2011) Kelp distribution in the northwest Atlantic Ocean under a changing climate. J Exp Mar Biol Ecol 400:90–98
Mieszkowska N, Kendall MA, Hawkins SJ, Leaper R, Williamson P, Hardman-Mountfort NJ, Southward AJ (2006) Changes in the range of some common rocky shore species in Britain—a response to climate change? Hydrobiologia 555:241–251
Molenaar F (1996) Seasonal growth and reproduction of North Atlantic red seaweeds. PhD dissertation, University of Groningen, Netherlands, p 111
Müller R, Wiencke C, Bischof K (2008) Interactive effects of UV radiation and temperature on microstages of Laminariales (Phaeophyceae) from the Arctic and North Sea. Clim Res 37:203–213
Müller R, Bartsch I, Laepple T, Wiencke C (2009) Impact of oceanic warming on the distribution of seaweeds in polar and cold-temperate waters. Bot Mar 52:617–638
Müller R, Bartsch I, Laepple T, Wiencke C (2011) Impact of oceanic warming on the distribution of seaweeds in polar and cold-temperate waters. In: Wiencke C (ed) Biology of polar benthic algae. De Gruyter, Berlin, pp 237–270
Nelson WA (2009) Calcified macroalgae—critical to coastal ecosystems and vulnerable to change: a review. Mar Freshwater Res 60:787–801
Pakker H, Breeman AM, Prud’homme van Reine WF, van den Hoek C (1995) A comparative study of temperature responses of Carribean seaweeds from different biogeographic groups. J Phycol 31:499–507
Pearson RG, Dawson TP (2003) Predicting the impacts of climate change on the distribution of species: are bioclimate envelope models useful? Global Ecol Biogeogr 12:361–371
Peters AF, Breeman AM (1992) Temperature responses of disjunct temperate brown algae indicate long-distance dispersal of microthalli across the tropics. J Phycol 28:428–438
Pianka ER (1966) Latitudinal gradients in species diversity: a review of concepts. Am Natural 100:33–46
Pivovarov S, Schlitzer R, Novikhin A (2003) River runoff influence on the water mass formation in the Kara Sea. In: Stein R, Fahl K, Fütterer DK, Galimov EM, Stepanets OV (eds) Siberian river run-off in the Kara Sea: characterisation, quantification, variability, and environmental significance. Elsevier, Amsterdam, pp 9–26
Poloczanska ES, Smith S, Fauconnet L, Healy J, Tibbetts IR, Burrows MT, Richardson AJ (2011) Little change in the distribution of rocky shore faunal communities on the Australian east coast after 50 years of rapid warming. J Exp Mar Biol Ecol 400:145–154
Provan J, Wattier RA, Maggs CA (2005) Phylogeographic analysis of the red seaweed Palmaria palmata reveals a Pleistocene marine glacial refugium in the English Channel. Mol Ecol 14:793–803
Rasher DB, Stout EP, Engel S, Kubanek J, Hay ME (2011) Macroalgal terpernes function as allelopathic agents against reef corals. Proc Natl Acad Sci USA 43:17726–17731
Rayner NA, Parker DE, Horton EB, Folland CK, Alexander LV, Rowell DP, Kent EC, Kaplan A (2003) Global analyses of sea surface temperature, sea ice, and night marine air temperature since the late nineteenth century. J Geophys Res 108(D14):4407
Rijnsdorp AD, Peck MA, Engelhard GH, Mollmann C, Pinnegar JK (2009) Resolving the effect of climate change on fish populations. ICES J Mar Sci doi:10.1093/icesjms/fsp056
Sagarin RD, Barry JB, Gilman SE, Baxter CH (1999) Climate-related change in an intertidal community over short and long time scales. Ecol Monogr 69:465–490
Sala E, Knowlton N (2006) Global marine biodiversity trends. Annu Rev Environ Resour 31:93–122
Sangil C, Sanson M, Afonso-Carillo J (2011) Spatial variation patterns of subtidal assemblages along a subtropical oceanic archipelago: Thermal gradient vs herbivore pressure. Estuar Coast Shelf Sci 94:322–333
Santelices B, Bolton JJ, Meneses I (2009) 6. Marine algal communities. In: Whitman JD, Roy K (eds) Marine macroecology. Chicago University Press, Chicago, pp 153–192
Schiel DR, Steinbeck JR, Foster MS (2004) Ten years of induced ocean warming causes comprehensive changes in marine benthic communities. Ecology 85:1833–1839
Schmittner A, Latif M, Schneider B (2005) Model projections of the North Atlantic thermohaline circulation for the 21st century assessed by observations. Geophys Res Lett 32:L23710. doi:10.1029/2005GL024368
Schofield O, Ducklow HW, Martinson DG, Meredith MP, Moline MA, Fraser WR (2010) How do polar marine ecosystems respond to rapid climate change? Science 328:1529–1523
Seidel DJ, Fu Q, Randel WJ, Reichler TJ (2008) Widening of the tropical belt in a changing climate. Nat Geosci 1:21–24
Serreze MC, Holland MM, Stroeve J (2007) Perspectives on the Arctic’s shrinking sea-ice cover. Science 315:1533–1536
Snoeijs PJM, Prentice IC (1989) Effects of cooling water discharge on the structure and dynamics of epilithic algal communities in the northern Baltic. Hydrobiologia 184:99–123
Solomon S, Qin D, Manning M, Chen Z, Marquis M, Averyt KB, Tignor M, Miller HL (eds 2007) Contribution of Working Group I to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change. Cambridge University Press, Cambridge, New York
Spalding MD, Fox HE, Allen GR, Davidson N, Ferdaña ZA, Finlayson M, Halpern BS, Jorge MA, Lombana A, Lourie SA, Martin KD, McManus E, Molnar J, Recchia CA, Robertson J (2007) Marine ecoregions of the world: a bioregionalization of coastal and shelf areas. BioScience 57:573–583
Spurkland T, Iken K (2011) Salinity and irradiance effects on growth and maximum photosynthetic quantum yield in subarctic Saccharina latissima (Laminariales, Laminariaceae). Bot Mar 54:355–365
Stammerjohn SE, Martinson DG, Smith RC, Yuan X, Rind D (2008) Trends in Antarctic annual sea ice retreat and advance and their relation to El Niño-southern oscillation and southern annual mode variability. J Geophys Res 113:C03S90
Titlyanov EA, Yakovleva IM, Titlyanova TV (2007) Interaction between benthic algae (Lyngbya bouillonii, Dictyota dichotoma) and scleractinian coral Porites lutea in direct contact. J Exp Mar Biol Ecol 342:282–291
Tittensor DP, Mora C, Jetz W, Lotze HK, Ricard D, van den Berghe E, Worm B (2010) Global patterns and predictors of marine biodiversity across taxa. Nature 466:1098–1101
tom Dieck I (1992) North Pacific and North Atlantic digitate Laminaria species (Phaeophyta): hybridization experiments and temperature responses. Phycologia 31:147–163
tom Dieck I, de Oliveira EC (1993) The section Digitatae of the genus Laminaria (Phaeophyta) in the northern and southern Atlantic: crossing experiments and temperature responses. Mar Biol 115:151–160
Tronholm A, Steen F, Tyberghein L, Leliaert F, Verbruggen H, Siguan MAR, De Clerk O (2010) Species delimitation, taxonomy, and biogeography of Dictyota in Europe (Dictyotales, Phaeophyceae). J Phycol 46:1301–1321
Turner J, Overland JE, Walsh JE (2007) An Arctic and Antarctic perspective on recent climate change. Int J Climatol 27:277–293
van den Hoek C (1982a) The distribution of benthic marine algae in relation to the temperature regulation of their life histories. Biol J Linn Soc 18:81–144
van den Hoek C (1982b) Phytogeographic distribution groups of benthic marine algae in the North Atlantic Ocean. A review of experimental evidence from life history studies. Helgoländer Meeresunters 35:53–214
van den Hoek C, Breeman AM (1989) Seaweed biogeography in the North Atlantic: Where are we now? In: Garbary DJ, South GR (eds) Evolutionary biogeography of the marine algae of the North Atlantic. Springer, Berlin, pp 55–86
van der Strate HJ, Boele-Bos SA, Olsen JL, van den Zande L, Stam WT (2002) Phylogeographic studies in the tropical seaweed Cladophoropsis membranacea (Chlorophyta, Ulvophyceae) reveal a cryptic species complex. J Phycol 38:572–582
van Oppen MJH, Olsen JL, Stam WT, van den Hoek C, Wiencke C (1993) Arctic-Antarctic disjunctions in the benthic seaweeds Acrosiphonia arcta (Chlorophyta) and Desmarestia viridis/willii (Phaeophyta) are of recent origin. Mar Biol 115:381–386
van Oppen MJH, Diekmann OE, Wiencke C, Stam WT, Olsen JL (1994) Tracking dispersal routes: Phylogeography of the Arctic-Antarctic disjunct seaweed Acrosiphonia arcta (Chlorophyta). J Phycol 30:67–80
Vaughan DG, Marshall GJ, Connolley WM, Parkinson C, Mulvaney R, Hodgson DA, King JC, Pudsey CJ, Turner J (2003) Recent rapid regional climate warming on the Antarctic Peninsula. Clim Change 60:243–274
Verbruggen H, Tyberghein L, Pauly K, Vlaeminck C, van Nieuwenhuyze K, Kooistra WHCF, Leliaert F, De Clerk O (2009) Macroecology meets macroevolution: evolutionary niche dynamics in the seaweed Halimeda. Global Ecol Biogeogr 18:393–405
Viejo RM, Martínez B, Arrontes J, Astudillo C, Hernández L (2011) Reproductive patterns in central and marginal populations of a large brown seaweed: drastic changes at the southern range limit. Ecography 34:75–84
Vroom PS, Timmers MAV (2009) Spatial and temporal comparison of algal biodiversity and benthic cover at Gardner Pinnacles, northwestern Hawai’ian islands. J Phycol 45:337–347
Vroom PS, Page KN, Kenyon JC, Brainard RE (2006) Algae-dominated reefs. Am Scientist 94:430–437
Vroom PS, Musburger CA, Cooper SW, Maragos JE, Page-Albins KN, Timmers MAV (2010) Marine biological community baselines in unimpacted tropical ecosystems: spatial and temporal analysis of reefs at Howland and Baker Islands. Biodivers Conserv 19:797–812
Wanders JBW (1977) The role of benthic algae in the shallow reef of Curaçao (Netherlands Antilles). 3. The significance of grazing. Aquat Bot 3:357–390
Weatherhead EC, Andersen SB (2006) The search for signs of recovery of the oregion layer. Nature 441:39–45
Wernberg T, Thomsen MS, Tuya F, Kendrick GA, Staehr PA, Toohey BD (2010) Decreasing resilience of kelp beds along a latitudinal temperature gradient: potential implications for a warmer future. Ecol Lett 13:685–694
Weslawski JM, Wiktor J, Kotwicki L (2010) Increase in biodiversity in the arctic rocky littoral, Sorkappland, Svalbard, after 20 years of climate warming. Mar Biodiv 40:123–130
Weslawski JM, Kendall MA, Wlodarska-Kowalczuk M, Iken K, Kedra M, Legezynska J, Sejr MK (2011) Climate change effects on Arctic fjord and coastal macrobenthic diversity—observations and predictions. Mar Biodiv 41:71–85
Wiencke C, Bartsch I, Bischoff B, Peters AF, Breeman AM (1994) Temperature requirements and biogeography of Antarctic, Arctic and amphiequatorial seaweeds. Bot Mar 37:247–259
Willig MR, Kaufman DM, Stevens RD (2003) Latitudinal gradients of biodiversity: pattern, process, scale, and synthesis. Annu Rev Ecol Evol Syst 34:273–309
Wiltshire KH, Malzahn M, Wirtz K, Greve W, Janisch S, Mangelsdorf P, Manly BFJ, Boersma M (2008) Resilience of North Sea phytoplankton spring bloom dynamics: an analysis of long-term data at Helgoland Roads. Limnol Oceanogr 53:1294–1302
Yarish C, Breeman AM, van den Hoek C (1984) Temperature, light and photoperiod responses of some Northeast American and west European endemic rhodophytes in relation to their geographic distribution. Helgoländer Meeresunters 38:273–304
Zacher K, Wulff A, Molis M, Hanelt D, Wiencke C (2007) Ultraviolet radiation and consumer effects on a field-grown intertidal macroalgal assemblage in Antarctica. Global Change Biol 13:1201–1215
Zacher K, Rautenberger R, Hanelt D, Wulff A, Wiencke C (2011) The abiotic environment of polar marine benthic algae. In: Wiencke C (ed) Biology of polar benthic algae. De Gruyter, Berlin, pp 9–21
Zachos JC, Dickens GR, Zeebe RE (2008) An early Cenozoic perspective on green house warming and carbon-cycle dynamics. Nature 451:279–283
Zardi GI, Nicastro KR, Canovas F, Costa JF, Serrão EA, Pearson GA (2011) Adaptive traits are maintained on steep selective gradients despite gene flow and hybridization in the intertidal region. PLoS One 6(e19402):1–12
Zuccarello GC, Buchanan J, West JA, Pedroche FF (2011) Genetic diversity of the mangrove-associated alga Bostrychia radicans/Bostrychia moritziana (Ceramiales, Rhodophyta) from southern Central America. Phycol Res 59:98–104
Žuljević A, Antolić B, Nikolić V, Isajlović I (2011) Review of Laminaria rodriguezii records in the Adriatic Sea. 5th European Phycological Congress, Abstract Book, p 194
Acknowledgements
We acknowledge the modeling group WCRP’s Working Group on Coupled Modelling (WGCM) for making available the WCRP CMIP3 multi-model dataset. Support of this dataset is provided by the Office of Science, U.S. Department of Energy. The authors thank R. Müller and C. Buchholz for helpful advice on an earlier draft of this chapter.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2012 Springer-Verlag Berlin Heidelberg
About this chapter
Cite this chapter
Bartsch, I., Wiencke, C., Laepple, T. (2012). Global Seaweed Biogeography Under a Changing Climate: The Prospected Effects of Temperature. In: Wiencke, C., Bischof, K. (eds) Seaweed Biology. Ecological Studies, vol 219. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-642-28451-9_18
Download citation
DOI: https://doi.org/10.1007/978-3-642-28451-9_18
Published:
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-642-28450-2
Online ISBN: 978-3-642-28451-9
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)