Abstract
Chemical and microbial characterizations of a rice paddy soil subjected to long-term heavy metal pollution (P) and nonpolluted (NP) soil are used to investigate whether the distribution of heavy metals (Cd, Cu, Pb, and Zn) regulates microbial community activity, abundance, and diversity at the microenvironment scale. The soils are physically fractionated into coarse-sand, fine-sand, silt, and clay fractions. Long-term heavy metal pollution notably decreases soil basal respiration (a measurement of the total activity of the soil microbial community) and microbial biomass carbon across the fractions, respectively. The coarse-sand fraction is more affected by pollution than the clay fraction and displayed a significantly lower respiration and dehydrogenase activity. The abundances and diversities of bacteria were less affected under pollution. Long-term heavy metal pollution decreased the microbial biomass, activity, and diversity in long-term exposure.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
9.1 Introduction
Paddy fields are the production bases of the world’s most important staple food crop (rice) (Yang and Hao 2011), and they also comprise an important artificial wetland ecosystem, which may have great significance in regional ecological (e.g., hydrological, thermal, and biotic) balance and material circulation (Yang and Zhang 2014). Soil is a complex ecosystem where living organisms play a key role in the maintenance of its properties. Soil biota comprises a huge diversity of organisms belonging to different taxonomic and physiologic groups, which interact at different levels within the community. Soil microorganisms constitute a source and sink for nutrients and are involved in numerous activities, such as transformation of C, N, P, and S, degradation of xenobiotic organic compounds, formation of soil physical structure, and enhancement of plants’ nutrient uptake (Gregorich et al. 1994; Seklemova et al. 2001). For these reasons, the importance of microorganisms in the maintenance of quality and productivity of agricultural soils is unquestionable (Lopes et al. 2011). The responsiveness of microorganisms to environmental factors implies that disturbances imposed by agricultural treatments may lead to alterations in the composition and activity of soil micro biota and, therefore, may affect soil quality (Gregorich et al. 1994; Shibahara and Inubushi 1997).
The paddy field is a unique agroecosystem consisting of diverse habitats of microorganisms and other living things (Tang et al. 2014). Soil organisms contribute to the maintenance of soil quality (physical, chemical, and biological indicators) by controlling the decomposition of plant and animal materials, biogeochemical cycling, and the formation of soil structure. Land-use conversion is a common and direct factor that affects soil quality (Zhang et al. 2009). Land-use conversion from paddy field to upland has led to declines in soil moisture, pH, organic carbon, nitrogen, and microbial biomass and activity, as well as changes in soil structure, aeration, and nutrient status (Cao et al. 2004; Nishimura et al. 2008), which had important effects on soil microbial community structure (Steenwerth et al. 2002; Zhang et al. 2009).
The soil microbial community and its diversity are strongly influenced by anthropogenic land-use conversions (Yang and Zhang 2014). Maintenance of soil quality and crop yields relies on the functions of soil microorganisms for organic matter decomposition, residue degradation, and nutrient transformations
Microorganisms play an important role in agricultural ecosystems, mainly in terms of sustainability and the quality of agricultural soils (Li et al. 2017). In rice cultivation less than half of the total rice biomass is edible, and the remaining parts consist of straw, stubble, and rice root. The decomposition rate of the straw residues above ground is faster than that of the roots below ground (Lu et al. 2003). The different decomposition rates are due to both the chemical composition of the residues and the microbial community involved in degrading these residues. Focusing on biological process changes in different residue sources can alter the decomposition process indicating that understanding the significance of biodiversity on decomposition is essential to assess the consequences of biodiversity change for carbon and nutrient cycles. Cellulose degradation is one of the most important biological processes because of the large amount of cellulose in plant dry weight (30–50%). This process can occur under aerobic and anaerobic conditions. Both microorganisms, i.e., bacteria and fungi, are actively involved in this process. Bacterial or fungal decomposition can result in different amounts and composition of decomposed products. Aerobic cellulolytic fungi are remarkably effective degraders in cellulolytic systems compared with aerobic bacteria. These two degrader microbial groups can either facilitate partition or inhibit interactions depending on the substrates they inhabit. Soil microbial diversity is declining worldwide primarily because of human-induced global changes, and at least some soil species are known to be vulnerable to these changes. Several studies (Do Thi Xian 2012) in agroecosystems have reported reductions in soil faunal biodiversity associated with increased management. This biodiversity loss reduces the efficiency by which ecological communities capture biologically essential resources produce biomass decompose and recycle biologically essential nutrients.
The nutrient availability of surface soils is usually greater than that of subsurface soil because of the input of crop root exudates, surface litter, and root detritus in agricultural systems. Furthermore fertilizer applications enlarge the nutrient gap between surface and subsurface soils (Li et al. 2014). Nutrient availability, pH, soil texture, temperature, and moisture content can vary considerably with soil depth. Differences in physical and chemical parameters along a soil depth gradient allow for the proliferation of diverse microbial communities. Deeper soil layers may contain microbial communities that have adapted to this environment, and these communities may be distinct from surface communities (Bai et al. 2015). Many environmental factors that influence microbial communities have been reported including pH (Griffiths et al. 2011), soil texture (Chodak and Niklinska 2010), nutrient availability (Bowles et al. 2014), water content (Moche et al. 2015; Praeg et al. 2014), and temperature. These environmental parameters usually exhibit spatial heterogeneity; thus, the distributions of the microbial community along a soil depth gradient will vary greatly with spatial location (Li et al. 2017).
Variability in microbial communities can be related to specific nutrient characteristics of soil, i.e., phosphorus, soil moisture, etc. The microbial community dynamics is significantly correlated with total carbon, moisture, available potassium, and pH in high-yielding paddy soil ecosystem, while in low-yield soil, variability in pH and nitrogen factors affect microbial activity. The high-yield soil microbes are probably more active to modulate soil fertility for rice production. Soil characteristics like total nitrogen, total potassium, and moisture are said to significantly affect enzymatic activities of paddy soil microbial diversity. The enzymatic variability in paddy soil is explained by moisture, potassium, available nitrogen, pH, and total carbon (Lou et al. 2016).
9.2 Influence of Heavy Metals on Paddy Soil Microbial Diversity and Enzymatic Activity
Soil microorganisms and enzymes are the primary mediators of soil biological processes, including organic matter degradation, mineralization, and nutrient recycling. They play an important role in maintaining soil ecosystem quality and functional diversity. Structural polysaccharides include cellulose, xylane, chitin, and polyphenol, while starch is the fundamental storage polysaccharide in plants. Once being incorporated into soil, these polysaccharides are hydrolyzed to oligosaccharides by polysaccharidases, e.g., xylanase for xylane and hemicellulose and amylase for starch. They are further degraded to monosaccharides by heterosidases, i.e., b-glucosidase for cellobiose, invertase for sucrose, and N-acetyl-b-d-glucosaminidase for chitooligosaccharides. Low-molecular-weight sugars are mineralized as energy sources by soil microbes. Organics N and P are simultaneously mineralized in the process of decomposition by other hydrolases, such as urease and phosphatase (Li et al. 2009). Accordingly, the activities of enzymes involved in soil organic C, N, and P cycles are considered to be useful indices, or representing modification of microbial communities, because this community composition determines the potential for soil enzyme syntheses (Huaidong et al. 2017). Effects of soil pollution on enzyme activities are complex. The response of different enzymes to the same pollutant may vary greatly, and the same enzyme may respond differently to different pollutants.
9.3 Heavy Metal Contamination
Heavy metal contamination in agricultural soils due to anthropogenic mining activities can result in adverse environmental effects, including soil quality degeneration, inhibition of crop growth, and potential risks to human health by food chain transfer (Li et al. 2014). Paddy fields, a unique agroecosystem, are of vital importance for cereal production in Asia (Li et al. 2012; Chen et al. 2014). Similarly, arsenic (As) contamination (highly toxic) in paddy fields has been of concern worldwide (Meharg and Rahman 2003). China is the world’s largest rice producer, accounting for 30% of the world’s total production (Kogel-Knabner et al. 2010). During rice cultivation, paddy fields are periodically flooded, for favoring reductive dissolution of iron (Fe) oxides (Takahashi et al. 2004; Yamaguchi et al. 2011), resulting in an increase in bioavailability as to rice (Xu et al. 2008; Wang et al. 2017a).
In various studies, soil metals have showed heterogeneous effects on soil enzymes. Copper, Mn, Pb, and Cd inhibited organic C-acquiring enzyme activity (b-glucosidase and invertase), total enzyme activity, and AcdP, while Cd and Zn enhance organic N-acquiring enzyme activities (NA-Gase and urease) and amylase activity, while none of these metals influence xylanase and AlkP activity (Li et al. 2009).
9.4 Soil Microbiomes
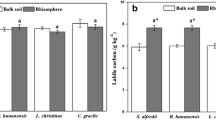
There is an increasing concern about rice (Oryza sativa L.) soil microbiomes under the influence of mixed heavy metal contamination. The bacterial diversity and community composition of soils containing heavy metal (Cd, Pb, and Zn) pollution in paddy fields have been studied. A wide range of bacterial operational taxonomic units (OTUs) in the bulk and rhizosphere soils of the paddy fields have been reported with the dominant bacterial phyla (greater than 1% of the overall community) including Proteobacteria, Actinobacteria, Firmicutes, Acidobacteria, Gemmatimonadetes, Chloroflexi, Bacteroidetes, and Nitrospirae. Rice rhizosphere soils display higher bacterial diversity indices. Total Cd and Zn in the soils show a negative correlation with biodiversity abundance (Huaidong et al. 2017). Similarly, total Pb, total Zn, pH, total nitrogen, and total phosphorus significantly affect the community structure. The activities of dehydrogenase and microbial biomass carbon are sensitive to the toxicity of these heavy metals and can be used as eco-indicators of soil pollution (Hu et al. 2014).
Copper (Cu) contamination is common in paddy fields due to wastewater irrigation and application of sludge and Cu-containing fungicides (Mao et al. 2015). It has a large effect on the soil microbial community structure (Chao-Rong and Zhang 2011). In various studies (Pennanen et al. 1996; Baath et al. 1998), Cu is considered as the most toxic metal around primary smelter. At a threshold level (100–200 μg/g), Cu concentration is responsible to induce toxic effects. Similarly, elevated Cu concentration, i.e., 1000 μg/g soil, has also been reported as a threshold value for a pollution effect on the phospholipid fatty acid (PLFA) patterns (Yao et al. 2003). The substrate utilization pattern depicts significant stress caused by heavy metal pollution. Such information can be useful in evaluating soil quality and support efforts for the recovery of soil ecosystems. Heavy metals like mercury (Hg) and lead (Pb) in paddy soil severely affect the soil ecosystem. They show their toxic effects on microbial biomass carbon (MBC) and microbial biomass nitrogen (MBN). Mercury is highly toxic than lead (Shang et al. 2012). When exposed to mercury, the microbial biomass carbon can result in significant reduction of up to 0.6 mg/kg soil, while microbial biomass nitrogen may reduce to 0.8 mg/kg soil. As to lead, the critical content is 150 mg/kg soil for microbial biomass carbon and 100 mg/kg soil for microbial biomass nitrogen. Various studies (Yang et al. 2016) suggest that the inclusion of native periphyton in paddy fields provides a promising buffer to minimize the effects of Cu and Cd pollution on rice growth and food safety. The microbial biomass, activity, and substrate utilization pattern of paddy soils (China) with different heavy metal concentrations in the vicinity of a Cu–Zn smelter has been investigated. The elevated metal levels negatively affect microbial biomass and basal respiration.
9.5 Biological Parameters
The two important biological parameters, i.e., the microbial biomass C (Cmic)/organic (Corg) ratio and metabolic quotient, are closely correlated to heavy metal stress (Yao et al. 2003). There is a significant decrease in their ratio and an increase in the metabolic quotient with increasing metal concentration.
High levels of vanadium have long-term, hazardous impacts on soil ecosystems and biological processes. The effects of V on soil enzymatic activities and the microbial community structure have also been explored (Xiao et al. 2017). The metal vanadium affects soil dehydrogenase activity (DHA), basal respiration (BR), and microbial biomass carbon (MBC), while urease activity is considered less sensitive to its stress. BR and DHA are more sensitive to V addition and hence can be used as biological indicators for polluted soil. The structural diversity of the microbial community can be severely affected by vanadium toxicity ranged between 254 and 1104 mg/kg after 1 week of incubation. There are also chances to develop microbial adaptation in soil community to resist the pollution. The diversity of paddy soil microbial community structure is expected to rise with the prolonged incubation time in response to vanadium concentration, i.e., 35–1104 mg/kg, indicating that some new V-tolerant bacterial species might have replicated under these conditions (Xiao et al. 2017).
The wide use of metal oxide nanoparticles (MNPs) inevitably increases their environmental release into soil, which consequently raises concerns about their environmental impacts and ecological risks. The negative effects of CuO nanoparticles are stronger than that of TiO2 on soil microbes. These nanoparticles may cause significant decline in soil microbial biomass, total phospholipid fatty acids (PLFAs), and enzyme activities, i.e., urease, phosphatase, and dehydrogenase. CuO NPs reduce the composition and diversity of the paddy soil microbial community. The bioavailability of CuO is considered to induce the major toxicity to microbes in the flooded paddy soil, as elevated Cu contents in the soil extractions and the microbial cells can be expected. It may also affect soil microbes indirectly by changing nutrient bioavailability. Hence, metal oxide nanoparticles may induce perturbations on the microbes in flooded paddy soil and pose potential risks to the paddy soil ecosystem (Xu et al. 2015). Therefore, attentions toward the effects of MNPs to the ecological environment should be given.
Some metal, i.e., Pb, treatment has a stimulating effect on soil enzymatic activities and microbial biomass carbon (Cmic) at low concentration, while at higher elevations, it shows an inhibitory effect. The degree of influence on enzymatic activities and Cmic by Pb is related to clay and organic matter contents of the soils. The Pb concentration which elevated up to 500 mg/kg induces ecological risks to soil microorganisms and plants. Therefore, soil enzymatic activities can be sensitive indicators to reflect environmental stress in soil–lead–rice system (Zeng et al. 2007).
9.6 Environmental Issues
Due to the emerging environmental issues related to e-waste, there is concern about the quality of paddy soils near e-waste workshops (Wu et al. 2017). The levels of heavy metals and polychlorinated biphenyls (PCBs) and their influence on the enzyme activity and microbial community of paddy soils have been explored (Tang et al. 2014). The heavy metal and PCB pollution do not differ significantly with the sampling point distance reaching from 5 to 30 m. The highest enzyme activity has been observed for urease compared to phosphatase and catalase. The contamination stress of heavy metals and PCBs might have a slight influence on microbial activity in paddy soils. This provides the baseline studies for enzyme activities and microbial communities in paddy soil under the influence of mixed contamination.
Application of various crop-protective measures, i.e., herbicide, fungicides, pesticides, and insecticides, also affect microbial characteristics in paddy soil. Butachlor enhances the activity of dehydrogenase at increasing concentrations. The soil dehydrogenase shows the highest activity at increased optimum application of butachlor in paddy soil. The optimum dose applications of these enhance crop productivity by improving enzymatic activities of microbes in paddy soil. It also affects soil respiration depending on concentrations of content applied (Min et al. 2001). The combined effects of cadmium and herbicide butachlor on enzyme activities and microbial community structure in a paddy soil have also been studied. Urease and phosphatase activities are significantly decreased by high butachlor concentration, i.e., 100 mg/kg soil. The combined effects of Cd and butachlor on soil urease and phosphatase activities depended largely on their addition concentration ratios (Jin-Hua et al. 2009). Chlorantraniliprole (CAP) is a newly developed insecticide widely used in rice fields in China. There have been few studies evaluating the toxicological effects of CAP on soil-associated microbes. In a recent study, the similar has been observed (Wu et al. 2017). According to this, the half-lives (DT50) of CAP in soil persist for 41–53 days. During incubation of soil, it has been found that CAP do not hamper MBC activities but inhibit CO2, acid phosphatase, and sucrose invertase activities (initial 14 days of soil incubation). The effects of CAP on microbial activities (microbial biomass carbon (MBC), basal soil respiration microbial metabolic quotient CO2, acid phosphatase, and sucrose invertase activities) in the soils need to be evaluated in detail.
9.7 Effects of Fertilization
Soil microorganisms are considered a sensitive indicator of soil health and quality. In cropping systems, soil microorganisms are strongly affected by crop management, including the application of fertilizers. Water and nitrogen (N) are considered the most important factors affecting rice production and play vital roles in regulating soil microbial biomass, activity, and community. The irrigation patterns and N fertilizer levels effect soil microbial community structure and yield of paddy rice (Li et al. 2012). In natural ecosystems, increased nitrogen (N) inputs decrease microbial biomass; microorganisms in soils under upland crops often benefit from mineral fertilizer input. Paddy rice soils, being flooded for part of the season, are dominated by different carbon (C) and nitrogen (N) cycle processes and microbial communities than soils under upland crops. In paddy rice systems, the application of inorganic fertilizers increases SOC and MBC contents, both of which are important indicators of soil health. The addition of mineral fertilizer significantly increases microbial biomass carbon content (MBC) by 26% in paddy rice soils. Mineral fertilizer applications also increase soil organic carbon (SOC) content by 13%. The higher crop productivity with fertilization likely led to higher organic C inputs, which in turn increased SOC and MBC contents. In a study, it has been observed that the positive effect of mineral fertilizer on MBC content does not differ between cropping systems with continuous rice and systems where paddy rice was grown in rotation with other crops (Geisseler et al. 2017). However, compared with upland cropping systems, the increase in the microbial biomass due to mineral fertilizer application is more pronounced in rice cropping systems, even when rice is grown in rotation with an upland crop. Differences in climate and soil oxygen availability explain the stronger response of soil microorganisms to mineral fertilizer input in paddy rice systems. Fertilization does not consistently select for specific microbial groups (e.g., gram-positive or gram-negative bacteria, fungi, actinomycetes) in paddy rice systems; however, it affects microbial community composition through changes in soil properties. Fertilizer applications at a rate of 450-59-187 kg/ha/year (N-P-K) may improve crop yields, SOC contents, and SOC stability in subtropical paddy soils (Xinyu et al. 2017). Soil organic matter (SOM) stability increased because of the fertilizer applications over the past 15 years resulting in increases of the fungal C/bacterial C ratios. The contents and ratios of amino sugars can be used as indicators to evaluate the impact of mineral fertilizer applications on SOM dynamics in subtropical paddy soils. Biochar amendments with high pH and surface area might be effective to mitigate emission of both N2O and CH4 from paddy soil (Wang et al. 2017b). Similarly, Lin et al. (2016) investigated the responses of antibiotic resistance genes (ARGs) and the soil microbial community in a paddy upland rotation system to mineral fertilizer (NPK) and different application dosages of manure combined with NPK. It has been observed that NPK application slightly affects soil ARG abundances. Soil ARG abundances can be increased by manure-NPK application, but manure dosage (2250–9000 kg/ha) is highly influential to induce any change in ARG abundance. Community-level physiological profile (CLPP) analysis depicts a sharp increase in ARGs by increasing manure dosage (4500–9000 kg/ha) induced but would not change the microbial community at large. However, 9000 kg/ha manure application produced a decline in soil microbial activity. The determination of antibiotics and heavy metals in soils suggests that the observed bloom of soil ARGs might associate closely with the accumulation of copper and zinc in soil.
9.8 Conclusions
Heavy metal pollution in the paddy fields showed Cd, Cu, Ni, and Zn contaminations, while the rice remained at a safe level. The long-term application of fertilizers, pesticides, and industrial activities is the main pollution source in soil properties and an plays an important role in the availability of most heavy metals to rice plants in the paddy fields. Heavy metals in soils and rice have clear spatial patterns. Such information, combined with soil properties, play rational site-specific management in paddy fields.
References
Baath E, Díaz-Raviña M, Frostegård Å, Campbell CD (1998) Effect of metal-rich sludge amendments on the soil microbial community. Appl Environ Microbiol 64(1):238–245
Bai Y, Müller DB, Srinivas G, Garrido-Oter R, Potthoff E, Rott M, Dombrowski N, Münch PC, Spaepen S, Remus-Emsermann M, Hüttel B, McHardy AC, Vorholt JA, Schulze-Lefert P (2015) Functional overlap of the Arabidopsis leaf and root microbiota. Nature 528(7582):364–369
Bowles TM, Acosta-Martínez V, Calderón F, Jackson LE (2014) Soil enzyme activities, microbial communities, and carbon and nitrogen availability in organic agro ecosystems across an intensively-managed agricultural landscape. Soil Biol Biochem 68:252–262
Cao ZH, Huang JF, Zhang CS, Li AF (2004) Soil quality evolution after land use change from paddy soil to vegetable land. Environ Geochem Health 26(2):97–103
Chao-Rong GE, Zhang QC (2011) Microbial community structure and enzyme activities in a sequence of copper-polluted soils. Pedosphere 21(2):164–169
Chen Y, Zuo R, Zhu Q, Sun Y, Li M, Dong Y, Ru Y, Zhang H, Zheng X, Zhang Z (2014) MoLys2 is necessary for growth, conidiogenesis, lysine biosynthesis, and pathogenicity in Magnaporthe oryzae. Fungal Genet Biol 67:51–57
Chodak M, Niklińska M (2010) Effect of texture and tree species on microbial properties of mine soils. Appl Soil Ecol 46(2):268–275
Do TX (2012) Microbial communities in paddy fields in the Mekong Delta of Vietnam, vol 2012, no. 101
Geisseler D, Linquist BA, Lazicki PA (2017) Effect of fertilization on soil microorganisms in paddy rice systems–A meta-analysis. Soil Biol Biochem 15:452–460
Gregorich EG, Monreal CM, Carter MR, Angers DA, Ellert B (1994) Towards a minimum data set to assess soil organic matter quality in agricultural soils. Can J Soil Sci 74(4):367–385
Griffiths RI, Thomson BC, James P, Bell T, Bailey M, Whiteley AS (2011) The bacterial biogeography of British soils. Environ Microbiol 13(6):1642–1654
Hu XF, Jiang Y, Shu Y, Hu X, Liu L, Luo F (2014) Effects of mining wastewater discharges on heavy metal pollution and soil enzyme activity of the paddy fields. J Geochem Explor 147:139–150
Huaidong HE, Waichin LI, Riqing YU, Zhihong YE (2017) Illumine-based analysis of bulk and rhizosphere soil bacterial communities in paddy fields under mixed heavy metal contamination. Pedosphere 27(3):569–578
Jin-Hua W, Hui D, Yi-Tong L, Guo-Qing S (2009) Combined effects of cadmium and butachlor on microbial activities and community DNA in a paddy soil. Pedosphere 19(5):623–630
Keogel-Knabner I, Amelung W, Cao Z, Fiedler S, Frenzel P, Jahn R, Kalbitz K, Keolbl A, Schloter M (2010) Biogeochemistry of paddy soils. Geoderma 157:1–14
Li YT, Rouland C, Benedetti M, Li FB, Pando A, Lavelle P, Dai J (2009) Microbial biomass, enzyme and mineralization activity in relation to soil organic C, N and P turnover influenced by acid metal stress. Soil Biol Biochem 41(5):969–977
Li Y-J, Chen X, Shamsi I, Fang P, Lin X-Y (2012) Effects of irrigation patterns and nitrogen fertilization on rice yield and microbial community structure in paddy soil. Pedosphere 22(5):661–672
Li Y, Zhang W, Zheng D, Zhou Z, Yu W, Zhang L, Feng L, Liang X, Guan W, Zhou J, Chen J, Lin Z (2014) Genomic evolution of Saccharomyces cerevisiae under Chinese rice wine fermentation. Genom Biol Evol 6(9):2516–2526
Li X, Sun J, Wang H, Li X, Wang J, Zhang H (2017) Changes in the soil microbial phospholipid fatty acid profile with depth in three soil types of paddy fields in China. Geoderma 290:69–74
Lin H, Sun W, Zhang Z, Chapman SJ, Freitag TE, Fu J, Ma J (2016) Effects of manure and mineral fertilization strategies on soil antibiotic resistance gene levels and microbial community in a paddy–upland rotation system. Environ Pollut 211:332–337
Lopes AR, Faria C, Prieto-Fernández Á, Trasar-Cepeda C, Manaia CM, Nunes OC (2011) Comparative study of the microbial diversity of bulk paddy soil of two rice fields subjected to organic and conventional farming. Soil Biol Biochem 43(1):115–125
Lu L, Roberts G, Simon K, Yu J, Hudson AP (2003) A protein required for respiratory growth of Saccharomyces cerevisiae. Curr Genet 43(4):263–272
Luo X, Fu X, Yang Y, Cai P, Peng S, Chen W, Huang Q (2016) Microbial communities play important roles in modulating paddy soil fertility. Sci Rep 6
Mao TT, Yin R, Deng H (2015) Effects of copper on methane emission, methanogens and methanotrophs in the rhizosphere and bulk soil of rice paddy. Catena 133:233–240
Meharg AA, Rahman MM (2003) Arsenic contamination of Bangladesh paddy field soils: implications for rice contribution to arsenic consumption. Environ Sci Technol 37(2):229–234
Min H, Ye YF, Chen ZY, Wu WX, Yufeng D (2001) Effects of butachlor on microbial populations and enzyme activities in paddy soil. J Environ Sci Health B 36(5):581–595
Moche M, Gutknecht J, Schulz E, Langer U, Rinklebe J (2015) Monthly dynamics of microbial community structure and their controlling factors in three floodplain soils. Soil Biol Biochem 90:169–178
Nishimura S, Yonemura S, Sawamoto T, Shirato Y, Akiyama H, Sudo S, Yagi K (2008) Effect of land use change from paddy rice cultivation to upland crop cultivation on soil carbon budget of a cropland in Japan. Agric Ecosyst Environ 125(1):9–20
Pennanen T, Frostegard ASA, Fritze H, Baath E (1996) Phospholipid fatty acid composition and heavy metal tolerance of soil microbial communities along two heavy metal-polluted gradients in coniferous forests. Appl Environ Microbiol 62(2):420–428
Praeg N, Wagner AO, Illmer P (2014) Effects of fertilisation, temperature and water content on microbial properties and methane production and methane oxidation in subalpine soils. Eur J Soil Biol 65:96–106
Seklemova E, Pavlova A, Kovacheva K (2001) Biostimulation-based bioremediation of diesel fuel: field demonstration. Biodegradation 12(5):311–316
Shang H, Yang Q, Wei S, Wang J (2012) The effects of mercury and lead on microbial biomass of paddy soil from southwest of China. Procedia Environ Sci 12:468–473
Shibahara F, Inubushi K (1997) Effect of organic matter application on microbial biomass and available nutrients in various types of paddy soils. Soil Sci Plant Nutr 43:191–203
Steenwerth KL, Jackson LE, Calderón FJ, Stromberg MR, Scow KM (2002) Soil microbial community composition and land use history in cultivated and grassland ecosystems of coastal California. Soil Biol Biochem 34(11):1599–1611
Takahashi Y, Minamikawa R, Hattori KH, Kurishima K, Kihou N, Yuita K (2004) Arsenic behavior in paddy fields during the cycle of flooded and non-flooded periods. Environ Sci Technol 38:1038–1044
Tang X, Hashmi MZ, Long D, Chen L, Khan MI, Shen C (2014) Influence of heavy metals and PCBs pollution on the enzyme activity and microbial community of paddy soils around an e-waste recycling workshop. Int J Environ Res Public Health 11(3):3118–3131
Wang N, Chang ZZ, Xue XM, Yu JG, Shi XX, Ma LQ, Li HB (2017a) Biochar decreases nitrogen oxide and enhances methane emissions via altering microbial community composition of anaerobic paddy soil. Sci Total Environ 581:689–696
Wang N, Xue XM, Juhasz AL, Chang ZZ, Li HB (2017b) Biochar increases arsenic release from an anaerobic paddy soil due to enhanced microbial reduction of iron and arsenic. Environ Pollut 220:514–522
Wu W, Dong C, Wu J, Liu X, Wu Y, Chen X, Yu S (2017) Ecological effects of soil properties and metal concentrations on the composition and diversity of microbial communities associated with land use patterns in an electronic waste recycling region. Sci Total Environ 601:57–65
Xiao XY, Wang MW, Zhu HW, Guo ZH, Han XQ, Zeng P (2017) Response of soil microbial activities and microbial community structure to vanadium stress. Ecotoxicol Environ Saf 142:200–206
Xinyu Z, Juan X, Fengting Y, Wenyi D, Xiaoqin D, Yang Y, Xiaomin S (2017) Specific responses of soil microbial residue carbon to ling term applications of mineral fertilizer to reddish paddy soils. Pedosphere. doi: https://doi.org/10.1016/S1002-0160(17)60335-7
Xu P, Chen F, Mannas JP, Feldman T, Sumner LW, Roossinck MJ (2008) Virus infection improves drought tolerance. New Phytol 180(4):911–921
Xu C, Peng C, Sun L, Zhang S, Huang H, Chen Y, Shi J (2015) Distinctive effects of TiO2 and CuO nanoparticles on soil microbes and their community structures in flooded paddy soil. Soil Biol Biochem 86:24–33
Yamaguchi N, Nakamura T, Dong D, Takahashi Y, Amachi S, Makino T (2011) Arsenic release from flooded paddy soils is influenced by speciation, Eh, pH, and iron dissolution. Chemosphere 83:925–932
Yang HQ, Hao YK (2011) Main restrictive factors and countermeasures of rice industry in China. Chin Agr Sci Bull 27:351–354
Yang D, Zhang M (2014) Effects of land-use conversion from paddy field to orchard farm on soil microbial genetic diversity and community structure. Eur J Soil Biol 64:30–39
Yang J, Tang C, Wang F, Wu Y (2016) Co-contamination of Cu and Cd in paddy fields: using periphyton to entrap heavy metals. J Hazard Mater 304:150–158
Yao H, Xu J, Huang C (2003) Substrate utilization pattern, biomass and activity of microbial communities in a sequence of heavy metal polluted paddy soils. Geoderma 115:139–148
Zeng LS, Liao M, Chen CL, Huang CY (2007) Effects of lead contamination on soil enzymatic activities, microbial biomass, and rice physiological indices in soil–lead–rice (Oryza sativa L.) system. Ecotoxicol Environ Saf 67(1):67–74
Zhang YB, Cao N, Su XG, Xu XH, Yang F, Yang ZM (2009) Effects of soil and water conservation measures on soil properties in the low mountain and hill area of Jilin province. Bull Soil Water Conserv 5:0–53
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer International Publishing AG, part of Springer Nature
About this chapter
Cite this chapter
Afzaal, M., Mukhtar, S., Malik, A., Murtaza, R., Nazar, M. (2018). Paddy Soil Microbial Diversity and Enzymatic Activity in Relation to Pollution. In: Hashmi, M., Varma, A. (eds) Environmental Pollution of Paddy Soils. Soil Biology, vol 53. Springer, Cham. https://doi.org/10.1007/978-3-319-93671-0_9
Download citation
DOI: https://doi.org/10.1007/978-3-319-93671-0_9
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-93670-3
Online ISBN: 978-3-319-93671-0
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)