Abstract
Tufted angiomas (TA) and kaposiform hemangioendothelioma (KHE) are classified as benign vascular tumors with locally aggressive potential. They share many similar histologic and clinical features and are believed to be two entities that lie along a spectrum (Le et al, Am J Surg Pathol 34:1563–1573, 2010; Osio et al, Arch Dermatol 146:758–763, 2010). Within the same tumor, there are focal areas of both KHE and TA histopathologic phenotype, and longitudinal transformation from TA to KHE has been described (Chu et al, Dermatology 206:334–337, 2003). A unique feature of these tumors is their potential to develop a severe, life-threatening coagulopathy called Kasabach-Merritt phenomenon (KMP) (Croteau SE and Gupta D, Semin Cutan Med Surg 35:147–52, 2016).
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
Genetic diagnosis | No |
Genetic etiology | None known |
Level of evidence: treatment | Low |
Evidence: | Case series, expert opinion |
Diagnosis
Tufted angiomas (TA) and kaposiform hemangioendothelioma (KHE) are classified as benign vascular tumors with locally aggressive potential. They share many similar histologic and clinical features and are believed to be two entities that lie along a spectrum [1, 2]. Within the same tumor, there are focal areas of both KHE and TA histopathologic phenotype, and longitudinal transformation from TA to KHE has been described [3]. A unique feature of these tumors is their potential to develop a severe, life-threatening coagulopathy called Kasabach-Merritt phenomenon (KMP) .
TA are slow-growing vascular tumors that develop predominantly during infancy or childhood but can occur at any stage of life [4]. They tend to be smaller in size and more localized to the epidermis and dermis. They have less risk of developing KMP and long-term musculoskeletal sequelae and may spontaneously resolve [5]. KHE can infiltrate deeper structures including bone, muscle, and fascia, extend to adjacent viscera and lymph nodes, and are associated with a higher risk for KMP. Tumors may decrease in size or become dormant over time, but do not completely disappear (Fig. 9.1).
KHE of face and infratemporal fossa with skull base invasion and bone loss causing repeated bouts of meningitis. (Photos and illustrations courtesy of Seattle Children’s Vascular Anomalies Program, Jonathan A. Perkins and Eden Palmer). (Images courtesy of Seattle Children’s Radiology, Giridar Shivaram, Eric Monroe and Kevin Koo)
Clinical Features
There are four main presentations for KHE/TA: (1) fulminant KHE with KMP in the neonate/young infant, (2) large cutaneous/noncutaneous KHE/TA with KMP, (3) KHE/TA without KMP, and (4) TA with chronic coagulopathy without thrombocytopenia [2]. KMP is associated with a mortality rate as high as 30%; thus, when findings of coagulopathy are present, they should prompt immediate treatment [6,11,40,9].
TA has variable clinical presentations ranging from deep red to purple papules or plaques or an indurated vascular stain with ill-defined borders (Fig. 9.2) [10]. There can be associated hypertrichosis with increased lanugo hairs and/or hyperpigmentation present. They enlarge slowly over time and are often warm and tender to touch.
KHE typically presents as a solitary, indurated, ill-defined, red-purple plaque in infancy or early childhood. They can have a nodular appearance, be warm to touch, have associated hypertrichosis or hyperhidrosis, and can have accompanying ecchymoses and telangiectasias. They can rapidly enlarge within weeks and have recurrent episodes of engorgement, swelling, pain, and purpura [11]. Multifocal KHE lesions have been reported, especially within the bone [12], but metastatic disease has not been observed. KHE has a predilection for the extremities but also occurs on the head and neck, trunk, and groin. KHE may also present in noncutaneous locations such as the retroperitoneum, mediastinum, and bone, and there have been reports of extension into adjacent viscera from these locations. The signs and symptoms of this vascular tumor are dependent on the extent of tumor and the tissue layers involved; for example, when KHE infiltrates muscle and fascial layers, there can be reduction in range of motion with risk of contracture development [13]. Visceral organ involvement in KHE is uncommon but portends a more severe clinical course. KHE is typically diagnosed in small children, with >90% of cases being apparent within the first year of life; identification of KHE in adults is being more commonly reported. Tumors in this setting tend to be less invasive and have not been associated with thrombocytopenia or coagulopathy thus far. Even after successful treatment, some amount of mass typically remains, and there is often some residual disease manifesting as skin discoloration, fibrosis, and occasionally lymphedema [14].
Kasabach-Merritt Phenomenon
TA and KHE can potentially develop Kasabach-Merritt phenomenon (KMP) [15, 16]. KMP presents with profound thrombocytopenia thought to result from intralesional platelet trapping typically accompanied by coagulopathy as evidenced by elevation in d-dimer, slight prolongation of PT and aPTT, and reduction in fibrinogen levels. The thrombocytopenia in KMP can be profound, generally with platelet counts less than 50,000/uL. In one large series, the median platelet count reported was 11,500/uL [6].
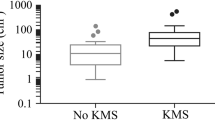
KMP is distinct from the coagulopathy that can accompany other vascular anomalies (e.g., venous malformation) that more closely resembles disseminated intravascular coagulation with a marked decrease in fibrinogen, prolongation of PT and aPTT, and a comparatively mild reduction in platelet count. KMP can arise in any patient with a lesion within the KHE/TA spectrum but is much more common with earlier onset of the tumors in the neonatal and infant setting and with KHE. Predictors of KMP include age and size and location of lesion. The proportion of infants who develop KMP is much higher than that of older children. KMP has not yet been reported in adult-onset KHE . The size and extent of the lesion has been correlated with increased risk of KMP; lesions >8 cm in greatest diameter and lesions involving muscle, bone, and the thoracic cavity are at increased risk [6, 7]. Although most patients manifest KMP at the time of presentation, 11% developed KMP later, generally within the first year after diagnosis. Changes such as, rapid lesion growth, deepening color, increasing firmness, and pain, should prompt a reevaluation for KMP.
Evaluation
The goal of TA/KHE evaluation is an accurate diagnosis and detection of KMP. This is accomplished with hematologic tests, imaging, and tissue biopsy if needed. Complete blood count will detect anemia and thrombocytopenia and a coagulation panel (PT, PTT, fibrinogen, and D-dimer) to identify a coagulopathy [9]. Imaging is important to evaluate lesion characteristics and extent. Magnetic resonance imaging (MRI) with and without gadolinium is the imaging modality of choice. MRI shows a diffusely enhancing T2 hyperintense infiltrate as well as decreased T1 signal with ill-defined margins (Fig. 9.3a, b ) [17]. There may be subcutaneous thickening, infiltrative margins into adjacent muscles and structures, and high flow vessels. Gadolinium-enhanced images demonstrate significant enhancement of the lesion. Understanding of the extent of the KHE/TA lesion will help inform treatment decision-making. CT can evaluate and characterize bone involvement, which is important in head and neck lesions, while ultrasound can demonstrate the intralesional flow characteristics (Fig. 9.1 ). Tissue biopsy is not always required for diagnosis if clinical appearance and imaging are characteristic, especially if KMP is present. Involved tissue, whether deep or superficial, has a characteristic appearance of irregular nodules with infiltrating growth leading to a dense hyaline stromal response [18]. The nodules of KHE/TA frequently coalesce and are formed by tightly packed capillaries focally accompanied by fascicles of moderately plump spindled lymphatic endothelial cells with eosinophilic-to-clear cytoplasm and bland nuclei that form elongated, slit-like lumina containing erythrocytes, reminiscent of Kaposi’s sarcoma (Fig. 9.4) [19]. Lumina containing platelet-rich microthrombi are easily found, and the spindled cells often curve around epithelioid nests that are rich in pericytes surrounding the microthrombi [18, 19]. Most tumors are full of thin-walled lymphatic vessels that stain negatively for GLUT-1 [20, 1, 18, 21]. KHE endothelial cells are positive for CD31, CD34, and FLI 1 in spindled areas as well as well canalized areas of KHE [18, 21]. Notably, the spindled endothelial cells of KHE also express lymphatic endothelial markers including podoplanin (recognized by the D240 antibody), LYVE-1, and PROX1, which is useful for histopathologic diagnosis.
Treatment
Treatment considers the clinical features of TA/KHE present, extent of involvement, presence of KMP , symptoms, and location to determine the best modality of therapy [22]. Generally accepted indications for initiating treatment include KMP, pain, or functional compromise due to an enlarging tumor. Treatment of lesions involving muscle or crossing joints may be appropriate in the hopes of reducing late complications of atrophy or contracture, although whether or not treatment reduces development of these late complications is an unresolved question.
Surgical Intervention
Complete KHE surgical resection , when possible in localized lesions, is curative. However, complete surgical resection may be impossible, especially when there is KMP and wide infiltration into surrounding structures. In cases of KHE with KMP, severe coagulopathy and associated hemorrhage may worsen with surgical resection [17]. When medical management fails to treat KHE, there may be a role for partial surgical resection or when lesions are imminently life-threatening and medical treatment response is slow [23]. Preoperative embolization may increase success of surgical resection by decreasing blood loss and preservation of normal tissues.
Radiation/Image-Guided Therapy
Intravascular embolization is used as an adjunct to surgical resection, since it does not have lasting benefit as a primary modality [17]. In severe or life-threatening cases prone to hemorrhage, arterial embolization may serve a temporizing role until medical therapy becomes effective. Radiation therapy has no role in TA/KHE treatment.
Pharmacologic Treatment
There are no prospective studies to guide TA/KHE medical management.
Corticosteroids and Vincristine
If KHE is not accompanied by KMP, then corticosteroids alone (oral prednisolone 2 mg/kg/day or the IV equivalent) are appropriate as starting therapy [9]. Corticosteroid monotherapy is not recommended for KMP [24]. Consensus guidelines recommend corticosteroids combined with vincristine infusion 0.05 mg/kg/week for KMP [25, 26, 9, 27]. Vincristine has been reported to be effective in improving KMP and reducing tumor size [25, 28]. The average time to hematologic response is 7.6 weeks (standard deviation of 5.2 weeks) [28]. Treatment duration is based on individual response, with a goal to wean steroids after 3–4 weeks and stop vincristine after 20–24 weeks. Other combination regimens incorporating vincristine for KHE management include vincristine with aspirin and ticlopidine [29] and vincristine with sirolimus [30]. Combination therapy attempts to reduce corticosteroid side effects. Common side effects associated with vincristine include constipation, peripheral neuropathy, and SIADH.
Sirolimus
Sirolimus has recently gained recognition for its potential efficacy in a wide range of vascular anomalies [31]. A phase 2 study of sirolimus for complicated vascular anomalies enrolled 13 patients with KHE, 10 of who had KMP [32]. Patients received sirolimus as a single drug at a starting dose of 0.8 mg/m2 q12 h, and dosing was adjusted to maintain a trough of 10–15 ng/ml. Patients could continue treatment for 12 months on study; continued therapy was at the discretion of the treating physician. All patients with KMP had a partial response at 6 months, defined as a greater than 20% reduction in size of the target vascular lesion by imaging, or improvement in target organ dysfunction by at least one grade, or improvement in quality of life instruments; one patient with KHE without KMP had progressive disease. Additional case reports suggest efficacy of sirolimus and other mTOR inhibitors in KHE, including correction of KMP, reduction of tumor size, and improvement of fibrosis [22,27,35]. Note that infants less than 3 months of age may require lower sirolimus doses to achieve target levels [36, 37]. Sirolimus is currently being compared to standard therapy with vincristine and corticosteroids in a prospective clinical trial (clinical trials.gov NCT00975819), which would be the first study of its kind for patients with vascular anomalies. In practice many specialists use combination therapy with sirolimus, vincristine, and nonsteroidal anti-inflammatory drugs, and lower-dose sirolimus may be effective [30]. Common side effects noted with sirolimus include hematologic toxicities, diarrhea, elevated blood lipids, and infection [32]. Limited data are available regarding long-term effects of this medication.
Propranolol
Propranolol , a beta-blocker, has revolutionized the therapy of infantile hemangiomas, but its role in other vascular anomalies is unclear. Chiu et al. reported a case series of 11 patients with KMP related to KHE/TA who were treated with propranolol; 36% responded, though specific response criteria were not defined [38]. Improvement was relatively slow, and therefore propranolol would not be appropriate for patients with aggressive disease or KMP. Based on a survey of expert opinion, propranolol 2–5 mg/kg/day may be considered for patients without KMP or as adjunctive therapy in patients who do not have a satisfactory response to first-line agents [27].
Interferon-Alpha
Interferon-alpha has demonstrated activity in KHE, but enthusiasm for this treatment has declined due to the risk of spastic diplegia in young infants [39]. Wu et al. reviewed outcomes of 12 children with KHE between the ages of 20 days and 8 months of age who were treated with interferon-alpha for 3–9 months. Initial dosage was 1 × 10^6 U/m2/day for the first week then 3 × 10^6 U/M2/day thereafter. Five patients had KMP and most of the patients had failed prior therapies. All patients with KMP achieved normalization of their platelet counts, and 75% of patients had at regression in the size of their tumors by at least 80%. The time to response to interferon-alpha treatment was 10 days to 5 weeks (mean 3.6 weeks). No severe adverse effects were observed, and no patient developed spastic diplegia [40]. Interferon-alpha remains an option for patients with disease refractory to other treatments, but families should be informed of the warnings associated with its use.
Aspirin
Aspirin is a rational therapy that is postulated to reduce intralesional platelet trapping and inflammation; however, data regarding its efficacy are mixed. Case reports exist in which the use of aspirin, either alone or in combination with ticlopidine, is associated with reduction of painful coagulopathy in tufted angioma [29, 41, 2]. In addition, low-dose aspirin (5 mg/kg/day) was reported to reduce tumor bulk and pain in two patients with tufted angioma who did not have Kasabach-Merritt phenomenon [42, 43]. Aspirin in conjunction with ticlopidine and vincristine is used by some in the treatment of KHE with KMP [29]. The most recent consensus guidelines suggest that aspirin 2–5 mg/kg/day can be considered as adjunctive therapy for KHE without KMP [9].
Supportive Care for Coagulopathy in Kasabach-Merritt Phenomenon
The primary management is directed at tumor treatment. Platelet transfusions should be reserved for clinically important bleeding or surgery, as the half-life of the transfused platelets is short and transfusions are associated with increased platelet trapping within the lesion and resultant pain and tumor growth [16, 9]. Conversely, administration of fresh frozen plasma or cryoprecipitate is recommended for fibrinogen <100 mg/dl and may help reduce bleeding symptoms. Antifibrinolytic agents have been used with variable efficacy [44], though the majority of experts only recommend use of these agents for bleeding not responsive to other therapy [9, 27]. Heparin and antiplatelet agents have not been shown to reduce platelet trapping and are not recommended [27].
References
Le Huu AR, Jokinen CH, Rubin BP, Mihm MC, Weiss SW, North PE, Dadras SS. Expression of prox1, lymphatic endothelial nuclear transcription factor, in Kaposiform hemangioendothelioma and tufted angioma. Am J Surg Pathol. 2010;34:1563–73.
Osio A, Fraitag S, Hadj-Rabia S, Bodemer C, de Prost Y, Hamel-Teillac D. Clinical spectrum of tufted angiomas in childhood: a report of 13 cases and a review of the literature. Arch Dermatol. 2010;146(7):758–63.
Chu CY, Hsiao CH, Chiu HC. Transformation between Kaposiform hemangioendothelioma and tufted angioma. Dermatology. 2003;206:334–7.
Jones EW, Orkin M. Tufted angioma (angioblastoma). A benign progressive angioma, not to be confused with Kaposi’s sarcoma or low-grade angiosarcoma. J Am Acad Dermatol. 1989;20(2 Pt 1):214–25.
Browning J, Frieden I, Baselga E, Wagner A, Metry D. Congenital, self regressing tufted angioma. Arch Dermatol. 2006;142:749–51.
Croteau SE, Liang MG, Kozakewich HP, Alomari AI, Fishman SJ, Mulliken JB, Trenor CC 3rd. Kaposiform hemangioendothelioma: atypical features and risks of Kasabach-Merritt phenomenon in 107 referrals. J Pediatr. 2013;162:142–7.
Gruman A, Liang MG, Mulliken JB, Fishman SJ, Burrows PE, Kozakewich HP, Blei F, Frieden IJ. Kaposiform hemangioendothelioma without Kasabach-Merritt phenomenon. J Am Acad Dermatol. 2005;52(4):616–22.
**Croteau SE, Gupta D. The clinical spectrum of kaposiform hemangioendothelioma and tufted angioma. Semin Cutan Med Surg. 2016;35:147–52.
***Drolet BA, Trenor CC 3rd, Brandao LR, Chiu YE, Chun RH, Dasgupta R, Garzon MC, Hammill AM, Johnson CM, Tlougan B, Blei F, David M, Elluru R, Frieden IJ, Friedlander SF, Iacobas I, Jensen JN, King DM, Lee MT, Nelson S, Patel M, Pope E, Powell J, Seefeldt M, Siegel DH, Kelly M, Adams DM. Consensus-derived practice standards plan for complicated Kaposiform hemangioendothelioma. J Pediatr. 2013;163:285–91.
Herron MD, et al. Tufted angiomas: variability of clinical morphology. Pediatr Dermatol. 2002;19:394–401.
Zukerberg LR, Nickoloff BJ, Weiss SW. Kaposiform hemangioendothelioma of infancy and childhood. An aggressive neoplasm associated with Kasabach-Merritt syndrome and lymphangiomatosis. Am J Surg Pathol. 1993;17(4):321–8.
Deraedt K, Vander Poorten V, Van Geet C, Renard M, De Wever I, Sciot R. Multifocal kaposiform haemangioendothelioma. Virchows Arch. 2006;448(6):843–6. Epub 2006 Apr 5.
Enjolras O, Mulliken JB, Wassef M, Frieden IJ, Rieu PN, Burrows PE, Salhi A, Léauté-Labreze C, Kozakewich HP. Residual lesions after Kasabach-Merritt phenomenon in 41 patients. J Am Acad Dermatol. 2000;42(2 Pt 1):225–35.
Hammill A, Mobberley-Schuman P, Adams D. Lymphoedema is a potential sequela of kaposiform hemangioendothelioma. Br J Dermatol. 2016;175(4):833–4.
Kelly M. Kasabach-Merritt phenomenon. Pediatr Clin N Am. 2010;57(5):1085–9.
Phillips WG, Marsden JR. Kasabach-Merritt syndrome exacerbated by platelet transfusion. J R Soc Med. 1993;86(4):231–2.
Szabo S, North PE. The histopathology and pathogenesis of vascular anomalies. In:Vascular tumors and developmental malformations: pathogenic mechanisms and molecular diagnosis. Molecular and translational medicine book series. New York: Humana Press (Springer); 2016. ISSN 2197-7860.
Lyons LL, North PE, Mac-Moune Lai F, Stoler MH, Folpe AL, Weiss SW. Kaposiform hemangioendothelioma: a study of 33 cases emphasizing its pathologic, immunophenotypic, and biologic uniqueness from juvenile hemangioma. Am J Surg Pathol. 2004;28:559–68.
North PE, Waner M, Buckmiller L, James CA, Mihm MC. Vascular tumors of infancy and childhood: beyond capillary hemangioma. Cardiovasc Pathol. 2006;15:303–17.
Arai E, Kuramochi A, Tsuchida T, Tsuneyoshi M, Kage M, Fukunaga M, Ito T, Tada T, Izumi M, Shimizu K, Hirose T, Shimizu M. Usefulness of D2-40 immunohistochemistry for differentiation between kaposiform hemangioendothelioma and tufted angioma. J Cutan Pathol. 2006;33:492–7.
North PE, Mizeracki A, Waner M, Mihm MC. GLUT1: a newly discovered marker for juvenile hemangiomas. Hum Pathol. 2000;31(1):11–22.
*Blei F, Karp N, Rofsky N, Rosen R, Greco MA. Successful multimodal therapy for kaposiform hemangioendothelioma complicated by Kasabach-Merritt phenomenon: case report and review of the literature. Pediatr Hematol Oncol. 1998;15:295–305.
**Drolet BA, Scott LA, Esterly NB, Gosain AK. Early surgical intervention in a patient with Kasabach-Merritt phenomenon. J Pediatr. 2001;138:756–8.
Yasui N, Koh K, et al. Kasabach-Merritt phenomenon: a report of 11 cases from a single institution. J Pediatr Hematol Oncol. 2013;35:554–8.
Haisley-Royster C, Enjolras O, Frieden IJ, Garzon M, Lee M, Oranje A, de Laat PC, Madern GC, Gonzalez F, Frangoul H, Le Moine P, Prose NS, Adams DM. Kasabach-merritt phenomenon: a retrospective study of treatment with vincristine. J Pediatr Hematol Oncol. 2002;24(6):459–62.
Wang Z, Li K, Yao W, et al. Steroid resistant Kaposiform Hemangioendothelioma: a retrospective study of 37 patients treated with vincristine and long-term follow up. Pediatr Blood Cancer. 2015;62:577–80.
***Tlougan BE, Lee MT, Drolet BA, Frieden IJ, Adams DM, Garzon MC. Medical management of tumors associated with Kasabach-Merritt phenomenon: an expert survey. J Pediatr Hematol Oncol. 2013;35(8):618–22.
Wang P, Zhou W, Tao L, Zhao N, Chen XW. Clinical analysis of Kasabach-Merritt syndrome in 17 neonates. BMC Pediatr. 2014;14:146. https://doi.org/10.1186/1471-2431-14-146.
Fernandez-Pineda I, Lopez-Gutierrez JC, Ramirez G, Marquez C. Vincristine-ticlopidine-aspirin: an effective therapy in children with Kasabach-Merritt phenomenon associated with vascular tumors. Pediatr Hematol Oncol. 2010;27:641–5.
Schaefer BA, Wang D, Merrow AC, Dickie BH, Adams DM. Long-term outcome for kaposiform hemangioendothelioma: a report of two cases. Pediatr Blood Cancer. 2017;64(2):284–6. https://doi.org/10.1002/pbc.26224. Epub 2016 Oct 4.
Hammill AM, Wentzel M, Gupta A, Nelson S, Lucky A, Elluru R, Dasgupta R, Azizkhan RG, Adams DM. Sirolimus for the treatment of complicated vascular anomalies in children. Pediatr Blood Cancer. 2011;57:1018–24.
**Adams DM, Trenor CC 3rd, Hammill AM, Vinks AA, Patel MN, Chaudry G, Wentzel MS, Mobberley-Schuman PS, Campbell LM, Brookbank C, Gupta A, Chute C, Eile J, McKenna J, Merrow AC, Fei L, Hornung L, Seid M, Dasgupta AR, Dickie BH, Elluru RG, Lucky AW, Weiss B, Azizkhan RG. Efficacy and safety of sirolimus in the treatment of complicated vascular anomalies. Pediatrics. 2016;137:1–10.
Matsumoto H, Ozeki M, Hori T, Kanda K, Kawamoto N, Nagano A, Azuma E, Miyazaki T, Fukao T. Successful Everolimus Treatment of Kaposiform Hemangioendothelioma With Kasabach-Merritt Phenomenon: clinical efficacy and adverse effects of mTOR inhibitor therapy. J Pediatr Hematol Oncol. 2016;38(8):e322–5.
Oza VS, Mamlouk MD, Hess CP, Mathes EF, Frieden IJ. Role of Sirolimus in advanced Kaposiform Hemangioendothelioma. Pediatr Dermatol. 2016;33(2):e88–92. https://doi.org/10.1111/pde.12787. Epub 2016 Feb 11.
Uno T, Ito S, Nakazawa A, Miyazaki O, Mori T, Terashima K. Successful treatment of Kaposiform hemangioendothelioma with everolimus. Pediatr Blood Cancer. 2015;62(3):536–8. https://doi.org/10.1002/pbc.25241. Epub 2014 Oct 12.
Mizuno T, Emoto C, Fukuda T, Mobberley-Schuman P, Hammel A, Adams D, Vinks A. Developmental pharmacokinetics of sirolimus; implications for dosing in neonates and infants with vascular anomalies. ISVAA abstract 106. 2016.
Shimano K, LongBoyle J, Mathes E, Meyer A, Rosbe K, Dowd C, Cooke D, Frieden I. Neonates require lower sirolimus doses to achieve therapeutic levels for treatment of vascular anomalies. ISVAA abstract 361. 2016.
Chiu YE, Drolet BA, Blei F, Carcao M, Fangusaro J, Kelly ME, Krol A, Lofgren S, Mancini AJ, Metry DW, Recht M, Silverman RA, Tom WL, Pope E. Variable response to propranolol treatment of kaposiform hemangioendothelioma, tufted angioma, and Kasabach-Merritt phenomenon. Pediatr Blood Cancer. 2012;59:934–8.
Barlow CF, Priebe CJ, Mulliken JB, Barnes PD, Mac Donald D, Folkman J, Ezekowitz RA. Spastic diplegia as a complication of interferon Alfa-2a treatment of hemangiomas of infancy. J Pediatr. 1998;132:527–30.
Wu HW, Wang X, Zhang L, et al. Interferon-alpha therapy for refractory kaposiform hemangioendothelioma: a single center experience. Sci Rep. 2016;6:36261.
Leaute-labreze C, et al. Tufted angioma associated with platelet trapping syndrome: response to aspirin. Arch Dermatol. 1997;133:1077–9.
DeKlotz CM, Langevin KK, Dohil M. On response of tufted angiomas to low-dose aspirin. Pediatr Dermatol. 2014;31:123–4.
**Javvaji S, Frieden IJ. Response of tufted angiomas to low-dose aspirin. Pediatr Dermatol. 2013;30:124–7.
Hanna BD, Bernstein M. Tranexamic acid in the treatment of Kasabach-Merritt phenomenon. Am J Pediatr Hematol Oncol. 1989;11(2):191–5.
To assist the reader in gaining familiarity with available evidence, the following rating system has been used to indicate key references for each chapter’s content:
***: Critical material. Anyone dealing with this condition should be familiar with this reference.
**: Useful material. Important information that is valuable in in clinical or scientific practice related to this condition.
*: Optional material. For readers with a strong interest in the chapter content or a desire to study it in greater depth.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer International Publishing AG, part of Springer Nature
About this chapter
Cite this chapter
Gupta, D., Geddis, A., Chun, R. (2018). Tufted Angioma and Kaposiform Hemangioendothelioma. In: Perkins, J., Balakrishnan, K. (eds) Evidence-Based Management of Head and Neck Vascular Anomalies. Springer, Cham. https://doi.org/10.1007/978-3-319-92306-2_9
Download citation
DOI: https://doi.org/10.1007/978-3-319-92306-2_9
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-92305-5
Online ISBN: 978-3-319-92306-2
eBook Packages: MedicineMedicine (R0)







