Abstract
Animal models are an important tool to study the pathophysiology of Machado-Joseph Disease (MJD). So far, animal models using simple organisms (like the round worm Caenorhabditis elegans or the fruit fly drosophila) but also mammalian models (mouse and even a non-human primate model) have been generated to study MJD. While simple organisms made an important contribution to the identification of pathophysiological mechanisms in MJD and were further used for modifier and screening purposes, mammalian models recapitulate major disease features of MJD in humans and are therefore a highly valuable tool for e.g. the validation of mechanisms or for pre-clinical validation of treatment approaches. Here we give an overview about the strategies which were used to model MJD and about the different models generated so far. We further highlight advantages of specific model organisms and describe the new findings which were made employing these animal models of MJD.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
15.1 Introduction
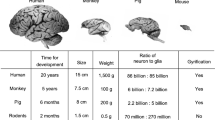
Animal models are indispensable especially for a late-onset disease like Machado-Joseph Disease (MJD), as they allow analysing pathogenic mechanisms and aspects of the disease which obviously cannot be studied in human MJD patients. For example, histopathological analyses of post-mortem human brain samples can be for obvious reasons only conducted at the end stage of a disease and therefore cannot reveal details about different disease stages. Sections and brain samples over the pathophysiological course of MJD can therefore only be gathered from animal models. In addition to this, animal models allow the assessment of disease modifiers or treatment approaches. Most commonly used model organisms contain an endogenous orthologue of the human ATXN3 gene (Table 15.1).
No naturally occurring animal models of MJD or expansions of the endogenous polyglutamine repeats beyond a disease-causing threshold have been reported in animals yet [1]. Animal models of MJD were therefore either generated by transfection with a viral construct, classical transgenic approaches or by the modification (knock-in) of the endogenous ATXN3 orthologue via the integration of an expanded polyglutamine repeat with surrounding sequence. Transgenic lines (in mice, flies, worms and even non-human primates) comprise of either the full-length ATXN3 cDNA, a fragment of it or even a large genomic YAC construct containing major parts of the ATXN3 locus.
15.2 Non-mammalian Models of MJD
Simple organisms, like the roundworm (Caenorhabditis elegans) or the fruit fly (Drosophila melanogaster) appear on the first sight not that much suitable to model a neurodegenerative disorder, like MJD. However during the recent years, a number of highly relevant studies were performed employing either C. elegans or Drosophila m. thereby demonstrating their high suitability for research in MJD.
15.2.1 Caenorhabditis elegans (Roundworm) Models of MJD
The round worm (Caenorhabditis elegans) is especially suitable to serve as disease model due to its size (about 1 mm in length), short generation time (about 4 days), and especially its transparency. This allows an easy visualization (and counting) of living (or degenerating) cells in vivo. C. elegans are in most cases self-fertilizing hermaphrodites or (rarely) males. It is known that C. elegans hermaphrodites consists of about 1000 cells in total from which 302 are neurons (divided into 118 distinct classes) and 56 are glial cell [2]. C. elegans is considered to be a “good compromise between complexity of vertebrates… and extreme simplicity of yeast” [2].
About 65% of human disease genes have homologs in C. elegans [3] including ATXN3. The endogenous worm ortholog of human ATXN3 (called atx-3) consists of four exons and does not contain a CAG repeat [4]. A detailed characterization of the C. elegans orthologue of ATXN3 revealed ubiquitously expression starting in late embryonic development and retaining during adulthood. Strong expression can be detected in the spermatheca (i.e. the organ that produces sperm in hermaphrodites) and vulval muscle as well as in neuronal dorsal and ventral cord and neurons of the head and tail [4]. As human ATXN3, C. elegans atx-3 possesses deubiquitinase activity in vitro against polyubiquitin chains with a minimum length of four ubiquitin residues. The knock-out of the gene does not induce an obvious phenotype in worms under normal growth conditions but lead to differential expression of genes involved in cell structural/motility, signal transduction and the ubiquitin proteasome system [4]. Known interactions in humans (e.g. between ATXN3 and p 97/VCP), likewise occur in C. elegans (i.e. between atx-3 and the C. elegans homologs of p 97/VCP namely CDC-48.1 and CDC-48.2) [5]. Interestingly, under challenging growth conditions atx-3 knock-out worms proved to have an enhanced stress response and display a better survival and tolerance against heat shock conditions possibly explained by the (pre-)activation of molecular chaperones upon the loss of atx-3 (dependent on DAF-16) and thereby a more effective activation of this process upon heat shock [6].
In order to induce aggregation and an MJD-like phenotype in worms, transgenic constructs (under control of the pan-neuronal unc-119 promoter) with either an extremely long polyglutamine repeat (130Q) within full-length ATXN3 or a C-terminal fragment of ATXN3 (aa284 ff with at least 63Q) are required. Full-length ATXN3 with 91Q or less, however, did not aggregate ([7], Table 15.2).
C-terminal fragments of ATXN3 induces neuronal dysfunction and behavioural symptoms (including uncoordinated locomotion and egg laying defects), interrupts synaptic transmission and causes morphological abnormalities of neuronal processes [7]. The fact that C-terminal fragments of ATXN3 induce a stronger phenotype than full-length ATXN3 has likewise previously been observed in mice [12]. Worms expressing full-length ATXN3 with an extremely long repeat (130Q) likewise developed movement deficits however not before reaching mid-age.
In another study, full-length ATXN3 with 130 glutamine repeats was able to induce an earlier phenotype in worms [8] including aggregation foci, lethargy and slightly reduced lifespan. This may be either due to the use of a different promoter (F25B3.3 promoter instead of unc-119) or a different isoform of ATXN3: While Khan et al. [7] employed the 1a short isoform of ATXN3 [10] (containing two ubiquitin -interacting motifs), Teixeira-Castro et al. [8] used the 3c isoform of ATXN3 (MJD1-1, [11]) harbouring an additional, third Ubiquitin -interacting motif.
As already observed in the study mentioned before, worms expressing a C-terminal fragment of ATXN3 exhibit a more severe phenotype than worms expressing the full-length protein. Motor dysfunction was highly associated with the aggregation of ATXN3: Likewise, the C-terminal fragment of ATXN3 was more aggregation prone than the full-length protein i.e. 75 glutamines induced aggregation in a C-terminal fragment of ATXN3 only but not in the full-length protein. Interestingly, aggregation of expanded ATXN3 occurred in a neuron-subtype-specific pattern rather than randomly indicating that the sequence surrounding the polyglutamine repeat confers a kind of cell-type specificity on ATXN3’s toxicity [8]. Furthermore, it was shown that the insulin/insulin growth-factor-1 (IGF-1)-like signalling pathway facilitated by heat-shock factor-1 (HSF-1) together with aging modify the proteotoxicity of expanded ATXN3 [8]. This age-dependency seems to be neuron-specific as the aggregation and toxicity of a comparable C-terminal fragment of ATXN3 turned out not to be age-dependent when expressed in the C. elegans body wall muscle cells instead of neurons [9]. A special strength of C. elegans as a small model organism is its suitability for screening procedures. While large genetic screens for disease modifiers of MJD have been conducted in drosophila (see below), C. elegans models of MJD turned out to be especially useful for an effective screening of compound libraries and validation of small molecules alleviating locomotive defects induced by mutant ATXN3. Using this approach, the serotonin reuptake inhibitor citalopram was identified and later even validated in a transgenic mouse model for MJD [13].
15.2.2 Drosophila melanogaster (Fly) Models of MJD
The fruit fly (Drosophila melanogaster) is a small organisms of about three mm in length. One invaluable advantage of using Drosophila m. as model organism is the availability of fly stocks allowing practically an infinite number of genetic manipulations (e.g. overexpression or downregulation of genes using transgenic flies or flies containing genomic duplications or deletions). The availability of suitable promoter (driver) lines, allow in addition goal-directed targeting of the expression of the gene of interest.
Although about 75% of all human disease genes have related sequences in drosophila [14], the fruit fly does not contain a natural orthologue of ATXN3. The lack of an endogenous copy of the ATXN3 gene excludes any knock-in and knock-out approaches in flies and may question the suitability of flies to model MJD as relevant target or binding partners of ATXN3 may be missing as well. Nevertheless, drosophila models expressing either a C-terminal fragment [15] or full-length [16] expanded human ATXN3 mimic some features of the disease in humans including intranuclear inclusions and neuronal degeneration. So far only flies expressing the short variant of the 1a isoform (containing a premature stop codon) are available and no flies expressing the 3c isoform (Table 15.3). The stability of the CAG repeat within a transgenic ATXN3 depends on the activity of the CREB-binding protein (CBP) in flies [17].
A severe and progressive neurodegeneration with adult onset is induced upon expression of ATXN3 (either in the flies’ eye or on the nervous system) with an expanded polyglutamine repeat (Q78 or Q84) only but not with a normal repeat (Q27) [15, 16]. On the contrary, co-expression of normal ATXN3 (Q27) delayed or even protected against the toxicity induced by either full-length or C-terminal expanded ATXN3 (Q78 or Q84) and normal ATXN3 further protected against the toxicity of expanded huntingtin and ataxin-1 [16]. This protective effect of normal ATXN3 depended on both its Ubiquitin -interactive motifs (UIM) and its deubiquitinase activity [16]. Interestingly, full-length ATXN3 even with an expanded repeat turned out to alleviate the toxicity of the C-terminal fragment of expanded ATXN3 [16]. In order to prevent the generation of ATXN3 fragments in vivo, Jung et al. [19] generated flies expressing full-length ATXN3 with six mutated putative Caspase cleavage sites (D171/208/217/223/225/228 N). The six mutations indeed prevented the cleavage of ATXN3 and mitigated its neurotoxicity, but without influencing its aggregation and formation of neuronal intranuclear inclusion [19].
Noteworthy, expanded C-terminal ATXN3 induce the formation of aggregates and behavioral changes not only when expressed in neuronal cells but comparable effects were likewise be observed upon its expression in glial cells [18]. In the search for factors alleviating the phenotype in flies, it was observed in a candidate-gene-based approach that the toxicity of ATXN3 is diminished by the co-expression of chaperones, like HSP70 [20, 21]. Large genome-wide screens further revealed that several chaperones and components of the ubiquitin pathway alleviated the phenotype of MJD flies induced by expanded ATXN3 by reducing its aggregation and thereby its toxicity [22]. Additional modifiers identified are implicated in nuclear export and transcription [22]. Interestingly, some of these modifiers not only impacted the toxicity of expanded ATXN3 but of mutant Tau as well. This finding was later confirmed in an independent modifier screen [23] and indicates their relevance for a broader range of neurodegenerative disorders beyond MJD [22].
The identification of another modifying pathway in drosophila models of MJD shed light on a novel component of the pathophysiology in MJD: The upregulation of muscleblind (mbl), a modifier of CUG repeat RNA toxicity, increased the toxicity of expanded ATXN3 thereby indicating that the CAG expansion within ATXN3 does not only lead to toxicity mediated by the (translated) polyglutamine repeat but by the (transcribed) CUG mRNA as well [24]. Importantly, the authors further demonstrated that an ATXN3 transgene loses partly its toxicity if a pure CAG repeat is interrupted by CAA although both CAG and CAA encode for glutamine and therefore the identical protein is encoded. Muscleblind seems to “act on the RNA to enhance polyQ toxicity” thereby accelerating the aggregation of ATXN3 [24].
ATXN2 is another highly interesting modifier identified in flies, as it causes (in its expanded form) another type of Spinocerebellar Ataxia: SCA2. The affected protein in SCA2, ATXN2, enhances in its normal state (when upregulated) the toxicity of ATXN3 and likewise mitigates (when downregulated) the neurodegeneration induced by expanded ATXN3 [25].
15.3 Mammalian (Mouse) Models of MJD
To learn more about the physiological function of ATXN3 in mammals, knock-out mice were generated by targeted mutagenesis [26]. ATXN3 knock-out mice had normal viability and fertility and showed no gross locomotor abnormalities, but contained a higher level of ubiquitinated proteins, underlining a deubiquitinating function of ATXN3 [26]. Attempting to create a MJD knock-in model, Switonski et al. [27] generated a MJD knock-out model due to alternative splicing events. Also these MJD knock-out mice were viable, fertile with no reduced life span or neurological abnormalities, but showed no higher amounts of ubiquitinated proteins [27].
The first mouse model to study MJD pathology was generated by targeting the expression of full-length human ATXN3 containing an expanded polyglutamine stretch (79Q) to Purkinje cells (L7 promoter, [12], Table 15.4). Up to an observation time of 23 weeks, these mice did not demonstrate any phenotype. Mice expressing a C-terminal ATXN3/polyglutamine fragment of 35 repeats (Q35C) in Purkinje cells as well did not develop a phenotype. Only when the polyglutamine repeat number was extended to 79, mice developed an ataxic phenotype starting at four weeks of age with wide-based hindlimb stance and falling down when moving [12]. Neuropathologically, Q79C mice presented with cerebellar atrophy with all three layers affected by neuronal cell loss [12]. Ikeda et al. concluded that the truncated ATXN3 protein is more potent in inducing Purkinje cell pathology than the full-length protein [12].
In order to generate a model including all regulatory elements, Cemal et al. [28] used a yeast artificial chromosome (YAC) spanning 250 kb of the human ATXN3 locus. Several different lines were established with 15 CAG repeats as control line (MJD15) and between 64 and 84 repeats resembling MJD disease lines (MJD64-84). Transgene expression levels equaled that of endogenous mouse ATXN3. Both the 1a long and the 3c isoforms of ATXN3 [11] are expressed in these mice, while 3c is the predominant isoform detected on protein level [57]. The observed behavioral phenotype of affected mice included wide gait, lowered pelvis, tremor, hypoactivity, body clasping and loss of weight. Neuropathological analyses revealed cell loss in several brain regions also primarily affected in humans like pons, deep cerebellar nuclei and cerebellum . Transgenic ATXN3 is predominately localized in the nucleus and accumulates in neuronal nuclear inclusion bodies (NIIs) in MJD disease lines. Severity of disease symptoms was increased with higher copy number and repeat length [28]. Dantrolene, a stabilizer of intracellular calcium signaling, was able to improve the motor coordinative performance of MJD84.2 mice and reduced neuronal cell loss in this model [29]. Using this mouse line it was also shown that Purkinje cell dysfunction in these mice is associated with alterations in the physiology of voltage-activated potassium channels. The administration of SKA-31, an activator of calcium-activated potassium channels, was able to correct Purkinje cell firing and improve motor function of SCA3/MJD84.2 mice [31]. A RNA interference approach using microRNA was able to reduce soluble levels as well as abnormal neuronal intranuclear accumulation of mutant ATXN3 protein in MJD84.2 mice [30].
Goti et al. [32] generated MJD transgenic mice using full length human ATXN3 cDNA containing either 20 CAG repeats for control lines (Q20) or 71 CAG repeats for disease lines (Q71). Transgene expression was controlled by the prion protein promoter from mouse. Starting at the age of 2 months homozygous Q71 mice developed a behavioral phenotype with tremor, ataxic limbs, abnormal posture (hunchback with low pelvic elevation and muscle wasting), impaired grip strength of fore- and hindlimbs, impaired rotarod performance, deteriorating footprint pattern and body weight loss. ATXN3 immunostaining was enriched in nuclei in several brain regions both in Q71 heterozygous and homozygous mice, but only the latter presented with large intranuclear inclusions . Furthermore the authors were able to detect a C-terminal ATXN3 cleavage fragment with high abundance in phenotypic Q71 mice and in affected brain regions of MJD patients [32]. Heterozygous Q71 mice bred poorly and homozygous mice were infertile due to gonadal failure secondary to a neuroendocrine dysfunction [58]. The generation of a mouse model lacking amino acids 190-220 (deltaQ71) narrowed a putative proteolytic cleavage site to a region N-terminal of amino acids 190 within ATXN3 [33]. Genetic reduction of the co-chaperone and ubiquitin ligase CHIP (C-terminus of Hsp70-interacting protein) in Q71 mice resulted in an increase of ATXN3 microaggregates and worsening of the neurological phenotype leading the authors to suggest that enhancing CHIP activity could be therapeutically beneficial in MJD [59].
In 2007, Bichelmeier and colleagues [34] not only demonstrated that the severity of a MJD phenotype increases with increasing CAG repeat number, but also that nuclear localization of mutant ATXN3 is a prerequisite for the development of symptoms : Transgenic mice expressing full-length ATXN3 with 70 CAG repeats under the control of the murine prion protein promoter (70Q) developed a strong neurological phenotype with tremor, wide based hind limbs, reduced activity, body weight and grooming. Purkinje cells in this model appeared shrunken and with an increased electron density. Expanding the CAG repeat size to 148 (148Q), mice showed comparable symptoms earlier in life leading to premature death already at three months of age. Transgenic mice in which ATXN3 with 148 CAG repeats was kept in the nucleus by attaching a nuclear localization signal (148Q.NLS) were even more severely and earlier affected. On the other hand, keeping ATXN3 out of the nucleus using a nuclear export signal (148Q.NES) led to only very mild or even missing behavioral symptoms . Moreover, inclusion bodies abundant in several brain regions in transgenic mice with 70, 148 and 148 polyglutamines with NLS were highly diminished in the 148Q.NES model [34]. Pharmaceutical upregulation of autophagy resulted in decrease of soluble mutant ATXN3 and number of inclusion bodies as well as in an improvement of rotarod performance in 70Q mice [35].
Another model using the mouse prion protein promoter and an ATXN3 construct with 22 and 79 polyglutamines (ataxin-3 -Q22 and ataxin-3 -Q79), respectively was generated by Chou et al. [36]. Transgenic ataxin-3 -Q79 mice developed a progressive neurological phenotype with forelimb clasping, impaired motor coordination, reduced locomotor activity and ataxic wide-based gait. Symptoms deteriorated with age leading to reduced pelvic elevation and hunch back posture at around one year of age. Purkinje cells exhibited morphological characteristics of degeneration. Intranuclear inclusion bodies were observed in neurons of the dentate nucleus , pontine nucleus and substantia nigra [36]. The authors identified transcriptional dysregulation of genes involved in glutamatergic neurotransmission, intracellular calcium signaling, MAP kinase pathways, regulators of neuronal survival or differentiation of GABAA/B receptor subunits and heat shock response in ataxin-3 -Q79 mice before ataxic symptoms became evident. These findings led to hypothesize that the ataxin-3 -Q79 transgene causes transcriptional downregulation by the induction of hypoacetylation and that the HDAC inhibitor sodium butyrate is able to reverse this transcriptional repression and thereby alleviates ataxic symptoms observed in ataxin-3 -Q79 mice. In follow up studies the authors were able to proof the efficacy of sodium butyrate in reversing transcriptional downregulation in the cerebellum of ataxin-3 -Q79 mice. Furthermore, the onset and severity of ataxic symptoms in these mice were delayed and ameliorated, respectively [37, 39]. Based on upregulated mRNA expression profiles of different apoptotic pathways, the authors also tested a second hypothesis: Ataxin-3 -Q79 mice were treated with a pro-apototic gene inhibitor leading to significantly ameliorated neuronal cell death in pontine nuclei [38]. In a third attempt, the same authors therapeutically targeted the ubiquitin -proteasome system. Transgenic mice were treated with an adenosine A2A receptor agonist leading to significantly increased chymotrypsin-like activity of the proteasome. This treatment resulted in reduced protein level of mutant ATXN3 and alleviated neuronal cell death and ataxic symptoms in treated ataxin-3 -Q79 mice [40].
To test whether a newly identified guanosine triphosphatase (CRAG) has the potential to be therapeutically effective in the treatment of polyglutamine diseases Torashima et al. [45] generated a new mouse model. In these mice the Purkinje-cell-specific promoter L7 controlled the expression of a truncated form of human ATXN3 starting at amino acid 287 with 69 glutamines (ataxin-3 [Q69]). Transgenic mice developed an ataxic phenotype with early onset, reduced weight, impaired rotarod performance and severe cerebellar atrophy. Inclusion bodies were present in the cytoplasm or extracellular of Purkinje cells [45]. Lentiviral vector-mediated expression of CRAG in Purkinje cells of this model led to clearance of inclusions and rescue of the ataxic phenotype [45]. Konno et al. [43] further analyzed this model and another study proved that Neuropeptide Y was able to alleviate motor coordinative abnormalities and neuropathological parameters in these mice [42].
The first conditional mouse model for MJD was generated by Boy et al. in 2009 [47]. Mice were generated employing the Tet-off system [60]. A promoter mouse line containing the prion protein promoter from hamster was cross bred to a responder mouse line expressing ATXN3 with 77 CAG repeats . In double transgenic mice containing both transgene constructs (Prp/MJD77) a factor (tTA) transcribed by the promoter construct is able to bind to the responder construct, thereby activating the expression of transgenic ATXN3. In the presence of tetracycline, this antibiotic binds to the tTA factor and thus inhibits its binding to the responder. In this way the expression of mutant ATXN3 can be turned off. Double transgenic Prp/MJD77 mice developed a progressive neurological phenotype characterized by deficits in motor coordination and balance, hyperactivity, reduced anxiety and reduced gain in body weight. ATXN3 positive intranuclear inclusion bodies were detected in brains of double transgenic mice and Purkinje cells presented with higher electron density indicating dark cell degeneration. Turning the expression of mutant ATXN3 off resulted in a complete reversal of the observed rotarod impairment as well as the reduced body weight gain proving that MJD symptoms although already manifested can be reversed [47]. Employing this model the suitability of riluzole as treatment for MJD has been assessed [46].
In 2010, Boy et al. [48] published a late onset MJD mouse model, using an ATXN3 construct with 148 CAG repeats under the control of a fragment of the huntingtin promoter (HDPromMJD148). Mutant ATXN3 is ubiquitiously expressed throughout the whole brain. Several main characteristics of the MJD disease in humans, like the late onset as well as intergenerational CAG repeat instability, are resembled in this model. Rotarod impairment as sign of decline of motor coordination occurs at around one year of age, whereas inclusion bodies are not detectable before 18 months of age, stressing that neuronal dysfunction is independent from the formation of inclusions . Electron microscopical analysis revealed that Purkinje cells , also in this MJD model, show dark cell degeneration [48].
Using the CMV promoter to control the expression of an ATXN3 construct with 83 and 94 CAG repeats , respectively, was the strategy of Silva-Fernandes et al. [49] to generate two more mouse models for MJD. CMVMJD94 mice presented with CAG repeat instability, neuronal dysfunction and impairment of motor function, whereas no phenotype was detectable in the CMVMJD83 model [49]. Using the CMVMJD94 model mitochondrial DNA damage and its suitability as a biomarker of initial stages of the SCA3 disease was dissected [61, 62]. Silva-Fernanades et al. also generated a third model in which the CAG stretch was expanded to 135 repeats (CMVMJD135) [53]. In these mice main characteristics of the human MJD disease, like intergenerational repeat length variation, progressive neurological deficits, reduced gain of body weight and formation of intranuclear inclusion bodies in different brain regions are recapitulated [53]. Hsp90 inhibition by administration of 17-DMAG resulted in reduced levels of ATXN3 protein and lower numbers of intranuclear inclusions in CMVMJD135 mice [53]. Using these mice the efficacy of valproic acid and citalopram as treatment for MJD was assessed [52, 13].
In 2011, Hübener et al. [54] reported on a MJD mouse model generated by gene trap integration, leading to an ATXN3 fusion protein containing 259 N-terminal amino acids, but lacking the C-terminal polyglutamine stretch and other regulatory regions. Initially ataxin-3 (gt/gt) mice develop without gross abnormalities. However, with 9 months of age a severe deterioration with impaired motor coordination and body weight loss was observed, leading to premature death with about 12 months of age. Phenotypic mice presented with extranuclear inclusion bodies and neuronal cell death of Purkinje cells and cells of the molecular layer of the cerebellum . Authors concluded that the N-terminal region of ATXN3 can contribute to MJD symptoms [54].
Knock-in technology presents a method to generate a model which expresses the mutant gene under control of the endogenous promoter in the proper genomic context including all regulatory elements. 2015 two different knock-in mouse models for MJD were published [56, 55]. In the first model a CAG repeat expansion was inserted into the murine ATXN3 locus by homologous recombination generating heterozygous Atxn3Q82/Q6 and homozygous Atxn3Q82/Q82 mice. Early accumulation of ATXN3 protein in intranuclear puncta as well as large neuritic inclusions in older mice and altered splicing of mutant ATXN3 were characteristics observed in this knock-in model. However, behavioral deficits or neuropathological changes are missing in these mice, leading the authors to promote this model as being especially suitable for analyses of pathogenic mechanisms preceding neuronal dysfunction or cell loss [56].
The second humanized knock-in model was generated by exchanging exons 7 through 11 of the mouse ATXN3 locus with human exon 7 through 11 containing 91 CAG repeats . This resulted in intergenerational CAG repeat instability, loss of Purkinje cells and impairment of motor coordination with late onset in Ki91 mice [55].
Injection of lentiviral vectors encoding transgenic ATXN3 constructs presents an alternative, fast and cost effective approach to generate MJD models. First studies using this technique involved rats as a MJD model organism [63, 64]. Lentiviral vectors expressing mutant ATXN3 constructs were injected in different brain regions led to behavioral deficits, neuronal cell loss and inclusion body formation [63]. Delivery of allele-specific siRNA via lentiviral vectors was able to silence mutant ATXN3 expression and decreased neuropathological abnormalities [64, 65, 44]. Targeting the mouse cerebellum with lentiviral-mediated expression of mutant ATXN3 resulted in the development of a behavioral phenotype, intranuclear inclusions and neuronal cell death abnormalities [66].
15.4 Non-human Primate Model of MJD
Very recently, the first transgenic non-human primate i.e. marmoset model of MJD has been generated [67]. Transgenic marmosets were generated using a lentiviral approach based on a construct containing 120 CAG interrupted by CAA (coding for glutamine as well) every 30 repeats within the full-length ATXN3 cDNA (3c isoform). However, studies employing non-human primates are expensive and time-consuming and only three monkeys of the founder generation reaching a certain expression level of the transgene (likely due to multiple insertions of the transgene in different chromosomes) developed symptoms and could be analysed yet. This model displays protein aggregates , neurodegeneration and degeneration of skeletal muscles as well as varying neurological symptoms (including grip strength deficits and reduced body weight gain) within 3–4 months of age. Although transmission of the transgene was confirmed, a stable transgenic line has not been generated yet, possibly complicated by mosaicism in the founder marmoset and integration into multiple and different chromosomes [67].
References
Matos CA, de Macedo-Ribeiro S, Carvalho AL (2011) Polyglutamine diseases: the special case of ataxin-3 and Machado-Joseph disease. Prog Neurobiol 95(1):26–48
Bessa C, Maciel P, Rodrigues AJ (2013) Using C. elegans to decipher the cellular and molecular mechanisms underlying neurodevelopmental disorders. Mol Neurobiol 48(3):465–489
Sonnhammer EL, Durbin R (1997) Analysis of protein domain families in Caenorhabditis elegans. Genomics 46(2):200–216
Rodrigues AJ, Coppola G, Santos C, Costa Mdo C, Ailion M, Sequeiros J, Geschwind DH, Maciel P (2007) Functional genomics and biochemical characterization of the C. elegans orthologue of the Machado-Joseph disease protein ataxin-3. FASEB J 21(4):1126–1136
Rodrigues AJ, Neves-Carvalho A, Ferro A, Rokka A, Corthals G, Logarinho E, Maciel P (2009) ATX-3, CDC-48 and UBXN-5: a new trimolecular complex in Caenorhabditis elegans. Biochem Biophys Res Commun 386(4):575–581
Rodrigues AJ, Neves-Carvalho A, Teixeira-Castro A, Rokka A, Corthals G, Logarinho E, Maciel P (2011) Absence of ataxin-3 leads to enhanced stress response in C. elegans. PLoS One 6(4):e18512
Khan LA, Bauer PO, Miyazaki H, Lindenberg KS, Landwehrmeyer BG, Nukina N (2006) Expanded polyglutamines impair synaptic transmission and ubiquitin-proteasome system in Caenorhabditis elegans. J Neurochem 98(2):576–587
Teixeira-Castro A, Ailion M, Jalles A, Brignull HR, Vilaça JL, Dias N, Rodrigues P, Oliveira JF, Neves-Carvalho A, Morimoto RI, Maciel P (2011) Neuron-specific proteotoxicity of mutant ataxin-3 in C. elegans: rescue by the DAF-16 and HSF-1 pathways. Hum Mol Genet 20(15):2996–3009
Christie NT, Lee AL, Fay HG, Gray AA, Kikis EA (2014) Novel polyglutamine model uncouples proteotoxicity from aging. PLoS One 9(5):e96835
Kawaguchi Y, Okamoto T, Taniwaki M, Aizawa M, Inoue M, Katayama S, Kawakami H, Nakamura S, Nishimura M, Akiguchi I, Kimura J, Narumiya S, Kakizuka A (1994) CAG expansions in a novel gene for Machado-Joseph disease at chromosome 14q32.1. Nat Genet 8(3):221–228
Goto J, Watanabe M, Ichikawa Y, Yee SB, Ihara N, Endo K, Igarashi S, Takiyama Y, Gaspar C, Maciel P, Tsuji S, Rouleau GA, Kanazawa I (1997) Machado-Joseph disease gene products carrying different carboxyl termini. Neurosci Res 28(4):373–377
Ikeda H, Yamaguchi M, Sugai S, Aze Y, Narumiya S, Kakizuka A (1996) Expanded polyglutamine in the Machado-Joseph disease protein induces cell death in vitro and in vivo. Nat Genet 13(2):196–202
Teixeira-Castro A, Jalles A, Esteves S, Kang S, da Silva Santos L, Silva-Fernandes A, Neto MF, Brielmann RM, Bessa C, Duarte-Silva S, Miranda A, Oliveira S, Neves-Carvalho A, Bessa J, Summavielle T, Silverman RB, Oliveira P, Morimoto RI, Maciel P (2015) Serotonergic signalling suppresses ataxin 3 aggregation and neurotoxicity in animal models of Machado-Joseph disease. Brain 138(Pt 11):3221–3237
Bier E (2005) Drosophila, the golden bug, emerges as a tool for human genetics. Nat Rev Genet 6:9–23
Warrick JM, Paulson HL, Gray-Board GL, Bui QT, Fischbeck KH, Pittman RN, Bonini NM (1998) Expanded polyglutamine protein forms nuclear inclusions and causes neural degeneration in Drosophila. Cell 93(6):939–949
Warrick JM, Morabito LM, Bilen J, Gordesky-Gold B, Faust LZ, Paulson HL, Bonini NM (2005) Ataxin-3 suppresses polyglutamine neurodegeneration in Drosophila by a ubiquitin-associated mechanism. Mol Cell 18(1):37–48
Jung J, Bonini N (2007) CREB-binding protein modulates repeat instability in a Drosophila model for polyQ disease. Science 315(5820):1857–1859
Kretzschmar D, Tschäpe J, Bettencourt Da Cruz A, Asan E, Poeck B, Strauss R, Pflugfelder GO (2005) Glial and neuronal expression of polyglutamine proteins induce behavioral changes and aggregate formation in Drosophila. Glia 49(1):59–72
Jung J, Xu K, Lessing D, Bonini NM (2009) Preventing Ataxin-3 protein cleavage mitigates degeneration in a Drosophila model of SCA3. Hum Mol Genet 18(24):4843–4852
Chan HY, Warrick JM, Gray-Board GL, Paulson HL, Bonini NM (2000) Mechanisms of chaperone suppression of polyglutamine disease: selectivity, synergy and modulation of protein solubility in Drosophila. Hum Mol Genet 9(19):2811–2820
Warrick JM, Chan HY, Gray-Board GL, Chai Y, Paulson HL, Bonini NM (1999) Suppression of polyglutamine-mediated neurodegeneration in Drosophila by the molecular chaperone HSP70. Nat Genet 23(4):425–428
Bilen J, Bonini NM (2007) Genome-wide screen for modifiers of ataxin-3 neurodegeneration in Drosophila. PLoS Genet 3(10):1950–1964
Voßfeldt H, Butzlaff M, PrüSSing K, Ní Chárthaigh RA, Karsten P, Lankes A, Hamm S, Simons M, Adryan B, Schulz JB, Voigt A (2012) Large-scale screen for modifiers of ataxin-3-derived polyglutamine-induced toxicity in Drosophila. PLoS One 7(11):e47452
Li LB, Yu Z, Teng X, Bonini NM (2008) RNA toxicity is a component of ataxin-3 degeneration in Drosophila. Nature 453(7198):1107–1111
Lessing D, Bonini NM (2008) Polyglutamine genes interact to modulate the severity and progression of neurodegeneration in Drosophila. PLoS Biol 6(2):e29
Schmitt I, Linden M, Khazneh H, Evert BO, Breuer P, Klockgether T, Wuellner U (2007) Inactivation of the mouse Atxn3 (ataxin-3) gene increases protein ubiquitination. Biochem Biophys Res Commun 362(3):734–739
Switonski PM, Fiszer A, Kazmierska K, Kurpisz M, Krzyzosiak WJ, Figiel M (2011) Mouse ataxin-3 functional knock-out model. Neuromolecular Med 13(1):54–65
Cemal CK, Carroll CJ, Lawrence L, Lowrie MB, Ruddle P, Al-Mahdawi S, King RH, Pook MA, Huxley C, Chamberlain S (2002) YAC transgenic mice carrying pathological alleles of the MJD1 locus exhibit a mild and slowly progressive cerebellar deficit. Hum Mol Genet 11(9):1075–1094
Chen X, Tang TS, Tu H, Nelson O, Pook M, Hammer R, Nukina N, Bezprozvanny I (2008) Deranged calcium signaling and neurodegeneration in spinocerebellar ataxia type 3. J Neurosci 28(48):12713–12724
Rodríguez-Lebrón E, Costa MD, Luna-Cancalon K, Peron TM, Fischer S, Boudreau RL, Davidson BL, Paulson HL (2013) Silencing mutant ATXN3 expression resolves molecular phenotypes in SCA3 transgenic mice. Mol Ther 21(10):1909–1918
Shakkottai VG, do Carmo Costa M, Dell’Orco JM, Sankaranarayanan A, Wulff H, Paulson HL (2011) Early changes in cerebellar physiology accompany motor dysfunction in the polyglutamine disease spinocerebellar ataxia type 3. J Neurosci 31(36):13002–13014
Goti D, Katzen SM, Mez J, Kurtis N, Kiluk J, Ben-Haïem L, Jenkins NA, Copeland NG, Kakizuka A, Sharp AH, Ross CA, Mouton PR, Colomer V (2004) A mutant ataxin-3 putative-cleavage fragment in brains of Machado-Joseph disease patients and transgenic mice is cytotoxic above a critical concentration. J Neurosci 24(45):10266–10279
Colomer Gould VF, Goti D, Pearce D, Gonzalez GA, Gao H, Bermudez de Leon M, Jenkins NA, Copeland NG, Ross CA, Brown DR (2007) A mutant ataxin-3 fragment results from processing at a site N-terminal to amino acid 190 in brain of Machado-Joseph disease-like transgenic mice. Neurobiol Dis 27(3):362–369
Bichelmeier U, Schmidt T, Hübener J, Boy J, Rüttiger L, Häbig K, Poths S, Bonin M, Knipper M, Schmidt WJ, Wilbertz J, Wolburg H, Laccone F, Riess O (2007) Nuclear localization of ataxin-3 is required for the manifestation of symptoms in SCA3: in vivo evidence. J Neurosci 27(28):7418–7428
Menzies FM, Huebener J, Renna M, Bonin M, Riess O, Rubinsztein DC (2010) Autophagy induction reduces mutant ataxin-3 levels and toxicity in a mouse model of spinocerebellar ataxia type 3. Brain 133(Pt 1):93–104
Chou AH, Yeh TH, Ouyang P, Chen YL, Chen SY, Wang HL (2008) Polyglutamine-expanded ataxin-3 causes cerebellar dysfunction of SCA3 transgenic mice by inducing transcriptional dysregulation. Neurobiol Dis 31(1):89–101
Chou AH, Chen SY, Yeh TH, Weng YH, Wang HL (2011) HDAC inhibitor sodium butyrate reverses transcriptional downregulation and ameliorates ataxic symptoms in a transgenic mouse model of SCA3. Neurobiol Dis 41(2):481–488
Chou AH, Lin AC, Hong KY, Hu SH, Chen YL, Chen JY, Wang HL (2011) p53 activation mediates polyglutamine-expanded ataxin-3 upregulation of Bax expression in cerebellar and pontine nuclei neurons. Neurochem Int 58(2):145–152
Chou AH, Chen YL, Hu SH, Chang YM, Wang HL (2014) Polyglutamine-expanded ataxin-3 impairs long-term depression in Purkinje neurons of SCA3 transgenic mouse by inhibiting HAT and impairing histone acetylation. Brain Res 1583:220–229
Chou AH, Chen YL, Chiu CC, Yuan SJ, Weng YH, Yeh TH, Lin YL, Fang JM, Wang HL (2015) T1-11 and JMF1907 ameliorate polyglutamine-expanded ataxin-3-induced neurodegeneration, transcriptional dysregulation and ataxic symptom in the SCA3 transgenic mouse. Neuropharmacology 99:308–317
Wang HL, Hu SH, Chou AH, Wang SS, Weng YH, Yeh TH (2013) H1152 promotes the degradation of polyglutamine-expanded ataxin-3 or ataxin-7 independently of its ROCK-inhibiting effect and ameliorates mutant ataxin-3-induced neurodegeneration in the SCA3 transgenic mouse. Neuropharmacology 70:1–11
Duarte-Neves J, Gonçalves N, Cunha-Santos J, Simões AT, den Dunnen WF, Hirai H, Kügler S, Cavadas C, Pereira de Almeida L (2015) Neuropeptide Y mitigates neuropathology and motor deficits in mouse models of Machado-Joseph disease. Hum Mol Genet 24(19):5451–5463
Konno A, Shuvaev AN, Miyake N, Miyake K, Iizuka A, Matsuura S, Huda F, Nakamura K, Yanagi S, Shimada T, Hirai H (2014) Mutant ataxin-3 with an abnormally expanded polyglutamine chain disrupts dendritic development and metabotropic glutamate receptor signaling in mouse cerebellar Purkinje cells. Cerebellum 13(1):29–41
Nóbrega C, Nascimento-Ferreira I, Onofre I, Albuquerque D, Hirai H, Déglon N, de Almeida LP (2013) Silencing mutant ataxin-3 rescues motor deficits and neuropathology in Machado-Joseph disease transgenic mice. PLoS One 8(1):e52396
Torashima T, Koyama C, Iizuka A, Mitsumura K, Takayama K, Yanagi S, Oue M, Yamaguchi H, Hirai H (2008) Lentivector-mediated rescue from cerebellar ataxia in a mouse model of spinocerebellar ataxia. EMBO Rep 9(4):393–399
Schmidt J, Schmidt T, Golla M, Lehmann L, Weber JJ, Hübener-Schmid J, Riess O (2016) In vivo assessment of riluzole as a potential therapeutic drug for spinocerebellar ataxia type 3. J Neurochem 138(1):150–162
Boy J, Schmidt T, Wolburg H, Mack A, Nuber S, Böttcher M, Schmitt I, Holzmann C, Zimmermann F, Servadio A, Riess O (2009) Reversibility of symptoms in a conditional mouse model of spinocerebellar ataxia type 3. Hum Mol Genet 18(22):4282–4295
Boy J, Schmidt T, Schumann U, Grasshoff U, Unser S, Holzmann C, Schmitt I, Karl T, Laccone F, Wolburg H, Ibrahim S, Riess O (2010) A transgenic mouse model of spinocerebellar ataxia type 3 resembling late disease onset and gender-specific instability of CAG repeats. Neurobiol Dis 37(2):284–293
Silva-Fernandes A, Costa Mdo C, Duarte-Silva S, Oliveira P, Botelho CM, Martins L, Mariz JA, Ferreira T, Ribeiro F, Correia-Neves M, Costa C, Maciel P (2010) Motor uncoordination and neuropathology in a transgenic mouse model of Machado-Joseph disease lacking intranuclear inclusions and ataxin-3 cleavage products. Neurobiol Dis 40(1):163–176
Duarte-Silva S, Neves-Carvalho A, Soares-Cunha C, Teixeira-Castro A, Oliveira P, Silva-Fernandes A, Maciel P (2014) Lithium chloride therapy fails to improve motor function in a transgenic mouse model of Machado-Joseph disease. Cerebellum 13(6):713–727
Duarte-Silva S, Silva-Fernandes A, Neves-Carvalho A, Soares-Cunha C, Teixeira-Castro A, Maciel P (2016) Combined therapy with m-TOR-dependent and -independent autophagy inducers causes neurotoxicity in a mouse model of Machado-Joseph disease. Neuroscience 313:162–173
Esteves S, Duarte-Silva S, Naia L, Neves-Carvalho A, Teixeira-Castro A, Rego AC, Silva-Fernandes A, Maciel P (2015) Limited effect of chronic valproic acid treatment in a mouse model of Machado-Joseph Disease. PLoS One 10(10):e0141610
Silva-Fernandes A, Duarte-Silva S, Neves-Carvalho A, Amorim M, Soares-Cunha C, Oliveira P, Thirstrup K, Teixeira-Castro A, Maciel P (2014) Chronic treatment with 17-DMAG improves balance and coordination in a new mouse model of Machado-Joseph disease. Neurotherapeutics 11(2):433–449
Hübener J, Vauti F, Funke C, Wolburg H, Ye Y, Schmidt T, Wolburg-Buchholz K, Schmitt I, Gardyan A, Driessen S, Arnold HH, Nguyen HP, Riess O (2011) N-terminal ataxin-3 causes neurological symptoms with inclusions, endoplasmic reticulum stress and ribosomal dislocation. Brain 134(Pt 7):1925–1942
Switonski PM, Szlachcic WJ, Krzyzosiak WJ, Figiel M (2015) A new humanized ataxin-3 knock-in mouse model combines the genetic features, pathogenesis of neurons and glia and late disease onset of SCA3/MJD. Neurobiol Dis 73:174–188
Ramani B, Harris GM, Huang R, Seki T, Murphy GG, Costa Mdo C, Fischer S, Saunders TL, Xia G, McEachin RC, Paulson HL (2015) A knockin mouse model of spinocerebellar ataxia type 3 exhibits prominent aggregate pathology and aberrant splicing of the disease gene transcript. Hum Mol Genet 24(5):1211–1224
Harris GM, Dodelzon K, Gong L, Gonzalez-Alegre P, Paulson HL (2010) Splice isoforms of the polyglutamine disease protein ataxin-3 exhibit similar enzymatic yet different aggregation properties. PLoS One 5(10):e13695
Colomer Gould VF, Goti D, Kiluk J (2006) A neuroendocrine dysfunction, not testicular mutant ataxin-3 cleavage fragment or aggregate, causes cell death in testes of transgenic mice. Cell Death Differ 13(3):524–526
Williams AJ, Knutson TM, Colomer Gould VF, Paulson HL (2009) In vivo suppression of polyglutamine neurotoxicity by C-terminus of Hsp70-interacting protein (CHIP) supports an aggregation model of pathogenesis. Neurobiol Dis 33(3):342–353
Gossen M, Bujard H (1992) Tight control of gene expression in mammalian cells by tetracycline-responsive promoters. Proc Natl Acad Sci U S A 89(12):5547–5551
Kazachkova N, Raposo M, Montiel R, Cymbron T, Bettencourt C, Silva-Fernandes A, Silva S, Maciel P, Lima M (2013) Patterns of mitochondrial DNA damage in blood and brain tissues of a transgenic mouse model of Machado-Joseph disease. Neurodegener Dis 11(4):206–214
Ramos A, Kazachkova N, Silva F, Maciel P, Silva-Fernandes A, Duarte-Silva S, Santos C, Lima M (2015) Differential mtDNA damage patterns in a transgenic mouse model of Machado-Joseph disease (MJD/SCA3). J Mol Neurosci 55(2):449–453
Alves S, Régulier E, Nascimento-Ferreira I, Hassig R, Dufour N, Koeppen A, Carvalho AL, Simões S, de Lima MC, Brouillet E, Gould VC, Déglon N, de Almeida LP (2008) Striatal and nigral pathology in a lentiviral rat model of Machado-Joseph disease. Hum Mol Genet 17(14):2071–2083
Alves S, Nascimento-Ferreira I, Auregan G, Hassig R, Dufour N, Brouillet E, Pedroso de Lima MC, Hantraye P, Pereira de Almeida L, Déglon N (2008) Allele-specific RNA silencing of mutant ataxin-3 mediates neuroprotection in a rat model of Machado-Joseph disease. PLoS One 3(10):e3341
Alves S, Nascimento-Ferreira I, Dufour N, Hassig R, Auregan G, Nóbrega C, Brouillet E, Hantraye P, Pedroso de Lima MC, Déglon N, de Almeida LP (2010) Silencing ataxin-3 mitigates degeneration in a rat model of Machado-Joseph disease: no role for wild-type ataxin-3? Hum Mol Genet 19(12):2380–2394
Nóbrega C, Nascimento-Ferreira I, Onofre I, Albuquerque D, Conceição M, Déglon N, de Almeida LP (2013) Overexpression of mutant ataxin-3 in mouse cerebellum induces ataxia and cerebellar neuropathology. Cerebellum 12(4):441–455
Tomioka I, Ishibashi H, Minakawa EN, Motohashi HH, Takayama O, Saito Y, Popiel HA, Puentes S, Owari K, Nakatani T, Nogami N, Yamamoto K, Noguchi S, Yonekawa T, Tanaka Y, Fujita N, Suzuki H, Kikuchi H, Aizawa S, Nagano S, Yamada D, Nishino I, Ichinohe N, Wada K, Kohsaka S, Nagai Y, Seki K (2017) Transgenic monkey model of the polyglutamine diseases recapitulating progressive neurological symptoms. eNeuro 4(2). pii: ENEURO.0250-16.2017
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer International Publishing AG
About this chapter
Cite this chapter
Schmidt, J., Schmidt, T. (2018). Animal Models of Machado-Joseph Disease. In: Nóbrega, C., Pereira de Almeida, L. (eds) Polyglutamine Disorders. Advances in Experimental Medicine and Biology, vol 1049. Springer, Cham. https://doi.org/10.1007/978-3-319-71779-1_15
Download citation
DOI: https://doi.org/10.1007/978-3-319-71779-1_15
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-71778-4
Online ISBN: 978-3-319-71779-1
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)