Abstract
Deep venous reflux, isolated or associated with deep venous obstruction, is an important cause of severe chronic venous insufficiency. The most frequent etiology of deep venous reflux is valve incompetence due to post-thrombotic syndrome (secondary reflux); primary reflux is less frequent and congenital is rare.
Deep venous reconstructive surgery is usually recommended for those cases not responding to conservative therapies. A precise patient selection and treatment strategy are essential. Different surgical techniques and outcomes are correlated to different etiologies. Deep venous surgery is safe and able to improve considerably the quality of life in patients affected by severe chronic venous insufficiency.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
- Chronic venous insufficiency
- Deep venous reflux
- Deep venous surgery
- Valve reconstruction
- Valvuloplasty
- Neovalve
- Post-thrombotic syndrome
- Venous obstruction
-
1.
Current surgical techniques for deep venous reconstruction are valvuloplasty, valve transposition, valve transplantation, and neovalve creation
-
2.
Deep venous obstruction should be corrected before valve reconstruction
-
3.
Neovalve creation seems to provide better valve competence after long-term follow-up compared to the other techniques for valvular reconstruction
Introduction
Deep venous reflux (DVR) is one of the main causes of chronic venous insufficiency (CVI) [1, 2]. It may be isolated or associated with other pathologies, such as deep venous obstruction ; or else associated to superficial venous reflux; or reflux of the perforators [3, 4]. CVI is the consequence of venous hypertension during ambulation; it is related to high volume/low velocity blood flow, which leads to microcirculatory disorders [5]. DVR tolerance varies according to the caliber of the veins and the overall venous volume, the efficiency of the muscle pump, the age and physical activity of the patient [6,7,8,9,10]. DVR may be axial or segmental [11]; CVI is more frequently correlated to the presence of axial reflux [12].
Various reflux patterns were defined at the VEIN TERM Consensus Conference [13]. Reflux is one of several determining elements in CVI [14], which also includes associated obstruction. CVI chiefly occurs in PTS , where reflux and obstruction are associated in two thirds of cases [15]. When not associated to obstruction or reflux in other districts, DVR is nevertheless a significant factor in CVI [16].
Correcting DVR must observe the principle of ensuring adequate blood flow , bearing in mind that the latter increases substantially during muscular activity. Past attempts to correct reflux, which employed ligation of the popliteal and femoral veins [17, 18], had a high failure rate because of the formation of a collateral network without valves. Failure was also reported [19] where banding was applied to reduce the vein caliber but was correlated to an increase in flow resistance. Hence, the best options would appear to be valve repair, where possible, the creation of a new valve, or a strategy to obtain a new venous axis with competent valves.
Deep Venous Reflux
On the basis of etiology, DVR can be distinguished into primary, secondary, or congenital [20,21,22]. Secondary reflux is by far the most frequent in that it includes post-thrombotic syndrome (PTS) valve insufficiency . Deep vein thrombosis leads to the destruction of the valve function in 40–70% of cases [23]. If rapid resolution of the thrombus occurs, the valve function may remain intact; the valve itself may present limited malfunction, and the vein wall at the thrombus site may develop simple thickening (Fig. 17.1). In other scenarios, valve destruction may be segmental; however, the valve apparatus, at sites distal or proximal, may be preserved. When the thrombotic process is not rapidly resolved and has been more extensive, valve destruction will be total and associated with axial reflux.
The term primary reflux is identified on CEAP classification [20] as a malfunction of the valve due to unknown causes, so such as to preserve the valve apparatus in a quasi-unaltered condition .
This malfunction may be due to malformations such as asymmetrical cusps or redundant leaflets which may be a congenital condition; alternatively, the malfunction might be linked to a small unrecognized venous thrombosis, thus making it in reality a secondary reflux.
Despite the fact that a congenital condition may be the cause of the malfunction, the CEAP classification currently reserves the term “congenital” to the extremely rare conditions of venous aplasia or hypoplasia [20, 22, 23]. The distinction between primary, congenital, and secondary, leaving aside etiology, in all events proves crucial since both techniques and outcomes vary widely when treating these three type of reflux [24].
Surgical Treatments
Surgical treatment of deep venous reflux essentially consists of two types of procedures: valvuloplasty or the creation of a non-refluxing segment.
Surgical Treatments in Primary DVR
Internal Valvuloplasty
As mentioned above, valvuloplasty is feasible only where the valve apparatus is intact or sufficiently preserved following the thrombotic episode. In such cases the valve malfunction presents a reflux of varying magnitude generally associated with the prolapse of one or both the free leaflets or their asymmetry or the widening of the valve annulus. The first surgical operation to correct an insufficiently functional valve was performed by Kistner in 1968 [25] and consists in stretching the leaflets, thus reducing the redundancy and the length of the free border of the leaflet itself (Fig. 17.2a, b). This first technique was subsequently modified by other authors who suggested various approaches in order to avoid direct damage to the valve apparatus during phlebotomy [26,27,28].
External Valvuloplasty
In order to avoid phlebotomy, external valvuloplasty was also proposed to establish valve competence by reducing the commissural angle with the application of external stitches. This technique presupposes perfect visibility of the site at which the cusps are inserted into the vein wall, but still implies the risk of damaging the valve apparatus by stretching it to excess [29,30,31]. The lower degree of technical precision is also associated with less satisfactory outcomes.
In view of the fact that the valve leaflets are often asymmetrical, direct frontal vision offered by phlebotomy prompts the choice of this technique. Valvuloplasty should also restore valve competence by maintaining the sail effects in the valve sinus, since the latter proves critical in ensuring correct valve function [32].
Surgical Treatments in Secondary and Congenital DVR
The surgical techniques that address secondary DVR are: vein transposition, vein transplant, neovalve, and artificial venous valve.
Vein Transposition
Where anatomical conditions permit, a devalvulated segment is relocated onto a competent valvulated segment. Transposition is normally performed at the inguinal level and was first described by Kistner in 1979 [33]. The most frequently performed version involves transposing the valvulated segment onto the deep femoral vein (DFV) or onto the great saphenous vein (GSV) .
Transposition onto the DFV: Technical Details
When transposing onto the DFV , the surgeon should isolate the femoral junction in the first tract of the common femoral vein (CFV). The tract should be long enough to allow control and should extend distally toward the femoral vein (FV) and the DFV.
The most proximal and competent valve in the DFV must be identified and the FV isolated for a tract long enough to enable us to transpose the segment without creating twists and tension.
The FV is divided at the proximal insertion sited at CFV level. The residual stump is closed off with a longitudinal suture. Thereafter the femoral vein is sutured at DFV level, downstream of a previously identified competent valve.
Any trabeculae in the FV due to post-thrombotic processes should be removed. Given that the two calibers are normally incompatible, an end-to-side anastomosis is more often performed (Fig. 17.3); however, for hemodynamic reasons, where possible an end-to-end anastomosis is preferable.
DFV is a multiaxial system, which explains why hemodynamic alterations are not caused by the descending branch interruption .
Transposition onto the Saphenous Vein: Technical Details
If a competent GSV be available, the transposition of the FV onto the GSV can be performed below the sapheno-femoral junction. In view of the fact that the GSV is located at subcutaneous level, it is better to transpose the GSV itself into a subfascial location. A segment 5–10 cm long should be harvested from the proximal GSV. The FV divides just below the junction with the DFV. An end-to-end anastomosis is performed between the GSV and the FV. The FV is often distended as a result of clamping and any discrepancy in caliber will quickly diminish after restoring flow. Still, the increased blood flow through the GSV may cause dilatation and precipitate valve insufficiency. Such a scenario can be avoided by applying a cuff below the competent saphenous valve, thus preventing postoperative dilatation.
The advantages of transposition are the relative technical ease of performance when a competent GSV is available and good long-term results. On the other hand, the disadvantages are the caliber mismatch between FV and GSV or DFV; adverse anatomical conditions in DFV. Further drawbacks may be related to the competent valve being only present in distal part of DFV, requiring extensive dissection. As mentioned prior, subsequent incompetence of the DFV and GSV due to the increase in caliber is another possible problem. Finally the risk of postoperative lymphocele which can be a difficult complication to treat.
Vein Transplant
Transplant aims at inserting a segment containing a competent valve inside an incompetent axis. In the first technique described by Raju in 1979 [34], the donor segment is the axillary vein (AV) ; in the version proposed by Taheri in 1982 [35], it is the brachial vein.
Technical Details
The main drawback of this technique may be incompetence in the donor segment and discrepancy in caliber between the two veins. Access to the AV can be gained by longitudinal incision at the summit of the armpit; this enables removal of a segment long enough for transplantation.
Vein dissection should be performed proximally to the ribcage level and distally as far as the incision will allow. There is no need to restore anatomical continuity since collateral pathways ensure good drainage of blood for the arm, and therefore complications resulting from removal of the AV are rare. First, test the segment for valve competence before removing the valve (or valves). In cases of incompetence, bench reconstructive surgery can be performed, but adds significant complexity to the case.
The harvested vein segment should be kept in a heparinized saline solution. Transplantation should be performed to the most compatible recipient segment, either in the FV or popliteal vein (PV) , depending on the caliber. If the PV is preferred, given its duplication sometimes, the competence of parallel veins should be verified in advance. If competence is not ascertained, the refluxing parallel veins should be ligated.
The popliteal vein can be accessed via a traditional medial incision. Posterior exposure is an option, but is technically more complex and provides limited exposure. Complexity of this technique is correlated with the double intervention (harvesting the arm vein and implantation in the lower limb).
It is essential to avoid creating twisting, tension, and stenosis at the suture site as this can predispose the vein to thrombosis. In preference to a continuous suture, an interrupted suture or two-half sutures should be performed. The proximal anastomosis should have a wider diameter than the distal one, and as large a distance as possible should be left between the valve cusps and the proximal anastomosis.
While the risks of wound complications and lymphocele are decreased by staying away from the groin area, the effectiveness of the procedure is diminished in the presence of multiple PV or DFV incompetence. It involves a surgical procedure on an unaffected a healthy upper extremity. The incidence of postoperative thrombosis is not uncommon.
Neovalve
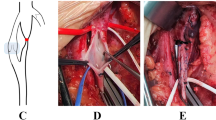
The neovalve is a technique which uses the principle of reconstructing a new autologous valve by refashioning the patient’s own vein wall. Raju and Hardy [36] proposed a de novo valve using a valvulated portion of the GSV or a tributary of the AV, which was inserted into the FV. They reported good results, despite a limited series. Plagnol [37] performed a neovalve in the terminal portion of the GSV; this portion of the GSV was invaginating into the CFV. Maleti [38] proposed a version in which the neovalve is obtained by dissecting the vein wall so as to fashion a flap or leaflet (Fig. 17.4). Thanks to the characteristic thickening of vein wall tissue, the neovalve is easier to perform in PTS, and it can also be performed in cases of valve agenesis [39]. Due to the anatomical variables in the vein wall and the diversity of post-thrombotic lesions, the configuration of the Maleti neovalve is variable in each case. The choice of the neovalve site, as well as the technical variations used in constructing it, should be based on high resolution duplex ultrasound (DUS) assessment . A significant portion of the technical aspects depends on the intraoperative findings after phlebotomy.
Post-thrombotic lesions have various features: slight thickening of the vein wall; uniform or otherwise; synechiae or septa; endoluminal fibrotic tissue which forms a double channel; notable thickening of the vein wall with fibrosis occupying a large portion of the lumen.
In all but the first of these conditions, endophlebectomy should be performed. The main risk with the neovalve technique is postoperative re-adhesion of the leaflet at the dissection site. Specific sutures can prevent this mode of failure.
Depending on the features of the vein wall, the neovalve may be bicuspid or have a single cusp. In order to prevent leakage, a valve with a single cusp should be fashioned deeper. It is now recognized that the shape of the valve itself determines physiological valve function. As the neovalve does not fully comply with nature’s model, the wash-out action performed by the sinus is lacking. The result is reduced movement in the flap, which in turn may provoke thrombosis in the valve sinus. In order to prevent this from happening, when applicable a leaflet is fashioned at the site of a tributary so as to create a competing flow.
An alternative technique has been suggested by other authors [40] whereby a portion of the vein wall is invaginated to create a flap; a PTFE patch is used to reconstruct the vein wall. The chief drawback of this technique is that, since the neovalve is open laterally, the leaflet cannot fragment the hydrostatic pressure. However, the reduced reflux volume will lead to partial functioning of the neovalve when combined with efficient ambulation.
The advantages of neovalve technique are that it creates an anti-reflux mechanism using the patient’s vein wall and offers a surgical alternative where transposition and transplant are not possible. The disadvantages of the technique are that it is technically challenging and has to be individualized. It frequently requires endophlebectomy and the reconstruction site can seldom be defined preoperatively.
Artificial Venous Valve
Over the years, various attempts have been made at creating a substitute venous valve. Research is still underway , but there is no current human application.
Indication for Treatment
Patient selection for deep valve reconstruction depends on a diagnostic protocol and a thorough clinical evaluation.
The diagnostic protocol involves DUS evaluation, air plethysmography [41], venography [11], and intravascular ultrasound (IVUS) [42].
DUS is essential for defining lower-limb venous abnormalities [43]. Since DUS is not sufficient to detect proximal obstructions—frequently present and to be treated first—further investigations are needed. DUS is able to detect the reflux, except where low buffering effects are present in the calf; in such cases the reflux can go undetected or underestimated. The reflux is not the only hemodynamic alteration correlated with valve malfunction; other features include modified volume and compliance, which may be underestimated in a standard DUS exam.
Patients with CVI should also be assessed on functional data such as calf pump efficiency [44]. Restoring valve competence at thigh level is not sufficient to maintain a low volume in the leg during ambulation if correction is not combined with efficient calf contraction.
Poor results in some series can be attributed to inappropriate patient selection.
Other parameters like VFI (venous filling index) , EF (ejection fraction) , RVF (residual volume fraction) are advisable in follow-up evaluation [45].
Any macrocirculatory disorders will have an impact at the microcirculatory level; correction of the same does not occur immediately following restoration of valve function. A microcirculatory evaluation is essential in monitoring the reversibility of microcirculatory lesions, and is also a key element in detecting the improvement brought about by pharmacological or compression therapy.
In patients selected for deep reflux correction, iliocaval and descending venography are indicated to rule out proximal obstruction when suspected with the use of IVUS .
The diagnostic protocol will be applied in any patient eligible for deep vein reconstruction with:
-
Severely impaired Quality of Life (QoL) despite compression therapy
-
Patients with C4–C6 affected by deep venous reflux
-
Patients with C3 disease and no superficial insufficiency has been detected;
-
Patients with C2 disease and multiple recurrences.
No further investigations and procedures are necessary [46, 47] in CVI patients who are able to maintain a good QoL and satisfactory conditions simply as a result of treatment to the superficial venous system or compression therapy.
CVI patients with malfunctioning hemodynamics in the leg , multiple recurrences in varicose veins that significantly affect QoL, but also young patients resistant to compression therapy deserve further investigation. By applying a selected and well-tolerated procedure we can considerably improve their condition [39, 48, 49].
Strategies
In primary, secondary and congenital reflux, operative treatment is important.
In primary varicose veins associated with superficial venous reflux, the competence of the deep venous system can sometimes be restored by treating the superficial system alone, thus reducing the overload of the deep system. However, the reduction of deep venous overload can restore the valve competence by reducing the diameter of the deep vein only when the valves are anatomically normal and with symmetrical, functional leaflets. Conversely, when the valves are dysplastic and present abnormal and asymmetrical leaflets, valve reconstruction should be considered since the reduction of deep venous overload is insufficient to restore the function of the valves. Thus a precise preoperative evaluation (DUS and phlebography) will allow for planning an appropriate treatment strategy.
Valve agenesis is characterized by the absence of valves throughout the deep venous system and usually manifests in young patients with severe CVI and impairment of QoL [50]. Superficial reflux and deep venous reflux due to valve agenesis are usually associated and ablation of the superficial system is usually not sufficient. In such cases, it is important to rule out any proximal venous obstruction and increase calf pump efficiency. The need to treat the deep system via a direct approach is reserved to patients in C4b–C6 and a neovalve construction is a good option.
PTS is a complex pathology characterized by two principal hemodynamic disorders: increased resistance to flow (obstruction), due to stenosis, intraluminal synechiae, rigidity of the venous wall [15] and reflux, due to valve damage [12] (Fig. 17.5). Usually obstructive lesions are in the proximal iliac and common femoral vein, while reflux is in the femoral, popliteal, and tibial veins.
Operative strategy in PTS [51] involves the treatment of proximal obstruction by means of venous stenting (Fig. 17.6) as a first step since the majority of the patients can improve without corrective reflux treatment. As a second step, consider the relief of common femoral vein obstruction by means of endophlebectomy, obstructive lesions in this crucial area of the leg.
Next the femoro-popliteal veins should be evaluated as common site for hemodynamic disorders due to reflux and obstruction. As a final step, in patients who show no improvement, direct deep venous reflux correction should be considered.
The diagnostic criteria needed to decide which strategy to apply are the following:
-
Presence and/or absence of proximal obstruction including occlusion.
-
Presence of axial reflux below the inguinal ligament, from groin to calf, via femoropopliteal axis or by superficial or profunda transfer, as well as their combination.
-
Presence and/or absence of proximal competence of the DFV.
-
In the case of DFV incompetence, identify single or multiple re-entry points into the PV.
-
Presence and competence of the great and small saphenous veins.
-
PV features (single and multiple channels) and their competence and/or incompetence.
-
Caliber of FV and PV.
-
Caliber and competence of the AV.
-
Presence of endoluminal fibrosis, determining double channel at femoropopliteal level.
Outcomes
The complication rate of deep venous system (DVS) treatments is particularly rare, and DVS surgery is safe [52]. The results are satisfactory, particularly in primary incompetence, despite the heterogeneity of patients. The less satisfactory results obtained treating the secondary incompetence are probably correlated with:
-
Inadequate understanding of deep venous pathophysiology
-
Inadequate imaging (in the past proximal obstruction was underestimated and consequently not previously treated)
-
Suboptimal patient selection
-
Patients with advanced CVI and non-reversible microcirculatory damage
The outcomes of deep venous reconstruction are limited to case series. Assessing the outcomes of deep vein surgery to correct reflux is complicated. Principally, we rely on the Villalta score in conjunction with VCSS. Most outcome literature to date is based on ulcer healing and pain reduction. A summary of the outcomes of each of the techniques is given in tables. Most experience has been with valvuloplasty with the internal and external techniques. The ability to achieve competent valves varies from 31.5% up to 79.8% but the clinical failure as measured by ulcer recurrence or nonhealing is 21–50% (Table 17.1). The experience with valve transposition and transplantation is more limited but has similar variability in long-term outcomes (Tables 17.2 and 17.3). There are only 3 case series with neovalve reconstruction but the outcomes seem to be relatively better with 68–100% achieving competent valve on follow-up and only 17% experiencing nonhealing or recurrence of ulcers (Table 17.4). The Portland team [65] has developed a bioprosthetic valve. However, at one-year follow-up valve competence was not evident, and the technology is still experimental.
Conclusions
The surgical treatment of deep venous reflux is safe and effective for treatment of selected patients with advanced venous disease and ulceration. Indeed, the clinical experience in this field is confined to only a few centers around the world and studies are based on case series. Nevertheless, surgical treatment addressing the deep vein system is gaining more attention, mostly because of new procedures which can help patients suffering from severe chronic vein insufficiency. It is possible that the variability in the outcomes in early series was affected by the presence of obstructive disease that was underdiagnosed. Our enhanced understanding of the complex interactions between reflux and obstruction, as well as the interactions between the superficial and deep venous systems will continue to evolve and improve our ability to deliver better therapy to patients with venous disease.
Abbreviations
- AV:
-
Axillary vein
- CFV:
-
Common femoral vein
- CVI:
-
Chronic venous insufficiency
- DFV:
-
Deep femoral vein
- DUS:
-
Duplex ultrasound
- DVR:
-
Deep venous reflux
- DVS:
-
Deep venous system
- EF:
-
Ejection fraction
- FV:
-
Femoral vein
- GSV:
-
Great saphenous vein
- IVUS:
-
Intravascular ultrasound
- PTFE:
-
Polytetrafluoroethylene
- PTS:
-
Post-thrombotic syndrome
- PV:
-
Popliteal vein
- QoL:
-
Quality of life
- RVF:
-
Residual volume fraction
- VCSS:
-
Venous clinical severity score
- VFI:
-
Venous filling index
References
Welch HJ, Faliakou EC, McLaughlin RL, et al. Comparison of descending phlebography with quantitative photoplethysmography, air plethysmography, and duplex quantitative valve closure time in assessing deep venous reflux. J Vasc Surg. 1992;16:913–20.
Nicolaides AN. Noninvasive assessment of primary and secondary varicose veins. In: Bernstein EF, editor. Noninvasive diagnostic techniques in vascular disease. 2nd ed. St. Louis: CV Mosby; 1982. p. 575–86.
Burnand KG, Lea Thomas M, O’Donnel TF, et al. The relationship between postphlebitic changes in the deep veins and the results of surgical treatment of venous ulcers. Lancet. 1976;1:936–8.
Holme TC, Negus D. The treatment of venous ulceration by surgery and elastic compression hosiery: a long term review. Phlebology. 1990;5:125–8.
Schmid-Schonbein GW, Takase S, Bergan JJ. New advances in the understanding of the pathophysiology of chronic venous insufficiency. Angiology. 2001;52(Suppl 1):S27–34.
Labropoulos N, Leon M, Nicolaides AN, et al. Venous reflux in patients with previous deep venous thrombosis : correlation with ulceration and other symptoms. J Vasc Surg. 1994;20:20–6.
Labropoulos N, Patel PJ, Tiongson JE, et al. Patterns of venous reflux and obstruction in patients with skin damage due to chronic venous disease. Vasc Endovasc Surg. 2007;41:33–40.
Lim KH, Hill G, Van Rij A. Deep venous reflux definitions and associated clinical and physiology significance. Lymphat Disord. 2013;1:325–32.
Marston W, Fish D, Unger J, et al. Incidence of and risk factors for iliocaval venous obstruction in patients with active or healed venous leg ulcers. J Vasc Surg. 2011;53:1303–8.
Danielsson G, Eklof B, Grandinetti A, et al. Deep axial reflux, an important contributor to skin changes or ulcer in chronic venous disease. J Vasc Surg. 2003;38:1336–41.
Kistner RL, Ferris EB, Raudhawa G, et al. A method of performing descending venography. J Vasc Surg. 1986;4:464–8.
Danielsson G, Arfvidsson B, Eklöf B, et al. Reflux from thigh to calf, the major pathology in chronic venous ulcer disease: surgery indicated in the majority of patients. Vasc Endovasc Surg. 2004;38:209–19.
Eklöf B, Perrin M, Delis KT, et al. VEINTERM Transatlantic Interdisciplinary Faculty. Updated terminology of chronic venous disorders: the vein-term transatlantic interdisciplinary consensus document. J Vasc Surg. 2009;49:498–501.
Adam DJ, Bello M, Hartshorne T, et al. Role of superficial venous surgery in patients with combined superficial and segmental deep venous reflux. Eur J Vasc Endovasc Surg. 2003;25:469–72.
Neglen P, Thrasher TL, Raju S. Venous outflow obstruction: an underestimated contributor to chronic venous disease. J Vasc Surg. 2003;38:879–85.
Vasdekis SN, Heather Clarke G, Nicolaides AN. Quantification of venous reflux by means of duplex scanning. J Vasc Surg. 1989;10:670–7.
Bauer G. The etiology of leg ulcers and their treatment by resection of the popliteal vein. J Int Chir. 1948;8:937.
Linton RR, Harry JB Jr. Posthrombotic syndrome of the lower extremity. Surgery. 1948;24:452.
Akesson H, Risberg B, Bjorgell O. External support valvuloplasty in the treatment of chronic deep vein incompetence of the legs. Int Angiol. 1999;18:233–8.
Eklöf B, Rutherford RB, Bergan JJ, et al. Revision of the CEAP classification for chronic venous disorders: consensus statement. J Vasc Surg. 2004;40:1248–52.
Nicolaides AN (chair) and the Executive Committee of the ad hoc committee, American Venous Forum, 6th annual meeting. February 22–25, 1994, Maui, Hawaii: Classification and grading of chronic venous disease in the lower limbs: a consensus statement. In: Gloviczki P, Yao JST, editors. Handbook of venous disorders. London, England: Chapman & Hall; 2001. p. 653–56.
Porter JM, Moneta GL. Reporting standards in venous disease: an update. International Consensus Committee on Chronic Venous Disease. J Vasc Surg. 1995;21:634–45.
Kahn SR, Comerota AJ, Cushman M, et al. The postthrombotic syndrome: evidence-based prevention, diagnosis and treatment strategies. A scientific statement from the American Heart Association. Circulation. 2014;130(18):1636–61.
Maleti O, Lugli M, Perrin M. Chirurgie du reflux veineux profond. Encyclopédie Médico-Chirurgicale (Elsevier Masson SAS, Paris), Techniques chirurgicales - Chirurgie vasculaire, 2009;43–163.
Kistner RL. Surgical repair of a venous valve. Straub Clin Proc. 1968;24:41–3.
Sottiurai VS. Technique in direct venous valvuloplasty. J Vasc Surg. 1988;8:646–8.
Tripathi R, Ktenedis KD. Trapdoor internal valvuloplasty – a new technique for primary deep vein valvular incompetence. Eur J Vasc Endovasc Surg. 2001;22:86–9.
Sottiurai VS. Supravalvular incision for valve repair in primary valvular insufficiency. In: Bergan JJ, Kistner RL, editors. Atlas of venous surgery. Philadelphia, PA: WB Saunders; 1992. p. 137–8.
Kistner RL. Surgical technique of external venous valve repair. Straub Found Proc. 1990;55:15–6.
Gloviczki P, Merrell SW, Bower TC. Femoral vein valve repair under direct vision without venotomy: a modified technique with angioscopy. J Vasc Surg. 1991;14:645–8.
Raju S, Berry MA, Neglén P. Transcommissural valvuloplasty: technique and results. J Vasc Surg. 2000;32:969–76.
Lurie F, Kistner RL, Eklof B, Kessler D. Mechanism of venous valve closure and role of the valve in circulation. A new concept. J Vasc Surg. 2003;38:955–61.
Kistner RL, Sparkuhl MD. Surgery in acute and chronic venous disease. Surgery. 1979;85:31–43.
Raju S. Axillary vein transfer for postphlebitic syndrome. In: Bergan JJ, Kistner RL, editors. Atlas of venous surgery. Philadelphia: WB Saunders; 1992. p. 147–52.
Taheri SA, Lazar L, Elias S, Marchand P, Heffner R. Surgical treatment of postphlebitic syndrome with vein valve transplant. Am J Surg. 1982;144:221–4.
Raju S, Hardy JD. Technical options in venous valve reconstruction. Am J Surg. 1997;173:301–7.
Plagnol P, Ciostek P, Grimaud JP, Prokopowicz SC. Autogenous valve reconstruction technique for post-thrombotic reflux. Ann Vasc Surg. 1999;13:339–42.
Maleti O, Lugli M. Neovalve construction in postthrombotic syndrome. J Vasc Surg. 2006;43:794–9.
Lugli M, Guerzoni S, Garofalo M, Smedile G, Maleti O. Neovalve construction in deep venous incompetence. J Vasc Surg. 2009;49:156–62.
Opie JC. Monocusp-novel common femoral vein monocusp surgery uncorrectable chronic venous insufficiency with aplastic/dysplastic valves. Phlebology. 2008;23:158–71.
Criado E, Farber MA, Marston WA, et al. The role of air plethysmography in the diagnosis of chronic venous insufficiency. J Vasc Surg. 1998;27:660–70.
Neglen P, Raju S. Intravascular ultrasound scan evaluation of the obstructed vein. J Vasc Surg. 2002;35:694–700.
Mattos MA, Summer D. Direct noninvasive tests (duplex scan) for the evaluation of chronic venous obstruction and valvular incompetence. In: Gloviczki P, Yao JST, editors. Handbook of venous disorders. London, England: Arnold; 2001. p. 120–31.
Araki CT, Back TL, Padberg FT, et al. The significance of calf muscle pump function in venous ulceration. J Vasc Surg. 1994;20:872–7.
Gillespie DL, Cordts PL, Hartono C, et al. The role of air plethysmography in monitoring results of venous surgery. J Vasc Surg. 1992;16:674–8.
Rutherford RB, Padberg FT Jr, Comerota AJ, et al. Venous severity scoring: an adjunct to venous outcome assessment. J Vasc Surg. 2000;31:1307–12.
Lamping DL, Schroter S, Kurz X, et al. Evaluation of outcomes in chronic venous disorders of the leg: development of a scientifically rigorous, patient-reported measure of symptoms and quality of life. J Vasc Surg. 2003;37:410–9.
Perrin M. Reconstructive surgery for deep venous reflux. A report on 144 cases. Cardiovasc Surg. 2000;8:246–55.
Sottiurai VS. Results of deep-vein reconstruction. Vasc Surg. 1997;31:276–8.
Plate G, Brudin L, Eklof B, Jensen R, Ohlin P. Physiologic and therapeutic aspects in congenital vein valve aplasia of the lower limb. Ann Surg. 1983;198(2):229–33.
Maleti O, Lugli M, Perrin M. Operative treatment in postthrombotic syndrome:an update. Phlebolymphology. 2014;21(3):131–7.
Lurie F, Kistner R, Perrin M, Raju S, Neglen P, Maleti O. Invasive treatment of deep venous disease. A UIP consensus. Int Angiol. 2010;29:199–204.
Masuda EM, Kistner RL. Long-term results of venous valve reconstruction: a four to twenty-one year follow-up. J Vasc Surg. 1994;19:391–403.
Raju S, Fredericks R, Neglen P, Bass JD. Durability of venous valve reconstruction for primary and post-thrombotic reflux. J Vasc Surg. 1996;23:357–67.
Sottiurai VS. Current surgical approaches to venous hypertension and valvular reflux. Int J Angiol. 1996;5:49–54.
Tripathi R, Sieunarine K, Abbas M, Durrani N. Deep venous valve reconstruction for non healing ulcers: techniques and results. ANZ J Surg. 2004;74:34–9.
Wang SM, Hu ZJ, Li SQ, et al. Effect of external valvuloplasty of the deep vein in the treatment of chronic venous insufficiency of the lower extremity. J Vasc Surg. 2006;44:1296–300.
Rosales A, Slagsvold CE, Kroese AJ, et al. External venous valveplasty (EVVp) in patients with primary chronic venous insufficiency (PVCI). Eur J Vasc Endovasc Surg. 2006;32:570–6.
Lethola A, Oinonen A, Sugano N, Alback A, Lepantalo M. Deep venous reconstructions: long-term outcome in patients with primary or post-thrombotic deep venous incompetence. Eur J Vasc Endovasc Surg. 2008;35:487–93.
Cardon JM, Cardon A, Joyeux A, Mangialardi N, Noblet D, Nguyen T, et al. La veine saphène interne comme transplant valvulé dans l’insuffisance veineuse post-thrombotique : résultats à long terme. Ann Chir Vasc. 1999;13:284–9.
Bry JD, Muto PA, O’Donnell TF, Isaaacson LA. The clinical and hemodynamic results after axillary-to-popliteal valve transplantation. J Vasc Surg. 1995;21:110–9.
Mackiewicz Z, Molski S, Jundzill W, Stankiewicz W. Treatment of postphlebitic syndrome with valve transplantation: 5 year experience. Eurosurgery ’95. Bologna Monduzzi 1995; 305–10.
Raju S, Neglen P, Doolittle J, et al. Axillary vein transfer in trabeculated postthrombotic veins. J Vasc Surg. 1999;29:1050–64.
Kabbani L, Escobar GA, Mansour F, Wakefield TW, Henke PK. Longevity and outcomes of axillary valve transplantation for severe lower extremity chronic venous insufficiency. Ann Vasc Surg. 2011 May;25(4):496–501.
Pavcnik D, Uchida B, Kaufman J, et al. Percutaneous management of chronic deep venous reflux: review of experimental work and early clinical with bioprosthetic valve. Vasc Med. 2008;13:75–84.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer International Publishing AG
About this chapter
Cite this chapter
Maleti, O., Lugli, M. (2018). Valve Reconstruction for Deep Venous Reflux. In: Chaar, C. (eds) Current Management of Venous Diseases . Springer, Cham. https://doi.org/10.1007/978-3-319-65226-9_17
Download citation
DOI: https://doi.org/10.1007/978-3-319-65226-9_17
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-65225-2
Online ISBN: 978-3-319-65226-9
eBook Packages: MedicineMedicine (R0)