Abstract
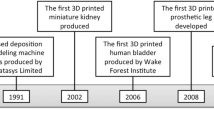
Current advances in imaging technology, virtual surgical planning, and 3D printing have potentially changed how we will use patient-specific information for treatment planning and customized treatment. Medical providers can not only view a 3D rendering of the patient’s anatomy on digital display, but that image can now be transferred as a physical model which not only aids in treatment planning but in patient education. The use of these technologies in craniofacial reconstruction is reported in the early 1990s (Gronet et al. 2003). These techniques have proven to provide surgeons confidence in executing their plan, reducing operating times, follow-up surgical revisions, and poor outcomes. In addition, they provide patient-centered care and better esthetic and functional outcomes (Grant et al. 2013) (Fig. 1). In this chapter, we will review some of the areas of application in craniofacial reconstruction and dentistry.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Current advances in imaging technology, virtual surgical planning, and 3D printing have potentially changed how we will use patient-specific information for treatment planning and customized treatment. Medical providers can not only view a 3D rendering of the patient’s anatomy on digital display, but that image can now be transferred as a physical model which not only aids in treatment planning but in patient education. The use of these technologies in craniofacial reconstruction was reported in the early 1990s (Gronet et al. 2003). These techniques have proven to provide surgeons confidence in executing their plan, reduced operating times, and better outcomes. In addition, they provide patient-centered care and better esthetic and functional outcomes (Grant et al. 2013) (Fig. 5.1). In this chapter, we will review some of the areas of application in craniofacial reconstruction and dentistry.
5.1 Craniofacial Imaging
Computed tomography is the preferred method of imaging for head and neck reconstructions. The Hounsfield scale enables identification of soft and hard tissues by their density; this allows for segmentation of the images for reconstruction of 3D models with minimal artifact but at the expense of radiation exposure to the patient (Gordon et al. 2014). In contrast, cone beam computed tomography (CBCT) has become more common in dental and medical practices; their low radiation exposure provides a unique opportunity to capture hard tissue images that have been used for endodontic diagnosis, airway visualization, orthognathic reconstructions, and dental implant planning (Gronet et al. 2003; Vannier 2003; Grant et al. 2013; Estrela et al. 2008). However, CBCT is subject to severe artifact from dental restorations, and lack the contrast to segment soft tissue to complement bone. In addition, due to the inconsistancy of contrast, the Hounsfield Scale does not apply to identify soft versus bone tissues.
Surface scanning has also been used to design and fabricate craniofacial devices. These are noninvasive and have applications in craniofacial planning (Sabol and Grant 2011). These scanning devices use laser, light, or some type of contact scanning technologies employing technologies such as stereo photogrammetry to increase accuracy, and are stationary or handheld (Knoops et al. 2017). The images captured are often used for registration to other medical images to provide more accurate virtual models for virtual planning and to design devices, medical models, and surgical guides. In addition, surface scanning has also been successfully used to fabricate maxillofacial prostheses (Sabol and Grant 2011; Grant et al. 2015).
5.2 Cranioplasty
Cranial defects can be caused by trauma, tumor, or decompressive craniotomy. Historically, the fabrication of a custom cranial implant involved an ambulatory patient, conventional impression techniques, fabricating an indirect stone model of the defect, and fabricating a mold for processing polymethyl methacrylate (PMMA) (Aquilino et al. 1988). Surgical placement involved extensive modification to get an acceptable fit with long hours in the operating room and use of self-curing acrylics to fill in the gaps. The initial use of 3D printing was to print the defect from which a custom wax implant could be fabricated and a mold for PMMA (Fig. 5.2). In this process, the patient does not have to be available to the laboratory, and the process enables more complicated craniofacial implants that fit the defect with minimal modification, cutting the fabrication time down by close to 75%, and operating time nearly in half (Gronet et al. 2003) (Fig. 5.2). This process has now evolved to digital design directly from medical imaging and fabrication of the cranial implant by milling PMMA and Polyetheretherketone (PEEK) implants or 3D-printed titanium and Polyethyl ketone ketone (PEKK) (Fig. 5.3).
5.3 Craniofacial Reconstruction
In trauma cases, 3D models may help to recognize the position and the direction of fractures, the number of bone fragments, and the degree of dislocation. Virtual planning can assist in a reconstruction plan with reestablishing contours and fabricating positioning and bending guides for plates and recontour bars. However, there are limitations that can result in surgical delay due to long model production with current additive manufacturing processes (Powers et al. 1998; Holck et al. 1999; McAllister 1998) (Fig. 5.4).
Virtual simulation and printed models from medical images provide solid models that simulate osteotomies and grafts, simulate segmental jaw movements, and facilitate preoperative construction of surgical guides, templates, and custom surgical devices (Ander et al. 1994; D’Urso et al. 1999; Kermer et al. 1998).
Guides can be designed and fabricated that allow the prebending of recontouring bars for mandibular stabilization prior to the surgical reduction, positioning guides that reapproximate bone sections for plating, cutting guides to move bone as needed, and customized devices to replace or stabilize sections of the mandible, zygoma, or orbit using biocompatible materials (Singarea et al. 2004). Using virtual surgical techniques, the surgical guides assist the surgeon in osteotomy cuts, implant placement, positioning of bone and soft tissue for reconstruction, and assistance in prebending of reconstruction plates (Fig. 5.5).
Recently, the limits of craniofacial reconstruction have been challenged with the success of full total face transplants. The same principles of virtual planning can be very useful in the selection of appropriate anatomical donors to approximate the correct dental occlusion and other anatomical reconstructions. (Murphy et al. 2015a; Sosin et al. 2016) (Fig. 5.6). Cutting guides can be designed to provide an intimate fit of bone margins of the donor anatomy to the recipient site. Current research in this area proposes navigational technologies and mastication simulation (Gordon et al. 2014; Murphy et al. 2015b).
In respect to the donors, a facial mask is required after the harvest of the transplant. Conventional fabrication of a total facial prostheses by a maxillofacial prosthodontist or anaplastologist at the time of the surgery can be disruptive and expensive. An alternative technique using 3D printing from medical imaging and photographs have been proposed, as they can be fabricated directly or with a mold, prior to the surgical intervention at a lower cost (Grant et al. 2014) (Fig. 5.7).
5.4 Dental Implant Guides
Dental implant placement is driven by the restorative plan it retains or supports. The purpose of a surgical guide is to assist the surgeon in the location and direction of the osteotomy prior to dental implant placement. The Academy of Prosthodontics defines a surgical template as a guide used to assist in proper surgical placement and angulations of dental implants (The Glossary of Prosthodontic Terms, 2017). Based on the amount of the operative restriction of the drill, the design of the surgical template can be classified as nonrestrictive, partially restrictive, or completely restrictive (Stumpel 2008; Misch and Dietsh-Misch 1999). Historically, surgical guides were fabricated conventionally on dental casts using a variety of techniques and materials including clear vacuum-formed matrix , free-form auto-polymerizing acrylic resin and acrylic resin duplicates of the available prosthesis or diagnostic wax-ups.
Recently, software has become more available that provides dental implant planning from CBCT using digital scans of diagnostic wax-ups or virtual restorations from intraoral scans or diagnostic casts. By registering the images, the restoration can be planned, and a surgical guide can be fabricated to limit the placement of the dental implant to accommodate the restorative plan (Fig. 5.8). In some instances, this workflow allows for “same day” implant retained restorations, even in more complicated cases requiring grafting (Cheng et al. 2008; Stapleton et al. 2014). Once the digital or virtual plans have been designed, the guides and the restorations can be produced with digital manufacturing—either additive or milled.
Most additive manufacturing technologies can be used to fabricate the surgical guides; however, there are some concerns of irritation from residual surface chemicals of some polymers. USP (US Pharmacopeia) Class VI judges the suitability of plastic material intended for use as containers or accessories for parenteral preparations. Suitability under USP Class VI is typically a base requirement for medical device manufacturers. It is recommended that materials compliant with this test be used for all surgical guides as well as medical/dental models available in a surgical setting. Most manufacturers of 3D printers will have a medical grade material that is Class VI compliant and offer a specific cleaning process for these items. However, the Food and Drug Administration (FDA) has cleared devices for surgical guides and limited intraoral use produced by 3D printed materials (Formlabs, Cambridge, MA), and it is expected that there may be similar trends in the future. This is further detailed in Chap. 10.
5.5 Maxillofacial Prosthetics
Trained maxillofacial prosthodontists along with technicians or anaplastologists have historically achieved the planning and fabrication of facial features through moulage and sculpting techniques. These techniques are usually uncomfortable to the patient and require several days to fabricate the prosthesis. In contrast, medical images provide the information that will allow for virtual planning and fabrication of facial prostheses. Software mirroring techniques can be used to “sculpt” missing ears or missing anatomy on the contralateral side and software that provides “electronic clay” type technologies (Geomagics Freeform, 3D Systems) allow for development of textures, accommodate attachments, and design molds for CAD/CAM or 3D printing (Jiao et al. 2004; Liacouras et al. 2011). Molds are then layered and colored with silicone since currently there are no commercial silicone printers that would allow for direct fabrication and color (Fig. 5.9). Recent advancements in lower-priced scanners and online maxillofacial design commercial sites are making these technologies more accessible to providers outside academic and military practices.
5.6 Other Craniofacial Applications
Aside from reconstruction and dental applications, benign tumors of the jaw usually present with localized expanding deformities. Other pathology and vascular lesions can be differentiated with contrast enhanced CT. Printed models provide vital information for planning and patient education, offering a physical model of the affected area (Fig. 5.10). In addition, using virtual surgical planning with a color-coding technique, specific structures such as teeth, nerves, and the extent of a tumor can be displayed, facilitating more detailed surgical planning (Santler et al. 1998).
Forensic reconstruction is another area of application of digital planning and 3D printing. An unpublished work by the Naval Postgraduate Dental School’s Craniofacial lab working with the Exploited Children’s section of the FBI validated soft tissue reconstruction software from CT images of complete and incomplete skulls (Fig. 5.11), suggesting that computer reconstruction can be valuable in skull reconstructions historically done by forensic artists.
5.7 Conclusions
Continual advances in medical imaging, reconstruction software, and 3D printing continue to aid and advance the field of craniofacial surgery and other medical specialties. Technology advances, including more user-friendly software will enhance utlization. As these technologies become increasingly available and affordable, adoption may eventually become routine.
References
Ander H, Zur Nedden D, Muhlbauer W. CT-guided stereolithography as a new tool in craniofacial surgery. Br J Plast Surg. 1994;47:60–4.
Aquilino SA, Jordan RD, White JT. Fabrication of an alloplastic implant for the cranial implant. J Prosthet Dent. 1988;59(1):68–71.
Cheng AC, Tee-Khin N, Siew-Luen C, Lee H, Wee AG. The management of a severely resorbed edentulous maxilla using a bone graft and a CAD/CAM-guided immediately loaded definitive implant prosthesis: a clinical report. J Prosthet Dent. 2008;99(2):85–90.
D’Urso PS, Barker TM, Earwaker WJ, Bruce LJ, Atkinson RL, Lanigan MW, Arvier JF, Effeney DJ. Stereolithography biomodelling in cranio-maxillofacial surgery: a prospective trial. J Craniomaxillofac Surg. 1999;27:30–7.
Estrela C, Bueno MR, Azevedo BC, Azevedo JR, Pécora JD. A new periapical index based on cone beam computed tomography. J Endod. 2008;34(11):1325–31.
Glossary of Prosthodontic Terms: ninth edition. 2017 May;117(5S):e1-e105.
Gordon CR, Murphy RJ, Coon D, Basafa E, Otake Y, Al Rakan M, Rada E, Susarla S, Swanson E, Fishman E, Santiago G, Brandacher G, Liacouras P, Grant G, Armand M. Preliminary development of a workstation for craniomaxillofacial surgical procedures: introducing a computer-assisted planning and execution system. J Craniofac Surg. 2014;25(1):273–83.
Grant GT, Liacouras P, Santiago G, Garcia JR, Al Rakan M, Murphy R, Armand M, Gordon CR. Restoration of the donor face after facial allotransplantation: digital manufacturing techniques. Ann Plast Surg. 2014;72(6):720–4.
Grant GT, Kondor S, Liacouras P. Maxillofacial imaging in the trauma patient. Atlas Oral Maxillofac Surg Clin North Am. 2013;21(1):25–36.
Grant GT, Liacouras P, Aitaholmes C, Garnes J, Wilson WO. Digital capture, design, and manufacturing of a facial prosthesis: clinical report of a pediatric patient. J Prosthet Dent. 2015;114(1):138–41.
Gronet PM, Waskewicz GA, Richardson C. Preformed Acrylic Cranial Implants using fused deposition modeling: a clinical report. J Prosthet Dent. 2003;90(5):429–33.
Holck DEE, Boyd EM Jr, Ng J, Mauffray RO. Benefits of stereolithography in orbital reconstruction. Ophthalmology. 1999;106:1214–8.
Jiao T, Zhang F, Huang X, Wang C. Design and fabrication of auricular prostheses by CAD/CAM system. Int J Prosthodont. 2004;17:460–3.
Kermer C, Rasse M, Lagogiannis G, Undt G, Wagner A, Millesi W. Colour stereolithography for planning complex maxillofacial tumour surgery. J Craniomaxillofac Surg. 1998;26:360–2.
Knoops PG, Beaumont CA, Borghi A, Rodriguez-Florez N, Breakey RW, Rodgers W, Angullia F, Jeelani NU, Schievano S, Dunaway DJ. Comparison of three-dimensional scanner systems for craniomaxillofacial imaging. J Plast Reconstr Aesthet Surg. 2017;70(4):441–9.
Liacouras P, Garnes J, Roman R, Grant GT. Auricular prosthetic design and manufacturing using computed tomography, 3D photographic imaging and rapid prototyping. J Prosthet Dent. 2011;105(2):80–2.
McAllister ML. Application of stereolithography to subperiosteal implants manufacture. J Oral Implantol. 1998;24:89–92.
Misch CE, Dietsh-Misch F. Diagnostic casts, preimplant prosthodontics, treatment prostheses, and surgical templates. In: Misch CE, editor. Contemporary implant dentistry. 2nd ed. St Louis, MO: Mosby; 1999. p. 135–50.
Murphy RJ, Gordon CR, Basafa E, Liacouras P, Grant GT, Armand M. Computer-assisted, Le Fort-based, face-jaw-teeth transplantation: a pilot study on system feasibility and translational assessment. Int J Comput Assist Radiol Surg. 2015b;10(7):1117–26.
Murphy RJ, Basafa E, Hashemi S, Grant GT, Liacouras P, Susarla SM, Otake Y, Santiago G, Armand M, Gordon CR. Optimizing hybrid occlusion in face-jaw-teeth transplantation: a preliminary assessment of real-time cephalometery as part of the computer-assisted planning and execution workstation for crani-omaxillofacial surgery. Plast Reconstr Surg. 2015a;136(2):350–62.
Powers DB, Edgin WA, Tabatchnick L. Stereolithography: a historical review and indications or use in the management of trauma. J Craniomaxillofac Trauma. 1998;4:16–23.
Sabol J, Grant GT. Digital image capture and rapid prototyping of the maxillofacial defect. J Prosthodont. 2011;20(4):310–4.
Santler G, Kärcher H, Ruda C. Indications and limitations of three-dimensional models in cranio-maxillofacial surgery. J Craniomaxillofac Surg. 1998;26:11–6.
Singarea S, Dichena L, Lu B, Yanpub L, Zhenyub G, Yaxionga L. Design and fabrication of custom mandible titanium tray based on rapid prototyping. Med Eng Phys. 2004;26(8):671–6.
Sosin M, Ceradini DJ, Hazen A, Levine JP, Staffenberg DA, Saadeh PB, Flores RL, Brecht LE, Bernstein GL, Rodriguez ED. Total face, eyelids, ears, scalp, and skeletal subunit transplant cadaver simulation: the culmination of aesthetic, craniofacial, and microsurgery principles. Plast Reconstr Surg. 2016;137(5):1569–81.
Stapleton BM, Lin WS, Ntounis A, Harris BT, Morton D. Application of digital diagnostic impression, virtual planning, and computer-guided implant surgery for a CAD/CAM-fabricated, implant-supported fixed dental prosthesis: a clinical report. J Prosthet Dent. 2014;112(3):402–8.
Stumpel LJ 3rd. Cast-based guided implant placement: a novel technique. J Prosthet Dent. 2008;100:61–9.
Vannier MW. Craniofacial computed tomography scanning: technology applications and future trends. Orthod Craniofac Res. 2003;6(Suppl. 1):23–30.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer International Publishing AG
About this chapter
Cite this chapter
Grant, G.T., Liacouras, P.C. (2017). Craniofacial Applications of 3D Printing. In: Rybicki, F., Grant, G. (eds) 3D Printing in Medicine. Springer, Cham. https://doi.org/10.1007/978-3-319-61924-8_5
Download citation
DOI: https://doi.org/10.1007/978-3-319-61924-8_5
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-61922-4
Online ISBN: 978-3-319-61924-8
eBook Packages: MedicineMedicine (R0)