Abstract
Germ cells, such as oocytes and spermatocytes, produce haploid gametes by a special type of cell division called meiosis. The reduction of chromosome number is achieved in meiosis I, in which homologous chromosomes (homologs) are paired and recombined with their counterparts and finally segregated from each other. How meiotic chromosomes behave in a different manner from mitotic chromosomes has been a fascinating problem for cellular and developmental biology. Cohesin and condensin are multi-subunit protein complexes that play central roles in sister chromatid cohesion and chromosome condensation (also segregation), respectively. Recent studies investigating the expression and function of cohesin and condensin in mammalian germ cells greatly advance our understanding of the molecular mechanism underlying the meiotic chromosomal events. Furthermore, accumulating evidence suggests that reduction of cohesin during prophase I arrest in mammalian oocytes is one of the major causes for age-related chromosome segregation error. This review focuses on the regulation and functions of cohesins and condensins during mammalian meiosis.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
1 Introduction
Meiosis is a special type of cell division that produces gametes (sperms and eggs) from spermatocytes and oocytes in animals. Unlike mitosis in which a single round of DNA replication is followed by a single round of cell division, two successive rounds of cell division follow a single round of DNA replication in meiosis. The actual reduction of chromosome number is accomplished in the first meiotic division (meiosis I). In meiosis I, homologous chromosomes (homologs) recombine with their partners at prophase. As a result, sister chromatid cohesion distal to chiasmata make physical connection between homologs by metaphase I. At the onset of anaphase I, homologs separate from each other by resolution of inter-sister arm cohesion while sister chromatids (sisters) are kept attached at the centromere regions. Finally, in the second meiotic division (meiosis II), sisters separate from each other by resolution of the centromeric cohesion (Miyazaki and Orr-Weaver 1994).
2 Sister Chromatid Cohesion by Cohesin
Cohesin is a multi-subunit protein complex which establishes and maintains sister chromatid cohesion in mitosis and meiosis (Peters et al. 2008; Nasmyth and Haering 2009). The cohesin complex consists of four subunits: two structural maintenance of chromosome proteins, SMC1 and SMC3, a kleisin subunit RAD21 (also called SCC1), and either one of STAG1/SA1 or STAG2/SA2 (Table 15.1). Since cohesin complex forms a tripartite ringlike structure, it is believed that the complex might hold sisters together by embracing the two nucleosomes in its ring (Gruber et al. 2003; Nasmyth 2011) (Fig. 15.1). Although cohesin binds to chromatin throughout interphase, it can establish sister chromatid cohesion only during S phase (Uhlmann and Nasmyth 1998). Cohesin maintains the sister chromatid cohesion from S phase until the onset of anaphase. However, most of cohesin is released from chromosomes prior to sister chromatid separation in a separase-independent pathway, which involves cohesin-associated proteins, Wapl, Pds5, and Sororin as well as phosphorylation of cohesin subunits by mitotic kinases (Shintomi and Hirano 2009; Nishiyama et al. 2010). Therefore, just before anaphase, only a small population of cohesin remains on the chromosomes mainly at centromere regions. At anaphase onset, anaphase-promoting complex/cyclosome (APC/C) associating with CDC20 ubiquitinates the target proteins, securin and cyclin B, thereby inducing the destruction of them by proteasome (Yu 2007). This leads to the activation of separase that cleaves the RAD21 subunit of cohesin complex, resulting in sister chromatid separation (Uhlmann et al. 2000; Waizenegger et al. 2000).
Models of chromosome cohesion and condensation by cohesin and condensin complexes. A cohesin complex is thought to embrace two nucleosomes from sister chromatids, while a condensin complex is thought to connect the DNA segments from the same chromatid. A transiently expressed meiotic cohesin may embrace two nucleosomes from non-sister chromatids in this hypothetical model
3 Meiosis-Specific Cohesin Subunits
In addition to the canonical mitotic cohesin subunits, specific variants of cohesin subunit are expressed in meiosis. In 1999, yeast Rec8 was first identified as a meiosis-specific cohesin subunit that is required for the reductional cell division (Klein et al. 1999; Watanabe and Nurse 1999). Thereafter, several meiosis-specific cohesin subunits have been found in various species. So far, SMC1β (Revenkova et al. 2001), STAG3 (Prieto et al. 2001), REC8 (Eijpe et al. 2003; Lee et al. 2003), and RAD21L (Gutiérrez-Caballero et al. 2011; Ishiguro et al. 2011; Lee and Hirano 2011) have been found as meiosis-specific cohesin subunits in mammals (Table 15.1). Remarkably, canonical mitotic cohesin subunits are also expressed, at least in some stages of meiosis (Eijpe et al. 2000; Prieto et al. 2002; Xu et al. 2004; Parra et al. 2004) (Fig. 15.1). Although a great deal of effort has been made to examine the expression pattern of cohesin subunits during meiosis, there are some contradictions (see Suja and Barbero 2009). The contradictions might have arisen due to the differences in the fixation and labeling methods, antibody specificity, and/or sensitivity for detection of signals in the immunofluorescence analyses. Here, I will talk about kleisin subunits as one of the examples of contradictions. One paper reported that RAD21 is expressed almost throughout whole meiosis and localizes along axial element/lateral elements (AE/LEs) in prophase I and mainly at centromeres in metaphase I and metaphase II (Xu et al. 2004). Another paper shows RAD21 being present on the AE/LEs in prophase I and at inter-chromatid arm region and around centromeres in metaphase I (Parra et al. 2004). And another paper reports that RAD21 is expressed only during a short period of prophase I and localizes along LEs (Lee and Hirano 2011). As for RAD21L, some reports (Gutiérrez-Caballero et al. 2011; Ishiguro et al. 2011) show the localization on chromosomes from prophase I to metaphase I, while other reports do so only from leptotene to mid pachytene (Lee and Hirano 2011; Ishiguro et al. 2014). Although it is difficult to determine which of these observations is correct, functional analyses might help in making that judgment call. For example, RAD21, if it were present on the AEs at leptotene stage, would not contribute to AE formation, since AEs are not formed in Rec8/Rad21L double KO mice (Llano et al. 2012).
Meiotic Cohesin Complexes
Immunoprecipitation analyses using mouse testis extracts in combination with immunofluorescence analyses of localization suggest that there are several types of meiotic cohesin complexes in addition to the canonical cohesin complexes. The possible combinations of subunits in cohesin complexes so far reported are as follows: RAD21-SMC1α-SMC3-STAG3, RAD21-SMC1β-SMC3-STAG3, RAD21L-SMC1α-SMC3-STAG3, RAD21L-SMC1β-SMC3-STAG3, REC8-SMC1α-SMC3-STAG3, and REC8-SMC1β-SMC3-STAG3. It is generally agreed that RAD21 and RAD21L associate with both SMC1α and SMC1β paralogs in combination with SMC3 and STAG3. However, opinions vary as to REC8-containing cohesin complexes: some groups argue that REC8 associates only with SMC1β but not SMC1α (Lee et al. 2003; Ishiguro et al. 2011; Lee and Hirano 2011), whereas another group insists that REC8 associates with both SMC1 paralogs (Revenkova et al. 2004). For settling this disagreement, it seems reasonable to suppose that REC8-SMC1α-containing complex, if any, would be present at an extremely low level, because simultaneous depletion of SMC1β and RAD21L, which removes all the cohesin complexes with the exception of REC8-SMC1α-containing complex from chromosome axes at leptotene, almost completely abolishes the AE formation (Biswas et al. 2016).
4 Roles of Cohesins in Meiotic Prophase I
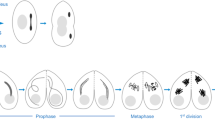
Prophase I proceeds in parallel with the assembly and disassembly of the synaptonemal complexes (SCs) (Page and Hawley 2004) (Fig. 15.2). After premeiotic S phase, axial elements (AEs) are formed along chromosomes at leptotene stage. The AEs on homologs start to be connected by transverse filaments by a mechanism called synapsis at zygotene stage. After synapsis the AEs are called the lateral elements (LEs). At pachytene stage, homologous AEs are connected along their entire length. Finally, the SC is disassembled at diplotene stage. Although the SC mediates the interaction between homologs transiently in prophase I, it is essential for the establishment of crossover recombination (de Vries et al. 2005), which holds association between homologs until anaphase I in conjunction with sister chromatid cohesion distal to chiasmata. The genetic studies using knockout (KO) mice showed that meiotic cohesins are involved in a variety of chromosomal events occurring in prophase I.
Pairing and synapsis of homologs at prophase I. At the leptotene stage, cohesins are linearly localized along chromosomes concurrently with formation of axial elements (AEs). Homologs are juxtaposed through elusive phenomenon called pairing. At the zygotene stage, a pair of AEs on homologs start to be connected by transverse filaments (TFs). This phenomenon is so called synapsis. The AEs on synapsed region are called lateral elements (LEs). At the pachytene stage, synapsis is completed, and the synaptonemal complex (SC) is formed on each pair of homologs. At the diplotene stage, the SC is disassembled, but homologs are kept connected through crossover recombination
4.1 Roles of Cohesins in AE Formation
One of the common phenotypes observed in the mice depleted of any meiosis-specific cohesin subunit is the defect in AE formation although the severity is different among the mutants. In Smc1β-, Rec8-, or Rad21L-deficient mice, the defect is only partial: shorter AEs are formed (Bannister et al. 2004; Revenkova et al. 2004; Xu et al. 2005; Herrán et al. 2011). On the other hand, no AEs are formed in spermatocytes depleted of STAG3 (Winters et al. 2014). The difference would be explained by the presence or absence of cohesin subunit paralogs that play a nonredundant role in AE formation. Indeed, the paralogs of SMC1β, RAD21L, and REC8 are localized along AEs whereas STAG3 paralogs, i.e., STAG1 or STAG2, are absent on AEs. Further supporting of this explanation, Rec8 and Rad21L double-knockout meiocytes completely abolished the AE formation (Llano et al. 2012). Thus, AE formation depends on several kinds of cohesins that function nonredundantly. Conversely, the linear assembly of cohesin on chromatin is not dependent on the AEs since thread-like signals of cohesin are detectable in Sycp3 KO spermatocytes in which AEs are not formed (Pelttari et al. 2001). Therefore, it seems reasonable to suppose that cohesins create axial cores along chromosomes, onto which AEs are later assembled (Fig. 15.2).
4.2 Roles of Cohesins in Meiotic Recombination
Meiotic recombination is initiated by the creation of double-strand breaks (DSBs) mediated by a topoisomerase-like protein, Spo11 (Keeney 2001). The DSBs are repaired and eventually converted into crossover or noncrossover recombination through a series of events including exchange of repair- or recombination-related proteins (Santucci-Darmanin et al. 2000). In Smc1β-, Rad21L-, Rec8-, and Stag3-deficient mice, γ-H2AX signals (a marker for double-strand breaks) and RAD51 or RPA signals (markers for recombination intermediates) are detected on the chromosomes in both male and female meiocytes (Bannister et al. 2004; Revenkova et al. 2004; Xu et al. 2005; Herrán et al. 2011; Fukuda et al. 2014). Thus, cohesins are dispensable for creation and initiation of DSB processing. The subsequent processing of DSBs to crossover recombination, however, requires cohesins in male meiosis since MLH1 foci, a marker for crossover recombination, are not detected in the cohesin-deficient spermatocytes. In this regard, female meiosis is far more complex. In oocytes, formation of crossover requires REC8 and STAG3 (Xu et al. 2005; Fukuda et al. 2014) but neither SMC1β nor RAD21L (Revenkova et al. 2004; Herrán et al. 2011). This issue will be discussed in a later Sect. 15.6.
4.3 Roles of Cohesins in Homolog Pairing
Synapsis and recombination of homologs are preceded by an elusive chromosomal phenomenon called recognition or pairing of homologs. How do homologs find their partners for the initiation of physical interaction? It has been widely believed that 3′ single-stranded DNA overhangs resulting from the resection of 5′ ends of DSBs are utilized for homolog searching based on the nucleotide sequences (Keeney 2001) (Fig. 15.3). However, recent studies suggest that homolog searching and pairing occurs independently of DSBs (Boateng et al. 2013). Moreover, it is argued that the paring is mostly dependent on RAD21L but only partly on REC8, since initial association of homologs and the subsequent pairing were impaired in Rad21L/Spo11 double KO but not in Rec8/Spo11 double KO mice (Ishiguro et al. 2014). This view is supported by a recent super-resolution microscopic study showing that RAD21L and REC8 are present at the connection sites between lateral elements and transverse filaments with RAD21L being interior to REC8 (Rong et al. 2016). Furthermore, ectopically expressed RAD21L promote adjacency of homologs in the somatic cells (Rong et al. 2017). The accumulating data suggest that RAD21L is a special type of cohesin subunit dedicated for interaction between homologs. Interestingly, when either RAD21L or REC8 is depleted, synapsis occurs in an abnormal fashion, i.e., between sisters or between non-homologous chromosomes (Xu et al. 2005; Herrán et al. 2011), implying that several types of cohesin are required for the proper synapsis between homologs after pairing.
In Drosophila, it has been recently proposed that two types of meiotic cohesin complexes are regulated and function differentially: one complex containing C(2)M, a presumptive ortholog of RAD21L, is dynamic and required for interhomolog interaction while another complex containing SOLO, a presumptive ortholog of REC8, is stable and required for sister chromatid interaction (Gyuricza et al. 2016). Therefore, contribution of different types of cohesin complex to specific process of establishing and maintaining meiotic chromosome cohesion might be a conserved mechanism in a wide variety of species. In mammalian meiosis, RAD21L-containing cohesin may transiently function to establish the interaction between homologs whereas REC8-containing cohesin may hold sisters together until the onset of anaphase II (Figs. 15.1 and 15.3).
5 Chromosome Cohesion and Separation in Meiosis
For the faithful homolog separation in meiosis I, it is essential to maintain the link between homologs until the onset of anaphase I. The link is made from the conjunction of sister chromatid cohesion and crossover recombination, both of which require meiotic cohesins (Fig. 15.3). The dissociation of arm cohesion leads to the homolog separation. As mentioned above, in mitotic prophase, most of cohesins are released from chromosome arms during sister chromatid resolution, which facilitates the subsequent sister chromatid separation. In contrast, total loss of cohesin from chromosome arms prior to metaphase I to anaphase I transition is deleterious for accurate chromosome separation in meiosis I although dissociation of some fraction of cohesin during prophase I might facilitate homolog separation (Brieño-Enríquez et al. 2016). Thus, meiotic cohesins, at least in part, must be resistant to the releasing factors such as Wapl, which had been reported to be localized on the SC at pachytene stage (Kuroda et al. 2005). Alternatively, the releasing factors might be suppressed or inactivated during meiosis.
Initiation of anaphase I requires the activation of anaphase-promoting complex (APC) by its associating activator CDC20 (Amanai et al. 2006; Jin et al. 2010). It is thought that APC activation subsequently induces the degradation of securin and cyclin B, which results in activation of separase, since both securin destruction and separase activation are required for homolog separation (Herbert et al. 2003; Terret et al. 2003; Kudo et al. 2006; Lee et al. 2006). The activated separase then cleaves REC8 on chromosome arms, thereby inducing homolog separation (Kudo et al. 2006, 2009) whereas REC8 on centromeres is protected by shugoshin (SGO2) (Lee et al. 2008). For the faithful chromosome separation in meiosis I, sister kinetochore should be oriented toward and connected to the same spindle poles. The so-called monopolar attachment requires kinetochore protein called MEIKIN, which has been recently identified as a functional homolog to budding yeast monopolin and fission yeast Moa1 (Kim et al. 2015). REC8-containing cohesin functions downstream to MEIKIN for mono-orientation of sister kinetochores; its cleavage at centromeres causes defect in mono-orientation (Tachibana-Konwalski et al. 2013).
6 Sexual Dimorphism in Cohesin KO Mice
Some of the meiosis-specific cohesin KO mice exhibit sexual dimorphism in the phenotype. Single knockout of either one of Smc1β, Rad21L, Rec8, or Stag3 arrests the spermatocytes at prophase I due to severe defects in synapsis and/or recombination (DNA repair). In contrast, the phenotypes in oocytes differ among these knockout mice. SMC1β-depleted oocytes are highly error prone but proceed to metaphase II (Revenkova et al. 2004). RAD21L-deficient females are fertile but develop an age-dependent sterility (Herrán et al. 2011). On the other hand, REC8-null neonatal ovaries are devoid of oocytes and ovarian follicles, indicating that REC8-depleted oocytes never proceed beyond prophase I (Bannister et al. 2004; Xu et al. 2005). Likewise, ovarian follicles are not found in Stag3 KO mice (Fukuda et al. 2014). Sexual dimorphism in meiotic cohesin mutants is partially attributed to the different stringency of control at pachytene checkpoint between spermatocytes and oocytes (Morelli and Cohen 2005; Burgoyne et al. 2009). Currently, however, the reason why variation in sexual dimorphism arises in a cohesin-subunit-dependent manner is unknown. Each cohesin subunit may have different degree of contribution to a particular event in male and female meiosis.
7 Age-Related Chromosome Error in Oocytes
It has been widely accepted that the rate of trisomy in human pregnancy increases with maternal age from ~2% among women under 25 years to ~35% among women over 40 years old (Hassold and Hunt 2001). Most of the trisomies originate from chromosome segregation errors during meiosis I in oocytes although there is variation in origin of trisomies depending on the type of chromosomes involved (Hassold and Hunt 2001). Recent studies using live cell imaging have investigated how chromosome segregation errors arise in aged oocytes and proposed two pathways. In one model, weakened centromere cohesion is a leading cause of premature sister chromatid separation in anaphase I in aged oocytes (Chiang et al. 2010). In another model, most of the errors are preceded by bivalent separation into univalent due to the intolerance of weakened arm cohesion against pulling force of spindle microtubules during metaphase I, which subsequently leads to premature separation of sisters in anaphase I (Sakakibara et al. 2015). In either case, aged deterioration of sister chromatid cohesion causes the premature separation of sisters in anaphase I. Thus, cohesin is one of the most probable molecules whose decay or destruction causes the age-related chromosome errors.
Several studies have approached the question whether cohesin is involved in the age-related meiotic chromosome errors. The first implication was provided by a study using conventional KO mice for Smc1β. The Smc1β-deficient female mice are sterile: the oocytes proceed to metaphase II but become aneuploidy due to premature loss of chromosome cohesion. In the Smc1β-deficient oocytes, the incidence of premature separation of both homologs and sisters is markedly increased as the mice ovulating grow up from 1 month old to 6 months old (Hodges et al. 2005). This suggests that depletion of SMC1β might accelerate or enhance the normal aging process. Thereafter, more direct evidence was provided by several studies using conditional KO strategies, which make meiotic cohesin genes being activated or inactivated at some critical stage(s) of oogenesis. Firstly, inactivation of Smc1β gene shortly after birth at dictyate arrest in oocytes didn’t affect chiasma positions and sister chromatid cohesion (Revenkova et al. 2010). Secondly, activation of an ectopic Rec8 transgene during the growing phase of the oocytes expressing artificially cleavable REC8 by TEV protease doesn’t prevent TEV-mediated bivalent separation (Tachibana-Konwalski et al. 2010). Thirdly, a recent study using drug-inducible Cre system also shows that REC8 can establish cohesion when it is expressed only during meiotic S phase but not when expressed in later meiotic stages (Burkhardt et al. 2016). These studies suggest that only the meiotic cohesin expressed prior to meiotic S phase can establish and maintain sister chromatid cohesion and also imply that there is little or no cohesin turnover during the meiotic arrest at prophase I and thereafter. Indeed, immunofluorescence analyses show that chromosome-associated REC8 present on both chromosome arms and centromeres is reduced in aged oocytes compared with young oocytes, although total REC8 protein levels are similar in both types of oocytes (Chiang et al. 2010: Lister et al. 2010). Furthermore, chromosome-associated Sgo2, the protector of centromeric cohesin, is also reduced in the aged oocytes (Lister et al. 2010).
It seems indubitable that reduction of cohesin during prophase I arrest contributes to chromosome segregation errors in aged oocytes. However, factors other than cohesin degradation are also needed to explain the increase in maternal age-related aneuploidy. As has been pointed out by Hunt and Hassold (2010), the age-dependent increase in aneuploidy and decrease in cohesin are not completely synchronized: the increase in aneuploidy is only evident in reproductively senescent females although the studies using naturally aged mice report a linear age-related decline in chromosome-associated cohesin. Furthermore, provided that only the cohesin expressed during or prior to meiotic S phase plays a role, how can meiotic cohesins in human oocytes keep chromosome cohesion tens of times longer than those in mouse oocytes? We haven’t got any clue to answer this question. In human oocytes, reduction of cohesin during prophase I arrest is still disputable (Garcia-Cruz et al. 2010; Tsutsumi et al. 2014).
8 Condensins in Mitosis
Condensin is a multi-subunit protein complex that is essential for chromosome condensation and segregation (Hirano 2016). Most of eukaryotes have two types of condensins, condensin I and condensin II. Both condensin complexes are composed of five subunits, that is, SMC2 and SMC4 common to condensin I and condensin II; CAP-D2, CAP-G, and CAP-H unique to condensin I; and CAP-D3, CAP-G2, and CAP-H2 unique to condensin II (Table 15.1). Both condensin complexes are essential for construction of mitotic chromosomes and thus development of embryos in many species. In contrast to cohesin that connects the DNA segment from different chromatids, condensin is thought to connect the DNA segment from the same chromatid, thereby contributing to chromosome compaction (Fig. 15.4).
9 The Expression and Localization of Condensins During Mammalian Gametogenesis
Only several studies have been conducted to elucidate the expression and localization of condensins during mammalian gametogenesis. In the first study, it has been shown that condensin I is localized mainly around telomeres and to a lesser extent along chromosome axes (Viera et al. 2007). Thereafter, the expression of both subunits of condensin I and condensin II has been reported in pig oocytes, but their localization on chromosomes has not been examined (Lisková et al. 2010). The first comprehensive study on both condensin I and II in mammalian meiosis has been conducted in mouse oocytes (Lee et al. 2011). Although both condensins are localized on mitotic and meiotic chromosomes, their dynamics are slightly modified in the oocytes. Prior to germinal vesicle breakdown (GVBD) (nuclear envelop disassembly), condensin I is present in the cytoplasm whereas condensin II is localized in the germinal vesicle (GV) (oocyte nucleus). It has been reported in somatic HeLa cells that condensin I and condensin II are present in the cytoplasm and in the nucleus at interphase, respectively (Ono et al. 2003). Thus, the localizations of condensins prior to nuclear envelop disassembly are similar in somatic cells and oocytes. After GVBD, condensin I localizes mainly around centromere regions of bivalent chromosomes while condensin II localizes along chromatid axes. After anaphase I, both condensin I and II are localized on chromosome arms. Why is only condensin II, but not condensin I, stably localized on arm region of bivalent chromosomes? The meiotic cohesin along chromosome arms might affect the loading of condensin I. It has been also suggested that meiotic cohesin along arms might contribute to the solidity of bivalent chromosome in place of condensin I (Lee 2013). Indeed, it has been proposed that that meiotic cohesin contributes not only to the cohesion but also to the organization of chromosome axes and loop (Novak et al. 2008; Haering and Jessberger 2012).
10 Role of Condensins in Construction of Bivalent Chromosomes in Oocytes
The role of condensins in mammalian oocytes has been investigated using two strategies. The first study using antibody injection suggests that both condensins I and II are essential for proper construction of bivalent chromosomes because injection of antibodies specific to condensin I or condensin II subunit affected the shape of chromosomes (Lee and Hirano 2011). However, in this strategy, the injected antibody might affect the other proteins localizing proximal to condensin I, thereby causing the defects in chromosome morphology indirectly. The second study using conditional knockout strategy has shown that knockout of condensin II subunit causes defects in chromosome compaction, chromatid disentanglement, and chromosome segregation during meiosis I while knockout of condensin I subunit hardly affects meiotic progression to metaphase II (Houlard et al. 2015). Therefore, it has been proposed that condensin I may function redundantly with condensin II and may be dispensable for meiotic maturation of oocytes. However, the conditional knockout strategy cannot exclude possibility that residual amount of condensin I due to insufficient depletion in the oocytes might influence the results. In spite of the discrepancy in the meiotic role of condensin I, both strategies argue that condensin II is essential for proper construction of bivalent chromosomes. The artificial inactivation of a condensin II subunit by TEV protease in oocytes further supports this view and suggests that condensin II is essential not only for formation but also for maintenance of bivalent chromosomes (Houlard et al. 2015).
11 Possible Roles of Condensins at Meiotic Prophase I
Although no studies have ever addressed the function of condensins at prophase I in mammals, the involvement of condensins in chromosome dynamics at prophase I has been reported in various species. In budding yeast, condensin I regulates various chromosome events including chromosome compaction, SC assembly, formation and processing of DSBs, repair at rDNA gene clusters, and resolution of recombination-dependent chromosome link (Yu and Koshland 2003; Li et al. 2014). In C. elegans, condensin I regulates the number and distribution of crossovers through construction of a higher-order chromosome structure (Mets and Meyer 2009). In a plant (A. thaliana), it has been suggested that condensin is involved in the regulation of crossover frequency (Smith et al. 2014). Thus, future studies are necessary to investigate possible roles of condensins at prophase I during mammalian gametogenesis.
12 Perspectives
In addition to the abovementioned roles, cohesins, condensins, and the related complexes are involved in multiple functions of chromosomes, such as genomic imprinting (Wendt et al. 2008), dosage compensation (Wood et al. 2010), and transvection (Hartl et al. 2008). Furthermore, the mutation of cohesin or condensin is one of the major causes for various congenital disorders in human (Peters et al. 2008; Martin et al. 2016). Notably, it has been reported that condensin I is recruited to chromosomes in a different manner in male and female pronuclei of mouse zygotes (Bomar et al. 2002). Thus, it is intriguing to search for unproved functions of cohesins and condensins in gametogenesis and early development of mammals.
References
Amanai M, Shoji S, Yoshida N, Brahmajosyula M, Perry AC (2006) Injection of mammalian metaphase II oocytes with short interfering RNAs to dissect meiotic and early mitotic events. Biol Reprod 75:891–898
Bannister LA, Reinholdt LG, Munroe RJ, Schimenti JC (2004) Positional cloning and characterization of mouse mei8, a disrupted allelle of the meiotic cohesin Rec8. Genesis 40:184–194
Biswas U, Hempel K, Llano E, Pendas A, Jessberger R (2016) Distinct roles of meiosis-specific cohesin complexes in mammalian spermatogenesis. PLoS Genet 12:e1006389
Boateng KA, Bellani MA, Gregoretti IV, Pratto F, Camerini-Otero RD (2013) Homologous pairing preceding SPO11-mediated double-strand breaks in mice. Dev Cell 24:196–205
Bomar J, Moreira P, Balise JJ, Collas P (2002) Differential regulation of maternal and paternal chromosome condensation in mitotic zygotes. J Cell Sci 115:2931–2940
Brieño-Enríquez MA, Moak SL, Toledo M, Filter JJ, Gray S, Barbero JL, Cohen PE, Holloway JK (2016) Cohesin removal along the chromosome arms during the first meiotic division depends on a NEK1-PP1g-WAPL axis in the mouse. Cell Rep 17:977–986
Burgoyne PS, Mahadevaiah SK, Turner JM (2009) The consequence of asynapsis form mammalian meiosis. Nat Rev Genet 10:207–216
Burkhardt S, Borsos M, Szydlowska A, Godwin J, Williams SA, Cohen PE, Hirota T, Saitoh M, Tachibana-Kowalski K (2016) Chromosome cohesion established by Rec8-cohesin in fetal oocytes is maintained without detectable turnover in oocytes arrested for months in mice. Curr Biol 26:678–685
Chiang T, Duncan FE, Schindler K, Schultz RM, Lampson MA (2010) Evidence that weakened centromere cohesion is a leading cause of age-related aneuploidy in oocytes. Curr Biol 20:1522–1528
de Vries FA, de Boer E, van den Bosch M, Baarends WM, Ooms M, Yuan L, Liu JG, van Zeeland AA, Heyting C, Pastin A (2005) Mouse Sycp1 functions in synaptonemal complex assembly, meiotic recombination, and XY body formation. Genes Dev 19:1376–1389
Eijpe M, Heyting C, Gross B, Jessberger R (2000) Association of mammalian SMC1 and SMC3 proteins with meiotic chromosomes and synaptonemal complexes. J Cell Sci 113:673–682
Eijpe M, Offenberg H, Jessberger R, Revenkova E, Heyting C (2003) Meiotic cohesin REC8 marks the axial elements of rat synaptonemal complexes before cohesins SMC1beta and SMC3. J Cell Biol 160:657–670
Fukuda T, Fukuda N, Agostinho A, Hernández-Hernández A, Kouznetsova A, Höög C (2014) STAG3-mediated stabilization of REC8 cohesin complexes promotes chromosome synapsis during meiosis. EMBO J 33:1243–1255
Garcia-Cruz R, Brieño MA, Roig I, Grossmann M, Velilla E, Pujol A, Cabero L, Pessarrodona A, Barbero JL, Garcia Caldés M (2010) Dynamics of cohesin proteins REC8, STAG3, SMC1β and SMC3 are consistent with a role in sister chromatid cohesion during meiosis in human oocytes. Hum Reprod 25:2316–2327
Gruber S, Haering CH, Nasmyth K (2003) Chromosomal cohesin forms a ring. Cell 112:765–777
Gutiérrez-Caballero C, Herrán Y, Sánchez-Martín MS, Suja JA, Barbero JL, Llano E, Pendáz AM (2011) Identification and molecular characterization of the mammalian kleisin RAD21L. Cell Cycle 10:1477–1487
Gyuricza MR, Manheimer KB, Apte V, Krishnan B, Joyce EF, Mckee BD, McKim KS (2016) Dynamic and stable cohesins regulate synaptonemal complex assembly and chromosome segregation. Curr Biol 26:1688–1698
Haering CH, Jessberger R (2012) Cohesin in determining chromosome architecture. Exp Cell Res 318:1386–1393
Hartl TA, Smith HF, Bosco G (2008) Chromosome alignment and transvection are antagonized by condensin II. Science 322:1384–1387
Hassold T, Hunt P (2001) To err (meiotically) is human: the genesis of human aneuploidy. Nat Rev Genet 2:280–291
Herbert M, Levasseur M, Homer H, Yallop K, Murdoch A, McDougall A (2003) Homologue disjunction in mouse oocytes requires proteolysis of securin and cyclin B1. Nat Cell Biol 5:1023–1025
Herrán Y, Gutiérrez-Caballero C, Sánchez-Martín M, Hernández T, Viera A, Barbero JL, de Álava E, de Rooij DG, Suja JA, Llano E, Pendás AM (2011) The cohesin subunit RAD21L functions in meiotic synapsis and exhibits sexual dimorphism in fertility. EMBO J 30:3091–3105
Hirano T (2016) Condensin-based chromosome organization from bacteria to vertebrates. Cell 164:847–857
Hodges CA, Revenkova E, Jessberger R, Hassold TJ, Hunt PA (2005) SMC1beta-deficient female mice provide evidence that cohesins are a missing link in age-related nondisjunction. Nat Genet 37:1351–1355
Houlard M, Godwin J, Metson J, Lee J, Hirano T, Nasmyth K (2015) Condensin confers the longitudinal rigidity of chromosomes. Nat Cell Biol 17:771–781
Hunt P, Hassold T (2010) Female meiosis: coming unglued with age. Curr Biol 20:R699–R702
Ishiguro K, Kim J, Fujiyama-Nakamura S, Kato S, Watanabe Y (2011) A new meiosis-specific cohesin complex implicated in the cohesin code for homologous pairing. EMBO Rep 12:267–275
Ishiguro K, Kim J, Shibuya H, Hernández-Hernández A, Suzuki A, Fukagawa T, Shioi G, Kiyonari H, Li XC, Schimenti J, Höög C, Watanabe Y (2014) Meiosis-specific cohesin mediates homolog recognition in mouse spermatocytes. Genes Dev 28:594–607
Jin F, Hamada M, Malureanu L, Jeganathan KB, Zhou W, Morbeck DE, van Deursen JM (2010) Cdc20 is critical for meiosis I and fertility of female mice. PLoS Genet 6:e1001147
Keeney S (2001) Mechanism and control of meiotic recombination initiation. Curr Top Dev Biol 52:1–53
Kim J, Ishiguro K-I, Nambu A, Akiyoshi B, Yokobayashi S, Kagami A, Ishiguro T, Pendas AM, Takeda N, Sakakibara Y, Kitajima T, Tanno Y, Sakuno T, Watanabe Y (2015) Meikin is a conserved regulator of meiosis-I-specific kinetochore function. Nature 512:466–471
Klein F, Mahr P, Galova M, Buonomo SB, Michelis C, Nairz K, Nasmyth K (1999) A central role for cohesins in sister chromatid cohesion, formation of axial elements, and recombination during yeast meiosis. Cell 98:91–103
Kudo NR, Wassmann K, Anger M, Schuh M, Wirth KG, Xu H, Helmhart W, Kudo H, McKay M, Maro B, Ellenberg J, de Boer P et al (2006) Resolution of chiasmata in oocytes requires separase-mediated proteolysis. Cell 126:135–146
Kudo NR, Anger M, Peters AHFM, Stemmann O, Theussi H-C, Helmhart W, Kudo H, Heyting C, Nasmyth K (2009) Role of cleavage by separase of the Rec8 kleisin subunit of cohesin during mammalian meiosis I. J Cell Sci 122:2686–2698
Kuroda M, Oikawa K, Ohbayashi T, Yoshida K, Yamada K, Mimura J, Matsuda Y, Fujii-Kuriyama Y, Mukai K (2005) A dioxin sensitive gene, mammalian WAPL, is implicated in spermatogenesis. FEBS Lett 579:167–172
Lee J (2013) Roles of cohesin and condensin in chromosome dynamics during mammalian meiosis. J Reprod Dev 59:431–436
Lee J, Hirano T (2011) RAD21L, a novel cohesin subunit implicated in linking homologous chromosomes in mammalian meiosis. J Cell Biol 192:263–276
Lee J, Iwai T, Yokota T, Yamashita M (2003) Temporally and spatially selective loss of Rec8 protein from meiotic chromosomes during mammalian meiosis. J Cell Sci 116:2781–2790
Lee J, Okada K, Ogushi S, Miyano T, Miyake M, Yamashita M (2006) Loss of Rec8 from chromosome arm and centromere region is required for homologous chromosome separation and sister chromatid separation, respectively, in mammalian meiosis. Cell Cycle 5:1448–1455
Lee J, Kitajima TS, Tanno Y, Yoshida K, Morita T, Miyano T, Miyake M, Watanabe Y (2008) Unified mode of centromeric protection by shugoshin in mammalian oocytes and somatic cells. Nat Cell Biol 10:42–52
Lee J, Ogushi S, Saitou M, Hirano T (2011) Condensins I and II are essential for construction of bivalent chromosomes in mouse oocytes. Mol Biol Cell 22:3465–3477
Li P, Jin H, H-G Y (2014) Condensin suppresses recombination and regulates double-strand break processing at the repetitive ribosomal DNA array to ensure proper chromosome segregation during meiosis in budding yeast. Mol Biol Cell 25:2934–2947
Lisková L, Susor A, Pivonkova K, Saskova A, Karabinova P, Kubelka M (2010) Detection of condensin I and II in maturing pig oocytes. Reprod Fertil Dev 22:644–652
Lister LM, Kouznetsova A, Hyslop LA, Kalleas D, Pace SL, Barel JC, Nathan A, Floros V, Adelfalk C, Watanabe Y, Jessberger R, Kirkwood TB, Höög C, Herbert M (2010) Age-related meiotic segregation errors in mammalian oocytes are preceded by depletion of cohesin and Sgo2. Curr Biol 20:1511–1521
Llano E, Herran Y, Garcia-Tunon I, Gutierrez-Caballero C, de Alava E, Barbero JL, Schimenti J, de Rooij DG, Sanchez-Martin M, Pendas AM (2012) Meiotic cohesin complexes are essential for the formation of the axial element in mice. J Cell Biol 197:877–885
Martin C-A, Murray JE, Carroll P, Leitch A, Mackenzie KJ, Halachev M, Fetit AE, Keith C, Bicknell LS, Fluteau A et al (2016) Mutations in genes encoding condensin complex proteins cause microcephaly through decatenation failure at mitosis. Genes Dev 30:2158–2172
Mets DG, Meyer BJ (2009) Condensins regulate meiotic DNA break distribution, thus crossover frequency, by controlling chromosome structure. Cell 139:73–86
Miyazaki WY, Orr-Weaver TL (1994) Sister-chromatid cohesion in mitosis and meiosis. Annu Rev Genet 28:167–187
Morelli MA, Cohen PE (2005) Not all germ cells are created equal: aspects of sexual dimorphism in mammalian meiosis. Reproduction 130:761–781
Nasmyth K (2011) Cohesin: a catenase with separate entry and exit gates? Nat Cell Biol 13:1170–1177
Nasmyth K, Haering CH (2009) Cohesin: its roles and mechanisms. Annu Rev Genet 43:525–558
Nishiyama T, Ladurner R, Schmitz J, Kreidl E, Schleiffer A, Bhaskara V, Bando M, Shirahige K, Hyman AA, Mechtler K, Peters JM (2010) Sororin mediates sister chromatid cohesion by antagonizing Wapl. Cell 143:737–749
Novak I, Wang H, Revenkova E, Jessberger R, Scherthan H, Hoog C (2008) Cohesin Smc1beta determines meiotic chromatin axis loop organization. J Cell Biol 180:83–90
Ono T, Losada A, Hirano M, Myers MP, Neuwald AF, Hirano T (2003) Differential contributions of condensin I and condensin II to mitotic chromosome architecture in vertebrate cells. Cell 115:109–121
Page SL, Hawley RS (2004) The genetics and molecular biology of the synaptonemal complex. Annu Rev Cell Dev Biol 20:525–558
Parra MT, Viera A, Gómez R, Page J, Benavente R, Santos JL, Rufas JS, Suja JA (2004) Involvement of the cohesin Rad21 and SCP3 in monopolar attachment of sister kinetochores during mouse meiosis I. J Cell Sci 117:1221–1234
Pelttari J, Hoja M-R, Yuan L, Liu J-G, Brundell E, Moens P, Santucci-Darmnin S, Jessberger R, Barbero JL, Heyting C, Höög C (2001) A meiotic chromosomal core consisting of cohesin complex proteins recruits DNA recombination proteins and promotes synapsis in the absence of an axial element in mammalian meiotic cells. Mol Cell Biol 21:5667–5677
Peters JM, Tedeschi A, Schmitz J (2008) The cohesin complex and its roles in chromosome biology. Genes Dev 22:3089–3114
Prieto I, Suja JA, Pezzi N, Kremer L, Martinez AC, Rufas JS, Barbero JL (2001) Mammalian STAG3 is a cohesin specific to sister chromatid arms in meiosis I. Nat Cell Biol 3:761–766
Prieto I, Pezzi N, Buesa J, Kremer L, Barthelemy I, Carreiro C, Roncal F, Martínez A, Gómez L, Fernández R, Martínez A, Barbero J (2002) STAG2 and Rad21 mammalian mitotic cohesins are implicated in meiosis. EMBO Rep 3:543–550
Revenkova E, Eijpe M, Heyting C, Gross B, Jessberger R (2001) Novel meiosis-specific isoform of mammalian SMC1. Mol Cell Biol 21:6984–6998
Revenkova E, Eijpe M, Heyting C, Hodges CA, Hunt PA, Liebe B, Scherthan H, Jessberger R (2004) Cohesin SMC1 beta is required for meiotic chromosome dynamics, sister chromatid cohesion and DNA recombination. Nat Cell Biol 6:555–562
Revenkova E, Herrmann K, Adelfalk C, Jessberger R (2010) Oocyte cohesin expression restricted to predictyate stages provides full fertility and prevents aneuploidy. Curr Biol 20:1529–1533
Rong M, Matsuda A, Hiraoka Y, Lee J (2016) Meiotic cohesin subunits RAD21L and REC8 are positioned at distinct regions between lateral elements and transverse filaments in the synaptonemal complex of mouse spermatocytes. J Reprod Dev 62:623–630
Rong M, Miyauchi S, Lee J (2017) Ectopic expression of meiotic cohesin RAD21L promotes adjacency of homologous chromosomes in somatic cells. J Reprod Dev. doi:10.1262/jrd.2016-171
Sakakibara Y, Hashimoto S, Nakaoka Y, Kouznetsova A, Höög C, Kitajima TS (2015) Bivalent separation into univalents precedes age-related meiosis I errors in oocytes. Nat Commun 6:7550
Santucci-Darmanin S, Walpita D, Lespinasse F, Desnuelle C, Ashley T, Paquis-Flucklinger V (2000) MSH4 acts in conjunction with MLH1 during mammalian meiosis. FASEB J 14:1539–1547
Shintomi K, Hirano T (2009) Releasing cohein from chromosome arms in early mitosis: opposing actions of Wapl-Pds5 and Sgo1. Genes Dev 23:2224–2236
Smith SJ, Osman K, Franklin CH (2014) The condensin complexes play distinct roles to ensure normal chromosome morphogenesis during meiotic division in Arabidopsis. Plant J 80:255–268
Suja JA, Barbero JL (2009) Cohesin complexes and sister chromatid cohesion in mammalian meiosis. Genome Dyn 5:94–116
Tachibana-Konwalski K, Godwin J, van der Weyden L, Champion L, Kudo NR, Adams DJ, Nasmyth K (2010) Rec8-containing cohesin maintains bivalents without turnover during the growing phase of mouse oocytes. Genes Dev 24:2505–2516
Tachibana-Konwalski K, Godwin J, Borsos M, Rattani A, Adams DJ, Nasmyth K (2013) Spindle assembly checkpoint of oocytes depends on a kinetochore structure determined by cohesin in meiosis I. Curr Biol 23:2534–2539
Terret ME, Wassmann K, Waizenegger I, Maro B, Peters JM, Verlhac MH (2003) The meiosis I-to-meiosis II transition in mouse oocytes requires separase activity. Curr Biol 13:1797–1802
Tsutsumi M, Fujiwara R, Nishizawa H, Ito M, Kogo H, Inagaki H, Ohye T, Kato T, Fujii T, Kurahashi H (2014) Age-related decrease of meiotic cohesins in human oocytes. PLoS One 9:e96710
Uhlmann F, Nasmyth K (1998) Cohesion between sister chromatids must be established during DNA replication. Curr Biol 8:1095–1101
Uhlmann F, Wernic D, Poupart MA, Koonin EV, Nasmyth K (2000) Cleavage of cohesin by the CD clan protease separin triggers anaphase in yeast. Cell 103:375–386
Viera A, Gomez R, Parra MT, Schmiesing JA, Yokomori K, Rufas JS, Suja JA (2007) Condensin I reveals new insights on mouse meiotic chromosome structure and dynamics. PLoS One 2:e783
Waizenegger IC, Hauf S, Meinke A, Peters JM (2000) Two distinct pathways remove mammalian cohesin from chromosome arms in prophase and from centromeres in anaphase. Cell 103:399–410
Watanabe Y, Nurse P (1999) Cohesin Rec8 is required for reductional chromosome segregation at meiosis. Nature 29:461–464
Wendt KS, Yoshida K, Itoh T, Bando M, Koch B, Schirghuber E, Tsutsumi S, Nagae G, Ishihara K, Mishiro T et al (2008) Cohesin mediates transcriptional insulation by CCCTC-binding factor. Nature 451:796–801
Winters T, McNicoll F, Jessberger R (2014) Meiotic cohesin STAG3 is required for chromosome axis formation and sister chromatid cohesion. EMBO J 33:1256–1270
Wood AJ, Severson AF, Meyer BJ (2010) Condensin and cohesin complexity: the expanding repertoire of functions. Nat Rev Genet 11:391–404
Xu H, Beasley M, Verschoor S, Inselman A, Handel MA, Mckay MJ (2004) A new role for the mitotic RAD21/SCC1 cohesin in meiotic chromosome cohesion and segregation in the mouse. EMBO Rep 5:378–384
Xu H, Beasley MD, Warren WD, van der Horst GT, McKay MJ (2005) Absence of mouse REC8 cohesin promotes synapsis of sister chromatids in meiosis. Dev Cell 8:949–961
Yu H (2007) Cdc20: a WD40 activator for a cell cycle degradation machine. Mol Cell 27:3–16
Yu HG, Koshland DE (2003) Meiotic condensin is required for proper chromosome compaction, SC assembly, and resolution of recombination-dependent chromosome linkages. J Cell Biol 163:937–947
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer International Publishing AG
About this chapter
Cite this chapter
Lee, J. (2017). The Regulation and Function of Cohesin and Condensin in Mammalian Oocytes and Spermatocytes. In: Kloc, M. (eds) Oocytes. Results and Problems in Cell Differentiation, vol 63. Springer, Cham. https://doi.org/10.1007/978-3-319-60855-6_15
Download citation
DOI: https://doi.org/10.1007/978-3-319-60855-6_15
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-60854-9
Online ISBN: 978-3-319-60855-6
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)