Abstract
Alphaviruses are enveloped, single-stranded, plus-strand RNA viruses belonging to the Togaviridae family. These are zoonotics and arthropod-borne viruses (mainly mosquitoes) that are distributed nearly worldwide. Many of the New World alphaviruses occur throughout Latin America: Venezuelan and eastern equine encephalitis viruses (VEEV, EEEV) in both North and South America, and western equine encephalitis virus (WEEV) from Canada to Argentina. Others such as Mayaro (MAYV), UNA (UNAV), and Aura (AURA) viruses have a more restricted distribution, and chikungunya virus (CHIKV) was recently introduced into Latin America. The evolutionary patterns show clades correlated with host/reservoirs (nonhuman primates, birds, and rodents) and distinctive human disease syndromes such as fever/rash/arthralgia and encephalomyelitis. The majority of alphaviruses cause at least mild febrile disease in humans and several produce severe, life-threatening diseases, whereas others are little studied epidemiologically and their public health importance is unknown. Recent studies in Latin America of “dengue-like” illness in several locations have revealed that many alphaviruses such as VEEV, EEEV, MAYV, and CHIKV are misdiagnosed as dengue; moreover, with the recent introduction of Zika virus, diagnosis based only on signs and symptoms is even more complicated in areas where these viruses are circulating simultaneously. This chapter describes important aspects of the alphaviruses in the region including evolution, outbreaks, vector–host, and eco-epidemiological/molecular determinants of their emergence from the New World viruses and chikungunya.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
The Togaviridae is a family of enveloped, single-stranded, plus-strand RNA viruses composed of the genera Alphavirus and Rubivirus. The rubella virus (the cause of German measles) is the only member of the latter genus [129]. The Alphaviruses are arthropod-borne viruses (mainly mosquitoes) with a nearly worldwide geographic distribution, having been reported from all continents except Antarctica and from many islands [16].
The genus Alphavirus includes 30 species grouped into 10 complexes based on antigenic and/or genetic similarities [75]. The Barmah Forest, Ndumu, Middelburg, and Semliki Forest complexes occur almost exclusively in the Old World. In the New World, the first alphaviruses to be isolated were western equine encephalitis virus (WEEV) in 1930, eastern equine encephalitis virus (EEEV) in 1933, and Venezuelan equine encephalitis virus (VEEV) in 1938 [129]. Other Alphaviruses found in Latin America include Mayaro (MAYV), Aura (AURAV), Una (UNAV), Trocara virus (TROV), and the recently introduced chikungunya virus (CHIKV) (Table 9.1). Others found in North America belonging to the WEE complex, Highlands J (HJV), and Fort Morgan viruses (FMV) [129] are recombinants resembling WEEV derived from ancestral EEEV and Sindbis (SINV) from the Old World.
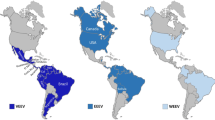
Many of the New World alphaviruses are widely distributed throughout the Americas. WEEV is found from Canada to Argentina, and EEEV and VEEV occur in both North America and South America. Other viruses such as MAYV and AURAV have a more restricted neotropical distribution [106].
Recent phylogenetic analyses depict the evolution of the alphaviruses (Fig. 9.1) based on structural protein genes. Clades or branches are clearly correlated with the antigenic complexes, host/reservoirs, and disease syndromes: the Semliki Forest virus complex is associated with nonhuman primates and human fever/rash/arthralgia (including chikungunya, Mayaro, Ross River, Semliki Forest). The WEEV/EEEV complexes associate with birds, and the VEEV complex is mostly related to rodents as reservoirs, and all three with human and equine encephalitis.
Phylogenetic tree showing the evolutionary hypothesis of Alphaviruses. The viruses in bold are neotropical and New World viruses; those in gray are the Old World viruses (including the mosquito-specific virus Eilat). Lines on right delimit the clades of viruses associated with a serogroup complex, vertebrate hosts, and symptoms (arthralgia and encephalomyelitis). (Modified from Weaver et al. [129])
The most recently described alphavirus, Eilat (EILV) , a “mosquito-specific” virus from Anopheles coustani mosquitoes in Israel [75], is a sister of the WEEV complex (see Fig. 9.1). In contrast to all other mosquito-borne viruses, it is unable to replicate in vertebrate cell lines. EILV has important implications for arbovirus evolution and may help elucidate the viral factors responsible for the virus–cell interactions of pathogenic alphaviruses, facilitate vaccine development, and help develop strategies to control or prevent alphavirus transmission [28, 74, 75, 138].
Figure 9.2 shows a cartoon of alphavirus transmission cycles . All alphaviruses except EILV are zoonotic: mosquitoes (family Culicidae) are the major vector group, especially the genera Aedes (chikungunya), Culex subgenus Melanoconion (VEEV, EEEV, WEEV), and Haemagogus and Sabethes for Mayaro [26, 72, 84, 112]. In concordance with the phylogenetic tree, enzootic transmission cycles involve nonhuman primates (chikungunya, Mayaro, others that produce arthralgias), birds (EEEV, WEEV), and rodents (VEEV, possibly Madariaga virus). Spillover transmission to humans occurs mainly in rural areas but can become urban for CHIKV and potentially others. Affected animals (including humans) usually generate insufficient viremia to participate in the transmission cycle (i.e., dead-end hosts). VEEV uses equids (horses, donkeys, mules) as amplification hosts (epizootics) to increase spillover to humans (epidemics) [126]. CHIKV is the only alphavirus known to utilize humans as amplification hosts and the urban mosquitoes [Aedes (Stegomyia) aegypti or Aedes (Stegomyia) albopictus] for transmission, resulting in major epidemics [47, 129]. Mayaro, similar to CHIKV in Africa, uses nonhuman primates as enzootic hosts , but their role is still not well understood [72].
Transmission cycles and mechanisms of human infection by alphaviruses. At the center is an enzootic cycle, typically involving avian, rodent, or nonhuman primates as amplification or reservoir hosts and mosquito vectors. Humans become infected via direct spillover when they enter enzootic habitats or when amplification results in high levels of circulation. Transmission to humans may involve the enzootic vector or bridge vectors with broader host preferences. Right panel: Secondary amplification involving domestic animals can increase circulation around humans, increasing their chance of infection via spillover. In the case of VEEV, mutations that enhance equine viremia are needed for secondary equine amplification. Left panel: CHIKV can use humans for amplification, resulting in urban epidemic cycles and massive outbreaks . (Modified from Weaver et al. [129] and Muñoz and Navarro [73])
The majority of alphaviruses can cause at least mild febrile disease in humans, with several producing severe, life-threatening diseases; however, many remain poorly studied epidemiologically with unknown public health importance.
Recent studies in several Latin American locations of “dengue-like” illness revealed that alphaviruses such as VEEV, EEEV, MAYV, and CHIKV account for a significant number of cases misdiagnosed clinically as dengue. Moreover, with the recent introduction of the Zika virus, diagnosis based only on signs and symptoms is even more complicated in areas where these viruses are circulating simultaneously.
A better understanding of alphavirus transmission cycles, molecular evolution, vector biology, virus–vector–host coevolution, and rapid diagnostics is needed to prevent alphavirus diseases.
Brief descriptions of the most important alphaviruses in Latin America follow.
2 Alphavirus and Encephalitis
2.1 Venezuelan Equine Encephalitis Virus
The Venezuelan equine encephalitis (VEE) complex includes major human and equine pathogens and consequently is the most thoroughly studied in Latin America [126]. VEEV was recognized first in 1936 and isolated soon thereafter from the brains of fatal equids in Venezuela. This virus is transmitted enzootically between mosquitoes and rodents, and equine-amplified epizootic cycles cause large outbreaks of encephalitis in humans and horses [49, 129]. Outbreaks in Mexico and South America (Colombia, Venezuela, and Peru) demonstrated that VEE is a reemerging disease [44] as a naturally emerging pathogen endemic to South and Central America, Mexico, and Florida [126] circulating among wild rodents and mosquitoes. The introduction of horses, a new and susceptible host, into the Americas during the colonial period triggered outbreaks in these animals and increased the exposure of humans .
Systematics and Geographic Distribution
The VEE complex is a sister of the eastern equine encephalitis (EEE) complex [90] and includes six subtypes. Only subtypes IAB and IC are traditionally considered epizootic strains that use equids for amplification via high-titer viremias. Other VEEV subtypes (ID and IE) are considered equine-avirulent, enzootic strains, although IE strains from recent Mexican epizootics appear to be equine-neurovirulent but incapable of generating high-titer equine viremia [35]. The remaining subtypes (II–VI) are also enzootic strains that generally circulate in sylvatic or swamp habitats and are considered incapable of equine amplification. In the United States, these include the Everglades virus in Florida and a variant of Tonate virus, Bijou Bridge, isolated in Colorado from cliff swallow bugs during the 1970s [17].
Geographic phylogenetic correlations of VEEV subtypes ID enzootic lineages or genotypes [1, 13, 93, 97, 124] suggest that geographic barriers explain their current distributions.
The major epidemic/epizootic subtype IAB and IC strains are highly pathogenic for horses, with case-fatality rates of 20% to 80%. The last major VEE outbreak occurred in 1995 in Venezuela and Colombia with 75,000 to 100,000 human cases, more than 300 of them fatal. In 1993, equine disease was associated with VEEV-IE in Mexico, and since 1993, human cases of VEEV ID-associated disease have occurred in Peru [33, 126].
VEEV infection usually causes flu-like symptoms, and encephalitis is rare in adults. Although the case-fatality rate is low (≤1%), neurological disease, including ataxia, disorientation, mental depression, and convulsions, can be detected in up to 14% of infected individuals, mainly children. High seroprevalence has been detected in humans in interepidemic/epizootic periods in Argentina [87, 88]. Neurological sequelae in humans are also common [34, 49, 94].
Evolution of Epizootic Strains from Enzootic Ancestors
Phylogenetic analysis of the VEE complex shows a close evolutionary relationship among IAB, IC, and ID strains and delineates six major lineages of enzootic VEEV, including five ID-like lineages and the subtype IE lineage. All epizootic strains from major outbreaks fall into one of three clades nested within one of these lineages, which is otherwise composed of enzootic ID strains from western Venezuela, Colombia, and northern Peru. These phylogenetic data support the hypothesis that epizootic VEEV strains have arisen on at least four occasions by mutation of enzootic ID strains and changes in host range. In further investigations, the occurrence of two mutations involving charge alterations on the surface of the E2 protein implies alterations in cellular receptor usage that influences pathogenesis as a mechanism of epizootic emergence [38, 91, 124,125,126].
Epizootic Transmission Cycle
The epizootic transmission cycle of VEEV is fairly well understood [91]. A feature common to all major outbreaks is the role of equids as highly efficient amplification hosts. Although the vertebrate host range of epizootic VEEV strains is wide and includes humans, rodents, bats, dogs, sheep, and some birds, major epidemics in the absence of equine cases have never occurred. Despite the repeated occurrence of epizootics near major cities such as Maracaibo (1995), in western Venezuela , interhuman mosquito-borne transmission has not been detected. However, the potential for urban transmission by a species such as Aedes aegypti, which is susceptible to infection after biting humans and exhibits behavioral traits such as multiple host feeding and peri-domesticity that augment its vector competence [43, 54, 80], or the continuous expansion of Aedes albopictus should be considered [12, 30, 68, 77], as human populations continue to expand and those of equines decline in rural areas in Latin America [85, 118]. Several small, atypical equine outbreaks were detected in Venezuela in the central llanos (2000–2003) with VEEV sequences showing high genomic stability 10 years after the 1995 outbreak. Cattle sero-surveys indicated the recent circulation of enzootic VEEV strains, and possibly of epizootic strains. Persistence of VEEV subtype IC strains and infection of horses at the end of the rainy season suggested the possibility of an alternative, cryptic transmission cycle involving survival through the dry season of infected vectors or persistently infected vertebrates [76].
Epizootic Vectors
Epizootic strains (subtype IAB and IC) of VEEV are opportunistic in their use of mosquito vectors during outbreaks. Field studies have indicated that more than one principal vector species can be involved in transmission [99, 122, 123, 136]. Although susceptibility to infection is a prerequisite for biological transmission, ecological and behavioral traits can be more important than susceptibility differences in vectorial capacity.
Although several mosquito species have been incriminated as VEEV vectors during epizootics, Aedes (Ochlerotatus) taeniorhynchus , a salt-marsh mosquito, may be the most important epizootic vector. This species is abundant in coastal areas including the Guajira Peninsula (Colombia and Venezuela), where many of the largest outbreaks have occurred, and virus isolations and susceptibility studies have documented its role in transmission [51, 107, 116, 136]. Culex (Deinocerites) spp. may also be VEEV vectors in coastal areas [37].
Psorophora confinnis and P. columbiae were probably important vectors during outbreaks in northern South America and in the 1971 epizootic/epidemic in northern Mexico and Texas [136]. Aedes (Ochlerotatus) sollicitans also exhibited extremely high infection rates in the coastal areas of Mexico and Texas in 1971 [136] and is capable of laboratory transmission following high-titer blood meals [116]. Non-mosquito arthropods (blackflies and ticks) have also been implicated as VEEV vectors but appear to be less important [55,56,57, 99].
Enzootic Transmission Cycle: Hosts and Vectors
Sylvatic rodents in the genera Sigmodon, Oryzomys, Zygodontomys, Heteromys, Peromyscus, and Proechimys are believed to be the principal reservoir hosts of most enzootic VEE complex viruses because they are frequently infected in nature, have high rates of immunity, and develop moderate- to high-titer viremia [23, 24, 126]. Spiny rats (Proechimys semispinosus) and cotton rats (Sigmodon hispidus) are the principal reservoir hosts of enzootic subtype ID viruses in Panama and also in Colombia and Venezuela [13, 14, 63, 79, 126]. Comparative studies in Venezuela and Colombia demonstrated a strong correlation between spiny rat (Proechimys chrysaeolus in Colombia) populations and levels of VEEV circulation [13]. Other mammals such as opossums (Didelphis marsupialis) are also frequently infected, and bats and shorebirds may be involved in the dispersal of enzootic viruses.
The most important enzootic vectors are members of the genus Culex, subgenus Melanoconion, Spissipes section [9, 25, 31, 71, 78, 101, 123, 126, 134], a diverse and taxonomically difficult group [78, 83, 98, 112]. The Spissipes section [98, 104, 112] includes most vectors of enzootic VEEV and EEEV in Latin America [18, 126], and seven species are proven vectors of VEE complex viruses. Studies of enzootic VEEV ecology have incriminated a single species or multiple species as the principal vector in a given location [9, 13, 31, 69, 123, 134]. A combination of enzootic vectors (transmission within forests) and epizootic vectors (potential exporters of the virus to open agricultural areas) were also studied in enzootic areas of the Catatumbo region in Venezuela [5, 67].
The restriction of most Melanoconion arbovirus vectors to the Spissipes section raises the question of what genetic, physiological, or ecological characteristics are shared by the members of this section that predispose them to transmit arboviruses. Recently, the use of ribosomal DNA sequences and phylogenetic methods have revealed evolutionary relationships among the Vomerifer and Pedroi groups of Spissipes [78]. Navarro and Weaver also detected two cryptic potential vector species under Culex pedroi supporting the hypothesis of differential vector capacity for VEEV and EEEV [31, 117].
Control and Prevention of VEE Outbreaks
Equine vaccination in enzootic countries, where progenitors of epizootic strains circulate and where recent outbreaks have been documented, can be effective if VEEV circulation is anticipated or recognized quickly during outbreaks. However, governmental responses to epizootics are often slow for reasons of veterinary and public heath surveillance deficiencies. The live-attenuated TC-83 vaccine is the most effective way to prevent and control epizootic VEEV transmission, and it is available throughout most of Latin America. However, some equids in South America are vaccinated with inactivated, multivalent alphavirus vaccines marketed in the United States. Immunity from these vaccines is slower to develop, is less durable, and requires frequent boosters. Therefore, public and veterinary health officials should strongly discourage the use of these inactivated vaccines in regions of Latin America with a history of VEE [126]. The protection of human populations relies principally on personal protection and avoidance of mosquito bites by limiting physical exposure and applying repellants containing the active ingredient diethylmethylbenzamide (DEET). Applying permethrin to clothing to enhance protection of individuals who reside or work near equine herds during epizootics, who contact tropical forest or swamp habitats where enzootic VEEV circulates, or during outbreaks is also effective. The rural–sylvan behavior of enzootic VEEV vectors renders the usual control based on ULV insecticide methods inappropriate and largely ineffective.
2.2 Eastern and Western Equine Encephalitis Viruses
2.2.1 Eastern Equine Encephalitis
Eastern equine encephalitis virus (EEEV) (Alphavirus, Togaviridae) is an single-stranded RNA mosquito-borne zoonotic pathogen transmitted throughout the Americas [105, 135]. In North America, EEEV circulates principally along the east cost of United States and Canada, and related strains have been also detected in northern Mexico and the Caribbean region. In North America, sporadic human cases averaging approximately five or six per year occur in swamp habitats where the enzootic cycle is involved mainly among birds of the Passeriformes order and the ornithophilic mosquito Culiseta melanura and Culex subgenus Melanoconion species in the southeast [105, 135]. EEEV infections in domestic animals are common; the case-fatality rates in both human and equine cases average about 50% to 70% or more.
South American eastern equine encephalitis virus (EEEV/SA) was first characterized in Argentina in 1933 [70]. New evidence shows that North American EEEV (EEEV/NA) and EEEV/SA variants have developed differences in the ecological, epidemiological, pathogenic, antigenic, and genetic profiles that allowed the classification of EEE/SA into a new species called Madariaga virus (MADV), named after the place of its first collection in Argentina. Consequently, MADV is composed of three distinct genetic lineages: one that circulates in Guatemala, Brazil, and Peru; a second lineage in Argentina, Brazil, Colombia, Ecuador, Guyana, Panama, Peru, Venezuela, and Trinidad; and a third represented by a single location isolated in Brazil [8].
Early reports suggest that MADV virus was human avirulent and causes equine epizootics with high mortality averaging approximately 70%; despite human exposure during epizootics, cases have never been detected in Panama and Argentina despite active surveillance [27, 70, 96]. Studies in Peru revealed that although the isolation of MADV in mosquitoes known to feed on humans was not uncommon, no MADV was isolated in acutely febrile patients [3]. Only three human cases of MADV infection had been recognized in Brazil (one) and in Trinidad (two) [21, 32] before the first documented epidemic was detected in Panama during 2010 [18].
Vector and Host
Culex (Melanoconion) taeniopus and Cx. (Mel.) pedroi are recognized as the main enzootic vectors of MADV in Central and South America, respectively [108, 117]. Forest-dwelling rodents and marsupials have been implicated as possible hosts based on serosurveys, although birds may also serve as hosts in the Amazon and southern regions of South America. Lizards have also been suggested as MADV hosts in Panama [22]. Recent serological studies in Panama suggest that the short-tailed cane mouse (Zygodontomys brevicauda) is a host for MADV, and humans active in pastures and farms where this rodent is abundant are at increased risk of infection [121].
Epidemiology
Human MADV infections in Panama are recognized after equine cases have been detected early in the rainy season (May–June). The distribution of cases is typically clustered in the Province of Darien close to the Colombian border. Severe cases are observed principally in children with a median age of 5.1 years, with a case-fatality rate around 10% [18].
With the exception of Panama, there is no report of human disease outbreaks attributed to MADV, and the lack of human cases in the rest of Latin America may result from (a) cross-protective immunity by heterologous alphavirus antibodies such as VEEV or (b) intrinsic characteristics of MADV strains, such as the inability to evade the interferon response [2, 3]. In addition to previous studies, in Panama recent evidence supports the effect of cross-protective immunity, as areas of high VEEV transmission appear to have reduced MADV transmission [121].
Clinical Characteristics
Symptomatic MADV cases present a prodromal phase, principally with fever and headache; vomiting and diarrhea occur less frequently. A neurological stage follows accompanied by disorientation, somnolence, seizures, and coma. Patients typically show elevated white cell counts and protein elevation in the cerebrospinal fluid (CSF). Severe cases can develop long-term neurological sequelae including seizures, hemiparesis, psychomotor retardation, and coma [18, 61].
Laboratory Diagnosis
Laboratory diagnosis is a challenge in endemic countries, where a high level of training to perform the viral isolation and serological assays is required. Co-circulation of multiple alphaviruses and antibody cross-reactions are common in Latin America. Furthermore, in endemic regions where multiple alphaviruses such as VEEV and EEEV (MADV) that cause similar clinical presentations are circulating, the interpretation of laboratory results is complex. The alphavirus IgM antibody response lasts around 2 to 3 months, and multiple diagnostic tools such as viral isolation, antibody tests, and viral RNA detection should be implemented [18, 62]. Although incidental laboratory infections with MADV have not been reported, diagnostic confirmation in endemic regions may require the use of live VEEV and EEEV in biosafety level 3 containment. In this case, neutralization tests for MADV and VEEV can be performed with EEEV chimeric viruses and TC-83 live-attenuated vaccine VEEV strain in biosafety level 2 facilities with results similar to those obtained with the wild-type strains [48, 121].
2.2.2 Western Equine Encephalitis
Western equine encephalitis virus (WEEV) is a recombinant alphavirus descended from Sindbis- and EEEV-like ancestors [40]. WEEV causes sporadic epizootics in the western United States and Canada, associated with increased rainfall in early spring followed by warmer-than-normal temperatures. Culex tarsalis is recognized as the principal vector in North America, and Aedes albifasciatus has been implicated in South America; passerine birds are the main enzootic hosts.
Circulation of WEEV in Latin America has been recognized during epizootics in Argentina, Uruguay, and Cuba [128]. A human fatal case was reported in Uruguay during 2009. However, the available evidence suggests that WEEV circulation is declining, with the last human case in North America reported during 1994, and the last detection in mosquito pools in 2008 [15].
3 Alphavirus and Arthralgias
3.1 Mayaro Virus
Mayaro virus (MAYV) is a unique, exceptional New World alphavirus, and, before the introduction of chikungunya virus (CHIKV) in 2013, represented the sole arthralgic alphavirus endemic to the Western Hemisphere. MAYV was first detected in forest workers in the county of Mayaro, Trinidad, in 1954 [6]. Since then, there is evidence of MAYV infection, either by serological detection or virus isolation, in several regions of Latin America, including Brazil, Colombia, Ecuador, Peru, Surinam, Bolivia, French Guiana, Trinidad, and Venezuela [4, 10, 11, 33, 39, 46, 50, 59, 66, 72, 81, 109,110,111, 113, 120, 137]. Although MAYV has not recently emerged sufficiently to result in major outbreaks, several acute undifferentiated febrile illness surveillance studies have clearly shown that the virus commonly infects humans, and that those infected most are infected as a result of occupational exposure [33, 120].
MAYV causes sporadic outbreaks, which have been localized primarily to regions of Brazil [11, 53, 64, 82, 120, 137], but has also been detected in Bolivia and Peru [33]. A pediatric infection was recently detected in Haiti, suggesting local enzootic (although no wild monkeys are present) or endemic circulation [139]. Typically, infection with MAYV is not fatal but cases usually present with fever, headache, retro-orbital pain, myalgia, vomiting, diarrhea, rash, and often persistent (<1 year) severe arthralgia [41, 100]. In this regard, MAYV may be more incapacitating than other common arboviruses such as dengue virus (DENV). Given the low economic impact of MAYV, there is very little vaccine development effort, the exception being an IRES-based attenuated live-attenuated vaccine [133]. The largest documented MAYV outbreak occurred in Belterra, Brazil, in 1978, with approximately 790 persons possibly affected and 55 confirmed cases: the virus was isolated from 43 cases [53, 86]. Since then, outbreaks have been very limited, until recently in Venezuela in 2010, where there was an outbreak of 77 suspected cases, of which 6 were detected by virus isolation from acute-phase sera [10].
Given the extent of the most recent MAYV outbreak in Venezuela, it is important to consider the possibility that Aedes (Steg) aegypti was involved in virus transmission, in addition to its enzootic vectors. Ae. aegypti-vectored arboviruses are among the most important arboviral pathogens, and previous studies suggest this is a moderately competent MAYV vector [60]. Ae. albopictus is a competent vector for several arboviruses [131], and in contrast to Ae. aegypti, it is found in periurban areas and more temperate regions. Ae. albopictus competency for MAYV has been examined experimentally, and results suggest it is not a competent vector unless bloodmeals are taken from highly viremic mice (>7 logs). If MAYV were to adapt for more efficient Ae. aegypti or Ae. albopictus transmission, it can present a significant global threat [140].
Phylogenetic studies of MAYV sequences show that the viruses can be further delineated into three genotypes, designated genotype D, L, and N [10, 89]. Genotype D includes isolates from Trinidad, Brazil, French Guiana, Surinam, Peru, and Bolivia; genotype L contains isolates from Brazil, but it is unclear if this genotype is still in circulation because it has not been detected since 1991; and genotype N consists of a single strain isolated from Peru in 2010 [10]. The single genotype N sequence is intermediary in phylogeny between genotypes D and L. It would be interesting to determine the prevalence of this strain in Peru and fully characterize its pathogenicity in mice relative to the other genotypes.
There is no evidence that the MAYV phylogeny is temporally structured, but it appears to be influenced to some extent by geography. Genotype D strains can be further delineated into smaller clades based on the geographic region of collection [10, 89]. Given this geographically structured phylogeny, we cannot exclude the possibility that there may be potential restrictions associated with vector competence, vector distributions, or alternative vertebrate amplification hosts that might affect this apparent population subdivision. However, sampling bias and the difficulty associated with isolating MAYV should be considered when proposing these conclusions. Additionally, recent studies provide evidence that MAYV strains concurrently circulating within Venezuela are undergoing regionally independent evolution, suggesting the absence of a single panmictic viral population, at least in Venezuela [10].
The MAYV ecological niche model based on localities of virus isolations [10, 89], vector distributions, and 19 climatic variables [58] predicts the suitable MAYV habitats in the following order of importance: eco-regions, followed by Haemagogus (Hg.) leucocelaenus, Hg. celeste, and Hg. clarki distributions. Figure 9.3 presents a map of the predicted geographic distribution of the virus. MAYV presents six distributional patterns across South America related to terrestrial eco-regions showing the high and low probabilities of predicted areas.
Previous findings suggest that MAYV circulates between canopy-dwelling Haemagogus mosquitoes and nonhuman primates [45, 72, 89], but very little is known about the enzootic transmission cycles that perpetuate MAYV.
The MAYV genome is highly conserved (i.e., 96.4%–100% nucleotide sequence identities and 97.7%–100% amino acid sequence identities), across the complete genome, among genotype D strains [10]. Given this level of sequence conservation, it is unlikely that there may be significant phenotypic variation among strains, but recent work has shown the presence of five positively selected sites across the genome and nonsynonymous mutations that delineate various genotypes as well as the Venezuelan 2010 outbreak strains [10]. Reverse genetic studies are necessary to determine if any of these substitutions can cause phenotypic alterations. These studies can also be used to compare virulence among historical and contemporaneous isolates.
3.2 Chikungunya Virus
Chikungunya virus (CHIKV) is an alphavirus in the family Togaviridae and a relative of other neotropical viruses such as Venezuelan, western, and eastern equine encephalitis (although most strains in Latin America are part of the species Madariaga virus) viruses, as well as Mayaro (MAYV) and Una viruses, its closest relatives in the New World [132].
As does MAYV, CHIKV causes an acute febrile disease typically accompanied by severe arthralgia that can persist for years [130]. However, differing from MAYV wherein human infections are thought to result mainly from direct spillover of enzootic strains , CHIKV causes disease via direct spillover as well as by entering a human–mosquito–human cycle in urban areas, typically involving transmission by the anthropophilic mosquito Aedes ae. and recently also by Ae. albopictus; this leads to major epidemics involving millions of persons with efficient spread via infected air travelers during recent outbreaks. Although CHIKV is rarely fatal, newborns infected during birth, as well as the elderly, especially those with complicating, underlying medical conditions, can have severe neurological disease,. However, in addition to CHIKV being a direct cause of extensive morbidity in all age groups because of its typically high attack rates, the debilitating and often chronic arthralgia results in extensive economic impacts when infected persons cannot work or care for their families [102].
Chikungunya virus is believed to have originated in sub-Saharan Africa in enzootic cycles involving nonhuman primates and sylvatic Aedes spp. mosquito vectors, and these cycles continue in many regions of that continent. The history of CHIKV in Latin America probably began centuries ago when sailing ships carried it from Africa to port cities around the world, including the Caribbean and Latin America [127]. In fact, the term “dengue” may have originally described CHIKV infections, with the terminology becoming confused over the centuries with what is now known as dengue fever [42]. During modern scientific history since CHIKV was first isolated and associated with febrile illness in 1952 [65, 95], CHIKV is believed to have emerged from the enzootic African cycle to initiate urban transmission on several occasions, beginning about a century ago when the Asian lineage was introduced into South and Southeast Asia and caused outbreaks first recognized in 1958 (Fig. 9.4) [127]. This Asian lineage , transmitted primarily by Ae. aegypti, has continued to cause sporadic outbreaks in Asia and Oceania ever since. The next major emergence of a strain into a stable urban cycle began in 2004 when an outbreak began in coastal Kenya [20] and spread into the Indian Ocean basin as well as into Asia to infect millions of persons. Following the importation of this Indian Ocean lineage (IOL) strain by tens of thousands of infected travelers, outbreaks were also detected in Italy [92] and France [36]. However, despite importations into permissive (naïve human populations and abundant Ae. aegypti) dengue-endemic regions of the Americas , no local transmission was detected in the Western Hemisphere until late 2013, when an Asian lineage strain was implicated in human infections on the island of St. Martin in the Caribbean. Subsequently, this strain spread to nearly all Caribbean islands and throughout tropical and subtropical regions of Latin America during 2014, with continued circulation in many regions as of 2016. Then, in 2014, another CHIKV strain was introduced into northeastern Brazil directly from Africa (a member of the East/Central/South African, or ECSA, lineage). The distributions of the two CHIKV strains (Asian and ECSA lineages) are not completely known, but the Asian strain has been detected by sequencing in the Caribbean, Central America, Mexico, Florida in the United States (briefly following introductions in 2014), and northern South America, whereas the ECSA strain has not been reported outside Brazil. Determination of the geographic ranges of the two strains could be important because many ECSA strains have the ability to adapt for more efficient transmission by Ae. albopictus via mutations in the E1 and E2 envelope glycoprotein genes [114], although Asian lineages are epistatically constrained from such adaptation [115]. However, the ECSA strain circulating in Brazil and possibly beyond may have a different epistatic constraint based on a different E2 residue (position 211) [115]. Reverse genetic studies are needed to more definitively assess this adaptive potential of the Brazilian ECSA strain because the ability to use Ae. albopictus as an efficient vector could allow CHIKV to extend its geographic range into rural and temperate regions of Latin America.
Evolution of CHIKV. a Maximum-likelihood tree of major CHIKV lineages based on concatenated open reading frames (ORFs). Branch lengths reflect genetic distance. Bootstrap values are labeled for major lineages and clades. b Maximum clade credibility (MCC) tree of the Asian CHIKV lineage based on the Skyride population model using all Asian lineage strains. Branch length is scaled to the sampling and divergence time, and the branches are color coded for sample location. Node bars representing 95% highest probability density (HPD) value of the node height are shown only for those with a posterior probability of 90 or higher. (Modified from Chen et al. [19])
Control of CHIKV in Latin America represents the same challenges imposed by dengue and now Zika viruses. Until a vaccine can be licensed (and several promising candidates are in late preclinical or early clinical stages of testing), vector control represents the only means of preventing infection and limiting spread. Although CHIKV has already spread extensively to many regions of Latin America and the Caribbean, and high seroprevalence [52, 103] as well as a drop in reported cases since 2014 (PAHO data) suggest that the epidemic has peaked in many regions, CHIKV infections continue to occur and outbreaks have not been reported in some areas with a history of dengue , suggesting continued spread. Unfortunately, past failures with the control of Ae. aegypti because of the wide range of challenges posed by this species do not bode well for this approach to CHIKV control [29]. The presence of MAYV in many parts of South America could also influence further CHIKV spread and possibly disease manifestations. These two alphaviruses exhibit some antigenic cross-reactivity [16] that could reduce viremia to affect pathogenesis and possibly vector transmission, which should be evaluated in the near future.
4 Challenges of Future Research
In VEEV, there are four major challenges that we believe can be solved using new approaches: (1) rapidly estimating the origin of a newly discovered VEEV strain ; (2) estimating its equine and/or human amplification and thus epidemic potential; (3) predicting the human virulence phenotype of a newly discovered VEEV strain. Phylogenetic relationships of a diverse collection of VEEV strains have proved useful for identification of the genetic features leading to epidemic spread to humans and livestock of this zoonotic pathogen. Also, (4) search for synapomorphic genetic, physiological, or ecological factors shared by Spissipes mosquitoes could explain their important role in transmitting arboviruses [34, 126].
Meanwhile, in EEEV several advances in the understanding of MADV pathogenesis have been achieved in the recent year. However, the available evidence is limited to in vitro studies. Genetic determinants of virulence are still unclear: animal models have failed to reproduce the natural history of disease, although cotton rats seem to be a promising model for evaluation of this question [7]. Basic epidemiological investigations are needed to understand the potential of MADV emergence in other Latin American countries; evaluation of cross-protective immunity is also important for vaccine design.
For MAYV and CHIKV, the degree and longevity of such cross-protection between both viruses should be further assessed not only to assist with predicting interactions that might limit circulation and spread but also to determine if an effective CHIKV vaccine could also limit disease caused by MAYV, which is probably indistinguishable from CHIKV infection and appears to be grossly underreported in Latin America [119].
Further work is warranted to truly understand the ecology of MAYV as a potentially emergent alphavirus. Of particular interest are (i) which vectors maintain enzootic transmission, (ii) what species are competent bridge vectors that facilitate transmission to humans, (iii) which nonhuman primate species serve as the primary amplification host, and last (iv) do other canopy-dwelling vertebrates, rodents, or birds have a role in transmission, or act as dead-end hosts only. These questions can be addressed through experimental infections in the laboratory or via field studies aimed at virus isolation and serological detection of MAYV among vectors and vertebrates in known endemic areas.
References
Aguilar PV, Adams AP, Suárez V, Beingolea L, Vargas J, Manock S, Freire J, Espinoza WR, Felices V, Diaz A, Liang X, Roca Y, Weaver SC, Kochel TJ (2009) Genetic characterization of Venezuelan equine encephalitis virus from Bolivia, Ecuador and Peru: identification of a new subtype ID lineage. PLoS Negl Trop Dis 3:e514. doi:10.1371/journal.pntd.0000514
Aguilar PV, Paessler S, Carrara A-S, Baron S, Poast J, Wang E, Moncayo AC, Anishchenko M, Watts D, Tesh RB, Weaver SC (2005) Variation in interferon sensitivity and induction among strains of eastern equine encephalitis virus. J Virol 79:11300–11310
Aguilar PV, Robich RM, Turell MJ, O’Guinn ML, Klein TA, Huaman A, Guevara C, Rios Z, Tesh RB, Watts DM, Olson J, Weaver SC (2007) Endemic eastern equine encephalitis in the Amazon region of Peru. Am J Trop Med Hyg 76:293–298
Aitken TH, Downs WG, Anderson CR, Sence L, Casals J (1960) Mayaro virus isolated from a Trinidadian mosquito, Mansonia Venezuelensis. Science 131:986
Alfonzo D, Grillet ME, Liria J, Navarro J-C, Weaver SC, Barrera R (2005) Ecological characterization of the aquatic habitats of mosquitoes (Diptera: Culicidae) in enzootic foci of Venezuelan equine encephalitis virus in western Venezuela. J Med Entomol 42:278–284. doi:10.1603/0022-2585(2005)042[0278:ECOTAH]2.0.CO;2
Anderson CR, Downs WG, Wattley GH, Ahin NW, Reese AA (1957) Mayaro virus: a new human disease agent. II. Isolation from blood of patients in Trinidad, B.W.I. Am J Trop Med Hyg 6:1012–1016
Arrigo NC, Adams AP, Watts DM, Newman PC, Weaver SC, Paige Adams A, Watts DM, Newman PC, Weaver SC (2010) Cotton rats and house sparrows as hosts for north and south american strains of eastern equine encephalitis virus. Emerg Infect Dis 16:1373–1380. doi:10.3201/eid1609.100459
Arrigo NC, Adams AP, Weaver SC (2010) Evolutionary patterns of eastern equine encephalitis virus in north versus South America suggest ecological differences and taxonomic revision. J Virol 84:1014–1025. doi:10.1128/JVI.01586-09
Auguste AJ, Adams AP, Arrigo NC, Martinez R, Travassos da Rosa APA, Adesiyun AA, Chadee DD, Tesh RB, Carrington CVF, Weaver SC (2010) Isolation and characterization of sylvatic mosquito-borne viruses in Trinidad: enzootic transmission and a new potential vector of Mucambo virus. Am J Trop Med Hyg 83:1262–1265. doi:10.4269/ajtmh.2010.10-0280
Auguste AJ, Liria J, Forrester NL, Giambalvo D, Moncada M, Long KC, Morón D, de Manzione N, Tesh RB, Halsey ES, Kochel TJ, Hernandez R, Navarro J, Weaver SC, de Manzione N, Tesh RB, Halsey ES, Kochel TJ, Hernandez R, Navarro J, Weaver SC (2015) Evolutionary and ecological characterization of Mayaro virus strains isolated during an outbreak, Venezuela, 2010. Emerg Infect Dis J 21:1742–1750. doi:10.3201/eid2110.141660
Azevedo RSS, Silva EVP, Carvalho VL, Rodrigues SG, Nunes Neto JP, Monteiro HAO, Peixoto VS, Chiang JO, Nunes MRT, Vasconcelos PFC, Nunes-Neto JP, Monteiro HAO, Peixoto VS, Chiang JO, Nunes MRT, Vasconcelos PFC (2009) Mayaro fever virus, Brazilian amazon. Emerg Infect Dis 15:1830–1832. doi:10.3201/eid1511.090461
Bargielowski IE, Lounibos LP, Shin D, Smartt CT, Carrasquilla MC, Henry A, Navarro JC, Paupy C, Dennett JA (2015) Widespread evidence for interspecific mating between Aedes aegypti and Aedes albopictus (Diptera: Culicidae) in nature. Infect Genet Evol 36:6–11. doi:10.1016/j.meegid.2015.08.016
Barrera R, Ferro C, Navarro J-C, Freier J, Liria J, Salas R, Ahumada M, Vasquez C, Gonzalez M, Kang W, Boshell J, Weaver SC (2002) Contrasting sylvatic foci of Venezuelan equine encephalitis virus in northern South America. Am J Trop Med Hyg 67:324–334
Barrera R, Torres N, Freier JE, Navarro JC, García CZ, Salas R, Vasquez C, Weaver SC (2001) Characterization of enzootic foci of Venezuelan equine encephalitis virus in western Venezuela. Vector Borne Zoonotic Dis 1:219–230. doi:10.1089/153036601753552585
Bergren NA, Auguste AJ, Forrester NL, Negi SS, Braun WA, Weaver SC (2014) Western equine encephalitis virus: evolutionary analysis of a declining alphavirus based on complete genome sequences. J Virol 88:9260–9267. doi:10.1128/JVI.01463-14
Calisher C, Karabatsos N (1988) Arbovirus serogroups: definition and geographic distribution. In: Montar T (ed) The arboviruses: epidemiology and ecology, vol I. CRC Press, Boca Raton, pp 19–57
Calisher CH, Lazuick JS, Justines G, Francy DB, Monath TP, Gutierrez E, Haven N (1981) Viruses isolated from Aedomyia squamipennis collected in Panama, Ecuador and Argentina: establishment of the Gamboa Serogroup. Am J Trop Med Hyg 30:219–223
Carrera J-P, Forrester N, Wang E, Vittor AY, Haddow AD, López-Vergès S, Abadía I, Castaño E, Sosa N, Báez C, Estripeaut D, Díaz Y, Beltrán D, Cisneros J, Cedeño HG, Travassos da Rosa AP, Hernandez H, Martínez-Torres AO, Tesh RB, Weaver SC (2013) Eastern equine encephalitis in Latin America. N Engl J Med 369:732–744. doi:10.1056/NEJMoa1212628
Chen R, Puri V, Fedorova N, Lin D, Hari KL, Jain R, Rodas JD, Das SR, Shabman RS, Weaver SC (2016) Comprehensive genome scale phylogenetic study provides new insights on the global expansion of chikungunya virus. J Virol 90:10600–10611. doi:10.1128/JVI.01166-16
Chretien J-P, Anyamba A, Bedno SA, Breiman RF, Sang R, Sergon K, Powers AM, Onyango CO, Small J, Tucker CJ, Linthicum KJ (2007) Drought-associated chikungunya emergence along coastal East Africa. Am J Trop Med Hyg 76:405–407
Corniou B, Ardoin P, Bartholomew C, Ince W, Massiah V (1972) First isolation of a South American strain of eastern equine virus from a case of encephalitis in Trinidad. Trop Geogr Med 24:162–167
Craighead JE, Shelokov A, Peralta PH (1962) The lizard: a possible host for eastern equine encephalitis virus in Panama. Am J Hyg 76:82–87
Deardorff ER, Estrada-Franco JG, Freier JE, Navarro-Lopez R, Travassos Da Rosa A, Tesh RB, Weaver SC, Da Rosa AT, Tesh RB, Weaver SC (2011) Candidate vectors and rodent hosts of Venezuelan equine encephalitis virus, Chiapas, 2006–2007. Am J Trop Med Hyg 85:1146–1153. doi:10.4269/ajtmh.2011.11-0094
Deardorff ER, Forrester NL, Travassos-da-Rosa AP, Estrada-Franco JG, Navarro-Lopez R, Tesh RB, Weaver SC (2009) Experimental infection of potential reservoir hosts with Venezuelan equine encephalitis virus, Mexico. Emerg Infect Dis 15:519–525. doi:10.3201/eid1504.081008
Deardorff ER, Weaver SC (2010) Vector competence of Culex (Melanoconion) taeniopus for equine-virulent subtype IE strains of Venezuelan equine encephalitis virus. Am J Trop Med Hyg 82:1047–1052. doi:10.4269/ajtmh.2010.09-0556
Díaz-González EE, Kautz TF, Dorantes-Delgado A, Malo-García IR, Laguna-Aguilar M, Langsjoen RM, Chen R, Auguste DI, Sánchez-Casas RM, Danis-Lozano R, Weaver SC, Fernández-Salas I (2015) First report of Aedes aegypti transmission of chikungunya virus in the Americas. Am J Trop Med Hyg 93:1325–1329. doi:10.4269/ajtmh.15-0450
Dietz WH, Galindo P, Johnson KM (1980) Eastern equine encephalomyelitis in Panama: the epidemiology of the 1973 epizootic. Am J Trop Med Hyg 29:133–140
Erasmus JH, Needham J, Raychaudhuri S, Diamond MS, Beasley DWC, Morkowski S, Salje H, Fernandez Salas I, Kim DY, Frolov I, Nasar F, Weaver SC (2015) Utilization of an Eilat virus-based chimera for serological detection of chikungunya infection. PLoS Negl Trop Dis 9:e0004119. doi:10.1371/journal.pntd.0004119
Fernández-Salas I, Danis-Lozano R, Casas-Martínez M, Ulloa A, Bond JG, Marina CF, Lopez-Ordóñez T, Elizondo-Quiroga A, Torres-Monzón JA, Díaz-González EE (2015) Historical inability to control Aedes aegypti as a main contributor of fast dispersal of chikungunya outbreaks in Latin America. Antivir Res 124:30–42. doi:10.1016/j.antiviral.2015.10.015
Fernandez Z, Moncayo AC, Carrara AS, Forattini OP, Weaver ASC (2003) Vector competence of rural and urban strains of Aedes (Stegomyia) albopictus (Diptera: Culicidae) from São Paulo state, Brazil for IC, ID, and IF subtypes of Venezuelan equine encephalitis virus. J Med Entomol 40:522–527
Ferro C, Boshell J, Moncayo AC, Gonzalez M, Ahumada ML, Kang W, Weaver SC (2003) Natural enzootic vectors of Venezuelan equine encephalitis virus, Magdalena Vally, Colombia. Emerg Infect Dis 9:49–54. doi:10.3201/eid0901.020136
Alice FJ (1956) Infeccao humana pelo virus “leste” da encefalite equina. Bol Inst Biol da Bahia 3:3–9
Forshey BM, Guevara C, Laguna-Torres VA, Cespedes M, Vargas J, Gianella A, Vallejo E, Madrid C, Aguayo N, Gotuzzo E, Suarez V, Morales AM, Beingolea L, Reyes N, Perez J, Negrete M, Rocha C, Morrison AC, Russell KL, Blair PJ, Olson JG, Kochel TJ, Blair PJ, Olson JG, Kochel TJ (2010) Arboviral etiologies of acute febrile illnesses in western South America, 2000–2007. PLoS Negl Trop Dis 4:2000–2007. doi:10.1371/journal.pntd.0000787
Gardner SN, McLoughlin K, Be NA, Allen J, Weaver SC, Forrester N, Guerbois M, Jaing C (2016) Characterization of genetic variability of Venezuelan equine encephalitis viruses. PLoS One 11:e0152604. doi:10.1371/journal.pone.0152604
Gonzalez-Salazar D, Estrada-Franco JG, Carrara AS, Aronson JF, Weaver SC (2003) Equine amplification and virulence of subtype IE Venezuelan equine encephalitis viruses isolated during the 1993 and 1996 Mexican epizootics. Emerg Infect Dis 9:161–168. doi:10.3201/eid0902.020124
Grandadam M, Caro V, Plumet S, Thiberge JM, Souarès Y, Failloux AB, Tolou HJ, Budelot M, Cosserat D, Leparc-Goffart I, Desprès P (2011) Chikungunya virus, southeastern France. Emerg Infect Dis 17:910–913. doi:10.3201/eid1705.101873
Grayson MA, Galindo P (1972) Experimental transmission of Venezuelan equine encephalitis virus by Deinocerites pseudes Dyar and Knab, 1909. J Med Entomol 9:196–200
Greene IP, Paessler S, Austgen L, Anishchenko M, Brault AC, Bowen RA, Weaver SC (2005) Envelope glycoprotein mutations mediate equine amplification and virulence of epizootic venezuelan equine encephalitis virus. J Virol 79:9128–9133. doi:10.1128/JVI.79.14.9128-9133.2005
Groot H, Morales A, Vidales H (1961) Virus isolations from forest mosquitoes in San Vicente de Chucuri, Colombia. Am J Trop Med Hyg 10:397–402
Hahn CS, Lustig S, Strauss EG, Strauss JH (1988) Western equine encephalitis virus is a recombinant virus. Proc Natl Acad Sci U S A 85:5997–6001
Halsey ES, Siles C, Guevara C, Vilcarromero S, Jhonston EJ, Ramal C, Aguilar PV, Ampuero JS (2013) Mayaro virus infection, Amazon Basin region, Peru, 2010-2013. Emerg Infect Dis 19:1839–1842. doi:10.3201/eid1911.130777
Halstead SB (2015) Reappearance of chikungunya, formerly called dengue, in the Americas. Emerg Infect Dis 21:557–561. doi:10.3201/eid2104.141723
Harrington LC, Edman JD, Scott TW (2001) Why do female Aedes aegypti (Diptera: Culicidae) feed preferentially and frequently on human blood? J Med Entomol 38:411–422. doi:10.1603/0022-2585-38.3.411
Hawley RJ, Eitzen EM (2001) Biological weapons—a primer for microbiologists. Annu Rev Microbiol 55:235–253. doi:10.1146/annurev.micro.55.1.235
Hoch AL, Peterson NE, LeDuc JW, Pinheiro FP (1981) An outbreak of Mayaro virus disease in Belterra, Brazil. III. Entomological and ecological studies. Am J Trop Med Hyg 30:689–698
Izurieta RO, Macaluso M, Watts DM, Tesh RB, Guerra B, Cruz LM, Galwankar S, Vermund SH (2011) Hunting in the rainforest and Mayaro virus infection: an emerging alphavirus in Ecuador. J Glob Infect Dis 3:317–323. doi:10.4103/0974-777X.91049
Johansson MA, Powers AM, Pesik N, Cohen NJ, Erin Staples J (2014) Nowcasting the spread of chikungunya virus in the Americas. PLoS One 9:e104915. doi:10.1371/journal.pone.0104915
Johnson BW, Kosoy O, Wang E, Delorey M, Russell B, Bowen RA, Weaver SC (2011) Use of sindbis/eastern equine encephalitis chimeric viruses in plaque reduction neutralization tests for arboviral disease diagnostics. Clin Vaccine Immunol 18:1486–1491. doi:10.1128/CVI.05129-11
Johnson KM, Martin DH (1974) Venezuelan equine encephalitis. Adv Vet Sci Comp Med 18:79–116
Karbaat J (1965) Arthropod-borne virus infections in Dutch soldiers stationed in Surinam. Trop Geogr Med 17:187–189
Kramer LD, Scherer WF (1976) Vector competence of mosquitoes as a marker to distinguish central American and Mexican epizootic from enzootic strains of Venezuelan encephalitis virus. Am J Trop Med Hyg 25:336–346
Kuan G, Ramirez S, Gresh L, Ojeda S, Melendez M, Sanchez N, Collado D, Garcia N, Mercado JC, Gordon A, Balmaseda A, Harris E (2016) Seroprevalence of anti-chikungunya virus antibodies in children and adults in Managua, Nicaragua, after the first chikungunya epidemic, 2014-2015. PLoS Negl Trop Dis 10:e0004773. doi:10.1371/journal.pntd.0004773
LeDuc JW, Pinheiro FP, Travassos da Rosa AP (1981) An outbreak of Mayaro virus disease in Belterra, Brazil. II. Epidemiology. Am J Trop Med Hyg 30:682–688
Liebman KA, Stoddard ST, Reiner RC, Perkins TA, Astete H, Sihuincha M, Halsey ES, Kochel TJ, Morrison AC, Scott TW (2014) Determinants of heterogeneous blood feeding patterns by Aedes aegypti in Iquitos, Peru. PLoS Negl Trop Dis 8:e2702. doi:10.1371/journal.pntd.0002702
Linthicum KJ, Gordon SW, Monath TP (1992) Comparative infections of epizootic and enzootic strains of Venezuelan equine encephalomyelitis virus in Amblyomma cajennense (Acari: Ixodidae). J Med Entomol 29:827–831
Linthicum KJ, Logan TM (1994) Laboratory transmission of Venezuelan equine encephalomyelitis virus by the tick Hyalomma truncatum. Trans R Soc Trop Med Hyg 88:126. doi:10.1016/0035-9203(94)90536-3
Linthicum KJ, Logan TM, Bailey CL, Gordon SW, Peters CJ, Monath TP, Osorio J, Francy DB, McLean RG, Leduc JW et al (1991) Venezuelan equine encephalomyelitis virus infection in and transmission by the tick Amblyomma cajennense (Arachnida: Ixodidae). J Med Entomol 28:405–409
Liria J, Navarro JC (2010) Modelo de nicho ecológico en Haemagogus Williston (Diptera: Culicidae), vectores del virus de la fiebre amarilla. Rev Biomédica 21:149–161
Llagonne-Barets M, Icard V, Leparc-Goffart I, Prat C, Perpoint T, André P, Ramière C (2016) A case of Mayaro virus infection imported from French Guiana. J Clin Virol 77:66–68. doi:10.1016/j.jcv.2016.02.013
Long KC, Ziegler SA, Thangamani S, Hausser NL, Kochel TJ, Higgs S, Tesh RB (2011) Experimental transmission of Mayaro virus by Aedes aegypti. Am J Trop Med Hyg 85:750–757. doi:10.4269/ajtmh.2011.11-0359
Luciany K, Abadía I, Cisneros J, Guerra I, Garcıa M, Martínez-Torres AO, Estripeaut D, Carrera J-P (2015) Case report : madariaga virus infection associated with a case of acute disseminated encephalomyelitis. Am J Trop Med Hyg 92:1130–1132. doi:10.4269/ajtmh.14-0845
Luciany K, Abadía II, Cisneros J, Guerra I, Garcıa M, Martínez-Torres AO, Estripeaut D, Carrera J-P, Garcı M, Luciany K, Abadía II, Cisneros J, Guerra I, Garcıa M, Martínez-Torres AO, Estripeaut D, Carrera J-P, Luciani K, Abadía II, Martínez-Torres AO, Cisneros J, Guerra I, García M, Estripeaut D, Carrera J-P (2015) Case report : madariaga virus infection associated with a case of acute disseminated encephalomyelitis. Am J Trop Med Hyg 92:1130–1132. doi:10.4269/ajtmh.14-0845
Grayson MA, Galindo P, Grayson MA, Galindo P (1969) Ecology of Venezuelan equine encephalitis virus in Panama. Am Vet Med Assoc 155:2141–2145
do Carmo Alves Martins V, de Souza Bastos M, Ramasawmy R, de Figueiredo RP, JBL G, WSM B, Nogueira ML, Nozawa S, Naveca FG, LTM F, MPG M (2014) Clinical and virological descriptive study in the 2011 outbreak of dengue in the Amazonas, Brazil. PLoS One 9:e100535. doi:10.1371/journal.pone.0100535
Mason PJ, Haddow AJ (1957) An epidemic of virus disease in Southern Province, Tanganyika territory, in 1952–1953. Trans R Soc Trop Med Hyg 51:238–240. doi:10.1016/0035-9203(57)90022-6
Medina G, Garzaro DJ, Barrios M, Auguste AJ, Weaver SC, Pujol FH (2015) Genetic diversity of Venezuelan alphaviruses and circulation of a Venezuelan equine encephalitis virus subtype IAB strain during an interepizootic period. Am J Trop Med Hyg 93:7–10. doi:10.4269/ajtmh.14-0543
Mendez W, Liria J, Navarro JC, Garcia CZ, Freier JE, Salas R, Weaver SC, Barrera R (2001) Spatial dispersion of adult mosquitoes (Diptera: Culicidae) in a sylvatic focus of Venezuelan equine encephalitis virus. J Med Entomol 38:813–821
Miller MJ, Loaiza JR (2015) Geographic expansion of the invasive mosquito Aedes albopictus across Panama—implications for control of dengue and chikungunya viruses. PLoS Negl Trop Dis 9:e0003383. doi:10.1371/journal.pntd.0003383
Mitchell CJ, Monath TP, Sabattini MS, Cropp CB, Daffner JF, Calisher CH, Jakob WL, Christensen HA (1985) Arbovirus investigations in Argentina, 1977–1980. II. Arthropod collections and virus isolations from argentine mosquitoes. Am J Trop Med Hyg 34:945–955
Monath TP, Sabattini MS, Mitchell CJ, Bowen GS (1985) Arbovirus investigations in Argentina, 1977–1980. Surveys 34:966–975
Monath TP, Sabattini MS, Pauli R, Daffner JF, Mitchell CJ, Bowen GS, Cropp CB (1985) Arbovirus investigations in Argentina, 1977-1980. IV. Serologic surveys and sentinel equine program. Am J Trop Med Hyg 34:966–975
Muñoz M, Navarro JC (2012) Mayaro: a re-emerging arbovirus in Venezuela and Latin America. Biomedica (Bogotá) 32:286–302. doi:10.1590/S0120-41572012000300017
Muñoz M, Navarro JC (2012) Virus Mayaro : un arbovirus reemergente en Venezuela y Latinoamérica. Biomédica (Bogotá) 32:286–302
Nasar F, Gorchakov RV, Tesh RB, Weaver SC (2015) Eilat virus host range restriction is present at multiple levels of the virus life cycle. J Virol 89:1404–1418. doi:10.1128/JVI.01856-14
Nasar F, Palacios G, Gorchakov RV, Guzman H, Da Rosa APT, Savji N, Popov VL, Sherman MB, Lipkin WI, Tesh RB, Weaver SC (2012) Eilat virus, a unique alphavirus with host range restricted to insects by RNA replication. Proc Natl Acad Sci U S A 109:14622–14627. doi:10.1073/pnas.1204787109
Navarro J-C, Medina G, Vasquez C, Coffey LL, Wang E, Suárez A, Biord H, Salas M, Weaver SC (2005) Postepizootic persistence of Venezuelan equine encephalitis virus, Venezuela. Emerg Infect Dis 11:1907–1915. doi:10.3201/eid1112.050533
Navarro J-C, Quintero L, Zorrilla A, González R (2013) Molecular tracing with mitochondrial ND5 of the invasive mosquito Aedes (Stegomyia) albopictus (Skuse) in Northern South America. J Entomol Zool Stud 1(4):32–39
Navarro J-C, Weaver SC (2004) Molecular phylogeny of the Vomerifer and pedroi groups in the Spissipes section of the subgenus Culex (Melanoconion). J Med Entomol 41:575–581
Navarro J, Medina G, Vasquez C, Coffey LL, Wang E, Suárez A, Biord H, Salas M, Weaver SC (2005) Postepizootic persistence of Venezuelan equine encephalitis. Emerg Infect Dis 11:1907–1915
Ortiz DI, Kang W, Weaver SC (2008) Susceptibility of Ae. aegypti (Diptera: Culicidae) to infection with epidemic (subtype IC) and enzootic (subtypes ID, IIIC, IIID) Venezuelan equine encephalitis complex alphaviruses. J Med Entomol 45:1117–1125. doi:10.1603/0022-2585(2008)45[1117:SOAADC]2.0.CO;2
Pauvolid-Correa A, Tavares FN, Alencar J, Silva Jdos S, Murta M, Serra-Freire NM, Pellegrin AO, Gil-Santana H, Guimaraes AE, Silva E (2010) Preliminary investigation of Culicidae species in south Pantanal, Brazil and their potential importance in arbovirus transmission. Rev Inst Med Trop Sao Paulo 52:17–24
Pauvolid-Corrêa A, Juliano RS, Campos Z, Velez J, Nogueira RMR, Komar N (2015) Neutralising antibodies for Mayaro virus in Pantanal, Brazil. Mem Inst Oswaldo Cruz 110:125–133. doi:10.1590/0074-02760140383
Pecor JE, Jones J, Turell MJ, Fernandez R, Carbajal F, O’Guinn M, Sardalis M, Watts D, Zyzak M, Calampa C, Klein TA (2000) Annotated checklist of the mosquito species encountered during arboviral studies in Iquitos, Peru (Diptera: Culicidae). J Am Mosq Control Assoc 16:210–218
Petersen LR, Powers AM (2016) Chikungunya: epidemiology. F1000Research 5:1–8. doi:10.12688/f1000research.7171.1
Pfeffer M, Dobler G (2010) Emergence of zoonotic arboviruses by animal trade and migration. Parasit Vectors 3:35. doi:10.1186/1756-3305-3-35
Pinheiro FP, Freitas RB, Travassos da Rosa JF, Gabbay YB, Mello WA, LeDuc JW (1981) An outbreak of Mayaro virus disease in Belterra, Brazil. I. Clinical and virological findings. Am J Trop Med Hyg 30:674–681
Pisano MB, Oria G, Beskow G, Aguilar J, Konigheim B, Cacace ML, Aguirre L, Stein M, Contigiani MS (2013) Venezuelan equine encephalitis viruses (VEEV) in Argentina: serological evidence of human infection. PLoS Negl Trop Dis 7:e2551. doi:10.1371/journal.pntd.0002551
Pisano MB, Ré VE, Díaz LA, Farías A, Stein M, Sanchez-Seco MP, Tenorio A, Almirón WR, Contigiani MS (2010) Enzootic activity of pixuna and Rio Negro viruses (Venezuelan equine encephalitis complex) in a neotropical region of Argentina. Vector Borne Zoonotic Dis 10:199–201. doi:10.1089/vbz.2008.0156
Powers AM, Aguilar PV, Chandler LJ, Brault AC, Meakins TA, Watts D, Russell KL, Olson J, Vasconcelos PFC, Da Rosa AT, Weaver SC, Tesh RB (2006) Genetic relationships among Mayaro and Una viruses suggest distinct patterns of transmission. Am J Trop Med Hyg 75:461–469. 75/3/461 [pii]
Powers AM, Brault AC, Shirako Y, Strauss EG, Kang W, Strauss JH, Weaver SC (2001) Evolutionary relationships and systematics of the alphaviruses. J Virol 75:10118–10131. doi:10.1128/JVI.75.21.10118-10131.2001
Powers AM, Oberste MS, Brault AC, Rico-Hesse R, Schmura SM, Smith JF, Kang W, Sweeney WP, Weaver SC (1997) Repeated emergence of epidemic/epizootic Venezuelan equine encephalitis from a single genotype of enzootic subtype ID virus. J Virol 71:6697–6705
Rezza G, Nicoletti L, Angelini R, Romi R, Finarelli A, Panning M, Cordioli P, Fortuna C, Boros S, Magurano F, Silvi G, Angelini P, Dottori M, Ciufolini M, Majori G, Cassone A (2007) Infection with chikungunya virus in Italy: an outbreak in a temperate region. Lancet 370:1840–1846. doi:10.1016/S0140-6736(07)61779-6
Rico-Hesse R, Weaver SC, de Siger J, Medina G, Salas RA (1995) Emergence of a new epidemic/epizootic Venezuelan equine encephalitis virus in South America. Proc Natl Acad Sci U S A 92:5278–5281. doi:10.1073/pnas.92.12.5278
Ronca SE, Dineley KT, Paessler S (2016) Neurological sequelae resulting from encephalitic alphavirus infection. Front Microbiol 7:959. doi:10.3389/fmicb.2016.00959
Ross RW (1956) The Newala Epidemic. III. The virus: isolation, pathogenic properties and relationship to the epidemic. J Hyg (Lond) 54:177–191. doi:10.1017/S0022172400044442
Sabattini MS, Daffner JFMT, Al E (1991) Localized eastern equine encephalitis in Santiago del Estero Province, Argentina, without human infection. Med Buenos Aires 51:3–8
Salas RA, Garcia CZ, Liria J, Barrera R, Navarro JC, Medina G, Vasquez C, Fernandez Z, Weaver SC (2001) Ecological studies of enzootic Venezuelan equine encephalitis in north-central Venezuela, 1997-1998. Am J Trop Med Hyg 64:84–92
Sallum MA, Forattini OP (1996) Revision of the Spissipes section of Culex (Melanoconion) (Diptera:Culicidae). J Am Mosq Control Assoc 12:517–600
Sanmartin C, Mackenzie RB, Trapido H, Barreto P, Mullenax CH, Gutierrez E, Lesmes C (1973) Venezuelan equine encephalitis in Colombia, 1967. Bol Oficina Sanit Panam 74:108–137
Santiago FW, Halsey ES, Siles C, Vilcarromero S, Guevara C, Silvas JA, Ramal C, Ampuero JS, Aguilar PV (2015) Long-term arthralgia after Mayaro virus infection correlates with sustained pro-inflammatory cytokine response. PLoS Negl Trop Dis 9:e0004104. doi:10.1371/journal.pntd.0004104
Scherer WF, Weaver SC, Taylor CA, Cupp EW, Dickerman RW, Rubino HH (1987) Vector competence of Culex (Melanoconion) taeniopus for allopatric and epizootic Venezuelan equine encephalomyelitis viruses. Am J Trop Med Hyg 36:194–197
Schilte C, Staikovsky F, Couderc T, Madec Y, Carpentier F, Kassab S, Albert ML, Lecuit M, Michault A (2013) Chikungunya virus-associated long-term arthralgia: a 36-month prospective longitudinal study. PLoS Negl Trop Dis. doi:10.1371/journal.pntd.0002137
Simmons G, Brès V, Lu K, Liss NM, Brambilla DJ, Ryff KR, Bruhn R, Velez E, Ocampo D, Linnen J, Latoni G, Petersen LR, Williamson PC, Busch MP (2016) High incidence of chikungunya virus and frequency of viremic blood donations during epidemic, Puerto Rico, USA, 2014. Emerg Infect Dis 22:1221–1228. doi:10.3201/eid2207.160116
Sirivanakarn S (1982) A review of the systematics and a proposed scheme of internal classification of the new world subgenus Melanoconion of Culex (Diptera, Culicidae). Mosq Syst 14:265–333
Smith DW, Mackenzie JS, Weaver SC (2009) Alphaviruses. In: Richman DD, Whitley RJ, Hayden FG (eds) Clinical virology. ASM Press, Washington, DC
Strauss JH, Strauss EG (1994) The alphaviruses: gene expression, replication, and evolution. Microbiol Rev 58:491–562
Suarez O, Bergold G (1968) Investigations of an outbreak of Venezuelan equine encephalitis in towns of eastern Venezuela. Am J Trop Med Hyg 17:875–880
Sunthorn S, Galindo P (1967) The isolation of eastern equine encephalitis from Culex (Melanoconion) taeniopus Dyar and Knab in Panama. Mosq News 27:74–76
Talarmin A, Chandler LJ, Kazanji M, de Thoisy B, Debon P, Lelarge J, Labeau B, Bourreau E, Vié JC, Shope RE, Sarthou JL (1998) Mayaro virus fever in French Guiana: isolation, identification, and seroprevalence. Am J Trop Med Hyg 59:452–456
Terzian ACB, Auguste AJ, Vedovello D, Ferreira MU, da Silva-Nunes M, Speranca MA, Suzuki RB, Juncansen C, Araujo JP, Weaver SC, Nogueira ML (2015) Isolation and characterization of Mayaro virus from a human in Acre, Brazil. Am J Trop Med Hyg 92:401–404. doi:10.4269/ajtmh.14-0417
de Thoisy B, Gardon J, Salas RA, Morvan J, Kazanji M (2003) Mayaro virus in wild mammals, French Guiana. Emerg Infect Dis 9:1326–1329. doi:10.3201/eid0910.030161
Torres-Gutierrez C, Sallum MAM (2015) Catalog of the subgenus Melanoconion of Culex (Diptera: Culicidae) for South America. Zootaxa 4028:1–50
Torres JR, Russell KL, Vasquez C, Barrera R, Tesh RB, Salas R, Watts DM (2004) Family cluster of Mayaro fever, Venezuela. Emerg Infect Dis 10:1304–1306. doi:10.3201/eid1007.030860
Tsetsarkin KA, Chen R, Yun R, Rossi SL, Plante KS, Guerbois M, Forrester N, Perng GC, Sreekumar E, Leal G, Huang J, Mukhopadhyay S, Weaver SC (2014) Multi-peaked adaptive landscape for chikungunya virus evolution predicts continued fitness optimization in Aedes albopictus mosquitoes. Nat Commun 5:4084. doi:10.1038/ncomms5084
Tsetsarkin KA, Chen R, Leal G, Forrester N, Higgs S, Huang J, Weaver SC (2011) Chikungunya virus emergence is constrained in Asia by lineage-specific adaptive landscapes. Proc Natl Acad Sci U S A 108:7872–7877. doi:10.1073/pnas.1018344108
Turell MJ, Ludwig GV, Beaman JR (1992) Transmission of Venezuelan equine encephalomyelitis virus by Aedes sollicitans and Aedes taeniorhynchus (Diptera: Culicidae). J Med Entomol 29:62–65
Turell MJ, O’Guinn ML, Jones JW, Sardelis MR, Dohm DJ, Watts DM, Fernandez R, Travassos da Rosa A, Guzman H, Tesh R, Rossi CA, Ludwig V, Mangiafico JA, Kondig J, Wasieloski LP, Pecor J, Zyzak M, Schoeler G, Mores CN, Calampa C, Lee JS, Klein TA (2005) Isolation of viruses from mosquitoes (Diptera: Culicidae) collected in the Amazon Basin region of Peru. J Med Entomol 42:891–898. doi:10.1603/0022-2585(2005)042[0891:IOVFMD]2.0.CO;2
Vasconcelos PF, Travassos da Rosa AP, Rodrigues SG, Travassos da Rosa ES, Dégallier N, Travassos da Rosa JF (2001) Inadequate management of natural ecosystem in the Brazilian Amazon region results in the emergence and reemergence of arboviruses. Cad saude publica / Minist da Saude, Fund Oswaldo Cruz, Esc Nac Saude Publica 17(suppl):155–164. doi:10.1590/S0102-311X2001000700025
Vasconcelos PFC, Calisher CH (2016) Emergence of human arboviral diseases in the Americas, 2000-2016. Vector Borne Zoonotic Dis 16:1–7. doi:10.1089/vbz.2016.1952
Vieira C, Silva D, Barreto E, Siqueira C, Colombo T, Ozanic K, Schmidt D, Drumond B, Mondini A, Nogueira ML, Bronzoni R (2015) Detection of Mayaro virus infections during a dengue outbreak in Mato Grosso, Brazil. Acta Trop 147:12–16. doi:10.1016/j.actatropica.2015.03.020
Vittor AY, Armien B, Gonzalez P, Carrera J-P, Dominguez C, Valderrama A, Glass GE, Beltran D, Cisneros J, Wang E, Castillo A, Moreno B, Weaver SC (2016) Epidemiology of emergent Madariaga encephalitis in a region with endemic Venezuelan equine encephalitis: initial host studies and human cross-sectional study in Darien, Panama. PLoS Negl Trop Dis 10:e0004554. doi:10.1371/journal.pntd.0004554
Walder R, Suarez OM, Calisher CH (1984) Arbovirus studies in the Guajira region of Venezuela: activities of eastern equine encephalitis and Venezuelan equine encephalitis viruses during an interepizootic period. Am J Trop Med Hyg 33:699–707
Walder R, Suarez OM, Calisher CH (1984) Arbovirus studies in southwestern Venezuela during 1973-1981. II. Isolations and further studies of Venezuelan and eastern equine encephalitis, Una, Itaqui, and Moju viruses. Am J Trop Med Hyg 33:483–491
Wang E, Barrera R, Boshell J, Ferro C, Freier JE, Navarro JC, Salas R, Vasquez C, Weaver SC (1999) Genetic and phenotypic changes accompanying the emergence of epizootic subtype IC Venezuelan equine encephalitis viruses from an enzootic subtype ID progenitor. J Virol 73:4266–4271
Wang E, Bowen RA, Medina G, Powers AM, Kang W, Chandler LM, Shope RE, Weaver SC (2001) Virulence and viremia characteristics of 1992 epizootic subtype IC Venezuelan equine encephalitis viruses and closely related enzootic subtype ID strains. Am J Trop Med Hyg 65:64–69
Weaver SC, Ferro C, Barrera R, Boshell J, Navarro JC (2004) Venezuelan equine encephalitis. Annu Rev Entomol 49:141–174. doi:10.1146/annurev.ento.49.061802.123422
Weaver SC, Forrester NL (2015) Chikungunya: evolutionary history and recent epidemic spread. Antivir Res 120:32–39. doi:10.1016/j.antiviral.2015.04.016
Weaver SC, Kang W, Shirako Y, Rumenapf T, Strauss EG, Strauss JH (1997) Recombinational history and molecular evolution of western equine encephalomyelitis complex alphaviruses. J Virol 71:613–623
Weaver SC, Klimstra WB, Ryman KD(2009) Togaviruses: general features. In: Mahy BWJ, van Regenmortel MHV (eds) Desk encyclopedia of plant and fungal virology. Elsevier/Academic Press, New York. doi: 10.1016/B978-012374410-4.00627-0
Weaver SC, Lecuit M (2015) Chikungunya virus infections. N Engl J Med 373:93–95. doi:10.1056/NEJMc1505501
Weaver SC, Reisen WK (2010) Present and future arboviral threats. Antivir Res 85:328–345. doi:10.1016/j.antiviral.2009.10.008
Weaver SC, Winegar R, Manger ID, Forrester NL (2012) Alphaviruses: population genetics and determinants of emergence. Antivir Res 94:242–257. doi:10.1016/j.antiviral.2012.04.002
Weise WJ, Hermance ME, Forrester N, Adams AP, Langsjoen R, Gorchakov R, Wang E, Alcorn MDH, Tsetsarkin K, Weaver SC (2014) A novel live-attenuated vaccine candidate for Mayaro fever. PLoS Negl Trop Dis 8:e2969. doi:10.1371/journal.pntd.0002969
Yanoviak SP, Aguilar PV, Lounibos LP, Weaver SC (2005) Transmission of a Venezuelan equine encephalitis complex alphavirus by Culex (Melanoconion) gnomatos (Diptera: Culicidae) in northeastern Peru. J Med Entomol 42:404–408
Zacks MA, Paessler S (2010) Encephalitic alphaviruses. Vet Microbiol 140:281–286. doi:10.1016/j.vetmic.2009.08.023
Zehmer RB, Dean PB, Sudia WD, Calisher CH, Sather GE, Parker RL (1974) Venezuelan equine encephalitis epidemic in Texas, 1971. Health Serv Rep 89:278–282
Zuchi N, da Silva Heinen LB, dos MAM S, Pereira FC, Slhessarenko RD (2014) Molecular detection of Mayaro virus during a dengue outbreak in the state of Mato Grosso, central-West Brazil. Mem Inst Oswaldo Cruz 109:820–823
Erasmus JH, Auguste AJ, Kaelber JT, Luo H, Rossi SL, Fenton K, Leal G, Kim DY, Chiu W, Wang T, Frolov I, Nasar F, Weaver SC (2017) A chikungunya fever vaccine utilizing an insect-specific virus platform. Nat Med 23:192–199. PMCID: PMCPMC5296253
Lednicky J, De Rochars VM, Elbadry M, Loeb J, Telisma T, Chavannes S, Anilis G, Cella E, Ciccozzi M, Okech B, Salemi M, Morris JG Jr (2016) Mayaro virus in child with acute febrile illness, haiti, 2015. Emerg Infect Dis 22:2000–2002
Laboratory studies of a Brazilian strain of Aedes albopictus as a potential vector of Mayaro and Oropouche viruses. J Am Mosq Control Assoc (1991) 7(1):89–93
Acknowledgments
We thank the Editors for the invitation to contribute this chapter. Projects Fonacit MC-2008000911-4 (Venez), P011617-UISEK (ECU).
Competing Interest
The authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer International Publishing AG
About this chapter
Cite this chapter
Navarro, JC., Carrera, JP., Liria, J., Auguste, A.J., Weaver, S.C. (2017). Alphaviruses in Latin America and the Introduction of Chikungunya Virus. In: Ludert, J., Pujol, F., Arbiza, J. (eds) Human Virology in Latin America. Springer, Cham. https://doi.org/10.1007/978-3-319-54567-7_9
Download citation
DOI: https://doi.org/10.1007/978-3-319-54567-7_9
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-54566-0
Online ISBN: 978-3-319-54567-7
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)