Abstract
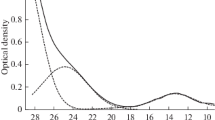
A laboratory study was conducted for the determination of dissolved metals (including aluminum, sodium, and lithium) in cryolitic melts by using a tin extraction-stripping technique. Results demonstrate that the dissolved metal is not only aluminum, but actually a combination of aluminum, sodium, and lithium whose concentrations are dependent on the cryolitic composition and temperature. The results are shown to be rational and self consistent. Differences in total metallic solubility for earlier work in the literature can be attributed to the variations in experimental techniques. Metallic aluminum solubility increased with increasing bath temperature and decreased with increasing cryolite ratio (CR) and LiF concentration. Metallic sodium solubility increased with increasing CR and bath temperature, and decreased with increasing LiF concentration. Metallic lithium solubility increased with increasing CR, LiF concentration, and bath temperature, but was an order of magnitude less than the other metallic species. When molar CR was greater than 2.5, metallic sodium was the dominant component in the total solubility.
Access provided by Autonomous University of Puebla. Download to read the full chapter text
Chapter PDF
Similar content being viewed by others
Keywords
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
References
M. Feinleib and B. Porter, J. Electrochem. Soc., Vol. 103 (4) (1956), 231–236.
M. M. Vetyukov and V. B. Vinokurov, The Soviet J. of Non-Ferrous Metals, (Eng. transi.), Vol. 44 (6) (1971), 35–39.
C. Mitchell and C. S. Samis, Trans, of Metall. Soc. AIME, Vol. 245 (1969), 1227–1234.
K. Yoshida and E. W. Dewing, Metall. Trans., Vol. 3 (1972), 1817–1821.
J. Thonstad and S. Roseth, Light Metals, (1976), 171–182.
H. Kvande, Light Metals, (1980), 171–182.
R. Odegard, A. Sterten, and J. Thonstad, Light Metals, (1987), 389–398; and Metall. Trans. B., Vol. 19B (1988), 449–457.
W. E. Haupin, J. Electrochem. Soc, Vol. 107 (1960), 232–236.
O. P. Ersimenko, M. M. Vetyukov, and A. S. Andrev, J. Appl. Chem. USSR (Eng. transi.), Vol. 40 (1967), p. 1613.
X. Wang and R. D. Peterson, Sn Extraction — Stripping Technique for Metal Solubility in Molten Salts, to be published.
K. Yoshida, T. Ishihara, and M. Yoko, Trans. Metall. Soc. AIME, Vol. 243 (1968), 231–237.
J. Thonstad, Can. J. Chem., Vol. 43 (1965), 3429–3432.
R. Odegard, On the Solubility and Electrochemical Behavior of Aluminum and Aluminum Carbide in Cryolitic melts, Ph.D. thesis, Universitetet I Trondheim, 1986.
K. Grjotheim, et al., eds, Aluminum Electrolysis. 2nd Edition, Aluminum-Verlag, Dusseldorf, 1982.
H. Kvande, Light Metals, (1986), p. 451–459.
M. M. Vetyukov, et al., Soviet Non-Ferrous Met. Research, Vol. 26 (4) (1978), p. 154.
A. M. Arthur, Metall. Trans, 5B (1974), 1225–1230.
X. Wang and R. D. Peterson, Dissolved Metals in Crvolitic Melts, MTL Report 1030, TIC 43–1597 (1990).
R. D. Peterson and X. Wang, Correlation of Metal Solubility with Current Efficiency of Hall Cells, AIME, Light Metal Division, New Orleans, 1991.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 The Minerals, Metals & Materials Society
About this chapter
Cite this chapter
Wang, X., Peterson, R.D., Richards, N.E. (2016). Dissolved Metals in Cryolitic Melts. In: Bearne, G., Dupuis, M., Tarcy, G. (eds) Essential Readings in Light Metals. Springer, Cham. https://doi.org/10.1007/978-3-319-48156-2_7
Download citation
DOI: https://doi.org/10.1007/978-3-319-48156-2_7
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-48155-5
Online ISBN: 978-3-319-48156-2
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)