Abstract
Radiotherapy is a commonly used local and regional treatment for cancer. Although important advances in radiation treatment delivery have been made in recent years, normal tissue damage remains a major cause of toxicity from radiotherapy and chemoradiotherapy regimens. Efforts to reduce normal tissue injury have included technical improvements to minimize normal tissue exposure to high doses of irradiation. Extensive preclinical research and a growing field of clinical research are focusing on the development of agents to protect normal tissues from the deleterious effects of irradiation. In this review, we discuss the characteristics of these agents, the research required to translate these agents into clinical trials, and highlight some challenges and successes in these efforts.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
Background
Radiotherapy is a commonly used treatment modality for cancer, with more than half of all cancer patients receiving radiotherapy during the course of their malignancy [1, 2]. With a few exceptions, radiotherapy is used for the local and regional treatment of cancer. In many cases, radiation is combined with surgery or chemotherapy to improve the likelihood of long-term local and regional control of cancers. Advancements in radiation treatment delivery and medical imaging have revolutionized the field of radiation oncology , providing a greater certainty about the location of tumor in the body and allowing more precise delivery of complex dose distributions. Altered fractionation schemes have allowed improved tumor control [3]. Collectively, these advancements in technology have driven improvements in local control by allowing an escalation of dose to tumor while minimizing the volume of normal tissue exposed to high radiation doses.
Despite these improvements, normal tissue injury remains a common problem in modern treatments. Improving the ability of normal tissues to tolerate radiotherapy may further reduce toxicity of treatment, thus improving efficacy by minimizing treatment breaks and improving adherence to therapy. Further, the ability to spare normal tissues to even a moderate degree may allow further dose escalation to tumor at a similar or reduced rate of toxicity, thus potentially improving disease control.
The development of agents to protect normal tissues from irradiation to enhance the therapeutic index has long been a goal of radiobiologists. Herein, we will describe the mechanisms of injury in normal tissues after irradiation and highlight methods to prevent and treat this damage. We will focus on agents that have successfully been translated into the clinic and agents that are currently in development.
Methods to Improve the Therapeutic Index of Radiotherapy
Normal tissue damage from irradiation can result in both acute and late toxicities. Acute toxicities manifest within days or weeks after treatment, whereas late toxicities manifest months or years after radiation. Acute toxicities are often reversible, but may negatively impact treatment compliance or require treatment interruption. Examples of acute toxicities include dermatitis, mucositis, and cystitis. Late toxicities are often chronic, progressive, and, in many cases, irreversible. Examples of late toxicities are proctitis, fibrosis, myelitis, and brain necrosis. Both acute and late toxicities are radiation dose limiting.
The therapeutic index is a concept that can be applied to any treatment modality, including radiotherapy. For radiotherapy, the therapeutic index is the ratio between the radiation dose that results in tumor control and the dose that results in toxicity. A larger therapeutic index is favorable because it affords the selection of a higher dose of radiation that in turn results in a greater chance of cure with a minimal chance of toxicity. In practice, this ratio is often small, necessitating the clinician to tolerate a moderate chance of substantial toxicity from a radiation treatment.
A number of strategies may be employed to increase this ratio, including increasing the tolerance of normal tissue to a radiation treatment or enhancing the radiation response in a tumor. Strategies to improve normal tissue tolerance to radiotherapy include altered fractionation schedules, improvements in targeting and delivery, and the use of radiation modifiers. Altered fractionation schedules take advantage of differential responses between tumor and normal tissues to allow “escalation” of dose to tumor with similar normal tissue toxicity . Technological advancements allow improved accuracy and precision of radiation treatments, minimizing the amount of normal tissue exposed to high doses of irradiation.
Perhaps the greatest opportunity for improving the therapeutic ratio in radiotherapy in the future is the development of radiation protectors , radiation mitigators , and effective treatments for radiation injury in normal tissues. A basic understanding of these strategies is helpful to understanding their clinical implementation. Each of these strategies is briefly described below.
Radiation results in ionization events , which lead to free radical production. Often, these ionizations occur in water molecules, although direct damage to DNA and other cellular structures may occur (Fig. 4.1). It is thought that DNA double strand breaks are the lethal event that occurs after exposure to ionizing radiation . In cells that survive radiation exposure , signal transduction may be initiated, leading to the activation of multiple pathways important in cell survival and growth. At the tissue level, the loss of cells or the activation of these pathways may result in changes in tissue function or activation of additional processes like inflammation and wound healing. Intervening in these processes may result in modulation of normal tissue damage after irradiation. The three major categories of agents that can minimize normal tissue injury after irradiation fall into three classes: radioprotectors, radiation mitigators, and treatment.
Sequence of events following radiation exposure . The chart is divided into three parts by dashed lines suggesting events and reactions that might be modified by radiation protectors (top), radiation mitigators, and treatment (bottom). Reproduced from Citrin et al. [5]
Radioprotectors
A radioprotector is an agent that prevents the damage caused by radiation, generally, by scavenging the free radicals that cause DNA oxidation and DNA double strand breaks [4]. Because these agents prevent the damage from occurring, they must be given before or at the time of the radiation exposure. Free radicals have an extremely short half-life and, as a result, a limited range for diffusion. Thus, radioprotectors must have the ability to cross the nuclear membrane and accumulate near DNA. This accumulation allows scavenging of radicals that would otherwise lead to lethal DNA damage.
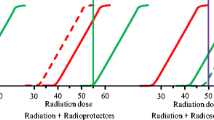
In order for a radioprotector to enhance the therapeutic ratio, the agent must selectively protect normal tissues from irradiation (Fig. 4.2). If the agent protects both normal and tumor tissues, there is no change in the ratio and hence, no benefit to the delivery of the agent [5]. The agent may selectively protect normal tissue through a variety of mechanisms. For example, the radioprotector may be activated or taken up by normal tissue more effectively than tumor tissue, leading to a higher concentration of the agent in normal tissue . Conversely, the agent may be cleared or metabolized more rapidly by tumor tissue, also leading to a higher concentration of the agent in normal tissue.
Effects of radioprotectors on the therapeutic window . (a) The chosen treatment dose (blue vertical line) delivers a high chance of tumor cure (black) with a small chance of normal tissue injury (red). (b) A nonselective radioprotector indiscriminately protects tumor and normal tissue shifting both the tumor cure and normal tissue injury curve to the right. Consequently, the therapeutic window remains unchanged. Shifted curves are shown in red and black. The original curves are shown in gray and pale red. (c) A true selective radioprotector exclusively protects normal tissue and, thus, shifts only the normal tissue injury curve to the right. This affords a larger therapeutic window such that a higher dose can be given to achieve increased tumor cure with equal or less injury. A lower dose producing the same tumor probability can also be given with less tissue injury. (d) Some nonselective radioprotectors can protect the normal tissue to a greater extent shifting the normal tissue curve further to the right than the tumor cure curve and resulting in an increased therapeutic window
Antioxidants are molecules that reduce cellular damage caused by free radicals . Some examples of antioxidants are ascorbic acid, polyphenols, and thiols. These low molecular weight antioxidants produce a more stable reactive species by donating a hydrogen atom to free radicals. Most radioprotectors are also antioxidants [5]. Of note, however, not every antioxidant has radioprotective effects [6, 7], as they are not all reactive toward the secondary species generated by radiation [7]. In addition to small molecule antioxidants, some antioxidants exist in the form of enzymes, such as superoxide dismutase, catalase, and glutathione peroxidase [5].
Amifostine
Amifostine is a thiol compound that scavenges free radicals, and it is the only FDA-approved radioprotector . Clinically, it has been administered to head and neck cancer patients receiving radiotherapy to prevent xerostomia (dry mouth) [8]. Amifostine is a prodrug that is only activated when dephosphorylated by alkaline phosphatase, a cell membrane protein [9]. It selectively protects normal tissue as it preferentially accumulates in normal tissue rather than tumor tissue [10]. It is thought that the hypovascularity and low pH of the tumor microenvironment limits the activation of amifostine. Furthermore, tumors have lower levels of alkaline phosphatase than normal tissues [11]. In addition to scavenging free radicals, amifostine metabolites induce hypoxia by increasing oxygen consumption [12, 13], which further protects tissues in which the metabolites concentrate.
Amifostine has been tested extensively in clinical trials in various cancer types for the prevention of both acute and late injury. At least 30 different studies have evaluated the use of amifostine in preventing oral mucositis, and the results from these studies have been conflicting. A systematic review of these studies found that data supporting the use of amifostine for oral mucositis was inconclusive [14]. In non-small lung cancer , several small studies showed that amifostine minimized esophagitis [15–17]. However, a study with a larger number of patients receiving chemoradiotherapy was unable to support a reduction in physician-assessed esophagitis; however, amifostine was reported to ameliorate patient-reported swallowing impairment and pain [18, 19]. Amifostine has been shown in some series to be effective against proctitis and dermatitis in patients with pelvic malignancies who underwent radiotherapy [20–22]. It has also been effective in reducing soft tissue [23] and lung fibrosis [23, 24] in patients receiving radiation.
Despite a large number of trials that have been conducted to evaluate amifostine, its usefulness is limited. Many of the studies were conducted with a small and heterogeneous patient population, and dosing schedules differed between studies. In addition, there are several limitations associated with the use of the drug. To be effective, amifostine must be given 15–30 min before radiation and is only approved for intravenous delivery, which may be logistically challenging. Systemic delivery of amifostine is also associated with several side effects, including nausea, vomiting, sleepiness, and low blood pressure. Finally there is much debate over the use of amifostine with radiotherapy and chemoradiotherapy not only because of its side effects but because there are concerns that it may reduce the effectiveness of radiation treatment.
Nitroxides
Nitroxides are recycling antioxidants that have been shown to prevent cytotoxicity induced by oxidative stress as well as by radiation. Nitroxides interconvert between the oxidized and reduced form. In their oxidized state, nitroxides are a stable free radical referred to as a nitroxide radical. These radicals undergo hydrogen reductions to generate hydroxylamine. Both nitroxide radicals and hydroxylamine have antioxidant functions [7, 25]; however only nitroxides exhibit radioprotective effects. In vitro studies using various cell types have shown that nitroxides can reduce DNA damage and cell death induced by radiation [26, 27]. More importantly, systematic administration of nitroxides to mice resulted in decrease lethality after total body irradiation exposures, further substantiating their therapeutic potential [28, 29].
One of the more clinically promising nitroxides is tempol (4-hydroxy-2,2,6,6-tetramethylpiperidine-1-oxyl). Tempol has been studied as a radioprotector in both topical and systemic applications. Topical application of tempol to the skin of guinea pigs exposed to single and fractionated doses of radiation was capable of ameliorating alopecia [30, 31]. Systemic administration of tempol was capable of reducing the lethality of total body irradiation exposures [28, 29]. Systemic administration of tempol has also been shown to protect salivary glands from radiation [32].
As mentioned previously, to enhance the therapeutic ratio, a radioprotector must selectively protect normal and not tumor tissue. The selectivity of tempol for normal tissues has been addressed in several preclinical studies. Systemic administration of tempol in tumor-bearing mice had no effect on tumor growth, and administration with radiation had no impact on the dose of radiation that results in cure in 50 % of tumors at 30 days after treatment [33]. However, systemic administration of tempol was capable of protecting salivary glands and the skin from irradiation [32].
The differential effect of tempol in normal and tumor tissue has been hypothesized to relate to faster reduction to the hydroxylamine metabolite in tumor compared to normal tissues [33]. This hypothesis was evaluated by temporally tracking nitroxide levels with magnetic resonance imaging (MRI) . Tempol in its oxidized form acts as contrast agent and can be imaged by MRI. As tempol reduces into hydroxylamine , the contrast enhancement decreases because in its reduced form, it does not provide T1 contrast. By following the intensity of contrast enhancement over time, it was deduced that tempol is reduced faster into its non-radioprotective form in the tumor compared to surrounding normal tissue (Fig. 4.3) [34]. The unique ability to image tempol may allow a determination of the optimal time of tempol delivery and may further allow the unique opportunity to test this relationship in each patient to be treated.
Results from a redox imaging experiment of tumor and salivary glands. (a) Concentration maps overlaid on T2-weighted images corresponding to the hind leg region of a mouse. The tumor and the adjacent leg muscle are outlined in red. (b) The average tempol concentration inside the muscle and tumor was plotted as a function of time after injection. The concentration of tempol was determined in three different tumor models: SCCVII, KHT, and HT-29. For each time point after injection (20 s intervals), the average concentration was determined for each tissue. (c) Using the same technique as used in a and b, the concentration of tempol was determined in nine noncancerous tissue compartments. The error bars represent the standard error of the mean, and the lines connect the data points. Note the difference in tempol concentration in tumor compared to normal tissues as a function of time. Reproduced from Davis et al. [34]
Clinical translation of tempol has met with initial success. In a phase I clinical trial, tempol was effective at reducing alopecia in patients who underwent whole-brain radiotherapy [35]. Pharmacokinetic studies found that tempol was only detectable in 50 % of plasma samples after topical application and that the levels were minimal in those in which it was detected, suggesting that tumor protection via systemic leak was not a major concern in patients treated with topical tempol.
Other Candidate Radioprotectors
Naturally occurring antioxidants have also been tested for their efficacy as radioprotectors. Antioxidants such as α-tocopherol (vitamin E) and β-carotene have been demonstrated to reduce various radiation-induced injuries including xerostomia [36] and mucositis [37, 38], and in combination with pentoxifylline , α-tocopherol has been shown to reduce lung fibrosis [39, 40]. The use of these nutritional antioxidants as radioprotectors has come under question due to concerns that these agents may also interfere with tumor control either through radioprotection or via enhancement in the rate of second malignancies. Combined α-tocopherol and β-carotene supplementation given during and after radiation was also shown to increase the local recurrence rate of head and neck tumors [37]. These findings highlight the need to consider the possibility of tumor radioprotection carefully.
One promising antioxidant that could be an effective radioprotector is superoxide dismutase (SOD) . SOD is an endogenous enzyme that converts superoxide into oxygen and hydrogen peroxide. Transgene expression of SOD in animal models via gene therapy has been shown to protect against mucositis [41], esophagitis [42], and pneumonitis [43, 44]. Furthermore in animal models , SOD was demonstrated to selectively protect normal and not tumor tissue [45]. The major concern with this approach is the ability of SOD to access the primary target of radaition, DNA.
Melatonin is a hormone that has been shown in a number of studies to have radioprotective effects . It has the ability to directly scavenge free radicals and to increase the expression levels of antioxidant enzymes, including SOD, glutathione peroxidase, and catalase. It also has been reported to increase the efficiency of mitochondrial function, thereby reducing ROS levels generated by the electron transport chain [46]. In animal models, melatonin has been demonstrated to protect mice against lethal total body exposures [47, 48] and to protect a number of organs from radiation injury [49]. In vitro studies have demonstrated that melatonin sensitizes cancer cell lines to irradiation [50, 51].
Despite the fact that preclinical data supports that melatonin is a selective radioprotector, a phase II trial did not confirm activity in this regard. A Radiation Therapy Oncology Group (RTOG) trial randomized patients with brain metastases into two groups: morning versus nighttime high-dose melatonin. Melatonin was given to the patients during and following radiation . Results from this study were compared to historical controls who received only whole-brain radiotherapy. It was concluded that melatonin improved neither overall survival nor neurocognitive function after radiotherapy [52]. More recently, several in vitro studies have shown that melatonin at pharmacologic doses may actually have oxidant effects [53–55]. This finding has yet to be confirmed in vivo; nevertheless, it suggests caution and careful consideration of dosing is necessary when combining melatonin with radiotherapy .
Radiation Mitigators
Radiation mitigators reduce normal tissue damage after exposure to radiation through a variety of mechanisms. Unlike radioprotectors, which prevent damage from occurring, radiation mitigators minimize damage by acting upon physiologic processes that occur after radiation exposure but before the clinical manifestation of injury [4]. Radiation mitigators may be used to ameliorate both acute and late toxicities. Acute radiation toxicities, such as dermatitis and mucositis, are often caused by the death of rapidly dividing cells [56]. Damage to the rapidly proliferating stem cell compartment of organs, such as the skin and small intestine, eventually leads to loss of differentiated, specialized cells and results in the manifestation of tissue damage [57, 58]. Therefore, mitigators that effectively prevent acute injury often promote stem cell survival and proliferation.
Although the cellular response to radiation is almost immediate, the expression of late toxicities may be delayed for months to years after radiation exposure. Radiation activates a myriad of different signaling pathways that initiate pro-inflammatory, profibrotic, and vascular injury responses. These responses continue long after the initial radiation exposure and result in altered tissue homeostasis, fibrosis, vascular damage, atrophy, and necrosis [59]. Many radiation mitigators target key molecules or processes in the pathways that lead to late radiation injury. It is impossible to review every radiation mitigator identified in the space allowed. Thus, we have highlighted clinically approved or notable examples in the sections below.
Total Body Exposures
Normal tissue stem cells are responsible for regenerating tissues damaged by radiation and other processes. Stem cells are highly sensitive to radiation and are typically depleted in the radiation-damaged tissues [58, 60, 61]. Thus, a great deal of research has evaluated cytokines and growth factors that have the capacity to promote stem cell survival and proliferation. These approaches have been explored primarily as a method to reduce acute toxicity after localized and total body exposures.
Total body exposures can cause death through bone marrow failure and loss of intestinal integrity (gastrointestinal syndrome) [56]. At extreme doses death results from damage to the central nervous system and vascular collapse [56]. Intestinal damage is also a cause of major morbidity among transplant patients treated with total body irradiation. Many mitigant strategies for total body exposures are therefore geared toward preventing bone marrow failure and gastrointestinal syndrome by stimulating stem cell function. Intestinal injury is also a major dose-limiting factor during abdominal and pelvic radiation.
One example of this approach is the use of granulocyte colony-stimulating factor (G-CSF) as a radiation mitigant. In nonhuman primates , administration of G-CSF at 6 h after a total body exposure led to increased hematopoietic recovery [62]. In mice, administration of two separate doses of G-CSF at 24 h and 30 min before total body irradiation increased survival rates. Interestingly, there was no effect on survival when G-CSF was given 24 h after irradiation [63] suggesting that for the G-CSF to be effective, it must be given close to when radiation DNA damage occurs. This type of treatment may be useful in the setting of accidental total body exposures or in therapeutic exposures that require irradiation of extensive marrow compartments that are uninvolved by tumor . The use of G-CSF with therapeutic radiation must be approached with caution as G-CSF has been reported to drive epithelial to mesenchymal transition and tumor progression [64]. G-CSF is considered an effective treatment for accidental total body exposures and is part of the US Strategic National Stockpile [65].
Another agent in the stockpile, entolimod , is a toll-like receptor 5 (TLR5) agonist that has been shown to have mitigant efficacy in animal models of lethal total body exposures , dermatitis, and mucositis [66, 67], with no evidence of impaired tumor response [66, 67]. Entolimod, formerly known as CBLB502 , is a Salmonella flagellin derivative that activates the NF-kB pathway by binding toll-like receptor 5 (TLR5). It is more potent but less immunogenic and toxic compared to purified flagellin [67]. At high radiation doses, delivery of entolimod in mice shortly before irradiation led to reduced gastrointestinal and hematological injury and subsequent improvement in survival. Enhanced survival was also observed with postirradiation delivery of entolimod but only at low radiation doses [67]. Efficacy of entolimod was further tested in nonhuman primates, and data showed that entolimod protected primates from hematopoietic and lymphoid organ damage and lethality incurred by radiation [67]. G-CSF and interleukin-6 (IL-6) are two potential biomarkers for entolimod efficacy in mitigating radiation-induced injury . In irradiated and nonirradiated animals, levels of these cytokines were stimulated by entolimod in a TLR5- and dose- dependent manner. Furthermore, inhibiting G-CSF and IL-6 with neutralizing antibodies blocked the radiation mitigating effects of entolimod suggesting that the two cytokines are major mediators of entolimod’s mechanism of action [68]. The discovery of these two biomarkers may aid in determining the most optimal, efficacious dose to use in humans. Currently, entolimod is being evaluated in clinical trials for its capacity to mitigate radiation injury.
Palifermin is a truncated recombinant human keratinocyte growth factor (KGF) . KGF is a mitogenic factor with diverse functions in proliferation, survival, differentiation, DNA repair, and reactive oxygen species detoxification [69]. Animal studies have shown that administration of palifermin can reduce xerostomia [70], mucositis [71], and gastrointestinal injury [72, 73] induced by radiation. Palifermin is FDA approved for prevention of severe mucositis in hematologic cancer patients who receive chemoradiotherapy prior to stem cell transplant [74]. Additionally, a phase II clinical trial for head and neck cancer found that delivery of palifermin to patients receiving concurrent chemotherapy and hyperfractionated radiotherapy minimized the incidence, severity, and duration of oral mucositis [75]. In the same study, it was concluded that palifermin had no effect on survival or progression-free survival [75], suggesting an absence of tumor-promoting effects . The selectivity of palifermin for normal tissue may depend on whether or not the fibroblast growth factor receptor 2b (FGFR2b) , a cognate receptor for KGF, is expressed in tumor tissues. Preclinical studies have shown that KGF-FGFR2b signaling can promote tumor and metastatic phenotypes in breast, lung, and gastric cancer [76].
Because inflammation and vascular damage are two immediate radiation responses that can persist and cause late tissue damage, there has been much interest in mitigators that target inflammatory pathways. One such pathway is the thrombomodulin (THBD)-activated protein C (APC) . THBD is a transmembrane glycoprotein that binds thrombin and activates thrombin activatable fibrinolysis inhibitor (TAFI) [77]. In addition, it cleaves and activates the protein C zymogen , which in turn inactivates blood coagulation factors V and VIII via proteolysis [78]. The anticoagulant APC also promotes fibrinolysis and induces anti-inflammatory and cytoprotective activities in endothelial, neuronal, and innate immune cells [78]. In clinical and preclinical studies, it has been shown that THBD is significantly reduced and never recovered in intestinal endothelial cells after radiation [79–81]. More interestingly, systematic delivery of soluble THBD and APC in mice exposed to total body irradiation resulted in protection from hematological injury and lethality [82]. Numerous candidate mitigants that expand hematopoiesis or gastrointestinal recovery after total body exposures have been studied, such as metformin, lysophosphatidic acid mimics, genistein, and GSK-3 inhibitors [83–89].
Dermatitis
Radiation dermatitis is an acute toxicity of radiotherapy that can cause pain, increase the risk of infection, and result in the need for treatment breaks. Radiation dermatitis typically begins as erythema and progresses with increasing skin dose to induration, dry desquamation, moist desquamation, and finally to ulceration [56]. A number of agents have been tested as treatment for radiation dermatitis in small randomized trials. Several of these agents have been found to reduce the severity of radiation dermatitis to some degree [90–94]. Despite these findings, these agents are not in widespread clinical use at this time, and many patients are only treated with topical emollients for symptomatic relief. Mometasone furoate and betamethasone , topical steroids , have been shown in randomized trials to reduce the severity of radiation dermatitis [95, 96].
Fibrosis
Fibrosis is a common type of late tissue injury associated with radiation. TGF-β is the predominant signaling pathway that drives fibrosis [59], and accordingly, many radiation mitigators that target this pathway have been evaluated. Neutralizing antibodies against TGF-β have been shown to minimize fibrosis in animal models following radiation [97]. Halofuginone , a small molecular inhibitor that targets TGF-β signaling, also had similar protective effects in mice and showed normal tissue selectivity as it had no effect on tumor radiosensitivity [98]. Downstream molecules of the TGF-β signaling pathway, such as Smad3, have also been demonstrated to be possible therapeutic targets [99, 100].
It is noteworthy to mention that TGF-β has opposing, dual roles in cancer. In the early stages of cancer, it inhibits tumorigenesis but promotes metastasis as the disease progresses [101, 102]. Therefore, determining the appropriate dosing schedule for anti-TGF-β agents may be crucial in achieving therapeutic gain. Currently, there are no TGF-β-targeting agents in clinical trials specifically for mitigation of radiation-induced fibrosis, although there are several for fibrotic disease and scarring [103].
Statins are HMG-CoA reductase inhibitors that are traditionally used in clinic to lower cholesterol levels. Data from preclinical studies however suggest that statins may also be effective as a radiation mitigator for late and acute injury. In mice that received whole-lung irradiation, lovastatin was shown to reduce thrombopenia and mRNA levels of several pro-inflammatory and profibrotic genes including TNFα, IL-6, TGF-β, and connective tissue growth factor (CTGF) [104]. Lovastatin in a separate study was also shown to attenuate lung fibrosis and increase survival [105]. Moreover, other statins such as pravastatin were shown to attenuate CTGF expression and intestinal radiation fibrosis [106]. The mechanism by which statins regulate late tissue injury is unclear, but it has been hypothesized that it may involve the Rho pathways as inhibition of this pathway resulted in similar protective effects as seen with pravastatin [107, 108]. Statins are also known to inactivate Rho-GTPases further supporting the hypothesis [109]. Whether or not statins are safe to use during tumor radiotherapy remains to be determined. Several reports have shown that statins enhance the radiation cytotoxic effects in tumor tissue [110, 111], but further evaluation is needed.
Activation of the PDGF pathway has been implicated in the progression of radiation fibrosis. PDGFR inhibition via imatinib and other small molecule inhibitors has been shown to be effective in preclinical models of pulmonary and dermal fibrosis [112, 113]. One of the targets of imatinib , c-Abl , is known to be a downstream signaling intermediate of TGF-β in fibroblasts [114]. Furthermore, morphologic transformation and activation of gene expression in TGF-β-stimulated fibroblasts are dependent on the activation of c-Abl, suggesting that reduction in fibrosis with imatinib treatment may partially involve inhibition of TGF-β signaling.
Another group of agents thought to have efficacy in fibrosis and nephropathy are angiotensin-converting enzyme (ACE) inhibitors , agents that are widely used to treat hypertension and heart failure. These agents act by preventing the generation of Angiotensin II which is a vasoconstrictor [115]. More recently, animal studies have shown that ACE inhibitors can ameliorate radiation-induced normal tissue injury. For example, captopril, an ACE inhibitor, reduced pulmonary endothelial dysfunction in irradiated rats [116]. Captopril and other ACE inhibitors were also shown to minimize lung fibrosis caused by radiation [117, 118]. In addition to lung injury, ACE inhibitors have been reported to protect other organs such as the kidney [119] and skin [120, 121] from radiation injury. Importantly, ACE inhibitors have been shown to reduce radiation lethality from lung and kidney toxicity after total body exposures [119]. As for the effects of ACE inhibitors on tumors , a study of ramipril in mice bearing A549 xenografts had no effect on tumor response [121]. The mechanism by which captopril mitigates radiation injury is unclear. It has been hypothesized that it maybe through inhibition of angiotensin II production as angiotensin II is known to promote the expression of TGF-β , a known pro-fibrogenic factor [122, 123].
Small molecule inhibitors account for many of the agents thought to be effective as radiation mitigators. Another area of growing interest for radiation mitigation is cell-based therapies . Cell-based therapies can be used to inhibit inflammatory processes or to repopulate the damaged organ. The infusion of mesenchymal stem cells , an expandable, multipotent stem cell found in mesenchymal tissues, has been reported to mitigate radiation fibrosis in the lung and skin [124, 125]. These cells may be derived from a number of mesenchymal tissues such as bone marrow and adipose tissues . The use of these cells as a therapeutic option is of particular interest given that they can be harvested, expanded in vitro, and stored for future use. Further, these cells are considered immune privileged due to low expression of MHC-II, allowing the use of donor-derived mesenchymal stem cells as therapy. Importantly, the timing of delivery of these cells may be important in their efficacy. Evidence from skin fibrosis models suggests that the interaction of bone marrow stromal cells with activated macrophages results in macrophage repolarization , with elaboration of anti-inflammatory IL-10 [125]. Thus, it is possible that effective treatment with bone marrow stromal cells for mitigation requires a substantial accumulation of macrophages in the irradiated tissues, which is often not seen until several weeks after treatment . Stem cell-based therapies for radiation-induced organ dysfunction have also been studied for other types of radiation injury, such as liver injury and osteonecrosis [126–128].
Central Nervous System Injury
Patients with multiple brain tumors that cannot be surgically resected are commonly treated with whole-brain radiotherapy and/or stereotactic radiosurgery . Unfortunately, patients can experience cognitive impairments from tumor progression or radiation-induced demyelination, vascular damage, and white matter necrosis [129]. Although oxidative stress and inflammation have been implicated in promoting radiation-induced brain injury [130], more recent data suggests that brain tumors themselves may produce inflammatory changes in the CNS microenvironment that are independent of radiation [131]. Accordingly, agents that inhibit these cellular processes including ACE inhibitors , peroxisome proliferator-activated receptor-у (PPARу) agonists , and vascular endothelial growth factor (VEGF) inhibitors have been evaluated in animal models, and preclinical evidence suggests that it may be effective as radiation mitigators for brain injury [130, 132–137].
Clinical trials to mitigate radiation toxicity in the CNS have largely focused on technical approaches to reduce radiation dose to critical structures or off-label use of pharmacotherapies commonly used to treat dementia. RTOG 04 utilized intensity-modulated external beam radiation planning techniques to reduce the dose of radiation delivered to the hippocampus of patients requiring whole-brain radiation for treatment of brain metastases, with the hippocampal sparing technique producing improved performance on a neurocognitive evaluation at 4 months following treatment [138]. Acetylcholinesterase inhibitors , such as donepezil , have been evaluated for differences in neurocognition, mood, and quality of life outcomes in adult and in pediatric patients with some improvement compared to baseline at 24 weeks after radiation treatment [139, 140]. NMDA receptor antagonists such as memantine have produced some improvements in cognitive function over time and delayed neurocognitive decline 24 weeks after whole-brain radiotherapy [141].
Despite the current data demonstrating short-term benefits with some radiation mitigators in the CNS, there is a severe lack of understanding about the underlying biological processes which produce acute and long-term neurodegenerative changes. Most clinical strategies have relied on the assumption that the underlying biological process of neurocognitive decline seen in other conditions such as dementia also holds true for radiation-induced CNS damage. However, this assumption may be misplaced or may not reflect the entire spectrum of radiation-induced changes. The goal of current scientific investigations is to determine how the DNA damage response from radiotherapy to the CNS compartment translates to altered structural and biochemical changes so that more effective, targeted therapies may be developed to afford patients long-term, sustained benefits after radiation.
Treatment
Agents that are given after the development of radiation-induced symptoms are characterized as treatments. In general, preventing normal tissue injury with the use of radioprotectors and mitigators is preferable since some of these toxicities are irreversible and only limited treatments of variable efficacy exist. Treatments for radiation injury may be used for a short duration, or ongoing treatment may be required for prolonged periods to maintain clinical benefit. A comprehensive discussion of treatment of radiation injury is outside the scope of this section; however a few examples are highlighted below. In addition to the examples provided, a number of treatments have been studied for gastrointestinal toxicity, dermatologic toxicity, and mucositis with varying degrees of efficacy.
Fibrosis is a particularly challenging toxicity of radiotherapy, often considered to be irreversible. One treatment that has shown efficacy in this setting is pentoxifylline combined with vitamin E. A double blind, placebo-controlled trial of pentoxifylline and vitamin E in patients with radiation fibrosis of the skin and subcutaneous tissues after treatment for breast cancer found a marked regression in fibrotic surface area with treatment [142]. These findings were confirmed in a study of patients with radiation fibrosis at multiple sites, who were found to have improved range of motion and reduced pain with pentoxifylline treatment [143]. Importantly, long-term treatment with pentoxifylline and vitamin E appears to be necessary, with the possibility of rebound effects if treatment is discontinued too early [144]. Studies of pentoxifylline have also been completed for patients suffering from radiation injury in a number of organs with variable success [145–149].
Radiation pneumonitis may occur in up to 15 % of patients treated with thoracic radiotherapy [150]. Pneumonitis is characterized by fever, cough, and dyspnea with radiographic changes corresponding to the radiated field and may occur within the first 18 months after irradiation. Radiation pneumonitis is a diagnosis of exclusion, meaning that infection, tumor spread, and other causes of lung inflammation must be excluded. Radiation pneumonitis evolves into radiation fibrosis over time, although symptomatic pneumonitis is not a prerequisite for developing radiation fibrosis. Glucocorticoids remain the most effective treatment for radiation pneumonitis but are not considered an effective treatment for radiation fibrosis [151, 152]. Oxygen is also used as needed to limit hypoxia.
In patients receiving radiotherapy to the brain or head and neck, radiation brain necrosis may occur as a side effect. Traditionally, radiation necrosis has been managed with glucocorticoids, with surgical resection reserved for patients in whom radiation necrosis causes persistent symptoms. More recently, bevacizumab, a VEGF inhibitor, has been reported to substantially ameliorate radiation-induced brain necrosis [153–156]. In a randomized trial of bevacizumab versus placebo in 14 patients with biopsy or radiographically confirmed radiation necrosis, treatment with bevacizumab improved neurologic symptoms and signs in all patients [154]. All patients treated with placebo were allowed to cross over into the bevacizumab arm. No response was seen after treatment with the placebo; however, after bevacizumab treatment , only two patients developed a recurrence of radiation necrosis .
Challenges
A number of challenges exist in the effective preclinical and clinical development of the agents for use as radioprotectors and radiation mitigators. One major concern is the ability of animal models to predict the behavior of the human condition. Animals may have different pharmacokinetic responses to drugs, which may alter the appropriate timing and dosing of agents. Conventional radiotherapy often continues daily for up to 8 weeks, a condition challenging to replicate in animal models of injury in which one to ten fractions are typically delivered. Animals also tend to be of similar genetic background with no comorbidities that may affect drug metabolism, drug penetration, and susceptibility to radiation toxicity. And it cannot be forgotten that mice have fundamental biological differences from humans.
Another factor complicating the study of these agents is the use of chemotherapy concurrently with radiotherapy in a growing number of cancers. As a result, testing of these agents as radioprotectors or mitigators often requires assessment of effect in the context of combined radiation and chemotherapy, not just radiotherapy alone. This increases the level of complexity of preclinical studies, increases cost substantially, and complicates analysis of efficacy. To ensure that candidate radiation protectors or mitigators do not impair tumor control, additional studies should always be completed in tumor models before clinical translation if it is expected that they will be delivered in close proximity to radiotherapy.
Perhaps the greatest challenge for the development of effective radiation protectors and radiation mitigators is determining which patients would benefit most from treatment . Unless an agent has minimal cost and minimal toxicity, it would be unreasonable to treat every patient prophylactically unless the toxicity was so severe it would cause death or serious chronic injury in a substantial number of patients. In order to determine which patients would benefit the most from the use of these agents, it is necessary to develop effective means of predicting who will develop injury.
Finally, the clinical translation of agents used to prevent, mitigate, or treat radiation injury can be difficult because of the challenges involved in designing clinical trials capable of determining efficacy. Patients included in these trials will have different comorbidities and genetic background, which may variably affect the likelihood or predisposition to developing toxicity. Patients will have tumors of varying sizes or with characteristics that require treatment of different volumes or doses with radiotherapy. With newer techniques such as intensity-modulated radiation therapy , dose delivered to organs becomes increasingly complex to consider as a variable in these studies. Perhaps most importantly, the endpoint of these studies and grading scales used to assess efficacy must be carefully chosen to accurately reflect clinical benefit.
Despite these challenges, the development of radiation protectors, radiation mitigators, and treatment for radiation injury holds great promise for the growing number of patients that survive aggressive cancer therapy . Encouraging progress has been made in reducing normal tissue injury from radiotherapy, but clearly additional work is needed.
References
Durante M, Loeffler JS (2010) Charged particles in radiation oncology. Nat Rev Clin Oncol 7(1):37–43
Moding EJ, Kastan MB, Kirsch DG (2013) Strategies for optimizing the response of cancer and normal tissues to radiation. Nat Rev Drug Discov 12(7):526–542
Fu KK, Pajak TF, Trotti A et al (2000) A Radiation Therapy Oncology Group (RTOG) phase III randomized study to compare hyperfractionation and two variants of accelerated fractionation to standard fractionation radiotherapy for head and neck squamous cell carcinomas: first report of RTOG 9003. Int J Radiat Oncol Biol Phys 48(1):7–16
Stone HB, Moulder JE, Coleman CN et al (2004) Models for evaluating agents intended for the prophylaxis, mitigation and treatment of radiation injuries. Report of an NCI Workshop, December 3-4, 2003. Radiat Res 162(6):711–728
Citrin D, Cotrim AP, Hyodo F et al (2010) Radioprotectors and mitigators of radiation-induced normal tissue injury. Oncologist 15(4):360–371
Camphausen K, Citrin D, Krishna MC et al (2005) Implications for tumor control during protection of normal tissues with antioxidants. J Clin Oncol 23(24):5455–5457
Xavier S, Yamada K, Samuni AM et al (2002) Differential protection by nitroxides and hydroxylamines to radiation-induced and metal ion-catalyzed oxidative damage. Biochim Biophys Acta 1573(2):109–120
Kouvaris JR, Kouloulias VE, Vlahos LJ (2007) Amifostine: the first selective-target and broad-spectrum radioprotector. Oncologist 12(6):738–747
Calabro-Jones PM, Fahey RC, Smoluk GD et al (1985) Alkaline phosphatase promotes radioprotection and accumulation of WR-1065 in V79-171 cells incubated in medium containing WR-2721. Int J Radiat Biol Relat Stud Phys Chem Med 47(1):23–27
Yuhas JM (1980) Active versus passive absorption kinetics as the basis for selective protection of normal tissues by S-2-(3-aminopropylamino)-ethylphosphorothioic acid. Cancer Res 40(5):1519–1524
Giatromanolaki A, Sivridis E, Maltezos E et al (2002) Down-regulation of intestinal-type alkaline phosphatase in the tumor vasculature and stroma provides a strong basis for explaining amifostine selectivity. Semin Oncol 29(6 Suppl 19):14–21
Purdie JW, Inhaber ER, Schneider H et al (1983) Interaction of cultured mammalian cells with WR-2721 and its thiol, WR-1065: implications for mechanisms of radioprotection. Int J Radiat Biol Relat Stud Phys Chem Med 43(5):517–527
Glover D, Negendank W, Delivoria-Papadopoulos M et al (1984) Alterations in oxygen transport following WR-2721. Int J Radiat Oncol Biol Phys 10(9):1565–1568
Nicolatou-Galitis O, Sarri T, Bowen J et al (2013) Systematic review of amifostine for the management of oral mucositis in cancer patients. Support Care Cancer 21(1):357–364
Werner-Wasik M, Axelrod RS, Friedland DP et al (2002) Phase II: trial of twice weekly amifostine in patients with non-small cell lung cancer treated with chemoradiotherapy. Semin Radiat Oncol 12(1 Suppl 1):34–39
Antonadou D (2002) Radiotherapy or chemotherapy followed by radiotherapy with or without amifostine in locally advanced lung cancer. Semin Radiat Oncol 12(1 Suppl 1):50–58
Komaki R, Lee JS, Milas L et al (2004) Effects of amifostine on acute toxicity from concurrent chemotherapy and radiotherapy for inoperable non-small-cell lung cancer: report of a randomized comparative trial. Int J Radiat Oncol Biol Phys 58(5):1369–1377
Movsas B, Scott C, Langer C et al (2005) Randomized trial of amifostine in locally advanced non-small-cell lung cancer patients receiving chemotherapy and hyperfractionated radiation: radiation therapy oncology group trial 98-01. J Clin Oncol 23(10):2145–2154
Sarna L, Swann S, Langer C et al (2008) Clinically meaningful differences in patient-reported outcomes with amifostine in combination with chemoradiation for locally advanced non-small-cell lung cancer: an analysis of RTOG 9801. Int J Radiat Oncol Biol Phys 72(5):1378–1384
Kouvaris J, Kouloulias V, Kokakis J et al (2002) The cytoprotective effect of amifostine in acute radiation dermatitis: a retrospective analysis. Eur J Dermatol 12(5):458–462
Singh AK, Menard C, Guion P et al (2006) Intrarectal amifostine suspension may protect against acute proctitis during radiation therapy for prostate cancer: a pilot study. Int J Radiat Oncol Biol Phys 65(4):1008–1013
Simone NL, Menard C, Soule BP et al (2008) Intrarectal amifostine during external beam radiation therapy for prostate cancer produces significant improvements in Quality of Life measured by EPIC score. Int J Radiat Oncol Biol Phys 70(1):90–95
Koukourakis MI, Panteliadou M, Abatzoglou IM et al (2013) Postmastectomy hypofractionated and accelerated radiation therapy with (and without) subcutaneous amifostine cytoprotection. Int J Radiat Oncol Biol Phys 85(1):e7–e13
Antonadou D, Coliarakis N, Synodinou M et al (2001) Randomized phase III trial of radiation treatment +/- amifostine in patients with advanced-stage lung cancer. Int J Radiat Oncol Biol Phys 51(4):915–922
Samuni A, Goldstein S, Russo A et al (2002) Kinetics and mechanism of hydroxyl radical and OH-adduct radical reactions with nitroxides and with their hydroxylamines. J Am Chem Soc 124(29):8719–8724
Soule BP, Hyodo F, Matsumoto K et al (2007) Therapeutic and clinical applications of nitroxide compounds. Antioxid Redox Signal 9(10):1731–1743
Soule BP, Hyodo F, Matsumoto K et al (2007) The chemistry and biology of nitroxide compounds. Free Radic Biol Med 42(11):1632–1650
Hahn SM, Tochner Z, Krishna CM et al (1992) Tempol, a stable free radical, is a novel murine radiation protector. Cancer Res 52(7):1750–1753
Hahn SM, Krishna CM, Samuni A et al (1994) Potential use of nitroxides in radiation oncology. Cancer Res 54 (7 Suppl):2006s–2010s
Cuscela D, Coffin D, Lupton GP et al (1996) Protection from radiation-induced alopecia with topical application of nitroxides: fractionated studies. Cancer J Sci Am 2(5):273–278
Goffman T, Cuscela D, Glass J et al (1992) Topical application of nitroxide protects radiation-induced alopecia in guinea pigs. Int J Radiat Oncol Biol Phys 22(4):803–806
Cotrim AP, Hyodo F, Matsumoto K et al (2007) Differential radiation protection of salivary glands versus tumor by Tempol with accompanying tissue assessment of Tempol by magnetic resonance imaging. Clin Cancer Res 13(16):4928–4933
Hahn SM, Sullivan FJ, DeLuca AM et al (1997) Evaluation of tempol radioprotection in a murine tumor model. Free Radic Biol Med 22(7):1211–1216
Davis RM, Mitchell JB, Krishna MC (2011) Nitroxides as cancer imaging agents. Anticancer Agents Med Chem 11(4):347–358
Metz JM, Smith D, Mick R et al (2004) A phase I study of topical Tempol for the prevention of alopecia induced by whole brain radiotherapy. Clin Cancer Res 10(19):6411–6417
Chitra S, Shyamala Devi CS (2008) Effects of radiation and alpha-tocopherol on saliva flow rate, amylase activity, total protein and electrolyte levels in oral cavity cancer. Indian J Dent Res 19(3):213–218
Bairati I, Meyer F, Gelinas M et al (2005) Randomized trial of antioxidant vitamins to prevent acute adverse effects of radiation therapy in head and neck cancer patients. J Clin Oncol 23(24):5805–5813
Ferreira PR, Fleck JF, Diehl A et al (2004) Protective effect of alpha-tocopherol in head and neck cancer radiation-induced mucositis: a double-blind randomized trial. Head Neck 26(4):313–321
Misirlioglu CH, Demirkasimoglu T, Kucukplakci B et al (2007) Pentoxifylline and alpha-tocopherol in prevention of radiation-induced lung toxicity in patients with lung cancer. Med Oncol 24(3):308–311
Jacobson G, Bhatia S, Smith BJ et al (2013) Randomized trial of pentoxifylline and vitamin E vs standard follow-up after breast irradiation to prevent breast fibrosis, evaluated by tissue compliance meter. Int J Radiat Oncol Biol Phys 85(3):604–608
Guo H, Seixas-Silva JA Jr, Epperly MW et al (2003) Prevention of radiation-induced oral cavity mucositis by plasmid/liposome delivery of the human manganese superoxide dismutase (SOD2) transgene. Radiat Res 159(3):361–370
Stickle RL, Epperly MW, Klein E et al (1999) Prevention of irradiation-induced esophagitis by plasmid/liposome delivery of the human manganese superoxide dismutase transgene. Radiat Oncol Investig 7(4):204–217
Epperly MW, Bray JA, Krager S et al (1999) Intratracheal injection of adenovirus containing the human MnSOD transgene protects athymic nude mice from irradiation-induced organizing alveolitis. Int J Radiat Oncol Biol Phys 43(1):169–181
Epperly MW, Travis EL, Sikora C et al (1999) Manganese [correction of Magnesium] superoxide dismutase (MnSOD) plasmid/liposome pulmonary radioprotective gene therapy: modulation of irradiation-induced mRNA for IL-I, TNF-alpha, and TGF-beta correlates with delay of organizing alveolitis/fibrosis. Biol Blood Marrow Transplant 5(4):204–214
Epperly MW, Defilippi S, Sikora C et al (2000) Intratracheal injection of manganese superoxide dismutase (MnSOD) plasmid/liposomes protects normal lung but not orthotopic tumors from irradiation. Gene Ther 7(12):1011–1018
Acuna-Castroviejo D, Martin M, Macias M et al (2001) Melatonin, mitochondria, and cellular bioenergetics. J Pineal Res 30(2):65–74
Vijayalaxmi MML, Reiter RJ et al (1999) Melatonin and protection from whole-body irradiation: survival studies in mice. Mutat Res 425(1):21–27
Blickenstaff RT, Brandstadter SM, Reddy S et al (1994) Potential radioprotective agents. 1. Homologs of melatonin. J Pharm Sci 83(2):216–218
Mihandoost E, Shirazi A, Mahdavi SR et al (2014) Can melatonin help us in radiation oncology treatments? Biomed Res Int 2014:578137
Jang SS, Kim WD, Park WY (2009) Melatonin exerts differential actions on X-ray radiation-induced apoptosis in normal mice splenocytes and Jurkat leukemia cells. J Pineal Res 47(2):147–155
Alonso-Gonzalez C, Gonzalez A, Martinez-Campa C et al (2015) Melatonin sensitizes human breast cancer cells to ionizing radiation by downregulating proteins involved in double-strand DNA break repair. J Pineal Res 58(2):189–197
Berk L, Berkey B, Rich T et al (2007) Randomized phase II trial of high-dose melatonin and radiation therapy for RPA class 2 patients with brain metastases (RTOG 0119). Int J Radiat Oncol Biol Phys 68(3):852–857
Zhang HM, Zhang Y (2014) Melatonin: a well-documented antioxidant with conditional pro-oxidant actions. J Pineal Res 57(2):131–146
Medina-Navarro R, Duran-Reyes G, Hicks JJ (1999) Pro-oxidating properties of melatonin in the in vitro interaction with the singlet oxygen. Endocr Res 25(3-4):263–280
Wolfler A, Caluba HC, Abuja PM et al (2001) Prooxidant activity of melatonin promotes fas-induced cell death in human leukemic Jurkat cells. FEBS Lett 502(3):127–131
Hall EJ, Giaccia AJ (2006) Radiobiology for the Radiologist. Lippincott Williams & Wilkins, Philadelphia
Blanpain C, Fuchs E (2006) Epidermal stem cells of the skin. Annu Rev Cell Dev Biol 22:339–373
Metcalfe C, Kljavin NM, Ybarra R et al (2014) Lgr5+ stem cells are indispensable for radiation-induced intestinal regeneration. Cell Stem Cell 14(2):149–159
Bentzen SM (2006) Preventing or reducing late side effects of radiation therapy: radiobiology meets molecular pathology. Nat Rev Cancer 6(9):702–713
Iglesias-Bartolome R, Patel V, Cotrim A et al (2012) mTOR inhibition prevents epithelial stem cell senescence and protects from radiation-induced mucositis. Cell Stem Cell 11(3):401–414
Mizumatsu S, Monje ML, Morhardt DR et al (2003) Extreme sensitivity of adult neurogenesis to low doses of X-irradiation. Cancer Res 63(14):4021–4027
Bertho JM, Frick J, Prat M et al (2005) Comparison of autologous cell therapy and granulocyte-colony stimulating factor (G-CSF) injection vs. G-CSF injection alone for the treatment of acute radiation syndrome in a non-human primate model. Int J Radiat Oncol Biol Phys 63(3):911–920
Uckun FM, Souza L, Waddick KG et al (1990) In vivo radioprotective effects of recombinant human granulocyte colony-stimulating factor in lethally irradiated mice. Blood 75(3):638–645
Cui YH, Suh Y, Lee HJ et al (2015) Radiation promotes invasiveness of non-small-cell lung cancer cells through granulocyte-colony-stimulating factor. Oncogene 34:5372–5382
Singh VK, Romaine PL, Seed TM (2015) Medical countermeasures for radiation exposure and related injuries: characterization of medicines, FDA-approval status and inclusion into the strategic national stockpile. Health Phys 108(6):607–630
Burdelya LG, Gleiberman AS, Toshkov I et al (2012) Toll-like receptor 5 agonist protects mice from dermatitis and oral mucositis caused by local radiation: implications for head-and-neck cancer radiotherapy. Int J Radiat Oncol Biol Phys 83(1):228–234
Burdelya LG, Krivokrysenko VI, Tallant TC et al (2008) An agonist of toll-like receptor 5 has radioprotective activity in mouse and primate models. Science 320(5873):226–230
Krivokrysenko VI, Shakhov AN, Singh VK et al (2012) Identification of granulocyte colony-stimulating factor and interleukin-6 as candidate biomarkers of CBLB502 efficacy as a medical radiation countermeasure. J Pharmacol Exp Ther 343(2):497–508
Finch PW, Rubin JS (2004) Keratinocyte growth factor/fibroblast growth factor 7, a homeostatic factor with therapeutic potential for epithelial protection and repair. Adv Cancer Res 91:69–136
Lombaert IM, Brunsting JF, Wierenga PK et al (2008) Keratinocyte growth factor prevents radiation damage to salivary glands by expansion of the stem/progenitor pool. Stem Cells 26(10):2595–2601
Farrell CL, Rex KL, Kaufman SA et al (1999) Effects of keratinocyte growth factor in the squamous epithelium of the upper aerodigestive tract of normal and irradiated mice. Int J Radiat Biol 75(5):609–620
Farrell CL, Bready JV, Rex KL et al (1998) Keratinocyte growth factor protects mice from chemotherapy and radiation-induced gastrointestinal injury and mortality. Cancer Res 58(5):933–939
Cai Y, Wang W, Liang H et al (2013) Keratinocyte growth factor pretreatment prevents radiation-induced intestinal damage in a mouse model. Scand J Gastroenterol 48(4):419–426
Spielberger R, Stiff P, Bensinger W et al (2004) Palifermin for oral mucositis after intensive therapy for hematologic cancers. N Engl J Med 351(25):2590–2598
Brizel DM, Murphy BA, Rosenthal DI et al (2008) Phase II study of palifermin and concurrent chemoradiation in head and neck squamous cell carcinoma. J Clin Oncol 26(15):2489–2496
Finch PW, Mark Cross LJ, McAuley DF et al (2013) Palifermin for the protection and regeneration of epithelial tissues following injury: new findings in basic research and pre-clinical models. J Cell Mol Med 17(9):1065–1087
Bouma BN, Marx PF, Mosnier LO et al (2001) Thrombin-activatable fibrinolysis inhibitor (TAFI, plasma procarboxypeptidase B, procarboxypeptidase R, procarboxypeptidase U). Thromb Res 101(5):329–354
Mosnier LO, Zlokovic BV, Griffin JH (2007) The cytoprotective protein C pathway. Blood 109(8):3161–3172
Wang J, Zheng H, Ou X et al (2002) Deficiency of microvascular thrombomodulin and up-regulation of protease-activated receptor-1 in irradiated rat intestine: possible link between endothelial dysfunction and chronic radiation fibrosis. Am J Pathol 160(6):2063–2072
Richter KK, Fink LM, Hughes BM et al (1998) Differential effect of radiation on endothelial cell function in rectal cancer and normal rectum. Am J Surg 176(6):642–647
Richter KK, Fink LM, Hughes BM et al (1997) Is the loss of endothelial thrombomodulin involved in the mechanism of chronicity in late radiation enteropathy? Radiother Oncol 44(1):65–71
Geiger H, Pawar SA, Kerschen EJ et al (2012) Pharmacological targeting of the thrombomodulin-activated protein C pathway mitigates radiation toxicity. Nat Med 18(7):1123–1129
Xu G, Wu H, Zhang J et al (2015) Metformin ameliorates ionizing irradiation-induced long-term hematopoietic stem cell injury in mice. Free Radic Biol Med 87:15–25
Deng W, Kimura Y, Gududuru V et al (2015) Mitigation of the hematopoietic and gastrointestinal acute radiation syndrome by octadecenyl thiophosphate, a small molecule mimic of lysophosphatidic acid. Radiat Res 183(4):465–475
Patil R, Szabo E, Fells JI et al (2015) Combined mitigation of the gastrointestinal and hematopoietic acute radiation syndromes by an LPA2 receptor-specific nonlipid agonist. Chem Biol 22(2):206–216
Lee CL, Lento WE, Castle KD et al (2014) Inhibiting glycogen synthase kinase-3 mitigates the hematopoietic acute radiation syndrome in mice. Radiat Res 181(5):445–451
Landauer M, Kohler PA, Kortek Y et al (2009) Long-term care benefits and services in Europe. Gerontology 55(5):481–490
Zhou Y, Mi MT (2005) Genistein stimulates hematopoiesis and increases survival in irradiated mice. J Radiat Res 46(4):425–433
Davis TA, Clarke TK, Mog SR et al (2007) Subcutaneous administration of genistein prior to lethal irradiation supports multilineage, hematopoietic progenitor cell recovery and survival. Int J Radiat Biol 83(3):141–151
Chan RJ, Mann J, Tripcony L et al (2014) Natural oil-based emulsion containing allantoin versus aqueous cream for managing radiation-induced skin reactions in patients with cancer: a phase 3, double-blind, randomized, controlled trial. Int J Radiat Oncol Biol Phys 90(4):756–764
Stefanelli A, Forte L, Medoro S et al (2014) Topical use of phytotherapic cream (Capilen(R) cream) to prevent radiodermatitis in breast cancer: a prospective historically controlled clinical study. G Ital Dermatol Venereol 149(1):107–113
Herst PM, Bennett NC, Sutherland AE et al (2014) Prophylactic use of Mepitel Film prevents radiation-induced moist desquamation in an intra-patient randomised controlled clinical trial of 78 breast cancer patients. Radiother Oncol 110(1):137–143
Kong M, Hong SE (2013) Topical use of recombinant human epidermal growth factor (EGF)-based cream to prevent radiation dermatitis in breast cancer patients: a single-blind randomized preliminary study. Asian Pac J Cancer Prev 14(8):4859–4864
Ryan JL, Heckler CE, Ling M et al (2013) Curcumin for radiation dermatitis: a randomized, double-blind, placebo-controlled clinical trial of thirty breast cancer patients. Radiat Res 180(1):34–43
Hindley A, Zain Z, Wood L et al (2014) Mometasone furoate cream reduces acute radiation dermatitis in patients receiving breast radiation therapy: results of a randomized trial. Int J Radiat Oncol Biol Phys 90(4):748–755
Ulff E, Maroti M, Serup J et al (2013) A potent steroid cream is superior to emollients in reducing acute radiation dermatitis in breast cancer patients treated with adjuvant radiotherapy. A randomised study of betamethasone versus two moisturizing creams. Radiother Oncol 108(2):287–292
Anscher MS, Thrasher B, Rabbani Z et al (2006) Antitransforming growth factor-beta antibody 1D11 ameliorates normal tissue damage caused by high-dose radiation. Int J Radiat Oncol Biol Phys 65(3):876–881
Xavier S, Piek E, Fujii M et al (2004) Amelioration of radiation-induced fibrosis: inhibition of transforming growth factor-beta signaling by halofuginone. J Biol Chem 279(15):15167–15176
Flanders KC, Sullivan CD, Fujii M et al (2002) Mice lacking Smad3 are protected against cutaneous injury induced by ionizing radiation. Am J Pathol 160(3):1057–1068
Roberts AB, Piek E, Bottinger EP et al (2001) Is Smad3 a major player in signal transduction pathways leading to fibrogenesis? Chest 120(1 Suppl):43S–47S
Massague J (2008) TGFbeta in Cancer. Cell 134(2):215–230
Massague J, Heino J, Laiho M (1991) Mechanisms in TGF-beta action. Ciba Found Symp 157:51–59, discussion 59-65
Akhurst RJ, Hata A (2012) Targeting the TGFbeta signalling pathway in disease. Nat Rev Drug Discov 11(10):790–811
Ostrau C, Hulsenbeck J, Herzog M et al (2009) Lovastatin attenuates ionizing radiation-induced normal tissue damage in vivo. Radiother Oncol 92(3):492–499
Williams JP, Hernady E, Johnston CJ et al (2004) Effect of administration of lovastatin on the development of late pulmonary effects after whole-lung irradiation in a murine model. Radiat Res 161(5):560–567
Haydont V, Gilliot O, Rivera S et al (2007) Successful mitigation of delayed intestinal radiation injury using pravastatin is not associated with acute injury improvement or tumor protection. Int J Radiat Oncol Biol Phys 68(5):1471–1482
Haydont V, Bourgier C, Vozenin-Brotons MC (2007) Rho/ROCK pathway as a molecular target for modulation of intestinal radiation-induced toxicity. Br J Radiol 80(1):S32–S40
Bourgier C, Haydont V, Milliat F et al (2005) Inhibition of Rho kinase modulates radiation induced fibrogenic phenotype in intestinal smooth muscle cells through alteration of the cytoskeleton and connective tissue growth factor expression. Gut 54(3):336–343
Liao JK, Laufs U (2005) Pleiotropic effects of statins. Annu Rev Pharmacol Toxicol 45:89–118
Miller AC, Kariko K, Myers CE et al (1993) Increased radioresistance of EJras-transformed human osteosarcoma cells and its modulation by lovastatin, an inhibitor of p21ras isoprenylation. Int J Cancer 53(2):302–307
Fritz G, Brachetti C, Kaina B (2003) Lovastatin causes sensitization of HeLa cells to ionizing radiation-induced apoptosis by the abrogation of G2 blockage. Int J Radiat Biol 79(8):601–610
Horton JA, Chung EJ, Hudak KE et al (2013) Inhibition of radiation-induced skin fibrosis with imatinib. Int J Radiat Biol 89(3):162–170
Abdollahi A, Li M, Ping G et al (2005) Inhibition of platelet-derived growth factor signaling attenuates pulmonary fibrosis. J Exp Med 201(6):925–935
Daniels CE, Wilkes MC, Edens M et al (2004) Imatinib mesylate inhibits the profibrogenic activity of TGF-beta and prevents bleomycin-mediated lung fibrosis. J Clin Invest 114(9):1308–1316
Brown NJ, Vaughan DE (1998) Angiotensin-converting enzyme inhibitors. Circulation 97(14):1411–1420
Ward WF, Kim YT, Molteni A et al (1988) Radiation-induced pulmonary endothelial dysfunction in rats: modification by an inhibitor of angiotensin converting enzyme. Int J Radiat Oncol Biol Phys 15(1):135–140
Molteni A, Moulder JE, Cohen EF et al (2000) Control of radiation-induced pneumopathy and lung fibrosis by angiotensin-converting enzyme inhibitors and an angiotensin II type 1 receptor blocker. Int J Radiat Biol 76(4):523–532
Kma L, Gao F, Fish BL et al (2012) Angiotensin converting enzyme inhibitors mitigate collagen synthesis induced by a single dose of radiation to the whole thorax. J Radiat Res 53(1):10–17
Medhora M, Gao F, Wu Q et al (2014) Model development and use of ACE inhibitors for preclinical mitigation of radiation-induced injury to multiple organs. Radiat Res 182(5):545–555
Matsuura-Hachiya Y, Arai KY, Ozeki R et al (2013) Angiotensin-converting enzyme inhibitor (enalapril maleate) accelerates recovery of mouse skin from UVB-induced wrinkles. Biochem Biophys Res Commun 442(1-2):38–43
Kohl RR, Kolozsvary A, Brown SL et al (2007) Differential radiation effect in tumor and normal tissue after treatment with ramipril, an angiotensin-converting enzyme inhibitor. Radiat Res 168(4):440–445
Wolf G, Mueller E, Stahl RA et al (1993) Angiotensin II-induced hypertrophy of cultured murine proximal tubular cells is mediated by endogenous transforming growth factor-beta. J Clin Invest 92(3):1366–1372
Hahn AW, Resink TJ, Bernhardt J et al (1991) Stimulation of autocrine platelet--derived growth factor AA-homodimer and transforming growth factor beta in vascular smooth muscle cells. Biochem Biophys Res Commun 178(3):1451–1458
Jiang X, Jiang X, Qu C et al (2015) Intravenous delivery of adipose-derived mesenchymal stromal cells attenuates acute radiation-induced lung injury in rats. Cytotherapy 17(5):560–570
Horton JA, Hudak KE, Chung EJ et al (2013) Mesenchymal stem cells inhibit cutaneous radiation-induced fibrosis by suppressing chronic inflammation. Stem Cells 31(10):2231–2241
Chen Y, Li Y, Wang X et al (2015) Amelioration of hyperbilirubinemia in gunn rats after transplantation of human induced pluripotent stem cell-derived hepatocytes. Stem Cell Reports 5(1):22–30
Landis CS, Zhou H, Liu L et al (2015) Liver regeneration and energetic changes in rats following hepatic radiation therapy and hepatocyte transplantation by (3)(1)P MRSI. Liver Int 35(4):1145–1151
Jin IG, Kim JH, Wu HG et al (2015) Effect of bone marrow-derived stem cells and bone morphogenetic protein-2 on treatment of osteoradionecrosis in a rat model., J Craniomaxillofac Surg
Greene-Schloesser D, Robbins ME, Peiffer AM et al (2012) Radiation-induced brain injury: a review. Front Oncol 2:73
Steeg PS, Camphausen KA, Smith QR (2011) Brain metastases as preventive and therapeutic targets. Nat Rev Cancer 11(5):352–363
Smart D, Garcia Glaessner A, Palmieri D et al (2015) Analysis of radiation therapy in a model of triple-negative breast cancer brain metastasis. J Clin Exp Metastasis 32:717–727
Ryu S, Kolozsvary A, Jenrow KA et al (2007) Mitigation of radiation-induced optic neuropathy in rats by ACE inhibitor ramipril: importance of ramipril dose and treatment time. J Neurooncol 82(2):119–124
Lee TC, Greene-Schloesser D, Payne V et al (2012) Chronic administration of the angiotensin-converting enzyme inhibitor, ramipril, prevents fractionated whole-brain irradiation-induced perirhinal cortex-dependent cognitive impairment. Radiat Res 178(1):46–56
Jenrow KA, Brown SL, Liu J et al (2010) Ramipril mitigates radiation-induced impairment of neurogenesis in the rat dentate gyrus. Radiat Oncol 5:6
Greene-Schloesser D, Payne V, Peiffer AM et al (2014) The peroxisomal proliferator-activated receptor (PPAR) alpha agonist, fenofibrate, prevents fractionated whole-brain irradiation-induced cognitive impairment. Radiat Res 181(1):33–44
Zhao W, Payne V, Tommasi E et al (2007) Administration of the peroxisomal proliferator-activated receptor gamma agonist pioglitazone during fractionated brain irradiation prevents radiation-induced cognitive impairment. Int J Radiat Oncol Biol Phys 67(1):6–9
Jiang X, Engelbach JA, Yuan L et al (2014) Anti-VEGF antibodies mitigate the development of radiation necrosis in mouse brain. Clin Cancer Res 20(10):2695–2702
Gondi V, Pugh SL, Tome WA et al (2014) Preservation of memory with conformal avoidance of the hippocampal neural stem-cell compartment during whole-brain radiotherapy for brain metastases (RTOG 0933): a phase II multi-institutional trial. J Clin Oncol 32(34):3810–3816
Shaw EG, Rosdhal R, D'Agostino RB Jr et al (2006) Phase II study of donepezil in irradiated brain tumor patients: effect on cognitive function, mood, and quality of life. J Clin Oncol 24(9):1415–1420
Castellino SM, Tooze JA, Flowers L et al (2012) Toxicity and efficacy of the acetylcholinesterase (AChe) inhibitor donepezil in childhood brain tumor survivors: a pilot study. Pediatr Blood Cancer 59(3):540–547
Brown PD, Pugh S, Laack NN et al (2013) Memantine for the prevention of cognitive dysfunction in patients receiving whole-brain radiotherapy: a randomized, double-blind, placebo-controlled trial. Neuro Oncol 15(10):1429–1437
Delanian S, Porcher R, Balla-Mekias S et al (2003) Randomized, placebo-controlled trial of combined pentoxifylline and tocopherol for regression of superficial radiation-induced fibrosis. J Clin Oncol 21(13):2545–2550
Okunieff P, Augustine E, Hicks JE et al (2004) Pentoxifylline in the treatment of radiation-induced fibrosis. J Clin Oncol 22(11):2207–2213
Delanian S, Porcher R, Rudant J et al (2005) Kinetics of response to long-term treatment combining pentoxifylline and tocopherol in patients with superficial radiation-induced fibrosis. J Clin Oncol 23(34):8570–8579
Gothard L, Cornes P, Brooker S et al (2005) Phase II study of vitamin E and pentoxifylline in patients with late side effects of pelvic radiotherapy. Radiother Oncol 75(3):334–341
Gothard L, Cornes P, Earl J et al (2004) Double-blind placebo-controlled randomised trial of vitamin E and pentoxifylline in patients with chronic arm lymphoedema and fibrosis after surgery and radiotherapy for breast cancer. Radiother Oncol 73(2):133–139
Magnusson M, Hoglund P, Johansson K et al (2009) Pentoxifylline and vitamin E treatment for prevention of radiation-induced side-effects in women with breast cancer: a phase two, double-blind, placebo-controlled randomised clinical trial (Ptx-5). Eur J Cancer 45(14):2488–2495
Delanian S, Chatel C, Porcher R et al (2011) Complete restoration of refractory mandibular osteoradionecrosis by prolonged treatment with a pentoxifylline-tocopherol-clodronate combination (PENTOCLO): a phase II trial. Int J Radiat Oncol Biol Phys 80(3):832–839
Venkitaraman R, Price A, Coffey J et al (2008) Pentoxifylline to treat radiation proctitis: a small and inconclusive randomised trial. Clin Oncol (R Coll Radiol) 20(4):288–292
Yazbeck VY, Villaruz L, Haley M et al (2013) Management of normal tissue toxicity associated with chemoradiation (primary skin, esophagus, and lung). Cancer J 19(3):231–237
Abratt RP, Morgan GW, Silvestri G et al (2004) Pulmonary complications of radiation therapy. Clin Chest Med 25(1):167–177
Monson JM, Stark P, Reilly JJ et al (1998) Clinical radiation pneumonitis and radiographic changes after thoracic radiation therapy for lung carcinoma. Cancer 82(5):842–850
Gonzalez J, Kumar AJ, Conrad CA et al (2007) Effect of bevacizumab on radiation necrosis of the brain. Int J Radiat Oncol Biol Phys 67(2):323–326
Levin VA, Bidaut L, Hou P et al (2011) Randomized double-blind placebo-controlled trial of bevacizumab therapy for radiation necrosis of the central nervous system. Int J Radiat Oncol Biol Phys 79(5):1487–1495
Wang Y, Pan L, Sheng X et al (2012) Reversal of cerebral radiation necrosis with bevacizumab treatment in 17 Chinese patients. Eur J Med Res 17:25
Tye K, Engelhard HH, Slavin KV et al (2014) An analysis of radiation necrosis of the central nervous system treated with bevacizumab. J Neurooncol 117(2):321–327
Acknowledgment
This research was supported by the Intramural Research Program of the National Institutes of Health, National Cancer Institute.
Disclosures and Conflicts of Interests
The authors have no conflicts of interests or relationships to disclose.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Chung, S.I., Smart, D.K., Chung, E.J., Citrin, D.E. (2017). Radioprotection as a Method to Enhance the Therapeutic Ratio of Radiotherapy. In: Tofilon, P., Camphausen, K. (eds) Increasing the Therapeutic Ratio of Radiotherapy. Cancer Drug Discovery and Development. Humana Press, Cham. https://doi.org/10.1007/978-3-319-40854-5_4
Download citation
DOI: https://doi.org/10.1007/978-3-319-40854-5_4
Published:
Publisher Name: Humana Press, Cham
Print ISBN: 978-3-319-40852-1
Online ISBN: 978-3-319-40854-5
eBook Packages: MedicineMedicine (R0)