Abstract
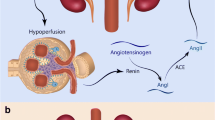
Renovascular hypertension occurs when an existing renal artery stenosis (RAS) leads to hypoperfusion of the juxtaglomerular apparatus of the kidney resulting in an increase in renin production with subsequent up regulation of the renin-angiotensin-aldosterone system. The most common pathology of RAS is atherosclerosis. Non-atherosclerotic etiologies of RAS include fibromuscular dysplasia (FMD), dissection, trauma, congenital hypoplastic syndromes and arteritis. Both medical management, and endovascular (angioplasty/stenting) or open revascularization strategies have been used to treat the hypertensive diathesis, however comparative data of the competing strategies is limited.
Recent randomized trials have demonstrated that medical management should be the first line therapy for patients with atherosclerotic RAS with revascularization, by either endovascular or open surgical technique, being reserved for patients with resistant hypertension (blood pressure greater than 140/90 despite maximum tolerated doses of three antihypertensive with one being a diuretic), non-cardiac flash pulmonary edema, or bilateral severe RAS (>90 %) associated with renal dysfunction (ischemic nephropathy).
Our recommended initial treatment strategy for atherosclerotic RAS mediated renovascular hypertension is optimal medical management. Renal artery angioplasty/stenting is safe with high degree of technical success, and should be considered as the first line of revascularization therapy. Open surgical revascularization is reserved for patients with renal artery anatomy not amenable to endovascular revascularization such as a renal artery occlusion and renal artery disease associated with extensive aortic occlusive or aneurysm disease requiring open repair.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
Introduction
Renovascular hypertension occurs when an existing RAS leads to hypoperfusion of the juxtaglomerular apparatus of the kidney resulting in an increase in renin production with subsequent up regulation of the renin-angiotensin-aldosterone system. Atherosclerosis accounts for more than 90 % of RAS and, affects approximately 6.8 % of elderly patients [1]. Other less common causes of RAS include FMD, arteritis, dissection, trauma, and congenital hypoplastic syndromes [2–4].
Renovascular hypertension exists in 1–5 % of hypertensive patients [5]. Historical features of patients with renovascular hypertension include one or more of the following: onset of hypertension in patients younger than 30 and older than 55, an abrupt increase in existing hypertension which was previously mild and well controlled, hypertension resistant to multi-drug therapy, unexplained episodes of “flash” pulmonary edema, unexplained deterioration in renal function in a non-diabetic patient [1]. Physical findings are few beyond a cuff blood pressure greater than 140/90. An epigastric bruit is found in a minority of patients and patients with atherosclerotic RAS may have associated extremity pulse deficits or bruits. The optimal treatment strategy is an area of controversy, particularly the comparative roles of best medical therapy versus revascularization by renal artery angioplasty/stenting. General goals of revascularization include the improvement in blood pressure control, the lowering of doses and number of antihypertensive medications, the preservation of renal function, and the prevention of cardiovascular events [6].
Search Strategy
A literature search of English language publications from 1990 to 2014 was used to identify published data on renovascular hypertension, RAS and treatment strategies. Primary database search was performed through PubMed. Terms used in the search were “renovascular hypertension” “atherosclerotic renal artery stenosis” “non-atherosclerotic renal artery stenosis” “Takaysu/renal artery stenosis” AND “interventions” “open surgery” “endovascular therapy” “medical management” “CORAL trial” “ASTRAL trial.” Six randomized controlled trials, ten cohort studies, one meta-analysis, one consensus statement, and 12 review articles were identified. The data was classified using the GRADE system (See Table 24.1).
Atherosclerotic Renal Artery Stenosis
Atherosclerotic RAS is more common than typically appreciated, and has a higher incidence in individuals with concomitant coronary artery (15–23 %), aortoiliac (28–38 %), and peripheral vascular disease (45–59 %). Atherosclerotic lesions typically involve the ostia and proximal renal artery [1]. Despite the prevalence of angiographic as well as hemodynamically significant RAS, the relative role of optimal medical therapy versus revascularization, particularly renal artery angioplasty/stenting is controversial.
Optimal Medical Therapy Versus Angioplasty/Stenting Plus Optimal Medical Therapy
Initial studies comparing medical versus endovascular management began with renal artery angioplasty alone. Three separate randomized trials (SNRASCG, EMMA, DRASTIC) all failed to demonstrate significant improvement in blood pressure or renal function with angioplasty alone when compared to optimal medical therapy [7–9]. The inadequacy of angioplasty alone for atherosclerotic RAS was first documented by van de Ven. He demonstrated that primary patency at 6 months was significantly better (75 % vs. 29 %) and restenosis rates were lower (14 % vs. 48 %) in patients subjected to angioplasty plus stenting when compared to angioplasty alone [10].
Based on these earlier studies, randomized trials were performed comparing optimal medical therapy with optimal medical therapy plus renal artery angioplasty/stenting. The STAR trial (Stent Placement and Blood Pressure and Lipid-Lowering for the Prevention of Progression of Renal Dysfunction Caused by Atherosclerotic Ostial Stenosis of the Renal Artery) and ASTRAL trial (Angioplasty and Stent for Renal Artery Lesions) were designed to determine whether optimal medical therapy plus renal artery angioplasty/stenting reduced adverse cardiovascular or renal events when compared to optimal medical therapy alone [11–13]. Both studies failed to demonstrate that renal artery angioplasty/stenting and optimal medical management were better than optimal medical management alone in reducing cardiovascular and renal events [12–14].
However both trials had serious design limitations. The STAR trial was criticized for being underpowered with only 140 patients and included many patients who may have had clinically insignificant RAS. While ASTRAL had a larger enrollment, 806 patients, only 59 % of patients randomized to angioplasty/stenting had angiographic evidence of RAS >70 %. In addition, the design of the trial excluded patients with RAS who in the opinion of the investigators would definitely benefit from renal artery angioplasty/stenting. This biased the results in general against angioplasty/stenting. Finally, there was concern over operator experience due to the low rate of technical success (78.6 %) of the angioplasty/stenting procedure [15, 16].
The Cardiovascular Outcomes in Renal Atherosclerotic Lesions (CORAL) study was a multicenter, randomized, controlled trial that compared optimal medical therapy with optimal medical therapy plus renal artery angioplasty/stenting in patients with atherosclerotic RAS. Inclusion criteria included patients with hypertension (SBP >155 mmHg or higher on ≥2 antihypertensive medications) and angiographic renal artery stenosis of at least 80–99 % or RAS greater than 60 % but less than 80 % with a systolic pressure gradient of at least 20 mmHg. The primary endpoint was the composite end point of death from cardiovascular or renal cause, myocardial infarction, stroke, hospitalization for heart failure, progressive renal insufficiency or need for permanent renal-replacement therapy. No significant difference in the primary composite endpoint was found between the medical and stenting/angioplasty groups (35.8 % and 35.1 %, P = 0.58). In addition, similar results were seen for the individual components of the composite endpoint [17].
CORAL as designed required that after randomization, patients were prohibited from crossing over to the competing arm for the duration of the study. By limiting crossovers, many patients with severe bilateral renal artery disease may have been treated outside the trial by renal artery angioplasty/stenting rather than subjecting them to the possibility of medical therapy only. This biased the study group towards patients with less severe renal artery disease and potentially excluded the subset of patients who may have received the most benefit from a renal artery intervention (i.e. severe bilateral renal artery stenosis associated with heart failure/flash pulmonary edema, malignant refractory hypertension or renal dysfunction) [18].
A recent meta-analysis of the six major randomized control trials (SNRASCG, EMMA, DRASTIC, STAR, ASTRAL, CORAL), concluded that renal artery angioplasty with or without stenting for atherosclerotic RAS was no better than optimal medical therapy alone in achieving blood pressure control or preserving renal function. However, it is important to note that in the angioplasty/stenting group a trend toward a lower rate of major events, including acute heart decompensation, renal dysfunction, stroke, and mortality was demonstrated [19].
Given the shortcomings of the available literature, particularly for patients who might truly benefit from renal artery angioplasty/stenting, a consensus document was produced in 2014 by the Society for Cardiovascular Angiography and Interventions (SCAI). The consensus is based on the major randomized trials, cohort studies, and the multi-societal guidelines recommendations from ACC/AHA 2005 Practice Guidelines for the management of patients with peripheral arterial disease.
The Consensus recommendations were divided into three categories based on varying levels of appropriateness for renal artery angioplasty/stenting: (1) Renal artery angioplasty/stenting represents appropriate care, (2) Renal artery angioplasty/stenting may represent appropriate care, and (3) Renal artery angioplasty/stenting rarely represents appropriate care. Patients deemed appropriate for renal artery angioplasty/stenting included those with cardiac disturbance syndrome or “flash” pulmonary edema, severe (>90 %) bilateral renal artery stenosis or stenosis to a solitary kidney, accelerated or resistant hypertension, or global renal ischemia associated with renal dysfunction. Patients in whom renal artery angioplasty/stenting may represent appropriate care include those with unilateral severe (90 %) renal artery stenosis, hypertension and prior episodes of unexplained congestive heart failure or patients at high risk for progressive ischemic nephropathy who could benefit from revascularization for stabilization of renal function. Finally, patients in whom renal artery angioplasty/stenting is rarely appropriate are those who have RAS with controlled blood pressure and normal renal function, mild to moderate renal artery stenosis, ischemic nephropathy already requiring hemodialysis greater than 3 months, and chronic total occlusions of the renal arteries [20]. Although not specifically addressed in this consensus statement, it is also important to emphasize that the “prophylactic” or “drive-by” renal artery angioplasty/stenting of clinically occult atherosclerotic RAS is rarely if ever appropriate [12–14].
Open Surgical Intervention
Surgical revascularization is considered for patients with anatomically challenging or high-risk atherosclerotic RAS not amenable to endovascular intervention. Blood pressure response rates in selected patients undergoing open surgical revascularization have been favorable, with up to 85 % being cured or showing significant improvement in the hypertensive diathesis [21]. Open surgical repair of atherosclerotic RAS is durable, with patency rates in one series as high as 97 % at a mean follow-up of 3 years [21]. In a large case series by Darling, 687 open renal artery reconstructions were performed over a 23-year period. The majority of procedures were performed in conjunction with aortic reconstruction (531/687), with an overall morbidity of 15.5 % and mortality of 5.5 %. In the subgroup of patients who underwent an isolated renal artery procedure, the morbidity was 14.1 % and mortality was 3.2 %. Primary graft patency at 5 years was 95 % [22].
Given the current use of renal artery angioplasty/stenting as the first line revascularization strategy for atherosclerotic RAS, the use of open renal artery revascularization for an isolated RAS is rare. However, a role for open surgery remains for patients with an occluded renal artery or arteries with early bifurcation, small diameter (<3 cm), or severe concentric calcification; or patients with other abdominal aortic pathology (i.e. aortoiliac occlusive disease, aneurysmal disease) that require concomitant open repair; or in selected patients who fail renal artery stenting or develop restenosis following intervention [20, 21].
Non-atherosclerotic Renal Artery Stenosis
Non-atherosclerotic RAS, primarily FMD, accounts for a significant portion of patients with renovascular hypertension [23]. Other non-atherosclerotic pathologies include renal artery dissection, aneurysm, arteritis, congenital hypoplastic syndromes and trauma. No randomized clinical trials for non-atherosclerotic RAS comparing optimal medical management to renal artery angioplasty/stenting exist, but the principles of medical and, endovascular or open revascularization are the same as for patients with atherosclerotic RAS. Optimal treatment is predicated on a variety of considerations including lesion pathology, patient age, severity of hypertension and associated renal dysfunction.
Optimal Medical Therapy Versus Angioplasty and Stenting (Non-atherosclerotic Renal Artery Stenosis)
The primary indication for renal revascularization in a patient with non-atherosclerotic renal artery disease is resistant hypertension [24]. When required in patients with FMD, renal angioplasty alone has been shown to be effective for most lesions isolated to the main renal artery. Three recent case series of renal angioplasty for FMD have documented reasonable results. Hypertension improvement was seen in 21–76 % of patients with a primary patency of the renal intervention being 50–71 % at 5 years [25–27]. Davies demonstrated that 71 % of patients maintained improvement or cure in hypertension at 5 years [28]. Stent placement is rarely if ever required for FMD and should be reserved for residual stenosis >30 % or a flow limiting dissection [26]. Other non-atherosclerotic renal artery pathologies are managed on a case-by-case basis, since depending on the anatomy and lesion, open or endovascular revascularization may be preferred.
Open Surgical Intervention (Non-atherosclerotic Renal Artery Stenosis)
Open surgical revascularization for FMD is uncommon and is reserved for patients with extension of FMD into secondary renal artery branches or associated with a renal artery aneurysm, or in patients who fail endovascular intervention. Overall cure and improvement rates in hypertension for open revascularization are 33 % to 63 % and 24 % to 57 % respectively. Five year primary graft patency ranges from 75 to 85 % [29–32]. In a recent series of 43 patients with non-atherosclerotic renal artery disease primary graft patency was 80 % at 5 years, and survival 78 % at 10 years. Open revascularization was associated with an improvement in blood pressure, less antihypertensive medication and improvement in renal function [33]. For patients with arteritis, and specifically Takayasu’s arteritis, open surgical revascularization is the primary revascularization strategy. Primary patency has been demonstrated as high as 79 % at 5 years, with a statistically significant reduction of anti-hypertensive medication requirements [34].
Recommendations
The management of renovascular hypertension is stratified by the RAS pathology: atherosclerotic and non-atherosclerotic.
For atherosclerotic RAS, optimal medical therapy is the appropriate first line therapy for most patients. Renal artery angioplasty/stenting should be considered for patients with significant RAS (>60 %) associated with “flash” pulmonary edema, resistant hypertension, or in selected patients with renal insufficiency and either severe (>90 %) bilateral renal artery stenosis or severe stenosis to a solitary functioning kidney. Open revascularization should be considered in patients who are reasonable surgical candidates who have unfavorable renal artery anatomy for angioplasty/stenting (i.e. early bifurcation, small diameter vessels (<3 cm); vessels with severe concentric calcification, renal artery occlusion) or those with associated renal artery aneurysms, complex aortic disease or concomitant aortic disease that requires open repair; or in selected patients with severe bilateral renal artery disease or a solitary kidney, especially if associated with renal dysfunction.
The optimal management of non-atherosclerotic renovascular hypertension varies and is largely dependent on RAS pathology. For patients with hypertension easily controlled with one or two antihypertensives, medical therapy is preferred. However, for patients with resistant hypertension revascularization should be considered. Balloon angioplasty alone is preferred for FMD confined to the main renal artery with open surgical revascularization employed for FMD associated renal artery aneurysms or branch vessel involvement; or patients who have failed endovascular management. Open surgical revascularization is also the primary therapy for selected non-FMD lesions, specifically renal artery stenosis secondary to Takayasu’s arteritis.
A Personal View of the Data
The prospective randomized trials to date have consistently failed to show any beneficial effect of renal artery angioplasty/stenting over optimal medical therapy alone for the treatment of renovascular hypertension due to atherosclerotic RAS. However, these trials by design excluded many patients who would have benefited the most from endovascular intervention. While ample evidence has demonstrated the safety and the high technical proficiency of renal artery angioplasty/stenting, the benefits in blood pressure control and preservation or improvement in renal function have been difficult to validate. Despite dissonance in the published data, we continue to be of the opinion that renal artery angioplasty/stenting has a beneficial role in a select subset of patients of with atherosclerotic RAS mediated renovascular hypertension.
Comparative data regarding non-atherosclerotic RAS is lacking. However, for FMD, simple angioplasty is safe and relatively effective. Unfortunately, the role of endovascular revascularization in non-FMD lesions is less certain. Data from our own institution indicates open revascularization is preferred for Takayasu’s arteritis and may be the optimal choice for most non atherosclerotic RAS. However, a comparator population is lacking for most non-FMD management leaving it up to physician judgment and experience to decide the appropriate treatment for the individual patient.
Recommendations
-
Renovascular Hypertension due to Atherosclerotic RAS
-
We recommend optimal medical therapy as first line treatment (evidence quality high; strong recommendation).
-
For patients with cardiac disturbance syndrome or “flash” pulmonary edema, severe (>90 %) bilateral renal artery stenosis or stenosis to a solitary kidney, accelerated or resistant hypertension (failure of >3 maximally tolerated medications including the use of a diuretic), or ischemic nephropathy, we recommend renal artery angioplasty/stenting (evidence quality moderate; strong recommendation).
-
For patients with unilateral severe (90 %) renal artery stenosis and resistant hypertension or prior episodes of congestive heart failure without a primary cardiac etiology or patients with progressive ischemic nephropathy who could benefit from revascularization for preservation of renal function, we recommend renal artery angioplasty/stenting as this group of patients may benefit (evidence quality moderate; moderate recommendation).
-
For patients with controlled blood pressure and normal renal function, mild to moderate RAS, chronic ischemic nephropathy already requiring hemodialysis greater than 3 months and chronic total occlusions of the renal arteries, we do not recommend renal artery angioplasty/stenting, and especially “prophylactic” or “drive-by” interventions on clinically occult atherosclerotic RAS (evidence quality high; strong recommendation).
-
Open surgical revascularization should be considered for those that meet criteria for revascularization but have lesion characteristics not amenable to endovascular management or have concomitant abdominal aortic pathology that requires open surgical repair (evidence quality strong; strong recommendation).
-
-
Renovascular Hypertension due to Non-atherosclerotic RAS: The optimal treatment for renovascular hypertension due to non-atherosclerotic RAS varies widely and largely depends on the pathology of the renal artery stenosis. Patients with resistant hypertension and FMD confined to the main renal artery are best managed initially by renal artery angioplasty. Other pathologies are managed on a case by case basis.
References
Kerut EK, Geraci SA, Falterman C, Hunter D, Hanawalt C, Giles TD. Atherosclerotic renal artery stenosis and renovascular hypertension: clinical diagnosis and indications for revascularization. J Clin Hypertens (Greenwich). 2006;8(7):502–9.
Olin JW, Froehlich J, Gu X, Bacharach JM, Eagle K, Gray BH, et al. The United States Registry for Fibromuscular Dysplasia: results in the first 447 patients. Circulation. 2012;125:3182–90.
Piercy KT, Hundley JC, Stafford JM, Craven TE, Nagaraj SK, Dean RH, et al. Renovascular disease in children and adolescents. J Vasc Surg. 2005;41:973–82.
Stanley JC, Criado E, Upchurch Jr GR, Brophy PD, Cho KJ, Rectenwald JE, et al. Pediatric renovascular hypertension: 132 primary and 30 secondary operations in 97 children. J Vasc Surg. 2006;44:1219–28.
Olin JW, Melia M, Young JR, Graor RA, Risius B. Prevalence of atherosclerotic renal artery stenosis in patients with atherosclerosis elsewhere. Am J Med. 1990;88(1N):46N–51.
Rundback JH, Sacks D, Kent KC, Cooper C, Jones D, Murphy T, et al. Guidelines for the reporting of renal artery revascularization in clinical trials. American Heart Association. Circulation. 2002;106:1572–85.
Webster J, Marshall F, Abdalla M, Dominicziak A, Edwards R, Isles GG, et al. Randomised comparison of percutaneous angioplasty vs continued medical therapy for hypertensive patients with atheromatous renal artery stenosis. J Hum Hypertens. 1998;12(5):329–35.
Plouin PF, Chatellier G, Darne B, Raynaud A. Blood pressure outcome of angioplasty in atherosclerotic renal artery stenosis: a randomized trail. Essai Multicentrique Medicaments vs Angioplastie (EMMA) Study Group. Hypertension. 1998;31(3):823–9.
van Jaarsveld BC, Krijnen P, Pieterman H, Derkx FH, Deinum J, Postma C, et al. The effect of ballon angioplasty on hypertension in atherosclerotic renal-artery stenosis. N Engl J Med. 2000;342(14):1007–14.
van de Ven PJ, Kaatee R, Beutler JJ, Beek FJ, Woittiez AJ, Buskens E, et al. Arterial stenting and balloon angioplasty in ostial atherosclerotic renovascular disease: a randomized trial. Lancet. 1999;353:282–6.
Mistry S, Ives N, Harding J, Fitzpatrick-Ellis K, Lipkin G, Kalra PA, et al. Angioplasty and STent for Renal Artery Lesions (ASTRAL trial): rationale, methods and results so far. J Human Hypertens. 2007;21(7):511–5.
Wheatley K, Ives N, Gray R, Kalra PA, Moss JG, Baigent C, et al. Revascularization versus medical therapy for renal-artery stenosis. N Engl J Med. 2009;361:1953–62.
Bax L, Woittiez AJ, Kouwenberg HJ, Mali WP, Buskens E, Beek FJ, et al. Stent placement in patients with atherosclerotic renal artery stenosis and impaired renal function: a randomized trial. Ann Intern Med. 2009;150:840–8.
Chrysochou C, Kalra PA. Current management of atherosclerotic renovascular disease–what have we learned from ASTRAL? Nephron Clin Pract. 2010;115(1):c73–81.
George JC, White CJ. Renal artery stenting: lessons from ASTRAL (Angioplasty and Stenting for Renal Artery Lesions). JACC Cardiovasc Interv. 2010;3(7):786–7.
Sarac TP. Influence and critique of the ASTRAL and CORAL Trials. Semin Vasc Surg. 2011;24(3):162–6.
Cooper CJ, Murphy TP, Cutlip DE, Jamerson K, Henrich W, Reid DM, et al; CORAL Investigators. Stenting and medical therapy for atherosclerotic renal-artery stenosis. N Engl J Med. 2014; 370(1):13–22.
Herrmann SM, Saad A, Textor SC. Management of atherosclerotic renovascular disease after Cardiovascular Outcomes in Renal Atherosclerotic Lesions (CORAL). Nephrol Dial Transplant. 2015;30(3):366–75.
Caielli P, Frigo AC, Pengo MF, Rossitto G, Maiolino G, Seccia TM, Calò LA, Miotto D, Rossi GP. Treatment of atherosclerotic renovascular hypertension: review of observational studies and a meta-analysis of randomized clinical trials. Nephrol Dial Transplant. 2015;30(4):541–53.
Parikh SA, Shishehbor MH, Gray BH, White CJ, Jaff MR. SCAI expert consensus statement for renal artery stenting appropriate use. Catheter Cardiovasc Interv. 2014;84(7):1163–71.
Cherr GS, Hansen KJ, Craven TE, Edwards MS, Liquish Jr J, Levy PJ, et al. Surgical management of atherosclerotic renovascular disease. J Vasc Surg. 2002;35:236–45.
Darling RC, Kreienberg PB, Chang BB, Paty PS, Lloyd WE, Leather RP, et al. Outcome of renal artery reconstruction: analysis of 687 procedures. Ann Surg. 1999;230(4):524–30; discussion 530–2.
Weaver FA, Kuehne JP, Papanicolaou G. A recent institutional experience with renovascular hypertension. Am Surg. 1996;62(3):241–5.
Davidson RA, Barri Y, Wilcox CS. Predictors of cure of hypertension in fibromuscular renovascular disease. Am J Kidney Dis. 1996;28:334–8.
Mousa AY, Campbell JE, Stone PA, Broce M, Bates MC, AbuRahma AF. Short- and long-term outcomes of percutaneous transluminal angioplasty/stenting of renal fibromuscular dysplasia over a ten-year period. J Vasc Surg. 2012;55(2):421–7.
Barrier P, Julien A, Guillaume C, Philippe O, Herve R, Francis J. Technical and clinical results after percutaneous angioplasty in nonmedial fibromuscular dysplasia: outcome after endovascular management of unifocal renal artery stenosis in 30 patients. Cardiovasc Intervent Radiol. 2010;33:270–7.
Thatipelli MR, Huettl EA, McKusick MA, Misra S. Angioplasty for renal artery fibromuscular dysplasia in older hypertensive patients. Angiology. 2009;60(6):714–8.
Davies MG, Saad WE, Peden EK, Mohiuddin IT, Naoum JJ, Lumsden AB. The long-term outcomes of percutaneous therapy for renal artery fibromuscular dysplasia. J Vasc Surg. 2008;48(4):865–71.
Marekovic Z, Mokos I, Krhen I, Goreta NR, Roncevic T. Long-term outcome after surgical kidney revascularization for fibromuscular dysplasia and atherosclerotic renal artery stenosis. J Urol. 2004;171(3):1043–5.
Anderson CA, Hansen KJ, Benjamin ME, Keith DR, Craven TE, Dean RH. Renal artery fibromuscular dysplasia: results of current surgical therapy. J Vasc Surg. 1995;22(3):207–16.
Reiher L, Pfeiffer T, Sandmann W. Long-term results after surgical reconstruction for renal artery fibromuscular dysplasia. Eur J Vasc Endovasc Surg. 2000;20(6):556–9.
Novick AC, Ziegelbaum M, Vidt DG, Gifford Jr RW, Pohl MA, Goormastic M. Trends in surgical revascularization for renal artery disease: ten years’ experience. JAMA. 1987;257(4):498–501.
Ham SW, Kumar SR, Wang BR, Rowe VL, Weaver FA. Late outcomes of endovascular and open revascularization for nonatherosclerotic renal artery disease. Arch Surg. 2010;145(9):832–9.
Weaver FA, Kumar SR, Yellin AE, Anderson S, Hood DB, Rowe V, et al. Renal revascularization in Takaysu arteritis-induced renal artery stenosis. J Vasc Surg. 2004;39(4):749–57.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Dunn, J.C., Ham, S.W., Weaver, F.A. (2017). In Patients with Renovascular Hypertension Is There a Role for Open or Endovascular Revascularization Compared to Medical Management?. In: Skelly, C., Milner, R. (eds) Difficult Decisions in Vascular Surgery. Difficult Decisions in Surgery: An Evidence-Based Approach. Springer, Cham. https://doi.org/10.1007/978-3-319-33293-2_24
Download citation
DOI: https://doi.org/10.1007/978-3-319-33293-2_24
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-33291-8
Online ISBN: 978-3-319-33293-2
eBook Packages: MedicineMedicine (R0)