Abstract
Agnathia/otocephaly complex (OMIM #202650) is a rare, congenital malformation characterized by multiple malformations involving anatomic structures originating from the first pharyngeal arch as a consequence of failed mesenchymal migration of the maxillary prominence and atrophy of the development of the mandibular prominences [1]. Features of agnathia/otocephaly complex are absence or hypoplasia of the mandible, microstomia, hypoglossia/aglossia, and variable anterior midline fusion of the ears (melotia, synotia) [2]. An incidence of less than 1 in 70,000 births has been estimated [3]. The complex has been linked with a heterozygous mutation of the PRRX1 gene on chromosome 1q24. Genotype/phenotype heterogeneity is possible, and the disease may be inherited in either autosomal recessive or autosomal dominant patterns. In addition, the environmental teratogens have been associated [2, 4]. Recently, it has been demonstrated that perturbations in the PRRX1 and OTX2 genes may alter DNA signaling pathways, suggesting a role in palatal development [2, 5, 6].
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
1 Introduction
Agnathia/otocephaly complex (OMIM 202650) is a rare congenital malformation characterized by multiple malformations involving anatomic structures originating from the first pharyngeal arch as a consequence of failed mesenchymal migration of the maxillary prominence and atrophy of the development of the mandibular prominences [1]. Features of agnathia/otocephaly complex are absence or hypoplasia of the mandible, microstomia, hypoglossia/aglossia, and variable anterior midline fusion of the ears (melotia, synotia) [2]. An incidence of less than 1 in 70,000 births has been estimated [3]. The complex has been linked with a heterozygous mutation of the PRRX1 gene on chromosome 1q24. Genotype/phenotype heterogeneity is possible, and the disease may be inherited in either autosomal recessive or autosomal dominant patterns. In addition, association with environmental teratogens have been described [2, 4]. Recently, it has been demonstrated that perturbations in the PRRX1 and OTX2 genes may alter DNA signaling pathways, suggesting a role in palatal development [2, 5, 6].
2 Case Description
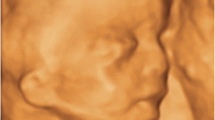
A 39-year-old nulliparous woman was referred for level II ultrasound examination for an unspecified complex facial anomaly detected on a routine third-trimester scan. The ultrasound examination was performed using transabdominal and transvaginal two-dimensional (2D) and three- and four-dimensional (3D/4D) volumetric probes. Digital 2D ultrasound imaging and 3D/4D volume datasets were stored onto an optical disk for offline analysis. Ultrasound examination showed findings consistent with congenital agenesis of the mandible (agnathia), proboscis, and hypotelorism (Fig. 17.1). Multiple skeletal anomalies were visualized, consisting of the absence of the right ulna and right fibula associated with shortening of the contralateral long bones, syndactyly of 5th finger of right hand, and bilateral clubfoot. A multicystic right dysplastic kidney, single umbilical artery, and polyhydramnios were additionally seen. 3D ultrasound volume datasets were transmitted to an expert at remote site (HW) and a physical model reconstruction on photopolymerized resin was constructed using postprocessing software. Specifically, Mimics v. 12, Materialise (Leuven, Belgium), was used for 3D virtual model reconstruction, and the model was exported into a standard triangular language (STL) format and converted into an “OBJ” extension for adjustment using 3D modeling polygonal software (Autodesk Mudbox, San Francisco, CA, USA).
A presumptive diagnosis of agnathia/otocephaly complex associated with multiple congenital anomalies was entertained (Fig. 17.2). Although prognosis of the disease was poor and the parents declined fetal magnetic resonance imaging (MRI) investigation, the 3D virtual model improved the parents’ understanding of the fetal anomalies and aided counseling. Termination of pregnancy was not performed due to legal limitations. Intrauterine fetal demise occurred shortly after ultrasound examination and gross pathology confirmed the antenatal diagnosis (Fig. 17.3). Interestingly, the anatomic details of the 3D virtual model substantially overlapped with the characteristic features of the agnathia/otocephaly complex.
3 Discussion
Agnathia/otocephaly complex may be isolated [7] or associated with other anomalies. Anencephaly and meningomyelocele [8], skeletal, genitourinary, and cardiovascular anomalies, and situs inversus have been reported [3]. However, the most common associated congenital anomaly is holoprosencephaly [9, 10].
The antenatal diagnosis of agnathia/otocephaly complex is challenging. Although it has most commonly been detected in the third trimester [4, 11, 12], detection at 12 weeks has also been reported [13]. 3D ultrasound ultrasound may aid the prenatal diagnosis of agnathia/otocephaly complex [4, 10–13] and a systematic look at the “CHIN” is advocated (“CHIN”: chin, headbone outline, inner head, nuchal translucency) [13]. 3D ultrasound has also proven to be of clinical value in the characterization of the disease and in planning neonatal treatment by the multispecialty team [14], although outcome is generally poor and newborns requires ventilatory support [1].
In addition to 3D virtual modeling of the anomaly that improves parents’ understanding and enhances prenatal counseling, 3D computed tomography (CT) scan has been used postmortem in a case of agnathia/otocephaly complex associated with organomegaly to obtain detailed anatomic information about this lethal disease [15].
This work was based on a previously published report [4].
References
Gekas J, Li B, Kamnasaran D. Current perspectives on the etiology of agnathia-otocephaly. Eur J Med Genet. 2010;53:358–66.
Herman S, Delio M, Morrow B, Samanich J. Agnathia-otocephaly complex: a case report and examination of the OTX2 and PRRX1 genes. Gene. 2012;494:124–9.
Faye-Petersen O, David E, Rangwala N, Seaman JP, Hua Z, Heller DS. Otocephaly: report of five new cases and a literature review. Fetal Pediatr Pathol. 2006;25:277–96.
Menezes GA, Araujo Junior E, Lopes J, Belmonte S, Tonni G, Werner H. Prenatal diagnosis and physical model reconstruction of agnathia-otocephaly with limb deformities (absent ulna, fibula and digits) following maternal exposure to oxymetazoline in the first trimester. J Obstet Gynecol Research. 2016;42:1016–20.
Sergouniotis PI, Urquhart JE, Williams SG, Bhaskar SS, Black GC, Lovell SC, Whitby DJ, Newman WG, Clayton-Smith J. Agnathia-otocephaly complex and asymmetric velopharyngeal insufficiency due to an in-frame duplication in OTX2. J Hum Genet. 2015;60:199–202.
Chassaing N, Sorrentino S, Davis EE, Martin-Coignard D, Iacovelli A, Paznekas W, Webb BD, Faye-Petersen O, Encha-Razavi F, Lequeux L, Vigouroux A, Yesilyurt A, Boyadjiev SA, Kayserili H, Loget P, Carles D, Sergi C, Puvabanditsin S, Chen CP, Etchevers HC, Katsanis N, Mercer CL, Calvas P, Jabs EW. OTX2 mutations contribute to the otocephaly-dysgnathia complex. J Med Genet. 2012;49:373–9.
Yang SH, Seo YS, Lee YS, Choi SJ, Kim YA, Kim JH. Prenatal sonographic diagnosis of isolated agnathia: a case report. Ultrasound Obstet Gynecol. 2003;22:190–3.
Tos T, Ceylaner S, Senel S, Aktas S, Alp Y. A case of otocephaly with anencephaly and meningomyelocele. Genet Couns. 2010;21:325–8.
Rolland M, Sarramon MF, Bloom MC. Astomia-agnathia-holoprosencephaly association. Prenatal diagnosis of a new case. Prenat Diagn. 1991;11:199–203.
Chaoui R, Heling KS, Thiel G, Karl K. Agnathia-otocephaly with holoprosencephaly on prenatal three-dimensional ultrasound. Ultrasound Obstet Gynecol. 2011;37:745–8.
Hisaba WJ, Milani HJ, Araujo Júnior E, Passos JP, Barreto EQ, Carvalho NS, Helfer TM, Pares DB, Nardozza LM, Moron AF. Agnathia-otocephaly: prenatal diagnosis by two- and three-dimensional ultrasound and magnetic resonance imaging. Case report. Med Ultrason. 2014;16:377–9.
Tantbirojn P, Taweevisit M, Sritippayawan S, Tanawattanacharoen S, Uerpairojkit B. Prenatal three-dimensional ultrasonography in a case of agnathia-otocephaly. J Obstet Gynaecol Res. 2008;34:663–5.
Huissoud C, La Mela Jumel A, Bisch C, Dijoud F, Pages O, Rudigoz RC. Take a look at the CHIN! --early diagnosis of isolated agnathia using two- and three-dimensional sonography. Fetal Diagn Ther. 2008;24:246–9.
Umekawa T, Sugiyama T, Yokochi A, Suga S, Uchida K, Sagawa N. A case of agnathia-otocephaly complex assessed prenatally for ex utero intrapartum treatment (EXIT) by three-dimensional ultrasonography. Prenat Diagn. 2007;27:679–81.
Rajan PV, Wing DA, Bocian M, McKeown A. Computed tomographic reconstruction of a fetus with the dysgnathia complex (agnathia-otocephaly). Prenat Diagn. 2007;27:130–2.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Werner, H., Tonni, G., Menezes, G.A., Araujo Júnior, E. (2017). 3D Virtual Model Reconstruction by 3D Ultrasound Volume Data Sets in a Case of Prenatally Diagnosed Agnathia/Otocephaly Complex Associated with Multiple Congenital Anomalies. In: Tonni, G., Sepulveda, W., Wong, A. (eds) Prenatal Diagnosis of Orofacial Malformations. Springer, Cham. https://doi.org/10.1007/978-3-319-32516-3_17
Download citation
DOI: https://doi.org/10.1007/978-3-319-32516-3_17
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-32514-9
Online ISBN: 978-3-319-32516-3
eBook Packages: MedicineMedicine (R0)