Abstract
The developing lung is vulnerable to adverse conditions during prenatal and postnatal development; the peripheral lung, including alveoli and small conducting airways, is especially vulnerable. Owing to a limited capacity for repair, the functional effects of lung injury or altered development during fetal or early postnatal life may persist into later life, affecting lung function and disease vulnerability. A range of environmental factors can likely cause epigenetic alterations in key genes regulating lung development, thus contributing to persistent changes in lung structure and/or function. New experimental evidence supports the effectiveness of cell-based therapies to prevent or repair neonatal lung injury, and clinical trials are now underway in very preterm infants at risk of developing bronchopulmonary dysplasia.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Development
- Fetus
- Alveoli
- Conducting airways
- Epithelium
- Surfactant
- Lung function
- Lung hypoplasia
- Tobacco smoking
- Fetal growth restriction
- Preterm birth
- Inflammation
- Oxidative stress
- Alcohol
- Respiratory infection
- Nutrition
- Mesenchymal stromal cells
Evidence for Developmental Programming: Epidemiological and Experimental Evidence
Like many organs, the lung can be affected during its period of development in such a way that its later structure and function are altered. This can occur as a result of altered structural development or altered cellular function as a result of genetic or epigenetic alterations. Many such alterations can impair lung function for much or all of the life span and can also increase the risk of later pulmonary illness. Developmental insults or challenges that have the potential to induce persistent alterations in lung structure or function include physical and metabolic changes in the developmental environment. As the lung undergoes development during both prenatal and early postnatal life, its development can be altered by environmental and/or epigenetic factors that affect the intrauterine environment or the environment during infancy and childhood. Altered development can include gross structural alterations such as lung hypoplasia caused by congenital diaphragmatic hernia (CDH) or oligohydramnios, for example, or more subtle changes that may lead to persistent oxidative stress or inflammation of lung tissue.
In this chapter, we review current knowledge of factors commonly operating during development that can persistently affect the structure and function of the lung , and the underlying processes. It has long been known that impaired lung function during early life is associated with impaired lung function later in life; this is known as “tracking” of lung function [1]. More recently, it has become accepted that impaired lung function during early life can be caused by environmental factors operating during fetal and early postnatal life, and that these can consequently lead to impaired lung function and increased disease risk much later in life: this has become known as “developmental programming” [2, 3]. These two concepts (tracking and developmental programming) are both manifestations of the same phenomenon, as both are caused by persistent changes in the structure and/or function of lung tissue. They are both likely a result of lung development proceeding along a genetically determined program, such that alterations in development at one stage cannot be corrected at a subsequent stage.
Evidence for developmental programming of the lung in humans has been derived from numerous epidemiological and clinical studies and in recent years has become supported and enhanced by an abundance of experimental evidence. Lung diseases of infancy and childhood thought to have fetal or neonatal origins include chronic lung disease of prematurity, lung hypoplasia , persistent pulmonary hypertension (PPH), and asthma. Pulmonary diseases of later life, such as chronic obstructive pulmonary disease (COPD), are thought to have their origins in early life [4]. Similarly, it is likely that accelerated lung aging may be a consequence of altered lung development , such as impaired alveolarization .
Fetal Causes of Altered Lung Development and Later Pulmonary Illness
Fetal Nutrition and Growth
Restricted fetal growth affects around 8–10 % of live births and is recognized as a significant cause of perinatal morbidity [5]. Multiple organs are known to be developmentally affected by fetal growth restriction (FGR), in that their structure and function can be persistently altered. One of the first studies to show a link between FGR and later pulmonary health was an epidemiological study of adult men in the UK [6]; that study showed statistical links between low birthweight (and weight at 1 year) on later lung function and death from respiratory illness. Since that study was published, similar studies from several countries have confirmed that FGR can have a persistent adverse effect on the developing lung [7]. These observations have led to numerous investigations of the underlying processes. The most common causes of FGR are placental insufficiency and a maternal diet that is restricted in calories or protein. Placental insufficiency usually occurs as a result of maternal vascular disease (e.g., hypertension or pre-eclampsia), multiple gestation or maternal tobacco smoking . In the presence of placental insufficiency and FGR, the fetus is likely to be hypoglycemic, hypolipidemic, acidemic, and hypoxemic [8, 9]. Thus, the etiology of altered lung development in the presence of FGR is likely to be multifactorial, and to date it has been difficult to isolate any given factor as being a cause of altered lung development .
Respiratory Effects of FGR Observed During Infancy and Childhood
During infancy, FGR has been associated with respiratory insufficiency and poor lung function [10–13]. It is now clear that this deficit in lung function persists into childhood and beyond, supporting the concept of “tracking” of lung function. School-age children who experienced FGR have poorer lung function, as evidenced by reduced expiratory flow rates and greater airway reactivity, compared to children of the same age born with normal birthweight [14–16]. Many infants with FGR are born preterm, and such infants are found to have even poorer lung function than FGR infants born at term [17]. In twin pairs of children and adolescents, it is apparent that the twin with the lower birthweight, indicative of FGR, has poorer lung function and increased airway reactivity [18, 19]. Such studies of twins are important because they eliminate many maternal and genetic factors.
Respiratory Effects of FGR Observed During Adult Life
Early studies on the relation between fetal growth and adult lung function only had access to data on birthweight. Therefore, such data could have included subjects who were of low birthweight as a result of preterm birth . Later studies have avoided the potentially confounding effects of preterm birth by only including subjects born at term [20]; such studies have in general confirmed a significant relationship between birthweight at term and respiratory illness during adulthood [21–24]. Nine studies of lung function in male and female subjects, performed in several different countries, formed the basis of a meta-analysis of birthweight effects on adult lung function [7]; the analysis revealed a small but significant reduction in adult forced expiratory volume in 1 s (FEV1) with decreasing birthweight. Taken together with lung function data from infants and children, the above studies provide strong evidence that FGR causes changes in lung development that impair lung function throughout postnatal life and may contribute to COPD in adult subjects of low birthweight [25, 26]. Although there are apparently no morphometric analyses of the lungs of adults exposed to FGR, data from animal studies suggest that impaired alveolarization and structural alterations to small conducting airways (see below) play an important mechanistic role.
Experimental Studies
Numerous animal studies using a variety of species have been performed in an attempt to elucidate the mechanisms by which FGR alters fetal lung development [10]. Experimentally, FGR has been induced by maternal food restriction (often protein restriction), mechanical constriction of the uterine or umbilical arteries, maternal hyperthermia or pre-pregnancy ablation of placentation sites.
Using sheep, a series of studies has shown that FGR, experimentally induced by placental insufficiency, has a profound effect on fetal lung development . In such models, the major factors leading to changes in organ development are likely to be fetal nutrient restriction and hypoxemia, as well as possible elevations in circulating corticosteroids. FGR was induced to coincide with the saccular and early alveolar stages of lung development, which occur before birth in sheep as in humans [27]. The effects of FGR on lung architecture were studied in the near-term fetus , 8-week-old lamb (childhood equivalent), and 2-year-old (adult) sheep. Interestingly, some changes not present before term became apparent with postnatal development. At 8 weeks of age, there were 31 % fewer alveoli in FGR lambs, compared to controls, and this reduction was still evident in adult sheep [28, 29]. In accordance with fewer alveoli, the alveoli of FGR animals were larger at both 8 weeks and 2 years. Thus, FGR induced by late-gestational nutrient restriction led to a permanent reduction in alveolarization , which resulted in a reduction in alveolar surface area relative to both lung volume and body weight [29]. It was also found that the inter-alveolar walls were significantly thicker than in control animals at 8 weeks (73 % thicker) and 2 years (47 % thicker) [29] due to an increased extracellular matrix ; as alveolar walls normally become progressively thinner with lung development, thicker alveolar walls suggest persistently inhibited development. Importantly, the blood–air barrier was also thicker: in adults, it was 43 % thicker after FGR than in control animals [29]. The combination of fewer alveoli, thicker alveolar walls, and a thicker blood–air barrier would be likely to reduce pulmonary compliance and diffusing capacity, as found at 8 weeks [30]. The persistence of such alterations in lung architecture into adulthood would be expected to impair adult lung function and to increase the rate of the age-related decline in lung function.
The findings in sheep are largely consistent with findings in other laboratory species following nutrient restriction coincident with lung development [31–33]. Using rodent models of FGR, it has been shown that the deposition of extracellular matrix proteins, including elastin, is impaired by FGR [32, 34], and that the impairment of alveolarization is likely to be mediated by alterations in retinoic acid receptors [35]. In addition to alterations in alveolar structure, pulmonary surfactant has been reported to be affected by FGR, although findings are not consistent between models [10].
The conducting airways may also be affected by FGR, although few structural studies have been undertaken. In near-term fetal sheep subjected to late-gestation FGR, it was found that airway walls were proportionally thinner than in controls (less cartilage and epithelium ); however, these changes had resolved by 8 weeks after birth [36]. There was also evidence that airway submucosal glands were attenuated in the FGR animals after birth [36].
The mechanisms by which FGR alters lung development , with some changes persisting into adult life, are still under investigation. In most cases, FGR results from placental insufficiency, which induces a complex and variable mix of physiological alterations in the fetus , including reduced nutrient supply, altered levels of growth factors , hypoxemia, and endocrine alterations, including elevated levels of circulating corticosteroids [37]. Each of these factors has the potential to alter aspects of prenatal lung development , including alveolarization . Owing to the postnatal lung’s limited ability to recover from structural deficits that occur during development, many of the alterations induced during fetal (and neonatal) life are likely to persist throughout life. There is now strong evidence that FGR induces epigenetic alterations in genes that are critical to lung development [38].
Amniotic Fluid Volume
The fetal lung develops in an expanded state; indeed, the volume of luminal liquid within the lung during fetal life (fetal lung liquid) is greater than functional residual capacity (FRC) in the postnatal air-filled lung [39]. A remarkable feature of fetal lung development is that the degree of lung expansion by fetal lung liquid has a profound, direct effect on lung growth. This is clearly demonstrated experimentally by the removal of lung liquid leading to cessation of lung growth and lung hypoplasia ; conversely, chronically increasing the degree of fetal lung expansion (e.g., by tracheal occlusion) leads to augmented growth of lung tissue [39].
An inadequate amount of amniotic fluid surrounding the fetus (oligohydramnios) is a common cause of fetal lung hypoplasia . Clinically, oligohydramnios is associated with respiratory insufficiency at birth and increased hospitalization during childhood [40]. Oligohydramnios can be caused by reduced production of amniotic fluid (usually due to fetal renal abnormalities) or by premature rupture of the fetal membranes. The lack of amniotic fluid allows the elastic uterine wall to “force” the fetal trunk into a state of increased flexion; this exaggerated trunk flexion increases intra-abdominal pressure, elevating the fetal diaphragm and compressing the fetal lung, as observed in the sheep and human fetus [41, 42]. Thoracic dimensions are reversibly reduced by experimental oligohydramnios [43]. The degree of lung hypoplasia at the time of birth is related to the duration and severity of the oligohydramnios. Oligohydramnios caused by reduced fetal urine production occurs from earlier in gestation than oligohydramnios resulting from membrane rupture; hence, the effects on the developing lung are likely to be more severe.
Attempts have been made to restore amniotic fluid volume, and hence fetal lung expansion, by infusing an appropriate fluid into the amniotic sac. In an ovine model of oligohydramnios, serial infusions of artificial amniotic fluid led to a reduction in lung hypoplasia and normalized airspace fraction [44]. A recent review of five randomized controlled trials suggests that amnioinfusion leads to a reduction in pulmonary hypoplasia [45]. It is likely that amnioinfusion improves fetal lung development by reducing the degree of fetal trunk flexion, allowing the fetal lungs to expand; it may also improve placental perfusion, resulting in improved oxygenation and nutrient delivery to the fetus [45].
Congenital Diaphragmatic Hernia
A less common cause of fetal lung hypoplasia is CDH, in which a defect in closure of the diaphragm (unilateral or more rarely bilateral) allows the lung to recoil and abdominal contents to enter the chest. The defect normally occurs during embryogenesis, resulting in the lung being unexpanded from an early stage in its development . CDH affects 1–4 per thousand live births, and usually has a poor prognosis; it is fatal in about 30 % of cases.
Fetal lung hypoplasia , if severe, can lead to respiratory distress or death at birth. The hypoplastic lung has a reduced number of airway generations, fewer alveoli , an underdeveloped vascular network, and hence a reduced capacity for gas exchange. The extent to which the hypoplastic lung can recover structurally after birth is unclear, as few studies have examined the potential for catch-up lung growth following intrauterine compromise. Given the limited ability of the postnatal lung to form new alveoli, it is likely that the lungs of survivors of CDH have reduced gas exchange, which may in turn limit exercise capacity. A recent study of almost 100 survivors of CDH aged up to 3 years showed that lung function was impaired; the extent of impairment was related to the degree of lung hypoplasia at birth and the duration of mechanical ventilation [46].
One of the major long-term complications of CDH is PPH, which is thought to be a result of poorly developed vasculature in the lung , and impaired vasodilator mechanisms [47]. PPH is evident in infants and children with CDH, and is likely to persist into adulthood, potentially contributing to adult lung disease; however, current survivors of CDH are still young and therefore there is little information available on the long-term outcomes of CDH survivors.
The fetal lungs can be expanded by occlusion of the trachea , due to the continued production of fetal lung liquid by the distal lung epithelium [39]. In animal studies, tracheal occlusion has been shown to increase pulmonary luminal volume, thereby rapidly accelerating lung tissue growth and alveolarization [48]. Currently, there is an ongoing clinical trial of tracheal occlusion performed endoscopically under local anesthesia in fetuses diagnosed with CDH [49].
Fetal Exposures
Fetal exposure to an increasing number of chemicals is now known to alter lung development , with long-term consequences. These include the products of maternal tobacco smoking , alcohol ingested by the mother, antenatal corticosteroids, and pro-inflammatory cytokines. Each of these exposures has been reported to alter lung development in the fetus , with potential long-term consequences for lung function and respiratory health.
Maternal Tobacco Smoking and Nicotine
Tobacco smoking during pregnancy has long been known to impact upon placental function, fetal lung development , and postnatal respiratory health in exposed offspring. Fetal exposure to the products of maternal smoking leads to an increased postnatal risk of pulmonary infections, wheezing, reduced lung function (low FEV1), and asthma [50–52]. The products of maternal smoking that are the most important causal factors in altering fetal lung development are likely to be nicotine and carbon monoxide, both of which readily cross the placenta, although many other chemicals may also play a role. Numerous experimental studies have been performed to identify the mechanisms by which maternal smoking alters lung development: these have largely focused on the role of whole smoke or nicotine administered to the mother [53]. Such studies have shown that fetal nicotine exposure permanently impairs alveolarization and reduces the ability of exposed offspring to protect themselves against environmental stressors. Nicotine has been shown to be genotoxic, to increase the production of reactive oxygen species (ROS), and to reduce the antioxidant capacity of the lung [54]. Thus, the fetal lungs develop in an environment of oxidant–antioxidant imbalance, with concomitant adverse effects of oxidants and nicotine on cell integrity [54]. As a result of fetal disturbances to lung development, exposed offspring are more prone to develop respiratory disease s such as asthma and emphysema later in life [53, 54]. Recent studies have shown that retinoic signalling in the fetal lung may be perturbed by maternal smoking [50], and that collagen deposition may be increased via upregulated transforming growth factor (TGF)-β signalling [55]. It is now considered likely that maternal smoking is capable of inducing alterations in the fetal epigenome that could result in life-long alterations in lung function [56].
Maternal Alcohol Consumption
Recent studies suggest that a substantial number of women consume alcohol during pregnancy, with 30 % of women in the USA reporting consumption of alcohol at some stage during their pregnancy and 8.3 % reporting consumption of four or more alcoholic drinks on one occasion at least once during pregnancy [57]. This is cause for concern, as experimental studies indicate that ethanol readily crosses the placenta [58]. Indeed, chronic maternal alcohol consumption can lead to a spectrum of developmental defects in the offspring, collectively termed fetal alcohol spectrum disorders (FASD). The incidence of FASD is estimated to be 1 in 100 live births, although the prevalence could be as high as 2–5 % [59, 60]. Fetal alcohol syndrome is the most severe form of FASD and is characterized by growth restriction, craniofacial dysmorphology, and central nervous system dysfunction. Although most research into the effects of fetal ethanol exposure has been focused on the developing brain, it has also been associated with congenital abnormalities of the respiratory tract [61] and an increased susceptibility to respiratory infection s in childhood [62]. In animal models, exposure to high levels of ethanol throughout gestation or at mid-gestation causes reduced fetal lung growth [63, 64]; however, moderate exposure in late gestation has no effect [65]. Despite a lack of effect on lung growth or alveolarization , moderate ethanol exposure in late gestation increases collagen deposition in the ovine fetal lung, decreases surfactant protein (SP) levels, and alters surfactant phospholipid composition [65–67]; such changes could compromise lung function after birth. Evidence also suggests that prenatal ethanol exposure may reduce the ability of the lungs to clear pathogens, as indicated by decreases in ciliary beat frequency [66] and pro-inflammatory cytokine gene expression [65], an increase in apoptosis of alveolar macrophages and a decrease in alveolar macrophage function both under normal conditions and in response to infection [68–70]. A recent study of prepubescent sheep exposed to ethanol in late gestation, however, reported no significant differences in lung growth, lung architecture, pro-inflammatory cytokine gene expression, and surfactant phospholipid composition, although SP-D mRNA levels were increased [67]. As ethanol exposure did not continue to birth or after birth, this study suggests that the developing lung has some capacity to undergo repair in the absence of further exposure. As SP-D has immunoregulatory roles and roles in surfactant phospholipid homeostasis [39], the observed increase in SP-D mRNA levels following ethanol exposure suggests that innate pulmonary immunity and lung function may be persistently altered. Further studies investigating the long-term effects of fetal ethanol exposure on the lung are warranted.
Antenatal Exposure to Corticosteroids
During the last several decades, corticosteroids have been routinely administered to pregnant women who show signs of preterm delivery (8–12 % of pregnancies). These agents, primarily betamethasone and dexamethasone, are administered during preterm labor to enhance lung maturation in the preterm fetus , thereby increasing its chances of survival. The principal beneficial effect of corticosteroids for preterm survival is the stimulation of surfactant production and release by the immature lung; this occurs as a result of an increase in the number and maturity of alveolar epithelial type ll cells [71]. It has long been recognized that, as well as stimulating surfactant production, corticosteroids inhibit secondary septation and alveolarization in the immature lung when administered before or during the process of alveolarization [72]. The effects of corticosteroids during development can be long lasting in many tissues [73], depending on the timing and duration of corticosteroid exposure, and the lung is also likely to be permanently affected, potentially as a result of epigenetic modifications [74]. This persistent effect has been clearly demonstrated in rats exposed to corticosteroids during early postnatal life [75] and is thought to result from a persistent imbalance between cell proliferation and cell death in secondary septa [76]. Because such a high proportion of infants who are born preterm (i.e., birth prior to 37 completed weeks of gestation) are exposed to corticosteroids, the adverse effects of corticosteroids on structural development of the lung, and hence on later lung function , are likely to be a significant factor in the etiology of respiratory illness and reduced lung function during all stages of life.
Intrauterine Inflammation (Chorioamnionitis)
In many pregnancies, the fetus is exposed to inflammation of the surrounding membranes (chorion and amnion) as a result of bacterial infection; this is often referred to as chorioamnionitis. Intrauterine inflammation is present in up to 60 % of pregnancies that result in preterm birth , and it is now considered that inflammation plays a causal role in the etiology of preterm birth [77]. In such pregnancies, the fetus is exposed to inflammatory mediators over prolonged periods, and it is now clear that these mediators can enter the fetal lungs [78]. Since the first demonstration that intrauterine inflammation improves respiratory function in the preterm neonate [79], numerous studies have investigated the impact of intrauterine inflammation on fetal lung development . Such studies have shown that lipopolysaccharide (LPS), a component of the Gram-negative bacterial cell wall, and pro-inflammatory cytokines accelerate the maturation of alveolar epithelial type ll cells in the preterm fetus, such that they produce more surfactant ; in addition, pulmonary vascular development is enhanced [80]. It has also been reported that intrauterine inflammation inhibits pulmonary elastin deposition and alveolarization [81]; these effects may be transient, as other studies have not shown a persistent effect of intrauterine inflammation on lung morphometry [82, 83]. The long-term effects of intrauterine inflammation on the lungs have received very little attention. However, a recent study in sheep has shown that it can lead to persistent changes in gene expression of pro-inflammatory cytokines in the postnatal lung, as well as reductions in protein expression of SP-A and -C [82]; together, these changes may impair innate immunity of the postnatal lung. It is clear that more research is needed regarding the pulmonary programming effects of intrauterine inflammation.
Neonatal Causes of Lung Disease or Altered Lung Development
Early postnatal environments that interfere with normal lung development and maturation may result in persistent alterations in lung structure and function, increasing the risk for respiratory disease in adult life. The first two years of life are an important time for lung development , and many studies have shown that a large proportion of adult respiratory diseases stem from events that occur during infancy and early childhood. In the sections below, we provide an overview of some of the factors in the neonatal period that can alter lung development and may be key contributors to adult lung disease.
Preterm Birth
Preterm birth, defined as birth prior to 37 completed weeks of gestation, affects approximately 11 % of pregnancies worldwide [84]. Preterm birth can be categorized into very preterm birth (<32 weeks’ gestation) and extremely preterm birth (<28 weeks’ gestation). As lung development occurs throughout gestation, and critical stages of lung development occur late in gestation, babies born preterm are born with structurally and functionally immature lungs. Very preterm infants are born during the saccular stage of lung development, which is prior to the formation of definitive alveoli . Extremely preterm babies can be born during the canalicular (pre-saccular) stage of lung development and therefore are born with a thick lung interstitium and prior to the production of surfactant . Owing to lung immaturity, infants born more than 6–8 weeks before term usually experience respiratory insufficiency at birth, referred to as the respiratory distress syndrome (RDS). Therefore, infants who are born very preterm or extremely preterm usually require respiratory support to ensure their survival; the degree of support and the length of stay in hospital are proportional to the degree of prematurity [85]. Respiratory support of preterm infants can involve supplemental oxygen therapy and/or assisted ventilation, including continuous positive airway pressure (CPAP), nasal intermittent positive pressure ventilation (nIPPV), or mechanical ventilation via an endotracheal tube. Improvements in the respiratory care of preterm infants, especially the introduction of exogenous surfactant therapy, more gentle ventilation strategies and more cautious use of supplemental oxygen, have resulted in an increase in their survival, in particular those who are born at earlier gestational ages. Despite the necessity of this respiratory intervention, the incidence of chronic lung disease or bronchopulmonary dysplasia (BPD) in this population remains high [86]. BPD is characterized by disrupted lung development, in particular alveolar simplification (fewer and larger alveoli), variable interstitial fibrosis , and increased airway smooth muscle (see Fig. 5.1) [87, 88].
Histological representation of the alveolar region of a lung from (a; ×25 magnification) a term infant, and (b; ×50 magnification) a surfactant -treated preterm infant with BPD. Photomicrographs of a preterm infant with “healed” BPD, demonstrating (c; ×30 magnification) diffuse alveolar septal fibrosis (bottom) with enlarged alveoli (top), and (d; ×100 magnification) hyperplastic smooth muscle (arrow) surrounding a bronchiole (modified from [196])
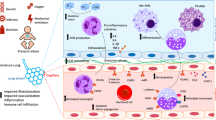
Although BPD is a multifactorial disease, major factors in its etiology are thought to be inflammation , oxidative stress , and inappropriate repair mechanisms due to prolonged exposure to high oxygen concentrations (hyperoxia) during supplemental oxygen therapy or mechanical ventilation [89–91]. Mechanical ventilation can contribute to lung injury by inducing inflammation as a result of repetitive tissue stretch (shear stress) and over-inflation (barotrauma) of incompliant lung tissue; particularly damaging is repetitive opening of collapsed alveoli in atelectatic regions of the surfactant deficient lung [92]. Numerous other fetal and neonatal factors associated with preterm birth can contribute to the pathogenesis of BPD or alterations in lung development; such factors include the severity of prematurity, exposure to antenatal or postnatal corticosteroids, prenatal infection or inflammation, postnatal infection, intrauterine growth restriction, and inadequate postnatal nutrition (see Fig. 5.2) [91, 93]. Regardless of etiology, it is now apparent that persistent changes within the lungs of preterm infants, especially those who developed BPD, lead to an increased risk of poor lung function in childhood and late adolescence, as indicated by reductions in FEV1, forced mid-expiratory flow (FEF25–75 %), peak expiratory flow (PEF), transfer across the lung of carbon monoxide (TLCO), and increased airway resistance [94–99]. Individuals who were born preterm also have an increased risk of reduced exercise capacity [97, 98], asthma [94, 99–101], COPD [102], and respiratory infection s [103–105] later in life. The contribution of hyperoxia, mechanical ventilation, and corticosteroids to altered lung development, and later lung disease following preterm birth will be discussed in more detail in the following sections.
Factors that are likely to contribute to respiratory distress syndrome (RDS) in infants who are born preterm, and factors that may lead to the subsequent development of chronic lung disease (CLD) [93]
Hyperoxia
Preterm infants are often administered hyperoxic gas to increase blood oxygen levels. Oxygen is added to inhaled gas to reach a target arterial oxygen saturation (SaO2) of 85–99 %, with the aim of preventing tissue hypoxia. In the past, the use of high oxygen concentrations (up to 100 % O2) was common, although concentrations were reduced once it became apparent that high concentrations could lead to tissue injury [106–108]. Currently, preterm babies are normally resuscitated using room air (21 % O2) and then the fraction of inspired oxygen (FiO2) is increased until adequate blood oxygenation levels are achieved [109]. However, as fetuses normally have an arterial partial pressure of oxygen (PaO2) of approximately 25–30 mmHg and a SaO2 of 70–80 %, exposure of an immature lung with an immature antioxidant system to even low supplemental oxygen concentrations may be detrimental. It is difficult to determine the contribution of neonatal hyperoxia alone to the long-term pulmonary outcomes of preterm birth and BPD. However, oxygen supplementation during the neonatal period in infants born very preterm has been identified as an independent risk factor for asthma in childhood [101]; further, prolonged oxygen therapy is associated with low FEV1 and the use of inhaled bronchodilators in very-low-birth-weight (preterm) children [96], suggesting an impact on the conducting airways .
Numerous experimental studies have shown that neonatal inhalation of hyperoxic gas can persistently alter the development of alveoli , conducting airways and pulmonary vasculature. Specifically, neonatal hyperoxia leads to fewer and larger alveoli (reviewed in [110]), altered alveolar epithelial cell differentiation [111], altered cellular composition of the bronchiolar epithelium [112–114], altered surfactant composition [111, 115], increased airway smooth muscle [116–119], decreased bronchiolar collagen [112], decreased pulmonary capillary density [120], and remodelling of the lung extracellular matrix [111, 121] and small pulmonary arteries [120]. Consequently, neonatal hyperoxia is associated with later reductions in lung function , including altered airway resistance and reactivity, dynamic lung compliance, tissue elastance and tissue damping, and pulmonary hypertension, as evidenced by right ventricular hypertrophy [111, 112, 120, 122]. Importantly, some of the effects of neonatal hyperoxia on the lung are long-lasting, with alterations in lung structure and function observed in early and advanced adulthood [111, 112, 118, 122–125]. Interestingly, recent studies and reviews have indicated that the oxygen concentration used and the duration of oxygen exposure are important determinants of lung injury [110, 111, 119].
Lung injury and alterations to lung structure that are induced by neonatal exposure to hyperoxia are thought to be due to oxidative stress and a subsequent pro-inflammatory response (see Fig. 5.3) (reviewed in [110]). Experimentally, neonatal treatments aimed at minimizing ROS and exploiting antioxidant defense, as well as treatments that target inflammatory cell influx into the lung and reduce the degree of lung inflammation , have shown promise in improving lung structure following neonatal hyperoxia [110]. Due to the importance of pulmonary vascular development in regulating alveolarization , treatments promoting angiogenesis following neonatal hyperoxia have also received much attention [110]. Recently, the use of stem/progenitor cells to repair or prevent lung injury has also been trialled in experimental models, and clinical trials for their use in preterm infants are currently underway, as discussed below.
Potential pathways whereby exposure of the developing lung to hyperoxia leads to alterations in the structure and function of the airways, pulmonary vasculature and alveoli , thus contributing to impaired lung function later in life. ASM airway smooth muscle , VEGF vascular endothelial growth factor [110]
Mechanical Ventilation
Owing to lung immaturity, preterm infants usually require assisted ventilation. Different ventilation strategies are employed depending on the requirement of the infant; these range from noninvasive strategies, including those involving constant airway pressure (i.e., CPAP using a constant or variable flow, or nasal cannula with low or high flow) or variable airway pressure (i.e., nIPPV, nasal positive airway pressure or nasal high-frequency ventilation), to invasive positive pressure ventilation requiring endotracheal intubation, including patient-triggered ventilation, volume target ventilation, and high-frequency oscillation [126]. It is now recognized that invasive ventilation strategies can increase the incidence of BPD and lung injury, which can increase the risk for persistent lung disease in infants born preterm. Furthermore, intubation is associated with trauma at the site of intubation (i.e., larynx and upper trachea ), as well as bacterial colonization, and sepsis [126]. Noninvasive ventilation strategies have been shown to reduce these adverse effects associated with mechanical ventilation, improve gas exchange, and reduce the incidence of BPD [127]. Thus, not only are noninvasive ventilation strategies now used for weaning infants off mechanical ventilation to reduce extubation failure, they are increasingly being used as primary methods of respiratory support for preterm infants with respiratory insufficiency. However, even noninvasive techniques have been associated with adverse effects, including pneumothorax, pulmonary over-inflation, increased work of breathing, decreased cardiac output, as well as nasal complications and gastrointestinal distension and perforation [126]. Large randomized controlled trials are required to determine the beneficial effects of different modes of noninvasive ventilation on long-term respiratory and non-respiratory outcomes.
Ventilator-induced lung injury is believed to be a result of either volutrauma or barotrauma, which is injury related to over-expansion of the lungs (due to high volumes or pressures), atelectrauma, which is injury caused by repeated opening of collapsed alveoli , or biotrauma, which is injury caused by up-regulation of pro-inflammatory responses to mechanical ventilation, or a combination of these factors [126]. In the clinical setting, it is difficult to determine the precise cause of lung injury in preterm infants due to the multifactorial and individualized nature of their care, both before and after birth. Experimentally, it has been shown in rodents and sheep that even brief mechanical ventilation (2–24 h) of the developing lung alone (i.e., without other confounding factors) leads to impaired formation of alveoli and lung microvessels, as indicated by decreases in secondary crest density, alveolar number, and CD31 staining, and increases in airspace and alveolar size; these changes in lung structure are likely due to disruption of elastin deposition in the lung [92, 128–130] and altered expression of other genes/proteins that regulate alveolar formation and angiogenesis, including platelet derived growth factor-A (PDGF-A), tenascin-C, and vascular endothelial growth factor receptor 2 (VEGF-R2) (see Fig. 5.4) [92, 131]. Studies have also identified numerous transcription factors and genes that are dysregulated by mechanical ventilation, contributing to lung injury and altered lung development ; likely factors include those involved in TGF-β signalling, of which elastin is a target, mechanotransduction and the immune response [132–134]. Furthermore, recent studies have shown that invasive mechanical ventilation in preterm lambs causes an increase in histone deacetylatase (HDAC) 1 and subsequent genome-wide histone hypoacetylation in the lung, indicating alterations in the epigenetic regulation of gene expression [135]. Insulin-like growth factor-1 (IGF-1) is one gene whose expression is increased in mechanically ventilated preterm lambs and in infants who have died from RDS or BPD, and which appears to be epigenetically modified by mechanical ventilation [135]. Further studies are required to identify other genes that are modified by mechanical ventilation and the specific pathways leading to their alteration [93]. A greater understanding of the mechanisms by which mechanical ventilation alters gene expression and leads to altered lung development will enable targeted interventions to be developed. In this regard, histone acetylation is increased and alveolar formation is improved when preterm lambs that are mechanically ventilated are treated with HDAC inhibitors [135].
Potential pathways whereby prolonged cyclic stretch of the lung caused by positive-pressure mechanical ventilation alters the formation of the pulmonary vasculature and alveoli . TGF-β transforming growth factor-β, VEGF-R2 vascular endothelial growth factor receptor 2 [92]
In addition to altering alveolar structure, a short period (2–12 h) of positive pressure ventilation of the immature (prenatal) sheep lung induces injury and remodelling of the small conducting airways (i.e., bronchioles), including epithelial cell detachment, alterations in the areas of epithelium and collagen in intact airways, and an increase in airway smooth muscle [129, 130, 136]. There is evidence that airway remodelling persists in the absence of further ventilation, with changes in epithelial area, epithelial cell proliferation and apoptosis, airway smooth muscle and collagen, and a decrease in the number of bronchiolar-alveolar attachments [129, 130, 136]. There is also evidence, however, that the fetal lung is capable of spontaneous repair following ventilation-induced injury, with normalization of alveolar structure in the absence of continued ventilation [129, 130], although this recovery could potentially be restricted to the fetal lung. Changes in alveolarization and airway wall structure following mechanical ventilation could contribute to the increased risk of poor lung function and obstructive lung disease reported in individuals who were born preterm. Indeed, airway reactivity is increased 48 h after brief mechanical ventilation of rat pups [137]. Further studies investigating the long-term effects of mechanical ventilation of the immature lung on respiratory health are needed.
Corticosteroids
Corticosteroids have been used in preterm infants with evolving or established BPD to reduce lung inflammation and improve gas exchange; corticosteroids are used to reduce the need for oxygen supplementation [91] and to decrease the risk of retinopathy of prematurity and death [138]. Postnatal corticosteroids, predominantly dexamethasone, are generally administered systemically and can be given early (≤7 days after birth) or late (>7 days). Although systemic corticosteroid therapy can have short-term benefits, it has been linked to adverse long-term outcomes, including detrimental effects on lung structure, neurodevelopment, cardiovascular function, and the gastrointestinal system [91, 138]. Although many of these effects have been linked to the prolonged use of high-dose corticosteroids (especially dexamethasone), studies have indicated that even low-dose corticosteroids may have adverse effects [91]. Inhaled corticosteroids have been trialled in preterm infants with the aim of reducing the side effects associated with systemic corticosteroid use, although these have had limited success [91, 138]. Studies investigating the optimal timing, dose, and type of corticosteroids given to preterm infants to improve short-term outcomes and reduce adverse long-term outcomes are warranted.
Nutrition
Adequate nutrition plays a key role in normal lung development . Experimentally, reduced nutrition during lung development before and after birth can affect alveolarization and bronchiolar structure [28, 31, 139]. The effects of undernutrition during fetal life on lung development have been described above. Lung development continues after birth and it has been shown in sheep that, at maturity, animals that experienced postnatal growth restriction have a smaller lung volume and a smaller pulmonary surface area, relative to lung and body size, than animals that grow normally [140]. Undernutrition during the early postnatal period has been shown to disrupt lung development, especially when combined with exposure to hyperoxia [112, 141, 142]. This is of particular importance in the context of very preterm infants who form most of their alveoli after birth. In addition, very preterm infants have a high risk of developing BPD, and it is known that malnutrition represents one of the many factors contributing to BPD [143]. Maintaining a careful balance of nutritional support for very and extremely preterm infants represents a considerable challenge. Interventions that are necessary to support the survival of these preterm infants, combined with developmental complications they may encounter (e.g., prolonged orotracheal intubation, medications that alter gastrointestinal motility, altered palatal groove formation, irritability due to neurological status, gastrointestinal immaturity) make feeding by mouth a problematic task; therefore, nutritional support is often provided via parenteral or enteral routes [144]. However, most extremely preterm infants receive minimal enteral feeding during the first two weeks of life, due to the abovementioned problems, and their nutritional requirements need to be supplemented with parenteral feeding. Recently, it was reported that a critical minimal amount of enteral feeding is required to prevent the development of BPD [145]. Preterm infants who developed BPD received less enteral feeding during the first two weeks of life, even though it was compensated by parenteral nutrition [145]. Furthermore, it has been reported that, compared to preterm infants without BPD, those with BPD have a greater energy expenditure (up to 25 % above total caloric needs), likely due to their increased work of breathing, stress, inflammation , medications, and the perceived need for catch-up growth [143, 144].
Another aspect of nutritional management of preterm infants that has been associated with an increased risk for BPD is fluid intake. It has been reported that high fluid intake during the first days after birth may increase the risk of developing BPD, through the persistence of a patent ductus arteriosus and a higher fluid content in the pulmonary interstitial tissue; these complications could contribute to decreased lung compliance and increase the need for respiratory support [143, 146]. Overall, it is suggested that adequate nutritional management of preterm infants with evolving or established BPD can be maintained by adopting enteral nutrition as soon as possible, avoiding excess fluid administration during the first days after birth, and progressively increasing energy intake [147].
The use of specific micronutrients, such as vitamins A, C, and E, inositol, iron, selenium, glutamine, cysteine, and methionine, may be beneficial in the nutritional management of preterm infants to prevent or lessen the severity of BPD (reviewed in [144]). Indeed, optimal feeding and growth in preterm infants remains a challenge, and overnutrition of the preterm infant to achieve catch-up growth may actually be detrimental to long-term health [148]. Although accelerated growth of the preterm infant has beneficial effects on neurodevelopment, it has also been associated with many metabolic and cardiovascular implications that present in childhood and adulthood [148]. Further research is needed in order to identify the optimal method of managing preterm nutrition with the goal of enhancing lung growth, preventing BPD, and avoiding long-term, potentially harmful programming effects of catch-up growth.
Although infants born preterm are faced with a vulnerable period during which nutrition can greatly affect their lung development and subsequent lung health, nutritional status can also impact upon the mature lung. Emphysema is commonly observed in patients with anorexia nervosa and was also noted at autopsies of humans who died from starvation [149]. These findings support the concept that lung structure/function is dependent upon nutritional status.
Respiratory Infections
The link between early-life respiratory infection s, particularly viral infections, and subsequent increased risk of recurrent wheezing and asthma is being increasingly investigated. Viral respiratory infections during infancy have been shown to contribute to the development of wheezing in childhood. In particular, the occurrence of symptomatic rhinovirus infection during infancy (<12 months of age) has been identified as the most significant risk factor for the development of wheezing 1 year after infection [150] and into early childhood [151]. Infection with respiratory syncytial virus (RSV) in infancy has also been linked with wheezing in pre-school children, albeit with a weaker relationship compared to rhinovirus infections [151]. A later study confirmed these findings, showing that bronchiolitis caused by viruses other than RSV was associated with a greater risk of recurrent wheezing (within the first 2 and 3 years after hospitalization for bronchiolitis) compared to RSV-associated wheezing [152]. Not only has rhinovirus infection been associated with recurrent wheezing, but rhinovirus infection during the first 3 years of life is also significantly associated with reduced lung function (lower FEV1, percent predicted FEV1, FEV0.5, FEF25–75 %, FEV1/forced vital capacity (FVC), and FEV0.5/FVC) at 8 years of age compared to children with non-wheezing rhinovirus illnesses and no rhinovirus illnesses [153]. Furthermore, this study showed that children with RSV-induced wheezing did not have significant alterations in any of the lung function measurements, indicating that infections in early childhood caused by rhinovirus, rather than RSV, are significant predictors of decreased lung function in childhood [153]. Not only are viral respiratory infections during early postnatal life associated with subsequent wheezing, but they often precede the development of childhood asthma. In a cohort of children that was followed up to 6 years of age after specific viral respiratory wheezing illnesses in early childhood (within the first 3 years of life), rhinovirus-associated wheezing illnesses were the most significant predictors of the subsequent development of asthma [154]. This, however, does not exclude RSV as a potential risk factor for asthma. Recent studies showed that severe RSV-induced bronchiolitis during infancy (<12 months of age) is associated with an increased risk of asthma at 6 and 7 years of age [155, 156]. Furthermore, RSV infection in infancy was shown to be associated with a 3.2-fold increase in the risk of wheezing and impaired lung function (lower FEV1, FVC, and FEV1/FVC, and increased respiratory system resistance) at age 6 [155]. Another recent study reported on a study population at 15–18 years of age, and compared patients hospitalized for bronchiolitis in infancy (<24 months of age) caused by RSV or rhinovirus to a control population [157]. This study showed that patients hospitalized in infancy for bronchiolitis had an increased risk of asthma in adolescence; of the bronchiolitis patients, those who had rhinovirus infection had a higher risk of self-reported asthma, but not doctor-diagnosed asthma, than those who had RSV infection [157]. Studies that have extended further into adulthood have found that hospitalization for bronchiolitis in infancy (<24 months of age) is associated with an increased risk of asthma, and an increased use of asthma medication at 28–31 years [158]. In that study, doctor-diagnosed asthma was present in adulthood in one-third of former bronchiolitis patients, whereas asthma was present in only 10 % of controls. An earlier study of the same cohort demonstrated that the prevalence of asthma at 28–31 years was similar to that recorded 10 years earlier at 18–21 years of age [159], indicating the early-onset and persistence of long-term sequelae following early-life respiratory infection. However, these studies did not differentiate between RSV and rhinovirus bronchiolitis. The link between early-life respiratory infections and subsequent long-term respiratory morbidity has also been highlighted in a follow-up study that showed an association between RSV bronchiolitis in the first year of life and increased prevalence of asthma or recurrent wheeze at 18 years of age; 39 % of RSV subjects vs. 9 % of control subjects [160]. A reduction in spirometric airway function was reported in the RSV cohort compared to controls, regardless of whether the RSV subjects presented with or without asthma or recurrent wheeze [160].
Bacterial colonization of the hypopharynx with S. pneumonia, H. influenza, or M. catarrhalis in healthy neonates at 4 weeks of age has been associated with an increased risk of pneumonia and bronchiolitis in early life (up to 3 years) independent of asthma [161]; this suggests that an altered microbiome of the neonatal airway may affect the maturation of the immune system, leading to an increased risk of pneumonia and bronchiolitis. Furthermore, radiologically confirmed pneumonia in the first 2 years of life has been associated with asthma or asthma-like symptoms in pre-school children (4–5 years of age) [162]. This is confirmed in a meta-analysis that showed an overall risk of major long-term respiratory sequelae following childhood pneumonia (before 5 years of age) in non-hospitalized children of 5.5 %; this risk was almost three times greater in children who had been hospitalized, with adenovirus pneumonia having the highest risk (approximately 55 %) [163]. Major respiratory sequelae were defined as restrictive lung disease, obstructive lung disease, and/or bronchiectasis, and the most common type of major outcome reported was restrictive lung disease [163]. These findings indicate that children diagnosed with pneumonia should be considered at risk of long-term respiratory sequelae, even after acute symptoms have resolved.
Environmental Exposures
Many studies have highlighted the deleterious effects of a suboptimal environment on lung development (reviewed in [164]). Harmful environmental exposures include both outdoor pollutants (e.g., industrial and motor vehicle emissions) and indoor pollutants (e.g., environmental tobacco smoke (ETS) and fungal spores). In particular, ETS exposure in early life has been identified as a significant risk factor for poor pulmonary outcome in later life. A recent meta-analysis indicated that passive exposure to ETS (in both pre- and postnatal life) increases the incidence of wheeze and asthma in children and adolescents by at least 20 % [165]. Maternal smoking was associated with a 52 % increase in the risk of wheeze and a 20 % increase in the risk of asthma at 5–18 years of age [165]. Not only has ETS exposure during infancy been associated with increased risk of wheeze and asthma, but a meta-analysis has also shown it to increase the risk of lower respiratory infection s, particularly bronchiolitis [166]. All types of passive ETS exposure (pre- and postnatal maternal, paternal, both parents, any household member smoking), but particularly maternal smoking, led to a significant increase in the risk of infants developing lower respiratory infections in the first 2 years of life [166]. This meta-analysis highlights the importance of limiting ETS exposure during both gestation and childhood to prevent the onset of wheezing, asthma, and respiratory infections. In contrast to the large number of reports on effects of ETS exposure, there are few reports on the effects of exposure to traffic-related air pollution during infancy and childhood and its effects on later respiratory morbidity. A recent systematic review and meta-analysis showed that early childhood exposure to traffic-related air pollution was associated with the development of asthma throughout childhood up to 12 years of age, with the magnitude of risk increasing with age [167]. However, further studies are needed to investigate the relation between effects of exposure at specific ages and adult outcomes.
Potential Targets for Cell-Based Therapies: Neonatal Treatments for Preterm Infants
The use of cell-based therapies to prevent or repair neonatal lung injury in very preterm infants has now evolved from experimental studies using animal models to phase I and II clinical trials in very preterm infants at risk of BPD. The rationale for the current clinical trials is based on knowledge gained from studies examining the perturbation of pulmonary stem cells in neonatal lung injury, as well as experimental studies in animal models that found stem cell-based therapies to be beneficial. Studies in animal models and in humans have shown that altered function or depletion of resident stem cells within the developing lung likely contributes to the pathogenesis of BPD. In rodent models of BPD, in which newborn pups are exposed to high concentrations of oxygen, lung epithelial cells, mesenchymal stromal cells (MSCs), and endothelial progenitor cells (EPCs) were shown to be significantly reduced in number and also displayed altered differentiation potential [123, 168–170]. Similar perturbations to resident stem cells have been reported in tracheal aspirates and umbilical cord blood samples taken from preterm infants [171–175]. The presence of MSCs in tracheal aspirates has been reported as an indicator of BPD development , and these MSCs were shown to demonstrate a lung-specific gene expression and to secrete pro-inflammatory cytokines; this suggests that the MSCs in the tracheal aspirates originated from the lungs and have the capacity to contribute to lung injury [171, 172]. In samples of umbilical cord blood from preterm infants, endothelial colony forming cells (ECFCs; a subset of EPCs) were shown to have an increased susceptibility to in vitro exposure to hyperoxic gas, and were found in reduced numbers among BPD infants compared to non-BPD preterm infants [173–175]. This suggests that a reduction in the levels of angiogenic progenitor cells may contribute to disrupted vasculogenic mechanisms during lung development and lead to impaired vascular and alveolar growth. In response to the findings of stem cell perturbation in BPD, experimental studies have focused on their therapeutic replacement with exogenous-derived stem cells (reviewed in [176]). One of the most commonly researched cell types is the MSC, which can be easily sourced from bone marrow, adipose tissue, muscle, peripheral blood, umbilical cord blood, umbilical cord Wharton’s jelly, and placenta. Numerous components of neonatal lung injury have been attenuated by the administration of MSCs, including inflammation , vascular damage, impaired alveolarization , and fibrosis [170, 177–188]. Furthermore, MSCs have been reported to have low engraftment and differentiation into the lung, suggesting a paracrine-mediated therapeutic effect. This has been supported by studies showing similar protective properties of cell-free conditioned media from MSCs in experimental models of neonatal lung injury. Other cell types (ECFCs, angiogenic cells, and amnion epithelial cells) have also been examined in experimental studies and have shown promise in their therapeutic benefit for neonatal lung injury [189–194].
The overall aim of experimental studies on neonatal lung regeneration has been to generate evidence to create a rationale for translating experimental findings into the clinic. Encouragingly, there has recently been translation of findings from animal studies into clinical trials in preterm infants, with the results of the first phase I clinical trial (NCT01297205) of stem cell therapy in preterm infants being published [195]. The authors assessed the safety and feasibility of human umbilical cord blood-derived MSCs, administered to nine very preterm infants who were at risk for developing BPD. The infants were born at 24–26 weeks of gestation (630–1030 g) and needed continuous ventilatory support that could not be decreased due to significant respiratory distress prior to enrolment into the study. Three patients were given a low dose of MSCs (1 × 107 cells/kg) via the trachea , and six were given a high dose (2 × 107 cells/kg). Treatment-related serious adverse effects were assessed for 84 days following transplantation of the MSCs. The authors reported no serious adverse effects or dose-limiting toxicity associated with transplantation of the MSCs. There was no significant difference in serious adverse effects between the low-dose and high-dose MSC group or between the MSC group and a matched-comparison group, except in BPD severity; the MSC group had significantly lower BPD severity than the matched-comparison group. Furthermore, the inflammatory markers matrix metalloproteinase (MMP)-9, interleukin (IL)-6, IL-8, tumor necrosis factor (TNF)-α, and TGF-β were reduced in tracheal aspirate fluid after MSC transplantation. The findings of this study are important in establishing the feasibility, safety, and efficacy of MSC treatment for BPD in preterm infants. Follow-up studies are currently underway to assess the long-term safety of MSC transplantation in the same preterm infants reported in the phase I clinical trial, at approximately 2 years of age (NCT01632475) and 5 years of age (NCT02023788). Furthermore, a randomized, double-blind, multicenter, phase II clinical trial (NCT0182957) has commenced that will investigate the efficacy and safety of a low dose of MSCs versus a control group for the treatment of BPD in preterm infants; this trial will be followed up with another study (NCT01897987) investigating the long-term safety and efficacy of the phase II trial. It is anticipated that findings from these current clinical trials will pave the way for the future treatment of neonatal lung diseases using stem cell-based therapies.
Conclusions
The lung develops during prenatal and early postnatal life, and is therefore vulnerable to adverse environmental conditions during the embryonic, fetal, and postnatal stages of lung development . The peripheral lung, including alveoli , the pulmonary microcirculation, and small conducting airways , is especially vulnerable to injury or altered development . Owing to the lung’s limited capacity for repair , lung injury, or altered lung development caused by a suboptimal fetal or early postnatal environment can persist into later life, adversely affecting adult lung function and increasing the risk of respiratory illness. Changes in lung structure can be due to a direct developmental effect or to changes in gene and protein expression caused by alterations in the nutritional, physical/mechanical, oxidative, or immunological environments. In addition, a range of environmental factors may induce epigenetic alterations in key genes that regulate lung development to cause persistent alterations in lung structure and/or function. Although there are currently no treatments capable of repairing the injured lung, there is now encouraging experimental evidence on the effectiveness of cell-based therapies to prevent or repair injury to the immature lungs, with clinical trials now underway in infants who are born very preterm and are at risk of developing chronic lung disease.
References
Martinez FD (2009) The origins of asthma and chronic obstructive pulmonary disease in early life. Proc Am Thorac Soc 6:272–277
Barker DJ, Thornburg KL (2013) Placental programming of chronic diseases, cancer and lifespan: a review. Placenta 34:841–845
Vickers MH (2014) Early life nutrition, epigenetics and programming of later life disease. Nutrients 6:2165–2178
Narang I, Bush A (2012) Early origins of chronic obstructive pulmonary disease. Semin Fetal Neonatal Med 17:112–118
Pallotto EK, Kilbride HW (2006) Perinatal outcome and later implications of intrauterine growth restriction. Clin Obstet Gynecol 49:257–269
Barker DJ, Godfrey KM, Fall C, Osmond C, Winter PD, Shaheen SO (1991) Relation of birth weight and childhood respiratory infection to adult lung function and death from chronic obstructive airways disease. BMJ 303:671–675
Lawlor DA, Ebrahim S, Davey Smith G (2005) Association of birth weight with adult lung function: findings from the British Women’s Heart and Health Study and a meta-analysis. Thorax 60:851–858
Cetin I, Alvino G (2009) Intrauterine growth restriction: implications for placental metabolism and transport. A review. Placenta 30:S77–S82
Pecks U, Brieger M, Schiessl B, Bauerschlag DO, Piroth D, Bruno B et al (2012) Maternal and fetal cord blood lipids in intrauterine growth restriction. J Perinat Med 40:287–296
Briana DD, Malamitsi-Puchner A (2013) Small for gestational age birth weight: impact on lung structure and function. Paediatr Respir Rev 14:256–262
Dezateux C, Lum S, Hoo AF, Hawdon J, Costeloe K, Stocks J (2004) Low birth weight for gestation and airway function in infancy: exploring the fetal origins hypothesis. Thorax 59:60–66
Minior VK, Divon MY (1998) Fetal growth restriction at term: myth or reality? Obstet Gynecol 92:57–60
Tyson JE, Kennedy K, Broyles S, Rosenfeld CR (1995) The small for gestational age infant: accelerated or delayed pulmonary maturation? Increased or decreased survival? Pediatrics 95:534–538
Kotecha SJ, Watkins WJ, Heron J, Henderson J, Dunstan FD, Kotecha S (2010) Spirometric lung function in school-age children: effect of intrauterine growth retardation and catch-up growth. Am J Respir Crit Care Med 181:969–974
Rona RJ, Gulliford MC, Chinn S (1993) Effects of prematurity and intrauterine growth on respiratory health and lung function in childhood. BMJ 306:817–820
Wjst M, Popescu M, Trepka MJ, Heinrich J, Wichmann HE (1998) Pulmonary function in children with initial low birth weight. Pediatr Allergy Immunol 9:80–90
Morsing E, Gustafsson P, Brodszki J (2012) Lung function in children born after foetal growth restriction and very preterm birth. Acta Paediatr 101:48–54
Nikolajev K, Heinonen K, Hakulinen A, Lansimies E (1998) Effects of intrauterine growth retardation and prematurity on spirometric flow values and lung volumes at school age in twin pairs. Pediatr Pulmonol 25:367–370
Nikolajev K, Koskela H, Korppi M (2008) Birth weight and adult lung function: a within-pair analysis of twins followed up from birth. World J Pediatr 4:222–226
Edwards CA, Osman LM, Godden DJ, Campbell DM, Douglas JG (2003) Relationship between birth weight and adult lung function: controlling for maternal factors. Thorax 58:1061–1065
Boezen HM, Vonk JM, van Aalderen WM, Brand PL, Gerritsen J, Schouten JP et al (2002) Perinatal predictors of respiratory symptoms and lung function at a young adult age. Eur Respir J 20:383–390
Canoy D, Pekkanen J, Elliott P, Pouta A, Laitinen J, Hartikainen AL et al (2007) Early growth and adult respiratory function in men and women followed from the fetal period to adulthood. Thorax 62:396–402
Narang I, Rosenthal M, Cremonesini D, Silverman M, Bush A (2008) Longitudinal evaluation of airway function 21 years after preterm birth. Am J Respir Crit Care Med 178:74–80
Rona RJ, Smeeton NC, Bustos P, Amigo H, Diaz PV (2005) The early origins hypothesis with an emphasis on growth rate in the first year of life and asthma: a prospective study in Chile. Thorax 60:549–554
Bush A (2008) COPD: a pediatric disease. COPD 5:53–67
Stocks J, Hislop A, Sonnappa S (2013) Early lung development: lifelong effect on respiratory health and disease. Lancet Respir Med 1:728–742
Alcorn DG, Adamson TM, Maloney JE, Robinson PM (1981) A morphologic and morphometric analysis of fetal lung development in the sheep. Anat Rec 201:655–667
Maritz GS, Cock ML, Louey S, Joyce BJ, Albuquerque CA, Harding R (2001) Effects of fetal growth restriction on lung development before and after birth: a morphometric analysis. Pediatr Pulmonol 32:201–210
Maritz GS, Cock ML, Louey S, Suzuki K, Harding R (2004) Fetal growth restriction has long-term effects on postnatal lung structure in sheep. Pediatr Res 55:287–295
Joyce BJ, Louey S, Davey MG, Cock ML, Hooper SB, Harding R (2001) Compromised respiratory function in postnatal lambs after placental insufficiency and intrauterine growth restriction. Pediatr Res 50:641–649
Das RM (1984) The effects of intermittent starvation on lung development in suckling rats. Am J Pathol 117:326–332
Joss-Moore LA, Wang Y, Yu X, Campbell MS, Callaway CW, McKnight RA et al (2011) IUGR decreases elastin mRNA expression in the developing rat lung and alters elastin content and lung compliance in the mature rat lung. Physiol Genomics 43:499–505
Lechner AJ (1985) Perinatal age determines the severity of retarded lung development induced by starvation. Am Rev Respir Dis 131:638–643
Rehan VK, Sakurai R, Li Y, Karadag A, Corral J, Bellusci S et al (2012) Effects of maternal food restriction on offspring lung extracellular matrix deposition and long term pulmonary function in an experimental rat model. Pediatr Pulmonol 47:162–171
Joss-Moore L, Carroll T, Yang Y, Fitzhugh M, Metcalfe D, Oman J et al (2013) Intrauterine growth restriction transiently delays alveolar formation and disrupts retinoic acid receptor expression in the lung of female rat pups. Pediatr Res 73:612–620
Wignarajah D, Cock ML, Pinkerton KE, Harding R (2002) Influence of intrauterine growth restriction on airway development in fetal and postnatal sheep. Pediatr Res 51:681–688
Thorn SR, Rozance PJ, Brown LD, Hay WW Jr (2011) The intrauterine growth restriction phenotype: fetal adaptations and potential implications for later life insulin resistance and diabetes. Semin Reprod Med 29:225–236
Joss-Moore LA, Albertine KH, Lane RH (2011) Epigenetics and the developmental origins of lung disease. Mol Genet Metab 104:61–66
Harding R, Hooper SB (1996) Regulation of lung expansion and lung growth before birth. J Appl Physiol 81:209–224
Chien LN, Chiou HY, Wang CW, Yeh TF, Chen CM (2014) Oligohydramnios increases the risk of respiratory hospitalization in childhood: a population-based study. Pediatr Res 75:576–581
Albuquerque CA, Smith KR, Saywers TE, Johnson C, Cock ML, Harding R (2002) Relation between oligohydramnios and spinal flexion in the human fetus. Early Hum Dev 68:119–126
Harding R, Hooper SB, Dickson KA (1990) A mechanism leading to reduced lung expansion and lung hypoplasia in fetal sheep during oligohydramnios. Am J Obstet Gynecol 163:1904–1913
Harding R, Liggins GC (1991) The influence of oligohydramnios on thoracic dimensions of fetal sheep. J Dev Physiol 16:355–361
Nicksa GA, Yu DC, Kalish BT, Klein JD, Turner CG, Zurakowski D et al (2011) Serial amnioinfusions prevent fetal pulmonary hypoplasia in a large animal model of oligohydramnios. J Pediatr Surg 46:67–71
Hofmeyr GJ, Eke AC, Lawrie TA (2014) Amnioinfusion for third trimester preterm premature rupture of membranes. Cochrane Database Syst Rev 3, CD000942
Panitch HB, Weiner DJ, Feng R, Perez MR, Healy F, McDonough JM et al (2014) Lung function over the first 3 years of life in children with congenital diaphragmatic hernia. Pediatr Pulmonol doi:10.1002/ppul.23082
Pierro M, Thebaud B (2014) Understanding and treating pulmonary hypertension in congenital diaphragmatic hernia. Semin Fetal Neonatal Med 19:357–363
Nardo L, Maritz G, Harding R, Hooper SB (2000) Changes in lung structure and cellular division induced by tracheal obstruction in fetal sheep. Exp Lung Res 26:105–119
Deprest J, Brady P, Nicolaides K, Benachi A, Berg C, Vermeesch J et al (2014) Prenatal management of the fetus with isolated congenital diaphragmatic hernia in the era of the TOTAL trial. Semin Fetal Neonatal Med 19:338–348
Manoli SE, Smith LA, Vyhlidal CA, An CH, Porrata Y, Cardoso WV et al (2012) Maternal smoking and the retinoid pathway in the developing lung. Respir Res 13:42
Stocks J, Dezateux C (2003) The effect of parental smoking on lung function and development during infancy. Respirology 8:266–285
Stocks J, Sonnappa S (2013) Early life influences on the development of chronic obstructive pulmonary disease. Ther Adv Respir Dis 7:161–173
Maritz GS, Harding R (2011) Life-long programming implications of exposure to tobacco smoking and nicotine before and soon after birth: evidence for altered lung development. Int J Environ Res Public Health 8:875–898
Maritz GS (2013) Perinatal exposure to nicotine and implications for subsequent obstructive lung disease. Paediatr Respir Rev 14:3–8
Dasgupta C, Xiao D, Xu Z, Yang S, Zhang L (2012) Developmental nicotine exposure results in programming of alveolar simplification and interstitial pulmonary fibrosis in adult male rats. Reprod Toxicol 34:370–377
Suter MA, Anders AM, Aagaard KM (2013) Maternal smoking as a model for environmental epigenetic changes affecting birthweight and fetal programming. Mol Hum Reprod 19:1–6
Ethen MK, Ramadhani TA, Scheuerle AE, Canfield MA, Wyszynski DF, Druschel CM et al (2009) Alcohol consumption by women before and during pregnancy. Matern Child Health J 13:274–285
Kenna K, Sozo F, De Matteo R, Hanita T, Gray SP, Tare M et al (2012) Alcohol exposure during late gestation: multiple developmental outcomes in sheep. J Dev Org Health Dis 3:224–236
May PA, Gossage JP, Kalberg WO, Robinson LK, Buckley D, Manning M et al (2009) Prevalence and epidemiologic characteristics of FASD from various research methods with an emphasis on recent in-school studies. Dev Disabil Res Rev 15:176–192
Public, editor (2005) Fetal alcohol spectrum disorder (FASD): a framework for action, 2003: National Library of Canada Cataloguing in Publication
Baumann P, Schild C, Hume RF, Sokol RJ (2006) Alcohol abuse—a persistent preventable risk for congenital anomalies. Int J Gynaecol Obstet 95:66–72
Johnson S, Knight R, Marmer DJ, Steele RW (1981) Immune deficiency in fetal alcohol syndrome. Pediatr Res 15:908–911
Inselman LS, Fisher SE, Spencer H, Atkinson M (1985) Effect of intrauterine ethanol exposure on fetal lung growth. Pediatr Res 19:12–14
Wang X, Gomutputra P, Wolgemuth DJ, Baxi L (2007) Effects of acute alcohol intoxication in the second trimester of pregnancy on development of the murine fetal lung. Am J Obstet Gynecol 197(269):e261–e264
Sozo F, O’Day L, Maritz G, Kenna K, Stacy V, Brew N et al (2009) Repeated ethanol exposure during late gestation alters the maturation and innate immune status of the ovine fetal lung. Am J Physiol Lung Cell Mol Physiol 296:L510–L518
Lazic T, Wyatt TA, Matic M, Meyerholz DK, Grubor B, Gallup JM et al (2007) Maternal alcohol ingestion reduces surfactant protein A expression by preterm fetal lung epithelia. Alcohol 41:347–355
Sozo F, Vela M, Stokes V, Kenna K, Meikle PJ, De Matteo R et al (2011) Effects of prenatal ethanol exposure on the lungs of postnatal lambs. Am J Physiol Lung Cell Mol Physiol 300:L139–L147
Gauthier TW, Ping XD, Gabelaia L, Brown LA (2010) Delayed neonatal lung macrophage differentiation in a mouse model of in utero ethanol exposure. Am J Physiol Lung Cell Mol Physiol 299:L8–L16
Gauthier TW, Ping XD, Harris FL, Wong M, Elbahesh H, Brown LA (2005) Fetal alcohol exposure impairs alveolar macrophage function via decreased glutathione availability. Pediatr Res 57:76–81
Gauthier TW, Young PA, Gabelaia L, Tang SM, Ping XD, Harris FL et al (2009) In utero ethanol exposure impairs defenses against experimental group B streptococcus in the term Guinea pig lung. Alcohol Clin Exp Res 33:300–306
Orgeig S, Morrison JL, Sullivan LC, Daniels CB (2015) The development of the pulmonary surfactant system. In: Harding R, Pinkerton KE (eds) The lung: development, aging and the environment, 2nd edn. Elsevier, London, pp 183–209
Tschanz SA, Damke BM, Burri PH (1995) Influence of postnatally administered glucocorticoids on rat lung growth. Biol Neonate 68:229–245
Seckl JR (2004) Prenatal glucocorticoids and long-term programming. Eur J Endocrinol 151(Suppl 3):U49–U62
Yu HR, Kuo HC, Chen CC, Sheen JM, Tiao MM, Chen YC et al (2014) Prenatal dexamethasone exposure in rats results in long-term epigenetic histone modifications and tumour necrosis factor-alpha production decrease. Immunology 143:651–660
Tschanz SA, Haenni B, Burri PH (2002) Glucocorticoid induced impairment of lung structure assessed by digital image analysis. Eur J Pediatr 161:26–30
Luyet C, Burri PH, Schittny JC (2002) Suppression of cell proliferation and programmed cell death by dexamethasone during postnatal lung development. Am J Physiol Lung Cell Mol Physiol 282:L477–L483
Goldenberg RL, Hauth JC, Andrews WW (2000) Intrauterine infection and preterm delivery. N Engl J Med 342:1500–1507
Moss TJ, Nitsos I, Kramer BW, Ikegami M, Newnham JP, Jobe AH (2002) Intra-amniotic endotoxin induces lung maturation by direct effects on the developing respiratory tract in preterm sheep. Am J Obstet Gynecol 187:1059–1065
Watterberg KL, Demers LM, Scott SM, Murphy S (1996) Chorioamnionitis and early lung inflammation in infants in whom bronchopulmonary dysplasia develops. Pediatrics 97:210–215
Galinsky R, Polglase GR, Hooper SB, Black MJ, Moss TJ (2013) The consequences of chorioamnionitis: preterm birth and effects on development. J Pregnancy 2013:412831
Kramer BW, Kallapur SG, Moss TJ, Nitsos I, Polglase GP, Newnham JP et al (2010) Modulation of fetal inflammatory response on exposure to lipopolysaccharide by chorioamnion, lung, or gut in sheep. Am J Obstet Gynecol 202(77):e71–e79
Atik A, Sozo F, Orgeig S, Suri L, Hanita T, Harding R et al (2012) Long-term pulmonary effects of intrauterine exposure to endotoxin following preterm birth in sheep. Reprod Sci 19:1352–1364
Kallapur SG, Nitsos I, Moss TJ, Kramer BW, Newnham JP, Ikegami M et al (2005) Chronic endotoxin exposure does not cause sustained structural abnormalities in the fetal sheep lungs. Am J Physiol Lung Cell Mol Physiol 288:L966–L974
Blencowe H, Cousens S, Oestergaard MZ, Chou D, Moller AB, Narwal R et al (2012) National, regional, and worldwide estimates of preterm birth rates in the year 2010 with time trends since 1990 for selected countries: a systematic analysis and implications. Lancet 379:2162–2172
Hintz SR, Bann CM, Ambalavanan N, Cotten CM, Das A, Higgins RD (2010) Predicting time to hospital discharge for extremely preterm infants. Pediatrics 125:e146–e154
Jensen EA, Schmidt B (2014) Epidemiology of bronchopulmonary dysplasia. Birth Defects Res A Clin Mol Teratol 100:145–157
Coalson JJ (2003) Pathology of new bronchopulmonary dysplasia. Semin Neonatol 8:73–81
Husain AN, Siddiqui NH, Stocker JT (1998) Pathology of arrested acinar development in postsurfactant bronchopulmonary dysplasia. Hum Pathol 29:710–717
Jobe AJ (1999) The new BPD: an arrest of lung development. Pediatr Res 46:641–643
Saugstad OD (2010) Oxygen and oxidative stress in bronchopulmonary dysplasia. J Perinat Med 38:571–577
Kinsella JP, Greenough A, Abman SH (2006) Bronchopulmonary dysplasia. Lancet 367:1421–1431
Mokres LM, Parai K, Hilgendorff A, Ertsey R, Alvira CM, Rabinovitch M et al (2010) Prolonged mechanical ventilation with air induces apoptosis and causes failure of alveolar septation and angiogenesis in lungs of newborn mice. Am J Physiol Lung Cell Mol Physiol 298:L23–L35
Albertine KH, Yoder BA (2015) Pulmonary consequences of preterm birth. In: Harding R, Pinkerton KE (eds) The lung: development, aging and the environment, 2nd edn. Elsevier, London, pp 311–327
Doyle LW (2006) Respiratory function at age 8–9 years in extremely low birthweight/very preterm children born in Victoria in 1991–1992. Pediatr Pulmonol 41:570–576
Doyle LW, Faber B, Callanan C, Freezer N, Ford GW, Davis NM (2006) Bronchopulmonary dysplasia in very low birth weight subjects and lung function in late adolescence. Pediatrics 118:108–113
Korhonen P, Laitinen J, Hyodynmaa E, Tammela O (2004) Respiratory outcome in school-aged, very-low-birth-weight children in the surfactant era. Acta Paediatr 93:316–321
Smith LJ, van Asperen PP, McKay KO, Selvadurai H, Fitzgerald DA (2008) Reduced exercise capacity in children born very preterm. Pediatrics 122:e287–e293
Kilbride HW, Gelatt MC, Sabath RJ (2003) Pulmonary function and exercise capacity for ELBW survivors in preadolescence: effect of neonatal chronic lung disease. J Pediatr 143:488–493
Gross SJ, Iannuzzi DM, Kveselis DA, Anbar RD (1998) Effect of preterm birth on pulmonary function at school age: a prospective controlled study. J Pediatr 133:188–192
Gessner BD, Chimonas MA (2007) Asthma is associated with preterm birth but not with small for gestational age status among a population-based cohort of Medicaid-enrolled children <10 years of age. Thorax 62:231–236
Mai XM, Gaddlin PO, Nilsson L, Finnstrom O, Bjorksten B, Jenmalm MC et al (2003) Asthma, lung function and allergy in 12-year-old children with very low birth weight: a prospective study. Pediatr Allergy Immunol 14:184–192
Brostrom EB, Akre O, Katz-Salamon M, Jaraj D, Kaijser M (2013) Obstructive pulmonary disease in old age among individuals born preterm. Eur J Epidemiol 28:79–85
Stoll BJ, Gordon T, Korones SB, Shankaran S, Tyson JE, Bauer CR et al (1996) Late-onset sepsis in very low birth weight neonates: a report from the National Institute of Child Health and Human Development Neonatal Research Network. J Pediatr 129:63–71
Webber S, Wilkinson AR, Lindsell D, Hope PL, Dobson SR, Isaacs D (1990) Neonatal pneumonia. Arch Dis Child 65:207–211
Doyle LW, Anderson PJ (2009) Long-term outcomes of bronchopulmonary dysplasia. Semin Fetal Neonatal Med 14:391–395
Campbell K (1951) Intensive oxygen therapy as a possible cause of retrolental fibroplasia; a clinical approach. Med J Aust 2:48–50
Northway WH Jr, Rosan RC, Porter DY (1967) Pulmonary disease following respirator therapy of hyaline-membrane disease. Bronchopulmonary dysplasia. N Engl J Med 276:357–368
Ryan H (1952) Retrolental fibroplasia; a clinicopathologic study. Am J Ophthalmol 35:329–342
Richmond S, Wyllie J (2010) European Resuscitation Council Guidelines for Resuscitation 2010 Section 7. Resuscitation of babies at birth. Resuscitation 81:1389–1399
Sozo F, O’Reilly M (2015) The effects of neonatal hyperoxia on lung development. In: Harding R, Pinkerton KE (eds) The lung: development, aging and the environment, 2nd edn. Elsevier, London, pp 329–349
Yee M, Chess PR, McGrath-Morrow SA, Wang Z, Gelein R, Zhou R et al (2009) Neonatal oxygen adversely affects lung function in adult mice without altering surfactant composition or activity. Am J Physiol Lung Cell Mol Physiol 297:L641–L649
O’Reilly M, Hansbro PM, Horvat JC, Beckett EL, Harding R, Sozo F (2014) Bronchiolar remodeling in adult mice following neonatal exposure to hyperoxia: relation to growth. Anat Rec (Hoboken) 297:758–769
Massaro GD, McCoy L, Massaro D (1986) Hyperoxia reversibly suppresses development of bronchiolar epithelium. Am J Physiol 251:R1045–R1050
Massaro GD, McCoy L, Massaro D (1988) Development of bronchiolar epithelium: time course of response to oxygen and recovery. Am J Physiol 254:R755–R760
White CW, Greene KE, Allen CB, Shannon JM (2001) Elevated expression of surfactant proteins in newborn rats during adaptation to hyperoxia. Am J Respir Cell Mol Biol 25:51–59
Belik J, Jankov RP, Pan J, Tanswell AK (2003) Chronic O2 exposure enhances vascular and airway smooth muscle contraction in the newborn but not adult rat. J Appl Physiol 94:2303–2312
Denis D, Fayon MJ, Berger P, Molimard M, De Lara MT, Roux E et al (2001) Prolonged moderate hyperoxia induces hyperresponsiveness and airway inflammation in newborn rats. Pediatr Res 50:515–519
O’Reilly M, Harding R, Sozo F (2014) Altered small airways in aged mice following neonatal exposure to hyperoxic gas. Neonatology 105:39–45
Hartman WR, Smelter DF, Sathish V, Karass M, Kim S, Aravamudan B et al (2012) Oxygen dose responsiveness of human fetal airway smooth muscle cells. Am J Physiol Lung Cell Mol Physiol 303:L711–L719
Ladha F, Bonnet S, Eaton F, Hashimoto K, Korbutt G, Thebaud B (2005) Sildenafil improves alveolar growth and pulmonary hypertension in hyperoxia-induced lung injury. Am J Respir Crit Care Med 172:750–756
Chetty A, Cao GJ, Severgnini M, Simon A, Warburton R, Nielsen HC (2008) Role of matrix metalloprotease-9 in hyperoxic injury in developing lung. Am J Physiol Lung Cell Mol Physiol 295:L584–L592
Dauger S, Ferkdadji L, Saumon G, Vardon G, Peuchmaur M, Gaultier C et al (2003) Neonatal exposure to 65% oxygen durably impairs lung architecture and breathing pattern in adult mice. Chest 123:530–538
Yee M, Vitiello PF, Roper JM, Staversky RJ, Wright TW, McGrath-Morrow SA et al (2006) Type II epithelial cells are critical target for hyperoxia-mediated impairment of postnatal lung development. Am J Physiol Lung Cell Mol Physiol 291:L1101–L1111
Yee M, White RJ, Awad HA, Bates WA, McGrath-Morrow SA, O’Reilly MA (2011) Neonatal hyperoxia causes pulmonary vascular disease and shortens life span in aging mice. Am J Pathol 178:2601–2610
Thibeault DW, Mabry S, Rezaiekhaligh M (1990) Neonatal pulmonary oxygen toxicity in the rat and lung changes with aging. Pediatr Pulmonol 9:96–108
Mahmoud RA, Roehr CC, Schmalisch G (2011) Current methods of non-invasive ventilatory support for neonates. Paediatr Respir Rev 12:196–205
Gupta S, Sinha SK, Donn SM (2009) Ventilatory management and bronchopulmonary dysplasia in preterm infants. Semin Fetal Neonatal Med 14:367–373
Allison BJ, Crossley KJ, Flecknoe SJ, Davis PG, Morley CJ, Harding R et al (2008) Ventilation of the very immature lung in utero induces injury and BPD-like changes in lung structure in fetal sheep. Pediatr Res 64:387–392
Brew N, Hooper SB, Allison BJ, Wallace MJ, Harding R (2011) Injury and repair in the very immature lung following brief mechanical ventilation. Am J Physiol Lung Cell Mol Physiol 301:L917–L926
Brew N, Hooper SB, Zahra V, Wallace M, Harding R (2013) Mechanical ventilation injury and repair in extremely and very preterm lungs. PLoS One 8, e63905
Bland RD, Mokres LM, Ertsey R, Jacobson BE, Jiang S, Rabinovitch M et al (2007) Mechanical ventilation with 40% oxygen reduces pulmonary expression of genes that regulate lung development and impairs alveolar septation in newborn mice. Am J Physiol Lung Cell Mol Physiol 293:L1099–L1110
Kompass KS, Deslee G, Moore C, McCurnin D, Pierce RA (2010) Highly conserved transcriptional responses to mechanical ventilation of the lung. Physiol Genomics 42:384–396
Hillman NH, Moss TJ, Kallapur SG, Bachurski C, Pillow JJ, Polglase GR et al (2007) Brief, large tidal volume ventilation initiates lung injury and a systemic response in fetal sheep. Am J Respir Crit Care Med 176:575–581
Wallace MJ, Probyn ME, Zahra VA, Crossley K, Cole TJ, Davis PG et al (2009) Early biomarkers and potential mediators of ventilation-induced lung injury in very preterm lambs. Respir Res 10:19
Joss-Moore LA, Lane RH, Albertine KH (2015) Epigenetics and the developmental origins of lung disease. In: Harding R, Pinkerton KE (eds) The lung: development, aging and the environment, 2nd edn. Elsevier, London, pp 299–308
O’Reilly M, Hooper SB, Allison BJ, Flecknoe SJ, Snibson K, Harding R et al (2009) Persistent bronchiolar remodeling following brief ventilation of the very immature ovine lung. Am J Physiol Lung Cell Mol Physiol 297:L992–L1001
Iben SC, Haxhiu MA, Farver CF, Miller MJ, Martin RJ (2006) Short-term mechanical ventilation increases airway reactivity in rat pups. Pediatr Res 60:136–140
Schulzke SM, Pillow JJ (2010) The management of evolving bronchopulmonary dysplasia. Paediatr Respir Rev 11:143–148
Massaro GD, McCoy L, Massaro D (1988) Postnatal undernutrition slows development of bronchiolar epithelium in rats. Am J Physiol 255:R521–R526
Maritz G, Probyn M, DeMatteo R, Snibson K, Harding R (2008) Lung parenchyma at maturity is influenced by postnatal growth but not by moderate preterm birth in sheep. Neonatology 93:28–35
Mataloun MM, Leone CR, Mascaretti RS, Dohlnikoff M, Rebello CM (2009) Effect of postnatal malnutrition on hyperoxia-induced newborn lung development. Braz J Med Biol Res 42:606–613
Mataloun MM, Rebello CM, Mascaretti RS, Dohlnikoff M, Leone CR (2006) Pulmonary responses to nutritional restriction and hyperoxia in premature rabbits. J Pediatr (Rio J) 82:179–185
Dani C, Poggi C (2012) Nutrition and bronchopulmonary dysplasia. J Matern Fetal Neonatal Med 25(Suppl 3):37–40
Biniwale MA, Ehrenkranz RA (2006) The role of nutrition in the prevention and management of bronchopulmonary dysplasia. Semin Perinatol 30:200–208
Wemhoner A, Ortner D, Tschirch E, Strasak A, Rudiger M (2011) Nutrition of preterm infants in relation to bronchopulmonary dysplasia. BMC Pulm Med 11:7
Guo MM, Chung CH, Chen FS, Chen CC, Huang HC, Chung MY (2015) Severe bronchopulmonary dysplasia is associated with higher fluid intake in very Low-birth-weight infants: a retrospective study. Am J Perinatol 30:155–162
Moya F (2014) Preterm nutrition and the lung. World Rev Nutr Diet 110:239–252
Harding JE, Derraik JG, Berry MJ, Jaquiery AL, Alsweiler JM, Cormack BE et al (2013) Optimum feeding and growth in preterm neonates. J Dev Org Health Dis 4:215–222
Jobe AH (2006) Let’s feed the preterm lung. J Pediatr (Rio J) 82:165–166
Midulla F, Pierangeli A, Cangiano G, Bonci E, Salvadei S, Scagnolari C et al (2012) Rhinovirus bronchiolitis and recurrent wheezing: 1-year follow-up. Eur Respir J 39:396–402
Lemanske RF Jr, Jackson DJ, Gangnon RE, Evans MD, Li Z, Shult PA et al (2005) Rhinovirus illnesses during infancy predict subsequent childhood wheezing. J Allergy Clin Immunol 116:571–577
Valkonen H, Waris M, Ruohola A, Ruuskanen O, Heikkinen T (2009) Recurrent wheezing after respiratory syncytial virus or non-respiratory syncytial virus bronchiolitis in infancy: a 3-year follow-up. Allergy 64:1359–1365
Guilbert TW, Singh AM, Danov Z, Evans MD, Jackson DJ, Burton R et al (2011) Decreased lung function after preschool wheezing rhinovirus illnesses in children at risk to develop asthma. J Allerg Clin Immunol 128:532–U147
Jackson DJ, Gangnon RE, Evans MD, Roberg KA, Anderson EL, Pappas TE et al (2008) Wheezing rhinovirus illnesses in early life predict asthma development in high-risk children. Am J Respir Crit Care Med 178:667–672
Zomer-Kooijker K, van der Ent CK, Ermers MJ, Uiterwaal CS, Rovers MM, Bont LJ et al (2014) Increased risk of wheeze and decreased lung function after respiratory syncytial virus infection. PLoS One 9, e87162
Bacharier LB, Cohen R, Schweiger T, Yin-Declue H, Christie C, Zheng J et al (2012) Determinants of asthma after severe respiratory syncytial virus bronchiolitis. J Allergy Clin Immunol 130(91-100), e103
Ruotsalainen M, Hyvarinen MK, Piippo-Savolainen E, Korppi M (2013) Adolescent asthma after rhinovirus and respiratory syncytial virus bronchiolitis. Pediatr Pulmonol 48:633–639
Backman K, Piippo-Savolainen E, Ollikainen H, Koskela H, Korppi M (2014) Increased asthma risk and impaired quality of life after bronchiolitis or pneumonia in infancy. Pediatr Pulmonol 49:318–325
Piippo-Savolainen E, Remes S, Kannisto S, Korhonen K, Korppi M (2004) Asthma and lung function 20 years after wheezing in infancy: results from a prospective follow-up study. Arch Pediatr Adolesc Med 158:1070–1076
Sigurs N, Aljassim F, Kjellman B, Robinson PD, Sigurbergsson F, Bjarnason R et al (2010) Asthma and allergy patterns over 18 years after severe RSV bronchiolitis in the first year of life. Thorax 65:1045–1052
Vissing NH, Chawes BLK, Bisgaard H (2013) Increased risk of pneumonia and bronchiolitis after bacterial colonization of the airways as neonates. Am J Respir Crit Care Med 188:1246–1252
Santos JC, Zhang L, Menegatti PK, Guasselli CS, Celso Filho CM, Maito LR et al (2011) Pneumonia in the first 2 years of life, and asthma in preschool-age children. Pediatr Int 53:576–580
Edmond K, Scott S, Korczak V, Ward C, Sanderson C, Theodoratou E et al (2012) Long term sequelae from childhood pneumonia; systematic review and meta-analysis. PLoS One 7, e31239
La Grutta S, Indinnimeo L, di Coste A, Ferrante G, Landi M, Pelosi U et al (2013) Environmental risk factors and lung diseases in children: from guidelines to health effects. Early Hum Dev 89(Suppl 3):S59–S62
Burke H, Leonardi-Bee J, Hashim A, Pine-Abata H, Chen Y, Cook DG et al (2012) Prenatal and passive smoke exposure and incidence of asthma and wheeze: systematic review and meta-analysis. Pediatrics 129:735–744
Jones LL, Hashim A, McKeever T, Cook DG, Britton J, Leonardi-Bee J (2011) Parental and household smoking and the increased risk of bronchitis, bronchiolitis and other lower respiratory infections in infancy: systematic review and meta-analysis. Respir Res 12:5
Bowatte G, Lodge C, Lowe AJ, Erbas B, Perret J, Abramson MJ et al (2014) The influence of childhood traffic-related air pollution exposure on asthma, allergy and sensitization: a systematic review and a meta-analysis of birth cohort studies. Allergy 70:245–256
Balasubramaniam V, Mervis CF, Maxey AM, Markham NE, Abman SH (2007) Hyperoxia reduces bone marrow, circulating, and lung endothelial progenitor cells in the developing lung: implications for the pathogenesis of bronchopulmonary dysplasia. Am J Physiol Lung Cell Mol Physiol 292:L1073–L1084
Irwin D, Helm K, Campbell N, Imamura M, Fagan K, Harral J et al (2007) Neonatal lung side population cells demonstrate endothelial potential and are altered in response to hyperoxia-induced lung simplification. Am J Physiol Lung Cell Mol Physiol 293:L941–L951
van Haaften T, Byrne R, Bonnet S, Rochefort GY, Akabutu J, Bouchentouf M et al (2009) Airway delivery of mesenchymal stem cells prevents arrested alveolar growth in neonatal lung injury in rats. Am J Respir Crit Care Med 180:1131–1142
Popova AP, Bozyk PD, Bentley JK, Linn MJ, Goldsmith AM, Schumacher RE et al (2010) Isolation of tracheal aspirate mesenchymal stromal cells predicts bronchopulmonary dysplasia. Pediatrics 126:e1127–e1133
Bozyk PD, Popova AP, Bentley JK, Goldsmith AM, Linn MJ, Weiss DJ et al (2011) Mesenchymal stromal cells from neonatal tracheal aspirates demonstrate a pattern of lung-specific gene expression. Stem Cells Dev 20:1995–2007
Borghesi A, Massa M, Campanelli R, Bollani L, Tzialla C, Figar TA et al (2009) Circulating endothelial progenitor cells in preterm infants with bronchopulmonary dysplasia. Am J Respir Crit Care Med 180:540–546
Baker CD, Ryan SL, Ingram DA, Seedorf GJ, Abman SH, Balasubramaniam V (2009) Endothelial colony-forming cells from preterm infants are increased and more susceptible to hyperoxia. Am J Respir Crit Care Med 180:454–461
Baker CD, Balasubramaniam V, Mourani PM, Sontag MK, Black CP, Ryan SL et al (2012) Cord blood angiogenic progenitor cells are decreased in bronchopulmonary dysplasia. Eur Respir J 40:1516–1522
O’Reilly M, Thebaud B (2012) Cell-based strategies to reconstitute lung function in infants with severe bronchopulmonary dysplasia. Clin Perinatol 39:703–725
Ahn SY, Chang YS, Kim SY, Sung DK, Kim ES, Rime SY et al (2013) Long-term (postnatal day 70) outcome and safety of intratracheal transplantation of human umbilical cord blood-derived mesenchymal stem cells in neonatal hyperoxic lung injury. Yonsei Med J 54:416–424
Aslam M, Baveja R, Liang OD, Fernandez-Gonzalez A, Lee C, Mitsialis SA et al (2009) Bone marrow stromal cells attenuate lung injury in a murine model of neonatal chronic lung disease. Am J Respir Crit Care Med 180:1122–1130
Chang YS, Choi SJ, Ahn SY, Sung DK, Sung SI, Yoo HS et al (2013) Timing of umbilical cord blood derived mesenchymal stem cells transplantation determines therapeutic efficacy in the neonatal hyperoxic lung injury. PLoS One 8, e52419
Chang YS, Choi SJ, Sung DK, Kim SY, Oh W, Yang YS et al (2011) Intratracheal transplantation of human umbilical cord blood-derived mesenchymal stem cells dose-dependently attenuates hyperoxia-induced lung injury in neonatal rats. Cell Transplant 20:1843–1854
Chang YS, Oh W, Choi SJ, Sung DK, Kim SY, Choi EY et al (2009) Human umbilical cord blood-derived mesenchymal stem cells attenuate hyperoxia-induced lung injury in neonatal rats. Cell Transplant 18:869–886
Hansmann G, Fernandez-Gonzalez A, Aslam M, Vitali SH, Martin T, Mitsialis SA et al (2012) Mesenchymal stem cell-mediated reversal of bronchopulmonary dysplasia and associated pulmonary hypertension. Pulm Circ 2:170–181
Sutsko RP, Young KC, Ribeiro A, Torres E, Rodriguez M, Hehre D et al (2013) Long-term reparative effects of mesenchymal stem cell therapy following neonatal hyperoxia-induced lung injury. Pediatr Res 73:46–53
Tropea KA, Leder E, Aslam M, Lau AN, Raiser DM, Lee JH et al (2012) Bronchioalveolar stem cells increase after mesenchymal stromal cell treatment in a mouse model of bronchopulmonary dysplasia. Am J Physiol Lung Cell Mol Physiol 302:L829–L837
Waszak P, Alphonse R, Vadivel A, Ionescu L, Eaton F, Thebaud B (2012) Preconditioning enhances the paracrine effect of mesenchymal stem cells in preventing oxygen-induced neonatal lung injury in rats. Stem Cells Dev 21:2789–2797
Zhang H, Fang J, Su H, Yang M, Lai W, Mai Y et al (2012) Bone marrow mesenchymal stem cells attenuate lung inflammation of hyperoxic newborn rats. Pediatr Transplant 16:589–598
Zhang H, Fang J, Wu Y, Mai Y, Lai W, Su H (2013) Mesenchymal stem cells protect against neonatal rat hyperoxic lung injury. Expert Opin Biol Ther 13:817–829
Zhang X, Wang H, Shi Y, Peng W, Zhang S, Zhang W et al (2012) Role of bone marrow-derived mesenchymal stem cells in the prevention of hyperoxia-induced lung injury in newborn mice. Cell Biol Int 36:589–594
Alphonse RS, Vadivel A, Fung M, Shelley WC, Critser PJ, Ionescu L et al (2014) Existence, functional impairment, and lung repair potential of endothelial colony-forming cells in oxygen-induced arrested alveolar growth. Circulation 129:2144–2157
Baker CD, Seedorf GJ, Wisniewski BL, Black CP, Ryan SL, Balasubramaniam V et al (2013) Endothelial colony-forming cell conditioned media promote angiogenesis in vitro and prevent pulmonary hypertension in experimental bronchopulmonary dysplasia. Am J Physiol Lung Cell Mol Physiol 305:L73–L81
Balasubramaniam V, Ryan SL, Seedorf GJ, Roth EV, Heumann TR, Yoder MC et al (2010) Bone marrow-derived angiogenic cells restore lung alveolar and vascular structure after neonatal hyperoxia in infant mice. Am J Physiol Lung Cell Mol Physiol 298:L315–L323
Hodges RJ, Jenkin G, Hooper SB, Allison B, Lim R, Dickinson H et al (2012) Human amnion epithelial cells reduce ventilation-induced preterm lung injury in fetal sheep. Am J Obstet Gynecol 206(448):e448–e455
Vosdoganes P, Hodges RJ, Lim R, Westover AJ, Acharya RY, Wallace EM et al (2011) Human amnion epithelial cells as a treatment for inflammation-induced fetal lung injury in sheep. Am J Obstet Gynecol 205(2):156; e26–33
Vosdoganes P, Lim R, Koulaeva E, Chan ST, Acharya R, Moss TJ et al (2013) Human amnion epithelial cells modulate hyperoxia-induced neonatal lung injury in mice. Cytotherapy 15:1021–1029
Chang YS, Ahn SY, Yoo HS, Sung SI, Choi SJ, Oh WI et al (2014) Mesenchymal stem cells for bronchopulmonary dysplasia: phase 1 dose-escalation clinical trial. J Pediatr 164(966-972), e966
Agrons GA, Courtney SE, Stocker JT, Markowitz RI (2005) From the archives of the AFIP: lung disease in premature neonates: radiologic-pathologic correlation. Radiographics 25:1047–1073
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2015 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Sozo, F., O’Reilly, M., Harding, R. (2015). Fetal and Neonatal Origins of Lung Disease. In: Bertoncello, I. (eds) Stem Cells in the Lung. Stem Cell Biology and Regenerative Medicine. Springer, Cham. https://doi.org/10.1007/978-3-319-21082-7_5
Download citation
DOI: https://doi.org/10.1007/978-3-319-21082-7_5
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-21081-0
Online ISBN: 978-3-319-21082-7
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)