Abstract
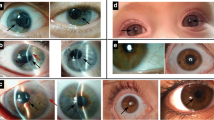
Aniridia is a rare, sight-threatening disorder that affects the iris, retina, optic nerve, lens, and cornea. Aniridia most often occurs as an isolated ocular abnormality without obvious systemic involvement, but may also be part of certain syndromes. Absence or hypoplasia of the iris and foveal hypoplasia are characteristic features that present from birth in patients with aniridia, usually resulting in photophobia, reduced visual acuity (normally 20/100–20/200) and nystagmus. Cataract, glaucoma, and aniridickeratopathy are frequently associated progressive ocular disorders with typically later onset. The prevalence of aniridia is about 1:80,000 with no known race or gender effect [1]. Approximately two-thirds of cases are inherited in an autosomal dominant fashion and one-third are sporadic. Aniridia is associated with PAX6 gene mutations. There is considerable phenotypic heterogeneity, but usually little difference between the two eyes.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
Aniridia in Brief
Aniridia is a rare, sight-threatening disorder that affects the iris, retina, optic nerve, lens, and cornea. Aniridia most often occurs as an isolated ocular abnormality without obvious systemic involvement, but may also be part of certain syndromes. Absence or hypoplasia of the iris and foveal hypoplasia are characteristic features that present from birth in patients with aniridia, usually resulting in photophobia, reduced visual acuity (normally 20/100–20/200) and nystagmus. Cataract, glaucoma, and aniridickeratopathy are frequently associated progressive ocular disorders with typically later onset. The prevalence of aniridia is about 1:80,000 with no known race or gender effect [1]. Approximately two-thirds of cases are inherited in an autosomal dominant fashion and one-third are sporadic. Aniridia is associated with PAX6 gene mutations. There is considerable phenotypic heterogeneity, but usually little difference between the two eyes.
Summary of Main Clinical Features in Aniridia
Iris
Iris hypoplasia is the most commonly seen ocular abnormality. Normally, only residual iris tissue is left in patients with aniridia. However, PAX6 mutation with no clinical evidence of defects in irides has been described [2].
Cornea
Aniridickeratopathy is thought to be caused by limbal stem cell deficiency secondary to PAX6 gene mutation [3]. It is a common feature, but often presents relatively late in the disease [4]. Corneal changes vary from mild peripheral vascularisation to pancorneal vascularisation, ulceration, keratinization, and opacification. Aniridickeratopathy can be painful and ultimately result in blindness [4]. Aniridickeratopathy may be triggered by surgical intervention for glaucoma or cataract [4].
Lens
The prevalence of cataract in aniridia varies from 50 % to over 90 % [1, 5]. Significant lens opacities typically develop from the teens. Lens subluxation or dislocation may also occur occasionally.
Glaucoma
Glaucoma develops in about half of patients with aniridia with mean onset in the teens [6]. Initially, patients are usually treated with intraocular pressure-lowering medications, but most eventually require surgery to control intraocular pressure.
Retina
Foveal hypoplasia is seen in about four out of five patients, whereas optic nerve hypoplasia is less common (about one in five cases) [7].
Dry Eyes
Dry eye disease is frequently observed in aniridia [8]. It is often caused by Meibomian gland dysfunction with stenosed Meibomian orifices [9]. Dry eye disease exacerbates the ocular surface problems associated with aniridic keratopathy [4].
Vision
Visual acuity is often between 20/100 and 20/200 [4], and nystagmus has been described in 95 % of the cases [7].
Systemic Features
Aniridia may be part of syndromes such as WAGR (Wilmstumour-Aniridia-Genital anomalies-Retardation) or Gillespie syndrome (cerebellar ataxia, partial aniridia, and developmental delay). In sporadic aniridia there is an increased risk of involvement of both the PAX6 and the WT1 gene, which strongly predisposes patients to developing Wilmstumour, a paediatric nephroblastoma. There is increasing awareness that even ‘isolated’ aniridia may have characteristic systemic features, such as reduced olfaction and auditory deficits [10, 11].
Future Avenues for Research: Examples
Genotype: Phenotype Correlations
Greater awareness of the huge variations in severity of aniridia is important to avoid misdiagnosed or undiagnosed mild presentations. The range of phenotypes described, with which PAX6 mutation is associated, is steadily increasing. The total number of unique DNA variants reported in the PAX6 locus-specific database is also increasing (359 at present). Studies correlating genotype with phenotype will facilitate diagnostics and make it easier to estimate a prognosis. The use of optical coherence tomography (OCT) for phenotypic characterization should be encouraged for three reasons: (1) its non-invasive nature; (2) few studies are hitherto performed; and (3) it gives valuable and quantifiable morphological information of the eye.
With recent advances involving nonsense mutation suppression drugs for certain types of aniridia [12], precise knowledge about the individual’s genotype may become therapeutically more important than ever. As pharmaceutical therapy is associated with adverse side effects, knowledge about the association between the genotype and phenotype will help guide the clinician as to when a possible intervention is advisable. However, the highest value in genetic testing is to determine whether there is an underlying WAGR deletion (i.e. deletion of PAX6 and WT1) due to the increased risk of cancer.
Aniridic Fibrosis Syndrome
Intraocular fibrosis has been noted after ocular surgery in 8 % of patients [13]. A characteristic feature of aniridic fibrosis syndrome is the development of fibrosis in the absence of clinically observable inflammation [13]. Activation of immature vessels in the rudimentary iris in patients with aniridia, as a consequence of the surgery, has been put forward as a potential mechanism. However, the syndrome is poorly understood. More research is needed to explore the underlying mechanisms, thereby paving the way for improvements in the clinical outcome of surgery for cataract, glaucoma, and aniridickeratopathy.
AniridicKeratopathy
There is undoubtedly room for improvement in the treatment of progressive ocular disorders such as glaucoma and cataract in patients with aniridia. For severe aniridickeratopathy caused by limbal stem cell deficiency, the potential for improvement may be significant. It is unclear whether limbal stem cell deficiency is primarily caused by reduced number of stem cells or unfavorable alterations in their microenvironment. Basic research on the interaction between limbal stem cells and their niche structures may provide valuable insight into the best therapeutic approach. Limbal stem cell deficiency may be treated by transplantation of either non-cultured [14] or ex vivo cultured tissue [15].
The use of cultured cells, rather than non-cultured cells, for treating limbal stem cell deficiency has some advantages [16]. If the cells are cultured, only a small biopsy is needed for producing a sufficiently large transplant [17], thus minimizing the risk of inducing stem cell failure in the donor eye [18]. The high rate of rejection following transplantation of non-cultured foreign tissue (i.e. limbal allograft) is associated with the presence of antigen-presenting cells, vessels, and lymphatics in the limbal region. If cultured cells are used, no or very few antigen-presenting cells are contained in the transplants, which reduces the risk of provoking an immune response [16, 19].
As limbal stem cell deficiency in aniridia is almost invariably bilateral, there is a need for a non-limbal cell source for ex vivo culture and transplantation. This avoids the need for immunosuppression and its many known adverse effects. Recently, several non-limbal autologous cell sources have shown promising results in treatment of limbal stem cell deficiency. These cells are derived from various locations, including oral mucosa, conjunctiva, epidermis, dental pulp, and hair follicles [16]. This field remains largely unexplored for patients with aniridia, apart from the transplantation of cultured oral mucosal cell sheets in four patients with aniridia [20, 21].
The costs related to the establishment and maintenance of a stem cell laboratory is a disadvantage of ex vivo based cell therapy. However, such facilities open up the possibility for genetic manipulation of cultured cells before they are transplanted to the patients. This is particularly relevant for patients with aniridia where PAX6 mutations are the cause of their limbal stem cell deficiency.
Recently, several non-cell based approaches to treat mild or moderate forms of limbal stem cell deficiency that do not require surgery have emerged. These include electro-stimulation, oxygen therapy, amniotic membrane extract, and limbal fibroblast conditioned medium [16]. These alternative methods of treating limbal stem cell deficiency have not yet been evaluated in clinical trials in aniridia. Such approaches may prove particularly useful for patients with aniridia as surgical intervention is associated with more complications in these patients compared to the general population.
Dry Eye Disease
There are very few studies on dry eye disease in patients with aniridia despite its high prevalence [8]. It is generally believed that increased lubrication has a beneficial effect on the development of aniridickeratopathy. Therefore, research on dry eye disease may serve a dual purpose: lessening dry eye disease symptoms and reducing the severity of aniridickeratopathy. Autologous serum may be effective in the treatment of some types of dry eye disease, but it does not address. Meibomian gland dysfunction, which is the most common form of dry eye disease. Meibomian gland dysfunction, if left untreated, may result of atrophy of the glands. Research on the many possible new strategies to treat Meibomian gland dysfunction, such as the use of Blephasteam (LaboratoiresThéa) and LipiFlow (TearScience), should be prioritized in patients with aniridia.
A Mutation-Independent Nonsense Mutation Suppression Strategy
Some mutations of PAX6 belong to the larger category entitled nonsense mutations, for which a novel therapeutic approach has recently been tested in a mouse model of aniridia [12]. The most successful results in this model were achieved through topical application of the drug formulation START (0.9 % sodium chloride, 1 % Tween 80, 1 % powdered ataluren, 1 % carboxymethylcellulose). Topical application has the benefit of reducing the risk of systemic adverse effects. Gregory-Evans and co-workers demonstrated that nonsense mutation suppression inhibited disease progression and, more remarkably, reversed retinal, lens, and corneal malformations [12]. It also restored electrical and behavioral responses of the retina. These findings suggest that START was able to suppress the nonsense mutation in order for full-length PAX6 protein to be synthesized [12]. More research, including additional animal studies, is warranted to fully explore the mechanism of action of this drug, in which ataluren is known to have nonsense suppression effect. Such efforts may lead to initiation of clinical trials applying nonsense mutation suppression strategies, which, if successful, may represent a paradigm shift in the therapy of aniridia.
Conclusion
Gene therapy and pharmaceutical therapy, such as nonsense suppressiondrugs, open up new exciting possibilities for research in aniridia. These advances, coupled with the emergence of strong international networks of clinicians, scientists, and patients, may pave the way for large, well-coordinated studies to significantly advance our knowledge and treatment of aniridia in the future.
References
Nelson LB, Spaeth GL, Nowinski TS, Margo CE, Jackson L (1984) Aniridia. A review. Surv Ophthalmol 28:621–642
Thomas S, Thomas MG, Andrews C et al (2014) Autosomal-dominant nystagmus, foveal hypoplasia and presenile cataract associated with a novel PAX6 mutation. Eur J Hum Genet EJHG 22:344–349
Nishida K, Kinoshita S, Ohashi Y, Kuwayama Y, Yamamoto S (1995) Ocular surface abnormalities in aniridia. Am J Ophthalmol 120:368–375
Hingorani M, Hanson I, van Heyningen V (2012) Aniridia. Eur J Hum Genet EJHG 20:1011–1017
Eden U, Lagali N, Dellby A et al (2014) Cataract development in Norwegian patients with congenital aniridia. Acta Ophthalmol 92:e165–e167
Netland PA, Scott ML, Boyle JW, Laudedale JD (2011) Ocular and systemic findings in a survey of aniridia subjects. J AAPOS Off Publ Am Assoc Pediatr Ophthalmol Strabismus Am Assoc Pediatr Ophthalmol Strabismus 15:562–566
Hingorani M, Williamson KA, Moore AT, van Heyningen V (2009) Detailed ophthalmologic evaluation of 43 individuals with PAX6 mutations. Invest Ophthalmol Vis Sci 50:2581–2590
Rivas L, Murube J, Rivas A, Murube E (2003) Impression cytology study of dry eyes in patients with congenital aniridia. Arch Soc Esp Oftalmol 78:615–622
Jastaneiah S, Al-Rajhi AA (2005) Association of aniridia and dry eyes. Ophthalmology 112:1535–1540
Sisodiya SM, Free SL, Williamson KA et al (2001) PAX6 haploinsufficiency causes cerebral malformation and olfactory dysfunction in humans. Nat Genet 28:214–216
Bamiou DE, Free SL, Sisodiya SM et al (2007) Auditory interhemispheric transfer deficits, hearing difficulties, and brain magnetic resonance imaging abnormalities in children with congenital aniridia due to PAX6 mutations. Arch Pediatr Adolesc Med 161:463–469
Gregory-Evans CY, Wang X, Wasan KM, Zhao J, Metcalfe AL, Gregory-Evans K (2014) Postnatal manipulation of Pax6 dosage reverses congenital tissue malformation defects. J Clin Invest 124:111–116
Tsai JH, Freeman JM, Chan CC et al (2005) A progressive anterior fibrosis syndrome in patients with postsurgical congenital aniridia. Am J Ophthalmol 140:1075–1079
Holland EJ, Djalilian AR, Schwartz GS (2003) Management of aniridic keratopathy with keratolimbal allograft: a limbal stem cell transplantation technique. Ophthalmology 110:125–130
Shortt AJ, Bunce C, Levis HJ et al (2014) Three-year outcomes of cultured limbal epithelial allografts in aniridia and Stevens-Johnson syndrome evaluated using the Clinical Outcome Assessment in Surgical Trials assessment tool. Stem Cells Trans Med 3:265–275
Utheim TP (2013) Limbal epithelial cell therapy: past, present, and future. Methods Mol Biology (Clifton NJ) 1014:3–43
Utheim TP, Lyberg T, Raeder S (2013) The culture of limbal epithelial cells. Methods Mol Biology (Clifton NJ) 1014:103–129
Jenkins C, Tuft S, Liu C, Buckley R (1933) Limbal transplantation in the management of chronic contact-lens-associated epitheliopathy. Eye (Lond) 7(Pt 5):629–633
Espana EM, Di Pascuale M, Grueterich M, Solomon A, Tseng SC (2004) Keratolimbal allograft in corneal reconstruction. Eye (Lond) 18:406–417
Burillon C, Huot L, Justin V et al (2012) Cultured autologous oral mucosal epithelial cell sheet (CAOMECS) transplantation for the treatment of corneal limbal epithelial stem cell deficiency. Invest Ophthalmol Vis Sci 53:1325–1331
Sotozono C, Inatomi T, Nakamura T et al (2013) Visual improvement after cultivated oral mucosal epithelial transplantation. Ophthalmology 120:193–200
Conflict of Interest
Tor Paaske Utheim is co-founder of the Norwegian Dry Eye Clinic, Oslo, Norway. The clinic is sponsored by Abbott Medical Optics, Abigo, Alcon, Allergan, BolPharma, Santen, TearScience, and Thea Laboratories. Utheim also holds patent applications on storage of cultured epithelial cells for treating limbal stem cell deficiency and age-related macular degeneration.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2015 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Utheim, T.P. (2015). Future Avenues of Research in Aniridia. In: Parekh, M., Poli, B., Ferrari, S., Teofili, C., Ponzin, D. (eds) Aniridia. Springer, Cham. https://doi.org/10.1007/978-3-319-19779-1_19
Download citation
DOI: https://doi.org/10.1007/978-3-319-19779-1_19
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-19778-4
Online ISBN: 978-3-319-19779-1
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)