Abstract
This chapter reviews investigations concerning the importance of veligers of the exotic bivalve Limnoperna fortunei in the diets of larval fish in the Río de la Plata basin. These studies have shown that of the 25 fish taxa studied, 18 consumed veligers of L. fortunei. These species included the most abundant members of Characiformes and Siluriformes. The relative frequency and biomass contribution of L. fortunei larvae differed strongly in pimelodid and Prochilodus lineatus larvae at different developmental stages and in different environments. Thus, as fish larvae grew, their diets shifted from veligers to other prey items. The fact that the earliest fish larvae are the most active consumers of veligers is particularly significant because these early larvae usually represent the most vulnerable life stage when mortality rates are the highest. In addition, field data and laboratory experiments indicate that small crustaceans have been largely replaced by L. fortunei veligers in diets of fish larvae, especially when veligers are abundant. Selectivity for feeding on veligers was recorded in the field and in laboratory experiments by manipulating prey density. Experiments also demonstrated that P. lineatus larvae grew to a significantly larger size with a high concentration of veligers in the diet. This new and abundant food resource appears to have a very important impact on the survival and growth of P. lineatus and probably other fish species as well.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Limnoperna fortunei
- Golden mussel
- Predation by fish
- Ecological impact
- Trophic interactions
- Fish diet
- Fish larvae
- Veligers
Introduction: Bivalve Invaders as Prey in Aquatic Ecosystems
One of the most important and extensively studied effects of introduced bivalves is their impact on trophic relationships and food web structures (Karatayev et al. 2007b; Bulté and Blouin-Demers 2008; García-Ortega et al. 2010). For example, several species of adult fish have incorporated the zebra mussel into their diets, but not always with beneficial results (French and Bur 1996; Molloy et al. 1997; García-Ortega et al. 2010). In South America, at least 50 fish species have been recorded to feed on adult Limnoperna fortunei, and this mussel has become an important food item for native fish of ecological and economic importance (see Chapter “Trophic Relationships of Limnoperna fortunei with Adult Fishes” in this volume; Boltovskoy and Correa 2015). Furthermore, the planktivorous larvae and juveniles of several fish species benefit from very high densities of the planktonic larval stages of L. fortunei (Paolucci et al. 2007).
Many planktivorous fish larvae in South American rivers are the product of reproductive migrations, in which mature adults migrate upstream to spawn, after which the larvae drift passively downstream until they reach a marginal wetland (Carolsfield et al. 2004). Even though these species migrate upstream for spawning, other aspects of these movements have species-specific traits, such as the extent and timing of migrations (Welcomme 1979; Fuentes and Espinach Ros 1998). This results in an ichthyoplankton characterized by a mix of different species and different developmental stages. In addition, larval feeding behaviors of these species are also different, depending on their developmental stage and environmental conditions (Rossi 2008). For example, larvae of the sábalo, Prochilodus lineatus, migrate from the main channel toward marginal lagoons while depending mostly on their yolk reserves for energy (Rossi 1992; Fuentes and Espinach Ros 1998; Paolucci 2002). Active feeding begins once they have reached marginal lagoons, which serve as nursery areas for several key fish species (Rossi 2008). In contrast, catfish larvae (Pimelodidae) and the larvae and juveniles of several ichthyophagous species start feeding in the channel shortly after hatching (Merigoux and Ponton 1998; Rossi 2001; Makrakis et al. 2008).
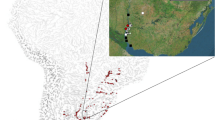
Location of sampling sites for studies investigating larval fish predation on veligers of Limnoperna fortunei. Red circles: data from Paolucci et al. (2007). (a Lower and Middle Paraná River. b Marginal environments); yellow circle: data from Rossi (2008); orange and blue circles: data from Paolucci (2010) (a Paraná and Paraguay rivers. c Salto Grande Reservoir)
Importance of Veligers in Larval Fish Diets
Using samples collected in 1996–1997, Rossi (2008) studied the trophic behavior of larval fish in the main and secondary channels of the Middle Paraná River (Fig. 1a) and found that veligers of L. fortunei were consumed by ten fish taxa (Table 1). Of these fishes, pimelodid larvae were the heaviest consumers of this new prey item. On a larger scale study of the impact of these veligers, Paolucci et al. (2007) analyzed fish larvae in the Middle and Lower Paraná during 2000–2001 and in subsidiary marginal lagoons in 2004 (Fig. 1a, b). They reported that L. fortunei veligers were actively consumed by 11 of 15 larval fish taxa surveyed (Table 1). At that time, L. fortunei was recorded in the guts of P. lineatus, Iheringichthys labrosus, Luciopimelodus pati, Sorubim lima, Pimelodus spp., and Parapimelodus valenciennis, as well as the larvae of other unidentified species of Anostomidae, Doradidae, Characiformes, and Pimelodidae (Fig. 2a–g). These first studies showed that veligers of L. fortunei were also present, although at low importance, in the gut contents of piscivorous species, such as Pseudoplatystoma sp. and Rhaphiodon vulpinus. In a subsequent analysis of the local ichthyoplankton, which included the Lower Paraguay River, Middle Paraná River, and the Salto Grande Reservoir (Fig. 1a, c), the list of consumers of veligers was extended to 18 out of a total of 25 analyzed taxa (Table 1; Paolucci 2010). In the Paraguay River and its marginal and lentic environments, several species of Clupeiformes, Pleuronectiformes, and Perciformes (Fig. 2i–k) were added to the list of predators. The temporal overlap between fish and mussel reproductive periods results in a stable food supply for the larval fishes and is a key factor in this relationship. In comparison, the zebra mussel, D. polymorpha, often has a shorter reproductive period (Karatayev et al. 2007a; see Chapter “Parallels and Contrasts between Limnoperna fortunei and Species of Dreissena” in this volume), and this may be one of the reasons that zebra mussel veligers are much less important in the diets of North American fish larvae (Banard et al. 2006).
Some of the larval fish species that feed on Limnoperna fortunei veligers. a Mesolarva of catfish, Iheringichthys labrosus. (b, d) Pimelodid protolarva. (c, e, h) Veligers of L. fortunei in gut contents. f Prochilodus lineatus protolarva. g Anostomid protolarva. i Flatfish, Catathyridium jenynsii. j Anchovy larva, Lycengraulis grossidens. k Sciaenid larva. (Modified from Paolucci 2010)
Feeding on Veligers by Larval Fish in the Main Channel Versus Marginal Environments
Some larval fish start feeding in the main river channel, while others forage in marginal water-bodies of the alluvial plain, and data from both environments were compared by Paolucci et al. (2007). These authors found that in the main channel of the Paraná River, eight taxa had L. fortunei larvae in their guts. Among these, pimelodid larvae such as I. labrosus, L. pati, and S. lima were by far the most active consumers of veligers (Fig. 3). Among the Characiformes in their study, only P. lineatus consumed L. fortunei in the main channel; however, veligers were the only identifiable food item recorded for all of these fish larvae. In the San Nicolás lagoons (a marginal environment connected to the river; Fig. 1a), seven out of ten taxa they investigated had L. fortunei in their guts. Approximately 20 % of the Characiformes consumed veligers. For the Siluriformes, the proportion was similar (23 %), but these fishes were comparatively scarce in these lagoons (ca. 10 % of all larvae collected).
Taxonomic composition, proportions of larvae with gut contents, and frequency of occurrence (FO) of Limnoperna fortunei for the dominant fish taxa (those comprising > 94 % of all the larvae retrieved) in the Paraná River and San Nicolás lagoons. (Based on data from Paolucci et al. 2007)
Differences in taxonomic compositions and feeding activities of larval fish assemblages between the main channel and marginal environments seem to be the main factors affecting the frequency of occurrence (FO) of L. fortunei in diets found by Paolucci et al. (2007; Fig. 3). The proportions of feeding larvae that had L. fortunei in their gut varied between 20 % (San Nicolás lagoons) and 56 % (Paraná River). In terms of biomass, however, the mean contributions of veligers in larval fish diets was smaller, ranging from 8 % (Paraná) to 12 % (San Nicolás) (mean weighted values), but contributions as high as 100 % were recorded for some taxa in the Paraná River. Larvae of the sábalo, P. lineatus, which is the most abundant species in the Paraná-Uruguay watershed (Sverlij et al. 1993; Bonetto 1998), were dominant in both environments (Fig. 3). In the Paraná River, only 3 % had gut contents, while in San Nicolás almost half of the specimens had food in their stomachs. On the other hand, for Siluriformes, proportions of nonempty guts in the Paraná were comparable to those in San Nicolás. Similar observations of differences between the main channel and secondary rivers have been made by Rossi (1992).
Average frequency of occurrence (FO) and total biomass (% contribution) of major prey items found in protolarvae, mesolarvae, and metalarvae, pooled data for the Paraguay River and Middle Paraná River (upper panels), and the Middle and Lower Paraná River and San Nicolás lagoons (lower panels). Letters denote significant differences between developmental stages at p < 0.01 (a) or p < 0.05 (b) (ANOVA, Duncan post-hoc test). (Based on data from Paolucci et al. 2007 and Paolucci 2010)
Ontogenetic Dietary Shift and Veliger Impact
It is well known that interactions between larval fish and their prey change over time (Lazzaro 1987; Merigoux and Ponton 1998), and field studies on the importance of L. fortunei veligers in the diets of larval fishes have also demonstrated ontogenetic changes (Paolucci et al. 2007; Rossi 2008; Paolucci 2010). In absolute terms, the FO and biomass of L. fortunei larvae in the guts of proto-, meso-, and metalarvae of mostly pimelodid species collected in the Lower and Middle Paraná and Paraguay rivers had more or less similar values (Paolucci et al. 2007; Paolucci 2010). However, as a percentage of total occurrence or biomass, size-related patterns in the diets of fish larvae were evident (Fig. 4). Protolarvae fed chiefly on L. fortunei veligers and cladocerans with fewer copepods and fish larvae (Fig. 4). Mesolarvae consumed veligers, cladocerans, and copepods in similar proportions, but exhibited increased frequencies of fish and insect larvae. Finally, metalarvae consumed veligers and cladocerans less often, but exhibited an increased amount of copepods and fish larvae in their diets. Because the biomass of copepods and fish larvae is 5–10 and 50 times greater, respectively, than that of veligers and cladocerans, the relative importance of veliger biomass dropped from 15 to 45 % in protolarvae and mesolarvae, to only 3 % in the metalarvae (Fig. 4). Thus, as larvae grew, their diet shifted from L. fortunei veligers to other larger prey items. Similar trends in the importance of veligers during the development of larval fish were observed by Rossi (2008) for pimelodid species such as S. lima, Pimelodus sp., and particularly Pseudopla-tystoma cf. corruscans. These results clearly highlight the importance of L. fortunei veligers as prey during the earliest developmental stages of larval fishes. The fact that the earliest fish larvae are the most active consumers of veligers is particularly significant because they usually represent the most vulnerable life history stage where mortality rates are the highest (Elliott and Persson 1978).
Mean selectivity index for the prey items assessed for the most abundant fish species present in San Nicolás lagoons (a) and in the Paraguay and Paraná rivers (b). Asterisks denote significant differences between proportions of the corresponding prey in the water column and in the gut contents at p < 0.05 (*), p < 0.01 (**), or p < 0.001 (***) (Chi-square tests). (Based on data from Paolucci et al. 2007 and Paolucci 2010)
Selectivity for Veligers by Fish Larvae as a Function of Prey Density: Field Evidence
Preliminary analyses of the feeding preferences of native larval fishes, such as P. lineatus and anostomid larvae, in marginal environments (Fig. 5a), showed that the veligers of L. fortunei were selected positively over cladocerans and copepods (Paolucci et al. 2007). The importance of L. fortunei in the diets of larval fishes was up to seven times higher than that expected based on the relative abundance of veligers in the zooplankton. A subsequent study of selectivity and feeding behavior of larval fish collected in the main channel recorded higher impact and preference values for veligers of L. fortunei in the Paraná River than in the Paraguay River (Fig. 5b; Paolucci et al. 2015). Comparison of the larval fish diet between the Paraguay and Paraná rivers indicates that average veliger FO was ca. five times more common in fishes caught in the Paraná River (68.4 % of which consumed L. fortunei vs.14.2 %; Fig. 6). In the Paraguay River, in contrast, cladocerans were usually favored over other prey, often accounting for significantly higher proportions of all items in the stomachs than in the water-column (Fig. 5b). Selectivity behaviors were not evenly distributed among taxa, but restricted to Characiformes, C. jenynsii, I. labrosus, and pimelodid larvae (Fig. 5b). In addition, selectivity toward veligers was positively correlated with the absolute abundance of veligers in the water column (Fig. 6); when more veligers were available, fish larvae relied on them more as prey. As seen in other studies (Deudero and Morales-Nin 2001; Graeb et al. 2004; Fulford et al. 2006), veliger density plays a central role in selectivity by the predators, mainly due to an increase in the rate of predator-prey encounters.
Contribution of the five main food items to the diet of fish larvae and zooplankton composition recorded in the Paraguay and Paraná Rivers. Statistically significant differences between the two rivers are denoted with asterisks (* p < 0.05; ** p < 0.01, Kruskal-Wallis test). (Modified from Paolucci 2010)
Effects of Developmental Stage and Veliger Density on Selectivity: Experimental Evidence
As in field studies for other fish species, comparisons of gut contents versus available food determined in laboratory experiments demonstrated that selectivity behavior was a function of prey density and the developmental stage of P. lineatus (Paolucci 2010; Paolucci et al. 2010a). For protolarvae, average proportions of veligers, small cladocerans, and nauplii were always higher in the gut contents than in the prey offered indicating selectivity for these items (positive values in Fig. 7) (small cladocerans and nauplii were the main prey of P. lineatus larvae before L. fortunei was present in South America; Rossi 1992). Mesolarvae preyed on veligers selectively only when these were very abundant in the experimental tanks (enriched veliger concentrations in Fig. 7); however, when veliger concentrations dropped, they were consumed less selectively, with gut contents yielding lower proportions of veligers. Mesolarvae were especially efficient at consuming small and medium-sized cladocerans, whose selectivity indices were almost invariably positive and statistically significant (Fig. 7). In contrast with younger larvae, metalarvae never selected veligers, regardless of their concentration in the experimental tanks, and they clearly favored small and medium-sized cladocerans. For P. lineatus, as well as for pimelodid larvae, the highest positive selectivity values were also associated with the highest proportions of veligers in the zooplankton.
Average selectivity index per prey item assessed for protolarvae (n = 15), mesolarvae (n = 15), and metalarvae (n = 15) in three experimental settings (enriched, normal, and low concentrations of veligers). Error bars denote confidence intervals at p < 0.05. Letters denote significant differences between available prey and gut contents at p < 0.05 (a), p < 0.01 (b), or p < 0.001 (c) (Chi-square tests) (Modified from Paolucci et al. 2010a)
Laboratory results and available field data for these fish species (Paolucci et al. 2007; Rossi 2008; Paolucci et al. 2010a) indicate that small crustaceans have been largely replaced by veligers, especially when veligers are abundant. This dietary switch, however, is restricted to the earliest larvae. As fishes grow larger and develop a larger mouth gape (Fig. 8a), the relative importance of large crustaceans increases. The replacement of crustaceans by veligers of L. fortunei in the diet of larval fishes is chiefly due to the fact that veligers are poor swimmers, with limited neuromuscular coordination and poor predator-avoidance behavior compared to crustacean zooplankton. Cladocerans, in turn, are slower and less agile than copepods. This gradient in predator-avoidance capabilities seems to be the main prey-selection factor during the earliest life stages of a fish. Indeed, for many fish species mollusc larvae have been reported to be preferred over crustaceans (Pepin and Penney 1997; Lehtiniemi et al. 2007), and cladocerans are generally preferred over copepods (Cooper and Goldman 1980; Vanderploeg et al. 1982; Clarke et al. 2004), sometimes regardless of prey size (Werner 1974). However, it is worth noting that several of these fish species also prey on the juvenile and adult stages of L. fortunei when they reach a larger developmental stage (see Chapter “Trophic Relationships of Limnoperna fortunei with Adult Fishes” in this volume).
a Correlation between total length and mouth width of the Prochilodus lineatus larvae used in the selectivity experiments (R = 0.917; p < 0.01; n = 45; modified from Paolucci et al. 2010a). Growth as b wet weight (mg) and c total length (mm) of larval fish fed three different diets: veliger-enriched (solid red line), natural (dashed blue line), and low-veliger (dotted green line). Vertical bars denote 95 % confidence intervals. Letters denote significant differences. (ANOVA, p < 0.01) as a between all diets; or (b) between at least one pair of diets. d weekly energetic costs of growth (black bars) and standard metabolic rate (yellow bars) according to the mean weight of the larval fish used in respiration experiments. Error bars denote 95 % confidence intervals of the combined energetic costs. (b, c, and d modified from Paolucci et al. 2010b)
Effects on Growth of Larval Fishes
Even in fish that are important predators of bivalves, some negative effects have been reported (French and Bur 1996; Nagelkerke and Sibbing 1996; Magoulick and Lewis 2002). These have been explained by the low caloric content of bivalve larvae compared to traditional food items, mainly due to the presence of shells that are not assimilated. However, growth experiments carried out using newly hatched P. lineatus larvae (with yolk-sac absorption completed) and different veliger concentrations showed positive rather than negative effects (Paolucci et al. 2010b). This experimental approach not only supported the idea that this new and abundant resource is selectively preyed upon by this larval fish, but also demonstrates that veligers of L. fortunei can significantly enhance the growth of P. lineatus larvae. Different veliger concentrations had significant effects on growth, for both total length and wet weight, of P. lineatus larvae; those fed a veliger-enriched diet had the highest growth performance, followed by those fed natural and low-veliger diets (Fig. 8b, c).
Similar to that observed in other studies with larval fishes (Halver 2001; Teshima et al. 2004), the enhanced growth rates observed by Paolucci et al. (2010b) may depend on the biochemical composition and caloric content of the veligers, as well as the energy costs involved in prey capture. Chemical composition analyses showed high protein and lipid contents for veligers with lipid contents being higher than those of cladocerans and copepods. This combination of high protein and fat contents, like that found in veligers of L. fortunei, has been highlighted by several authors as important in the diet of larval fish (Sargent et al. 1999; Lazo 2000; Rønnestad et al. 2007). While protein is the most important body component and accounts for over 50 % of the ash free dry weight (AFDW) in these organisms, lipids provide necessary energy during the fast-paced larval fish development period. Mostly as a consequence of high lipid content (17 % of the AFDW), a significantly higher specific caloric content was found in veligers also (24.88 ± 1.81 kJ/g dry weight) followed by cladocerans and copepods (Paolucci et al. 2010b). In addition to high energy density, veligers had a higher dry biomass than crustacean prey of the same or greater total length, and consequently veligers had comparatively higher total energy content sufficient to support the costs of growth and standard metabolic rate (Fig. 8d).
The energy density of veligers of L. fortunei is slightly higher than that recorded for adults of the invasive bivalve Dreissena polymorpha and other bivalve larvae (between 17.3 and 22.7 kJ/g) (Blaber 1979). In addition to the biochemical composition, the reduced energetic costs associated with the capture of slower prey, such as veligers, in comparison with faster prey, such as cladocerans, and especially copepods, may also have had an effect on larval growth. The results of these physiological studies combined with observations made during experimental and field investigations imply that selective feeding on slow and easy-to-capture prey results in a lower energetic cost of feeding and can result in a positive energetic impact that could enhance growth rates of larval fishes (Lazzaro 1987; Lankford and Targett 1997).
Impacts at Population and Community Levels
All these results suggest that fish species whose larvae have been observed to feed on veligers of L. fortunei have greatly benefited from its presence, and the impact of this new resource on fish populations is most likely very important. It is noteworthy that fish species whose diets rely heavily on L. fortunei are among the most abundant and ecologically important in the Paraná-Paraguay river system (Sverlij et al. 1993; Espinach Ros and Fuentes 2001). For example, deposit-feeding adults of P. lineatus constitute the main food item of larger ichthyophagous species (Sverlij et al. 1993). Thus, feeding conditions for P. lineatus may strongly affect abundances of many other fish species. Consequently, the effects of these shifts in the feeding behavior of larval fishes are conceivably not restricted to the organisms directly involved in the interactions, but may have cascading effects both up and down trophic webs (MacIsaac et al. 1999; Yan et al. 2001; Clarke et al. 2004). Insofar as the new interactions modify established grazing pressures, they can strongly affect species composition and size structure of the zooplankton community, which in turn may change phytoplankton abundance and composition (Strecker and Arnott 2008). Indirect impacts on other fishes may also derive from these ecological rearrangements through the direct consumption of veligers (Paolucci et al. 2007), or from changes in the availability of other food items.
Larval Fish Predation as Biological Control
Whether or not grazing on veligers by larval fish is able to curtail the growth of mussel populations is a question of major interest. Sylvester et al. (2007) suggested that adult mussels will not be controlled by fish predation, but a similar estimate for veligers is more complicated. Several key elements remain unknown; in particular, the reproductive output of L. fortunei on a basin-wide scale is unclear. Assuming conservative densities of 1000 ind/m3 for veligers (Boltovskoy et al. 2009) and around 3 ind/m3 for fish larvae (Fuentes and Espinach Ros 1998), and an ingestion rate of 2 veligers/h (Paolucci et al. 2010a), one could speculate that on a steady-state basis P. lineatus consumes daily between 10 and 20 % of the standing stock of veligers. This figure could probably be doubled to include other species of fish that consume veligers (Paolucci et al. 2007; Paolucci et al. 2010a). However, the reproductive period of L. fortunei (around September–April) is much longer than that of most fishes (between November–December and February–March (cf. Rossi et al. 2007), and this must decrease the long-term impact significantly. Thus, although these estimates are very rough, they agree with the conclusion of Sylvester et al. (2007) who concluded that predation impact on the geographical spread of L. fortunei is probably minor, and the potential for predation to control the spread of the bivalve seems limited.
References
Banard C, Martineau C, Frenette JJ, Dodson JJ, Vincent WF (2006) Trophic position of zebra mussel veligers and their use of dissolved organic carbon. Limnol Oceanogr 51:1473–1484
Blaber SJM (1979) The biology of filter feeding teleosts in Lake St Lucia, Zululand. J Fish Biol 15:37–59
Boltovskoy D, Correa N (2015) Ecosystem impacts of the invasive bivalve Limnoperna fortunei (golden mussel) in South America 746:81–95
Boltovskoy D, Sylvester F, Otaegui A, Leytes V, Cataldo D (2009) Environmental modulation of the reproductive activity of the invasive mussel Limnoperna fortunei in South America. Austral Ecol 34:719–730
Bonetto AA (1998) Panorama sinóptico sobre la ictiofauna, la pesca y la piscicultura en los ríos de la cuenca del Plata con especial referencia al Paraná. Revista de Ictiología 6:3–15
Bulté G, Blouin-Demers G (2008) Northern map turtles (Graptemys geographica) derive energy from the pelagic pathway through predation on zebra mussels (Dreissena polymorpha). Freshw Biol 53:497–508
Carolsfield J, Harvey B, Ross C, Baer A (2004) Migratory fish of South America: biology, fisheries and conservation status. World Bank, Victoria, pp 1–380
Clarke LR, Letizia PS, Bennett DH (2004) Autumn-to-Spring energetic and diet changes among Kokanee from North Idaho lakes with and without Mysis relicta. N Am J Fish Manag 24:597–608
Cooper SD, Goldman CR (1980) Opossum shrimp (Mysis relicta) predation on zooplankton. Can J Fish Aquat Sci 37:909–919
Deudero S, Morales-Nin B (2001) Prey selectivity in planktivorous juvenile fishes associated with floating objects in the western Mediterranean. Aquac Res 32:481–490
Elliott JM, Persson L (1978) The estimation of daily rates of food consumption for fish. J Animal Ecol 47:977–991
Espinach Ros A, Fuentes CM (2001) Recursos pesqueros y pesquerías de la Cuenca del Plata. In: Bezzi S, Akselman R, Boschi EE (eds) Síntesis del estado de las pesquerías marítimas argentinas y de la Cuenca del Plata. Años 1997–1998. Instituto Nacional de Investigación y Desarrollo Pesquero, Mar del Plata, pp 353–358
French JRP, Bur MT (1996) The effect of zebra mussel consumption on growth of freshwater drum in Lage Erie. J Freshw Ecol 11:283–289
Fuentes CM, Espinach Ros A (1998) Variación de la actividad reproductiva del sábalo, Prochilodus lineatus (Valenciennes, 1847), estimada por el flujo de larvas en el rıo Paraná Inferior. Nat Neotrop 29:25–32
Fulford RS, Rice JA, Miller TJ, Binkowski FP, Dettmers JM, Belonger B (2006) Foraging selectivity by larval yellow perch (Perca flavescens): implications for understanding recruitment in small and large lakes. Can J Fish Aquat Sci 63:28–42
García-Ortega A, Verreth J, Vermis K, Nelis HJ, Sorgeloos P, Verstegen M (2010) Laboratory investigation of daily food intake and gut evacuation in larvae of African catfish Clarias gariepinus under different feeding conditions. Aquac Int 18:119–134
Graeb BDS, Dettmers JM, Wahl DH, Cáceres CE (2004) Fish size and prey availability affect growth, survival, prey selection, and foraging behavior of larval yellow perch. Transactions Am Fish Soc 133:504–514
Halver JE (2001) My 50 years in fish nutrition, 1949–99. Aquac Res 32:615–622
Karatayev AY, Boltovskoy D, Padilla DK, Burlakova LE (2007a) The invasive bivalves Dreissena polymorpha and Limnoperna fortunei: parallels, contrasts, potential spread and invasion impacts. J Shellfish Res 26:205–213
Karatayev AY, Padilla DK, Minchin D, Boltovskoy D, Burlakova LE (2007b) Changes in global economies and trade: the potential spread of exotic freshwater bivalves. Biol Invasions 9:161–180
Lankford TE, Targett TE (1997) Selective predation by juvenile weakfish: post-consumptive constraints on energy maximization and growth. Ecology 78:1049–1061
Lazo J (2000) Conocimiento actual y nuevas perspectivas en el desarrollo de dietas para larvas de peces marinos. In: Avances en Nutrición Acuícola. V Simposium Internacional de Nutrición Acuícola, Mérida (Venezuela)
Lazzaro X (1987) A review of planktivorous fishes: their evolution, feeding behaviours, selectivities, and impacts. Hydrobiologia 146:97–167
Lehtiniemi M, Hakala T, Saesmaa S, Viitasalo M (2007) Prey selection by the larvae of three species of littoral fishes on natural zooplankton assemblages. Aquat Ecol 41:85–94
MacIsaac HJ, Grigorovich IA, Hoyle JA, Yan ND, Panov VE (1999) Invasion of Lake Ontario by the Ponto–Caspian predatory cladoceran Cercopagis pengoi. Can J Fish Aquat Sci 56:1–5
Magoulick DD, Lewis LC (2002) Predation on exotic zebra mussels by native fishes: effects on predator and prey. Freshw Biol 47:1908–1918
Makrakis MC, Nakatani K, Bialetzki A, Gomes LC, Sanches PV, Baumgartner G (2008) Relationship between gape size and feeding selectivity of fish larvae from a neotropical reservoir. J Fish Biol 72:1690–1707
Merigoux S, Ponton D (1998) Body shape, diet and ontogenetic diet shifts in young fish of the Sinnamary River, French Guiana, South America. J Fish Biol 52:556–569
Molloy DP, Karatayev AY, Burlakova EB, Kurandina DP, Laruelle F (1997) Natural enemies of zebra mussels: predators, parasites, and ecological competitors. Rev Fish Sci 5:27–97
Nagelkerke LAJ, Sibbing FA (1996) Efficiency of feeding on zebra mussel (Dreissena polymorpha) by common bream (Abramis brama), white bream (Blicca bjoerkna), and roach (Rutilus rutilulus): the effects of morphology and behavior. Can J Fish Aquat Sci 53:2847–2861
Paolucci EM (2002) Actividad reproductiva del sábalo Prochilodus lineatus (Valenciennes, 1847) y otras especies de interés comercial y deportivo en el río Uruguay inferior, estimada por la abundancia de estadios larvales en la deriva. MSc Thesis, Universidad de Buenos Aires (Argentina), pp 1–74
Paolucci EM (2010) Impacto del molusco invasor Limnoperna fortunei sobre el ecosistema: interacción trófica y efectos sobre las larvas de peces nativos. PhD Thesis, Universidad de Buenos Aires (Argentina), pp 1–162
Paolucci EM, Cataldo DH, Fuentes CM, Boltovskoy D (2007) Larvae of the invasive species Limnoperna fortunei (Bivalvia) in the diet of fish larvae in the Paraná River, Argentina. Hydrobiologia 589:219–233
Paolucci EM, Cataldo DH, Boltovskoy D (2010a) Prey selection by larvae of Prochilodus lineatus (Pisces): indigenous zooplankton versus larvae of the introduced bivalve Limnoperna fortunei. Aquat Ecol 44:255–267
Paolucci EM, Thuesen EV, Cataldo DH, Boltovskoy D (2010b) Veligers of an introduced bivalve (Limnoperna fortunei) are a new food resource that enhances growth of larval fish in the Paraná River (South America). Freshw Biol 55:1831–1844
Paolucci EM, Almada P, Cataldo D, Boltovskoy D (2015) Native fish larvae take advantage of introduced mussel larvae: field evidence of feeding preferences on veligers of the introduced freshwater bivalve Limnoperna fortunei. Hydrobiologia 745:211–224
Pepin P, Penney RW (1997) Patterns of prey size and taxonomic composition in larval fish: are there general size-dependent models? J Fish Biol 51:84–100
Rønnestad I, Kamisaka Y, Conceição LEC, Morais S, Tonheim SK (2007) Digestive physiology of marine fish larvae: hormonal control and processing capacity for proteins, peptides and amino acids. Aquaculture 268:82–97
Rossi LM (1992) Evolucion morfológica del aparato digestivo de postlarvas y prejuveniles de Prochilodus lineatus (Val., 1847) (Pisces, Curimatidae) y su relacion con la dieta. Revue d’Hydrobiologie Tropicale 25:159–107
Rossi LM (2001) Ontogenetic diet shifts in a neotropical catfish, Sorubim lima (Schneider) from the River Paraná System. Fish Manag Ecol 8:141–152
Rossi LM (2008) Ecologia trófica de peces de importancia económica del Río Parana durante su ontogenia temprana. PhD Thesis, Universidad Nacional de La Plata (Argentina), pp 1–174
Rossi L, Cordiviola E, Parma MJ (2007) Fishes. In: Iriondo MH, Paggi JC, Parma MJ (eds) The middle Parana River. Limnology of a subtropical wetland. Springer, Berlin, pp 305–325
Sargent J, Bell G, McEvoy L, Tocher D, Estevez A (1999) Recent developments in the essential fatty acid nutrition of fish. Aquaculture 177:191–199
Strecker AL, Arnott SE (2008) Invasive predator, Bythotrephes, has varied effects on ecosystem function in freshwater lakes. Ecosystems 11:490–503
Sverlij SB, Espinach Ros A, Orti G (1993) Sinopsis de los datos biológicos y pesqueros del sábalo Prochilodus lineatus (Valenciennes, 1847). Food and Agriculture Organization, Rome, pp 1–64
Sylvester F, Boltovskoy D, Cataldo D (2007) Fast response of freshwater consumers to a new trophic resource: Predation on the recently introduced Asian bivalve Limnoperna fortunei in the lower Parana River, South America. Austral Ecology 32:403–415
Teshima SI, Koshio S, Ishikawa M, Alam MS, Hernandez LHH (2004) Effects of protein and lipid sources on the growth and survival of red sea bream Pagrus major and Japanese flounder Paralichthys olivaceus receiving micro-bound diets during larval and early juvenile stage. Aquac Nutr 10:279–287
Vanderploeg HA, Bowers JA, Chapelski O, Soo HK (1982) Measuring in situ predation by Mysis relicta and observations on underdispersed microdistributions of zooplankton. Hydrobiologia 93:109–119
Welcomme RL (1979) Fisheries ecology of floodplain rivers. Longman, London, pp 1–317
Werner EE (1974) The fish size, prey size, handling time relation in several sunfishes and some implications. J Fish Res Board Can 31:1531–1536
Yan ND, Blukacz A, Sprules WG, Kindy PK, Hackett D, Girard RE, Clark BJ (2001) Changes in zooplankton and the phenology of the spiny water flea, Bythotrephes, following its invasion of Harp Lake, Ontario, Canada. Can J Fish Aquat Sci 58:2341–2350
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2015 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Paolucci, E., Thuesen, E. (2015). Trophic Relationships of Limnoperna fortunei with Larval Fishes. In: Boltovskoy, D. (eds) Limnoperna Fortunei. Invading Nature - Springer Series in Invasion Ecology, vol 10. Springer, Cham. https://doi.org/10.1007/978-3-319-13494-9_12
Download citation
DOI: https://doi.org/10.1007/978-3-319-13494-9_12
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-13493-2
Online ISBN: 978-3-319-13494-9
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)