Abstract
Potentially overlooked and neglected sources of healthcare-acquired pathogens are non-intrusive soft and hard surfaces located in clinical settings. Microbes can survive on bedding, uniforms, trays, bed rails and other such surfaces for days to months. Furthermore, on some of these surfaces, such as patient bedding, the microorganisms proliferate as textiles are an excellent substrate for bacterial and fungal growth. Additionally the temperature and humidity conditions present between the patients and these textiles are appropriate for microorganism multiplication. Bed making in hospitals can release large quantities of microorganisms into the air, which contaminate the surroundings. Thus soft and hard surfaces that are in direct or indirect contact with the patients can serve as a source of healthcare-acquired pathogens.
Copper oxide impregnated materials have potent intrinsic biocidal properties. This manuscript reviews the laboratory and clinical studies that demonstrate that soft and hard surfaces containing copper oxide particles reduce bioburden and healthcare-acquired infection rates.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others

Keywords
- Copper Oxide
- Clostridium Difficile Infection
- Antibiotic Resistant Bacterium
- Biocidal Property
- Microbial Proliferation
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
5.1 Hospital Textiles as a Source of Healthcare-Acquired Pathogens
Textile products are widely used in the hospital environment. They range from simple cleaning wipes to advanced barrier fabrics used in operating rooms. Some of the products are in direct contact with the patients, such as blankets, sheets, pyjamas, towels, gowns, and pillowcases. Others are used by the healthcare personnel, such as uniforms, surgical gowns, face masks, and head and shoe covers. Some products are present in the patient wards, such as drapes, table covers and privacy curtains.
Textiles in general are an excellent substrate for microbial proliferation when in contact with the human body. The very large surface area, the capacity to retain oxygen, and the moisture and temperature conditions present between the skin and the textiles provide ideal environment for microbial proliferation. Humans shed bacteria directly from their skin, nasal cavities, genitalia area, and sweat onto the textiles they use [1]. Bacterial shedding is greater in patients than in healthy individuals [2, 3]. In addition, hospital textiles come in contact with spillages and body exudates, such as blood, stool, urine, nasopharyngeal secretions and vomit, all of which can contain large amounts of bacteria and serve as a bacterial nutrient source. Heavy microbial colonization of sheets, patient pajamas, healthcare worker uniforms, and privacy curtains, including by antibiotic resistant bacteria, has been reported [4–29]. Contamination of clean laundry occurs shortly after use [14]. Without washing, bacteria, fungi and viruses can remain viable on textiles, under ambient temperature and humidity, for very prolonged periods of time; even months [19, 29–36] (See also Chap. 2). The higher the bacterial titer spiked onto the fabrics, the longer the bacteria can survive [31]. Unfortunately, some microorganisms remain viable even after industrial laundry [7, 37–39], and contaminated laundry can lead to cross-contamination of clean laundry [40].
While proliferating on the textiles, some microorganisms secrete unpleasant volatile molecules creating foul odors [41]. But, more importantly, some microorganisms that multiply or remain on the textiles can be a source of healthcare-acquired pathogens [7, 42] (Fig. 5.1). These pathogens can be transmitted from one part of the host’s body to another [43]. They can also be the source of direct or indirect infection of patients and hospital personnel, as discussed below.
Contamination and healthcare-acquired infections (HAI) of patients and hospital personnel via contaminated towels, gowns, sheets, cleaning wipes and other hospital textiles with Methicillin-resistant Staphylococcus aureus (MRSA) [11, 24, 28, 44–47], Vancomycin-resistant enterococcus (VRE) [19, 24, 48], Carbapenem-resistant Acinetobacter [20], multidrug-resistant Acinetobacter baumannii [25, 28, 49], multidrug-resistant Pseudomonas aeruginosa [24, 50], Bacillus cereus [51–55], Cryptosporidium [56], Microsporum canis [57], Norwalk gastroenteritis [58], Klebsiella pneumonia [28], Rhizopus [59], Salmonella gastroenteritis [60], Salmonella typhimurium [61], Sarcoptes scabiei [62], or Streptococcus pyogenes [37, 63] have previously been reported (Table 5.1). Viruses can also survive on textiles for days and thus be a source of contamination ([33, 64–67] and Chap. 2).
When handling contaminated textiles hospital, personnel can contaminate their gloves with micro-organisms and then contaminate other surfaces, such as door knobs, and even patients directly [3, 11, 20–22, 24, 25, 49, 68, 69]. Furthermore, studies have shown that when the personnel change the bed linens or patients garments, large quantities of micro-organisms are released into the air, which then contaminate the immediate and non-immediate surroundings in the same room as well as throughout the building via the air conditioning system [3, 46, 70–72]. Healthcare workers who touch the aerosol contaminated surfaces can then contaminate patients [3, 73]. Airborne transmission of pathogens such as Mycobacterium tuberculosis and Aspergillus niger is well documented [3]. It has also been implicated in healthcare-acquired outbreaks of A. baumannii, P. aeruginosa, Scedosporium prolificans, S. aureus, MRSA, and other Staphylococci spp [74–82].
5.2 Biocidal Textiles as a Tool to Fight Healthcare-Acquired Infections
Based on the above, it has been hypothesized that endowing the hospital textiles, especially those that come in contact with the patients, such as patient’s sheets, pillowcases, robes, and pyjamas, with biocidal properties, would help reduce HAI by reducing an important source of microbes contributing to endogenous, indirect-contact, and aerosol transmission of healthcare-acquired pathogens [42]. Nicas and Sun [83] by using an integrated mathematical model of the infection risk in a health-care environment, also concluded that biocidal textiles have the potential to substantially reduce HAI.
Biocidal textiles should have potent broad spectrum antimicrobial, antifungal and antiviral efficacies. They should be highly effective against antibiotic resistant micro-organisms, especially those already circulating in the hospital environment causing HAI outbreaks, such as MRSA, VRE, and Carbapenem-resistant K. pneumoniae (CRKP). Additionally they should not enable the development of resistant micro-organisms to the biocidal compound. They should not be affected by washings and continue to be efficacious for the life of the product. Obviously, they should be safe to humans following continuous dermal exposure [42] (Table 5.2).
In the last 20 years, the development of biocidal textiles in general and specifically for the use in the hospital environment has gained momentum, and different biocidal compounds are being explored for this purpose [84–87]. Biocides can by chemically or physically attached to the natural or synthetic fibres from which the textile products are made or to the surface of the finished textile products. Surface applications usually have a lower persistence over time, as the active ingredient is lost due to friction and washing. Other biocides are introduced earlier during the production stage by impregnating them in the polymeric fibres used in the textile industry. Some of the biocidal active ingredients being studied are Cliniweave®, organofunctional silane, citric acid, copper, silver, zinc, triclosan, quaternary ammonium compounds, chitosan and zeolite. Some of the above active ingredients have been found not to be appropriate for use in hospital related applications (e.g. [88, 89]).
Only a few clinical trials have been performed to determine the efficacy of biocidal textiles in clinical settings. Most of the studies examined the capacity of the biocidal textiles to reduce microbial contamination [4, 90–94]. These studies, which included personnel uniforms, patient linens, scrubs, blankets, privacy curtains, cloths and mops, found statistically significant lower bioburden levels than those found in the matched non-biocidal controls. One study, performed with only 10 workers that used silver containing jackets and pants, did not find any significant difference in the extent of microbial contamination between the silver containing textiles and control textiles [95]. It may be that a larger sample size was required to prove the silver containing fabric’s efficacy. Also, a randomized controlled study that compared the bacterial contamination of uniforms of healthcare workers when using a regular textile and two textile containing antimicrobial finishes, did not find any decrease in the bioburden levels in the antimicrobial textiles [96]. Unfortunately, the identity and nature of the antimicrobial components in the scrubs tested is not clear. Interestingly, in a recent study in which copper-coated films (21 × 39.7 cm) were attached to bed sheets used by a heavily MRSA-colonized patient found 20–130 MRSA colony forming units (cfu) in these films as opposed to 6,600–11,000 cfu on the surface of the non-film-coated control sheet areas [97].
The capacity of antimicrobial cleaning cloths to reduce bioburden and HAI was demonstrated. For example, a recent study examined the capacity of copper treated cleaning cloths in neutralizing the bacterial virus MS2. This virus serves as a non-pathogenic surrogate virus to clinically relevant viruses such as hepatitis A, enteroviruses, poliovirus or novovirus, due to its structure and environmental stability. Ninety percent of the absorbed virus in the cloths were killed, reducing significantly potential cross contamination during cleaning [98]. More dramatically, by introducing peracetic acid sporicidal wipes, the Clostridium difficile infections rates in an acute London trust were reduced by 72 % during the monitored 18 months as compared to the previous period [99].
In contrary to lab conditions, during in vivo use, continual re-inoculation with pathogens occurs. Since the killing of the microorganisms is not instant, the expectation is not to obtain a sterile fabric, but a fabric that prevents microbial proliferation and reduces the bioburden levels significantly. The concept that such textiles can reduce HAI, to the best of my knowledge, has been demonstrated in only one clinical trial (to be discussed below) and obviously more trials are needed to clearly establish the capacity of biocidal textiles to help in the fight against HAI.
Furthermore, as HAI are spreading into the community (e.g. [100]), the use of biocidal textiles and biocidal hard surfaces may not only significantly contribute to the reduction of HAI, but may also confer protection in other environments where at-risk individuals run the risk of contracting infections such as in long term care facilities.
5.3 Biocidal Textiles Containing Copper Oxide
Copper is one of the several materials that are being explored as a potent wide spectrum biocide to be used in hard and soft surfaces in clinical settings for the reduction of HAI. The biocidal mechanisms of copper are discussed in Chap. 6 of this Book. Different copper compounds that were applied to different textile fibres or polymers via different techniques, demonstrated potent in vitro biocidal efficacy including against antibiotic resistant bacteria [4, 101–116]. Most of these studies were conducted in the academia and the only technology that has generated textile products widely used commercially is the technology based on the impregnation of copper oxide particles into products [4, 108–110, 117–123].
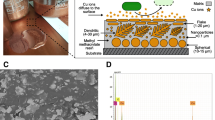
Copper oxide has been chosen as the active copper form to be introduced into textiles due to two main reasons: it is a non-soluble form of copper and it is highly reactive with potent wide spectrum biocidal properties [124]. As can be seen in Fig. 5.2, the copper oxide particles are an integral part of the polymeric fibers, as they are homogenously distributed throughout the polymeric matrix. This is very important for biocidal textiles as even when some of the external polymeric fiber material disintegrates due to friction, repeated use and laundry resulting in loss of the surface copper oxide particles, there are always “new” copper oxide particles that “reach” the surface of the fiber, endowing the fiber with biocidal properties for the life of the fiber.
Copper oxide impregnated textiles. (a) Scanning electronic microscope (SEM) picture of a cross section of a synthetic fiber impregnated with copper oxide particles (white dots). (b) SEM picture of a woven fabric in which the copper oxide impregnated polyester yarn is found only in the weft of the textile. (c) An X-ray photoelectron spectrum analysis of a white dot shown in (b), demonstrating that it is copper. (d and e) Pictures showing hospital textiles containing copper oxide in use in clinical settings (e.g. the beige and brown linens and the green uniform and blanket)
The biocidal efficacy is not affected by repeated use, home or industrial washings [109] (Fig. 5.3). This is in contrast to coating technologies in which the active material is only bound to the external layer of the fiber. Once this externally bound active material is removed from the surface of the fiber due to friction or laundry, the fiber losses its bioactive characteristics.
The biocidal potency of a hospital linen containing 1 % copper oxide particles (w/w) was tested by an independent laboratory (AminoLabs, Rehovot, Israel) by using the American Association of Textile Chemists and Colorists (AATCC) test method (TM) 100. The linen was washed following the AATCC TM 150. Candida albicans and Trichophyton mentagrophytes were exposed to the fabric for 24 h. Staphylococcus aureus and Escherichia coli were exposed to the fabric for 4 h. The results shown are the mean of duplicate samples. The titers of each microorganism before being exposed to the fabric samples are shown by the arrow.
The copper oxide impregnated products possess broad-spectrum antimicrobial properties, including against antibiotic resistant bacteria [4, 108–110, 117, 119, 125, 126]. These products include biocidal fabrics [4, 108, 109, 117], anti-fungal socks [108, 118, 121, 123, 127], anti-viral masks and filters [119, 125, 126, 128], anti-dust mite mattress-covers [108, 129], and non-porous biocidal countertops (see next section).
A preliminary pilot study with 30 patients, who slept overnight on regular sheets and then overnight on sheets containing copper-oxide, demonstrated a statistically significant lower (~50 %) bacterial colonization on the copper-oxide containing sheets than on regular-sheets [4]. Similar statistically significant lower titers of gram positive and gram negative bacteria were recovered from copper oxide containing sheets than regular sheets (n = 40), immediately after 7 h of patient’s use [130].
Importantly, in a clinical trial in the Reuth Medical Center (Tel Aviv, Israel), in which the regular non-biocidal linens in a chronic care head injury ward were replaced with the biocidal copper oxide impregnated linens, the rates of HAI per 1,000 hospitalization days (HD) were reduced by 24 % (P < 0.05). Accordingly there was a 32.8 % reduction in total number of days of antibiotics administration per 1,000 HD (P < 0.0001) and there was a 47 % reduction in the number of fever days (>38.5 °C) per 1,000 HD (P < 0.01) [130]. The study was conducted in a chronic care head injury ward as most of the patients hospitalized in this ward are high risk patients typically immunocompromised. Unfortunately the most common medical complication which afflicts them is a HAI [131–133]. Based on the successful result of this trial, the Reuth Medical Center has now changed all their linens in all wards to the copper oxide impregnated linens.
There is no reason to believe that reducing bioburden in other wards or clinical settings by using biocidal linens would not reduce bioburden and HAI. The use of biocidal textiles should be a complementary approach to fight HAI in medical institutions as well as long term care facilities, where the risks of acquiring an infection are high. However, additional studies with other patient populations and different wards should further test this notion. Currently, a study is being conducted at Sentara Norfolk General Hospital (Norfolk, Va.), where a critical care unit that shares the same nursing staff will test the biocidal fabrics in one side of the unit for 6 months before switching them to the other unit for another 6 months.
5.4 Non-porous Solid Biocidal Surfaces Containing Copper Oxide
Elemental copper and copper alloys have been registered by the USA Environmental Protection Agency (EPA) as antimicrobial substances with approval to make public health claims that the copper‐oxide impregnated surfaces kill greater than 99.9 % of gram negative and gram positive bacteria within 2 h of exposure. The approvals were obtained after demonstrating in independent laboratories potent biocidal efficacy following Good Laboratory Practices (GLP) testing. The significant contribution of metallic copper surfaces to the reduction of bioburden in clinical settings [134–140] and to reduction of HAI [141] has recently been demonstrated and is discussed in Chap. 4.
Recently, similarly to the elemental copper and copper alloys, also non-porous hard surfaces containing copper oxide particles (Fig. 5.4) have been registered by the EPA as antimicrobial surfaces and allowed to make public health claims (EPA Registration number 84542–7). The approval is based on GLP testing demonstrating the ability to kill specific disease‐causing bacteria: MRSA, Staphylococcus aureus, Enterobacter aerogenes, P. aeruginosa and Escherichia coli (O157:H7). The product is approved for use in a wide range of applications, including health care. The samples were tested in various environmental conditions, cleaning protocols, and for efficacy after repeated exposure. The non-porous copper infused surfaces also underwent extensive American Society for Testing and Materials (ASTM) standard testing to support mechanical performance claims, allowing making efficacy claims that the copper‐oxide impregnated surfaces kill greater than 99.9 % of gram negative and gram positive bacteria within 2 h of exposure between routine cleaning and sanitizing steps and even after repeated exposure [142].
Copper oxide impregnated non-porous surfaces. (a) A nurse top station in which the non-porous countertops are impregnated with copper oxide particles. (b) A sink made with non-porous copper oxide impregnated material. (c) SEM picture of the non-porous material shown in (b), demonstrating the homogenous distribution of the copper oxide particles on the material surface. (d) An X-ray photoelectron spectrum analysis of a white dot particle shown in (c), demonstrating the presence of copper
Currently seven hospitals have already installed the non-porous copper infused surfaces – six in the USA and one in Israel, with the aim of further reducing the rates of HAI. The Sentara Leigh Hospital, which is one of the 11 acute care Sentara Hospitals in Virginia, has outfitted the copper-infused countertops, bed rails, and over the bed tables in their new 129 bed-tower in all patient care areas, including nursing units, visitor lounges, and patient rooms. In early 2014, the hospital also introduced copper-infused hospital gowns, pillowcases, and towels in the new tower. They will compare the rates of HAI, such as urinary catheter-associated and central line blood stream infections, with the infection rates in a similar existing 129-bed tower, in which no copper containing products will be utilized.
In conclusion, the introduction of soft and hard surfaces containing biocidal copper oxide particles in clinical settings may be an important adjunct for the reduction of bioburden and HAI. Furthermore, as HAI are now spreading out from the hospital environment into the community, the use of biocidal textiles, such as those impregnated with copper oxide, and hard surfaces containing a high percentage of copper, may not only significantly contribute to the reduction of HAI, but may also confer protection when used in homes for the elderly and in other environments where immune compromised individuals are at high risk of contracting infections.
Abbreviations
- AATCC:
-
American Association of Textile Chemists and Colorists
- cfu:
-
Colony forming units
- EPA:
-
USA Environmental Protection Agency
- GLP:
-
Good laboratory practices
- HAI:
-
Healthcare-acquired infections
- HD:
-
Hospitalization days
- MRSA:
-
Methicillin-resistant Staphylococcus aureus
- SEM:
-
Scanning electronic microscope
- TM:
-
Test Method
- VRE:
-
Vancomycin-resistant enterococcus
References
Noble WA (1981) Dispersal of microorganisms from skin. In: Microbiology of human skin, 2nd edn. Lloyd-Luke Ltd, London, pp 77–85
Coronel D et al (2001) Infection et contamination bacterienne de l’environnement des patients: les draps. Reanimation 10S:43–44
Beggs CB (2003) The airborne transmission of infection in hospital buildings: fact or fiction? Indoor Built Environ 12:9–18
Gabbay J et al (2006) Copper oxide impregnated textiles with potent biocidal activities. J Ind Text 35(4):323–335
Nordstrom JM, Reynolds KA, Gerba CP (2012) Comparison of bacteria on new, disposable, laundered, and unlaundered hospital scrubs. Am J Infect Control 40(6):539–543
Fijan S, Sostar-Turk S, Cencic A (2005) Implementing hygiene monitoring systems in hospital laundries in order to reduce microbial contamination of hospital textiles. J Hosp Infect 61(1):30–38
Fijan S, Turk SS (2012) Hospital textiles, are they a possible vehicle for healthcare-associated infections? Int J Environ Res Public Health 9(9):3330–3343
Perry C, Marshall R, Jones E (2001) Bacterial contamination of uniforms. J Hosp Infect 48(3):238–241
Bureau-Chalot F, Piednoir E, Camus J, Bajolet O (2004) Microbiologic quality of linen and linen rooms in short-term care units. J Hosp Infect 56(4):329–331
Hota B (2004) Contamination, disinfection, and cross-colonization: are hospital surfaces reservoirs for nosocomial infection? Clin Infect Dis 39(8):1182–1189
Boyce JM, Potter-Bynoe G, Chenevert C, King T (1997) Environmental contamination due to methicillin-resistant Staphylococcus aureus: possible infection control implications. Infect Control Hosp Epidemiol 18(9):622–627
Gould FK, Freeman R (1993) Nosocomial infection with microsphere beds. Lancet 342(8865):241–242
Kniehl E, Becker A, Forster DH (2005) Bed, bath and beyond: pitfalls in prompt eradication of methicillin-resistant Staphylococcus aureus carrier status in healthcare workers. J Hosp Infect 59(3):180–187
Burden M et al (2011) Newly cleaned physician uniforms and infrequently washed white coats have similar rates of bacterial contamination after an 8-hour workday: a randomized controlled trial. J Hosp Med 6(4):177–182
Babb JR, Davies JG, Ayliffe GA (1983) Contamination of protective clothing and nurses’ uniforms in an isolation ward. J Hosp Infect 4(2):149–157
Malnick S, Bardenstein R, Huszar M, Gabbay J, Borkow G (2008) Pyjamas and sheets as a potential source of nosocomial pathogens. J Hosp Infect 70(1):89–92
Reiss-Levy E, McAllister E (1979) Pillows spread methicillin-resistant staphylococci. Med J Aust 1(3):92
Wiener-Well Y et al (2011) Nursing and physician attire as possible source of nosocomial infections. Am J Infect Control 39(7):555–559
Noskin GA, Bednarz P, Suriano T, Reiner S, Peterson LR (2000) Persistent contamination of fabric-covered furniture by vancomycin-resistant enterococci: implications for upholstery selection in hospitals. Am J Infect Control 28(4):311–313
Das I et al (2002) Carbapenem-resistant Acinetobacter and role of curtains in an outbreak in intensive care units. J Hosp Infect 50(2):110–114
Ohl M et al (2012) Hospital privacy curtains are frequently and rapidly contaminated with potentially pathogenic bacteria. Am J Infect Control 40(10):904–906
Trillis F III, Eckstein EC, Budavich R, Pultz MJ, Donskey CJ (2008) Contamination of hospital curtains with healthcare-associated pathogens. Infect Control Hosp Epidemiol 29(11):1074–1076
Palmer R (1999) Bacterial contamination of curtains in clinical areas. Nurs Stand 14(2):33–35
Morgan DJ et al (2012) Transfer of multidrug-resistant bacteria to healthcare workers’ gloves and gowns after patient contact increases with environmental contamination. Crit Care Med 40(4):1045–1051
Morgan DJ et al (2010) Frequent multidrug-resistant Acinetobacter baumannii contamination of gloves, gowns, and hands of healthcare workers. Infect Control Hosp Epidemiol 31(7):716–721
Treakle AM et al (2009) Bacterial contamination of health care workers’ white coats. Am J Infect Control 37(2):101–105
Osawa K et al (2003) Significance of methicillin-resistant Staphylococcus aureus (MRSA) survey in a university teaching hospital. J Infect Chemother 9(2):172–177
Pilonetto M et al (2004) Hospital gowns as a vehicle for bacterial dissemination in an intensive care unit. Braz J Infect Dis 8(3):206–210
Traore O, Springthorpe VS, Sattar SA (2002) A quantitative study of the survival of two species of Candida on porous and non-porous environmental surfaces and hands. J Appl Microbiol 92(3):549–555
Neely AN, Maley MP (2000) Survival of enterococci and staphylococci on hospital fabrics and plastic. J Clin Microbiol 38(2):724–726
Neely AN (2000) A survey of gram-negative bacteria survival on hospital fabrics and plastics. J Burn Care Rehabil 21(6):523–527
Neely AN, Orloff MM (2001) Survival of some medically important fungi on hospital fabrics and plastics. J Clin Microbiol 39(9):3360–3361
Dixon GJ, Sidwell RW, McNeil E (1966) Quantitative studies on fabrics as disseminators of viruses. II. Persistence of poliomyelitis virus on cotton and wool fabrics. Appl Microbiol 14(2):183–188
Coughenour C, Stevens V, Stetzenbach LD (2011) An evaluation of methicillin-resistant Staphylococcus aureus survival on five environmental surfaces. Microb Drug Resist 17(3):457–461
Huang R, Mehta S, Weed D, Price CS (2006) Methicillin-resistant Staphylococcus aureus survival on hospital fomites. Infect Control Hosp Epidemiol 27(11):1267–1269
Lankford MG et al (2006) Assessment of materials commonly utilized in health care: implications for bacterial survival and transmission. Am J Infect Control 34(5):258–263
Brunton WA (1995) Infection and hospital laundry. Lancet 345(8964):1574–1575
Orr KE, Holliday MG, Jones AL, Robson I, Perry JD (2002) Survival of enterococci during hospital laundry processing. J Hosp Infect 50(2):133–139
Hellickson LA, Owens KL (2007) Cross-contamination of Clostridium difficile spores on bed linen during laundering. Am J Infect Control 35:E32–E33
Gerhardts A, Mucha H, Hofer D (2012) Testing linen disinfection procedures in practice with phage-charged-bioindicators. Int J Health Care Qual Assur 25(6):519–531
Hofer D (2006) Antimicrobial textiles, skin-borne flora and odour. Curr Probl Dermatol 33:67–77
Borkow G, Gabbay J (2008) Biocidal textiles can help fight nosocomial infections. Med Hypotheses 70(5):990–994
Kropec A et al (1993) Exogenous or endogenous reservoirs of nosocomial Pseudomonas aeruginosa and Staphylococcus aureus infections in a surgical intensive care unit. Intensive Care Med 19(3):161–165
CDC (2007) Fact sheet. http://www.cdc.gov/ncidod/hip/aresist/ca_mrsa_public.htm
Creamer E, Humphreys H (2008) The contribution of beds to healthcare-associated infection: the importance of adequate decontamination. J Hosp Infect 69(1):8–23
Shiomori T et al (2002) Evaluation of bedmaking-related airborne and surface methicillin-resistant Staphylococcus aureus contamination. J Hosp Infect 50(1):30–35
Sexton T, Clarke P, O’Neill E, Dillane T, Humphreys H (2006) Environmental reservoirs of methicillin-resistant Staphylococcus aureus in isolation rooms: correlation with patient isolates and implications for hospital hygiene. J Hosp Infect 62(2):187–194
Bonten MJ et al (1996) Epidemiology of colonisation of patients and environment with vancomycin-resistant enterococci. Lancet 348(9042):1615–1619
Munoz-Price LS et al (2012) Associations between bacterial contamination of health care workers’ hands and contamination of white coats and scrubs. Am J Infect Control 40(9):e245–e248
Panagea S, Winstanley C, Walshaw MJ, Ledson MJ, Hart CA (2005) Environmental contamination with an epidemic strain of Pseudomonas aeruginosa in a Liverpool cystic fibrosis centre, and study of its survival on dry surfaces. J Hosp Infect 59(2):102–107
Barrie D, Wilson JA, Hoffman PN, Kramer JM (1992) Bacillus cereus meningitis in two neurosurgical patients: an investigation into the source of the organism. J Infect 25(3):291–297
Barrie D, Hoffman PN, Wilson JA, Kramer JM (1994) Contamination of hospital linen by Bacillus cereus. Epidemiol Infect 113(2):297–306
Birch BR et al (1981) Bacillus cereus cross-infection in a maternity-unit. J Hosp Infect 2(4):349–354
Sasahara T et al (2011) Bacillus cereus bacteremia outbreak due to contaminated hospital linens. Eur J Clin Microbiol Infect Dis 30(2):219–226
Dohmae S et al (2008) Bacillus cereus nosocomial infection from reused towels in Japan. J Hosp Infect 69(4):361–367
Reif JS, Wimmer L, Smith JA, Dargatz DA, Cheney JM (1989) Human cryptosporidiosis associated with an epizootic in calves. Am J Public Health 79(11):1528–1530
Shah PC, Krajden S, Kane J, Summerbell RC (1988) Tinea corporis caused by Microsporum canis: report of a nosocomial outbreak. Eur J Epidemiol 4(1):33–38
Gustafson TL, Kobylik B, Hutcheson RH, Schaffner W (1983) Protective effect of anticholinergic drugs and psyllium in a nosocomial outbreak of Norwalk gastroenteritis. J Hosp Infect 4(4):367–374
Duffy J et al (2010) Zygomycosis outbreak associated with hospital linens. In Fifth decennial international conference on healthcare associated infections, Atlanta, USA
Standaert SM, Hutcheson RH, Schaffner W (1994) Nosocomial transmission of Salmonella gastroenteritis to laundry workers in a nursing home. Infect Control Hosp Epidemiol 15(1):22–26
Datta N, Pridie RB (1960) An outbreak of infection with Salmonella typhimurium in a general hospital. J Hyg (Lond) 58:229–240
Thomas MC, Giedinghagen DH, Hoff GL (1987) An outbreak of scabies among employees in a hospital-associated commercial laundry. Infect Control 8(10):427–429
Takahashi A et al (1998) Streptococcus pyogenes hospital-acquired infection within a dermatological ward. J Hosp Infect 40(2):135–140
Sidwell RW, Dixon GJ, Westbrook L, Forziati FH (1970) Quantitative studies on fabrics as disseminators of viruses. IV. Virus transmission by dry contact of fabrics. Appl Microbiol 19(6):950–954
Sidwell RW, Dixon GJ, McNeil E (1967) Quantitative studies on fabrics as disseminators of viruses. 3. Persistence of vaccinia virus on fabrics impregnated with a virucidal agent. Appl Microbiol 15(4):921–927
Borg MA, Portelli A (1999) Hospital laundry workers–an at-risk group for hepatitis A? Occup Med (Lond) 49(7):448–450
Keeffe EB (2004) Occupational risk for hepatitis A: a literature-based analysis. J Clin Gastroenterol 38(5):440–448
Cozad A, Jones RD (2003) Disinfection and the prevention of infectious disease. Am J Infect Control 31(4):243–254
Sattar SA et al (2001) Transfer of bacteria from fabrics to hands and other fabrics: development and application of a quantitative method using Staphylococcus aureus as a model. J Appl Microbiol 90(6):962–970
Greene VW, Bond RG, Michaelsen GS (1960) Air handling systems must be planned to reduce the spread of infection. Mod Hosp 95:136–144
SOLBERG CO (1965) A study of carriers of Staphylococcus aureus with special regard to quantitative bacterial estimations. Acta Med Scand Suppl 436:1–96
Noble WC, Davies RR (1965) Studies on the dispersal of staphylococci. J Clin Pathol 18:16–19
Gastmeier P et al (2006) Where should one search when confronted with outbreaks of nosocomial infection? Am J Infect Control 34(9):603–605
Bernards AT et al (1998) Methicillin-resistant Staphylococcus aureus and Acinetobacter baumannii: an unexpected difference in epidemiologic behavior. Am J Infect Control 26(6):544–551
Allen KD, Green HT (1987) Hospital outbreak of multi-resistant Acinetobacter anitratus: an airborne mode of spread? J Hosp Infect 9(2):110–119
Cox RA, Conquest C, Mallaghan C, Marples RR (1995) A major outbreak of methicillin-resistant Staphylococcus aureus caused by a new phage-type (EMRSA-16). J Hosp Infect 29(2):87–106
Walter CW, Kundsin RB, Brubaker MM (1963) The incidence of airborne wound infection during operation. JAMA 186:908–913
Farrington M, Ling J, Ling T, French GL (1990) Outbreaks of infection with methicillin-resistant Staphylococcus aureus on neonatal and burns units of a new hospital. Epidemiol Infect 105(2):215–228
Rutula WA (1983) Environmental study of a methicillin-resistant Staphylococcus aureus epidemic in a burn unit. J Clin Microbiol 18:683–688
Lidwell OM et al (1982) Effect of ultraclean air in operating rooms on deep sepsis in the joint after total hip or knee replacement: a randomised study. Br Med J (Clin Res Ed) 285(6334):10–14
Mortimer EA Jr, Wolinsky E, Gonzaga AJ, Rammelkamp CH Jr (1966) Role of airborne transmission in staphylococcal infections. Br Med J 1(5483):319–322
Walter EA, Bowden RA (1995) Infection in the bone marrow transplant recipient. Infect Dis Clin N Am 9(4):823–847
Nicas M, Sun G (2006) An integrated model of infection risk in a health-care environment. Risk Anal 26(4):1085–1096
Giannossa LC et al (2013) Metal nanoantimicrobials for textile applications. Nanotechnol Rev 2(3):307–331
Papaspyrides CD, Pavlidou S, Vouyiouka SN (2009) Development of advanced textile materials: natural fibre composites, anti-microbial, and flame-retardant fabrics. Proc Inst Mech Eng Part L J Mater Des Appl 223(2):91–102
Coman D, Oancea S, Vrinceanu N (2010) Biofunctionalization of textile materials by antimicrobial treatments: a critical overview. Rom Biotechnol Lett 15(1):4913–4921
Windler L, Height M, Nowack B (2013) Comparative evaluation of antimicrobials for textile applications. Environ Int 53:62–73
Yazdankhah SP et al (2006) Triclosan and antimicrobial resistance in bacteria: an overview. Microb Drug Resist 12(2):83–90
Kalyon BD, Olgun U (2001) Antibacterial efficacy of triclosan-incorporated polymers. Am J Infect Control 29(2):124–125
Bearman GM et al (2012) A crossover trial of antimicrobial scrubs to reduce methicillin-resistant Staphylococcus aureus burden on healthcare worker apparel. Infect Control Hosp Epidemiol 33(3):268–275
Renaud FNR, Dore J, Freney HJ, Coronel B, Dusseau JY (2006) Evaluation of antibacterial properties of a textile product with antimicrobial finish in a hospital environment. J Ind Text 36(1):89–94
Taylor L, Phillips P, Hastings R (2009) Reduction of bacterial contamination in a healthcare environment by silver antimicrobial technology. J Infect Control 10(1):6–12
White WC, Bellfield R, Ellis J, Vandendaele IP (2007) Controlling the spread of nosocomial infections in hospital wards by the use of antimicrobials on textiles, facilities and devices. In Medtex 07 – fourth international conference and exhibition on healthcare and medical textiles, Bolton, UK
Schweizer M et al (2012) Novel hospital curtains with antimicrobial properties: a randomized, controlled trial. Infect Control Hosp Epidemiol 33(11):1081–1085
Gross R, Hubner N, Assadian O, Jibson B, Kramer A (2010) Pilot study on the microbial contamination of conventional vs. silver-impregnated uniforms worn by ambulance personnel during one week of emergency medical service. GMS Krankenhhyg Interdiszip 5(2):25–41
Burden M et al (2013) Bacterial contamination of healthcare workers’ uniforms: a randomized controlled trial of antimicrobial scrubs. J Hosp Med 8(7):380–385
Niiyama N et al (2013) Use of copper alloy for preventing transmission of methicillin-resistant Staphylococcus aureus contamination in the dermatology ward. Acta Derm Venereol 93(3):294–300
The Hohenstein Institute (2014) Scientists prove benefit of textiles with antiviral and antibacterial effect. Textile World. http://textileworld.com/Articles/2014/January/Scientists_Prove_Benefit_Of_Textiles_With_Antiviral_And_Antibacterial_Effect
Carter Y, Barry D (2011) Tackling C difficile with environmental cleaning. Nurs Times 107(36):22–25
Chatterjee SS, Otto M (2013) Improved understanding of factors driving methicillin-resistant Staphylococcus aureus epidemic waves. Clin Epidemiol 5:205–217
El-Nahhal IM et al (2013) Nanostructured copper oxide-cotton fibers: synthesis, characterization, and applications. Int Nano Lett 2:14
Priniotakis G, Chronis I, Tzerachoglou A, Souli M, Westbroek P (2013) Copper coated textiles with potent antimicrobial activity against multidrug-resistant nosocomial pathogens, 13th AUTEX World Textile Conference, Dresden, Germany
Sharifahmadian O, Salimijazi HR, Fathi MH, Mostaghimi J, Pershin L (2012) Study of the antibacterial behavior of wire arc sprayed copper coatings. J Therm Spray Technol 22(2–3):371–379. doi:10.1007/s11666-012-9842-2
Singh G, Beddow JA, Joyce E, Mason T (2013) Production and efficacy testing of antimicrobial fabrics for use in hospitals. Antimicrobial Resistance and Infection Control 2(1): 184.
Perelshtein I et al (2009) CuO-cotton nanoparticles: formation, morphology and antibacterial activity. Surf Coat Technol 204:54
Rio L et al (2012) Comparison of methods for evaluation of the bactericidal activity of copper-sputtered surfaces against methicillin-resistant Staphylococcus aureus. Appl Environ Microbiol 78(23):8176–8182
Grace M, Chand N, Bajpai SK (2009) Copper alginate-cotton cellulose (CACC) fibers with excellent antibacterial properties. J Eng Fibers Fabr 4(3):24–35
Borkow G, Gabbay J (2004) Putting copper into action: copper-impregnated products with potent biocidal activities. FASEB J 18(14):1728–1730
Borkow G, Gabbay J (2006) Endowing textiles with permanent potent biocidal properties by impregnating them with copper oxide. JTATM 5(1):1–3
Borkow G, Okon-Levy N, Gabbay J (2010) Copper oxide impregnated wound dressings: biocidal and safety studies. Wounds 22:310–316
Anita S, Ramachandran T, Rajendran R, Koushik CV, Mahalakshmi M (2011) A study of the antimicrobial property of encapsulated copper oxide nanoparticles on cotton fabric. Text Res J 81:1081–1088
Osorio-Vargas P et al (2011) Antimicrobial Cu-functionalized surfaces prepared by bipolar asymmetric DC-pulsed magnetron sputtering (DCP). J Photochem Photobiol A 220:70–76
Chattopadhyay DP, Patel BH (2010) Effect of nanosized colloidal copper on cotton fabric. J Eng Fibers Fabr 5:1–6
Grace M, Bajpai SK, Chand N (2009) Copper (II) ions and copper nanoparticles-loaded chemically modified cotton cellulose fibers with fair antibacterial properties. J Appl Polym Sci 11:757–766
Berendjchi A, Khajavi R, Yazdanshenas MS (2011) Fabrication of superhydrophobic and antibacterial surface on cotton fabric by doped silica-based sols with nanoparticles of copper. Nanoscale Res Lett 6:594–602
Rtimi S, Baghriche O, Pulgarin C, Lavanchy CJ, Kiwi J (2013) Growth of TiO2/Cu films by HiPIMS for accelerated bacterial loss of viability. Surf Coat Technol 232:804–813
Borkow G, Gabbay J (2009) An ancient remedy returning to fight microbial, fungal and viral infections. Curr Chem Biol 3(3):272–278
Borkow G, Zatcoff RC, Gabbay J (2009) Reducing the risk of skin pathologies in diabetics by using copper impregnated socks. Med Hypotheses 73:883–886
Borkow G, Zhou SS, Page T, Gabbay J (2010) A novel anti-influenza copper oxide containing respiratory face mask. PLoS One 5(6):e11295
Borkow G (2012) Using copper to fight microorganisms. Curr Chem Biol 6(2):93–103
Borkow G, Mellibovsky JC (2012) Resolution of skin maladies of the trapped Chilean miners: the unplanned underground copper-impregnated antifungal socks “trial”. Arch Dermatol 148(1):134–136
Gargiulo ME, Del Carmen-Elias A, Borkow G (2012) Analysis of the effect of wearing copper oxide impregnated socks on tinea pedis based on “before and after” pictures – a statistical follow-up tool. Open Biol J 5:17–22
Zatcoff RC, Smith MS, Borkow G (2008) Treatment of tinea pedis with socks containing copper-oxide impregnated fibers. Foot (Edinb) 18(3):136–141
Borkow G, Gabbay J (2005) Copper as a biocidal tool. Curr Med Chem 12(18):2163–2175
Borkow G et al (2007) Neutralizing viruses in suspensions by copper oxide based filters. Antimicrob Agents Chemother 51(7):2605–2607
Borkow G, Lara HH, Covington CY, Nyamathi A, Gabbay J (2008) Deactivation of human immunodeficiency virus type 1 in medium by copper oxide-containing filters. Antimicrob Agents Chemother 52(2):518–525
Borkow G (2013) Protection of soldiers’ feet by copper oxide impregnated socks. Adv Mil Technol 8:101–108
Borkow G et al (2011) Prevention of human immunodeficiency virus breastmilk transmission with copper oxide: proof-of-concept study. Breastfeed Med 6:165–170
Mumcuoglu KY, Gabbay J, Borkow G (2008) Copper oxide impregnated fabrics for the control of house dust mites. Int J Pest Manag 54(3):235–240
Lazary A, Weinberg I, Vatine J-J, Jefidoff A, Bardenstein R, Borkow G, Ohana N (2014) Reduction of healthcare-associated infections in a long-term care brain injury ward by replacing regular linens with biocidal copper oxide impregnated linens. Int J Infect Dis 24:e23–e29. doi:10.1016/j.ijid.2014.01.022
Lazary A, Kupferman Y, Orly A, Lerman Y (2003) Survival of patients remaining in vegetative state. In: 2nd world congress of the international society of physical and rehabilitation medicine – ISPRM. Prague, Czech Republic
The Multi-Society Task Force on PVS (1994) Medical aspects of the persistent vegetative state (1). The Multi-Society Task Force on PVS. N Engl J Med 330(21):1499–1508
Wolach B et al (1993) Some aspects of the humoral and neutrophil functions in post-comatose nawareness patients. Brain Inj 7(5):401–410
Casey AL et al (2010) Role of copper in reducing hospital environment contamination. J Hosp Infect 74(1):72–77
Mikolay A et al (2010) Survival of bacteria on metallic copper surfaces in a hospital trial. Appl Microbiol Biotechnol 87(5):1875–1879
Marais F, Mehtar S, Chalkley L (2010) Antimicrobial efficacy of copper touch surfaces in reducing environmental bioburden in a South African community healthcare facility. J Hosp Infect 74(1):80–82
Schmidt MG et al (2013) Copper continuously limits the concentration of bacteria resident on bed rails within the intensive care unit. Infect Control Hosp Epidemiol 34(5):530–533
Schmidt MG et al (2012) Sustained reduction of microbial burden on common hospital surfaces through introduction of copper. J Clin Microbiol 50(7):2217–2223
Karpanen TJ et al (2012) The antimicrobial efficacy of copper alloy furnishing in the clinical environment: a crossover study. Infect Control Hosp Epidemiol 33(1):3–9
Vessey A (2013) Copper–a weapon in the war on pathogens. Health Estate 67(8):65–69
Salgado CD et al (2013) Copper surfaces reduce the rate of healthcare-acquired infections in the intensive care unit. Infect Control Hosp Epidemiol 34(5):479–486
Monk AB, Kanmukhla V, Trinder K, Borkow G (2014) Potent bactericidal efficacy of copper oxide impregnated non-porous solid surfaces. BMC Microbiol 14:57–71
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Borkow, G. (2014). Biocidal Hard and Soft Surfaces Containing Copper Oxide Particles for the Reduction of Healthcare-Acquired Pathogens. In: Borkow, G. (eds) Use of Biocidal Surfaces for Reduction of Healthcare Acquired Infections. Springer, Cham. https://doi.org/10.1007/978-3-319-08057-4_5
Download citation
DOI: https://doi.org/10.1007/978-3-319-08057-4_5
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-08056-7
Online ISBN: 978-3-319-08057-4
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)