Abstract
Although esophagectomy is an effective treatment of early esophageal cancer, due to high rates of morbidity and potential mortality, and also, importantly, the potentially reduced quality of life, endoscopic therapy represents a favorable approach where appropriate. Endoscopic resection therapies have been successfully applied to selected patients with early tumors of the esophagus with equivalent oncological outcomes. This chapter will address the selection of tumors appropriate for endoscopic resection and review the technical approaches and outcomes of the two main technical approaches, including Endoscopic Mucosal Resection (EMR) and Endoscopic Submucosal Dissection (ESD).
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
Introduction
Esophageal cancer has exponentially increased annually in the contemporary era, with a shift in anatomical and histological subtype from esophageal squamous cell carcinoma to adenocarcinoma in North America and Western Europe [1]. Although the overall 5-year relative survival rate is only approximately 20%, patients with early-stage disease have a better chance of survival, and cure rates of successfully treated early esophageal cancer approach 90%.
The conventional treatment of localized disease consists of esophagectomy, open initially and currently minimally invasively. Although traditional surgical resection is an effective treatment of early esophageal cancer, due to high rates of morbidity 50–60%, and potential mortality of 1–3%, and also, importantly, the potentially reduced quality of life, endoscopic therapy represents a favorable approach where appropriate [2]. Indeed, endoscopic resection therapies have been successfully applied to selected patients with early tumors of the esophagus with equivalent oncological outcomes. This chapter will address the selection of tumors appropriate for endoscopic resection and review the technical approaches and outcomes of the two main technical approaches, including Endoscopic Mucosal Resection (EMR) and Endoscopic Submucosal Dissection (ESD).
Selecting the Appropriate Tumors for Endoscopic Resection
To justify an organ-sparing endoscopic resection approach for cure, two main criteria must be met: (1) negligible rate of occult lymph node metastasis and (2) ability to resect the lesion completely en bloc with negative margins, particularly the deep margin. Staging investigations are often required to eliminate clinically apparent regional/distant disease, including cross-sectional imaging, endoscopic ultrasound, and PET scans.
There is an exponentially increased incidence of nodal disease with advancing T stage. Consequently, it is essential to appropriately stage the patient ab initio to delineate patients suitable for endotherapy versus those necessitating a formal resection. The esophageal wall comprises four layers, with the mucosa being the most superficial layer. The mucosa encompasses the epithelium, lamina propria, and muscularis mucosa. Immediately beyond is the submucosa, which is made of connective tissue, including blood vessels, lymphatics, and Meissner’s plexus. The submucosa connects the mucosa to the muscularis propria, made of inner circular and outer longitudinal muscle layers along with the Auerbach plexus. The deepest level comprised of connective tissue is called the adventitia.
The incidence of nodal metastases directly correlates to the subclassification of T1a and T1b disease, with invasion confined to the epithelium (m1), laminal propria (m2), or muscularis propria (m3) for T1a disease, and lesions infiltrating the submucosa (T1b) can be further categorized into sm1 (inner third), sm2 (middle third) and sm3 (outer third) (Table 1 and Fig. 1).
For decades, oncological surgical resection was the conventional curative therapy for malignancies of the esophagus. Endoscopic therapy of early carcinomas is now increasingly established as the gold standard, with the main challenge still to diagnose and carry out endoscopic therapy in good time before metastasis. It is also important to note that tumors over 2 cm with adverse features of tumor biology, such as poor differentiation, have higher incidences of metastases, with late diagnosis still common due to the indolent nature of the disease. Some scoring algorithms have been devised as simple metrics to predict those at risk of lymph node metastasis (LNM) to aid decision-making in patients with T1 esophageal adenocarcinoma undergoing endoscopic resection [3].
Diagnostic and Staging Work-Up
To facilitate patient stratification, pre-treatment evaluation of esophageal cancer consists of locoregional staging and evaluation for distant metastases. Locoregional staging assesses both the degree of extension of the tumor into the esophageal wall and the nodal status. As already alluded to, as tumor depth progresses, so does the risk of LNM. From older surgical series, the rate of LNM in treatment-naive resected esophageal adenocarcinoma ranges between 0 and 7%. Most series have a clinically negligible rate of regional disease on pathology for pathology T1a tumors [4]. In comparison, this risk increases to up to 27% for T1b adenocarcinoma.
The Society of Thoracic Surgeons has published guidelines on staging patients with esophageal cancer [5]. Fluorodeoxyglucose (FDG) positron-emission tomography (PET) scans can improve overall staging by detecting metastatic disease in up to 15–20% of patients and should complement conventional CT imaging to identify metastatic disease [6]. In addition, FDG-PET has also been shown to have prognostic value that can be applied to patient management and aid in developing emerging therapies.
EUS aids locoregional staging and can guide treatment planning of esophageal cancer in the absence of distant metastases. In early esophageal cancer, T staging may help select appropriate cases for minimally invasive treatment using EMR and ESD techniques. Puli and colleagues reported the sensitivity and specificity of EUS for staging esophageal cancer to be 81.6 and 99.4% in T1 tumors [7]. The specificity and the sensitivity for identifying lymph node disease are better when EUS is combined with fine-needle aspiration (FNA) or fine needle biopsy (FNB) compared to EUS alone [8].
There are occasions where additional studies may be worthwhile before therapies are initiated, such as bronchoscopy considered for tumors in the upper and middle esophagus to rule out airway invasion. Although staging laparoscopy has been suggested for locally advanced esophagogastric junction adenocarcinoma, in early esophageal disease, the risk of peritoneal involvement is nominal, and thus this diagnostic test can be avoided.
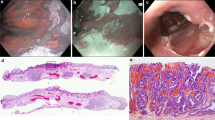
Chromoendoscopy and advanced endoscopic imaging techniques have the potential to improve the workup of patients with early esophageal neoplastic lesions. It involves using dye stains such as Lugol’s iodine for squamous cell cancer, acetic acid for Barrett’s esophagus, and Narrow Band Imaging (NBI), enabling detailed lesion assessment and extent (Figs. 2 and 3).
Lugol’s solution highlights glycogen-rich nonkeratinized squamous epithelium. An abnormal staining pattern is present in conditions that deplete glycogen in squamous cells, including dysplastic and early neoplastic lesions. Lugol’s iodine has a sensitivity of 91–100% and a specificity of 40–95% for detecting squamous neoplasia in the esophagus.
Acetic acid improves the diagnostic yield for identifying Barrett’s esophagus specialized columnar epithelium in comparison to random biopsies (57% vs. 26%, P = 0.12) [9, 10].
NBI has been combined with magnification endoscopy to predict the invasion depth of superficial squamous cell cancer [11]. Lugol’s chromoendoscopy is a simple technique in which the liquid is sprayed into the mucosa through an injection spray catheter via the endoscope’s working channel. The iodine-potassium iodide forms a dark brown complex with the physiological squamous epithelium, which allows the delineation of dysplastic carcinoma cells that do not stain, and this helps with the sometimes-difficult differentiation from healthy tissue, which is essential for R0 resection. NBI can also enhance the mucosa and the underlying vascular pattern, providing greater convenience for evaluation.
Endoscopic Mucosal Resection (EMR)
Endoscopic mucosal resection (EMR) is a technique that was originally pioneered and introduced in 1978 in Japan for early gastric cancer [12]. In the contemporary era, endoscopic techniques have replaced the traditional process of esophagectomy as the preferred treatment modality for high-grade dysplastic Barrett’s, intramucosal cancer [13], and in cases of early invasive cancer, with the decision influenced by the risk of LNM.
“Early carcinoma of the esophagus” is not uniformly defined and refers in the following to in situ and T1 tumors in non-metastatic disease. Notwithstanding higher cure rates are achieved with esophagectomy, the decision on treatment strategy must be borne in mind with the significant treatment-related morbidity and mortality, even in very experienced high volume tertiary referral centers, between 2 and 7% and up to 20% in others [14, 15]. In addition to this, high-risk cardiovascular patients may not be able to tolerate the anesthesia or esophagectomy itself, leaving very few options available for these patients.
Curative endoscopic resection for mucosal carcinomas is possible in adenocarcinoma, analogous to early squamous cell carcinoma. Consequently, endoscopic resection has evolved to become the first-line therapy for managing superficial early esophageal neoplasia. The term superficial, however, is in some ways confusing, because it is not directly related to histology or invasiveness of a GI cancer but simply describes the endoscopic appearance of a lesion, which looks to be restricted to superficial layers of the GI tract. EMR is an established simpler and faster technique compared to endoscopic submucosal dissection (ESD) but is limited by its inability to resect large lesions en bloc. In essence, piecemeal EMR of large lesions includes a high rate of recurrence and arduous tissue specimen histologic evaluation for accurate staging and margin assessment for R0 resections.
There is a significant risk of leaving disease behind with up to 30% local recurrence with EMR of lesions > 1 cm [16]. Alternatively, ESD, on the other hand, is technically more complex, and traditionally has been associated with a higher rate of adverse events but facilitates en bloc resection regardless of lesion size, reducing risk for recurrence and facilitating precise histologic staging. Ultimately, the optimal endoscopic technique should be selected based on organ location, type of neoplastic lesion, and local expertise. The role of ESD has expanded in Eastern regions beyond squamous cell lesions in the esophagus and gastric cancer to include superficial Barrett’s esophagus (BE). However, there is controversy in Western regions over the use of ESD for BE. Thus, focusing on practical considerations for formulating the most appropriate endoscopic resection approach for each patient must be applied.
Although there are several approaches for EMR, we prefer the band ligation technique (Fig. 4). After a lesion is sucked into the overtube, a rubber band is released to form a pseudopolyp. Once the ligation device is detached, the pseudopolyp is removed at the base with a diathermy snare under or above the rubber band. The standard multi-bander ligation devices necessitate the removal of the endoscope to disassemble the ligation device and reintroduce the endoscope to remove the pseudopolyp with a standard polypectomy snare. Although some endoscopists recommend a prior submucosal injection to facilitate elevation of the mucosa [17], we have found that this is not necessary in the vast majority of cases and do not perform this technical step routinely. The specimen is then fixed for the pathologist. For larger lesions, several sequential EMRs are required, complicating accurate pathological assessment of mucosal margin status.
The recommendation for endoscopic resection for m1 to m3 adenocarcinoma remains steadfast and is made based on patient factors and the risk of metastatic disease with high-risk disease or poor prognostic factors such as adverse biology [17, 18]. In the case of sm1 carcinomas, a deep infiltration of < 500 μm is required for endoscopic resection, otherwise, there is a borderline case between surgical and endoscopic therapy [19]. The Japan Esophageal Society provides primary endoscopic resection for m1/m2 carcinomas. In the case of m3 carcinomas, the general condition essentially determines whether surgical, endoscopic, or radio/chemotherapy is indicated. The recommendations apply regardless of the histopathological typing/grading [20].
Endoscopic Submucosal Dissection (ESD)
Endoscopic submucosal dissection (ESD) was developed in Japan as an alternative to the limitations of the well-established modality of EMR for the curative treatment for early GI cancer facilitating en bloc resection and, therefore, histopathologic assessment [21].
The European Society of Gastrointestinal Endoscopy (ESGE) guidelines which have been updated as of June 2022 advocate ESD as the first option for superficial esophageal squamous cell carcinoma (SCC) with a series of 15 studies on ESD for superficial SCC finding en bloc resection rates of 83–100%, complete resection rates of 78–100%, and low rates of local recurrence of 0–2.6% [22]. Consequently, ESD is the preferred treatment modality for M1 (intraepithelial) and M2 (invasion into the lamina propria) disease.
To ensure the correct patient cohort is selected, tumor morphology should be employed to predict tumor depth. Paris classification lesions 0-IIa, 0-IIb, and 0-IIc are typically intramucosal and the Japan Esophageal Society guidelines have advocated Paris 0-II lesions with m1–m2 invasion and < 2/3 circumferential extent as absolute indications for endoscopic resection. The rationale for this recommendation is based on the negligible risk of LNM for M1 and M2 disease as well as the morbidity and mortality of esophagectomy.
The role of ESD in Barrett’s esophagus-related adenocarcinoma is limited due to the high efficacy of EMR. Endoscopic ablation of Barrett’s esophagus is a gold standard treatment for patients with high-grade dysplasia in the absence of visible lesions. Endoscopic resection is the treatment of choice for patients with visible or flat neoplasia. EMR is a technically limited resection as it is piecemeal resection which hinders determination of negative margins. A systematic review compared outcomes of ESD and EMR and there were no significant differences in local recurrence rates, positive margins, lymph node positivity, complications, or patients requiring surgery [23, 24].
ESD is performed with a standard, single accessory-channel endoscope. Carbon dioxide is used for insufflation. Special equipment necessary for ESD is a transparent cap, submucosal injection needle and solutions, ESD knives, coagulation devices, and endoclips. Typical ESD is accomplished in a stepwise manner, including marking the lesion, incision and submucosal dissection with simultaneous hemostasis. Pre-resection definition of the border of esophageal neoplasms is essential to avoid compromising the margins. Chromoendoscopy or NBI, as illustrated above, can be useful for pre-procedural assessment. An argon plasma coagulation (APC) or ESD knife using a soft coagulation current can be applied to mark the resection borders with dots around the lesion (Fig. 5). This should be at least 1 mm away from the margin of the invasive component of the tumor. If resecting early cancer within a field of Barrett’s esophagus, it is not necessary to resect all of the flat columnar mucosa, which can be addressed by subsequent ablative therapies at a separate setting.
After the resection borders are marked, an injectable liquid can be injected beneath the mucosa and submucosa off of the muscularis propria by an injection needle through the endoscopic channel to create a cushion. Although several elevating liquids have been proposed, such as hyaluronic acid, we have generally used cheaper options, such as glycerol 10% or a readily available intravenous starched-based volume expander (Fig. 6).
A circumferential incision is made along the marginal markings. For esophageal ESDs we usually mark the distal extent of the lesion first to define the end point (Fig. 7), followed by the proximal incision (Fig. 8). Then, the mucosal incision is imitated with an uncovered needle knife or a short blunt-tip knife prior to generating a submucosal tunnel.
Starting at the proximal cut, we begin a submucosal tunnel to cut the distal mucosal margin (Fig. 9). The entire lesion is stripped or peeled from the muscularis propria by ESD knives in the submucosal space. During this step, the submucosal injection needle and ESD knives are used interchangeably to lift the lesion and dissect the submucosal tissue (Fig. 10).
EMR/ESD Complications
Bleeding
A potential complication of ESD is bleeding, which can occur during the procedure or as a delayed phenomenon. Therefore, meticulous and prophylactic coagulation and prompt identification and treatment are essential for successful patient outcomes, with various tools available to do so. All ESD knives have at least a limited capacity for hemostasis on small bleeding points. The ball tip knives typically have improved hemostatic efficiency compared with standard needle knives [25]. Hybrid knives have been demonstrated to reduce the need for hemostatic devices and regular hemostasis compared with conventional knives [26]. It is important to balance the use of these coagulation devices with potential thermal injury.
For larger vessels and associated bleeds, specific hemostatic devices may be necessary. The Coagrasper (Olympus, Tokyo, Japan) is a monopolar forceps which is commonly used during ESD. Clips are not widely used for bleeding intraprocedural as they can interfere with continued dissection. They are thus reserved for uncontrolled bleeding or at the end of dissection.
Perforation
There is also an increased risk of perforation with ESD compared to EMR. Risk estimates range from 0 to 6.9% in esophageal ESD, and 2.4 to 9.6% in gastric ESD [27, 28]. Perforation may be visualized as a definite defect during the procedure or delayed after the procedure is diagnosed clinically or on radiograph or CT demonstrating pneumoperitoneum.
The use of conservative decompression with nasogastric or nasoduodenal tubes to divert gastrointestinal fluid and nutritional support during healing post-repair is also advocated for the successful management of these complications. A chest drain may also be warranted for pneumothoraxes or defects in the pleura resulting in effusions. Small defects of approximately 1 cm can generally be treated by through the scope clips, while over-the-scope clips may be considered for holes less than 2 cm [29], and fully covered self-expanding metallic stents are always a salvage option.
Strictures
Strictures are common after any esophageal resection, with the phenomenon more frequently encountered post esophageal ESD than other GI tract areas. Esophageal strictures occur in patients who undergo more than a 75% circumference ESD resection of the esophagus. The multimodal treatment of strictures includes one or more sessions of endoscopic balloon dilatation which may be combined with local injection of steroids (triamcinolone, betamethasone) or implantation of a temporal esophageal stent.
Specimen Processing and Histological Evaluation
After specimen retrieval, it is placed on the cork board and orientated. After fixation and sectioning, it is essential to critically assess pathology for (a) mucosal and deep margins, and (b) histological features predicting occult lymph node metastasis to provide a consistent and accurate diagnosis (Fig. 11).
Several factors, such as maintenance of proper orientation, meticulous macroscopic examination, accurate mapping of the lesion, and appropriate morphologic diagnosis are the main concerns. It is important to mention that the maintenance of orientation is crucial in slicing, histological analysis, and reporting. The factors relevant to prognosis and further treatment decisions, include histologic type, the size of the lesion, depth of invasion, association conditions (ulcer/scar), lymphovascular/venous invasion, and cut margin status (horizontal and vertical), which should always be carefully evaluated and reported.
After careful analysis by an appropriately experienced pathologist and discussion at the tumor board, any cases with positive margins should be considered for an esophagectomy [30]. There is no robust data available on surveillance endoscopy for such cases [31, 32]. However, mitigating circumstances such as those who are heavily co-morbid may be more appropriately managed with a watch and wait approach.
Conclusions
Endoscopic resection of early esophageal cancer is a feasible and safe treatment strategy in appropriately selected early esophageal lesions. EMR and ESD are acceptable endoscopic treatment modalities for these early esophageal cancer lesions. ESD requires technical expertise but is associated with higher rates of en bloc, R0, and curative resections and lower recurrence rates compared to EMR. It is paramount to ensure sufficient training and institutional support for obtaining safe and high-quality resections. Ultimately, the choice of resection technique is made individually and not least depending on the circumstances of the treating center. Hybrid techniques are being evaluated to provide an optimal approach to combine the advantages of ESD and EMR.
References
Brown LM, Devesa SS, Chow WH. Incidence of adenocarcinoma of the esophagus among white Americans by sex, stage, and age. J Natl Cancer Inst. 2008;100(16):1184–7.
Zheng H, Kang N, Huang Y, Zhao Y, Zhang R. Endoscopic resection versus esophagectomy for early esophageal cancer: a meta-analysis. Transl Cancer Res. 2021;10(6):2653–62.
Lee L, Ronellenfitsch U, Hofstetter WL, Darling G, Gaiser T, Lippert C, et al. Predicting lymph node metastases in early esophageal adenocarcinoma using a simple scoring system. J Am Coll Surg. 2013;217(2):191–9.
Barbour AP, Jones M, Brown I, Gotley DC, Martin I, Thomas J, et al. Risk stratification for early esophageal adenocarcinoma: analysis of lymphatic spread and prognostic factors. Ann Surg Oncol. 2010;17(9):2494–502.
Varghese TK Jr, Hofstetter WL, Rizk NP, Low DE, Darling GE, Watson TJ, et al. The society of thoracic surgeons guidelines on the diagnosis and staging of patients with esophageal cancer. Ann Thorac Surg. 2013;96(1):346–56.
Yang GY, Wagner TD, Jobe BA, Thomas CR. The role of positron emission tomography in esophageal cancer. Gastrointest Cancer Res. 2008;2(1):3–9.
Puli SR, Reddy JB, Bechtold ML, Antillon D, Ibdah JA, Antillon MR. Staging accuracy of esophageal cancer by endoscopic ultrasound: a meta-analysis and systematic review. World J Gastroenterol. 2008;14(10):1479–90.
Facciorusso A, Crinò SF, Muscatiello N, Gkolfakis P, Samanta J, Londoño Castillo J, et al. Endoscopic ultrasound fine-needle biopsy versus fine-needle aspiration for tissue sampling of abdominal lymph nodes: a propensity score matched multicenter comparative study. Cancers (Basel). 2021;13(17).
Dawsey SM, Fleischer DE, Wang GQ, Zhou B, Kidwell JA, Lu N, et al. Mucosal iodine staining improves endoscopic visualization of squamous dysplasia and squamous cell carcinoma of the esophagus in Linxian China. Cancer. 1998;83(2):220–31.
Hoffman A, Korczynski O, Tresch A, Hansen T, Rahman F, Goetz M, et al. Acetic acid compared with i-scan imaging for detecting Barrett’s esophagus: a randomized, comparative trial. Gastrointest Endosc. 2014;79(1):46–54.
Inoue H, Kaga M, Ikeda H, Sato C, Sato H, Minami H, et al. Magnification endoscopy in esophageal squamous cell carcinoma: a review of the intrapapillary capillary loop classification. Ann Gastroenterol. 2015;28(1):41–8.
Faivre J, Bory R, Moulinier B. Benign tumors of oesophagus: value of endoscopy. Endoscopy. 1978;10(4):264–8.
Westerterp M, Koppert LB, Buskens CJ, Tilanus HW, ten Kate FJ, Bergman JJ, et al. Outcome of surgical treatment for early adenocarcinoma of the esophagus or gastro-esophageal junction. Virchows Arch. 2005;446(5):497–504.
Birkmeyer JD, Siewers AE, Finlayson EV, Stukel TA, Lucas FL, Batista I, et al. Hospital volume and surgical mortality in the United States. N Engl J Med. 2002;346(15):1128–37.
Heitmiller RF, Redmond M, Hamilton SR. Barrett’s esophagus with high-grade dysplasia. An indication for prophylactic esophagectomy. Ann Surg. 1996;224(1):66–71.
Nelson DB, Dhupar R, Katkhuda R, Correa A, Goltsov A, Maru D, et al. Outcomes after endoscopic mucosal resection or esophagectomy for submucosal esophageal adenocarcinoma. J Thorac Cardiovasc Surg. 2018;156(1):406-13.e3.
Gockel I, Hoffmeister A. Endoscopic or surgical resection for gastro-esophageal cancer. Dtsch Arztebl Int. 2018;115(31–32):513–9.
Donlon NE, Elliott JA, Donohoe CL, Murphy CF, Nugent T, Moran B, et al. Adverse biology in adenocarcinoma of the esophagus and esophagogastric junction impacts survival and response to neoadjuvant therapy independent of anatomic subtype. Ann Surg. 2020;272(5):814–9.
Hoeppner J, Plum PS, Buhr H, Gockel I, Lorenz D, Ghadimi M, et al. Surgical treatment of esophageal cancer-indicators for quality in diagnostics and treatment. Chirurg. 2021;92(4):350–60.
Lian J, Chen S, Zhang Y, Qiu F. A meta-analysis of endoscopic submucosal dissection and EMR for early gastric cancer. Gastrointest Endosc. 2012;76(4):763–70.
Ono H, Kondo H, Gotoda T, Shirao K, Yamaguchi H, Saito D, et al. Endoscopic mucosal resection for treatment of early gastric cancer. Gut. 2001;48(2):225–9.
Pimentel-Nunes P, Libânio D, Bastiaansen BAJ, Bhandari P, Bisschops R, Bourke MJ, et al. Endoscopic submucosal dissection for superficial gastrointestinal lesions: European society of gastrointestinal endoscopy (ESGE) guideline—update 2022. Endoscopy. 2022;54(6):591–622.
Sgourakis G, Gockel I, Lang H. Endoscopic and surgical resection of T1a/T1b esophageal neoplasms: a systematic review. World J Gastroenterol. 2013;19(9):1424–37.
Pan L, Liu X, Wang W, Zhu L, Yu W, Lv W, et al. The influence of different treatment strategies on the long-term prognosis of T1 stage esophageal cancer patients. Front Oncol. 2021;11:700088.
Harlow C, Sivananthan A, Ayaru L, Patel K, Darzi A, Patel N. Endoscopic submucosal dissection: an update on tools and accessories. Ther Adv Gastrointest Endosc. 2020;13:2631774520957220.
Huang R, Yan H, Ren G, Pan Y, Zhang L, Liu Z, et al. Comparison of o-type hybridknife to conventional knife in endoscopic submucosal dissection for gastric mucosal lesions. Medicine (Baltimore). 2016;95(13):e3148.
Noguchi M, Yano T, Kato T, Kadota T, Imajoh M, Morimoto H, et al. Risk factors for intraoperative perforation during endoscopic submucosal dissection of superficial esophageal squamous cell carcinoma. World J Gastroenterol. 2017;23(3):478–85.
Ding X, Luo H, Duan H. Risk factors for perforation of gastric endoscopic submucosal dissection: a systematic review and meta-analysis. Eur J Gastroenterol Hepatol. 2019;31(12):1481–8.
Paspatis GA, Arvanitakis M, Dumonceau JM, Barthet M, Saunders B, Turino SY, et al. Diagnosis and management of iatrogenic endoscopic perforations: European society of gastrointestinal endoscopy (ESGE) position statement—update 2020. Endoscopy. 2020;52(9):792–810.
Wang WP, Ni PZ, Yang JL, Wu JC, Yang YS, Chen LQ. Esophagectomy after endoscopic submucosal dissection for esophageal carcinoma. J Thorac Dis. 2018;10(6):3253–61.
Wang AY, Hwang JH, Bhatt A, Draganov PV. AGA clinical practice update on surveillance after pathologically curative endoscopic submucosal dissection of early gastrointestinal neoplasia in the United States: commentary. Gastroenterology. 2021;161(6):2030–40.
Parikh MP, Thota PN, Raja S, Murthy S, Ahmad U, Gupta NM, et al. Outcomes of endoscopic submucosal dissection in esophageal adenocarcinoma staged T1bN0 by endoscopic ultrasound in non-surgical patients. J Gastrointest Oncol. 2019;10(2):362–6.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2023 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Donlon, N.E., Ferri, L. (2023). Endoscopic Management of Early Esophageal Cancer. In: Schlottmann, F., Ferri, L., Molena, D., Patti, M.G. (eds) Esophageal Cancer. Springer, Cham. https://doi.org/10.1007/978-3-031-39086-9_7
Download citation
DOI: https://doi.org/10.1007/978-3-031-39086-9_7
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-39085-2
Online ISBN: 978-3-031-39086-9
eBook Packages: MedicineMedicine (R0)