Abstract
Cancer has been one of the leading causes of increasing mortality rate globally. It is the most prominent cause of death worldwide and in most cases incurable if there is delay in its diagnosis. The etiology of cancer is mostly dependent on its exposure to carcinogens consistently. The Renin-Angiotensin system (RAS) plays a significant role in the field of cancer biology that affects the growth of the tumor, and its dissemination either directly or indirectly. Targeting the RAS and by activating the immunostimulatory pathways, the RAS inhibitors (RASi) can enhance cancer immunotherapy by improving cancer treatment. Currently, researchers are more interested in using bioactive compounds from medicinal plants in anticancer therapy since it has no side effects. Plant-derived bioactive compounds having anticancer properties are generally non-toxic or are less toxic. Several phytochemicals have potential anticancer properties with effects on signaling pathways and cellular processes. This review focuses on the RAS in lung and liver cancer. It also highlights the extraction of bioactive compounds having anticancer effects from medicinal plants by using bioinformatic databases to treat liver and lung cancers.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
Introduction
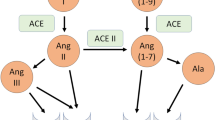
Angiotensin-converting enzyme (ACE) is the central unit of the Renin-Angiotensin system (RAS). It controls the blood pressure and regulates the volume of body fluids. ACE enhances the blood pressure and thereby, causes the constricting of blood vessels. ACE helps in the hydrolyzation of peptides by removing a dipeptide from C-terminus end.
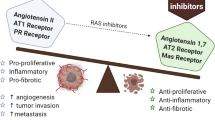
The RAS has been viewed as ‘Endocrine RAS’ due to its association with the endocrine system. It plays a crucial role in regulation of the blood pressure. In the endocrine system of RAS, an enzyme produced by the kidney, renin acts on circulation of the Angiotensinogen (AGT) protein [1]. Renin helps in cleaving the AGT protein by producing a particular fragment of 10 amino acids, Angiotensin I (Ang I). The inactive decapeptide form of Ang I later gets converted to the active octapeptide, Angiotensin II (Ang II) by removal of the His-Leu dipeptide. Ang II binds to its specific cell surface receptors, AT1 and AT2, belonging to the seven transmembrane, G-protein receptor superfamily [1]. The AT1 receptor plays a significant role in mediating the classical actions like sodium retention, cell proliferation and growth, and vasodilation of Ang II. Simultaneously, the AT2 receptor helps in promoting the apoptosis, inhibiting cell growth, cell differentiation, and vasodilation that may counterbalance the effect of Ang II on the AT1 receptor [2].
Angiotensin-converting enzyme 2 (ACE2) plays a pivotal role in protecting the inflammation and fibrogenesis of lung and liver [3, 4]. Cirrhosis and advanced level liver fibrosis are the major risk factors resulting in hepatocellular carcinoma (HCC) [5]. In addition, Idiopathic pulmonary fibrosis (IPF) has been reported to be associated with increased risk of lung cancers [6]. The occurrence of dysplastic or atypical changes in the fibrosis may progress to malignancy, thereby, resulting in the development of cancer in the major fibrosis region [7]. ACE2 is one of the most important parts of the RAS.
Both ACE2 and Ang 1-7 provides protection in liver fibrosis against liver injury and progress of cirrhosis [8]. Since RAS participates in regulation of hepatic fibrosis, tissue modelling, and hepatic inflammation post liver injury, the role of ACE2 in liver disease has been of special interest in comparison to other organs. RAS helps in inducing transforming growth factor β1 (TGF-β1) and activating the hepatic stellate cells [9]. The treatment of the liver disease with angiotensin receptor blockers and Angiotensin-converting enzyme inhibitors (ACEIs) can attenuate the progression of fibrosis in human studies [10]. In addition, ACE2 supplementation is reported to prevent bile duct liver fibrosis [4, 10].
Similarly, ACE2 mediated by the effects of Mas oncogene, peptide product of ACE2 known as the ANG1-7 receptor plays a major protective role in diseases related to lungs [3, 11,12,13]. ACE2 has been reported to be downregulated in fibrotic condition of neonatal and adult human lungs [3, 12, 13]. They also reported a significant reduction of ACE2 and identification of their protective effects in the IPF human lung [3]. They also demonstrated the regulation of ACE2 in the alveolar epithelial survival cells by balancing both proapoptotic Ang II and its antiapoptotic product, Ang 1-7 [14].
HCC being one of the most frequently occurring cancers has a high mortality rate. Several treatment methods have been reported for treating HCC but most of them also have side effects. Alternatively, the use of plant products not only helps in the prevention but also deals with the co-treatment of HCC. Abdel-Hamid et al. [15] reported the results of a study on the management of HCC by using herbal medicinal products. They demonstrated the mechanism of action and pathways involved in the herbal products and also their bioactive molecules involved in the co-treatment as well as prevention of the HCC. They also reported that these herbal products and their bioactive molecules inhibit the development and progression of liver cancer in various ways including enhancing the effects of several chemotherapeutic drugs, suppressing the chronic inflammation and oxidative stress, protection against the liver carcinogens, and inhibition of metastasis and cancerous cell growth. Another study by Chen et al. [16] reported the anticancer mechanism and the mode of action of the formulations of some Chinese herbal medicines. They reported the efficacy of these herbal medicines as an alternative therapeutic therapy for liver cancers.
A study by Li et al. [17] reported the effect of Traditional Chinese medicine (TCM) in lung cancers, especially non-small cell lung cancer (NSCLC). They demonstrated that TCM has a profound effect compared to other standard treatments in the treatment of lung cancer. Similarly, another study was reported by Chota et al. [18] on the potential treatment of lung cancer by using an African herbal medicinal plant, Dicoma anomala.
Yuan et al. [19] reported the role of several bioactive compounds extracted from plant products in the treatment of cancer. They also demonstrated the mechanism and mode of action of their anticancer effect. A recent study by Chavda et al. [20] reported the use of several natural bioactive components in anticancer therapy. They also reported the encapsulation of natural bioactive molecules in several drug delivery methods to improve their anticancer effect. A new drug delivery system that helps in entrapping the natural bioactive compounds was also developed by them.
This present review mainly focuses on the Renin-Angiotensin system (RAS) and also on the enzymes and inhibitors associated with RAS present in both lung and liver cancers. The treatment of lung and liver cancer using the medicinal plant products is also highlighted. This study also reviews the phytochemicals and bioactive compounds extracted from various medicinal plants by the use of bioinformatic databases and tools and their use in the treatment of liver and lung cancers.
Liver Cancer—Hepatocellular Carcinoma
Zhang et al. [21] reported that liver cancers are reported to be ranked sixth worldwide for most incident cases in the year 2020. It is considered to be the third main leading cause of cancer-associated death with 830,180 mortality per year and 905,677 new reported cases approximately. Based on the estimation of the World Health Organisation (WHO) approximately more than 1.3 million individuals are predicted to die from liver cancers by the year 2040 [21].
Among all the primary liver cancers occurring, hepatocellular carcinoma (HCC) is the one which causes tumor in 75–85% cases [22]. HCC is known to be one of the most malignant lethal tumors commonly occurring over the globe. Molecular studies have revealed that the source of HCC is the mature hepatocytes [23]. They are often diagnosed at a late or advanced stage of cancer and hence, there are very limited options available for their effective treatment at that stage [24].
During the past decade, only one systemic drug, Sorafenib has been reported to be clinically to be effective against advanced HCC. It has been approved by the Food and Drug Administration (FDA) and is the standard drug for treating advanced HCC [21].
In recent years, there has been an advancement in the treatment of advanced HCC. Several molecular targeted drugs as well as immune checkpoint inhibitors (ICIs) have been approved as therapeutic drugs for treating advanced HCC. Moreover, Lee et al. [25] reported that a combination of two drugs, bevacizumab and atezolizumab, brought about a significant change in the treatment of advanced HCC. They demonstrated that the incorporation of ICIs into advanced HCC therapies and also their combination with the molecular targeted therapy is emerging as a novel tool in the enhancement of immune responses.
Liver cirrhosis is the most significant risk factor for liver cancers. Although there has been progress in both diagnosis as well as treatment, surgical resection or orthotopic liver transplantation (OLT) is the only cure [23]. Once the degree of differentiation in the liver worsens, the prognosis for liver cancer patients becomes a worse. Hence, restoring the differentiation state may bring about an improvement in the prognosis. The treatment of differentiation of liver cancer is done for reversing the dedifferentiation method of hepatocyte cells to liver cancerous cells by using drugs. Hence, this can be used as a novel treatment tool for HCC in improving the prognosis, reducing the reoccurrence, and restoring the normal characteristics of the liver [23].
Lung Cancer
Lung cancer is considered as the leading cause of the death of cancer patients [26]. Mortality data has been collected and published by the National Center for Health Statistics (NCHS). In the year 2022, approximately 609,360 deaths from cancer and 1,918,030 new cases are projected in the US. Among them, lung cancer has been projected to cause most of the deaths, accounting for 350 deaths each day [27]. Immunotherapy is now a line of treatment for early stages of lung cancer which has drastically changed the quality of care for the patients with metastatic non-small-cell lung cancer (NSCLC) [26].
Slebe et al. [28] reported that currently immune oncology (IO) therapy has become a significant treatment method for patients living with a NSCLC. However, they revealed that only a limited number of patients benefit from this IO therapy. Hence, biomarkers and predictive biomarkers providing insights into the functional biological pathways at tumor microenvironment (TME) level have the capability of elevating the impact of IO therapy. This may lead to the development of improved and novel drug strategies. They also reported that by using several highly-specific radiolabeled tracers, immune positron emission tomography (immunoPET) could provide these biomarkers, to investigate the main targets in TME with immunoPET imaging. In addition, immunoPET has the ability to provide the pharmacodynamic biomarkers to help in the development of novel drugs and predictive biomarkers to guide in the decision making of innovative and clinical treatment strategies in lung cancer [28].
Angiotensin Converting Enzyme Inhibitors (ACEI)
Angiotensin-converting enzyme inhibitors (ACEI) are drugs that are prescribed mainly for treating heart problems like stroke, heart failure, coronary artery disease (CAD), heart attacks, and others involving high blood pressure (hypertension), hardening of connective tissues and skin (Scleroderma), diabetes, migraines, chronic kidney disease etc. [29, 30]. These drugs help to relax the arteries and veins and lower the blood pressure. The ACE inhibitors block the ACE from converting to Angiotensin II, thus preventing their production in the body. They can narrow the blood vessels and thereby, results in increasing the blood pressure and forcing the heart to work harder.
ACEI includes Captopril, Benazepril (Lotensin), Fosinopril, Enalapril (Vasotec), Lisinopril (Zestril, Prinvil), Perindopril, Ramipril (Altace), Moexipril, Quinapril (Accupril) and Trandolapril. The exact mechanism of action of ACEIs is still not known completely. Although, they interfere with the Renin-Angiotensin-Aldosterone system, the efficacy of ACE inhibitors is indirectly related to renin level in blood.
Hypertension
ACE inhibitors lower both the diastolic and systolic blood pressure mean arterial blood pressure effectively in normotensive and hypertensive subjects [30,31,32]. These drugs have been reported to have antihypertensive effects in several randomized clinical [33]. The Eighth Joint National Commission (JNC8) in the year 2014 reported evidence-based guidelines to treat hypertension in adults. They recommended ACE inhibitors as one of the four classes of drugs for initial therapy in adults with hypertension [34]. Currently, the guidelines published by the European Society of Cardiology (ESC) and the American Heart Association/American College of Cardiology (AHA/ACC) recommend the ACEI as first-line antihypertensive therapeutic drugs. They are mainly recommended for patients with cardiovascular diseases and diabetes mellitus [35, 36]. The ACE inhibitors are proven to be very effective drugs in White race people and less effective in case of Black race people in clinical practice [30].
Post Myocardial Infarction (MI)
Over the past few decades, there have been multiple randomized trials conducted on mortality due to ACE inhibitors post MI [30, 37, 38]. A majority of the trials conducted have reported a remarkable decrease in the mortality rate as well as slow progression of congestive heart failure after the MI patients were treated with ACEI [30, 39]. The clinical practice for patients with heart failure or dysfunctional left ventricle is treatment with ACEI without delay post MI. It is suggested that all the patients be treated with ACEI initially based on the functional assessment of the left ventricle with a review before proceeding further [40].
Heart Failure
ACE inhibitors improve heart failure by lowering the preload, afterload, and systolic wall stress, resulting in increase of cardiac output without much increase in the heart rate [30, 41, 42]. ACEIs play a significant role in excreting salts by reducing the production of antidiuretic and aldosterone hormones, and augmentation of renal blood flow. The ACE inhibitors also reduce cardiac myocyte hypertrophy. Several large, randomized, prospective placebo-controlled trials have been reported since the 1980s. They revealed that patients with heart failure when treated with ACEIs reduces the overall mortality rate with reduction in ejection fraction [30, 43]. They proved that ACEIs also reduce mortality in asymptomatic patients with dysfunction in their left ventricles [30, 44]. Hence, these trials proved that ACE inhibitors can be recommended strongly as first-line therapeutic drugs for patients suffering from heart failure [45].
Diabetes
Due to diabetes mellitus associated neuropathy, the elevation of glomerular capillary pressure and the Renin-Angiotensin-Aldosterone system have been reported to increase the progression of renal dysfunction [30, 46]. A large, randomized, prospective placebo-controlled trial has proved that ACEIs reduce the neuropathy progression in diabetes mellitus insulin-dependent patients. They also reduce the combined endpoints of transplantation, dialysis, and death significantly [30, 47]. Recent recommendations prescribe ACEIs as first-choice therapy in patients with high blood pressure having no history of CAD, which improves the heart function and decreases the incidence of MI [48].
Chronic Kidney Disease (CKD)
ACE inhibitors are the first-line therapeutic drugs used in CKD patients. The effect of ACEI has proved to be more superior compared to placebo-controlled treatment on slowing the CKD progression and reducing proteinuria [49].
Proteinuria or Nephrotic Syndrome
ACEIs have been reported to reduce the glomerular capillary pressure by dilating the efferent arterioles selectively as well as lowering the arterial pressure [30, 50]. The use of ACEIs has been proven to prevent microalbuminuria progression to proteinuria [30, 51]. The ACE inhibitors provide protection for longer duration against proteinuria progression and development. Hence, they maintain stability in renal function in untreated patients with renal function impairment [30, 51].
Post-transplant Glomerulonephritis
The use of ACEIs is the mainstay and first-line drug in patients suffering from glomerular diseases. It gradually slows down the decreasing proteinuria and glomerular filtration rate (GFR) [30, 52]. Moreover, the Renin-Angiotensin-Aldosterone inhibitors can prolong the survival of graft in patients suffering from post-transplant glomerulonephritis [30, 53].
Bioinformatics Study on Angiotensin Enzymes in Liver and Lung Cancer
Bioinformatic Tools Used in Lung Cancer
Patients with lung cancer are reportedly more susceptible to severe acute respiratory syndrome Coronavirus 2 (SARS-CoV-2) infection that began spreading during the Coronavirus disease 2019 (COVID-19) pandemic. The SARS-CoV-2 virus enters the host’s body through the Angiotensin-converting enzyme 2 (ACE2) receptor. Samad et al. [54] reported the identification of ACE2 protein as a potential biomarker in the SARS-CoV2 infection and its association with lung cancer by using bioinformatic tools and computational analyses.
ACE2 Expression Analysis Using the Cancer Genome Atlas (TCGA) Datasets
ACE2 transcriptional expression analysis was explored in multiple cancers by applying an online server tool, Gene expression profiling interactive analysis 2 (GEPIA2) that is available publicly using the datasets, “The cancer genome atlas (TCGA)” [55]. They studied the data of mRNA expression of ACE2 gene in lung cancer using TCGA dataset through UALCAN website [56, 57]. Further, they analyzed the mRNA expression of the ACE2 gene using another tool, Oncomine and the TCGA datasets [58, 59]. Both web-platforms, UALCAN and Oncomine are user friendly and are used for revealing the gene expression of the TCGA data and also relative expression of a particular query gene set.
Mutational and Survival Analyses of AEC2 Gene in Lung Cancer
The Genotype 2 outcome server was used to perform the survival analysis of the AEC2 gene in lung cancer. This server provides Kaplan–Meier plots using TCGA data. The position of mutational analysis was performed using cBioPortal in lung cancer through TCGA data [60].
COVID-19 Gene Identification and Co-expression Profiling of AEC2 Gene Using the Comparative Toxicogenomics Database (CTD)
The co-related genes of lung squamous carcinoma (LUSC) and lung adenocarcinoma (LUAD) in Visualization platform and R2 genomics analysis were collected using the TCGA dataset [61]. Then using the comparative toxicogenomics database (CTD) all the COVID -19 genes were downloaded [62] in text format and visualized by FunRich software.
Protein–Protein Interaction Using the GeneMANIA Database of Co-expressed Genes
An extensive database, GeneMANIA was used that provided a flexible interface for the gene function, proteomic, and genomic data query. This database has functional associated data that includes co-expression, gene and protein interactions, and pathways [63, 64].
Gene Ontology Analysis and Functional Pathway of Common Co-related Genes with COVID-19 Associated Lung Cancer
Kyoto encyclopedia of genomes and gene (KEGG) database is used for high-level function and biological system utilities through high-throughput technologies and information related to genome sequencing at molecular level [65]. Gene ontology analysis was predicted for computation of biological system. Gene ontology was performed by using web portal, Enrichr. In addition, the analysis of commonly co-related genes of the COVID-19 pandemic and lung cancer was done for their enrichment functional pathway [66].
Bioinformatic Tools Used in Liver Cancer
ACE2 is the SARS-CoV-2 receptor causing COVID-19 pandemic and Transmembrane serine protease 2 (TMPRSS2) is the coreceptor. The abnormality in the hepatic function due to COVID-19 infection suggested bystander or specific liver disease. The liver cells are widely used as SARS-CoV-2 infection models in vitro since they can express the viral ACE2 receptor. Desquilles et al. [67] demonstrated the analysis of TMPRSS2 and ACE2 expressions and their localization in non-tumorous livers and human cancerous livers. They reported that the differentiated liver cancers displayed the potential relation between metabolic breakdown and dysfunction of AEC2 gene using transcriptomic datasets.
Datasets Used in HCC Liver Cancers
The Désert’s microarray meta dataset was used. This is composed of many human HCCs. In this particular dataset, the HCCs that express an activated β-catenin transcriptomic program was identified with 5 genes including LGR5, VNN1, GHUL, HAL and ODAM, predicting activated Catenin-β1 (CTNNB1) mutations with specificity as well as high sensitivity [68]. The TCGA dataset was composed of many HCCs and non-tumorous samples. The methylation, RNA sequencing and mutation data were extracted using TCGA Abiolinks R package [68].
Plant Extracts for Treating ACE in Liver and Lung Cancer
Plant Extracts for Treating ACE in HCC
Bioactive compounds obtained from plant products have been successfully tested for treating the HCC as an alternative to chemotherapeutic drugs. The bioactive molecules from plants reported for treating liver cancer are listed in Table 9.1.
Plant Extracts for Treating ACE in Lung Cancer
Plant extracts have been reported to give significant results when used for treatment of several disorders either acute or chronic, and even cancer of the lungs. Bioactive compounds reported from plants for treating lung cancer are listed in Table 9.2.
Bioinformatics Work Using Plant Extracts for Treating Lung and Liver Cancer
A meticulous and detailed search has been performed on several electronic databases including PubMed, Scopus, Google Scholar, and Web of Science to retrieve relevant literature. A proper and detailed query set was designed on the databases. During the search, all the phrases, keywords, and relevant terms were used including “Natural medicinal plants,” “Lung cancer,” “Liver cancer,” “carcinoma,” “cancerous,” “hepatocellular carcinoma,” “Anticancer,” “Anticancer plants;” “Anticancer herbs,” “Renin-Angiotensin,” “Angiotensin-converting enzymes,” “in vivo activity,” “in vitro activity,” and “Animal models” [69].
The number of articles found to be relevant to our search were finalized. The data obtained after the curation, analysis, and extraction through the designing of phrases/keywords combination with their inclusion criteria were noted [69].
The inclusion criteria were based on two sets. The first set of criteria is the ‘general criteria’. In this criterion, the articles chosen for the manuscript had reported both the anticancer activity of traditional plants and the anticancer role of pure compounds or their extracts from the plants. The second criteria included the selection of specific plants having anticancer properties with a detailed list of phytochemicals present in them. Articles in current journals on the anticancer activity of relevant plants were selected. The in vivo and in vitro anticancer activities of the medicinal plants and the antitumor/anticancer activities of the bioactive compounds extracted from them was reported [69].
An in-silico study by Putra et al. [70] reported on the anticancer potential of several Indonesian herbal plants. They first used databases like Pubchem, Protein data bank (PDB), Marvin Sketch software, and Ramachandran plot to retrieve the protein and ligand. They later performed molecular docking by using the AutoDock tools software, and visualized the docking analysis using the Discovery Studio 4.0 for evaluating the binding value of Gibbs free energy and their amino acid residues. They reported three bioactive compounds, physcionin, berberine, and pinostrobin that showed potential anticancer properties against the lung cancer.
Conclusion
Cancer, being a public health problem has a profound impact on the health of people in both developing and developed countries across the globe. Clinical studies provided enough compelling evidences that Ang II helps in regulating all the hallmarks of several cancers, including liver and lung cancer. To overcome the problems involved in the use of currently available drugs for the treatment of cancer, there is a need to look for certain effective and novel anticancer agents that can improve the efficacy with minimum side effects. In the present review, bioinformatic databases were used to obtain the potential bioactive compounds from medicinal plants to treat lung and liver cancers. Later, anti-cancerous as well as bioactive medicinal compound libraries from several endemic plants from different regions can be developed by using high-throughput techniques.
References
Abdul-Hafez A, Mohamed T, Omar H et al (2018) The renin angiotensin system in liver and lung: impact and therapeutic potential in organ fibrosis. J Lung Pulm Respir Res 5(1):00160
Filippatos G, Tilak M, Pinillos H et al (2001) Regulation of apoptosis by angiotensin II in the heart and lungs (review). Int J Mol Med 7(3):273–280. https://doi.org/10.3892/ijmm.7.3.273
Li X, Molina-Molina M, Abdul-Hafez A et al (2008) Angiotensin converting enzyme-2 is protective but downregulated in human and experimental lung fibrosis. Am J Physiol Lung Cell Mol Physiol 295(1):L178–L185. https://doi.org/10.1152/ajplung.00009.2008
Osterreicher CH, Taura K, De Minicis S et al (2009) Angiotensin-converting-enzyme 2 inhibits liver fibrosis in mice. Hepatology 50(3):929–938. https://doi.org/10.1002/hep.23104
Dhar D, Baglieri J, Kisseleva T, Brenner DA (2020) Mechanisms of liver fibrosis and its role in liver cancer. Exp Biol Med (Maywood) 245(2):96–108. https://doi.org/10.1177/1535370219898141
Ballester B, Milara J, Cortijo J (2019) Idiopathic pulmonary fibrosis and lung cancer: mechanisms and molecular targets. Int J Mol Sci 20(3):593. https://doi.org/10.3390/ijms20030593
Park J, Kim DS, Shim TS et al (2001) Lung cancer in patients with idiopathic pulmonary fibrosis. Eur Respir J 17(6):1216–1219. https://doi.org/10.1183/09031936.01.99055301
Warner FJ, Lubel JS, McCaughan GW et al (2007) Liver fibrosis: a balance of ACEs? Clin Sci (Lond) 113(3):109–118. https://doi.org/10.1042/cs20070026
Abbas G, Silveira MG, Lindor KD (2011) Hepatic fibrosis and the reninangiotensin system. Am J Ther 18(6):e202–e208. https://doi.org/10.1097/mjt.0b013e3181df8df5
Mak KY, Chin R, Cunningham SC et al (2015) ACE2 therapy using adeno-associated viral vector inhibits liver fibrosis in mice. Mol Ther 23(9):1434–1443. https://doi.org/10.1038/mt.2015.92
Wagenaar GT, El Laghmani H, Fidder M et al (2013) Agonists of MAS oncogene and angiotensin II type 2 receptors attenuate cardiopulmonary disease in rats with neonatal hyperoxia-induced lung injury. Am J Physiol Lung Cell Mol Physiol 305(5):L341–L351. https://doi.org/10.1152/ajplung.00360.2012
Oarhe CI, Dang V, Dang M et al (2015) Hyperoxia downregulates angiotensin-converting enzyme-2 in human fetal lung fibroblasts. Pediatr Res 77(5):656–662. https://doi.org/10.1038/pr.2015.27
Mohamed TL, Nguyen HT, Abdul-Hafez A et al (2016) Prior hypoxia prevents downregulation of ACE-2 by hyperoxia in fetal human lung fibroblasts. Exp Lung Res 42(3):121–130. https://doi.org/10.3109/01902148.2016.1157712
Uhal BD, Li X, Xue A et al (2011) Regulation of alveolar epithelial cell survival by the ACE-2/angiotensin 1–7/Mas axis. Am J Physiol 301(3):L269–L274. https://doi.org/10.1152/ajplung.00222.2010
Abdel-Hamid NM, Abass SA, Mohamed AA, Hamid DM (2018) Herbal management of hepatocellular carcinoma through cutting the pathways of the common risk factors. Biomed Pharmacother 107:1246–1258. https://doi.org/10.1016/j.biopha.2018.08.104
Chen F, Zhong Z, Tan HY et al (2020) Uncovering the anticancer mechanisms of Chinese herbal medicine formulas: therapeutic alternatives for liver cancer. Front Pharmacol 11:293. https://doi.org/10.3389/fphar.2020.00293
Li Z, Feiyue Z, Gaofeng L (2021) Traditional Chinese medicine and lung cancer—from theory to practice. Biomed Pharmacother 137:111381. https://doi.org/10.1016/j.biopha.2021.111381
Chota A, George BP, Abrahamse H (2020) Potential treatment of breast and lung cancer using Dicoma anomala, an African medicinal plant. Molecules 25(19):4435. https://doi.org/10.3390/molecules25194435
Yuan M, Zhang G, Bai W et al (2022) The role of bioactive compounds in natural products extracted from plants in cancer treatment and their mechanisms related to anticancer effects. Oxid Med Cell Longev 2022:1429869. https://doi.org/10.1155/2022/1429869
Chavda VP, Patel AB, Mistry KJ et al (2022) Nano-drug delivery systems entrapping natural bioactive compounds for cancer: recent progress and future challenges. Front Oncol 12:867655. https://doi.org/10.3389/fonc.2022.867655
Zhang H, Zhang W, Jiang L, Chen Y (2022) Recent advances in systemic therapy for hepatocellular carcinoma. Biomark Res 10:3. https://doi.org/10.1186/s40364-021-00350-4
Bray F, Ferlay J, Soerjomataram I et al (2018) Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 68(6):394–424
Song J, Zhou H, Gu D, Xu Y (2022) Hepatocellular carcinoma differentiation: research progress in mechanism and treatment. Front Oncol 11:790358. https://doi.org/10.3389/fonc.2021.790358
Sim HW, Knox J (2018) Hepatocellular carcinoma in the era of immunotherapy. Curr Probl Cancer 42(1):40–48. https://doi.org/10.3322/caac.21492
Lee MS, Ryoo BY, Hsu CH et al (2020) Atezolizumab with or without bevacizumab in unresectable hepatocellular carcinoma (GO30140): an open-label, multicentre, phase 1b study. Lancet Oncol 21(6):808–820. https://doi.org/10.1016/s1470-2045(20)30156-x
Mithoowani H, Febbraro M (2022) Non-small-cell lung cancer in 2022: a review for general practitioners in oncology. Curr Oncol 29(3):1828–1839. https://doi.org/10.3390/curroncol29030150
Siegel RL, Miller KD, Fuchs HE, Jemal A (2022) Cancer statistics, 2022. CA Cancer J Clin 72(1):7–33. https://doi.org/10.3322/caac.21708
Slebe M, Pouw JEE, Hashemi SMS et al (2022) Current state and upcoming opportunities for immunoPET biomarkers in lung cancer. Lung Cancer 169:84–93. https://doi.org/10.1016/j.lungcan.2022.05.017
Nasution SA (2006) The use of ACE inhibitor in cardiovascular disease. Acta Med Indones 38(1):60–64
Herman LL, Padala SA, Ahmed I, Bashir K (2022) StatPearls [Internet]. Angiotensin converting enzyme inhibitors (ACEI), Treasure Island (FL), StatPearls Publishing LLC
Vidt DG, Bravo EL, Fouad FM (1982) Medical intelligence drug therapy: captopril. N Engl J Med 306(4):214–219
Todd PA, Heel RC (1986) Enalapril. A review of its pharmacodynamic and pharmacokinetic properties, and therapeutic use in hypertension and congestive heart failure. Drugs 31(3):198–248. https://doi.org/10.2165/00003495-198631030-00002
Messerli FH, Bangalore S, Bavishi C, Rimoldi SF (2018) Angiotensin-converting enzyme inhibitors in hypertension: to use or not to use? J Am Coll Cardiol 71(13):1474–1482. https://doi.org/10.1016/j.jacc.2018.01.058
James PA, Oparil S, Carter BL, et al (2014) 2014 evidence-based guideline for the management of high blood pressure in adults: report from the panel members appointed to the Eighth Joint National Committee (JNC 8). JAMA e311(5):507–520. https://doi.org/10.1001/jama.2013.284427
Whelton PK, Carey RM, Aronow WS et al (2018) 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA guideline for the prevention, detection, evaluation, and management of high blood pressure in adults: a report of the American College of Cardiology/American Heart Association Task Force on clinical practice guidelines. J Am Coll Cardiol 71(19):e127–e248. https://doi.org/10.1161/hyp.0000000000000066
Williams B, Mancia G, Spiering W et al (2018) ESC Scientific Document Group. 2018 ESC/ESH guidelines for the management of arterial hypertension. Eur Heart J 39(33):3021–3104. https://doi.org/10.1093/eurheartj/ehy339
Swedberg K, Held P, Kjekshus J et al (1992) Effects of the early administration of enalapril on mortality in patients with acute myocardial infarction. Results of the Cooperative New Scandinavian Enalapril Survival Study II (CONSENSUS II). N Engl J Med 327(10):678–684. https://doi.org/10.1056/NEJM199209033271002
Ambrosioni E, Borghi C, Magnani B (1995) The effect of the angiotensin-converting-enzyme inhibitor zofenopril on mortality and morbidity after anterior myocardial infarction. The survival of myocardial infarction long-term evaluation (SMILE) study investigators. N Engl J Med 332(2):80–85. https://doi.org/10.1056/nejm199501123320203
Pfeffer MA (1995) Left ventricular remodeling after acute myocardial infarction. Annu Rev Med 46:455–466. https://doi.org/10.1146/annurev.med.46.1.455
O’Gara PT, Kushner FG, Ascheim DD et al (2013) American College of Cardiology Foundation/American Heart Association Task Force on practice guidelines. 2013 ACCF/AHA guideline for the management of ST-elevation myocardial infarction: a report of the American College of Cardiology Foundation/American Heart Association Task Force on practice guidelines. Circulation 127(4):e362–e425. https://doi.org/10.1161/CIR.0b013e3182742c84
Gavras H, Faxon DP, Berkoben J, Brunner HR, Ryan TJ (1978) Angiotensin converting enzyme inhibition in patients with congestive heart failure. Circulation 58(5):770–776. https://doi.org/10.1161/01.cir.58.5.770
Dzau VJ, Colucci WS, Williams GH, Curfman G, Meggs L, Hollenberg NK (1980) Sustained effectiveness of converting-enzyme inhibition in patients with severe congestive heart failure. N Engl J Med 302(25):1373–1379. https://doi.org/10.1056/nejm198006193022501
Yusuf S, Pitt B, Davis CE, Hood WB, Cohn JN (1991) Effect of enalapril on survival in patients with reduced left ventricular ejection fractions and congestive heart failure. N Engl J Med 325(5):293–302. https://doi.org/10.1056/nejm199108013250501
Pfeffer MA, Braunwald E, Moyé LA et al (1992) Effect of captopril on mortality and morbidity in patients with left ventricular dysfunction after myocardial infarction. Results of the survival and ventricular enlargement trial. The SAVE Investigators. N Engl J Med 327(10):669–677. https://doi.org/10.1056/nejm199209033271001
Ponikowski P, Voors AA, Anker SD et al (2016) ESC Scientific Document Group. 2016 ESC guidelines for the diagnosis and treatment of acute and chronic heart failure: The Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC) developed with the special contribution of the Heart Failure Association (HFA) of the ESC. Eur Heart J 37(27):2129–2200. https://doi.org/10.1093/eurheartj/ehw128
Chikawa I, Brenner BM (1984) Glomerular actions of angiotensin II. Am J Med 76(5B):43–49. https://doi.org/10.1016/0002-9343(84)90882-9
Lewis EJ, Hunsicker LG, Bain RP, Rohde RD (1993) The effect of angiotensin-converting-enzyme inhibition on diabetic nephropathy. The Collaborative Study Group. N Engl J Med 329(20):1456–1462. https://doi.org/10.1056/nejm199311113292004
Zhang Y, Ding X, Hua B, Liu Q, Chen H, Zhao XQ, Li W, Li H (2020) Real-world use of ACEI/ARB in diabetic hypertensive patients before the initial diagnosis of obstructive coronary artery disease: patient characteristics and long-term follow-up outcome. J Transl Med 18(1):150. https://doi.org/10.1186/s12967-020-02314-y
Silvariño R, Rios P, Baldovinos G, Chichet MA, Perg N, Sola L, Saona G, De Souza N, Lamadrid V, Gadola L (2019) Is chronic kidney disease progression influenced by the type of renin-angiotensin-system blocker used? Nephron 143(2):100–107. https://doi.org/10.1159/000500925
Anderson S, Rennke HG, Brenner BM (1986) Therapeutic advantage of converting enzyme inhibitors in arresting progressive renal disease associated with systemic hypertension in the rat. J Clin Invest 77(6):1993–2000. https://doi.org/10.1172/jci112528
Ravid M, Lang R, Rachmani R, Lishner M (1996) Long-term renoprotective effect of angiotensin-converting enzyme inhibition in non-insulin-dependent diabetes mellitus. A 7-year follow-up study. Arch Intern Med 156(3):286–289
Rigat B, Hubert C, Alhenc-Gelas F, Cambien F, Corvol P, Soubrier F (1990) An insertion/deletion polymorphism in the angiotensin I-converting enzyme gene accounting for half the variance of serum enzyme levels. J Clin Invest 86(4):1343–1346. https://doi.org/10.1172/jci114844
Pazik J, Ostrowska J, Lewandowski Z, Mróz A, Perkowska-Ptasińska A, Baczkowska T, Durlik M (2008) Renin-angiotensin-aldosterone system inhibitors and statins prolong graft survival in post-transplant glomerulonephritis. Ann Transplant 13(4):41–45
Samad A, Jafar T, Rafi JH (2020) Identification of angiotensin-converting enzyme 2 (ACE2) protein as the potential biomarker in SARS-CoV-2 infection-related lung cancer using computational analyses. Genomics 112(6):4912–4923. https://doi.org/10.1016/j.ygeno.2020.09.002
Tang Z, Kang B, Li C et al (2019) GEPIA2: an enhanced web server for large-scale expression profiling and interactive analysis. Nucleic Acids Res 47(W1):W556–W560. https://doi.org/10.1093/nar/gkz430
Chandrashekar DS, Bashel B, Balasubramanya SAH et al (2017) UALCAN: a portal for facilitating tumor subgroup gene expression and survival analyses. Neoplasia (United States). https://doi.org/10.1016/j.neo.2017.05.002
Saha SK, Kader MA, Samad KA et al (2020) Prognostic and clinico-pathological significance of BIN1 in breast cancer. Inf Med Unlocked 19:100327. https://doi.org/10.1016/j.imu.2020.100327
Rhodes DR, Kalyana-Sundaram S, Mahavisno V et al (2007) Oncomine 3.0: genes, pathways, and networks in a collection of 18,000 cancer gene expression profiles. Neoplasia (New York, NY) 9:166. https://doi.org/10.1593/neo.07112
Barman UD, Saha SK, Kader MA et al (2020) Clinicopathological and prognostic significance of GPC3 in human breast cancer and its 3D structure prediction. Netw Model Anal Heal Inf Bioinf 9:1–18
Cerami E, Gao J, Dogrusoz U et al (2012) The cBio cancer genomics portal: an open platform for exploring multidimensional cancer genomics data. Cancer Discov 2:401–404. https://doi.org/10.1158/2159-8290.CD-12-0095
Koster J, Volckmann R, Zwijnenburg D et al (2019) R2: genomics analysis and visualization platform. https://doi.org/10.1158/1538-7445.sabcs18-2490
Mattingly CJ, Colby GT, Forrest JN, Boyer JL (2003) The comparative toxicogenomics database (CTD). Environ Health Perspect 111:793–795. https://doi.org/10.1289/ehp.6028
Warde-Farley D, Donaldson SL, Comes O et al (2010) The GeneMANIA prediction server: biological network integration for gene prioritization and predicting gene function. Nucleic Acids Res. https://doi.org/10.1093/nar/gkq537
Karim M, Samad A, Adhikari UK et al (2020) A multi-omics analysis of bone morphogenetic protein 5 (BMP5) mRNA expression and clinical prognostic outcomes in different cancers using bioinformatics approaches. Biomedicines 8(2):19. https://www.mdpi.com/2227-9059/8/2/19
Coperchini F, Chiovato L, Croce L et al (2020) The cytokine storm in COVID-19: an overview of the involvement of the chemokine/chemokine-receptor system. Cytokine Growth Factor Rev. https://doi.org/10.1016/j.cytogfr.2020.05.003
Kuleshov MV, Jones MR, Rouillard AD et al (2016) Enrichr: a comprehensive gene set enrichment analysis web server 2016 update. Nucleic Acids Res 44:W90–W97. https://doi.org/10.1093/nar/gkw377
Desquilles L, Cano L, Ghukasyan G et al (2022) Well-differentiated liver cancers reveal the potential link between ACE2 dysfunction and metabolic breakdown. Sci Rep 12:1859. https://doi.org/10.1038/s41598-021-03710-0
Desert R, Rohart F, Canal F et al (2017) Human hepatocellular carcinomas with a periportal phenotype have the lowest potential for early recurrence after curative resection. Hepatology 66:1502–1518. https://doi.org/10.1002/hep.29254
Khan T, Ali M, Khan A et al (2020) Anticancer plants: a review of the active phytochemicals, applications in animal models, and regulatory aspects. Biomolecules 10:47. https://doi.org/10.3390/biom10010047
Putra WK, Agustin F, Rochmatika L, Salma WO (2019) Potential of Indonesian medicinal plants as anti-cancer: in silico study. MJBMB 1:152–154. https://doi.org/10.1016/j.jep.2005.01.041
Akhtar T, Sheikh N (2016) Chemopreventive prospective of dietary spices against hepatocellular carcinoma. Curr Sci 110:579–583. https://doi.org/10.18520/cs/v110/i4/579-583
Darvesh AS, Aggarwal BB, Bishayee A (2012) Curcumin and liver cancer: a review. Curr Pharm Biotechnol 13:218–228. https://doi.org/10.2174/138920112798868791
Mann CD, Neal CP, Garcea G et al (2009) Phytochemicals as potential chemopreventive and chemotherapeutic agents in hepatocarcinogenesis. Eur J Cancer Prev 18(1):13–25. https://doi.org/10.1097/cej.0b013e3282f0c090
Gopalakrishnan A, Varuna PP, Gopinath D et al (2014) Ethnomedicine in cancer therapy: a review. W J Pharma Res 3:305–319
Platel K, Srinivasan K (1996) Influence of dietary spices or their active principles on digestive enzymes of small intestinal mucosa in rats. Int J Food Sci Nutr 47:55–59. https://doi.org/10.3109/09637489609028561
Fatehi M, Farifteh F, Fatehi-Hassanabad Z (2004) Antispasmodic and hypotensive effects of Ferula asafoetida gum extract. J Ethnopharmacol 91(2–3):321–324. https://doi.org/10.1016/j.jep.2004.01.002
Li Y, Martin RC (2011) Herbal medicine and hepatocellular carcinoma: applications and challenges. Evid Based Complement Alternat Med 2011:1–14. https://doi.org/10.1093/ecam/neq044
Perng DS, Tsai YH, Cherng J et al (2016) Discovery of a novel anticancer agent with both anti-topoisomerase i and ii activities in hepatocellular carcinoma sK-hep-1 cells in vitro and in vivo: Cinnamomum verum component 2-methoxycinnamaldehyde. Drug Des Devel Ther 10:141. https://doi.org/10.2147/dddt.s93599
Tavakkol AJ, Brook A, Mousavi SH (2008) Study of cytotoxic and apoptogenic properties of saffron extract in human cancer cell lines. Food Chem Toxicol 46(11):3443–3447. https://doi.org/10.1016/j.fct.2008.08.018
Srivastava R, Ahmed H, Dixit R (2010) Crocus sativus L.: a comprehensive review. Pharmacogn Rev 4(8):200–208. https://doi.org/10.4103/0973-7847.70919
Aggarwal BB (2010) Targeting inflammation-induced obesity and metabolic diseases by curcumin and other nutraceuticals. Annu Rev Nutr 30:173–199. https://doi.org/10.1146/annurev.nutr.012809.104755
Boyd J, Babish J, Stoewsand G (1982) Modification by beet and cabbage diets of aflatoxin B1-induced rat plasma α-foetoprotein elevation, hepatic tumorigenesis, and mutagenicity of urine. Food Chem Toxicol 20(1):47–52. https://doi.org/10.1016/S0278-6915(82)80008-2
Godlewski C, Boyd J, Sherman W et al (1985) Hepatic glutathione S-transferase activity and aflatoxin B1-induced enzyme altered foci in rats fed fractions of brussels sprouts. Cancer Lett 28(2):151–157. https://doi.org/10.1016/0304-3835(85)90070-9
Sadek I, Abdel-Salam F, Al-Qattan K (1995) Chemopreventive effects of cabbage on 7, 12-dimethylbenz (a)-anthracene-induced hepato-carcinogenesis in toads (Bufo viridis). J Nutr Sci Vitaminol 41(1):163–168. https://doi.org/10.3177/jnsv.41.163
Kassie F, Uhl M, Rabot S et al (2003) Chemoprevention of 2-amino-3-methylimidazo [4, 5-f] quinoline (IQ)-induced colonic and hepatic preneoplastic lesions in the F344 rat by cruciferous vegetables administered simultaneously with the carcinogen. Carcinogenesis 24(2):255–261. https://doi.org/10.1093/carcin/24.2.255
Tanaka T, Iwata H, Niwa K et al (1988) Inhibitory effect of ellagic acid on N-2-fluorenylacetamide-induced liver carcinogenesis in male ACI/N rats. Cancer Sci 79(12):1297–1303. https://doi.org/10.1111/j.1349-7006.1988.tb01559.x
Lagopoulos L, Sunahara GI, Würzner H et al (1991) The correlation of body growth with diethylnitrosamine-induced hepatocarcinogenesis in relation to serum insulin and somatomedin-C. Carcinogenesis 12(2):211–215. https://doi.org/10.1093/carcin/12.2.211
Kim SH, Lee CS (1992) The effect of caffeine on diethylnitrosamine initiated hepatic altered foci in a mid-term induction system. In Vivo 6(2):223–226
Hosaka S, Kawa S, Aoki Y et al (2001) Hepatocarcinogenesis inhibition by caffeine in ACI rats treated with 2-acetylaminofluorene. Food Chem Toxicol 39(6):557–561. https://doi.org/10.1016/s0278-6915(00)00175-7
Li J, Chen X, Dong X et al (2006) Specific COX-2 inhibitor, meloxicam, suppresses proliferation and induces apoptosis in human HepG2 hepatocellular carcinoma cells. J Gastroenterol Hepatol 21(12):1814–1820. https://doi.org/10.1111/j.1440-1746.2006.04366.x
Lea MA, Xiao Q, Sadhukhan AK et al (1993) Inhibitory effects of tea extracts and (−)-epigallocatechin gallate on DNA synthesis and proliferation of hepatoma and erythroleukemia cells. Cancer Lett 68(2–3):231–236. https://doi.org/10.1016/0304-3835(93)90151-x
Nishida N, Fukuda Y, Komeda T et al (1994) Amplification and overexpression of the cyclin D1 gene in aggressive human hepatocellular carcinoma. Cancer Res 54(12):3107–3110
Chen C, Yu R, Owuor ED, Kong ANT (2000) Activation of antioxidant-response element (ARE), mitogen-activated protein kinases (MAPKs) and caspases by major green tea polyphenol components during cell survival and death. Arch Pharmacal Res 23(6):605. https://doi.org/10.1007/bf02975249
Takeda H, Tsuji M, Matsumiya T, Kubo M (2002) Identification of rosmarinic acid as a novel antidepressive substance in the leaves of Perilla frutescens Britton var. acuta Kudo (Perillae Herba). Nihon Shinkei Seishin Yakurigaku Zasshi 22(1):15–22
Wei H, Saladi R, Lu Y et al (2003) Isoflavone genistein: Photoprotection and clinical implications in dermatology. J Nutr 133:3811S-3819S. https://doi.org/10.1093/jn/133.11.3811s
Ovadje P, Ammar S, Guerrero JA et al (2016) Dandelion root extract affects colorectal cancer proliferation and survival through the activation of multiple death signalling pathways. Oncotarget 7(45):73080–73100. https://doi.org/10.18632/oncotarget.11485
Jones NP, Schulze A (2012) Targeting cancer metabolism-aiming at a tumour’s sweet-spot. Drug Discov Today 17(5–6):232–241. https://doi.org/10.1016/j.drudis.2011.12.017
Duarte J, Pérez-Palencia R, Vargas F et al (2001) Antihypertensive effects of the flavonoid quercetin in spontaneously hypertensive rats. Br J Pharmacol 133(1):117–124. https://doi.org/10.1038/sj.bjp.0704064
Galisteo M, García-Saura MF, Jiménez R et al (2004) Effects of chronic quercetin treatment on antioxidant defence system and oxidative status of deoxycorticosterone acetate-salt-hypertensive rats. Mol Cell Biochem 259(1–2):91–99. https://doi.org/10.1023/b:mcbi.0000021360.89867.64
Kim KA, Yim JE (2015) Antioxidative activity of onion peel extract in obese women: a randomized, double-blind, placebo controlled study. J Cancer Prev 20(3):202–207. https://doi.org/10.15430/JCP.2015.20.3.202
Jahfar M, Vijayan K, Azadi P (2003) Studies on a polysaccharide from the fruit rind of Punica granatum. Res J Chem Environ 7:43–50
Li Y, Guo C, Yang J et al (2006) Evaluation of antioxidant properties of pomegranate peel extract in comparison with pomegranate pulp extract. J Food Chem 96(2):254–260. https://doi.org/10.1016/j.foodchem.2005.02.033
Mirdehghan SH, Rahemi M (2007) Seasonal changes of mineral nutrients and phenolics in pomegranate (Punica granatum L.) fruit. Scientia Horticulturae 111(2):120–127. https://doi.org/10.1016/j.scienta.2006.10.001
Nuncio-Jáuregui N, Calín-Sánchez Á, Vázquez-Araújo L et al (2015) Processing pomegranates for juice and impact on bioactive components. Beverages Food Sci Technol, 629–636. https://doi.org/10.1016/B978-0-12-404699-3.00076-7
Boyer J, Liu RH (2004) Apple phytochemicals and their health benefits. Nutr J 3:5. https://doi.org/10.1186/1475-2891-3-5
Wu CH, Ho YS, Tsai CY et al (2009) In vitro and in vivo study of phloretin-induced apoptosis in human liver cancer cells involving inhibition of type II glucose transporter. Int J Cancer 124(9):2210–2219. https://doi.org/10.1002/ijc.24189
Khan N, Syed DN, Ahmad N, Mukhtar H (2013) Fisetin: a dietary antioxidant for health promotion. Antioxid Redox Signal 19(2):151–162. https://doi.org/10.1089/ars.2012.4901
Chen RJ, Kuo HC, Cheng LH et al (2018) Apoptotic and nonapoptotic activities of pterostilbene against cancer. Int J Mol Sci 19(1):287. https://doi.org/10.3390/ijms19010287
Qian YY, Liu ZS, Yan HJ et al (2018) Pterostilbene inhibits MTA1/HDAC1 complex leading to PTEN acetylation in hepatocellular carcinoma. Biomed Pharmacother 101:852–859. https://doi.org/10.1016/j.biopha.2018.03.022
Siemann E, Creasy L (1992) Concentration of the phytoalexin resveratrol in wine. Am J Enol Viticult 43:49–52
Brown L, Kroon PA, Das DK et al (2009) The biological responses to resveratrol and other polyphenols from alcoholic beverages. Alcohol Clin Exp Res 33(9):1513–1523. https://doi.org/10.1111/j.1530-0277.2009.00989.x
Singh CK, George J, Ahmad N (2013) Resveratrol-based combinatorial strategies for cancer management. Ann N Y Acad Sci 1290(1):113–121. https://doi.org/10.1111/nyas.12160
Wu CP, Ohnuma S, Ambudkar SV (2011) Discovering natural product modulators to overcome multidrug resistance in cancer chemotherapy. Curr Pharm Biotechnol 12(4):609–620. https://doi.org/10.2174/138920111795163887
Sak K (2012) Chemotherapy and dietary phytochemical agents. Chemotherapy Res Pract 2012:1–11. https://doi.org/10.1155/2012/282570
Hassan B (2019) Plants and Cancer treatment prevention. https://doi.org/10.5772/intechopen.90568
Rodriguez S, Skeet K, Mehmetoglu-Gurbuz T et al (2021) Phytochemicals as an alternative or integrative option, in conjunction with conventional treatments for hepatocellular carcinoma. Cancers (Basel) 13(22):5753. https://doi.org/10.3390/cancers13225753
Mondal A, Guria T, Maity TK Bishayee A (2016) A novel tetraenoic fatty acid isolated from Amaranthus spinosus inhibits proliferation and induces apoptosis of human liver cancer cells. Int J Mol Sci 17(10):1604. https://doi.org/10.3390/ijms17101604
Kim DK, Baek JH, Kang CM et al (2000) Apoptotic activity of ursolic acid may correlate with the inhibition of initiation of DNA replication. Int J Cancer 87(5):629–636
Shyu MH, Kao TC, Yen GC (2010) Oleanolic acid and ursolic acid induce apoptosis in HuH7 human hepatocellular carcinoma cells through a mitochondrial-dependent pathway and downregulation of XIAP. J Agric Food Chem 58(10):6110–6118. https://doi.org/10.1021/jf100574j
Kim GH, Kan SY, Kang H et al (2019) Ursolic acid suppresses cholesterol biosynthesis and exerts anti-cancer effects in hepatocellular carcinoma cells. Int J Mol Sci 20(19):4767. https://doi.org/10.3390/ijms20194767
Ko H, Huh G, Jung SH et al (2020) Diospyros kaki leaves inhibit HGF/Met signaling-mediated EMT and stemness features in hepatocellular carcinoma. Food Chem Toxicol 142:111475
Durmaz I, Guven EB, Ersahin T et al (2016) Liver cancer cells are sensitive to Lanatoside C induced cell death independent of their PTEN status. Phytomedicine 23(1):42–51. https://doi.org/10.1016/j.phymed.2015.11.012
Dasgupta A, Dey D, Ghosh D et al (2019) Astrakurkurone, a sesquiterpenoid from wild edible mushroom, targets liver cancer cells by modulating Bcl-2 family proteins. IUBMB Life 71(7):992–1002. https://doi.org/10.1002/iub.2047
Weifeng T, Feng S, Xiangji L et al (2011) Artemisinin inhibits in vitro and in vivo invasion and metastasis of human hepatocellular carcinoma cells. Phytomedicine 18(2–3):158–162. https://doi.org/10.1016/j.phymed.2010.07.003
Jung KH, Rumman M, Yan H et al (2018) An ethyl acetate fraction of Artemisia capillaris (ACE-63) induced apoptosis and anti-angiogenesis via inhibition of PI3K/AKT signaling in hepatocellular carcinoma. Phytother Res 32(10):2034–2046. https://doi.org/10.1002/ptr.6135
Dai Q, Yin Q, Wei L et al (2016) Oroxylin A regulates glucose metabolism in response to hypoxic stress with the involvement of Hypoxia-inducible factor-1 in human hepatoma HepG2 cells. Mol Carcinog 55(8):1275–1289. https://doi.org/10.1002/mc.22369
Wei L, Dai Y, Zhou Y et al (2017) Oroxylin A activates PKM1/HNF4 alpha to induce hepatoma differentiation and block cancer progression. Cell Death Dis 8(7):e2944. https://doi.org/10.1038/cddis.2017.335
Peng W, Hu C, Shu Z et al (2015) Antitumor activity of tatariside F isolated from roots of Fagopyrum tataricum (L.) Gaertn against H22 hepatocellular carcinoma via up-regulation of p53. Phytomedicine 22(7–8):730–736. https://doi.org/10.1016/j.phymed.2015.05.003
Mohamed GA, Al-Abd AM, El-Halawany AM et al (2017) New xanthones and cytotoxic constituents from Garcinia mangostana fruit hulls against human hepatocellular, breast, and colorectal cancer cell lines. J Ethnopharmacol 198:302–312. https://doi.org/10.1016/j.jep.2017.01.030
Yoon JS, Kim HM, Yadunandam AK et al (2013) Neferine isolated from Nelumbo nucifera enhances anti-cancer activities in Hep3B cells: molecular mechanisms of cell cycle arrest, ER stress induced apoptosis and anti-angiogenic response. Phytomedicine 20(11):1013–1022. https://doi.org/10.1016/j.phymed.2013.03.024
Li M, Zhang M, Zhang ZL et al (2017) Induction of apoptosis by berberine in hepatocellular carcinoma HepG2 Cells via downregulation of NF-κB. Oncol Res 25(2):233–239. https://doi.org/10.3727/096504016x14742891049073
Kato R, Matsui-Yuasa I, Azuma H, Kojima-Yuasa A (2014) The synergistic effect of 10-acetoxychavicol acetate and sodium butyrate on the death of human hepatocellular carcinoma cells. Chem Biol Interact 212:1–10
Jiang F, Mu J, Wang X et al (2014) The repressive effect of miR-148a on TGF beta-SMADs signal pathway is involved in the glabridin-induced inhibition of the cancer stem cells-like properties in hepatocellular carcinoma cells. PLoS ONE 9(5):e96698. https://doi.org/10.1371/journal.pone.0096698
Hsieh MJ, Lin CW, Yang SF et al (2014) Glabridin inhibits migration and invasion by transcriptional inhibition of matrix metalloproteinase 9 through modulation of NF-κB and AP-1 activity in human liver cancer cells. Br J Pharmacol 171(12):3037–3050. https://doi.org/10.1111/bph.12626
Rawat D, Shrivastava S, Naik RA et al (2018) An overview of natural plant products in the treatment of hepatocellular carcinoma. Anticancer Agents Med Chem 18(13):1838–1859. https://doi.org/10.2174/1871520618666180604085612
Park JY, Ko JA, Kim DW et al (2016) Chalcones isolated from Angelica keiskei inhibit cysteine proteases of SARS-CoV. J Enzyme Inhib Med Chem 31(1):23–30. https://doi.org/10.3109/14756366.2014.1003215
Poofery J, Khaw-on P, Subhawa S et al (2020) Potential of Thai herbal extracts on lung cancer treatment by inducing apoptosis and synergizing chemotherapy. Molecules 25(1):231. https://doi.org/10.3390/molecules25010231
Kim DW, Seo KH, Curtis-Long MJ et al (2014) Phenolic phytochemical displaying SARS-CoV papain-like protease inhibition from the seeds of Psoralea corylifolia. J Enzyme Inhib Med Chem 29(1):59–63. https://doi.org/10.3109/14756366.2012.753591
Yeh CF, Wang KC, Chiang LC et al (2013) Water extract of licorice had anti-viral activity against human respiratory syncytial virus in human respiratory tract cell lines. J Ethnopharmacol 148(2):466–473. https://doi.org/10.1016/j.jep.2013.04.040
Tahir AH, Javed MM, Hussain Z (2020) Nutraceuticals and herbal extracts: a ray of hope for COVID-19 and related infections. Int. J. Funct. Nutr. 1(2):6. https://doi.org/10.3892/ijfn.2020.6
Park JY, Jeong HJ, Kim JH et al (2012) Diarylheptanoids from Alnus japonica inhibit papain-like protease of severe acute respiratory syndrome coronavirus. Bioorg Med Chem 5(11):2036–2042. https://doi.org/10.1248/bpb.b12-00623
Boukhatem MN, Setzer WN (2020) Aromatic herbs, medicinal plant-derived essential oils, and phytochemical extracts as potential therapies for coronaviruses: future perspectives. Plants 9(6):800. https://doi.org/10.3390/plants9060800
Ho TY, Wu SL, Chen JC et al (2007) Emodin blocks the SARS coronavirus spike protein and angiotensin-converting enzyme 2 interaction. Antivir Res 74(2):92–101. https://doi.org/10.1016/j.antiviral.2006.04.014
Miki K, Nagai T, Suzuki K et al (2007) Anti-influenza virus activity of biflavonoids. Bioorg Med Chem Lett 17(3):772–775. https://doi.org/10.1016/j.bmcl.2006.10.075
Boozari M, Hosseinzadeh H (2020) Natural products for COVID-19 prevention and treatment regarding to previous coronavirus infections and novel studies. Phytother Res 35(2):864–876. https://doi.org/10.1002/ptr.6873
Cho JK, Curtis-Long MJ, Lee KH et al (2013) Geranylated flavonoids displaying SARS-CoV papain-like protease inhibition from the fruits of Paulownia tomentosa. Bioorg Med Chem 21(11):3051–3057. https://doi.org/10.1016/j.bmc.2013.03.027
Park JY, Kim JH, Kim YM et al (2012) Tanshinones as selective and slow-binding inhibitors for SARS-CoV cysteine proteases. Bioorg Med Chem 20(19):5928–5935. https://doi.org/10.1016/j.bmc.2012.07.038
Chaotham C, Pongrakhananon V, Sritularak B, Chanvorachote P (2014) A Bibenzyl from Dendrobium ellipsophyllum inhibits epithelial-to-mesenchymal transition and sensitizes lung cancer cells to anoikis. Anticancer Res 34(4):1931–1938
Pinkhien T, Petpiroon N, Sritularak B, Chanvorachote P (2017) Batatasin III inhibits migration of human lung cancer cells by suppressing epithelial to mesenchymal transition and FAK-AKT signals. Anticancer Res 37(11):6281–6289. https://doi.org/10.21873/anticanres.12079
Hlosrichok A, Sumkhemthong S, Sritularak B et al (2018) A bibenzyl from Dendrobium ellipsophyllum induces apoptosis in human lung cancer cells. J Nat Med 72(3):615–625. https://doi.org/10.1007/s11418-018-1186-x
Zhu GF, Guo HJ, Huang Y et al (2015) Eriodictyol, a plant flavonoid, attenuates LPS-induced acute lung injury through its antioxidative and anti-inflammatory activity. Exp Ther Med 10(6):2259–2266. https://doi.org/10.3892/etm.2015.2827
Park JY, Kim JH, Kwon JM et al (2013) Dieckol, a SARS-CoV 3CLpro inhibitor, isolated from the edible brown algae Ecklonia cava. Bioorg Med Chem 21(13):3730. https://doi.org/10.1016/j.bmc.2013.04.026
Kuo MY, Liao MF, Chen FL et al (2011) Luteolin attenuates the pulmonary inflammatory response involves abilities of antioxidation and inhibition of MAPK and NFκB pathways in mice with endotoxin-induced acute lung injury. Food Chem Toxicol 49(10):2660–2666. https://doi.org/10.1016/j.fct.2011.07.012
Aguilar JL, Rojas P, Marcelo A et al (2002) Anti-inflammatory activity of two different extracts of Uncaria tomentosa (Rubiaceae). J Ethnopharmacol 81(2):271–276. https://doi.org/10.1016/s0378-8741(02)00093-4
Song YH, Kim DW, Curtis-Long MJ et al (2014) Papain-like protease (PLpro) inhibitory effects of cinnamic amides from Tribulus terrestris fruits. Biol Pharm Bull 37(6):1021–1028. https://doi.org/10.1248/bpb.b14-00026
Runfeng L, Yunlong H, Jicheng H et al (2020) Lianhuaqingwen exerts anti-viral and anti-inflflammatory activity against novel coronavirus (SARS-CoV-2). Pharmacol Res 156:104761. https://doi.org/10.1016/j.phrs.2020.104761
Jo S, Kim S, Shin DH, Kim MS (2020) Inhibition of SARS-CoV 3CL protease by flavonoids. J Enzyme Inhib Med Chem 35(1):145–151. https://doi.org/10.1080/14756366.2019.1690480
Mortaz E, Adcock IM, Folkerts G et al (2013) Garssen, probiotics in the management of lung diseases. Mediat Inflamm 2013:751068. https://doi.org/10.1155/2013/751068
Gengaihi S, Baker DHA (2017) Grape seeds extract as brain food: a review. Int J Pharm Clin Res 9(1):77–85. https://doi.org/10.25258/ijpcr.v9i1.8270
Yu MS, Lee J, Lee JM et al (2012) Identifification of myricetin and scutellarein as novel chemical inhibitors of the SARS coronavirus helicase, nsP13. Bioorg Med Chem Lett 22(12):4049–4054. https://doi.org/10.1016/j.bmcl.2012.04.081
Ryu YB, Park SJ, Kim YM et al (2010) SARS-CoV 3CLpro inhibitory effects of quinone-methide triterpenes from Tripterygium regelii. Bioorg Med Chem Lett 20(6):1873–1876. https://doi.org/10.1016/j.bmcl.2010.01.152
Weng JR, Lin CS, Lai HC et al (2019) Antiviral activity of Sambucus FormosanaNakai ethanol extract and related phenolic acid constituents against human coronavirus NL63. Virus Res 273:197767. https://doi.org/10.1016/j.virusres.2019.197767
Benarba B, Pandiella A (2020) Medicinal plants as sources of active molecules against COVID-19. Front Pharmacol 11:1189. https://doi.org/10.3389/fphar.2020.01189
Rahman MM, Bibi S, Rahaman MS et al (2022) Natural therapeutics and nutraceuticals for lung diseases: traditional significance, phytochemistry, and pharmacology. Biomed Pharmacother 150:113041. https://doi.org/10.1016/j.biopha.2022.113041
Acknowledgements
The authors thank Vellore Institute of Technology, Vellore for providing facilities to carry out the work.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2023 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Mondal, H., Chandrasekaran, N., Mukherjee, A., Thomas, J. (2023). Bioinformatics Study on Renin Angiotensin in Lung, and Liver Cancer Using Plant-Based Extracts. In: Bhullar, S.K., Tappia, P.S., Dhalla, N.S. (eds) The Renin Angiotensin System in Cancer, Lung, Liver and Infectious Diseases. Advances in Biochemistry in Health and Disease, vol 25. Springer, Cham. https://doi.org/10.1007/978-3-031-23621-1_9
Download citation
DOI: https://doi.org/10.1007/978-3-031-23621-1_9
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-23620-4
Online ISBN: 978-3-031-23621-1
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)