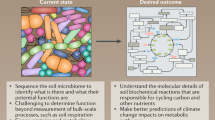
Abstract
The biogeochemical cycling of macronutrients, micronutrients, and other components necessary for the development of plants and animal life is governed by the soil microbiome. As we focus our research efforts on one of the most serious issues affecting our planet, knowing and anticipating how climate change will affect soil microbiomes and the ecosystem services they provide is a huge challenge and significant potential. Studies predict that factors related to climate change, such as elevated atmospheric [CO2] and heat, will function together to change ecosystem features and processes, influencing species distributions and, presumably, organism interactions. On the other hand, it is harder to forecast how the microbial populations that control ecological processes would respond. In complex ecosystems, organisms interact with thousands of different species, some of which are useful, some of which are poisonous, and some of which have little to no impact. In this chapter, we examine the present level of knowledge about the effects of climate change on soil microorganisms in various climate-sensitive soil ecosystems, as well as prospective approaches that soil microorganisms may be used in to help lessen the detrimental effects of climate change.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
Introduction
Although soil is one of the planet's most diverse environments, it is also one of the least understood in terms of the identification and ecological functions of the microbiota. A significant amount of the annual CO2 flow to the atmosphere is caused by the activities of heterotrophic soil organisms, which also serve as the greatest repository of organic carbon (C) in the terrestrial biosphere. Global temperatures have risen in step with constantly rising CO2 levels. According to the most recent US national climate assessment [1,2,3,4], the climate is expected to continue to change with more unpredictable and intense weather patterns. Since soil microbes play a major part in the cycling of nutrients and soil organic carbon (SOC), they also play a significant part in the production and consumption of greenhouse gases like CO2, CH4, and N2O. However, due to unknown modifications in soil carbon and nitrogen stores, as well as variations in microbial responses between different soil locations, it has been challenging to predict whether soil will act as a source or sink of greenhouse gases under future climate scenarios [5,6,7,8]. Therefore, despite the fact that soil microbial ecology is crucial for predicting future climate impacts, integrating it with landscape-scale climate models is still difficult. The fact that soil microbes would mineralize more SOC and significantly increase greenhouse gas (CO2 and CH4) emissions, aggravating warming trends, is one of the main concerns with climate change [9, 10]. This is concerning since the overall amount of soil carbon, including that found in permafrost, is thought to be around 3,300 pentagrams (Pg), which is around five times more than the amount of CO2 present in the atmosphere today [11, 12]. The future growth or decline of this stock of soil carbon is, however, highly unknown according to climate models. Measuring variations in soil respiration has been the main source of empirical data for field studies on climate change. Determining how bulk soil carbon reserves vary with climatic changes is also necessary in order to enhance models of soil carbon-climate feedback [13, 14].
Soil microbes perform the dual functions of mineralizing SOC and stabilizing carbon inputs into organic forms. The net flux of CH4 and CO2 to the atmosphere is controlled by the balance between these two processes. The microbial carbon utilization efficiency is the portion of the carbon substrate that is kept in the microbial biomass as opposed to that which is respired as CO2. Climate change has increased heterotrophic respiration of SOC globally, which has increased atmospheric CO2 inputs [15]. However, higher soil carbon inputs resulting from increased plant growth [16] and autotrophic fixation by soil microbes could offset soil carbon losses to the atmosphere. Additionally, the amount, content, and chemistry of plant litter as well as any pre-existing SOC affect how sensitively SOC decomposes at different temperatures [17]. Thus, even within certain biomes, the local biogeochemical environment has a significant impact on how organisms respond metabolically to climate. In order to improve climate change models, it is imperative to develop a mathematical understanding of the microbial ecology that drives ecosystem carbon use efficiency and the feedback with climate forcing.
Effects of the Soil Microbiome on the Characteristics of Emerging Ecosystems
The majority of soil microorganisms have developed coping mechanisms to deal with shifting environmental conditions because soil habitats are dynamic systems. The resident microorganisms often adapt, go dormant, or perish when the environment changes. Depending on their genetic and physiological conditions, soil microorganisms respond to environmental stress in various ways [18].
The degree of disruption and the amount of time required to control gene transcription and translation, as well as to amass mutations or new genes through horizontal gene transfer, determine how quickly an organism adapts to change. Quantifying microbial physiological responses, such as drought resistance, dormancy, or reactivation, nonetheless, continues to be a significant challenge in modeling ecological responses to change at the moment [19].
The stability and resistance of the microbial community to future perturbations may change as the community's structure does. The ability of a single species to adapt will be impacted by the interactions between microbial populations in communities as a result of climate change [20]. As a result of differences in how various species react to temperature increases, for instance, their dispersal patterns may shift. It is possible to predict how the soil microbiome will react to various climate change scenarios by focusing on specific functional traits in the soil microbiome, such as the prevalence of fast-growing, opportunistic “r-strategists” as opposed to slow-growing “K-strategists,” as well as environmental characteristics [21].
In order to establish a useful baseline for comparison as the climate changes, high-throughput sequencing has proven crucial in exposing the microbial diversity and composition in distinct soil ecosystems. However, it is now understood that compositional data does not always guide function Not every participant in a group, or even every cell within a population, is operational at all times [22]. The complicated interplay of gene regulation primarily controls which genes are expressed and access to resources, controls activity. The soil microbiome's phenotypic response to climate change is impacted by variations in soil moisture, temperature, and local atmospheric chemistry.
Microbial gene expression is induced by the interaction of the heterogeneous genetic potential within the soil microbiome with environmental changes. The metaphenome, which is the microbiome's collective phenotypic output, produces elemental cycling at the ecosystem level [19]. Soil microbiome management in response to climate change and the improvement of climate models depend heavily on our understanding of the factors that link small-scale microbial traits to larger-scale ecosystem responses.
The underlying bacterial-scale mechanisms that regulate environment responses to climate change are currently poorly defined. Instead of reacting to average environmental conditions, soil microorganisms react to sudden microscale variables that set off biochemical pathways, microbial reflexes, and metabolic relationships. Temporal pauses in biogeochemical responses to sudden environmental change are common as soil microorganisms acclimatize. Contrarily, a slow change, like a rise in temperature, gives evolution more time to select for organisms or genotypes that permit endurance to the stress circumstances brought on by the heat. The response of the community is also influenced by its historical background.
Influence of the Soil Environment on Microbial Responses to Climate Change
It is challenging to generalize the effects of climate change on soil microbiomes across various soil ecosystems due to the vast differences amongst soils in terms of their biotic and abiotic features. There are variations in biogeochemistry within a certain soil class that control the kinds of microorganisms that are present, including pH [23] and salinity [24]. Furthermore, the microbial dwellings and niches [25] that are created in the soil are influenced by its morphology and water content, which has a domino effect on the metabolism of nutrients and carbon. To better understand how species relationships and metabolism are impacted by climate change, it is necessary to study the fine-scale dispersion and interconnectivity of microbial communities in soils [26]. This data is crucial for understanding carbon cycling because how soil bacteria species distribute carbon eventually defines whether or not it persists in soil and how changes in climate alter such processes [27]. It is well recognized that population of microbes communicate and respire CO2, N2O, and CH4 in different soil niches, but the energetics and thermodynamic parameters of the organic carbon electron acceptors that run microbial metabolism are poorly understood in the context of the soil environment. The description of the physiological response surface, or metaphenome, of the microbial communities living in the soils of our planet is the current challenge.
Effects of Environmental Change
There are many physiological and community responses that soil microorganisms adopt to adapt to the changing environmental conditions brought on by climate change. Due to the varied expected climate change variables across geographic locations, it is impossible to generalize across diverse terrestrial ecosystems, which is why we present some instances to provide context.
Raised Carbon Dioxide (CO2)
Data from a number of eCO2 field studies has been useful in understanding how microbes may come to this impending climate change. Data from a number of eCO2 field studies has been useful in understanding how microbes may come to this impending climate change. In order to contrast prolonged exposure to increased and atmospheric CO2 levels, FACE (free-air CO2 enrichment) experiments been arranged across a variety of ecoregions. The microbiota has changed with eCO2, according to several investigations. Ecosystem-specific responses in addition to typical soil bacterial responses, like with eCO2, acido-bacterial rates increase, found by a one-decade cross-biome investigation [28]. eCO2 led to a shift in archaea and fungus and bacterial strain species in Australian grasslands. Researchers are being diligent to comprehend how ecological characteristics of microbial communities are mirrored by phylogenetic shifts. A foundation for incorporating microbial physiology into ecosystem ecology is provided by a gene-based approach.
By examining the abundance of particular genes in metagenomes, changes in the potential roles played by the soil microbiome under eCO2 have also been identified. By examining the abundance of particular genes in metagenomes, changes in the potential roles played by the soil microbiome under eCO2 have also been identified [29]. In the BioCON grassland experiment, eCO2-stimulated increases in gene families linked to decomposition, nitrogen fixation, and dissimilatory nitrate reduction were observed, while fewer abundances of gene families linked to glutamine formation and anaerobic ammonium oxidation were found. Genes of microbes involved in breakdown, nitrogen fixation, carbon fixation, CH4 metabolism, nitrogen mineralization, and denitrification were all upregulated in arid grasslands exposed to eCO2 [30].
Understanding the changes in gene activities related to the cycling of organic matter in soil (SOM) allows for a better comprehension of how eCO2 affects microorganisms. However, it is still problematic to provide information for globally terrestrial ecosystem models because eCO2 trials have not been conducted widely with duplicate data sets.
The quantification of carbon exchange between the atmosphere and the soil is a key scientific area of plant–microbe connections. Plant biomass, carbon uptake by roots, and soil microbial activity can all be improved by eCO2. An important scientific area for measuring carbon exchange in between environment and the topsoil is plant–microbe relationship. Equivalent CO2 can improve soil microbial activity, carbon sharing to roots, and plant biomass [31,32,33].
The frequency and pattern of carbon imports to the rhizosphere are influenced by how various species of plants react to elevated CO2. The eCO2-induced rise in rhizodeposition can ‘prime’ the microbial breakdown of existing SOC [34]. Priming is the process of speeding the degradation of old SOC by introducing new microbiological feedstock, such as production of litter and/or root exudates, both of which could be accelerated by elevated CO. A review consolidating meta-examination and demonstration uncovered that eCO2 at first invigorates photosynthesis and carbon contributions to soil. In any case, over decadal timescales, eCO2 expanded the microbial deterioration of SOM [35, 36]. Anticipating the balance between carbon gathering through mineral affiliation and soil aggregation [37] and sped up decay via priming [38] stays an extra test. This is on the grounds that adjustments of soil carbon stocks are hard to find [39] and the basic science managing SOM deterioration has not been found. The soil may become drier as temperature increases together with a rise in the soil's wetness brought on by elevated CO2 [28]. In the Australian grassland study [28], when eCO2 was linked with warming, there was a decline, even though overall fungal richness expanded under elevated CO2. The supply of water and micronutrients, that affect photosynthesis, microbial breakdown, and the net buildup of carbon sequestration, also affects the indelible effects of elevated CO2 on soil C reserves. Predicting the responses of soil ecosystems' microbiota composition to variations in CO2 necessitates a comprehension of how such changes react with other significant environmental parameters such as temperature, precipitation, and nutrients (such as phosphorus).
Elevated Temperature
The growth rates and outputs of pure microbial cultures are impacted by temperature. The expression of heat shock proteins and alterations in the lipid content of cell membranes, which diminish membrane integrity, are two physiological reactions of microbes to elevated temperature. The growth rates and outputs of pure microbial cultures are impacted by temperature. The expression of heat shock proteins and alterations in the lipid content of cell membranes, which diminish membrane integrity, are two physiological reactions of microbes to elevated temperature. Although technological developments in sequence analysis and functional gene assays have showed colony and functional gene alterations in result of higher temperatures in the fieldwork, evaluating the temperature sensitivity of soil microbes in situ has proven to be more challenging [39, 40]. The biome being examined also influences how the soil microbiome reacts to rising temperatures (for instance, distinguishing between woodland and grassland). For illustration, temperature rise has been demonstrated to have differential effects on soil fungi in various coniferous forest ecosystems, leading to either stimulation [41] or suppression of fungal biomass and activity. These variations are likely caused by variations in soil moisture and/or vegetation at various points [42, 43]. A long-term soil warming experiment was carried out at the Harvard Forest Ecological Research Station Long Term Ecological Research site, wherein soil was thawed by 5 °C above ambient temperature for up to 26 years in order to assess the effects of prolonged soil warming on the soil microbiome of temperate forests [38, 43].
Short-term reductions in microbial biomass and temperature adaptation of soil respiration were implicated for the apparent acclimation of soil respiration [13]. The physiological adaptations of various populations must yet be measured in a field setting. To fill this knowledge vacuum and measure microbial population changes in the field, new isotopic techniques are now available.
The interaction between drought, heat, and plant type ultimately decides how tolerant bacterial communities are to extreme heat. On Wyoming grasslands, the Prairie Heating and CO2 Enrichment (PHACE) experiment investigated the effects of twelve years of elevated CO2 coupled with warming [44]. Under eCO2 itself and in conjunction with warming, genetic variants in the recycling of nitrogen and carbon were amplified. However, heat alone suppressed nitrogen turnover. Variations in precipitation being magnified by the favorable flora community response, which resulted in a rise in biomas [45]. The enhanced plant biomass thereby largely countered the rising carbon loss via respiration, even while warming accelerated both the carbon intake into soil and soil respiration. Collectively, those actions would work to diminish the global warming's positive feedback loop and halt soil C loss. To sum up, whereas most climate analysis shows positive feedback as a result of warming due to increased soil respiration and a decrease in soil storage, there are confounding experimental data that are mostly ecosystem dependent [7, 8, 41].
References
Reidmiller DR, Avery CW, Easterling DR, Kunkel KE, Lewis KL, Maycock TK,Stewart BC (2017) Impacts, risks, and adaptation in the United States: Fourth national climate assessment, volume II (USGCRP, 2018). https://doi.org/10.7930/NCA4.2018
Shaban H, Fazeli-Nasab B, Alahyari H, Alizadeh G, Shahpesandi S (2015) An overview of the benefits of compost tea on plant and soil structure. Adv Biores 6(61):154–158. https://doi.org/10.15515/abr.0976-4585.6.1.154158
Ali-Soufi M, Shahriari A, Shirmohammadi E, Fazeli-Nasab B (2017) Investigation of biological properties and microorganism identification in susceptible areas to wind erosion in Hamoun wetlands. In: Congress on restoration policies and approaches of Hamoun international wetland Zabol, pp 231–240
Ali-Soufi M, Shahriari A, Shirmohammadi E, Fazeli-Nasab B (2017) Seasonal changes biological characteristics of airborne dust in Sistan plain, Eastern Iran. In: International conference on loess research. Gorgan University of Agricultural Sciences and Natural Resources, Gorgan, Iran
Friedlingstein P, Cox P, Betts R, Bopp L, von Bloh W, Brovkin V, Cadule P, Doney S, Eby M, Fung I (2006) Climate–carbon cycle feedback analysis: results from the C4MIP model intercomparison. J Clim 19(14):3337–3353. https://doi.org/10.1175/JCLI3800.1
Wang K, Peng C, Zhu Q, Zhou X, Wang M, Zhang K, Wang G (2017) Modeling global soil carbon and soil microbial carbon by integrating microbial processes into the ecosystem process model TRIPLEX-GHG. J Adv Model Earth Syst 9(6):2368–2384. https://doi.org/10.1002/2017MS000920
Ali-Soufi M, Shahriari A, Shirmohammadi E, Fazeli-Nasab B (2017) Identification and isolation of associated microorganisms with airborne dust loaded over Sistan plain. In: 15th Iranian Soil Science Congress, Isfahan University of Technology, Isfahan, Iran, Congress COI: SSCI15, Article COI: SSCI15_895
Ali-Soufi M, Shahriari A, Shirmohammadi E, Fazeli-Nasab B (2019) Investigation of dust microbial community and identification of its dominance species in northern regions of sistan and baluchestan province. J Water Soil Sci (Science and Technology of Agriculture and Natural Resources) 23(1):309–320. https://doi.org/10.29252/jstnar.23.1.23
Wieder WR, Bonan GB, Allison SD (2013) Global soil carbon projections are improved by modelling microbial processes. Nat Clim Chang 3(10):909–912. https://doi.org/10.1038/nclimate1951
Rasouli H, Popović-Djordjević J, Sayyed RZ, Zarayneh S, Jafari M, Fazeli-Nasab B (2020) Nanoparticles: a new threat to crop plants and soil rhizobia? In: Hayat S, Pichtel J, Faizan M, Fariduddin E (eds) Sustainable agriculture reviews 41: nanotechnology for plant growth and development. Springer International Publishing, Cham, pp 201–214. https://doi.org/10.1007/978-3-030-33996-8_11
Tarnocai C, Canadell J, Schuur EA, Kuhry P, Mazhitova G, Zimov S (2009) Soil organic carbon pools in the northern circumpolar permafrost region. Glob Biogeochem Cycles 23(2). https://doi.org/10.1029/2008GB003327
Bruhwiler L, Basu S, Butler JH, Chatterjee A, Dlugokencky E, Kenney MA, McComiskey A, Montzka SA, Stanitski D (2021) Observations of greenhouse gases as climate indicators. Clim Change 165(1):1–18. https://doi.org/10.1007/s10584-021-03001-7
Bradford MA, Davies CA, Frey SD, Maddox TR, Melillo JM, Mohan JE, Reynolds JF, Treseder KK, Wallenstein MD (2008) Thermal adaptation of soil microbial respiration to elevated temperature. Ecol Lett 11(12):1316–1327. https://doi.org/10.1111/j.1461-0248.2008.01251.x
Dhuldhaj UP, Malik N (2022) Global perspective of phosphate soliloquizing microbes and phosphatase for improvement of soil, food and human health. Cell, Mol Biomed Rep 2(3):173–186. https://doi.org/10.55705/cmbr.2022.347523.1048
Bond-Lamberty B, Bailey VL, Chen M, Gough CM, Vargas R (2018) Globally rising soil heterotrophic respiration over recent decades. Nature 560(7716):80–83. https://doi.org/10.1038/s41586-018-0358-x
Rustad L, Campbell J, Marion G, Norby R, Mitchell M, Hartley A, Cornelissen J, Gurevitch J (2001) A meta-analysis of the response of soil respiration, net nitrogen mineralization, and aboveground plant growth to experimental ecosystem warming. Oecologia 126(4):543–562. https://doi.org/10.1007/s004420000544
Nottingham AT, Whitaker J, Turner BL, Salinas N, Zimmermann M, Malhi Y, Meir P (2015) Climate warming and soil carbon in tropical forests: insights from an elevation gradient in the Peruvian Andes. Bioscience 65(9):906–921. https://doi.org/10.1093/biosci/biv109
Schimel J, Balser TC, Wallenstein M (2007) Microbial stress-response physiology and its implications for ecosystem function. Ecology 88(6):1386–1394. https://doi.org/10.1890/06-0219
Evans SE, Wallenstein MD (2014) Climate change alters ecological strategies of soil bacteria. Ecol Lett 17(2):155–164. https://doi.org/10.1111/ele.12206
Berg MP, Kiers ET, Driessen G, Van Der Heijden M, Kooi BW, Kuenen F, Liefting M, Verhoef HA, Ellers J (2010) Adapt or disperse: understanding species persistence in a changing world. Global change Biol 16(2):587–598. https://doi.org/10.1111/j.1365-2486.2009.02014.x
De Vries FT, Shade A (2013) Controls on soil microbial community stability under climate change. Front Microbiol 4:265. https://doi.org/10.3389/fmicb.2013.00265
Thompson LR, Sanders JG, McDonald D, Amir A, Ladau J, Locey KJ, Prill RJ, Tripathi A, Gibbons SM, Ackermann G (2017) A communal catalogue reveals Earth’s multiscale microbial diversity. Nature 551(7681):457–463. https://doi.org/10.1038/nature24621
Jansson JK, Hofmockel KS (2018) The soil microbiome—from metagenomics to metaphenomics. Curr Opin Microbiol 43:162–168. https://doi.org/10.1016/j.mib.2018.01.013
Fierer N, Jackson RB (2006) The diversity and biogeography of soil bacterial communities. Proc Natl Acad Sci 103(3):626–631. https://doi.org/10.1073/pnas.0507535103
Schimel JP, Schaeffer SM (2012) Microbial control over carbon cycling in soil. Front Microbiol 3:348. https://doi.org/10.3389/fmicb.2012.00348
Cordero OX, Datta MS (2016) Microbial interactions and community assembly at microscales. Curr Opin Microbiol 31:227–234. https://doi.org/10.1016/j.mib.2016.03.015
Dunbar J, Eichorst SA, Gallegos-Graves LV, Silva S, Xie G, Hengartner N, Evans RD, Hungate BA, Jackson RB, Megonigal JP (2012) Common bacterial responses in six ecosystems exposed to 10 years of elevated atmospheric carbon dioxide. Environ Microbiol 14(5):1145–1158. https://doi.org/10.1111/j.1462-2920.2011.02695.x
Hayden HL, Mele PM, Bougoure DS, Allan CY, Norng S, Piceno YM, Brodie EL, DeSantis TZ, Andersen GL, Williams AL (2012) Changes in the microbial community structure of bacteria, archaea and fungi in response to elevated CO2 and warming in an Australian native grassland soil. Environ Microbiol 14(12):3081–3096. https://doi.org/10.1111/j.1462-2920.2012.02855.x
Allison S (2012) A trait-based approach for modelling microbial litter decomposition. Ecol Lett 15(9):1058–1070. https://doi.org/10.1111/j.1461-0248.2012.01807.x
Tu Q, He Z, Wu L, Xue K, Xie G, Chain P, Reich PB, Hobbie SE, Zhou J (2017) Metagenomic reconstruction of nitrogen cycling pathways in a CO2-enriched grassland ecosystem. Soil Biol Biochem 106:99–108. https://doi.org/10.1016/j.soilbio.2016.12.017
Yu H, Deng Y, He Z, Van Nostrand JD, Wang S, Jin D, Wang A, Wu L, Wang D, Tai X (2018) Elevated CO2 and warming altered grassland microbial communities in soil top-layers. Front Microbiol 9:1790. https://doi.org/10.3389/fmicb.2018.01790
Bréchet LM, Lopez‐Sangil L, George C, Birkett AJ, Baxendale C, Castro Trujillo B, Sayer EJ (2018) Distinct responses of soil respiration to experimental litter manipulation in temperate woodland and tropical forest. Ecol Evol 8(7):3787–3796. https://doi.org/10.1002/ece3.3945
Abbasi-Moghadam J, Shahriari A, Fazeli-Nasab B (2017) Investigation of bacteria and fungi populations associated with airborne dust during ‘’wind of 120 days’’ blowing in the urban areas of Sistan plain. In: 15th Iranian soil science congress. Isfahan University of Technology, Isfahan, Iran, Congress COI: SSCI15, Article COI: SSCI15_687
Qiao N, Schaefer D, Blagodatskaya E, Zou X, Xu X, Kuzyakov Y (2014) Labile carbon retention compensates for CO2 released by priming in forest soils. Glob Change Biol 20(6):1943–1954. https://doi.org/10.1111/gcb.12458
Van Groenigen KJ, Qi X, Osenberg CW, Luo Y, Hungate BA (2014) Faster decomposition under increased atmospheric CO2 limits soil carbon storage. Science 344(6183):508–509. https://doi.org/10.1126/science.1249534
Drake JE, Gallet-Budynek A, Hofmockel KS, Bernhardt ES, Billings SA, Jackson RB, Johnsen KS, Lichter J, McCarthy HR, McCormack ML (2011) Increases in the flux of carbon belowground stimulate nitrogen uptake and sustain the long-term enhancement of forest productivity under elevated CO2. Ecol Lett 14(4):349–357. https://doi.org/10.1111/j.1461-0248.2011.01593.x
Lal R (2004) Soil carbon sequestration impacts on global climate change and food security. Science 304(5677):1623–1627. https://doi.org/10.1126/science.1097396
Kuzyakov Y (2010) Priming effects: interactions between living and dead organic matter. Soil Biol Biochem 42(9):1363–1371. https://doi.org/10.1016/j.soilbio.2010.04.003
Scharlemann JP, Tanner EV, Hiederer R, Kapos V (2014) Global soil carbon: understanding and managing the largest terrestrial carbon pool. Carbon Manag 5(1):81–91. https://doi.org/10.4155/cmt.13.77
Zhang B, Chen S, He X, Liu W, Zhao Q, Zhao L, Tian C (2014) Responses of soil microbial communities to experimental warming in alpine grasslands on the Qinghai-Tibet Plateau. PLoS ONE 9(8):e103859. https://doi.org/10.1371/journal.pone.0103859
Heimann M, Reichstein M (2008) Terrestrial ecosystem carbon dynamics and climate feedbacks. Nature 451(7176):289–292. https://doi.org/10.1038/nature06591
Zhu X, Liu M, Kou Y, Liu D, Liu Q, Zhang Z, Jiang Z, Yin H (2020) Differential effects of N addition on the stoichiometry of microbes and extracellular enzymes in the rhizosphere and bulk soils of an alpine shrubland. Plant Soil 449(1):285–301. https://doi.org/10.1007/s11104-020-04468-6
Yang N, Zhang Y, Li J, Li X, Ruan H, Bhople P, Keiblinger K, Mao L, Liu D (2022) Interaction among soil nutrients, plant diversity and hypogeal fungal trophic guild modifies root-associated fungal diversity in coniferous forests of Chinese Southern Himalayas. Plant Soil 1–14. https://doi.org/10.1007/s11104-022-05646-4
Bafana A (2013) Diversity and metabolic potential of culturable root-associated bacteria from origanum vulgare in sub-himalayan region. World J Microbiol Biotechnol 29(1):63–74. https://doi.org/10.1007/s11274-012-1158-3
Carol Adair E, Reich PB, Trost JJ, Hobbie SE (2011) Elevated CO2 stimulates grassland soil respiration by increasing carbon inputs rather than by enhancing soil moisture. Global Change Biol 17(12):3546–3563. https://doi.org/10.1111/j.1365-2486.2011.02484.x
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2023 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Nazir, A., Farooq, M., Farooq, B., Anjum, S., Yousuf, S. (2023). Soil Microbial Community and Climate Change Drivers. In: Parray, J.A. (eds) Climate Change and Microbiome Dynamics. Climate Change Management. Springer, Cham. https://doi.org/10.1007/978-3-031-21079-2_8
Download citation
DOI: https://doi.org/10.1007/978-3-031-21079-2_8
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-21078-5
Online ISBN: 978-3-031-21079-2
eBook Packages: Earth and Environmental ScienceEarth and Environmental Science (R0)