Abstract
This chapter discusses concerning land use shifting influences to the soil microorganisms dynamic, especially in Indonesia where the biggest tropical rain forest established. Indonesia is among the region with largest tropical rain forest in the world. The country is also rich in plants biodiversity associated with the biophysical and the climate conditions forming the tropical rain forest. The high of plant diversity of Indonesia forest is illustrated by Malik et al. (Jurnal Ilmiah Pendidikan Sains 1:35–42, 2020), in Kalimantan in a hectar of forest can be identified more than 150 species.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
Introduction
Kusmana and Hikmat [1] summarized, despite the fact large of terrestrial region of Indonesia is only about 1.3% from total of the earth, 25% of world seed plants (spermatophytes) species are distributed in Indonesia. Hence, Indonesia is positioned as the 7th world plant biodiversity with about 20,000 numbers of species. Among the 20,000 species, 40% are endemic species (origin) of Indonesia. The most abundance family is belong to Orchidaceae that is reached 4,000 species, followed by Dipterocarpaceae with 386 species numbers (70% of dipterocarps population in the world), Myrtaceae and Moraceae (each 500 species numbers); Ericaceae (737 species), involved Rhododendron and Naccinium with 287 and 239 species numbers, respectively. Malik et al. [2] noticed that Indonesia is the producer of 75% of world rattan.
Malik et al. [2] reviewed forest of Indonesia colonized by the world highest palm family (Arecaceae), for instance there is 122 species numbers of bamboo. Kusmana and Hikmat [1] also reported that Indonesia has a high diversity of ferns about 4,000 species numbers, rattan about 332 species numbers involved of big stems of Genera Calamus (204 species) and Genera of Daemonorops (86 species). On the other hand, Indonesia also noticed as the center of distribution of Vavilov (biodiversity of cultivated plants) such as Syzygium aromaticum, Nephelium spp., Musa spp., Durio spp., and Myristica fragrans (ref).
However, along with the increase of population and development, lots of forests convert into many land utilization caused a deforestation phenomenon. Referring to [3], sometimes deforestation is planned for infrastructure development even it gave several negative impacts to the environment. It was reported, forest coverage of Indonesia during in 12 years (1985–1997) was drastically reduced from 119 million ha to 95 million ha [4]. Several activities such as intensive forest management, illegal logging, mining, agriculture, transmigration, forest fire, and land grabbing are indicated as the major reason for deforestation and forest degradation [5]. In addition, the excessive oilpalm plantation and mining activities in the forest area lead to enormously increase of forest vulnerability [4].
Plant is a sessile organism hence they need assistance from various microbes living around them for reaching nutrients, growth factors, and safeguard against pathogens. Plants actively initiate in assembling a favorable environment to invite beneficial microbes colonized around their root system. On the other hand, vegetation (species, stage of growth, etc.) determine structure and composition of soil microbes [6]. Various studies shown that many plant-microbes association have a remarkable impact on germination of seeds; vigor of seedlings; plant nutrition; plant disease; as well as plant growing, development and yield [7].
Berg and Smalla [8] have summarized from earlier studies, essentially every plant species requires a set of microorganism communities in its rhizosphere, both to support its growth (nutrient availability and growth factors) and its specific health (biocontrol and anti-pathogens). Therefore, it is crucial to consider knowledge on the plant-rhizosphere community interrelation in developing strategy for soil treatments, multi-species cropping, and crop rotations. The characteristic of plant species is vital for biological control applications. Moreover, it is also important to recognize the existing specific association among plants and microbes in correlation to issues of nature conservation. It means that once a plant species distinct, soil rhizosphere community will be disturbed.
Pitman and Jorgensen [9] discovered approximately 22–47% of the world’s plants are threatened with extinction. Unknown microbial diversity may be impacted when plants become extinct. Improved understanding on specific interactions among plants with microorganisms in their rhizosphere is useful for reforestation activities that include replanting degraded forests and woodlands with native tree stock. It is also reported, the interactions among microorganism and plant are crucial issues influencing the invasive species competition with the indigenous flora. Hence, the influence of climate-change on interaction among vegetations and microorganisms, i.e. on plant diseases, is also urgent to be calculated.
The activities causing to forest coverage changing is presumed to give many alterations to the underground organisms, involved soil microorganisms. Furthermore, this chapter discussing review results on the dynamic and function of forest coverage related to development and planning from previous publications (journals, IOP proceedings, books, reports, etc.), especially focused on (1) the dynamic of soil microbe under forest harvesting/tree cutting; (2) the role of soil microbes to the succession of pioneer in the secondary forest, involved to the invasive alien species distribution, (3) the alteration of soil microbes population due to land use change from natural to monoculture plantation, (4) responsibility of soil microorganisms on the mining land and the limitations to reclamation achievement.
The Dynamic of Soil Microbes Under Forest Harvesting/Tree Cutting
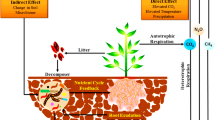
Plants are the initiator in rhizosphere configuration and controlling the composition and structure of root-microbial communities by releasing diverse organic compounds from photosynthesis [10]. It is estimated at 10–30% of photo-synthate [11], collectively labeled as root exudates [12] released to the root zone, for attracting soil microorganism and creating an unique environment known as the rhizosphere [13].
The rhizosphere recognized as the confine zone around and impacted by roots, is a hotspot for a variety of organisms and is the most dynamic ecosystems [14, 15]. In the rhizosphere is colonized by nematodes, arthropods, protozoa, algae, archaea, bacteria, fungi, oomycetes, and viruses [16, 15]. Most of them compose the complexity of food web using the large proportion of nutrients supplied by the plant, involved root exudates, border cells, mucilage [7]. The root exudates is a major driving force, with functions to attract and deter soil microbes hence the structure, size, and array of rhizosphere colonization match with the types, growth, and the stage of plant development [17, 7, 6]. Berg and Smalla [8] concluded, the rhizosphere is the important area for plant nutrition, health and productivity. Rhizosphere determine nutrient cycling in terrestrial ecosystems and ecosystem functioning.
Therefore, tree harvesting is perhaps the most harmful to trees since it removes all plant portions that operate as photosynthetic patches. This is an important process in the manufacturing of root exudates. Kӧgel-Knabner [18] found a half portion of root exudates is released as sugars, the main source of carbon for soil microbes [19]. Furthermore, tree felling is thought to influence the rhizosphere's interaction between plants and microbes.
Earlier studies, on a larger scale, the practice of forest harvesting conducted by clear-cutting. It removes in excess of the tree bole, which remarkable decreasing the total content of soil nitrogen and biomass of microbes (Johnson and Curtis 2001). This resulted a niche selecting some sensitive taxa and alter structure of soil community [20], which can be considered as an environmental screening [21]. The loss of susceptible microbes due to tree harvesting may support the colonization of better-adapted microbes, it shift the microbial community hence modify the process of decomposition [22, 23].
Specifically, the reset of soil community is due to forest harvesting contributes large amount of soil organic compound into soil. Referring to [24] huge of available organic C should facilitate copiotrophs microorganisms. Tate [25] divided soil microbes into two groups. Copiotrophs microbes group is opportunist, when resource conditions are plentiful, they prefer to ingest unstable soil organic C pools, then aggressively grow. In contrast, oligotrophic group have slower growth speed and are incapable to compete with the copiotrophs in poor nutrient circumstances [25].
Study on short rotation coppice monoculture plantation of Callyandra callothyrsus in Majalengka District, Indonesia by Widyati et al. [10] found cutting decreased the below ground sugars flux by 80% and lead to decrease the soil pH rapidly. The depletion of total soil sugar is hypothesized as the strategy for C. calothyrsus to survive and regenerate after being cut. Sugar deficiency causes major alteration in the size and composition of rhizosphere community. Another survival strategy for limiting adjacent competitor populations in the rhizosphere of callyandra is to increase soil acidity [10].
The Role of Soil Microbes to the Succession of Pioneer in the Secondary Forest, Involved to the Invasive Alien Species Distribution
Once forested land opened due to harvesting, fire, or other catastrophes, this is the opportunity for a new plant to occupy this new habitat. Vegetation formation in the earth is started with seed dispersal and establishment of seedlings in soil. A seed reach the new habitat by seed dispersal vectors, such as animals, wind, water, or human being. Nelson [26] reported the impact of environment and microbial interactions in plant development take place initially in germination and early growth stages. The microbiome developing throughout seed sprouting and spreads to seedlings and diverse organs of full-grown plants after a long time may contain microbes that were picked up along the way [26]. Afterwards, an extensive range of biotic (plant traits) and abiotic (soil properties) variables determine the diversity of structure and function of the microbial communities in the new rhizosphere assemblage [8].
From the seed stage onward, interactions between plants and microorganisms have been documented, the interaction is known as seed microbiome [26]. Furthermore, [26] classified seed microbiome into endophytic and epiphytic microbiota. Endophytic microbiota are microbes living inside seed tissues and inherited to its descendant through progeny process during seedlings development, while epiphytic microbiota are microbes inhabiting outside seed and may or may not be adopted to inner tissues of seeds and transmitted either vertically to their seedlings or horizontally to other plants [26]. Previous studies reported that seed-associated bacterial distinct due to species of plant [27], plant traits [28], stages of seed development [29], topographical locations [30], and the existence of plant pathogen [31]. Links et al. [27] explained seed endophytic bacteria deliver almost the entire species assemblage from where the seed microbiome recruited, it indicated that in some plant species the seed endophytic were substantially conserved. The seed endophytic microbiota is frequently dissimilar with the soil bacteria colonized the plants rhizosphere [32]. It is indicated that, the microbes colonizing the seed is predominantly brought from the parent plant environment [26], it carried away from the habitat where the origin of the host plants grow [33]. It is not clearly explained, either local site characteristics or host genotypes assembly the bacterial seed microbiome [33, 30].
Plants have an impact on soil microbial populations; every plant type is presumed to form a distinguish rhizosphere communities. Root exudates are the main force to carry out the selection process [8]. The type of vegetation determines the conformation of substances released by roots, which determines the relative abundance of microorganisms surrounding the roots [34]. To shape their own rhizosphere, plants allocate nutrients for the desired microbes, in the contrary it deliver unique antimicrobial metabolites to get rid the unwanted microorganisms.
The interested phenomenon on succession is invasive plant occupation, which has remarkable effects on the society of soil microorganisms [35]. The invasive plant species generally characterized by their capacity to grow rapidly, hence they will immediately replace the origin vegetation composition [36]. In the new ecosystem, these exotic plants will change the net primary productivity (NPP) and nutrient cycling processes [37]. Because there is an intently link among the plant aboveground and belowground subsystems, hence the alteration in species plant dominant in a community will simultaneously affect interactions among plants and microorganisms in the rhizosphere. Afterward, it determine the nutrient cycle processes [38]. It is due to, substances released by plant root facilitate the rhizosphere association, which in reciprocate they decompose organic matter to provide nutrients to the plants [39]. Zhang et al. [40] found more peculiar fixed carbon released in the rhizosphere of Spartina alterniflora Loisel., an invasive species, than it found in the native plants. In consequence, the carbon turnover effectiveness at the plant-soil boundary increase with the intention of achieving successful invasion.
Significant modification on the assembly of soil microbial associations, biomass, and their activities due to plant infiltrations determine the fundamental ecosystem behaviors such as decomposition of soil organic matter and nutrient cycling [41]. Stefanowicz et al. [42] convinced that the invader plants change belowground microbial performance significantly only in two growing seasons. The modification of soil environment is the effort of the invasive species to construct their proper niche to support the growth and successfully conquer the new habitat [42].
Stefanowicz et al. [42] summarized, the various alterations due to invasion of alien species can be classified into: impact on soil physic-chemicals (nutrients and pH), impact on soil communities (soil bacteri and fungy, ectomycorrhiza and endo-mycorrhizal fungi), impact on microbial activities (enzymatic and respiration). Characteristic of the plant invaders such as crown formation, rooting architecture, or chemical content of tissue define the distinction in the reactions of soil to plant invasion [42]. Root exudate is a selection tool in a rhizosphere because a root exudate with a certain composition is only suitable for the structure of a particular microbial community, otherwise, that composition can be a killing machine for other microbial groups [43]. Thus, it can be understood that the introduction of new plant species into a habitat, massively, will lead to dramatic reformation of the community of underground microbes as consequences of the powerfull reciprocal influence.
It is widely recognized that invasive plants brought negative impacts to the indigenous plant communities, even the invader often completely eliminate native species and change the habitat to a monodominant communities [44]. The shift in plant composition by exotic plant species interfere the linkage between above-ground communities [45], it modify soil chemical properties (pH, N content, N mineralization processes) due to revolution in the structure of microbial communities that control the main biogeochemical cycles in the habitat.
Every exotic species has a unique consequence to the physico-chemical characteristics of the soil in its new habitat [46]. Study on invasive species showed, they caused alteration on soil physical attributes, especially the soil porosity, temperature, water-holding capacity, and moisture [47]. This is due to the changes in the vegetation type in the habitat which has different in tissue biomass characteristics, rooting depth, leaf area index, and transpiration rate [48]. Modification in soil moisture and root exudate composition result in changes in the rhizospheric microbial flora to promotethe further invasiveness [49]. More over, the invasion also influence the chemical characteristics of soil due to the shifting of soil organic matter input, paterns of cycling of carbon and nitrogen, and soil pH. Invasive species also found to release of some allelopathic substances [50, 51, 52].
The belowground microbial community strongly determines the invasive capacity of exotic plant species [53]. Li et al. [53] reported one of most destructive invasive weeds in China, Ageratina adenophora, which formed a single species community rapidly. The existence of A. adenophora resulted in shifting of microbial composition either in the bulk soil or rhizosphere, for example Bradyrhizobium replace Aeromicrobium [53], the specific microbes rule in N-cycling processes. Li et al. [53] confirmed that A. adenophora change the soil pH of the rhizosphere environment to impose homogenous microbial communities. They selected appropriate microbial communities in providing their obligations in soils to encourage their invasiveness.
In Indonesia there are several invasive species incidences that caused alteration on the habitat dramatically. In Batukahu National Park, there were 10 identified invasive plant species member of 10 genera and five families [54]; which were classified as 40% herbs, and 30% each shrubs and grasses [54].
The most phenomenal invasive species in Indonesia is Vachellia nilotica (L.) P.J.H. Hurter & Mabb commonly known as thorny acacia, is notorious for its ability to conquer diverse environment, especially grassland (Fig. 4.1.) After being introduced for the first time in the 1969, to the Baluran National Park (BNP), Indonesia, the tree currently has invaded wild bull habitat of the national park more than half area [55]. BNP is the biggest Bull (Bos sondaicus) in Indonesia with 1500–2000 ha of savannah ecosystem [56]. The invasion of V. nilotica threatened the population of the bulls due to the invasive species eradicated the bulls feeding plants.
Another terrific invasive plant in Indonesia is Merremia peltata causing serious hazards to the regeneration of indigenous plant [57]. Both the opened areas and the bared land, before planting for estate and agriculture, in entire regions of Indonesia are susceptible to be invaded by this species [57]. This species has a large underground tuber. They climb and cover all over crowns of the woody plants, hence it disturb the photosynthesis process [57]. Merremia is classified into a fast-growing plant that is regenerate by rooting their nodes, or by resprouting and rooting the broken stem fragments [58]. The species dispersal also occurred by seeds that is unconsciously carried away by human activities or as a result of soil displacements [59]. Yudaputra [57] estimated that currently, M. peltata have influenced or perhaps destroyed the habitat of 30.4% of total terrestrial ecosystem of Indonesia.
Unfortunately, the study on the influents of invasive plant species in Indonesia on the microbial population and biogeochemical process in soil is lacking. Due to each plant species need a specific collective microbes forming their own microbiome, the gaps of the information is inspiration to conduct further studies.
Alteration of Soil Microbes Population Due to Land Use Shifting from Natural Forest to Monoculture Plantation
In natural forests, the presence of various types of plants growing together in a site will complement each other so that nutrient absorption becomes more efficient [60]. Multispecies swards have shown a variety of diversity benefits on aboveground performance, including yield, nitrogen contents, and even soil-legacy effects on a subsequent crop [61]. Diverse plant functional attributes in multispecies vegetations resulted in complementarity of resource acquisition [62, 63], such as growing season [64], rooting depth [65], and N2 fixation capability [65]. Importantly, [61] explained that these plant species diversity beneficial impacts resulted from interactions across the plant species and are thus more than merely the comparative contribution of each species (their identity effect).
The large-scale development of monoculture forest plantation will eventually replace the ecosystem's community. Plantation species are typically chosen for their highly adaptable traits, which are comparable characteristics to those of invasive plants. Monoculture cultivation's success in an ecosystem has replaced native plant dominance with exotic species. Due to changes in the content of plant root rhizodeposition into the rhizosphere, these alien species modify the network between aboveground and belowground communities in new settings [45]. Because the root exudate generated by new plantations has a different composition than the original soil environment, it alters the structure and function of the soil community of rhizosphere. Consequently, massive planting of new species as monoculture commonly drastically changes the important characteristic of soil such as pH, component of nitrogen and carbon, rate of mineralization and nitrification, and portion of essential elements such as potassium (K), calcium (Ca), and magnesium (Mg) (Table 4.1) [66].
The biotic and abiotic properties in soil can be modified by plant, and this will give impact to other plants that subsequently grow in this ecosystem. In multi-species plantation the effect of a plant type to the belowground ecosystem will be very complex [69]. It is depend on what it function and abundance in the ecosystem, it is also determined by species composition exist in the ecosystem and the characteristics of the soil [69]. Previous study carried out by Fox et al. [61] found that soil microbial community structures were highly driven by plant species identity. The difference physiology of plant species such as structures, differing root biomass, and symbiotic N2‐fixation induced soil physicochemical change.
Furthermore, poor species diversity on monoculture changes the rhizosphere microbial community [70]. Since, soil microbe abundance, composition, and diversity are strongly affected by plant species [71], changes in plant composition from multi- to monoculture modify the rhizosphere properties. Intensive monoculture activities over a long period lead to nutrient depletion because plants with the same growth rate in even-age forests require large amounts of the same nutrients [72]. As a result, they will release the same root exudate to invite microbes for helping grow and improving fitness. This continuous process will give negative impacts on soil function and yield sustainability due to different performance of their new rhizosphere composition.
Soil microbes have vital rule in a variety of ecological activities, including organic matter decomposition, nitrogen cycling, and plant productivity [73, 74]. The study of how different plant species and their configurations, such as forbs, grasses, and legumes, regulate their collaborated microbial association is receiving more consideration (e.g., [75, 76]). Within a particular soil type, distinct plant species found to assemblage-distinguished configuration of microbial colonization [77]. The diverse physiologies and features of different species, such as root architectures and activities, root productivity and array of rhizodeposition, are fundamental determinants of such variations [61] (Fig. 4.2).
After plants were dead or harvested, these changes in the soil microbiome mediated by plant left as “legacy” and determine the other plant species that grow subsequently (plant-soil feedbacks) [78]. The kind of soil-transferred legacy effects varies depending on different parameters, such as the prior plant, climate conditions, and soil type [79]. Rhizodeposits and litter attributes of plants determine soil microorganism [78]. Legacy effect is strongly defined by the amount and type of transmitted-persistance residue in the soil when the previous crop is removed [61]. The persistence best adapted decomposers to plant residues under the prevailing situations [80] may be assisted by the retaining of such plant excess in the soil environment, keeping this crucial macronutrient accessible in the habitat.
These kinds of legacy effects are likely to have wider ecological consequences. Plant legacy effects on the microbiome may effect on competition among plants, establishment and succession of plant, and the composition of the overlying plants [81]. Plants legacy can either negatively or positively effect succeeding plant species. Negatively effects occur when there are plant pathogens congregations in the soil and positively effect through the build-up of beneficial microbes [78]. So, that why one of negative effects of monoculture plantations is the occurrence of soil pathogens because its legacy may be the accumulation of pathogens.
The same species of plant has the same root system so the area of competition in the absorption of nutrients and water will be stronger [82]. The root competition of the same species of plants occurs three to five times greater than if they compete with different species [83]. To conquer the neighboring plants, they will release allellochemical, the compounds released frequently have impact either increase the growth of soil-borne pathogens or prevent the growth of advantageous microbes [84]. Similar plants will release similar allellochemicals, and there will be more buildup over time with recurrent plantation. The formation of numerous diseases known as replanting disease has been linked to one of the important chemicals in the allelopathic system: phenolic acid [85]. The phenolic acid level in soil was 400 percent higher in a continuous monoculture rye plantation than in a diverse cropping system, resulting in a decreased actinomycetes population [86]. Actinomycetes play a pivotal role in the rhizosphere, such as preventing plants from various soil-borne pathogens [87].
Role of Soil Microbes on Mining Land and the Limitations to Reclamation Achievement
Indonesia has the biggest deposit of mineral in the world, such as second position for gold and third for nikkel of the global supply (ESDM 2016). Indonesia also has 34.8 billion tons of coal deposit (the 8th position) (ESDM 2021). In one hand, mining sectors are the enormous source for the country income. On the other hand, minning results significant ecological effects such as soil erosion, holes formation, and biodiversity loss. Soil and water on ex-mining sites contaminated due to the chemicals used in the ore purification processes. Ex-mining sites are characterized by poor in macronutrients but rich in heavy metals, acidic soil reaction and inappropriate soil texture and moisture. Nikkel, tin, and coal mining are among the harmful to the forest area, due to those are operated in opened pit mining (OPM) that remove all of soil layers above the ore deposits, included the vegetation. The removal of vegetation brings immense consequence to the elimination of the origin soil microbiome, the essential actors in soil functioning and biogeochemical cycling.
The most tremendous consequence of OPM is the incident of acid mine drainage that is much more detrimental to the environment. The OPM systems reveal layers of rock containing sulfide compounds, expose to atmospheric oxygen hence it undergoes oxidation. This oxidation process will cause the previously inert rock to become reactive and release very strong sulfuric acid to the environment. Consequently, it will quickly acidify the surrounding waters and soil. The study conducted by Widyati [88] on ex-coal mining soil in South Sumatera, Indonesia, soil pH may decrease up to 2.8. This condition may dissolves metals, immobilizes various macro elements hence they are not available to plants, which can result in the death of various aquatic biota, as well.
Referring to Akcil and Koldas [89] mining of nickel, gold, and copper, is accompanied by acid drainage problems, that is in long-term destruct water bodies and life. When sulfide-containing rocks are exposed to oxygen and water, it resulted a phenomenon called acid-mine drainage (AMD) [89]; released sulfuric acid solution that will be polluted the surface water (rainwater, pond water) and shallow subsurface water. Once AMD is happened, extremely acidic water rich in heavy metals will be continually formed and transported follow the water movement [89]. The AMD phenomenon can be illustrated in the following reactions (Fig. 4.3) [89]:
-
(1)
2FeS 2 (S) + 7O 2 (g) + 2H 2 O (l) →2Fe 2+ (aq) + 4SO 4 2− (aq) + 4H + (aq)
The initial reaction is the sulfide mineral such as pyrite (FeS2) reacts with atmospheric oxygen and in the moist condition will dissolve ferrous (Fe2+) ion. The ferrous will be immediately oxidized into ferric (Fe3+) ion (reaction 2). AMD formation will be rapider in the region with high rainfall, like in Indonesia.
-
(2)
4Fe 2+ (aq) + O 2 (g) + 4H + (aq) →4Fe 3+ (aq) + 2H 2 O (l)
The rate of acid generation is strongly determined by the chemical, biological and physical attributes of the rocks and environments. Waste rock dump permeability is particularly the important physical factors. High permeability of dumping rock facilitates excessive oxygen access, which is contributes to rapid chemical reaction rates [89]. The acid environment favor the colonization of bacteria Acidithiobacillus ferrooxidans and the bacteria will be most favorable when the water pH is less than 3.2 [89]. The bacteria is classified as lithotrophs (“litho” means “rock”) groups that are getting energy rock weathering. It is also classified as chemotroph organisms that get energy from oxidation of inorganic compound i.e. FeS2 [90]. Bacteria A. ferrooxidans rapidly release lead and zinc from the rocks [90]. Removing soil layers rich in soil organic matter (top soil and sub soil) due to mining excavation give advantages to the bacteria group, and rapidly colonize in the habitat.
This reactions undergo either spontaneously or being catalyzed by A. ferrooxidans. The cathion Fe3+ will oxydize much more pyrite and release more ion responsible in acidifying the environment.
-
(3)
FeS 2 (S) + 14Fe 3+ (aq) + 8H 2 O (l) → 15Fe 2+ (aq) + 2SO 4 2- (aq) + 16H + (aq)
Other problem inherited by mining operation is talling, that can be highly diverse in their physic-chemical characteristics, generally is described as sandy or silty soil, and toxic peculiarities. Tailings from ore-metal minings are constantly not only sulphidic but also rich in residual metals and metalloids (mainly Arsenic) [90]. In many places of the world, surface stabilization by revegetation (i.e. phytostabilization) is essential to decrease the negative effects of legacy tailings. However, phytostabilization of sulphidic-based metal tailings through phytoremediation is limited by the tailings’ incapacity to facilitate the growth of vegetations [90]. Phytoremediation is a technology employing plant activities to absorb and eliminate elemental contaminants or decline their concentration in soil [91]. Avoidance and tolerance are two defense schemes employed by plants to deal with heavy metals poisonous in soils [92]. It is highly recommended to apply phytoremediation in ex-opened pit mining area with unsteady structure and high soil erosion, or on tailing of metal extraction [93]. The application of phytoremediation needs heavy metal detoxification as precondition process [94].
Beneficial microbes found in association with plants playing as phytoremediation activities. Earlier studies showed alteration in community structure of roots of pioneer grown in tailing containing Pb and Zn and improvement on microbial biomass [90]. Soil microbes can be engaged to assist in improving ex-mining land, directly or indirectly. Directly, microbial communities help in biogeochemical cycling of tailings. In the oxidized layer of neutralized base metal tailings can be colonized by microbial with significant biomass. However, the microbial diversity (mainly bacteria) is lower than it in the unpolluted soils [93]. The soil microbes population can be improved by inoculation. Introduction of sulphate-reducing bacteria inoculum to the ex-coal mining soils, have been improved the pH and soil nutrients [88], hence improve the seedlings planted as revegetation [95]. The bacteria reduced SO42− into S2 that is immobile [88].
Indirectly, favorable microbes in the rhizosphere of revegetation plants facilitate the revegetation process in a variety of manners. For example, arbuscular mycorrhizas acting as a prohibiting barricade for heavy metal uptake by absorption, adsorption, or chelation process [96]. (2) Microorganisms promote immobilize the metal ions by adsorbing metals to their cell walls, creating chelators, and stimulating precipitation processes [97]. They can also help with phytostabilization by increasing root surface and depth, as well as acting as a separation barricade to protect shoots from ion translocation from roots [98]. (3) Microbes directly stimulate root multiplication, promote plant development, increase plant tolerance to heavy metal, and improve plant health.
The group of plant growth-promoting rhizobacteria (PGPR) can be employed in ex-mining revegetation because their ability to enhance plant growth and fitness, improve plant nutrition, and the most important is their protection to plants from heavy metal uptake and translocation [97]. This is performed through producing organic acids, enzymes, siderophores, antibiotics, and phytohormones, among other chemicals [97].
Future Strategies
As one of most populated country in the world (more than 270 millions), Indonesia, will encounter food, energy, and water security in the future. The situation may be aggravated by environment destruction and climate change. It is need tight collaboration among all stakes in formulating smart strategies to deal with the challenges, included strengthen knowledge on importance of soil microbial to improve land productivity, to clean pollution, as well as to enhance land revegetation.
-
a.
Optimize land utilization in food, water and energy nexus to preserve deforestation.
Cultivation of mixed crops that produce food, bioenergy and species that quickly increase water catchment needs to be developed to prevent expansion of deforestation and optimize land productivity. In addition, the use of local varieties needs to be expanded for restoring biodiversity, also reducing destruction of the microbiome due to “strange rhizophere assemblage” by invasive exotic species.
-
b.
Rhizosphere engineered for environment friendly agriculture.
Plants rhizosphere can be engineered to produce substances for increasing nutrient availability, for defensing from biotic and abiotic pressures, or for promoting the growth of beneficial bacteria. Rhizosphere engineering can involve inoculation of beneficial microbial populations to the selected plants. Soil amendment can be applied to enhance the fitness of root associated bacterial communities. Hence, the rhizosphere favor selected bacteria collaborative synergically in consortia appropriate for barricading roots from pathogens. Rhizosphere engineering with various activities of PGPR improve the soil aggregation, soil health and fertility, hence facilitate plant growth better and increase the productivity.
-
c.
Ex-mining rehabilitation and revegetation employ beneficial microbes
The crucial step in ex-mining reclamation process is soil amendment to provide favorable environment for revegetation planting. To improve revegetation succeed, both organic and inorganic ameliorants can be added to the contaminated soil. Inorganic amendment is aimed to modify metal toxicity, reduce heavy metal bioavailability through adjustment soil reaction [99]. While, organic amendment is intended to increase the organic matter content. Those soil organic improvements add essential nutrients of the soil, improve physic, chemical and biological soil attributes, improve water-holding capacity which can benefit plant colonization in ex-mining sites. Earlier study on augmentation the ex-coal mining with material consists of raw organic matter, such as paper mills sludge, in a huge dosage (50%) successfully depleted the population of bacteria Thiobacillus thiooxidans in the ex-mining soil [88], that is recognized as biocatalyzer of AMD.
Another key method for maximizing the success of ex-mine land revegetation is species selection. The selected species should be tolerant to heavy metal environments, have a dense roots system and have capability to preserve soil structure, and prevent soil erosion, [92]. Qualification of selected plants for ex-mining revegetation such as fast growing for building large canopy in in a short period of time. It will assist land to modify microclimates, rapidly. They also produce lots of biomass that can be supplied to soil as organic matter. On the other hand, the selected plant should be effortless to cultivate in the field [91, 100]. The most familiar pioneer is acacias which have the ability to rehabilitate soils by absorbing and storing heavy metals like zinc (Zn), lead (Pb), copper (Cu), cadmium (Cd), and chromium (Cr) in their leaves, shoots, and roots [93]. Including microbes in ex-mining revegetation activities for example microbes enabling nitrogen fixation [101] that will improve not only soil remediation, soil amendment, but also assist plant to grow better in the severe environment.
References
Kusmana C, Hikmat A (2015) The Biodiversity of Flora in Indonesia. Jurnal Pengelolaan Sumberdaya Alam dan Lingkungan 5:187–198. e-ISSN: 2460-5824. https://doi.org/10.19081/jpsl.5.2.187
Malik AA, Prayudha JS, Annreany R, Sari MW, Walid A (2020) Keanekaragaman hayati flora di Taman Nasional bukit Barisan SelatanResor Merpas Bintuhan Kabupaten Kaur. Jurnal Ilmiah Pendidikan Sains. 1:35–42
Contreras-Hermosilia A (2000) The underlying causes of forest decline. CIFOR Occasional Paper 2000, No. 30. Center for International Forestry Research, Bogor, Indonesia
Sumargo W, Nanggara SG, Nainggolan FA, Apriani I (2011) Potret Keadaan Hutan Indonesia. Edisi Pertama. Forest Watch Indonesia. pp 52
DJPKTL [Direktorat Jenderal Planologi. Kehutanan dan Tata Lingkungan] (2017) Deforestasi Indonesia Periode 2014–2015. Direktorat Inventarisasi dan Pemantauan Sumber Daya Hutan, Direktrorat Jenderal Planologi Kehutanan dan Tata Lingkungan, Kementerian Lingkungan Hidup dan Kehutanan, Jakarta
Widyati E (2017) Understanding the business in the rhizosphere: how do plants and soil microbes make transactions. Dee Publish. Yogyakarta Indonesia. (in Indonesian)
Mendes R, Garbeva P, Raaijmakers JM (2013) The rhizosphere microbiome: significance of plant beneficial, plant pathogenic, and human pathogenic microorganisms. FEMS Microbiol Rev 37:634–663. https://doi.org/10.1111/1574-6976.12028
Berg G, Smalla K (2009) Plant species and soil type cooperatively shape the structure and function of microbial communities in the rhizosphere. FEMS Microbiol Ecol 68:1–13. https://doi.org/10.1111/j.1574-6941.2009.00654.x
Pitman NCA, Jorgensen PM (2002) Estimating the size of the threatened world flora. Science 298:989
Widyati E, Irianto RSB, Susilo A (2022) Rhizosphere upheaval after tree cutting: soil-sugar flux and microbial behavior. Commun Integr Biol 15:105–114. https://doi.org/10.1080/19420889.2022.2068110
Gunina A, Kuzyakov Y (2015) Sugars in soil and sweets for microorganisms: review of origin, content, composition. Soil Biol Biochem 90:87–100. https://doi.org/10.1016/j.soilbio.2015.07.021
Hütsch BW, Augustin J, Merbach W (2002) Plant rhizodeposition - An important source for carbon turnover in soils. J. Plant Nut. Soil Sci. 165:397–407. https://doi.org/10.1002/1522-2624(200208)165:4(397::%3cAID-JPLN397%3e3.0.CO;2-C
Dundek P, Holík L, Rohlík T, Vranová V, Rejšek K et al (2011) Methods of plant root exudates analysis: a review. Acta Universitatis Agriculturae et Silviculturae Mendelianae Brunensis 3:241–246. https://doi.org/10.11118/actaun201159030241
Hinsinger P, Bengough AG, Vetterlein D, Young IM (2009) Rhizosphere: biophysics, biogeochemistry and ecological relevance. Plant Soil 321:117–152
Raaijmakers JM, Paulitz TC, Steinberg C, Alabouvette C, Mo€enne-Loccoz Y (2009) The rhizosphere: a playground and battlefield for soilborne pathogens and beneficial microorganisms. Plant Soil 321:341–361
Bonkowski M, Villenave C, Griffiths B (2009) Rhizosphere fauna: the functional and structural diversity of intimate interactions of soil fauna with plant roots. Plant Soil 321:213–233
Bakker PAHM, Berendsen L, Doombos RF, Winterman PCA, Pieterse CMJ (2014) The rhizosphere revisited: root microbiomic. Front Plant Sci 4:1–8. https://doi.org/10.3389/fpls.2013.00165
Kӧgel-Knabner I (2002) The macromolecular organic composition of plant and microbial residues as inputs to soil organic matter. Soil Biol Biochem 34:139–162. PII:S0038-07170100158-4
Sasse J, Matinoia E, Northen T (2018) Feed your friends: to plant exudates shape the microbiome? Trends Plant Sci 23:25–41
Dvorsky M, Dolezal J, De Bello F, Klimesova J, Klimez L (2011) Vegetation types of East ladakh: species and growth form composition along main environmental gradients. Appl Veg Sci 14:132–147
Zhang X, Liu S, Li X, Wang J, Ding Q et al (2016) Changes of soil prokaryotic communities after clear-cutting in a kars forest: evidences for cutting-based disturbance promoting deterministic processes. FEMS Microbiol Ecol 92:1–12. https://doi.org/10.1093/femsec/fiw026
Hernesmaa A, Bjӧrkӧlf K, Jørgensen KS, Haahtela K, Romantschuk M (2008) Potential impacts of clear-felling on microbial activities in boreal humus and mineral soil layers. Boreal Environ Res 13:525–538
Hernesmaa A, Bjӧrkӧlf K, Kiikkilӓ O, Fritze H, Haahtela K et al (2005) Structure and function of microbial communities in the rhizosphere of Scot pine after tree-felling. Soil Biol Biochem 37:777–785
Fierer N, Bradford MA, Jackson RB (2007) Toward an ecological classification of soil bacteria. Ecology 88:1354–1364
Tate R (2000) Soil microbiology. 2nd ed. John Wiley and Sons, New York, New York, USA
Nelson EB (2018) The seed microbiome: origins, interactions, and impacts. Plant Soil 422:7–34. https://doi.org/10.1007/s11104-017-3289-7
Links MG, Demeke T, Grafenhan T, Hill JE, Hemmingsen SM, Dumonceaux TJ (2014) Simultaneous profiling of seed-associated bacteria and fungi reveals antagonistic interactions between microorganisms within a shared epiphytic microbiome on Triticum and Brassica seeds. New Phytol 202:542–553
Barret M, Brian M, Bonneau S, Preveaux A, Valiere S, Bouchez O, Hunault G, Simoneau P, Jacques MA (2015) Emergence shapes the structure of the seed microbiota. Appl Environ Microbiol 81:1257–1266
Liu Y, Zuo S, Zou YY, Wang JH, Song W (2013) Investigation on diversity and population succession dynamics of endophytic bacteria from seeds of maize (Zea mays L., Nongda108) at different growth stages. Ann Microbiol 63:71–79
Klaedtke S, Jacques M, Raggi L, Préveaux A, Bonneau S, Negri V, Chable V, Barret M (2016) Terroir is a key driver of seed-associated microbial assemblages. Environ Microbiol 18:1792–1804. https://doi.org/10.1111/1462-2920.12977
Rezki S, Campion C, Iacomi-Vasilescu B, Preveaux A, Toualbia Y, et al (2016) Differences in stability of seed-associated microbial assemblages in response to invasion by phytopathogenic microorganisms. PeerJ 4. https://doi.org/10.7717/peerj.1923
van Overbeek LS, Franke AC, Nijhuis EHM, Groeneveld RMW, da Rocha UN, Lotz LAP (2011) Bacterial communities associated with Chenopodium album and Stellaria media seeds from arable soils. Microb Ecol 62:257–264
Johnston-Monje D, Lundberg DS, Lazarovits G, Reis VM, Raizada MN (2016) Bacterial populations in juvenile maize rhizospheres originate from both seed and soil. Plant Soil 405:337–355
Somers E, Vanderleyden J, Srinivisam M (2004) Rhizosphere bacterial signaling: a love parade beneath our feet. Crit Rev Microbiol 30:205–240
Van der Putten WH, Klironomos JN, Wardle DA (2007) Microbial ecology of biological invasions. ISME J 1:28–37
Liang MQ, Zhang CF, Peng CL, Lai ZL, Chen DF, Chen ZH (2011) Plant growth, community structure, and nutrient removal in monoculture and mixed constructed wetlands. Ecol Eng 37:309–316. https://doi.org/10.1016/j.ecoleng.2010.11.018
Guedes BS, Olsson BA, Sitoe AA, Egnell G (2018) Net primary production in plantations of Pinus taeda and Eucalyptus cloeziana compared with a mountain miombo woodland in Mozambique. Glob Ecol Conserv 15:e00414. https://doi.org/10.1016/j.gecco.2018.e00414
Carvalhais LC, Dennis PG, Badri D, Kidd BN, Vivanco JM (2015) Linking jasmonic acid signaling, root exudates, and rhizosphere. Microbiomes 28:1049–1058. https://doi.org/10.1094/MPMI-01-15-0016-R
Kulmatiski A, Beard KH, Stevens JR, Cobbold SM (2008) Plant-soil feedbacks: a metaanalytical review. Ecol Lett 11:980–992. https://doi.org/10.1016/j.soilbio.2017.06.003
Zhang P, Nie M, Li B, Wu J (2017) The transfer and allocation of newly fixed C by invasive Spartina alterniflora and native Phragmites australis to soil microbiota. Soil Biol Biochem 113:231–239. https://doi.org/10.1016/j.soilbio.2017.06.003
Zubek S, Majewska ML, Błaszkowski J et al (2016) Invasive plants affect arbuscular mycorrhizal fungi abundance and species richness as well as the performance of native plants grown in invaded soils. Biol Fertil Soils 52:879–893. https://doi.org/10.1007/s00374-016-1127-3
Stefanowicz AM, Stanek M, Majewska ML, Nobis M, Zubek S (2019) Invasive plant species identity affects soil microbial communities in a mesocosm experiment. Appl Soil Ecol 136:168–177. https://doi.org/10.1016/j.apsoil.2019.01.004
Coats VC, Rumpho ME (2014) The rhizosphere microbiota of plant invaders: An overview of recent advances in the microbiomics of new-excotic plants. Front Microbiol 5:368–377. https://doi.org/10.3389/fmicb.2014.00368
Sun F, Ou Q, Yu H, Li N, Peng C (2019) The invasive plant Mikania micrantha affects the soil foodweb and plant-soil nutrient contents in orchards. Soil Biol Biochem 139:107630. https://doi.org/10.1016/j.soilbio.2019.107630
Sanon A, Andrianjaka ZN, Prin Y, Bally R, Thioulouse J, Comte G, Duponnois R (2009) Rhizosphere microbiota interfers with plant- plant interactions. Plant Soil 321:259–278. https://doi.org/10.1007/s11104-009-0010-5
Gibbons S, Lekberg Y, Mummey DL, Sangwan N, Ramsey PW, Gilbert JA (2017) Invasive plants rapidly reshape soil properties in a grassland ecosystem. mSystems 2:e00178–16. https://doi.org/10.1128/mSystems.00178-16
Lone PA, Dar JA, Subashree K, Raha D, Pandey PK, Ray T, Khare PK, Khan ML (2019) Impact of plant invasion on physical, chemical and biological aspects of ecosystems: a review. Trop Plant Res 6:528–544. https://doi.org/10.22271/tpr.2019.v6.i3.067
Levine JM, Vila M, Antonio CMD, Dukes JS, Grigulis K, Lavorel S (2003) Mechanisms underlying the impacts of exotic plant invasions. Proc R Soc Lond, B, Biol Sci 270:775–781
Si C, Liu X, Wang C, Wang L, Dai Z, Qi S, Du D (2013) Different degrees of plant invasion significantly affect the richness of the soil fungal community. PLoS ONE 8:e85490. https://doi.org/10.1371/journal.pone.0085490
Ehrenfeld JG (2003) Effects of exotic plant invasions on soil nutrient cycling processes. Ecosystems 6:503–523. https://doi.org/10.1007/s10021-002-0151-3
Hawkes CV, Wren IF, Herman DJ, Firestone MK (2005) Plant invasion alters nitrogen cycling by modifying the soil nitrifying community. Ecol Lett 8:976–985
Thorpe AS, Thelen GC, Diaconu A, Callaway RM (2009) Root exudate is allelopathic in invaded community but not in native community: field evidence for the novel weapons hypothesis. J Ecol 97:641–645
Li Q, Wan F, Zhao L (2022) Distinct soil microbial communities under Ageratina adenophora invasions. (Abstract). Plant Biol 24. https://doi.org/10.1111/plb.13387
Mukaromah L, Imron MA (2019) Invasive plant species in the disturbed forest of batukahu nature reserve, Bali, Indonesia. Biotropia 27:22–32. https://doi.org/10.11598/btb.2020.27.1.933
Zahra S, Hofstetter RW, Waring K, Gehring C (2020) Review: the invasion of Acacia nilotica in Baluran National Park, Indonesia, and potential future control strategies. Biodiversitas 21:104–116
Radiansyah AD, Susmianto A, Siswanto W, Tjitrosoedirdjo S, Djohor DJ et al (2015) Strategi Nasional dan Arahan Rencana Aksi Pengelolaan Jenis Asing Invasif di Indonesia. Deputi Bidang Pengendalian Kerusakan Lingkungan dan Perubahan Iklim, Jakarta. pp 2–3. ISBN 978-602-72942-2-6
Yudaputra A (2022) Future spatial prediction of invasive plant Merremia peltata in Indonesia. 2nd ISeNREM 2021. IOP Conf Series: Earth and Environmental Science 50:012084. https://doi.org/10.1088/1755-1315/950/1/012084
Paynter Q, Harman H, Waipara N (2006) Prospects for biological control of Merremia peltata. Landcare Research Contract Report: LC0506/177. Landcare Research, Auckland, New Zealand
Kirkham WS (2005) Valuing invasions: understanding the Merremia peltata invasion in postcolonial Samoa. University of Texas at Austin, Austin, TX, USA
Liu CLC, Kuchma O, Krutovsky KV (2018) Mixed-species versus monoculture in plantation forestry: development, benefits, ecosystem services and perspecitves for the future. Glob Ecol Conserv 15:e00419. https://doi.org/10.1016/j.gecco.2018.e00419
Fox A, Lüscher A, Widmer F (2020) Plant species identity drives soil microbial community structures that persist under a following crop. Ecol Evol 10:8652–8668. https://doi.org/10.1002/ece3.6560
Hooper DU, Chapin FS, Ewel JJ, Hector A, Inchausti P, Lavorel S, Wardle DA (2005) Effects of biodiversity on ecosystem functioning: a consensus of current knowledge. Ecol Monogr 75:3–35. https://doi.org/10.1890/04-0922
Loreau M, Naeem S, Inchausti P, Bengtsson J, Grime J, Hector A, … Wardle D (2001) Biodiversity and ecosystem functioning: current knowledge and future challenges. Science 294:804–808. https://doi.org/10.1126/science.1064088
Husse S, Lüscher A, Buchmann N, Hoekstra NJ, Huguenin-Elie O (2017) Effects of mixing forage species contrasting in vertical and temporal nutrient capture on nutrient yields and fertilizer recovery in productive grasslands. Plant Soil 420:505–521. https://doi.org/10.1007/s11104-017-3372-0
Hoekstra NJ, Suter M, Finn JA, Husse S, Lüscher A (2015) Do belowground vertical niche differences between deep- and shallow-rooted species enhance resource uptake and drought resistance in grassland mixtures? Plant Soil 394:21–34. https://doi.org/10.1007/s11104-014-2352-x
Simba YR, Kamweya AM, Mwangi PN, Ochora JM (2013) Impact of the invasive shrub, Lantana camara L. on soil properties in Nairobi National Park, Kenya. Int J Biodivers Conserv 5:803–809. https://doi.org/10.5897/IJBC2013.0623
The Center of Statistic Council of Indonesia (BPS) (2022) Luas tanaman perkebunan Indonesia. https://www.bps.go.id/indicator/54/131/1/luas-tanaman-perkebunan-menurut-provinsi.html
The Ministry of Forestry of Indonesia (KLHK) (2022) Statistik Kehutanan Indonesia 2020. https://www.menlhk.go.id/site/single_post/4697/statistik-2020
Kostenko O, Bezemer TM (2020) Abiotic and biotic soil legacy effects of plant diversity on plant performance. Front Ecol Evol 8:87. https://doi.org/10.3389/fevo.2020.00087
Gajda A, Martyniuk S (2005) Microbial biomass C and N and activity of enzymes in soil under winter wheat grown in different crop management systems. Pol J Environ Stud 14:159–163
Ushio M, Kitayama K, Balser TC (2010) Tree species effects on soil enzyme activities through effects on soil physicochemical and microbial properties in a tropical montane forest on Mt. Kinabalu, Borneo. Pedobiologia 53:227–233
Acosta-Martinez V, Zobeck TM, Allen V (2004) Soil microbial, chemical and physical properties in continuous cotton and integrated crop-livestock systems. Soil Sci Soc Am J 68:1875–1884
Schnitzer SA, Klironomos JN, HilleRisLambers J, Kinkel LL, Reich PB, Xiao K, … Scheffer M (2011) Soil microbes drive the classic plant diversity–productivity pattern. Ecology 92:296–303. https://doi.org/10.1890/10-0773.1
Wagg C, Bender SF, Widmer F, van der Heijden MGA (2014) Soil biodiversity and soil community composition determine ecosystem multifunctionality. Proc Nat Acad Sci 111:5266–5270. https://doi.org/10.1073/pnas.1320054111
Ladygina N, Hedlund K (2010) Plant species influence microbial diversity and carbon allocation in the rhizosphere. Soil Biol Biochem 42:162–168. https://doi.org/10.1016/j.soilbio.2009.10.009
Zhou Y, Zhu H, Fu S, Yao Q (2017) Variation in soil microbial community structure associated with different legume species is greater than that associated with different grass species. Front Microbiol 8:1007 https://doi.org/10.3389/fmicb.2017.01007
Leff JW, Bardgett RD, Wilkinson A, Jackson BG, Pritchard WJ, De Long JR, Fierer N (2018) Predicting the structure of soil communities from plant community taxonomy, phylogeny, and traits. ISME J 12:1794–1805. https://doi.org/10.1038/s41396-018-0089-x
Hannula SE, Heinen R, Huberty M, Steinauer K, De Long JR, Jongen R, Bezemer TM (2021) Persistence of plant-mediated microbial soil legacy effects in soil and inside roots. Nat Commun 12:5686. https://doi.org/10.1038/s41467-021-25971-z
Anderson RL (2011) Synergism: a rotation effect of improved growth efficiency. In: Sparks DL (ed) Advances in agronomy, vol 112. Elsevier Academic Press Inc., San Diego, CA, pp 205–226
Allison SD, Lu Y, Weihe C, Goulden ML, Martiny AC, Treseder KK, Martiny JBH (2013) Microbial abundance and composition influence litter decomposition response to environmental change. Ecology 94:714–725. https://doi.org/10.1890/12-1243.1
Kardol P, De Deyn GB, Laliberté E, Mariotte P, Hawkes CV (2013) Biotic plant–soil feedbacks across temporal scales. J Ecol 101:309–315. https://doi.org/10.1111/1365-2745.12046
Foxx AJ, Fort F (2019) Root and shoot competition lead to contrasting competitiveoutcomes under water stress: a systematic review and meta-analysis. PLoS ONE 14:e0220674
Rubio G, Walk T, Ge Z, Yan X, Liao H, Lynch J (2001) Root gravitropism and below-ground competition among neighbouring plants: a modelling approach. Ann Bot 88:924–940. https://doi.org/10.1006/anbo.2001.1530
Pollock JA, Kogan LA, Thorpe AS, Holben WE (2011) Catechin, a root exudate of the invasive Centaurea stoebe Lam. (Spotted Knapweed) exhibits bacteriostatic activity against multiple soil bacterial populations. J Chem Ecol 37:1044–1053. https://doi.org/10.1007/s10886-011-0005-6
Wu L, Wang J, Huang W, Wu H, Chen J, Yang Y, Zhang Z, Lin W (2015) Plant-microbe rhizosphere interactions mediated by Rehmannia glutinosa root exudates under consecutive monoculture. Sci Rep 5:15871. https://doi.org/10.1038/srep15871
Liu XB, Herbert SJ, Hashemi AM, Zhang X, Ding G (2006) Effects of agricultural management on soil organic matter and carbon transformation—A review. Plant Soil Environ 52:531–543. https://doi.org/10.17221/3544-PSE
Bhatti AA, Haq S, Bhat RA (2017) Actinomycetes benefaction role in soil and plant health. Microb Pathog 111:458–467. https://doi.org/10.1016/j.micpath.2017.09.036
Widyati E (2006) Bioremediation of ex-coal mining soil use sludge of pulp and paper to enhance its land revegetation. PhD thesis. IPB University
Akcil A, Koldas S (2006) Acid mine drainage (AMD): causes, treatment and case studies. J Clean Prod 14:1139–1145. https://doi.org/10.1016/j.jclepro.2004.09.006
Li X, Bond P, Van Nostrand J, Zhou J, Huang L (2015) From lithotroph- to organotroph-dominant: directional shift of microbial community in sulphidic tailings during phytostabilization. Sci Rep 5:12978. https://doi.org/10.1038/srep12978
Berti WR, Cunningham SD (2000) “Phytostabilization of metals”. In: Raskin I, Ensley BD (eds) Phytoremediation of toxic metals: using plants to clean-up the environment. John Wiley & Sons, Inc., New York, NY, pp 71–88
Yan A, Ming Y, Tan SN, Yusof MLM, Ghosh S, Chen Z (2020) Phytoremediation: a promising approach for revegetation of heavy metal-polluted land. Front Plant Sci. https://doi.org/10.3389/fpls.2020.00359
Yahya A, Vijayanathan J, Ishak MF, Kadir WRWA (2018) Reversing soil degradation via phytoremediation techniques in an ex-tin mine and gold mine in Peninsular Malaysia. https://www.researchgate.net/publication/327716844_Reversing_soil_degradation_via_phytoremediation_techniques_in_an_ex-tin_mine_and_gold_mine_in_Peninsular_Malaysia. Accessed 19 May 2022
Thakur S, Singh L, Wahid ZA, Siddiqui MF, Atnaw SM, Din MFM (2016) Plant-driven removal of heavy metals from soil: uptake, translocation, tolerance mechanism, challenges, and future perspectives. Environ Monit Assess 188:206. https://doi.org/10.1007/s10661-016-5211-9
Sembiring YRV, Andriyanto M, Siagian N, Widyati E, Azwir (2016) Isolasi bakteri pereduksi sulfat untuk memperbaiki sifat kimia tanah bekas tambang batubara dan pengaruhnya terhadap karet (hevea brasiliensis) di polibeg. Jurnal Penelitian Karet 34(2):165–174
Hall J (2002) Cellular mechanisms for heavy metal detoxification and tolerance. J Exp Bot 53:1–11. https://doi.org/10.1093/jexbot/53.366.1
Ma Y, Prasad M, Rajkumar M, Freitas H (2011) Plant growth promoting rhizobacteria and endophytes accelerate phytoremediation of metalliferous soils. Biotechnol Adv 29:248–258. https://doi.org/10.1016/j.biotechadv.2010.12.001
Göhre V, Paszkowski U (2006) Contribution of the arbuscular mycorrhizal symbiosis to heavy metal phytoremediation. Planta 223:1115–1122. https://doi.org/10.1007/s00425-006-0225-0
Burges A, Alkorta I, Epelde L, Garbisu C (2018) From phytoremediation of soil contaminants to phytomanagement of ecosystem services in metal contaminated sites. Int J Phytoremediat 20:384–397. https://doi.org/10.1080/15226514.2017.1365340
Marques AP, Rangel AO, Castro PM (2009) Remediation of heavy metal contaminated soils: phytoremediation as a potentially promising clean-up technology. Crit Rev Environ Sci Technol 39:622–654. https://doi.org/10.1080/10643380701798272
Suter M, Connolly J, Finn JA, Loges R, Kirwan L, Sebastià MT, Lüscher A (2015) Nitrogen yield advantage from grass–legume mixtures is robust over a wide range of legume proportions and environmental conditions. Global Change Biol 21:2424–2438. https://doi.org/10.1111/gcb.12880
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2023 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Widyati, E. et al. (2023). Microbial Community Dynamics Due to Land Use Change: Some Circumstances in the Tropical Rain Forest of Indonesia. In: Parray, J.A. (eds) Climate Change and Microbiome Dynamics. Climate Change Management. Springer, Cham. https://doi.org/10.1007/978-3-031-21079-2_4
Download citation
DOI: https://doi.org/10.1007/978-3-031-21079-2_4
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-21078-5
Online ISBN: 978-3-031-21079-2
eBook Packages: Earth and Environmental ScienceEarth and Environmental Science (R0)