Abstract
Rainbow trout (Oncorhynchus mykiss) was introduced in most lakes of Patagonia, generating concern about its impact on the receiving communities. However, the information about the effects of trout in stocked lakes of this region is still fragmentary. We conducted a comprehensive comparative study of fishless and stocked lakes of the arid southern Patagonia to analyse the extent of trout effects on the community composition, primary productivity and food webs, considering also contrasting climatic conditions (wet and dry years). Rainbow trout shaped the community of pelagic crustaceans, affecting both species’ dominance and size structure. Stocked lakes also showed substantial differences in phytoplankton structure, and higher abundances of microplanktonic cyanobacteria, probably caused by top-down effects. These changes were more pronounced during dry periods, indicating the concomitant effects of trout introduction and climate fluctuations. Trout also generated alterations in food webs, forcing their main prey (amphipods) to shift from predominantly pelagic to benthic/littoral feeders. Differences in stable isotope values also indicate changes in the habitat coupling when trout were present. We discuss results from a conservation perspective, considering the potential consequences for waterbirds, mainly for the critically endangered hooded grebe (Podiceps gallardoi), which have their reproductive habitat in the analysed aquatic systems.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
Patagonia is a vast region that covers 790,000 km2 in Argentine continental territory, from the wet Andes to the dry Atlantic coast and from the temperate Colorado River to the north to the cold Tierra del Fuego to the south. This area can be divided into two subregions: Patagonian forest and Patagonian steppe (Morello et al. 2012) (see Chap. 1).
The Patagonian steppe encompasses approximately 20% of the Argentinean territory and is characterised by regular reliefs and by its arid to semiarid climate (Cabrera 1976), with a mean annual precipitation below 300 mm year−1 (Jobbágy et al. 1995; Paruelo et al. 1998). An extensive system of elevated basaltic plateaus holds most of the freshwater of this subregion, including lakes larger than 300 km2 and thousands of shallow lakes (see Chap. 10). These plateaus were formed by tectonic activity during the Cenozoic (Panza and Franchi 2002) and include natural depressions that collect both rainfall and snowfall (Pereyra et al. 2002). These aquatic systems, which are naturally fishless, are characterised by a high diversity of morphometric and limnological characteristics (Lancelotti et al. 2009a), playing a crucial ecological role, since they constitute one of the few sources of water available to numerous species of animals, particularly for a diverse aquatic bird community. For this reason, Patagonian basaltic plateaus were declared relevant areas for bird conservation in Argentina (Di Giacomo 2005; Scott and Carbonell 1986). Particularly, this system constitutes the breeding habitat for Podiceps gallardoi Rumboll 1974, an endemic and critically endangered diving bird (Fjeldså 1986; Roesler et al. 2012; BirdLife International 2021). Moreover, other endemic organisms (e.g. vertebrates, invertebrates and algae) were recorded in these aquatic environments (Canevari et al. 1999; Menu-Marque et al. 2000; Ortubay et al. 2006; Coesel et al. 2017; Manzo et al. 2019) (see Chaps. 10, 11 and 15).
Exotic flora and fauna introductions have been common practices in Patagonia for more than a century. In particular, the introduction of salmonids and also the translocation of native fish species have been reported for many water bodies in this region (Perotti et al. 2005) (see Chap. 13). The introduction of Oncorhynchus mykiss (Walbaum 1792) in naturally fishless lakes of the Patagonian plateaus began around 1940, but over the last 20 years, the number of stocked lakes increased substantially, mainly for developing aquaculture and sport fisheries (Lancelotti et al. 2009a). These introductions have generated concerns about their potential consequences on the receiving communities in general and on the hooded grebe in particular (Lancelotti et al. 2009b, 2010b; Roesler et al. 2012).
The impact of climate variability on water systems is especially critical for the Patagonian region. Regional climate models project around 10–30% less precipitation over northern Patagonia by the end of the century, being the Patagonian forest subregion the most threatened one by this decline (Barros et al. 2014; Pessacg et al. 2020). Both the decrease of snowfall and the rise of temperature produce glacier recession and regional hydrological changes (Masiokas et al. 2008; Pasquini et al. 2008; Barros et al. 2014) (see Chap. 4). Moreover, the surface area of some lakes of the extra-forest Patagonian plains underwent a dramatic reduction since 2000 (Tejedo 2003; Llanos et al. 2016; Scordo et al. 2018a), thus increasing the desertification processes (Scordo et al. 2018b). Particularly, the lakes of the highland plateaus in the Patagonian steppe have shown a decrease in the water level of many shallow lakes and even the complete evaporation of some others (Roesler et al. 2012; Lancelotti et al. 2020).
The aim of this chapter is to summarise the current knowledge and ecological impacts associated with non-native fish introductions in lakes of the arid southern Patagonia. We provide an overview of the available information on planktonic and macroinvertebrate communities and on food webs in lakes of the two main Patagonian plateaus, the Buenos Aires Plateau and the Strobel Plateau, also assessing the influence of O. mykiss introductions and climate variability on these water bodies.
2 Characterisation of the Lakes of the Patagonian Steppe
2.1 Topography
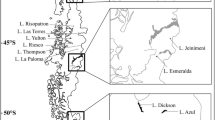
The studied plateaus are located in the central and northwestern area of Santa Cruz Province, in the arid Patagonian steppe of Argentina (Fig. 14.1).
Basaltic plateau system from Patagonia (grey areas, adapted from D’Orazio et al. (2000)) and geographic location of the studied lakes (white circles) in the Buenos Aires Plateau and in the Strobel Plateau, in Santa Cruz Province, Argentina
Topographically, the Strobel Plateau is relatively uniform with a mild upwards slope towards the northwest, varying in altitude from 1200 m a.s.l. westwards to 700 m a.s.l. eastwards (Lancelotti 2009). It has an extension of 3000 km2 holding >1500 natural depressions fed by rainfall and snowmelt (Lancelotti et al. 2009a). It represents an extensive lacustrine system, including small temporary ponds as well as a few large lakes (∼100 km2). Most of these lakes are closed depressions, but a few of them are connected by temporary watercourses (Lancelotti 2009). On the other hand, the Buenos Aires Plateau also reaches its maximum altitude westwards (∼1800 m a.s.l. with Mount Zeballos peaking at 2700 m a.s.l.) to decrease eastwards (700 m a.s.l.) (Roesler 2016). This plateau covers a total area of 3650 km2 holding more than 150 endorheic shallow lakes.
2.2 Climate
The region is characterised by its extremely cold, windy and dry climate (Cabrera 1976). It presents a great variation in the photoperiod over the year, with temperatures varying between −22.4 and 38.5 °C (Lancelotti et al. 2020). The water regime is mostly determined by the balance between precipitation and evaporation, favoured by the high irradiance and strong winds that can reach up to 150 km h−1 of speed (Correa 1998; Paruelo et al. 1998). Due to the frequent and strong westerly wind storms year round, even the deeper lakes are continuously mixed, preventing thermocline formation (Lancelotti 2009). All small lakes (<30 km2) remain frozen from early autumn throughout late spring. This very dynamic lacustrine system is controlled by climatic variables, mostly precipitation (Lancelotti 2009; Lancelotti et al. 2010a, b). The scarce annual precipitation is the only source of water for these lakes, and thus it has a direct effect on their dynamics and limnological characteristics (Lancelotti et al. 2010a).
2.3 Limnological Characterisation
The lakes of this region are naturally fishless and range from oligotrophic to eutrophic (Izaguirre et al. 2018; Porcel 2020). Myriophyllum quitense Kunth is the dominant macrophyte, occurring in most low salinity lakes of this region (Lancelotti 2009). From a limnological point of view, the lakes of the Strobel Plateau have been classified into four general categories (Lancelotti et al. 2009a): turbid lakes, large vegetated lakes, large unvegetated lakes and ponds. In large vegetated lakes, macrophytes cover from 30% to 80% of the lake surface area, whereas in large unvegetated lakes, macrophytes cover less than 5% of the lake surface area. Small ponds (<9 ha) are usually completely covered by plants (~90%) (see Chap. 10). The turbid environments can be separated into two categories: organic and inorganic turbid lakes. The first group includes phytoplankton turbid systems, which could have small patches of macrophytes, and the second one presents environments with higher values of conductivity (>2 mS cm−1) and is devoid of macrophytes (Izaguirre et al. 2018; Saad et al. 2019; Porcel 2020). This lake classification also applies to the water bodies of the Buenos Aires Plateau (Izaguirre et al. 2018; Porcel 2020). In addition, in this plateau, another category was described: clear mesohaline lake (“Honda”), which is a hard water lake because of its high concentration of calcium (bi)carbonate (Izaguirre et al. 2018; Porcel 2020), although these lake types are poorly represented on these aquatic systems. The main limnological features of the lake regimes of these plateaus are summarised in Table 14.1.
3 Fish Introduction
Biological invasions are recognised as a threat to biodiversity, generating alterations at multiple ecological levels (Mack et al. 2000; Simon and Townsend 2003). Fish introduction is a common and often successful tool for managing commercial or recreational fisheries (Wiley 2006; Gozlan 2008). However, this practice is among the main causes of impact in aquatic environments (Bahls 1992; McNaught et al. 1999; Sala et al. 2000; Parker and Schindler 2006; Schabetsberger et al. 2009). Particularly, the introduction of top predators into fish-lacking habitats has triggered trophic cascades and ecosystem alterations: changes in the abundance and composition of species, bioavailability and recirculation of nutrients, changes in primary production and shifts in the behaviour of organisms in the receiving lake (Carpenter and Kitchell 1993; Mittelbach et al. 1995; Scheffer 1998; Schindler et al. 2001; Schindler and Parker 2002). For example, the introduction of strictly planktivorous fish can lead to a reduction in the grazing pressure of zooplankton on phytoplankton, triggering an increase in algal biomass (Carpenter et al. 1985). Although there are fewer examples of the effect of generalist fish, which can feed on both pelagic and benthic prey, it is known that they can create a strong coupling between habitats (e.g. Schindler and Scheuerell (2002), and Vander Zanden and Vadeboncoeur (2002)). Likewise, the effects of generalist fish appear to depend on the introduced species and the trophic structure of the environment.
Salmonids were widely introduced around the world, and the available information suggests that their impacts may be severe and rapid (Crowl et al. 1992; Townsend 2003). The size-selective feeding behaviour of salmonids can change size structure and species composition of zooplankton and macroinvertebrate communities (Anderson 1980; Newman and Waters 1984; Gliwicz and Pijanowska 1989; Knapp et al. 2001). Frequently, predation on the native fauna by introducing salmonids causes ecological changes in higher levels, causing the elimination of amphibian and reptile populations (Tyler et al. 1998; Knapp 2005).
In rivers and lakes of Patagonia, the introductions of salmonids began at the onset of the twentieth century (Macchi et al. 1999). By the 1980s, all five Patagonian provinces had their own hatcheries, continuing the spread of salmonids up to the present (Pascual et al. 2002), being nowadays a widespread activity (see details in Chaps. 11 and 13). Particularly, in lakes of the Patagonian steppe region, the stocking and translocation of exotic fish have generated local extinctions of cladocerans species, reduction in zooplankton size and a general homogenisation of the systems (Modenutti and Balseiro 1994; Ortubay et al. 2006; Reissig et al. 2006; Buria et al. 2007), also causing the impoverishment of bird and amphibian fauna (Perotti et al. 2005; Cuello et al. 2006; Ortubay et al. 2006) (see Chap. 15). Specifically, some water bodies of the Patagonian basaltic plateaus, which are naturally fishless (Lancelotti 2009; Lancelotti et al. 2017), were stocked with O. mykiss during the last decade. In the present chapter, we discuss all the findings related to fish introductions in shallow lakes of the Strobel Plateau so far published. These studies have evidenced changes at several organisation levels in the aquatic communities due to O. mykiss introductions.
4 Influence of Fish Introduction on Autotrophic and Heterotrophic Planktonic Communities
The introduction of omnivorous fish in shallow water environments is associated with both top-down and bottom-up controls that promote the structuring of aquatic food webs. In the first type of control, fish can reduce by predation the abundance of larger zooplankton thus enhancing phytoplankton biomass (Blanco et al. 2008). In the latter case, phytoplankton development is triggered by an increase of nutrients in the water column, which can occur by direct nutrient excretion by fish (Vanni 1996, 2002) and/or sediment resuspension generated by fish feeding on benthic organisms (Schauss et al. 2002). Both processes can contribute to the shift and/or permanently turbid state of lentic water bodies (Blanco 2001).
The influence of fish introduction on the microbial planktonic communities in naturally fishless lakes was investigated in water bodies of the arid Patagonia since the beginning of the twenty-first century. One of the first studies was carried out by Reissig et al. (2006), who surveyed 18 lakes located in 4 zones of the Patagonian steppe, including fishless lakes, water bodies with only native fish and lakes where exotic fish were introduced. Besides the contrasting zooplankton structure that was evidenced between fishless lakes and fish stocked lakes – aspect that is addressed further in this chapter – this study also revealed evident differences in the phytoplankton structure. Fish stocked lakes showed higher phytoplankton abundance and a predominance of the net fraction (>20 μm) over nanoplankton; conversely, fishless lakes presented lower phytoplankton abundances and a higher proportion of nanoplankton. Another important difference was the relative abundance of cyanobacteria, which was notoriously higher in fish stocked lakes; some of the registered taxa of this group were Aphanocapsa delicatissima, Dolichospermum flos-aquae, Microcystis aeruginosa and Oscillatoria tenuis. Considering that the increase of cyanobacteria is an undesirable change in a water body, this finding was the first alert of the potential effect of the fish introduction in Patagonian lakes, providing valuable evidence for the conservation of the aquatic systems of the region.
In general, lakes from Patagonian plateaus range from mesotrophic to eutrophic and are characterised by higher phytoplankton biomass in comparison with those of the forest region (Izaguirre et al. 1990; Izaguirre 1993; Díaz et al. 2000). For the lakes located in the basaltic Patagonian plateaus, the ecological investigations on the phytoplankton communities started in 2007, and the first results were published by Izaguirre and Saad (2014). This study included 12 lakes from the Strobel Plateau and revealed differences between fishless and fish stocked lakes. The highest total phytoplankton biovolumes were found in fish stocked lakes, with a higher contribution of cyanobacteria, accounting for 84% of the total biovolume in one lake.
Within the framework of the same research project, the microbial plankton communities of the lakes located at the main basaltic plateaus of Santa Cruz (Strobel and Buenos Aires) were surveyed over a 7-year period. Particularly, some water bodies of the Strobel Plateau were visited during several late spring and summer campaigns. One of the published studies involved 14 lakes, which were sampled in December 2011 and March 2013 and were classified into three types according to the lake regime and the presence or absence of fish: fishless clear unvegetated lakes, fishless clear vegetated lakes and phytoplankton turbid fish stocked lakes (Saad et al. 2019). The study was focused on the autotrophic planktonic communities of all size fractions (pico, nano and micro), and the picoplanktonic fraction was assessed by flow cytometry. One of the main findings of the study was that the lakes exhibited differences in the limnological variables mainly in relation to the lake regime and the presence or absence of fish. On the other hand, in general, the abundances of autotrophic picoplankton and phytoplankton >20 μm were higher in fish stocked lakes, in agreement with the pattern observed in the studies previously described. The higher phytoplankton biomass in fish stocked lakes was attributed to a probable top-down effect. On the other hand, all three types of lakes presented phycoerythrin-rich picocyanobacteria (PE-rich Pcy) and picoeukaryotes (Peuk), but phycocyanin-rich picocyanobacteria (PC-rich Pcy) were only present in fish stocked lakes. In general, PC-rich Pcy are more efficient in more turbid and eutrophic aquatic environments, whereas PE-rich Pcy prevail in clear oligotrophic lakes, as was discussed by many authors (e.g. Stomp et al. (2007), Schiaffino et al. (2013), and Izaguirre et al. (2014)); thus, the presence of PC-rich Pcy in fish stocked lakes would be consistent with this pattern since these lakes presented higher turbidity (Fig. 14.2).
It is important to point out that the lower transparency in fish stocked was not attributed solely to the presence of fish, because also the resuspension of sediments during lower water levels may be important.
In a later study conducted in lakes of the Strobel Plateau, Porcel (2020) found lower mean abundances of bacterioplankton in fishless lakes than in fish stocked lakes; and among the stocked lakes, those clear vegetated presented higher abundances than the organic turbid. As for photosynthetic picoplankton, slightly higher abundances were found in turbid organic lakes with fish introduction. Although the three types of autotrophic picoplankton were present (Pcy-rich Pcy, PE-rich Pcy and Peuk), opposing the results obtained by Saad et al. (2019), PC-rich Pcy were more abundant in fishless clear vegetated lakes. The difference with the pattern previously observed was attributed to the water level decrease that occurred in the lakes during the years of sampling (2016 and 2017), which was accompanied by an increase in their turbidity. In the Buenos Aires basaltic Patagonian plateau, where all lakes lack fish, studies on the microbial planktonic communities were also carried out in lakes with contrasting regimes (clear vegetated and turbid) using flow cytometry (Porcel et al. 2019). In that paper, the effect of the water level decrease was also explored, showing higher abundances of heterotrophic bacterioplankton in more eutrophic and turbid lakes and the lowest values of autotrophic picoplankton in clear oligotrophic vegetated lakes. In these water bodies, PE-rich Pcy cells were more frequent than PC-rich Pcy cells. The water level decrease led to a switch from a clear vegetated regime to a turbid regime in some lakes, and under these conditions, an increase in heterotrophic picoplankton, PC-rich Pcy and Peuk was observed.
5 Influence of Fish Introduction on Zooplankton and Macroinvertebrate Communities
The zooplankton communities of lakes and ponds of the Patagonian steppe have a remarkable biogeographical importance, due to the dominance of endemic species of calanoid copepods (Boeckella and Parabroteas), cladocerans (Daphnia, Macrothrix and Ilyocryptus) and rotifers (Brachionus, Keratella and Notholca) that are well adapted to the harsh environmental conditions of this arid zone (José de Paggi 1990; Menu-Marque et al. 2000; Adamowicz et al. 2004; Reissig et al. 2006; Marinone and Menu Marque 2010; Izaguirre et al. 2018). The regional zooplankton species richness in the studied plateau lakes of Santa Cruz Province encompasses 22 rotifers and 17 microcrustacean species, varying in individual lakes from 0 to 10 and 1 to 7, respectively, with mean local species richness of 5 rotifers and 4 microcrustaceans (Porcel et al. 2020). Despite the hostile environment, the occurrence of heterogeneous types of water bodies promotes a high planktonic diversity (Porcel et al. 2020).
The fishless lakes hold the largest representatives of the macrozooplankton species, including Boeckella longicauda, Boeckella brasiliensis, Boeckella poppei (≤3.3 mm) and Parabroteas sarsi (≤6.5 mm) among the copepods, and Daphnia dadayana, Daphnia menucoensis, Daphnia commutata and Daphnia “obtusa group” (2–3.5 mm) among the cladocerans (Reissig et al. 2006; Lancelotti et al. 2017). Similar species composition and size structure can be found in turbid organic lakes with suspended inorganic matter, which provide UV radiation protection and visual refuge to zooplankters from predatory fish (Marinone et al. 2006). Additionally, Boeckella meteoris is frequent in turbid inorganic lakes (Lancelotti 2009). The frequently co-occurring D. dadayana and D. commutata and different species of Boeckella share most of the fishless extra-forest water bodies; however, the large D. dadayana are better adapted to coexist with the big predatory copepod P. sarsi (Marinone and Menu Marque 2010).
The highest total abundance and biomass of zooplankton and macrozooplankton were recorded in the fishless environments, where large microcrustaceans are dominant (Reissig et al. 2006; Lancelotti et al. 2017; Izaguirre et al. 2018). Conversely, in clear lakes with high predation pressure by fish, zooplankton communities encompass distinctly smaller crustacean species such as Boeckella michaelseni, Boeckella gracilipes, Boeckella meteoris (≤1.7 mm), Ceriodaphnia spp. and Bosmina spp., several genera of the Chydoridae family (0.3–1.3 mm) and rotifers (0.1–0.2 mm), which become dominant, thus decreasing total zooplankton biomass values and narrowing the size spectrum towards a higher frequency of smaller sizes (Reissig et al. 2006; Lancelotti et al. 2017). Trout predation on the larger macrozooplankton, such as Daphnia, could favour rotifer development (Izaguirre et al. 2018) by releasing them from exploitative competition and mechanical interference (Porcel et al. 2020). However, the presence of fish influences rotifer diversity but not microcrustacean diversity (Porcel et al. 2020).
Concerning the diverse macroinvertebrates that live in the water bodies of the Patagonian steppe, the community structure differs in the relative abundance of Ostracoda, Amphipoda (Hyalella), Chironomidae larvae and the coleopteran Haliplus depending on the presence or absence of fish (Lopez et al. 2021). It is remarkable that pelagic Hyalella, the larger crustacean in these lakes and the primary prey of trout, are absent in stocked lakes as compared to their high abundance in fishless lakes (Lancelotti et al. 2017). Additionally, a recent research on food webs evidenced that insect larvae of the families Leptophlebiidae (Ephemeroptera) and Chironomidae (Diptera) appeared only in fishless lakes (Porcel 2020; Porcel et al. submitted). The extinction and decline of aquatic insects due to fish introduction have been extensively covered in literature, as is the case of Ephemeroptera populations (Luecke 1990; Caudill 2003; Epanchin et al. 2010). Consequently, the decrease in the abundance of insect larvae could suppress the grazing pressure on the periphytic community, thus enhancing the development of algal biomass and changing algal species composition (Townsend 2003) as observed in some water bodies of the Strobel Plateau (Izaguirre and Saad 2014; Saad et al. 2019). The changes in the zooplankton and macroinvertebrate communities observed in stocked lakes are summarised in Fig. 14.3.
6 Influence of Fish Introduction on Food Webs
The introduction of non-native species could cause structural and functional alterations of food webs (Townsend 2003; Vander Zanden et al. 1999b). Stocked fishes, such as salmonids, are often top predators that either replace native top predators or increase the species richness of top predators, which can alter trophic relationships through cascading effects affecting the basal resources (Carpenter and Kitchell 1996; Scheffer et al. 2001; Eby et al. 2006). Additionally, a reduction in native species or the presence of non-native fishes can produce novel species interactions that have not coevolved (Tagliaferro et al. 2014) and could lead to a system with unstable trophic characteristics (Vander Zanden et al. 1999a).
Shallow lakes are dynamic ecosystems with a spatial structure characterised by a deep pelagic (open water) zone and a shallow benthic/littoral zone (Scheffer 1998), coupled with biological, physical and chemical processes (Schindler and Scheuerell 2002). In some water bodies, pelagic zone processes are described by complex interactions, which may include both the littoral and benthic zones of the lake (Pace 1993; France 1995a; Jeppesen et al. 1997; Covich et al. 1999; Rothhaupt 2000; Schindler and Scheuerell 2002; Rautio and Vincent 2007). Consequently, the magnitude of food web alterations, in this case of fish introductions, may depend on multiple factors related to the particular characteristics of each water body (Carpenter and Kitchell 1993; Pace et al. 1999; Polis 1999; Polis et al. 2000; Beisner et al. 2003). Fish are the main determinants of the food web structure and dynamics in shallow lakes (Jeppesen et al. 1997) and play an important role as habitat couplers because of their high mobility and inter-habitat omnivory (Schindler and Scheuerell 2002). In this context, fish would integrate both the benthic and the pelagic food webs, which could alter nutrient and energy pathways of the stocked lakes (Vander Zanden et al. 1999b; Schindler and Scheuerell 2002; Vander Zanden and Vadeboncoeur 2002).
Despite the abundant information about the negative effects of fish introductions on native freshwater ecosystems, very few studies give a clear image of how receiving communities might have looked before introductions, and scarce data are available on food web structure before these practices began, compromising the proposal of management tools for the conservation of the environment. Shallow lake systems of basaltic plateaus of Patagonia, with stocked and fishless lakes, offer the opportunity to evaluate the effects of the introduction of O. mykiss on the structure and functioning of the food web.
Food webs of a naturally fishless lake (“El Cervecero”) of the Buenos Aires Plateau, which was one of the most important habitats for the reproduction of P. gallardoi, were described through the stable isotope analysis (SIA) (Adami 2016). This aquatic community comprised eight taxonomic groups, dominated by crustaceans, insects, molluscs and leeches (Hirudinea). They occupied three trophic levels, including the most conspicuous groups of organisms, such as Lymnaea or Dytiscidae larvae which are potential grebe’s prey (Beltrán et al. 1992). The main sources of energy in this system seem to depend on benthic resources. Apparently, the structure of this community could be explained by its high macrophyte coverage and its low depth, favouring water transparency (allowing the proliferation of benthos) and providing refuge for zooplankton against predation. In this environment, P. sarsi (in the pelagic pathway) and Hirudinea (in the benthic pathway) were the only organisms occupying the third trophic level. The author suggested that P. sarsi was clearly separate from the rest of the organisms indicating that this group is sustained by pelagic sources of matter and energy, even in an environment dominated by macrophytes. A remarkable difference between the aquatic communities of both plateaus is that in “El Cervecero” (Buenos Aires Plateau), aquatic snails are dominant and amphipods are absent. Conversely, snails are scarce in most lakes of the Strobel Plateau and amphipods are abundant (Adami 2016).
The presence of predators can induce anti-predatory responses, altering the morphology, behaviour and spatial distribution of their prey (Simon and Townsend 2003; Ferrari et al. 2010; Lancelotti et al. 2017). These changes in prey behaviour are energy costly, and organisms face trade-offs between the benefits of a reduced predation risk and mortality and a potentially lower fitness associated with foraging or mating (Preisser et al. 2005). In the Strobel Plateau, amphipods represent the main trout prey (Bandieri 2011; Lancelotti et al. 2015). A study based on SIA in more than 12 water bodies showed that in the absence of trout, amphipods depend on both littoral and pelagic resources (Lancelotti 2009), being as much abundant in the water column as in littoral zones of fishless lakes, while in stocked lakes, they are restricted to littoral and benthic areas (Lancelotti, unpublished data).
A natural experiment with SIA comparing fishless and stocked lakes of the Strobel Plateau elucidated ecological questions about non-native fish introductions. These findings evidenced that the presence of O. mykiss has a marked influence on the food web structure and nutrient cycles of the analysed lakes (Porcel 2020), as shown in Fig. 14.4. Fish modify the trophic structure of the aquatic community of stocked lakes in this plateau, as demonstrated by a clear shift in the total isotopic niche size of the stocked lakes, towards more positive values of δ13C and δ15N (Fig. 14.4).
Simplified diagram of aquatic food webs in stocked and fishless organic lakes, based on a “natural experiment” carried out in the Strobel Plateau. The y-x axes show the δ15N and δ13C isotope values of the most abundant groups. δ15N values reflect a consumer’s trophic level (Post 2002). δ13C values show contrasting carbon isotope signals depending on the habitat: organisms inhabiting littoral and benthic zones tend to have more positive δ13C compared to those that inhabit pelagic zones (France 1995b). The aquatic community is represented by three trophic levels (TL) composed of the main taxonomic groups that inhabit these environments and the basal levels (BL): biofilm and particulate organic matter (POM), corresponding to the pelagic and littoral/benthic pathways. The big arrow indicates a clear shift in values of δ15N and δ13C between food webs with and without O. mykiss. The thin arrow shows the shift in the spatial distribution of amphipods towards littoral zone of the stocked lakes as an anti-predatory response. The circular arrow represents the increase in organic matter resuspension and nutrient recycling by fish presence
These differences in isotopic values between fishless and stocked lakes were explained by the presence of trout but also by multiple factors related to natural variations among the analysed environments such as changes in water levels, drainage patterns and substrate composition (Vanni 1996; Schindler and Scheuerell 2002; Feniova et al. 2016). The increase in basal δ15N values together with the higher concentrations of dissolved organic carbon and higher conductivity registered in the stocked lakes was related to the presence of fish. Fish introductions increase organic matter resuspension and nutrient recycling. The microbiological decomposition of organic matter results in the initial release of isotopically light nitrogen like ammonia, and the residual organic matter becomes isotopically heavier (Macko and Estep 1985). Decomposition outcomes are observed in an accumulation of isotopically heavy organic nitrogen at the deeper depths, which can then be incorporated by the periphyton, grazing invertebrates and finally their predators such as fish (Estep and Vigg 1985). Additionally, the use of community metrics evidenced that the organisms belonging to lakes with O. mykiss had a more uniform species distribution, indicating greater trophic redundancy (Porcel 2020). This finding was related to the fact that fish and the big carnivorous copepod (P. sarsi) occupied a similar trophic role, resulting in an unaltered trophic dimension, but with a higher number of components. Other studies, also based on community metrics, reported that the introduction of a new predator caused a reduction of prey species, thus deriving on a homogenisation of energy flow pathways to top predators in tidal creeks in the Bahamas (Layman et al. 2007). Similarly, Vidal et al. (2020) suggested that fish feeding in the pelagic zone in Tasmanian lakes caused the reduction of trophic diversity of zooplankton.
Porcel (2020) also focused on individual- and population-level impacts by fish introductions in stocked lakes of the Strobel Plateau. The findings showed that O. mykiss generate changes in the spatial distribution and/or diet of amphipods (main prey of trout), inducing the use of alternative carbon sources (Fig. 14.4). In stocked lakes, amphipods showed higher dependency of benthic/littoral resources, which could respond to anti-predatory responses to fish presence, using macrophyte beds or rocks as refuges against predators (Porcel et al., “unpublished data”). Similar changes in the spatial distribution of prey following the introduction of fish were observed in other aquatic systems (Gliwicz and Rowan 1984; Fraser and Huntingford 1986; Simon and Townsend 2003; Stuart-Smith et al. 2008; Martínez-Sanz et al. 2010; McHugh et al. 2012). For example, the introduction of Salvelinus fontinalis in high-altitude European lakes altered the distributional pattern of the copepod Cyclops abyssorumtatricus, shifting to a near-bottom maximum, which is not typically seen in fishless lakes. The ability of C. abyssorumtatricus to alter its depth distribution has allowed its persistence in stocked lakes, from which other species have disappeared (Gliwicz and Rowan 1984).
7 Joint Influence of Fish Introduction and Climate Change
The lakes located in the Patagonian plateaus exhibit a very dynamic hydrology associated with changes in precipitation and snowfall, which account for changes in their limnological features (e.g. macrophyte cover and turbidity; Lancelotti 2009; Lancelotti et al. 2010a). In general, lakes are very sensitive to the changes in water level, thus constituting excellent sentinels of climate change (e.g. Adrian et al. (2009), and Schindler (2009)). Particularly, some shallow lakes of Argentina are good examples of the switch in lake regime associated with documented variations in water levels (e.g. O’Farrell et al. (2011), and Sánchez et al. (2014)).
In Patagonia, some climatological studies have projected a decrease in precipitation and an increase in temperature (Barros et al. 2014). This may trigger a reduction in the discharge of rivers and a water level decrease in lentic water bodies (Lancelotti et al. 2020). In particular, for the Strobel Plateau, the study by Izaguirre et al. (2018) reported a marked reduction in the water level of many arheic shallow lakes, some of which dried up completely in 2016. This decrease in the hydrometric level produced an increase in conductivity, and several lakes changed their state, from a clear vegetated to a turbid condition, with higher chlorophyll a, and declining biomass of submerged macrophytes.
A strong decrease in most arheic basins in many lakes of the Patagonian plateaus was observed during the field studies from 2006 to 2016, also confirmed by the study of time series of climate data and satellite images of the Strobel Plateau (Lancelotti et al. 2020). For the lakes of the Buenos Aires Plateau, the water level decrease also triggered the shift from clear vegetated to turbid states (Porcel et al. 2019).
The joint effect of fish introduction and water level decrease was first explored for the lakes of the Patagonian plateaus by Izaguirre et al. (2018). This study included data obtained in 31 lakes of the Strobel and Buenos Aires Plateaus, which were surveyed from 2007 to 2016. Fishless and fish stocked lakes were compared under contrasting hydrological conditions (relatively stable water levels and decreasing water levels). The reduction in precipitation, with the concomitant effect in the water level, was established as a key driver shaping both macrophytes and plankton communities. On the other hand, it was also reported that fishless lakes presented higher zooplankton and macrozooplankton biomass and lower phytoplankton biomass, in coincidence with the other studies already mentioned. Since both fish introduction and water level decrease may provoke an increase in phytoplankton biomass and turbidity, it was hypothesised that under a scenario of climate change, the reduction in precipitation may enhance the effects of fish introduction, as illustrated in Fig. 14.5.
As it was previously mentioned, fish stocked lakes develop higher biomass of autotrophic pico-, nano- and microplankton than fishless lakes, and this is emphasised during periods of low hydrometric levels (Saad et al. 2019). In the lakes of the Buenos Aires Plateau, Porcel et al. (2019) also observed a gradual increase in conductivity, turbidity, nutrients and DOC associated with the drought process that occurred from 2015 to 2017, also affecting the picoplankton structure.
8 Potential Effects of Trout and Climate on Hooded Grebe
Climate change and fish introduction in lakes naturally lacking them seem to exert a synergic effect on the limnological conditions of lakes, which constitute the prime habitats for Patagonian waterbird endemic species, including some threatened and near-threatened species (see Chap. 15). The concomitant effects of climate fluctuations and trout could have drastic consequences on the hooded grebe survival. The dry periods reduce the habitat availability (number of lakes) and quality (i.e. macrophyte cover and robustness) (Izaguirre et al. 2018; Lancelotti et al. 2020). These effects could be maximised due to alterations of food webs and nutrient recycling caused by trout, which could result in trophic cascades and habitat loss for hooded grebe. Additionally, the reduction of suitable habitat for hooded grebe during dry periods increases habitat overlap with trout. These two species are generalist predators that potentially depend on the same prey spectrum (Porcel 2020). Therefore, the co-occurrence of both species could originate competitive interactions, resulting particularly negative during the breeding season of hooded grebe. Trout can lead to the reduction of zooplankton size and abundance and also the relocation of amphipods (one of the main prey species for trout and grebe) from open waters to benthic/littoral zones. All these alterations could have substantial negative effects, from a nutritional and bioenergetic point of view, on adults, fledgling and chicks of hooded grebe. Besides, there are records of the desertion of active colonies due to an insufficient food supply in lakes (Fjeldså 1986).
The dry conditions observed during the last 10 years produced a significant reduction of lake availability, including most of the reproductive habitats for hooded grebe, also increasing the spatial overlap between trout and grebe’s habitats (Lancelotti et al. 2020). Dry conditions were also proposed as the potential cause of macrophyte degradation, with the consequent failure of the reproduction of hooded grebe during the last three breeding seasons (Roesler’s “personal communication”). Besides, the introduction of trout has been listed among the primary threats for hooded grebe (Roesler 2016).
9 Conclusion
In this chapter, we provide an overview and a synthesis of the current information about the ecological impacts associated with O. mykiss introductions in lakes of the arid southern Patagonia. The information herein reported is expected to contribute to understand the functioning of these ecosystems and, above all, to generate management tools based on biological and ecological data. We gathered fundamental data regarding the characterisation of these aquatic environments, also showing key information to understand the local and regional processes and mechanisms that structure communities, from primary producers and bacteria to top predators. Furthermore, food web studies specifically showed the effects of trout introduction on nutrient and energy pathways and their potential effects on these environments.
Preserving the water bodies of the basaltic plateaus in their natural condition is crucial for the conservation of biodiversity. Particularly, changes in the plankton and macroinvertebrates communities due to the presence of fish in lakes of the Strobel Plateau could, in turn, influence other predatory organisms, such as the aquatic birds. Under a climate change scenario, the adverse effects of the introduction of fish on the structure of aquatic communities could be magnified. For this reason, in recent years, restoration measures have been proposed and even applied in lakes stocked with O. mykiss, such as in the “Islote” Lake, where trout removal using gill nets was monitored by limnological studies to evaluate the “recovery” capacity of the lake. While this represents a significant first step, a permanent trout eradication initiative is needed in several lakes of the Strobel Lake Plateau to transform the present aquaculture into a more sustainable scheme in terms of habitat conservation.
References
Adami MA (2016) Trophic structure of a shallow lake key for the reproduction of the Hooded Grebe (Podiceps gallardoi). Graduate Thesis, Universidad Nacional de la Patagonia San Juan Bosco
Adamowicz SJ, Hebert PDN, Marinone MC (2004) Species diversity and endemism in the Daphnia of Argentina: a genetic investigation. Zool J Linn Soc Lond 140:171–205
Adrian R, Reilly CMO, Zagarese H et al (2009) Lakes as sentinels of climate change. Limnol Oceanogr 54:2283–2297
Anderson RS (1980) Relationships between trout and invertebrate species as predators and the structure of the crustacean and rotiferan plankton in mountain lakes. In: Kerfoot WC (ed) Evolution and ecology of zooplankton communities. University Press of New England, Hanover, pp 635–641
Bahls P (1992) The status of fish populations and management of high mountain lakes in the western United States. Northwest Sci 66:183–193
Bandieri L (2011) Diet and growth of rainbow trout (Oncorhynchus mykiss) in shallow lakes of the Strobel Lake plateau, Santa Cruz. Graduate Thesis. Universidad Nacional de la Patagonia San Juan Bosco
Barros VR, Boninsegna JA, Camilloni IA et al (2014) Climate change in Argentina: trends, projections, impacts and adaptation. Wiley Interdiscip Rev Clim Change 6:151–169
Beisner B, Haydon D, Cuddington K (2003) Alternative stable states in ecology. Front Ecol Environ 1:376–382
Beltrán J, Bertonatti C, Johnson A et al (1992) Updates on the distribution, biology and conservation status of the Hooded Grebe (Podiceps gallardoi). El Hornero 13:193–199
BirdLife International (2021) Species factsheet: Podiceps gallardoi. Downloaded from http://www.birdlife.org. Accessed 17 Feb 2021
Blanco S (2001) Estudio Experimental sobre la Influencia delos Nutrientes en la Ecología Trófica de los Peces de unaLaguna Somera. Master Thesis. Universidad de León, España
Blanco S, Fernández-Aláez M, Bécares E (2008) Efficiency of top-down control depends on nutrient concentration in a Mediterranean shallow lake: A mesocosm study. Marine and Freshwater Research 59:922–930
Buria L, Albariño R, Villanueva V et al (2007) Impact of exotic rainbow trout on the benthic macroinvertebrate community from Andean-Patagonian headwater streams. Arch Hydrobiol 168:145–154
Cabrera AL (1976) Argentine phytogeographic regions. In: Enciclopedia argentina de agricultura y jardinería. Tomo II. Fascículo 1. ACME, Buenos Aires, p 1–85
Canevari P, Blanco DE, Bucher EH et al (1999) Wetlands of Argentina: classification, current situation, conservation and legislation, vol 46. Wetlands International, Buenos Aires
Carpenter SR, Kitchell JF (1993) The trophic cascade in lakes. Cambridge Univ Press, Cambridge
Carpenter SR, Kitchell JF, Hodgson JR (1985) Cascading trophic interactions and lake productivity. Bioscience 35:634–639
Carpenter SR, Kitchell JF (1996) The trophic cascade in lakes. Cambridge University Press
Caudill CC (2003) Empirical evidence for nonselective recruitment and a source-sink dynamic in a mayfly meta population. Ecology 84:2119–2132
Coesel PF, Porcel S, Van Geest A et al (2017) Remarkable desmid species from the southern Patagonian highlands. Fottea 17:89–95. https://doi.org/10.5507/fot.2016.019
Correa MN (1998) Patagonian flora. Key for the determination of the families of Phanerogams of the Patagonian Flora. Secretaría de Agricultura, Ganadería y Pesca de la Nación, Buenos Aires
Covich AP, Palmer MA, Crowl TA (1999) The role of benthic invertebrate species in freshwater ecosystems. Bioscience 49:119–127
Crowl TA, Townsend CR, McIntosh AR (1992) The impact of introduced brown and rainbow trout on native fish: the case of Australasia. Rev Fish Biol Fish 3:217–241
Cuello M, Bello M, Kun M et al (2006) Feeding habits and their implication for the conservation of the endangered semiaquatic frog Atelognathus patagonicus (Anura, Neobatrachia) in a northwestern Patagonian pond. Phyllomedusa 5:67–76
D’Orazio M, Agostini S, Mazzarini F et al (2000) The Pali Aike Volcanic Field, Patagonia: slab-window magmatism near the tip of South America. Tectonophysics 4:407–427
Di Giacomo AS (2005) Important areas for the conservation of birds in Argentina. In: Priority sites for conservation of biodiversity. Aves Argentinas/Asociación Ornitológica del Plata, Buenos Aires
Díaz M, Pedrozo F, Baccala N (2000) Summer classification of southern hemisphere temperate lakes (Patagonia, Argentina). Lakes Reserv Res Manag 5:213–229
Eby LA, Roach WJ, Crowder LB et al (2006) Effects of stocking-up freshwater food webs. Trends Ecol Evol 21:576–584
Epanchin PN, Knapp RA, Lawler SP (2010) Nonnative trout impact an alpine-nesting bird by altering aquatic-insect subsidies. Ecology 91:2406–2415
Estep ML, Vigg S (1985) Stable carbon and nitrogen isotope tracers of trophic dynamics in natural populations and fisheries of the Lahontan Lake System, Nevada. Can J Fish Aquat Sci 42:1712–1719
Feniova IY, Rzepecki M, Zilitinkevicz NS et al (2016) Experimental impacts of fish on small and large cladocerans under eutrophic conditions. Inland Water Biol 9:375–381. https://doi.org/10.1134/S1995082916040040
Ferrari MCO, Wisenden BD, Chivers DP (2010) Chemical ecology of predator–prey interactions in aquatic ecosystems: a review and prospectus. Can J Zool 88:698–724
Fjeldså J (1986) Feeding ecology and possible life history tactics of the Hooded grebe Podiceps gallardoi. Ardea 74:40–58
France RL (1995a) Carbon-13 enrichment in benthic compared to planktonic algae: food web implications. Mar Ecol Progr Ser 115:205–207
France RL (1995b) Differentiation between littoral and pelagic food webs in lakes using stable carbon isotopes. Limnol Oceanogr 40:1310–1313
Fraser DE, Huntingford FA (1986) Feeding and avoiding predation hazard: the behavioral response of the prey. Ethology 73:56–68
Gliwicz Z, Pijanowska J (1989) The role of predation in zooplankton succession. In: Sommer U (ed) Plankton ecology. Springer Verlag, Heidelberg, pp 253–296
Gliwicz ZM, Rowan MG (1984) Survival of Cyclops abyssorum tatricus (Copepoda, Crustacea) in alpine lakes stocked with planktivorous fish. Limnol Oceanogr 29:1290–1299
Gozlan RE (2008) Introduction of non-native freshwater fish: is it all bad? Fish 9:106–115
Izaguirre I (1993) Comparative analysis of the phytoplankton of six lentic environments from the Province of Chubut (Argentina). Physis (Buenos Aires) 48:7–23
Izaguirre I, Saad FJ (2014) Phytoplankton from natural water bodies of the Patagonian Plateau. Adv Limnol 65:309–319. https://doi.org/10.1127/1612-166X/2014/0065-0048
Izaguirre I, Del Giorgio P, O’Farrell I et al (1990) Classification of 20 Andean-Patagonian water bodies (Argentina) based on the structure of summer phytoplankton. Cryptogam Algol 11:31–46
Izaguirre I, Unrein F, Modenutti B et al (2014) Photosynthetic picoplankton in Argentina lakes. Adv Limnol 65:343–357. https://doi.org/10.1127/1612-166X/2014/0065-0050
Izaguirre I, Lancelotti J, Saad FJ et al (2018) Influence of fish introduction and water level decrease on lakes of the arid Patagonian plateaus with importance for biodiversity conservation. Glob Ecol Conserv 14:e00391. https://doi.org/10.1016/j.gecco.2018.e00391
Jeppesen E, Jensen JP, Søndergaard M et al (1997) Top-down control in freshwater lakes: the role of nutrient state, submerged macrophytes and water depth. Hydrobiologia 342(343):151–164
Jobbágy EG, Paruelo JM, León RJC (1995) Estimation of the precipitation regime from the distance to the mountain range in northwestern Patagonia. Ecol Austral 5:47–53
José de Paggi S (1990) Ecological and biogeographical remarks on the rotifer fauna of Argentina. Rev Hydrobiol Trop 23:297–311
Knapp RA (2005) Effects of nonnative fish and habitat characteristics on lentic herpetofauna in Yosemite National Park, USA. Biol Conserv 121:265–279
Knapp RA, Matthews KR, Sarnelle O (2001) Resistance and resilience of alpine lake fauna to fish introductions. Ecol Monogr 71:401–421
Lancelotti JL (2009) Limnological characterisation of shallow lakes in the Province of Santa Cruz and effects of the introduction of Rainbow Trout (Oncorhynchus mykiss) on the receiving communities. PhD Thesis, Universidad Nacional del Comahue
Lancelotti JL, Pozzi LM, Yorio PM et al (2009a) Fishless shallow lakes of Southern Patagonia as habitat for waterbirds at the onset of trout aquaculture. Aquat Conserv: Mar Freshw Ecosyst 505:497–505. https://doi.org/10.1002/aqc.1018
Lancelotti JL, Pozzi LM, Márquez F et al (2009b) Waterbird occurrence and abundance in the Strobel Plateau, Patagonia Argentina. El Hornero 24:13–20
Lancelotti JL, Pascual MA, Gagliardini A (2010a) A dynamic perspective of shallow lakes of arid Patagonia as habitat for waterbirds. In: Meyer PL (ed) Ponds: formation characteristics and uses. Nova Science Publishers, New York, pp 83–102
Lancelotti JL, Pozzi LM, Yorio PM et al (2010b) Precautionary rules for exotic trout aquaculture in fishless shallow lakes of Patagonia: minimizing impacts on the threatened hooded grebe (Podiceps gallardoi). Aquat Conserv: Mar Freshw Ecosyst 8:1–8. https://doi.org/10.1002/aqc.1067
Lancelotti JL, Bandieri LM, Pascual MA (2015) Diet of the exotic rainbow trout in the critical habitat of the threatened Hooded Grebe. Knowl Manag Aquat Ecosyst 416(26):1–11. https://doi.org/10.1051/kmae/2015022
Lancelotti JL, Marinone MC, Roesler CI (2017) Rainbow trout effects on zooplankton in the reproductive area of the critically endangered hooded grebe. Aquat Conserv: Mar Freshw Ecosyst 27:128–136. https://doi.org/10.1002/aqc.2629
Lancelotti JL, Pessacg NL, Roesler IC et al (2020) Climate variability and trends in the reproductive habitat of the critically endangered hooded grebe. Aquat Conserv: Mar Freshw Ecosyst 30:554–564. https://doi.org/10.1002/aqc.3240
Layman CA, Quattrochi JP, Peyer CM et al (2007) Niche width collapse in a resilient top predator following ecosystem fragmentation. Ecol Lett 10:937–944. https://doi.org/10.1111/j.1461-0248.2007.01087.x. PMID: 17845294
Llanos E, Behr S, González J et al (2016) Report on the variations of Lake Colhue Huapi by remote sensing and its relationship with precipitation. Technical report. Instituto Nacional de Tecnología Agropecuaria, Trelew, pp 1–8
Lopez ME, Saad J, Izaguirre I et al (2021) Aquatic macroinvertebrates from littoral vegetated lakes in arid Patagonia: the influence of fish introduction on community structure. Aquat Conserv: Mar Freshw Ecosyst:1–13. https://doi.org/10.1002/aqc.3522
Luecke C (1990) Changes in abundance and distribution of benthic macroinvertebrates after introduction of cutthroat trout into a previously fishless lake. Trans Am Fish Soc 119:1010–1021
Macchi PJ, Cussac VE, Alonso MF et al (1999) Predation relationships between introduced salmonids and the native fish fauna in lakes and reservoirs in northern Patagonia. Ecol Freshw Fish 8:227–236. https://doi.org/10.1002/aqc.696
Mack RN, Simberloff D, Lonsdale WM et al (2000) Biotic invasions: causes, epidemiology, global consequences, and control. Ecol Appl 10:689–710
Macko SA, Estep MLF (1985) Microbial alteration of stable nitrogen and carbon isotopic compositions of organic matter. Org Geochem 6:787–790
Manzo LM, Epele LB, Grech MG et al (2019) Wetland genesis rules invertebrate spatial patterns at Patagonian ponds (Santa Cruz, Argentina): a multiscale perspective. Ecol Eng 126:43–54. https://doi.org/10.1016/j.ecoleng.2018.10.026
Marinone MC, Menu Marque SA (2010) Planktonic microcrustacean (Cladocera and Copepoda) assemblages from inland waters of the province of Santa Cruz, Argentina. In: Corbella H, Maidana NI (eds) 1ª Reunión Internodos PIPA, 1er Workshop Argentino del Proyecto PotrokAike Maar Lake Sediment Archive Drilling Project. Proyecto Editorial PIPA, Buenos Aires, pp 41–44. http://www.pasado.uni-bremen.de/Files/abstracts%20PIPA-PASADO-small.pdf
Marinone MC, Menu Marque S, Añón Suárez D et al (2006) UVR radiation as a potential driving force for zooplankton community structure in Patagonian lakes. Photochem Photobiol 82:962–971. https://doi.org/10.1562/2005-09-09-RA-680
Martínez-Sanz C, García-Criado F, Aláez F et al (2010) Effects of introduced salmonids on macroinvertebrate communities of mountain ponds in the Iberian system of Spain. Limnetica 29:221–232
Masiokas M, Villalba R, Luckman BH et al (2008) 20th-century glacier recession and regional hydroclimatic changes in the northwestern Patagonia. Glob Planet Chang 60:85–100. https://doi.org/10.1016/j.gloplacha.2006.07.031
McHugh P, McIntosh A, Howard S et al (2012) Niche flexibility and trout–galaxiid co-occurrence in a hydrologically diverse riverine landscape. Biol Invasions 14:2393–2406
McNaught AS, Schindler DW, Parker BR et al (1999) Restoration of the food web of an alpine lake following fish stocking. Limnol Oceanogr 44:127–136. https://doi.org/10.4319/lo.1999.44.1.0127
Menu-Marque S, Morrone JJ, Locascio de Mitrovich C (2000) Distributional patterns of the South American species of Boeckella (Copepoda: Centropagidae): a track analysis. J Crustac Biol 20:262–272. https://doi.org/10.1651/0278-0372(2000)020[0262:DPOTSA]2.0.CO;2
Mittelbach GG, Turner AM, Hall DJ et al (1995) Perturbation and resilience: a long-term, whole-lake study of predator extinction and reintroduction. Ecology 76:2347–2360
Modenutti BE, Balseiro EG (1994) Zooplankton size spectrum in four lakes of the Patagonian Plateau. Limnologica 24:51–56
Morello J, Matteucci S, Rodríguez A et al (eds) (2012) Argentine ecoregions and ecosystem complexes, First edition. Facultad de Arquitectura, Diseño y Urbanismo, GEPAMA Grupo de Ecología del Paisaje y Medio Ambiente. Universidad de Buenos Aires, Buenos Aires
Newman RM, Waters TF (1984) Size-selective predation on Gammarus pseudolimnaeus by trout and sculpins. Ecology 65:1535–1545
O’Farrell I, Izaguirre I, Chaparro G et al (2011) Water level as the main driver of the alternation between a free-floating plant and a phytoplankton dominated state: a long term study in a floodplain lake. Aquat Sci 73:275–287
Ortubay S, Cussac V, Battini M et al (2006) Is the decline of birds and amphibians in a steppe lake of northern Patagonia a consequence of limnological changes following fish introduction? Aquat Conserv: Mar Freshw Ecosyst 16:93–105
Pace ML (1993) Heterotrophic microbial processes. In: Carpenter SR, Kitchell JF (eds) The trophic cascade in lakes. Cambridge University Press, Cambridge, pp 252–277
Pace ML, Cole JJ, Carpenter SR et al (1999) Trophic cascades revealed in diverse ecosystems. Trends Ecol Evol 14:483–488
Panza JL, Franchi MR (2002) Extra-Andean Cenozoic Basaltic Magmatism. In: Haller MJ (ed) Geología y Recursos Naturales de Santa Cruz. Relatorio del XV congreso Geológico Argentino, El Calafate, p 201–236
Parker BR, Schindler DW (2006) Cascading trophic interactions in an oligotrophic species-poor alpine lake. Ecosystems 9:157–166
Paruelo JM, Beltrán A, Jobággy E et al (1998) The climate of Patagonia: general patterns and controls on biotic processes. Ecol Austral 8:85–101
Pascual M, Macchi P, Urbanski J et al (2002) Evaluating potential effects of exotic freshwater fish from incomplete species presence – absence data. Biol Invasions 4:101–113
Pasquini AI, Lecomte KL, Depetris PJ (2008) Climate change and recent water level variability in Patagonian proglacial lakes, Argentina. Glob Planet Change 63:290–298. https://doi.org/10.1016/j.gloplacha.2008.07.001
Pereyra FX, Fauqué L, González Díaz EF (2002) Geomorphology. In: Haller MJ (ed) Geología y Recursos Naturales de Santa Cruz. Relatorio del XV Congreso Geológico Argentino, El Calafate, p 325–352
Perotti MG, Diéguez MC, Jara FG (2005) State of knowledge of wetlands in northern Patagonia (Argentina): relevant aspects and importance for the conservation of regional biodiversity. Rev Chil Hist Nat 78:723–737
Pessacg N, Flaherty S, Solman S et al (2020) Climate change in northern Patagonia: critical decrease in water resources. Theor Appl Climatol 140:807–822. https://doi.org/10.1007/s00704-020-03104-8
Polis GA (1999) Why are parts of the world green? Multiple factors control productivity and distribution of biomass. Oikos 83:3–15
Polis GA, Sears ALW, Huxel GR et al (2000) When is a trophic cascade a trophic cascade? Trends Ecol Evol 15:473–475
Porcel S (2020) Structure of planktonic communities in lakes of basaltic plateaus of Patagonia: influence of environmental variables and the introduction of exotic fish. PhD Thesis, Universidad de Buenos Aires
Porcel S, Saad JF, García CAS et al (2019) Microbial planktonic communities in lakes from a Patagonian basaltic plateau: influence of the water level decrease. Aquat Sci 81:51. https://doi.org/10.1007/s00027-019-0647-y
Porcel S, Chaparro G, Marinone MC et al (2020) The role of environmental, geographical, morphometric and spatial variables on plankton communities in lakes of the arid Patagonian plateaus. J Plankton Res 42(2):173–187. https://doi.org/10.1093/plankt/fbaa004
Post DM (2002) Using stable isotopes to estimate trophic position: models, methods, and assumptions. Ecology 83:703–718
Preisser EL, Bolnick DI, Benard MF (2005) Scared to death? The effects of intimidation and consumption in predator–prey interactions. Ecology 86:501–509
Rautio M, Vincent WF (2007) Isotopic analysis of the sources of organic carbon for zooplankton in shallow subarctic and artic waters. Ecography 30:77–87
Reissig M, Trochine C, Queimaliños C et al (2006) Impact of fish introduction on planktonic food webs in lakes of the Patagonian Plateau. Biol Conserv 132:437–447. https://doi.org/10.1016/j.biocon.2006.04.036
Roesler I (2016) Conservation of the Hooded Grebe (Podiceps gallardoi): factors that affect the viability of its populations. Argentina. PhD Thesis, Universidad de Buenos Aires
Roesler CI, Imberti S, Casañas H et al (2012) Hooded Grebe Podiceps gallardoi population decreased by eighty per cent in the last twenty-five years. Bird Conserv Int 22:371–382. https://doi.org/10.1017/S0959270912000512
Rothhaupt KO (2000) Plankton population dynamics: food web interactions and abiotic constraints. Freshw Biol 45:105–109
Rumboll MA (1974) Una nueva especie de Maca (PODICIPITIDAE)
Saad FJ, Porcel S, Lancelotti JL et al (2019) Both lake regime and fish introduction shape autotrophic planktonic communities of lakes from the Patagonian Plateau (Argentina). Hydrobiologia 831:133–145. https://doi.org/10.1007/s10750-018-3660-6
Sala OE, Chapin FS, Armesto JJ et al (2000) Global biodiversity scenarios for the year 2100. Science 287:1770–1774. http://www.ncbi.nlm.nih.gov/pubmed/10710299
Sánchez ML, Lagomarsino L, Allende L et al (2014) Changes in the phytoplankton structure in a Pampean shallow lake in the transition from a clear to a turbid regime. Hydrobiologia 752:65–76. https://doi.org/10.1007/s10750-014-2010-6
Schabetsberger R, Luger MS, Drozdowski G et al (2009) Only the small survive: monitoring long-term changes in the zooplankton community of an Alpine lake after fish introduction. Biol Invasions 11:1335–1345. https://doi.org/10.1007/S10530-008-9341-Z
Scheffer M (1998) Ecology of shallow lakes. Chapman & Hall, New York
Scheffer M, Carpenter SR, Foley JA et al (2001) Catastrophic shifts in ecosystems. Nature 413(6856):591–596
Schiaffino MR, Gasol JM, Izaguirre I et al (2013) Picoplankton abundance and cytometric group diversity along a trophic and latitudinal lake gradient. Aquat Microb Ecol 68(3):231–250. https://doi.org/10.3354/ame01612
Schindler DW (2009) Lakes as sentinels and integrators for the effects of climate change on watersheds, airsheds, and landscapes. Limnol Oceanogr 54:2349–2358
Schindler DW, Parker BR (2002) Biological pollutants: alien fishes in mountain lakes. Water Air Soil Pollut: Focus 2:379–397. https://doi.org/10.1023/A:1020187532485
Schindler DE, Scheuerell MD (2002) Habitat coupling in lake ecosystems. Oikos 98:177–189
Schindler DE, Knapp RA, Leavitt PR (2001) Alteration of nutrient cycles and algal production resulting from fish introductions into mountain lakes. Ecosystems 4:308–321
Scordo F, Seitz C, Zilio M et al (2018a) Evolution of water resources in the “Bajo de Sarmiento” (Extra Andean Patagonia): natural and anthropic impacts. Anu Inst Geociênc 40:106–117
Scordo F, Bohn VY, Piccolo MC et al (2018b) Mapping and monitoring lakes intra-annual variability in semi-arid regions: a case of study in Patagonian Plains (Argentina). Water 10(7):889. https://doi.org/10.3390/w10070889
Scott DA, Carbonell M (1986) A directory of neotropical wetlands. IUCN Conservation Monitoring Centre, Gland
Schaus MH, Vanni MJ, Wissing TE (2002) Biomass dependent diet shifts in omnivorous gizzard shad: implicationsfor growth, food web, and ecosystem effects. Transactionsof the American Fisheries Society 131:40–54
Simon KS, Townsend CR (2003) Impacts of freshwater invaders at different levels of ecological organisation, with emphasis on salmonids and ecosystem consequences. Freshw Biol 48:982–994
Stomp M, Huisman J, Vörös L et al (2007) Colourful coexistence of red and green picocyanobacteria in lakes and seas. Ecol Lett 10:290–298
Stuart-Smith RD, White RWG, Barmuta LA (2008) A shift in the habitat use pattern of a lentic galaxiid fish: an acute behavioural response to an introduced predator. Environ Biol Fish 82:93–100
Tagliaferro M, Arismendi I, Lancelotti JL et al (2014) A natural experiment of dietary overlap between introduced Rainbow Trout (Oncorhynchus mykiss) and native Puyen (Galaxias maculatus) in the Santa Cruz River, Patagonia. Environ Biol Fish 98:1311–1325. https://doi.org/10.1007/s10641-014-0360-6
Tejedo AG (2003) Soil degradation in the surroundings of Lake Colhué Huapi, Escalante, province of Chubut. In: Primer Congreso de la Ciencia Cartográfica y VII Semana Nacional de la Cartografía. Centro Argentino de Cartografía, Buenos Aires
Townsend CR (2003) Individual, population, community, and ecosystem consequences of a fish invader in New Zealand streams. Conserv Biol 17:38–47
Tyler TJ, Liss WJ, Hoffman RL et al (1998) Experimental analysis of trout effects on survival, growth, and habitat use of two species of ambystomatid salamanders. J Herpetol 32:345–349
Vander Zanden MJ, Vadeboncoeur Y (2002) Fishes as integrator of benthic and pelagic food webs in lakes. Ecology 83:2152–2161
Vander Zanden MJ, Shuter B, Lester N et al (1999a) Patterns of food chain length in lakes: a stable isotope study. Am Nat 154:406–416
Vander Zanden MJ, Casselman JM, Rasmussen JB (1999b) Stable isotope evidence for the food web consequences of species invasions in lakes. Nature 401:464–467
Vanni MJ (1996) Nutrient transport and recycling by consumers in lake food webs: implications for algal communities. In: Polis GA, Winemiller KO (eds) Food webs: integration of patterns and dynamics. Chapman and Hall, New York, pp 81–95
Vanni MJ (2002) Nutrient cycling by animals in freshwater ecosystems. Annu Rev Ecol Syst 33:341–370
Vidal N, Trochine C, Amsinck SL et al (2020) Interaction between non-native predatory fishes and native galaxiids (Pisces: Galaxiidae) shapes food web structure in Tasmanian lakes. Inland Waters 10:212–226. https://doi.org/10.1080/20442041.2020.1724047
Walbaum JJ (1792) Petri Artedi Sueci Genera Piscium. In quibus systema totum ichthyologiae proponitur cum classibus, ordinibus, generum characteribus, specierum differentiis, observationibus plurimis. Redactis Speciebus 242 ad Genera 52. ichthyologiae pp 59
Wiley RW (2006) Diversifying trout fishing opportunity in Wyoming: history, challenges, and guidelines. Fisheries 31:548–553
Acknowledgements
Most of the information reviewed in this chapter derives from studies that were possible thanks to the logistic support and the facilities provided by the Proyecto Macá Tobiano in Strobel and Buenos Aires Plateaus, Rodríguez family (Ea. Lago Strobel), Laguna Verde Lodge and Jurassic Lake Lodge in the Strobel Plateau and the facilities provided by the “Parque Nacional Patagonia” in the Buenos Aires Plateau. We thank the collaboration of I. Roesler, G. Tell, F. Unrein, R. Sinistro, A. Rua, C. Sabio y García, M.C. Diéguez, I. O’Farrell, V. Rago and the technicians and volunteers of the Proyecto Macá Tobiano/Programa Patagonia (Aves Argentinas) during the field trips to the plateaus. We also thank M.L. Fogel and the EDGE institute (UC Riverside) for their support of the project that allowed the isotopic analysis research. The cited studies were financed by the following grants: FONCYT PICT 32732 and FONCYT PICT 2013-0794 of the Argentine Fund for Scientific and Technical Investigations; Proyecto Macá Tobiano (Aves Argentinas/Ambiente Sur), Programa Patagonia Aves Argentinas (by Toyota Argentina, PAE, BirdLife International Preventing Extinction Programme (B. Olewine and S. & B. Thal), ICFC Canada, Toyota Environmental Activities Grant Programme, ZSL-EDGE, Secretaría de Ambiente de Santa Cruz and Patagonia Inc.); Technical Advisory Agreement IEASA-CONICET-Macá Tobiano; University of California’s EDGE Institute; and the Tonolli Award 2017 awarded by the International Society of Limnology.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Porcel, S., Saad, J.F., Marinone, M.C., Izaguirre, I., Lancelotti, J. (2022). Influence of the Fish Introduction in Lakes of the Arid Southern Patagonia. In: Mataloni, G., Quintana, R.D. (eds) Freshwaters and Wetlands of Patagonia. Natural and Social Sciences of Patagonia. Springer, Cham. https://doi.org/10.1007/978-3-031-10027-7_14
Download citation
DOI: https://doi.org/10.1007/978-3-031-10027-7_14
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-10026-0
Online ISBN: 978-3-031-10027-7
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)