Abstract
This article comprises a variety of comparisons between the several approaches employed for total hip arthroplasty (THA), highlighting both the strengths and weaknesses attributed to each approach. Moreover, it provides additional information regarding staged and simultaneous bilateral THA, as well as the most suitable methods for the treatment of femoral fractures in the elderly. Most of the comparisons included in this work provide details that juxtapose one of the most utilized surgical approaches, the DAA, with other conventional approaches and novel technique of supercapsular percutaneously assisted approach (SuperPATH). Additionally, we provide information on comparisons of the relative outcomes of conventional approaches and robotic THA, as well as an examination of the capsulectomy and capsulotomy techniques.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
1 Introduction
THA procedure can be performed via a variety of approaches, each presenting specific strengths, as well as weaknesses, mainly correlated to the methodology used to access the hip joint in order to ultimately implant the prosthetic components. The most commonly used approach is the direct anterior approach, and its growing popularity is attributed to the muscle-sparing technique that characterizes it, which allows for superior cosmetic appearance, as well as inferior intraoperative blood loss, shorter hospital stay, and decreased pain perceived by the patient in the initial stages following the surgical procedure [1]. Because of the aforementioned positive outcomes correlated to the DAA, this particular approach has been repeatedly analyzed and compared to other conventional approaches (CAs), some of which are summarized throughout this work. Some of the other analyzed approaches include the SuperPATH, robotic THA, hemiarthroplasty, capsular repair, capsulectomy, and proximal femoral nail antirotation.
In patients presenting bilateral arthritis, the THA procedure can be performed in either one or two stages, thus a brief but comprehensive summary of the relative advantages and downsides correlated to the performance of staged bilateral or simultaneous bilateral THA has been included.
2 Comparison of Minimum 2-Year Outcomes Following DAA and PA in Primary THA
The data gathered in [2] were retrospectively collected and reviewed and included patients that had undergone primary total hip replacement surgery with the senior surgeon (BGD) and had a minimum 2-year patient-recorded outcomes (PROs), in the period ranging from 2008 to 2016. The PROs employed during the follow-up process consisted of a variety of tests including the Harris Hip Score (HHS), the Forgotten Joint Score-12 (FJS-12), the visual analogue scale (VAS), and patient satisfaction. Additionally, the Veterans RAND 12-Item Health Survey (VR-12 Physical and VR-12 Mental) and Heath Survey Short (SF-12 Physical and SF-12 Mental) were used to analyze both the physical and mental state of the patients.
All the patients included in the study underwent THA through either DAA or PA. For DAA hip arthroplasties—in which acetabular reaming and cup component positioning was performed with the use of fluoroscopic guidance—a traction table was utilized to allow for the capsule to be opened in a “T” shape fashion and closed with absorbable sutures with the patient positioned in the supine position [2]. For THAs performed through posterior approach (PA), the patient was placed in the lateral position to enable the identification of external rotators and their subsequent removal for exposure while simultaneously preserving the piriformis tendon when practicable. The capsule was subsequently identified and incised in an inverted “L” shape fashion and preserved during the surgical procedure to achieve transosseous repair, which was then performed with non-absorbable sutures [3, 4] to reattach the external rotators.
All patients followed a precise postoperative rehabilitation protocol, which consisted of physical therapy and at-home care for 1–2 weeks. An additional 6–8-week long rehabilitation program was then planned to improve patients’ strength and range of motion, alongside postoperative follow-up appointments consisting of radiographic evaluation at the 2-week, 3-month, and annual time points [2]. A total of 707 THAs were conducted during the length of the study, among which 470 underwent THA through the DAA and 237 through the posterior approach. Of all cases, 415 out of the 470 cases reported for DAA met the minimum 2-year follow-up, while a greater percentage was recorded for the PA, 215 out of the initial 237. A total of 16 complications were observed in the DAA group, among which 9 patients with superficial infections, 5 resolved with oral antibiotics, and the remaining 4 with irrigation and debridement. Moreover, 1 case of transient femoral nerve palsy and 2 cases of intraoperative femur fractures were observed and resolved over time. Only 3 out of the 16 previously mentioned complications led to revisions, 2 of which were relative to loosening and 1 to periprosthetic femur fracture. Regarding the PA group, a total of ten complications were recorded, among which three ulterior cases of superficial infections, all resolved with oral antibiotics, and a deep infection that required revision surgery. Furthermore, two patients experienced dislocations, which did not require an ensuing surgery following reduction under anesthesia, one patient was diagnosed with deep vein thrombosis, whereas two had sciatic nerve injury. Only one patient required revision surgery due to the loosening of the implant. According to the data gathered throughout the study, the DAA group reported remarkably better results regarding VR-12 Mental, VR-12 Physical, SF-12 Mental, and SF-12 Physical in addition to higher scores of patient satisfaction. The worse outcomes for the PA group might have been due to the potential decrease in THA stability postoperatively since, using this approach, external rotators are inevitably severed while the abductor muscles are preserved. Despite the numerous limitations of this study—among which the lesser length of the follow-up period for the PA group and the non-randomized, retrospective design chosen—it is possible to report favorable outcomes for both groups at a minimum 2-year follow-up, and achievement of superior quality of life accomplishments for the DAA group when compared with the scores registered for the same parameters in the PA group [2].
3 Direct Anterior Approach and Other Conventional THA Approach Comparisons
Total hip replacement is performed through six CAs, namely, the anterior, anterolateral, lateral transtrochanteric, lateral transgluteal, posterior, and posterolateral. Additionally, such approaches have been slightly altered for minimal invasiveness and utilization of shorter incisions to achieve complete visibility of the anatomical landmarks while sparing the underlying muscle tissues to ultimately obtain enhanced patient-reported results in terms of decreased pain, prompter rehabilitation process, and satisfaction regarding the cosmetic appearance of the site of surgery. However, not every patient undergoing THA is a good candidate for a minimally invasive procedure. In fact, patients presenting a body mass index over 30, muscular thighs, or grave deformities could not be subjected to an 8 cm incision, but the procedure could potentially be performed through a slightly reduced incision compared to the traditional one, ranging from 20 to 25 cm [6,7,8,9,10,11,12,13,14,15,16,17,18].
Among the several approaches previously listed, the direct anterior approach (DAA) has been described as beneficial, due to the axis followed during the incision, which dissects the internervous and intermuscular planes, therefore sparing both the sartorius and the tensor fasciae latae and leading to a decreased tissue trauma [19,20,21]. However, the advantages of this technique compared to other CAs are still uncertain, thus increasing the need for a more meticulous analysis of the immediate outcomes following THA performed via DAA compared to other conventional approaches to ultimately treat disorders affecting the hip, as well as fractures.
Data gathered throughout the analysis performed in [5] reported a 15.1-min longer procedure and higher values for intraoperative blood loss—amounting to 51.5 ml—in THA through DAA compared to other conventional approaches. Nonetheless, the visual analogue scale for pain was inferior by 0.8 points 1 day postoperatively, and the values obtained for Harris Hip Score were higher by 2.8 points 3 months following the procedure performed via direct anterior approach, presumably correlated to the incision length, which was 2.9 cm shorter compared to other approaches. Finally, radiological outcomes reported a 4.3° lower anteversion angle, as well as a 1.6° lower inclination angle of the acetabular cup for the DAA compared to other conventional approaches [5], therefore indicating a higher propensity in reaching an excessively flat inclination angle with respect to the optimal values indicated for the inclination of the acetabular cup and for anteversion, which range from 40° to 50° and from 10° to 25°, respectively [22].
Based on the overall results gathered throughout the study, it is possible to assess that the THA performed through direct anterior approach displayed superior short-term results compared to other conventional approaches, specifically regarding reduced postoperative pain, shorter incision, and increased results in terms of performance up to 3 months following the procedure.
4 Direct Anterior and Posterolateral THA Approaches’ Comparison
The incidence of dislocation after performing THA through the DAA has been demonstrated to be particularly low; moreover, this approach is associated with a faster recovery in the early period following the procedure [24,25,26,27]. However, advocates of the posterolateral approach (PLA) primarily focus on the higher incidence of complications and early revision occurring following THA through the DAA [28,29,30]. Therefore, the study conducted in [23] ultimately aims at establishing the influence of each of the two aforementioned approaches for THA on the perioperative outcomes and the early results regarding the functionality of the patients. To do so, nine publications including a total of 22,698 patients were analyzed. The DAA cohort comprised 2947 patients, while the population size included in the PLA group consisted of 19,751 individuals.
The mean difference (MD) indicated for the Harris Hip Score (HHS) within 6 months after the surgical procedure was 3.82 for the DAA group, and it was substantially higher than the one indicated for the PLA cohort, showing a statistically significant difference between the two groups. In contrast, the MD indicated for the HHS after 6 months was – 0.17 for the DAA cohort, and no significant difference was recorded between the two analyzed cohorts. The MD reported for the length of hospital stay of the DAA group was – 0.5, significantly lower than the one indicated for the PLA cohort. The MD calculated for length of procedure and loss of blood for the DAA cohort were 19.73 ml and 125.19 ml, respectively, higher compared to the ones recorded for the PLA group and displaying significant differences between the two groups.
The incidence of complications was recorded in seven studies, including a total of 566 patients and revealing a greater rate in the DAA during the follow-up interval. The radiographic results regarding the position of the femoral component after the surgical procedure were analyzed in two studies, involving a total of 133 patients, and displaying an analogous proportion of neutral placement between the two groups. Five studies included, instead, information concerning the inclination angle of the acetabular cup component, which was not statistically significant between the two cohorts and indicated a MD of 0.75 for the DAA group. Furthermore, two studies included data regarding the anteversion angle of the acetabular component, which was significantly inferior for the DAA (with a MD of – 4.30) compared to the PLA group.
In summary, the DAA cohort displayed earlier recovery of their functions compared to the PLA group, despite exhibiting a greater incidence of early complications and a longer intraoperative time (increased by a mean of 19.73 min), alongside a greater volume of intraoperative blood loss, by a mean of 125.19 ml. Finally, the position of the femoral component was analogous between the two groups; however, the anteversion angle displayed in the DAA group was decreased by a mean of 4.3° [23].
5 Postoperative Complication Comparison of the Direct Anterior and the Lateral THA Approaches
Total hip arthroplasty is one of the most commonly performed surgical procedures to treat conditions including osteoarthrosis (OA), osteonecrosis of the femoral head (ONFH), and femoral neck fractures (FNFs), ultimately yielding excellent results in terms of relief from the pain and improvement of the functionality of the patients. The lateral approach (LA) also includes the anterolateral approach, also called Watson-Jones , and the direct lateral approach, or Hardinge , and has been developed to optimize intraoperative visualizations of both the proximal femur and acetabulum while simultaneously preserving the soft tissue surrounding the posterior surface of the hip joint, thus resulting in a lower incidence of dislocation, ranging between 0.43% and 0.70% [34]. However, such approach has been associated with greater early postoperative pain, heterotopic ossification, and damage to the superior gluteal nerve, alongside longer hospitalization and rehabilitation period [32].
The direct anterior approach (DAA) is considered a variation of the Smith-Peterson anterior approach and is commonly correlated to decreased postoperative pain and shorter hospitalization and rehabilitation process; however, it is associated with severe complications including femoral fractures occurring intraoperatively, lesions of the lateral femoral cutaneous nerve, and early revision [35, 36].
As a consequence, the choice regarding the appropriate surgical approach used to perform the THA procedure remains controversial; therefore, the study conducted in [31] performed a review of the applications of the DAA and LA while also focusing on the assessment of the related complications occurring postoperatively. Thirteen articles were ultimately included in the study, analyzing a total of 24,853 hips, 9575 of which were subjected to DAA, and 15,278 to LA.
The incidence of surgical infection was reported in six studies, two of which [38, 39] indicated superficial infections, one [37] disclosed deep infections, and three [40,41,42] reported superficial, as well as deep infections. Overall, no statistically significant difference was indicated between the DAA (1966 hips) and LA groups (1356 hips), as the incidence of surgical site infections was 2.59% and 2.14%, respectively [31].
The postoperative dislocation rate was analyzed in six studies [37,38,39, 41, 43, 44] including a total of 23,028 hips, showing an incidence of 0.77% for the DAA group, and 0.18% for the LA group, thus indicating a substantially higher incidence for the DAA cohort. Four studies [37, 38, 41, 52, 53] analyzed the rate of malposition of the prosthetic component, comprising a total of 210 hips in the DAA group and 371 in the LA cohort. The obtained results indicated a significantly lower incidence for the DAA group, corresponding to 36.19%, compared to the LA cohort, 54.86%.
The rate of periprosthetic fractures was evaluated in five studies [40, 41, 44,45,46], which included 6953 hips in the DAA group and 9173 in the LA cohort, and reported an incidence of 1.05% and 0.41%, respectively, thus suggesting a greater rate for the DAA group.
Four articles [37, 39, 41, 44] examined the rate of prosthesis loosening for both the DAA cohort, comprising 7019 hips, and the LA group, composed of 9237. The reported results indicated a higher rate for the DAA group, 0.61%, compared to the 0.37% observed in the LA cohort.
The rate of nerve damages was analyzed in four studies [38, 40, 41, 47] and indicated a substantially higher rate for the DAA group (1478 hips), 0.95%, compared to the LA cohort (468 hips), 0%.
Only two studies [39, 41] evaluated the rate of heterotopic ossification for the DAA group, composed of 74 hips, and the LA one, composed of 102, showing no statistically significant difference between the two cohorts, mainly attributed to the small size of the analyzed sample.
The discrepancy in leg length was examined in four publications [39, 41, 42, 48], including 1661 hips in the DAA cohort and 1055 in the LA one, ultimately showing a significantly lower rate for the DAA cohort (1.87%) compared to the LA group (2.37%). The rate of Trendelenburg gait was analyzed in three articles [39, 40, 42], including a total of 416 hips in the DAA group, and 712 in the LA one, exhibiting an incidence of 1.68% and 4.78%, respectively, thus suggesting a significantly higher trend in the LA group.
The rate of reintervention was examined in six studies [39,40,41,42,43,44], ultimately displaying no significant difference as the evaluated incidence was 2.70% for the DAA group, composed of 3596 hips, and 2.11% for the LA cohort, composed of 6028.
Infections occurring at the wound site during the DAA have been associated with a variety of factors that increase its incidence, including a higher body mass index for the patients (BMI ≥35 kg/m2) [49,50,51]. However, the study didn’t indicate any significant differences compared to the LA approach, thus suggesting that the BMI of the included population was inferior to 35 kg/m2.
In general, the incidence of dislocation observed in the DAA group was significantly higher compared to the LA; however, malposition of the prosthetic component was significantly lower in the DAA group, thus suggesting that the higher dislocation rates observed after the DAA are not correlated to malposition, rather to the release of the tendon and capsule surrounding the hip. The rate of fractures occurring after implantation of the prosthesis and loosening of the latter was substantially higher in the DAA group compared to the LA one, presumably due to the complexity of achieving optimal exposure for the preparation of the femur and subsequent implantation of the prosthesis. The DAA group also demonstrated a higher incidence of nerve damages compared to the LA group, whereas a lower rate of leg discrepancy was indicated for the DAA group showing that the supine position of the patient during such approach led to more precise placement of the implant and consequent control of the length of the limb. Similarly, the DAA procedure impacted the gait mechanics to a lesser extent compared to the LA, mainly because of its muscle-sparing nature that allowed for the preservation of the hip musculature, thus leading to a lower incidence of Trendelenburg gait. As per the rate of heterotopic ossification and reinterventions, no significant differences were observed between the two groups [31].
6 Direct Anterior Approach Comparison to Conventional THA Approaches Using Radiological Analysis
End-stage hip osteoarthritis (OA) is treated through THA, which is considered the most efficacious treatment and can be performed via a variety of approaches. The approach selected to perform the surgery dictates which tissues will be sectioned to reach the joint, the structures that should be avoided, and the difficulties that the surgeon will face when attempting to correctly position the implant [55, 56].
During the DAA procedure, the sartorius, rectus femoris, and iliopsoas are held in position through the use of retractors, while the tensor fasciae latae is mobilized on the opposite side, allowing for optimal exposure of the acetabulum following incision. During the PA, the gluteus maximus is split and the external rotators are detached to ultimately access the acetabulum. When performing THA through LA, the pelvis of the patient is elevated in correspondence to the anterior superior iliac spine to generate enough surface to displace the femur during exposure of the acetabular cavity [55, 63].
Among the conventional approaches previously listed, the DAA is achieving popularity [57], and its recognition is attributable to the conjecture regarding prosthesis stability and satisfaction of patients—ranging from 89% to 95%—alongside a more rapid rehabilitation period and reduced pain following surgery [58,59,60, 63].
The achievement of appropriate positioning of the femoral stem and acetabular cup—which will substantially minimize their component’s wear—constitutes one of the major challenges during the THA procedure, as positive clinical outcomes can be achieved by the positioning of the rotation center of the hip at an inclination of 40° and an anteversion of 20° [61, 62]. Correct positioning of the prosthesis could be potentially accomplished via the use of robotic-assisted surgery or intraoperative fluoroscopy [54].
7 Variation in Short-Term Outcomes Based on the Surgical THA Approach
Surgical variations of THA are performed to improve the functionality and reduce the pain experienced by the patients in the early postoperative period [65].
The primary goal of the systematic review performed in [64] was to compare the short-term outcomes following the most frequently used THA approaches, namely, the DAA, PA, DL, and AL, up to the 12th week following the surgical procedure, further considering the minimally important clinical difference (MCID) to ultimately establish whether the observed differences were clinically important, set to 1.9 for the visual analogue scale (VAS) [66]—characterized by a score ranging from 0 to 10—and to 7–10 for the Harris Hip Score (HHS) [67], which ranged from 0 to 100. The postoperative data used for the comparison included the functionality of the patients at 6 and 12 weeks and the pain scores—calculated using the VAS and the HHS—gathered at day 1 (POD 1) and 2 (POD 2), as well as 2 and 6 weeks. The overall consumption of opioids after the procedure was also registered when available. Moreover, the incidence of complications postoperatively was recorded, including data regarding reinterventions, the occurrence of fractures during the procedure, aggravation of the wound, deep infections, as well as dislocations [64].
The DAA showed superior outcomes calculated via the HHS during the follow-up performed at the sixth week postoperatively compared to the DL and the PA; nonetheless, the results didn’t reach the set range of 7–10 identified to achieve the MCID. Moreover, no statistical difference was indicated in the HSS during the follow-up procedure at the 12th week following the various surgical approaches when compared to the DAA [64].
In terms of postoperative pain, the VAS scores recorded on day 2 and after 2 weeks showed inferior results in the DAA cohort compared to DL; however, the calculated differences—corresponding to 0.9 and 1.3—didn’t reach the set value of 1.7 established to achieve the MCID. The data recorded for length of hospitalization indicated a shorter timeframe following the AL approach when compared to the DAA, whereas no significant differences were observed when comparing the DL and PA to the DAA [64].
Five studies reported the overall opioid consumption of the patients, while one examined the same parameters during the follow-up performed at the second week. No significant differences were observed in the study performed by Barrel et al. [74] regarding the opioid consumption in the DAA and PA groups on the first and second day following the procedure; instead, the study performed by Taunton [68] documented a higher consumption in the PA when compared to the DAA. Similarly, a higher opioid consumption during the second-week follow-up was observed by Cheng et al. [69] in the PA compared to DAA. Lower overall consumption of opioids was additionally demonstrated in the study performed by Brismar [70] and Nistor [71] for the DAA when compared to the DL. Furthermore, Mjaaland [72] observed a lower consumption on the day of the surgical procedure for the DAA compared to the DL; however, no other differences were recorded in the data gathered for the corresponding analyses. Finally, no differences were observed in the overall postoperative consumption when comparing the AL and the DL approaches in the study performed by Martin et al. [73]. The complication rates were recorded in 19 out of the 25 analyzed studies, reporting a total of 20 reinterventions, 21 aggravations at the wound site, 24 fractures occurred during the procedure, 12 dislocations, and 8 deep infections, ultimately indicating no significant differences between the various analyzed approaches.
In summary, the analyzed data indicated no relevant differences in the early period following THA among the various approaches, as well as differences in the complication rates and pain scores; however, the data recorded for opioid consumption indicated a lower trend following the DAA [64].
8 SuperPATH
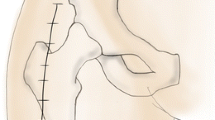
The supercapsular percutaneously assisted approach (SuperPATH) is a modification of the anterior and posterior approaches [75]. This minimally invasive procedure has been initially outlined by Stephen Murphy in 2004 and has been correlated with a variety of advantages compared to other conventional approaches. In fact, the SuperPATH approach utilizes a reduced superficial incision and doesn’t foresee the dislocation of the femoral head, thus preserving the muscles and tendons, as well as the capsule, and only applying a minimal amount of stretch to the aforementioned structures. This approach only involves the release of the piriformis tendon, which is—unlike what experienced for the anterior approach, during which the piriformis tendon inevitably retracts posteriorly—then repaired in its natural position, a technique that ulteriorly decreases the dislocation rates to a 0.2—0.3% range. Figure 1a depicts the approach to the hip capsule. The incision is started at the extremity of the greater trochanter and continued proximally. The subcutaneous fat is then incised and electrocauterized, to prevent excessive bleeding, followed by incision of the gluteus maximus. The bursa of the posterior segment of the gluteus medius is incised, and the latter is then retracted anteriorly to allow the visualization of the piriformis tendon, followed by additional incision and retraction of the gluteus minimus anteriorly. Exposure of the capsule is achieved via the use of several retractors. Figure 1b shows the preparation of the capsule, which is incised in line with the superficial incision [143].
8.1 Short-Term Effect Comparison of Direct Anterior Approach and SuperPATH in THA
Among the various CAs, two have been outlined as minimally invasive, DAA and SuperPATH. The increasing demand regarding the performance of minimally invasive procedures originated from the dissatisfaction of previously operated patients with the cosmetic appearance of the site where surgery was executed, alongside the goal of a prompter rehabilitation process and decreased costs [77]. The results gathered in [76] have indicated the superiority in outcomes of THA SuperPATH compared to DAA, as it exhibited better results in terms of decreased operation time, length of incision, intraoperative loss of blood, and severity of pain in the initial stage following the procedure. The DAA was characterized, in fact, by a 12.8-min longer operation time compared to SuperPATH constituting a significant advantage for the latter as the prolonged time of surgery is correlated to a more elevated rate of superficial infection—augmenting by about 6% for every 10-min increase in operational time [78]—as well as perioperative complications including higher readmission rates, wound dehiscence, and kidney problems [79]. Moreover, the incision performed during the direct anterior approach was approximately 4.3 cm longer, and the intraoperative blood loss recorded was 59 ml higher than the one observed for SuperPATH, attributed to bleedings of branches of the lateral circumflex femoral artery, exposed during the DAA procedure [76]. Moreover, the DAA registered a 0.8 points higher mean pain VAS 1 day following surgery, which might be attributable to the innervation of the area subjected to the procedure, as THA through DAA is performed in an area highly innervated by branches of the cutaneous lateral femoral nerve, femoral nerve, and obturator nerve, while only branches from Th12 and iliohypogastric nerves are exposed while performing THA through SuperPATH. The mean HHS recorded 3 months postoperatively ranged from 85.9 to 94.6 points for the DAA, and from 72.3 to 89.6 for SuperPATH; however, no differences in HHS were observed 3, 6, and 12 months after surgery. In conclusion, the comparison of data indicated superior short-term results for THA performed through SuperPATH, but both approaches resulted equivalent in acetabular cup position and functional outcome of the surgical procedure [76].
9 Simultaneous Bilateral THA Outcomes’ Performance: A Single Surgeon Performance
Patients presenting bilateral arthritis of the hip frequently undergo THA, either in one or two stages. Previous research has shown that these two procedures shared similar outcomes in terms of complications, both prior and following the surgery, and revision rates; however, the transfusion rates were significantly higher in one-stage procedures, whereas the length of hospital stay was longer, and the intraoperative blood loss and cost of the surgery were considerably higher for bilateral surgeries performed in two stages [81,82,83,84,85]. The previously mentioned data were obtained from studies characterized by several limitations, including a small number of participants, absence of reported post-discharge results, and integration of statistics obtained by different surgeons; therefore, the review conducted in [80] aimed at analyzing the reported outcomes of simultaneous bilateral THA performed via direct anterior approach by a single surgeon (WJH).
The patients were divided into two groups, one subjected to simultaneous bilateral THA via DAA and a second one consisting of participants undergoing staged bilateral THA through the same approach, with a mean time between the procedures of 31.5 months. The same technique was used throughout all the procedures, involving the insertion of a cementless tapered femoral stem without the use of fluoroscopy, and patients were administered with cefazolin—or an analogous antibiotic if the patient presented severe allergies—intravenously, to prevent the spread of bacteria, and with aspirin to preclude the risk of deep vein thrombosis.
Results showed a significantly shorter mean value for length of stay for the group undergoing simultaneous bilateral THA (1.8 days) compared to the one subjected to the staged bilateral procedure (2.8 days); however, the rate of transfusion of packed red blood cells amounted to 3.5% for the simultaneous bilateral cohort. Moreover, the simultaneous bilateral group displayed a percentage of 0.39% for infections at the site of surgery or following implantation of the prosthesis and formation of hematoma, and 0.77% for periprosthetic fractures, subsequent surgery, and readmissions. The negligible complication rates encountered throughout the previously analyzed study highlight the safety of the simultaneous bilateral procedure for younger patients with suitable indications, therefore presenting a lower body mass index and fewer health conditions [80].
10 Conventional and Robotic-Assisted THA Outcomes’ Follow-Up Comparisons
Despite the growing success of THA, multiple complications—including aseptic loosening or malpositioning of the prosthetic component—keep on arising [87, 88]. To avoid incurring in any complications, the demand for robotic-assisted THA has been concomitantly increasing. Two robotic systems have received approval by the FDA (Food and Drug Administration) for the performance of THA: the ROBODOC, which assists specifically with installation of the acetabular component following the input of patient’s information obtained through a computed tomographic scan—to generate a three-dimensional virtual design of the anatomy of the latter—as well as preparation of the femoral canal, and the Mako, which uses computed tomographic (CT)-guided navigation to develop an initial plan regarding the performance of the surgical procedure, to subsequently aid in the preparation of the acetabulum and the positioning of the cup, alongside osteotomy of the femoral head and the replication of the offset and length of the leg [86, 97]. Figure 2 shows the THA procedure performed with the Mako platform, which is one of the two robotic systems approved by the FDA [144].
Robotic THA has exhibited superior results in terms of accuracy in the placement of the implant [89, 90]; however, the related costs considerably increase compared to the one indicated for the conventional procedure. Additionally, more accurate positioning of the implant is not always an indicator of enhanced patient-reported outcome measures (PROMs). Thus, the goal of the study performed in [86] was to establish whether robotic-assisted THA generated enhanced patient-reported outcomes and decreased the incidence of dislocation and complications compared to manually conducted THA.
Analysis of seven articles including a total of 658 patients with 335 of whom underwent robotic THA while the remaining 323 were subjected to the manual surgical procedure. Thirteen different PROMs were recorded in the seven articles, mainly including the HHS [91,92,93,94, 95, 96] and the WOMAC scores [91, 92, 96]. Four of the seven studies didn’t show any substantial differences between the manually performed and the robotic-assisted procedures, whereas three reported enhanced PROMs in the robotic THA group during one of the evaluations performed postoperatively [91, 93, 96]. Robotic-assisted THA resulted in more accurate positioning of the implant; however, despite this favorable report, a greater incidence of dislocations was found in the robotic-assisted group compared to the one undergoing manual THA [93,94,95], thus indicating that implant stability depends on a variety of factors aside from components’ positioning. Moreover, data regarding operative times for robotic THA were considerably higher—107.1 ± 29.1 min—compared to the manually performed procedure, 82.4 ± 23.4 min [93].
In summary, robotic THA is currently evolving, but, according to available data extrapolated from other articles, the PROMs obtained for robotic THA are analogous to the ones obtained for manual THA, further highlighting the greater efficiency of robotic THA in terms of implant positioning which, however, does not seem to be an indicator of fewer incidence of complications or revisions [86].
11 Capsular Repair and Capsulectomy
The THA procedure could be performed via capsulectomy, consisting in the excision of the joint capsule, and then replacing with a pseudocapsule with no active neurophysiological roles, as no nerve endings are present in the recently operated area [99,100,101,102,103,104,105,106,107,108, 117], or reparation, in the case of capsulotomy, which could potentially result in increased postoperative pain and decreased range of motion (ROM) due to the reconstruction of the capsule over the previously installed prosthetic component [101, 103,104,105, 107,108,109,110,111,112,113,114, 117].
11.1 Comparison of Capsular Repair and Capsulectomy in THA
Dislocation constitutes one of the main complications following THA [115, 116]; however, the right approach to use to prevent such complications is still uncertain. Therefore, the systematic review conducted in [98] evaluated the various outcomes correlated to capsulectomy and capsular repair, especially regarding the incidence of dislocation, length of the procedure, and blood loss, and the results were further evaluated based on the approach used during the surgical procedure.
A total of 31 articles were included in the study, comprising 17,272 patients and 17,481 hips. The mean age of the patients at the time of the procedure was 62.6 years, and the mean follow-up period was 37.7 months. All the patients included in the study were subjected to THA, and 209 out of the 17,272 underwent bilateral THA, which consisted of the same procedure performed via the same approach on both sides. Capsular repair was performed in 7928 instances (45.4%), whereas capsulectomy took place in the remaining 9553 cases (54.5%).
The difference in blood loss was statistically significant, and the cohort subjected to capsular repair displayed a lower value, with an average of 465.2 ml, compared to the value indicated in the capsulectomy procedure, which corresponded to 709.2 ml. However, the procedure was substantially longer in the capsular repair group, with an average time of 102.5 min, compared to the capsulectomy cohort that resulted in operational time averaged to 96.08 min. Moreover, 345 dislocations were reported in the 17,481 THA surgeries analyzed, thus indicating a dislocation incidence of 1.97%. In particular, the rate of dislocation observed after the capsulectomy procedure corresponded to 3.06%, whereas the one identified for the capsular repair was significantly lower, corresponding to 0.65%.
The THA surgery was performed via the anterior approach in 2142 hips, 1718 of which were then subjected to capsular repair, whereas 424 underwent capsulectomy. The overall incidence of dislocation observed after the anterior approach was 1.3% (28 instances). More specifically, a dislocation rate of 3.7% (16/424) was observed in the capsulectomy group, while a significantly lower rate was determined for the capsular repair group, corresponding to 0.69% (12/1718).
The lateral approach was utilized on 6189 hips, 2308 of which underwent capsular repair, and 3881 of which were subjected to capsulectomy. The overall incidence of dislocation recorded for the THA procedure performed via the lateral approach was 2.86% (166 cases). In particular, the dislocation rate observed in the capsulectomy cohort was 3.89% (151/3381), and, similarly to the results observed for the anterior approach, the dislocation rate recorded for the patients subjected to capsular repair was substantially lower, corresponding to 0.64%.
The posterior approach was performed on 9150 hips, with a comprehensive incidence of dislocation of 1.65% (151 instances). 3902 hips were subjected to capsular repair through the same approach, whereas 5248 underwent capsulectomy. Again, the rate of dislocation was significantly higher in the capsulectomy cohort, with a percentage of 2.4% (126/5248), compared to the 0.64% (25/3902) found in the capsulotomy group.
In summary, the capsular repair procedure showed a significantly inferior amount of blood loss during the surgical procedure; however, it was associated with a longer operational time that could potentially constitute a disadvantage when treating elderly patients with comorbidities, as every additional minute spent in the operating room substantially increases the risk of adverse events. Capsular repair was also associated with inferior dislocation rates for all the analyzed approaches, ranging from 0.64% to 0.69%, compared to the much higher percentages observed for capsulectomies, corresponding to 3.7% after the procedure performed via the anterior approach, 3.89% after the lateral approach, and 2.4% after the posterior approach [98].
12 Fractures
The optimal treatment of extracapsular fractures to the proximal femur is an ongoing effort; however, the use of proximal femoral nail antirotation (PFNA) might constitute a viable option for the management of such fractures because of the relatively low incidence of complications compared to other techniques [118]. The PFNA device consists of a small intramedullary nail characterized by a helical blade design, which results extremely advantageous for the direct fixation of the head of the femur and the compaction of the trabecular bone, ultimately speeding up the fracture healing process [119].
Femoral neck fractures are usually treated with internal fixation in young, more active patients. In contrast, this procedure is not indicated for older patients mainly because of their slow fracture healing process requiring a longer hospital stay, which could potentially lead to ulterior complications [122, 123].
In the elderly, the more suitable procedure for the treatment of femoral fractures is hemiarthroplasty (HA), which foresees the replacement of only half of the impaired hip joint [120]. Figure 3a depicts the prosthesis used for the HA procedure, whereas Fig. 3b shows the implant used for THA surgery [145].
This procedure has several advantages compared to the THA surgery, including inferior length of surgery and decreased intraoperative blood loss [121]. During the HA procedure, the damaged femoral head is replaced with a prosthetic component that increases the stability of the femur and simultaneously restores the functionality of the hip [4]. The preparation of the femur in HA proceeds similarly to the THA surgery; in fact, the femoral canal is hollowed out prior to the insertion of the metal stem; however, in contrast to THA (in which the entire joint is replaced), only the femoral head is substituted. HA has been correlated to a variety of complications, which are analogous to the THA procedure. Such complications include infection, formation of blood clots, dislocation, and loosening of the femoral stem.
What follows would be additional information on the comparison of hip replacement and PFNA for the treatment of intertrochanteric fractures, and the attempts for optimal procedure search for the treatment of femoral neck fractures in the elderly.
12.1 Hip Replacement and Proximal Femoral Nail Antirotation Procedures’ Outcome Comparisons for Elderly with Intertrochanteric Fractures
Intertrochanteric fractures are particularly common among older people (95%), mainly because of the higher incidence of osteoporosis, which increases the likelihood of incurring fractures following minor traumatic events [123, 124]. The conservative treatment of the latter requires patients to remain in bed for a prolonged period of time, thus increasing the risk of experiencing serious complications, such as pneumonia, deep vein thrombosis, and infections of the urinary tract. Therefore, intertrochanteric fractures are usually treated with surgery [125, 126]—which accelerates functional recovery and increases the life quality of the patients [127]—in particular hip replacement (HR) and PFNA. Nonetheless, the choice regarding the appropriate approach to use is still being debated [128,129,130].
The technology involved in the performance of HR is particularly advanced, especially regarding prosthesis stability, which allows for a faster postoperative motion recovery [131, 132]. However, the procedure is significantly longer and characterized by a higher blood loss rate and a more extensive surgical incision, factors that might increase the risk of incidence of comorbidities in the elderly [133]. An alternative approach, the proximal femoral nail antirotation, was delineated based upon prior ameliorations of technologies relative to internal fixation. It presents various benefits compared to HR, including a shorter operation time, potential preservation of the head and neck of the femur, as well as good fixation effect favoring a better healing process [134]. However, internal fixations are frequently correlated to a greater risk of complications, including metal malfunction and puncture of the femoral head, alongside higher reported fatality rates (amounting to approximately 21.4%), presumably related to the longer time spent in bed after surgery, and deferral of activities involving weight load [135]. Based on these premises, HR exhibits more benefits relative to the treatment of intertrochanteric fractures in the elderly. In general, the choice of appropriate treatment for intertrochanteric fractures in the elderly should be based on a careful analysis of the patients’ clinical characteristics, alongside a perioperative process of muscle strengthening to ultimately reduce intraoperative hemorrhage and avoid the incidence of ulterior complications [122].
12.2 Hemiarthroplasty and THA Procedure Comparisons for Femoral Neck Fracture Treatment
Femoral neck fractures (FNF) comprise fractures of the head of the femur up until the base of the femoral neck occurring as a result of exposure to torsion, which ultimately threatens the blood supply and limits the bone healing process [136, 137]. Such fractures are often categorized using Garden’s classification, ranging from types I and II—which include stable fractures with no displacement or lesser degree of displacement—to types III and IV, which integrate unstable fractures resulting from the shifting of the fracture end, thus causing more severe damages [137,138,139]. Figure 4 illustrates the four different stages of Garden’s classification, which is employed to categorize FNFs based on the degree of displacement [145].
In the elderly, femoral neck fractures are commonly listed as type III or IV in Garden’s classification and present several downsides, such as venous thromboembolism and falling pneumonia. These fractures could be treated via either THA, which foresees the replacement of both the femoral head and the acetabulum of the hip and provides, thus, better functional results, or hemiarthroplasty, which only replaces the head of the femur, therefore presenting several benefits including decreased procedural trauma and blood loss, alongside complications such as the increased risk of elevated pain following the surgery and wear of the cartilage of the acetabulum [136]. As a result, the appropriate surgical technique to use for the treatment of FNFs is still disputed [140, 141].
As mentioned above, hemiarthroplasty presents many benefits compared to THA, including reduced trauma, shorter procedure, and inferior intraoperative loss of blood; however, it is associated with a longer hospital stay and increased risk of revision surgery, as the acetabulum is not replaced during the procedure, thus potentially leading to a higher incidence of prosthesis dislocation, deterioration of the cartilage surrounding the acetabulum, and infections derived from the sterility of the prosthesis. Moreover, hemiarthroplasty presents a significantly higher risk of contracting pneumonia and incurring in renal failure, whereas no differences with THA were identified with regard to the incidence of other complications such as myocardial infarct, venous thromboembolism, and infection [136].
13 Hemiarthroplasty and THA Procedural Differences of Elderly Orthogeriatric Patients
In order to establish the most suitable method for the treatment of femoral neck fractures (FNF), surgeons must carefully analyze a wide range of factors, including the individual necessities of the patients, as well as the presence of comorbidities and ambulatory capacity, to ultimately determine whether the patients should be subjected to hemiarthroplasty or THA. Thus, the study performed in [142] aimed to identify the differences in the results obtained after THA or hemiarthroplasty for the treatment of FNFs in the elderly, solely subjected to orthogeriatric co-management.
The 5554 patients elected for the study were further divided into two groups, one comprising 4662 patients undergoing hemiarthroplasty, with an average age of 85, and a second one consisting of 892 patients treated with THA, with a mean age of 79. However, some of the patients (54.8%) were excluded from some of the performed analyses because of lack of information; therefore, each examination ultimately displayed the overall number of included patients. The main observed parameters included ambulatory ability 120 days after the fracture, complications associated with surgery, as well as fatalities recorded during hospitalization or within the first 120 days following the procedure, and the quality of life calculated 7 and 120 days after the surgery, measured using the EQ-5D-3L questionnaire. Moreover, some independent variables that could have potentially impacted the results of the procedures were included, namely, the American Society of Anesthesiologists (ASA) classification, with a grade ranging from 1 to 5, the Identification of Seniors At Risk (ISAR) score, as well as other factors such as sex, age, length of hospitalization, presence of additional injuries, and anticoagulation.
The patients included in the hemiarthroplasty group observed to be significantly less healthy compared to the THA cohort, presenting an ASA grade of 3 or higher in 80% of the cases, whereas the same score was indicated in approximately 58% of the cases in the THA group. Likewise, 85% of the patients undergoing hemiarthroplasty received an ISAR score of 2 or higher at the time of hospital admission, whereas a lower percentage of the patients undergoing THA, corresponding to 58%, obtained analogous scores. The incidence of fatalities following the surgical procedure was greater in the hemiarthroplasty cohort (6%) than in the THA group (3%); moreover, the ambulatory capability was superior in the patients belonging to the THA group (28%) at the 120-day postoperative follow-up, determined after a careful examination of the EQ-5D-3L questionnaire. Nonetheless, the incidence of complications correlated to the surgical procedure was inferior following hemiarthroplasty (4%) compared to the 8% indicated after THA, which is further observed to increase to 10% postoperatively during 120-day follow-ups. Instead, the rate of readmissions was determined to be statistically insignificant, with a rate of 5% for the hemiarthroplasty cohort and 7% for the THA one; similarly, no substantial differences were found regarding the ambulatory ability of patients 120 days after the two surgical procedures and before the injury. Finally, the quality of life at 120 days following the surgery was substantially higher in the THA group (0.9) compared to the other analyzed cohort (0.81).
In conclusion, THA is more indicated for patients presentin;g a superior health status and requiring greater mobility, supported by enhanced ambulatory capacity and quality of life achieved after the surgery. Instead, hemiarthroplasty is advised for patients presenting multimorbidity to preclude the necessity of further procedures and the incidence of ulterior complications [142].
References
Post ZD, et al. Direct anterior approach for total hip arthroplasty indications, technique, and results. J Am Acad Orthop Surg. 2014;22:595–603. https://journals.lww.com/jaaos/fulltext/2014/09000/direct_anterior_approach_for_total_ip.7.aspx
Maldonado DR, et al. Direct anterior approach versus posterior approach in primary total hip replacement: comparison of minimum 2-year outcomes. HIP Int. 2019;31(2):166–73. https://doi.org/10.1177/1120700019881937.
Taunton MJ, Trousdale RT, Sierra RJ, et al. John Charnley award: randomized clinical trial of direct anterior and miniposterior approach THA: which provides better functional recovery? Clin Orthop Relat Res. 2018;476:216–29.
Petis S, Howard JL, Lanting BL, et al. Surgical approach in primary total hip arthroplasty: anatomy, technique and clinical outcomes. Can J Surg. 2015;58:128–39.
Lazaru P, et al. Direct anterior approach (DAA) vs. conventional approaches in total hip arthroplasty: a RCT meta-analysis with an overview of related meta-analyses. PLoS One. 2021;16(8):e0255888. https://doi.org/10.1371/journal.pone.0255888.
Sculco TP, Jordan LC, Walter WL. Minimally invasive total hip arthroplasty: the hospital for special surgery experience. Orthop Clin North Am. 2004;35:137–42. https://doi.org/10.1016/S0030-5898(03)00116-0. PMID: 15062699
Szendroi M, Sztrinkai G, Vass R, Kiss J. The impact of minimally invasive total hip arthroplasty on the standard procedure. Int Orthop. 2006;30:160–71. https://doi.org/10.1007/s00264-005-0049-8. PMID: 16552579
Wall SJ, Mears SC. Analysis of published evidence on minimally invasive total hip arthroplasty. J Arthroplast. 2008;23:55–8. https://doi.org/10.1016/j.arth.2008.06.010. PMID: 18922374
Moreau P. Minimally invasive total hip arthroplasty using hueter’s direct anterior approach. Eur J Orthop Surg Traumatol. 2018;28(5):771–9. https://doi.org/10.1007/s00590-018-2158-2. PMID: 29511824
Kayani B, Konan S, Chandramohan R, Haddad FS. The direct superior approach in total hip arthroplasty. Br J Hosp Med (Lond). 2019;80(6):320–4. https://doi.org/10.12968/hmed.2019.80.6.320. PMID: 31180766
Galakatos GR. Direct anterior total hip arthroplasty. Mo Med. 2018;115(6):537–41. PMID: 30643349.
Sculco TP, Boettner F. Minimally invasive total hip arthroplasty: the posterior approach. Instr Course Lect. 2006;55:205–14. PMID: 16958456
Basad E, Ishaque B, Stu ̈rz H, Jerosch J. The anterolateral minimally invasive approach for total hip arthroplasty: technique, pitfalls, and way out. Orthop Clin North Am. 2009;40(4):473–viii. https://doi.org/10.1016/j.ocl.2009.05.001. PMID: 19773052
Swanson TV. Posterior single-incision approach to minimally invasive total hip arthroplasty. Int Orthop. 2007;31(Suppl 1):S1–5. https://doi.org/10.1007/s00264-007-0436-4. PMID: 17653544
Ilchmann T. Approaches for primary total hip replacement. Hip Int. 2014;24(Suppl 10):S2–6. https://doi.org/10.5301/hipint.5000163. PMID: 24970034
Wojciechowski P, Kusz D, Kopeć K, Borowski M. Minimally invasive approaches in total hip replacement. Chir Narzadow Ruchu Ortop Pol. 2008;73(3):207–176. PMID: 18847028
Capuano N, Del Buono A, Maffulli N. Tissue preserving total hip arthroplasty using superior capsulotomy. Oper Orthop Traumatol. 2015;27(4):334–41. https://doi.org/10.1007/s00064-013-0242-7. PMID: 25900826
Migliorini F, Biagini M, Rath B, Meisen N, Tingart M, Eschweiler J. Total hip arthroplasty: minimally invasive surgery or not? Meta-analysis of clinical trials. Int Orthop. 2019;43(7):1573–82. https://doi.org/10.1007/s00264-018-4124-3. PMID: 30171273
Parratte S, Pagnano MW. Muscle damage during minimally invasive total hip arthroplasty: cadaver-based evidence that it is significant. Instr Course Lect. 2008;57:231. PMID: 18399584
Meneghini RM, Pagnano MW, Trousdale RT, et al. Muscle damage during MIS total hip arthroplasty. Clin Orthop Relat Res. 2006;453:293. https://doi.org/10.1097/01.blo.0000238859.46615.34. PMID: 17006366
Kennon RE, Keggi JM, Wetmore RS, et al. Total hip arthroplasty through a minimally invasive anterior surgical approach. J Bone Joint Surg. 2003;85:39. https://doi.org/10.2106/00004623-200300004-00005. PMID: 14652392
Tan SC, Teeter MG, Del BC, et al. Effect of taper design on Trunnionosis in metal on polyethylene total hip arthroplasty. J Arthroplast. 2015;30:1269–72. https://doi.org/10.1016/j.arth.2015.02.031. PMID: 25773576
Sun X, et al. Direct anterior approach versus posterolateral approach in total hip arthroplasty: a meta-analysis of results on early post-operative period. J Orthop Surg Res. 2021;16(1):1–8. https://doi.org/10.1186/s13018-021-02218-7.
Berend KR, Lombardi AV Jr, Seng BE, et al. Enhanced early outcomes with the anterior supine intermuscular approach in primary total hip arthroplasty. J Bone Joint Surg Am. 2009;91(Suppl. 6):107–20.
Sheth D, Cafri G, Inacio MC, et al. Anterior and anterolateral approaches for tha are associated with lower dislocation risk without higher revision risk. Clin Orthop Relat Res. 2015;473:3401–8.
Rodriguez JA, Deshmukh AJ, Rathod PA, et al. Does the direct anterior approach in THA offer faster rehabilitation and comparable safety to the posterior approach? Clin Orthop Relat Res. 2014;472:455–63.
Taunton MJ, Mason JB, Odum SM, et al. Direct anterior total hip arthroplasty yields more rapid voluntary cessation of all walking aids: a prospective, randomized clinical trial. J Arthroplast. 2014;29(Suppl):169–72.
de Steiger RN, Lorimer M, Solomon M. What is the learning curve for the anterior approach for total hip arthroplasty? Clin Orthop Relat Res. 2015;473(12):3860.
Seng BE, Berend KR, Ajluni AF, et al. Anterior-supine minimally invasive total hip arthroplasty: defining the learning curve. Orthop Clin North Am. 2009;40:343.
Jewett BA, Collis DK. High complication rate with anterior total hip arthroplasties on a fracture table. Clin Orthop Relat Res. 2011;469:503.
Huang X-t, et al. Comparisons between direct anterior approach and lateral approach for primary total hip arthroplasty in postoperative orthopaedic complications: a systematic review and meta-analysis. Orthop Surg. 2021;13(6):1707–20. https://doi.org/10.1111/os.13101.
Petis S, Howard JL, Lanting BL, Vasarhelyi EM. Surgical approach in primary total hip arthroplasty: anatomy, technique and clinical outcomes. Can J Surg. 2015;58:128–39.
Hardinge K. The direct lateral approach to the hip. J Bone Joint Surg Br. 1982;64:17–9.
Kwon MS, Kuskowski M, Mulhall KJ, Macaulay W, Brown TE, Saleh KJ. Does surgical approach affect total hip arthroplasty dislocation rates. Clin Orthop Relat Res. 2006;447:34–8.
Connolly KP, Kamath AF. Direct anterior total hip arthroplasty: literature review of variations in surgical technique. World J Orthop. 2016;7:38–43.
Meermans G, Konan S, Das R, Volpin A, Haddad FS. The direct anterior approach in total hip arthroplasty: a systematic review of the literature. Bone Joint J. 2017;99:732–40.
Chen AF, Chen CL, Low S, et al. Higher acetabular anteversion in direct anterior total hip arthroplasty: a retrospective case-control study. HSS J. 2016;12:240–4.
Pogliacomi F, De Filippo M, Paraskevopoulos A, Alesci M, Marenghi P, Ceccarelli F. Mini-incision direct lateral approach versus anterior mini invasive approach in total hip replacement: results 1 year after surgery. Acta Biomed. 2012;83:114–21.
Hürlimann M, Schiapparelli FF, Rotigliano N, Testa E, Amsler F, Hirschmann MT. Influence of surgical approach on heterotopic ossification after total hip arthroplasty—is minimal invasive better? A case control study. BMC Musculoskelet Disord. 2017;18:27.
Mjaaland KE, Kivle K, Svenningsen S, Nordsletten L. Do postoperative results differ in a randomized trial between a direct anterior and a direct lateral approach in THA. Clin Orthop Relat Res. 2019;477:145–55.
Aggarwal VK, Elbuluk A, Dundon J, et al. Surgical approach significantly affects the complication rates associated with total hip arthroplasty. Bone Joint J. 2019;101-B:646–51.
Hart A, Wyles CC, Abdel MP, Perry KI, Pagnano MW, Taunton MJ. Thirty-day major and minor complications following total hip arthroplasty-a comparison of the direct anterior, lateral, and posterior approaches. J Arthroplast. 2019;34:2681–5.
Sheth D, Cafri G, Inacio MC, Paxton EW, Namba RS. Anterior and anterolateral approaches for THA are associated with lower dislocation risk without higher revision risk. Clin Orthop Relat Res. 2015;473:3401–8.
Fleischman AN, Tarabichi M, Magner Z, Parvizi J, Rothman RH. Mechanical complications following total hip arthroplasty based on surgical approach: a large, single-institution cohort study. J Arthroplast. 2019;34:1255–60.
Zomar BO, Bryant D, Hunter S, Howard JL, Vasarhelyi EM, Lanting BA. A randomised trial comparing spatio-temporal gait parameters after total hip arthroplasty between the direct anterior and direct lateral surgical approaches. Hip Int. 2018;28:478–84.
Restrepo C, Mortazavi SM, Brothers J, Parvizi J, Rothman RH. Hip dislocation: are hip precautions necessary in anterior approaches. Clin Orthop Relat Res. 2011;469:417–22.
Takada R, Jinno T, Miyatake K, et al. Direct anterior versus anterolateral approach in one-stage supine total hip arthroplasty. Focused on nerve injury: a prospective, randomized, controlled trial. J Orthop Sci. 2018;23:783–7.
Mjaaland KE, Kivle K, Svenningsen S, Pripp AH, Nordsletten L. Comparison of markers for muscle damage, inflammation, and pain using minimally invasive direct anterior versus direct lateral approach in total hip arthroplasty: a prospective, randomized, controlled trial. J Orthop Res. 2015;33:1305–10.
Watts CD, Houdek MT, Wagner ER, Sculco PK, Chalmers BP, Taunton MJ. High risk of wound complications following direct anterior total hip arthroplasty in obese patients. J Arthroplast. 2015;30:2296–8.
Christensen CP, Karthikeyan T, Jacobs CA. Greater prevalence of wound complications requiring reoperation with direct anterior approach total hip arthroplasty. J Arthroplast. 2014;29:1839–41.
Purcell RL, Parks NL, Gargiulo JM, Hamilton WG. Severely obese patients have a higher risk of infection after direct anterior approach total hip arthroplasty. J Arthroplast. 2016;31:162–5.
Brun OL, Sund HN, Nordsletten L, Röhrl SM, Mjaaland KE. Component placement in direct lateral vs minimally invasive anterior approach in total hip arthroplasty: radiographic outcomes from a prospective randomized controlled trial. J Arthroplast. 2019;34:1718–22.
Gromov K, Greene ME, Huddleston JI, et al. Acetabular dysplasia and surgical approaches other than direct anterior increases risk for malpositioning of the acetabular component in total hip arthroplasty. J Arthroplast. 2016;31:835–41.
Maciąg B, et al. Systematic review of radiological analysis of total hip replacement via direct anterior approach in comparison to other approaches – study protocol. 2020; https://doi.org/10.21203/rs.3.rs-88637/v1.
Ilchmann T. Approaches for primary total hip replacement. Hip Int. 2014;24:S2–6. [CrossRef]
Graves SC, Dropkin BM, Keeney BJ, Lurie JD, Tomek IM. Does Surgical Approach Affect Patient-reported Function After Primary THA? Clin Orthop Relat Res. 2016;474:971–81. [CrossRef]
Smith-Petersen MN. A new supra-articular subperiosteal approach to the hip joint. J Bone Joint Surg Am. 1917;2:592–5.
Jelsma J, Pijnenburg R, Boons HW, Eggen PJ, Kleijn LL, Lacroix H, Noten HJ. Limited benefits of the direct anterior approach in primary hip arthroplasty: a prospective single centre cohort study. J Orthop. 2017;14:53–8. [CrossRef] [PubMed]
Kyriakopoulos G, Poultsides L, Christofilopoulos P. Total hip arthroplasty through an anterior approach. EFORT Open Rev. 2018;3:574–83. [CrossRef]
Wang Z, Hou J-Z, Wu C-H, Zhou Y-J, Gu X-M, Wang H-H, Feng W, Cheng Y-X, Sheng X, Bao H-W. A systematic review and meta-analysis of direct anterior approach versus posterior approach in total hip arthroplasty. J Orthop Surg Res. 2018 Sep 6;13(1):229. https://doi.org/10.1186/s13018-018-0929-4. PMID: 30189881; PMCID: PMC6127950.
Harrison CL, Thomson AI, Cutts S, Rowe PJ, Riches PE. Research synthesis of recommended acetabular cup orientations for total hip arthroplasty. J Arthroplast. 2014;29:377–82. [CrossRef] [PubMed]
Board T, Bhaskar D, Rajpura A. Current concepts in acetabular positioning in total hip arthroplasty. Indian J Orthop. 2017;51:386–96. [CrossRef] [PubMed]
Moretti VM Zachary DP. Surgical approaches for total hip arthroplasty. Indian J Orthop, Medknow Publications & Media Pvt Ltd. 2017. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5525517/.
Gazendam A, et al. Short-term outcomes vary by surgical approach in total hip arthroplasty: a network meta-analysis. Arch Orthop Trauma Surg. 2021; https://doi.org/10.1007/s00402-021-04131-4.
Trousdale WH, Taunton MJ, Mabry TM, et al. Patient slasty. J Arthroplast. 2017;32:1164–70. https://doi.org/10.1016/j.arth.2016.10.006.
Danoff JR, Goel R, Sutton R, et al. How much pain is significant? Defining the minimal clinically important difference for the visual analog scale for pain after total joint arthroplasty. J Arthroplast. 2018;33:S71–S75 e2. https://doi.org/10.1016/j.arth.2018.02.029.
Achten J, Parsons NR, Edlin RP, et al. A randomised controlled trial of total hip arthroplasty versus resurfacing arthroplasty in the treatment of young patients with arthritis of the hip joint. BMC Musculoskelet Disord. 2010;11:8. https://doi.org/10.1186/1471-2474-11-8.
Taunton MJ, Trousdale RT, Sierra RJ, et al. John Charnley award: randomized clinical trial of direct anterior and miniposterior approach THA: which provides better functional recovery? Clin Orthop Relat Res. 2018;476:216–29. https://doi.org/10.1007/s11999.0000000000000112.
Cheng TE, Wallis JA, Taylor NF, et al. A prospective randomized clinical trial in total hip arthroplasty—comparing early results between the direct anterior approach and the posterior approach. J Arthroplast. 2017;32:883–90. https://doi.org/10.1016/j.arth.2016.08.027.
Brismar BH, Hallert O, Tedhamre A, Lindgren JU. Early gain in pain reduction and hip function, but more complications following the direct anterior minimally invasive approach for total hip arthroplasty: a randomized trial of 100 patients with 5 years of follow up. Acta Orthop. 2018;89:484–9. https://doi.org/10.1080/17453674.2018.1504505.
Nistor D-V, Caterev S, Bolboacă S-D, et al. Transitioning to the direct anterior approach in total hip arthroplasty. Is it a true muscle sparing approach when performed by a low volume hip replacement surgeon? Int Orthop (SICOT). 2017;41:2245–52. https://doi.org/10.1007/s00264-017-3480-8.
Mjaaland KE, Kivle K, Svenningsen S, Nordsletten L. Do postoperative results differ in a randomized trial between a direct anterior and a direct lateral approach in THA? Clin Orthop Relat Res. 2019;477:145–55. https://doi.org/10.1097/CORR.0000000000000439.
Martin R, Clayson PE, Troussel S, et al. Anterolateral minimally invasive total hip arthroplasty: a prospective randomized controlled study with a follow-up of 1 year. J Arthroplast. 2011;26:1362–72. https://doi.org/10.1016/j.arth.2010.11.016.
Barrett WP, Turner SE, Leopold JP. Prospective randomized study of direct anterior vs postero-lateral approach for total hip arthroplasty. J Arthroplast. 2013;28:1634–8. https://doi.org/10.1016/j.arth.2013.01.034.
Ortho, ATX, ATX Ortho. SuperPATH or superior approach to the hip in total hip replacement. ATX Orthop. 2021; https://www.atxortho.com/superior/approach-to-the-hip-in-total-hip-replacement/
Ramadanov N, et al. Comparison of short-term outcomes between direct anterior approach (DAA) and Superpath in total hip replacement: a systematic review and network meta-analysis of randomized controlled trials. J Orthop Surg Res. 2021;16(1) https://doi.org/10.1186/s13018-021-02315-7.
Sculco TP, Jordan LC, Walter WL. Minimally invasive total hip arthroplasty: the hospital for special surgery experience. Orthop Clin North Am. 2004;35:137–42.
Wills BW, Sheppard ED, Smith WR, Staggers JR, Li P, Shah A, Lee SR, Naranje SM. Impact of operative time on early joint infection and deep vein thrombosis in primary total hip arthroplasty. Orthop Traumatol Surg Res. 2018;104(4):445–8. https://doi.org/10.1016/j.otsr.2018.02.008.
Surace P, Sultan AA, George J, Samuel LT, Khlopas A, Molloy RM, Stearns KL, Mont MA. The association between operative time and short-term complications in total hip arthroplasty: an analysis of 89,802 surgeries. J Arthroplast. 2019;34(3):426–32. https://doi.org/10.1016/j.arth.2018.11.015.
Inoue D, et al. Outcomes of simultaneous bilateral total hip arthroplasty for 256 selected patients in a single surgeon’s practice. Bone Joint J. 2021;103-B(7 Supple B):116–21. https://doi.org/10.1302/0301-620x.103b7.bjj-2020-2292.r1.
Parvizi J, Tarity TD, Sheikh E, Sharkey PF, Hozack WJ, Rothman RH. Bilateral total hip arthroplasty: one-stage versus two-stage procedures. Clin Orthop Relat Res. 2006;453:137–41.
Kim Y-H, Kwon O-R, Kim J-S. Is one-stage bilateral sequential total hip replacement as safe as unilateral total hip replacement? J Bone Joint Surg Br. 2009;91-B(3):316–20.
Stavrakis AI, SooHoo NF, Lieberman JR. Bilateral total hip arthroplasty has similar complication rates to unilateral total hip arthroplasty. J Arthroplast. 2015;30(7):1211–4.
Alfaro-Adrián J, Bayona F, Rech JA, Murray DW. One- or two-stage bilateral total hip replacement. J Arthroplast. 1999;14(4):439–45.
Shao H, Chen C-L, Maltenfort MG, Restrepo C, Rothman RH, Chen AF. Bilateral total hip arthroplasty: 1-stage or 2-stage? A meta-analysis. J Arthroplast. 2017;32(2):689–95.
Sweet MC, et al. Comparison of outcomes after robotic-assisted or conventional total hip arthroplasty at a minimum 2-year follow-up. JBJS Rev. 2021;9(6) https://doi.org/10.2106/jbjs.rvw.20.00144.
Ulrich SD, Seyler TM, Bennett D, Delanois RE, Saleh KJ, Thongtrangan I, Kuskowski M, Cheng EY, Sharkey PF, Parvizi J, Stiehl JB, Mont MA. Total hip arthroplasties: what are the reasons for revision? Int Orthop. 2008;32(5):597–604. Epub 2007 Apr 19
Bozic KJ, Kurtz SM, Lau E, Ong K, Vail TP, Berry DJ. The epidemiology of revision total hip arthroplasty in the United States. J Bone Joint Surg Am. 2009;91(1):128–33.
Wasterlain AS, Buza JA 3rd, Thakkar SC, Schwarzkopf R, Vigdorchik J. Navigation and robotics in total hip arthroplasty. JBJS Rev. 2017;5(3):01874474-201703000-00005.
Chen AF, Kazarian GS, Jessop GW, Makhdom A. Robotic technology in orthopaedic surgery. J Bone Joint Surg Am. 2018;100(22):1984–92.
Bargar WL, Parise CA, Hankins A, Marlen NA, Campanelli V, Netravali NA. Fourteen year follow-up of randomized clinical trials of active robotic-assisted total hip arthroplasty. J Arthroplast. 2018;33(3):810–4. Epub 2017 Oct 6
Lim SJ, Ko KR, Park CW, Moon YW, Park YS. Robot-assisted primary cementless total hip arthroplasty with a short femoral stem: a prospective randomized short-term outcome study. Comput Aided Surg. 2015;20(1):41–6. Epub 2015 Aug 13
Honl M, Dierk O, Gauck C, Carrero V, Lampe F, Dries S, Quante M, Schwieger K, Hille E, Morlock MM. Comparison of robotic-assisted and manual implantation of a primary total hip replacement. A prospective study. J Bone Joint Surg Am. 2003;85(8):1470–8.
Nakamura N, Sugano N, Sakai T, Nakahara I. Does robotic milling for stem implantation in cementless THA result in improved outcomes scores or survivorship compared with hand rasping? Results of a randomized trial at 10 years. Clin Orthop Relat Res. 2018;476(11):2169–73.
Domb BG, Chen JW, Lall AC, Perets I, Maldonado DR. Minimum 5-year outcomes of robotic-assisted primary total hip arthroplasty with a nested comparison against manual primary total hip arthroplasty: a propensity score-matched study. J Am Acad Orthop Surg. 2020;28(20):847–56.
Banchetti R, Dari S, Ricciarini ME, Lup D, Carpinteri F, Catani F, Caldora P. Comparison of conventional versus robotic-assisted total hip arthroplasty using the Mako system: an Italian retrospective study. J Health Soc Sci. 2018;3(1):37–48.
Mart J-PS, et al. Robotics in total hip arthroplasty: a review of the evolution, application and evidence base. EFORT Open Rev. 2020; British editorial society of bone and joint surgery, https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7784137/
Miranda L, et al. Capsular repair vs capsulectomy in total hip arthroplasty. Br Med Bull. 2021;139(1):36–47. https://doi.org/10.1093/bmb/ldab011.
Woolson ST, Rahimtoola ZO. Risk factors for dislocation during the first 3 months after primary total hip replacement. J Arthroplast. 1999;14:662–8.
Borrero S, Kent Kwoh C, Sartorius J, et al. Brief report: gender and total knee/hip arthroplasty utilization rate in the VA system. J Gen Intern Med. 2006;21:S54–7.
Williams D, Petruccelli D, Winemaker M, et al. Total joint replacement clinical outcomes: gender differences. Orthop Proc. 2010;92-B:137.
Kazley JM, Banerjee S, Abousayed MM, et al. Classifications in brief: garden classification of femoral neck fractures. Clin Orthop Relat Res. 2018;476:441–5.
Barıshan FC, Akesen B, Atıcı T, et al. Comparison of hemiarthroplasty and total hip arthroplasty in elderly patients with displaced femoral neck fractures. J Int Med Res. 2018;46:2717–30.
Koo K-H, Song H-R, Ha Y-C, et al. Role of thrombotic and fibrinolytic disorders in the etiology of Perthes’ disease. Clin Orthop Relat Res. 2002;399:162–7.
Smith-Petersen MN. A new supra-articular subperiosteal approach to the hip joint. J Bone Joint Surg. 1917;s2–15:592–5.
Boll KL. Total hip replacement using Müller’s method. Ugeskr Laeger. 1990;152:1987–9.
Müller ME. Total hip prostheses. Clin Orthop Relat Res. 1970;72:46–68.
Charnley J. Arthroplasty of the hip. A new operation. Lancet. 1961;1:1129–32. https://doi.org/10.1016/s0140-6736(61)92063-3. PMID: 15898154
Smith-Petersen MN. Approach to and exposure of the hip joint for mold arthroplasty. J Bone Joint Surg Am. 1949;31A:40–6.
Lindholm RV, Puranen J, Kinnunen P. The Moore vitallium femoral-head prosthesis in fractures of the femoral neck. Acta Orthop Scand. 1976;47:70–8. https://doi.org/10.3109/17453677608998976.
Ludloff K. Zur blutigen Einrenkung der angeborrenen Huftluxation. Zeischr Orthop Chir. 1908;22:272–6.
Camenzind RS, Stoffel K, Lash NJ, et al. Direct anterior approach to the hip joint in the lateral decubitus position for joint replacement. Oper Orthop Traumatol. 2018;30:276–85.
Khan RJK, Fick D, Khoo P, et al. Less invasive total hip arthroplasty. J Arthroplast. 2006;21:1038–46.
Lachiewicz PF, Poon ED. Revision of a total hip arthroplasty with a Harris-Galante porous-coated acetabular component inserted without cement. A follow-up note on the results at five to twelve years. J Bone Joint Surg. 1998;80:980–4.
Tripuraneni KR, Munson NR, Archibeck MJ, et al. Acetabular abduction and dislocations in direct anterior vs posterior total hip arthroplasty: a retrospective, matched cohort study. J Arthroplast. 2016;31:2299–302.
Schneeberger AG, Schulz RF, Ganz R. Blood loss in total hip arthroplasty. Lateral position combined with preservation of the capsule versus supine position combined with capsulectomy. Arch Orthop Trauma Surg. 1998;117:47–9. https://doi.org/10.1007/s004020050189.
Ometti M, et al. Capsulectomy vs capsulotomy in total hip arthroplasty. Clinical outcomes and proprioception evaluation: study protocol for a randomized, controlled, double blinded trial. J Orthop. 2019. Elsevier, https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6818366/
Gardenbroek T, et al. The Proximal Femur Nail antirotation: an identifiable improvement in the treatment of unstable pertrochanteric fractures? J Trauma. 2021. U.S. National Library of Medicine. https://pubmed.ncbi.nlm.nih.gov/21818023/
Twittercreator. Ernst Raaymakers. For trochanteric fracture, pertrochanteric, simple. https://surgeryreference.aofoundation.org/orthopedic-trauma/adult trauma/proximal-femur/trochanteric-fracture-pertrochanteric-simple/nailing.
Roland J. Hemiarthroplasty: procedure, recovery, complications, and more. Healthline. 2018. Healthline Media. https://www.healthline.com/health/hemiarthroplasty#hemiarthroplasty-vs-thr
Hip Hemiarthroplasty. Florida Orthopaedic Institute, https://www.floridaortho.com/specialties/hip-thigh/hip-hemiarthroplasty/.
Junming C. et al, Comparison of clinical outcomes with hip replacement versus PFNA in the treatment of intertrochanteric fractures in the elderly. Medicine. 2021;100(9) https://doi.org/10.1097/md.0000000000024166.
Tucker A, Donnelly KJ, Rowan C, et al. Is the best plate a nail? A review of 3230 unstable intertrochanteric fractures of the proximal femur. J Orthop Trauma. 2018;32:53–60.
Dung TT, Hieu ND, Son LM, et al. Primary cementless bipolar long stem hemiarthroplasty for unstable osteoporotic intertrochanteric fracture in the elderly patients. Open Access Maced J Med Sci. 2019;7:4342–6.
Sik CW, Hoon AJ, Joon-Hyuk K, et al. Cementless bipolar hemi- arthroplasty for unstable intertrochanteric fractures in elderly patients. Clin Orthop Surg. 2010;2:221–6.
Choi HJ, Kim E, Shin YJ, et al. The timing of surgery and mortality in elderly hip fractures: a retrospective, multicenteric cohort study. Indian J Orthop. 2014;48:599–604.
Wu K, Xu Y, Lei Z, et al. Which implant is better for beginners to learn to treat geriatric intertrochanteric femur fractures: a randomised controlled trial of surgeons, metalwork, and patients. J Orthop Translat. 2020;21:18–23.
Park-Wyllie LY, Mamdani MM, Juurlink DN, et al. Bisphosphonate use and risk of subtrochanteric or femoral shaft fractures in older women. JAMA. 2011;305:783–9.
Min BW, Lee KJ, Oh JK, et al. The treatment strategies for failed fixation of intertrochanteric fractures. Injury. 2019;50:1339–46.
Parker MJ, Cawley S. Sliding hip screw versus the Targon PFT nail for trochanteric hip fractures: a randomised trial of 400 patients. Bone Joint J. 2017;99-B:1210–5.
Lee YK, Ha YC, Chang BK, et al. Cementless bipolar hemiarthroplasty using a hydroxyapatite-coated long stem for osteoorotic unstable interochanteric fractures. Arthrpolasty. 2011;26:626–32.
Chai W, Kong X, Yang M, et al. Robot-assisted total hip arthroplasty for arthrodesed hips. Ther Clin Risk Manag. 2020;16:357–68.
Luo X, He S, Zeng D, et al. Proximal femoral nail antirotation versus hemiarthroplasty in the treatment of senile intertrochanteric fractures: case report. Int J Surg Case Rep. 2017;38:37–42.
Kulachote N, Sa-Ngasoongsong P, Sirisreetreerux N, et al. Predicting factors for return to prefracture ambulatory level in high surgical risk elderly patients sustained intertrochanteric fracture and treated with proximal femoral nail anti-rotation (PFNA) with and without cement augmentation. Geriatr Orthop Surg Rehabil. 2020;11:1–8.
Schuetze K, Ehinger S, Eickhoff A, et al. Cement augmentation of the proximal femur nail antirotation: is it safe? Arch Orthop Trauma Surg. 2020; https://doi.org/10.1007/s00402-020-03531-2. [Online ahead of print]
Xu K. Dual mobility Total hip arthroplasty versus bipolar hemiarthroplasty in treating patients with displaced femoral neck fractures: a systematic review and meta-analysis. 2020; https://doi.org/10.37766/inplasy2020.4.0085.
Guyen O. Hemiarthroplasty or total hip arthroplasty in recent femoral neck fractures? Orthop Traumatol Surg Res. 2019;105(1):95–101.
Florschutz AV, Langford JR, Haidukewych GJ, Koval KJ. Femoral neck fractures: current management. J Orthop Trauma. 2015;29(3):121–9.
Miller BJ, Callaghan JJ, Cram P, Karam M, Marsh JL, Noiseux NO. Changing trends in the treatment of femoral neck fractures: a review of the American Board of Orthopaedic surgery database. J Bone Joint Surg Am. 2014;96(17):e149.
Riggs BL, Melton LJ 3rd. The worldwide problem of osteoporosis: insights afforded by epidemiology. Bone. 1995;17(5):505–11.
Kannan A, Kancherla R, McMahon S, Hawdon G, Soral A, Malhotra R. Arthroplasty options in femoral-neck fracture: answers from the national registries. Int Orthop. 2012;36(1):1–8.
Pass B, et al. Differences of hemiarthroplasty and total hip replacement in orthogeriatric treated elderly patients: a retrospective analysis of the registry for geriatric trauma DGU®. Eur J Trauma Emerg Surg; 2021. https://doi.org/10.1007/s00068-020-01559-y.
Quitmann H. Supercapsular percutaneously assisted (Superpath) approach in total hip arthroplasty – operative Orthopädie Und Traumatologie. SpringerLink, Springer Medizin; 2019. https://springerlink.bibliotecabuap.elogim.com/article/10.1007/s00064-019-0597-5
Tarwala R, Dorr LD. Robotic assisted total hip arthroplasty using the MAKO platform. Curr Rev Musculoskelet Med. 2011;4:151. https://doi.org/10.1007/s12178-011-9086-7.
Palm H. Hip fracture: the choice of surgery. In: Falaschi P, Marsh D, editors. Orthogeriatrics. Practical issues in geriatrics. Cham: Springer; 2021. https://doi.org/10.1007/978-3-030-48126-1_9.
Author information
Authors and Affiliations
Rights and permissions
Copyright information
© 2023 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Tokgöz, E. (2023). Surgical Approach Comparisons in Total Hip Arthroplasty. In: Total Hip Arthroplasty. Springer, Cham. https://doi.org/10.1007/978-3-031-08927-5_3
Download citation
DOI: https://doi.org/10.1007/978-3-031-08927-5_3
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-08926-8
Online ISBN: 978-3-031-08927-5
eBook Packages: EngineeringEngineering (R0)