Abstract
Fly ash (FA) is an inevitable byproduct from the coal-fired thermal power plants that need timely, effective and safe disposal in many developing countries. It is an amorphous ferro-alumino silicate material similar to soil having practically all the elements except organic carbon, nitrogen and phosphorous. Although in many developed countries its use has reached saturation but technologically-starved poor countries are still lagging far behind in its resourceful use. Its use in cement-concrete, and land and mine filling have been widely accepted but in agriculture, this chemically heterogeneous material deserves cautious consideration. At low concentration, FA alters soil physicochemical properties and thus, acts as soil ameliorant or conditioner. However, its use at higher rate is restricted due to presence of heavy metals that affect soil biosphere and limits plant growth. Hence, remediation of toxic metal ions for sustainable agricultural intervention is a prerequisite in FA-contaminated soils or dumpsites. Like phytoremediation, earthworms with unique accumulation, extraction, transformation, conversion, degradation and stimulation properties could also be engaged in remediation of FA. In this chapter, attempts have been made to elucidate various mechanisms and processes involved in vermiremediation, and the advantages, disadvantages and future prospects of this innovative technology.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
10.1 Introduction
With the burgeoning global population, the demands for food have increased tremendously over last few decades beyond the yielding ability of many crops. Increase in the current global food production for feeding the teaming millions is the greatest challenge before us (Dwibedi 2018). The pressure on land for higher productivity per unit area and time is increasing day by day, resulting in more dependence on chemical fertilizers, synthetic pesticides, hormones and probiotics at the cost of environmental health and sustainability. The land is degrading and becoming less productive which needs bio-physical amelioration for bringing back to its pristine conditions. Furthermore, the greed for energy, under the veil of pseudo civilization, prosperity and economic development, has been driving us towards peril (Dwibedi and Sahoo 2017).
Although the global primary energy consumption in 2018 recorded sharp decline in coal share (27%), it still ranks next to petroleum oil (34%) (International Energy Agency 2020). However, other alternative energy sources such as nuclear and hydrothermal power require sophisticated technologies and huge initial investments that are beyond the reach of many developing countries. Therefore, production of ash (bottom and fly ash), is an inevitable byproduct from the coal-fired thermal power plants that need safe, timely and effective disposal. Combustion of pulverized sub-bituminous coal (lignite) in thermal power plants results in generation of this end-residue (Basu et al. 2009). Fly ash (FA) is an amorphous ferro-alumino silicate material similar to soil with all the elements except organic carbon, P and N (Tripathy and Sahu 1997; Pandey and Singh 2010; Pandey 2020a, b, c, d). It has been categorized ‘under high volume low effect waste under Hazardous Waste (Management and Handling and Trans-boundary Movement) Rules, 2008’ (Parab et al. 2012). Its production along with power generation in thermal power plants over decades of economic developments, both by developed and developing countries has been a necessary evil. This problematic ‘solid waste’ across the globe has now acquired the status of ‘resource material’ due to innovative uses in cement-concrete, land and mine filling, agriculture, etc. Its utilization in European countries is almost 100% while in developing countries like India lower percentage is being utilized in spite of its higher production (Dwibedi and Sahoo 2017).
10.2 Properties of FA
The physical, chemical and mineralogical properties of FA (Fisher et al. 1978; Page et al. 1979; Adriano et al. 1980; Carlson and Adriano 1993; Pandey 2020a) depend on the quality of coal, extent of thermal combustion and storage-handling methods. Therefore, ash compositions vary with burning of anthracite, bituminous and lignite coals. Elements present in coal are intense in FA. Physically, FA is very fine with mean diameter of <10 μm, light in texture. It has low to moderate bulk density (BD) and more surface area. Its water holding capacity is of 49–66% on the weight basis (Sharma and Kalra 2006). Its pH ranges from 4.5 to 12 largely depending on the S content in the coal. FA is chemically heterogeneous in nature as it contains variable proportions of different trace and heavy metals such as Be, B, Cd, Cr, Co, Hg, Mo, Mn, Pb and oxides Al, Ca, Fe and Si.
Incorporation of FA alters physicochemical properties of soil and works as soil conditioner or modifier (Kalra et al. 1998; Pandey and Singh 2010; Pandey 2020b). It alters the texture of soil (Kalra et al. 2000), reduces BD and increases porosity, water holding capacity and aeration due to its silty nature. Kuchawar et al. (1997) and Bhaisare et al. (1999) have shown an increase in cation-exchange capacity (CEC) as a result of FA amendment in soil. It also improves soil bacteria count and enzyme activity viz. dehydrogenase, urease and alkaline phosphatase that promote plant growth (Yeledhalli et al. 2007). Comparative physicochemical properties of soil and FA, and also FA in combination with press mud (PM) have been depicted under Table 10.1 (Singh and Pandey 2013).
According to the Intergovernmental Panel on Climate Change (IPCC), lime application for soil amelioration releases carbon dioxide (CO2) gas which ultimately adds to global warming. In United States of America, the Environment Protection Authorities (EPA) has estimated emission of 9 Tg (teragram = 1.012 g = 106 t) of CO2 from an approximate 20 Tg of agricultural lime applied in 2001. FA could be the befitting substitute for it minimizing global warming process (West and McBride 2005). It has also been estimated that 1 tonne of FA has the ability to sequester up to 26 kg of CO2 (i.e. 38.46 tonnes of FA per tonne of CO2 sequestered).
10.3 Verms as Bioreactor
Earthworms, regarded as the intestine of earth (Aristotle), are the terrestrial invertebrates, belonging to the phylum Annelida, and class Oligochaeta and they have more than 3000 species across the globe (Berridge 2020). They act as bioreactors in recycling the organic wastes to reusable plant nutrients at a very low or marginal cost of production and because of that, they act as ‘farmers’ friends’. Wastes from the agricultural field after harvest, and urban and rural solid organic wastes can very well be used in vermicomposting. Vermicomposting of agricultural residues and its effects on plant growth, microbial population and nutrient transformation at different concentrations in soil rhizosphere have been studied with much attention and interest.
10.4 Research Status on FA Use and Vermiremediation
The research on FA use began in late 1970s to evaluate its suitability for improving soil environment and increasing crop productivity (Dwibedi and Sahoo 2017). In developed countries, its utilization is more than 70% but in developing countries; it is still less than 5%. FA may be applied as soil amendment along with organic substrates such as farmyard manure, compost and microbial culture. A lot of research on use of FA in agricultural crops such as rice, maize, grams, beans, vegetables, etc. in pot culture and field trials has already been conducted. Its far-reaching consequences on soil bio-physicochemical properties have also been evaluated in long-term experiments. The role of FA in reclamation of acidic and sodic soils has been well acclaimed. Its utilization in agriculture has been a proven support as it improves physicochemical properties of soil resulting in better fertility and increased crop yield (Rautaray et al. 2003). However, heavy metal accumulation with FA amendment is a great concern. Researchers are in view of its application in lower concentrations as soil microbial population and availability of plant nutrients are affected at higher concentrations.
Earthworms are the ecological engineers having profound role in amelioration of soil physical, chemical and biological properties (Shi et al. 2017). The significant role played by earthworms in soil fertility enhancement, biodiversity restoration and detoxification of contaminated soil was studied since early 1800s (Edwards 2004) while much stress on soil remediation was given during 1980s (Sinha et al. 2010). In the recent past, ‘vermiremediation’, a new approach has been invoked (Gupta and Garg 2009). Attempts have also been made to study the composting behaviour of earthworms at varying levels of FA substrates to prepare vermi-ash.
10.4.1 FA Impact on Soil Characteristics
FA has tremendous potential as a valuable resource in agriculture, building, road and bridge construction and other related areas. Its soil amending and nutrient-enriching properties contribute to agricultural production (Pandey 2020c). It contains considerable quantities of both macro and micronutrients (Singh et al. 1997) which when applied to soil sustain crop growth and development, even in poor soils. As mentioned above, FA is deficient in N, P and organic matter and hence, its amendments with organic materials or microbial inoculants help in plant growth. Its possible agricultural applications such as liming material, fertilizer and physical amendment have been illustrated by many researchers. For effective and efficient vermiremediation of FA, it is imperative to understand the effects of FA on soil properties and agricultural crops as remediated land may simultaneously or subsequently be brought under cultivation. A brief review of the earlier studies on FA use in agriculture is hereunder for general reference.
FA is helpful in increasing the physical properties of soil that ultimately improve soil fertility and enhance crop yield (Rautaray et al. 2003). FA amendment in sunflower fields decreases BD of the soil (Pani et al. 2015). Wong and Wong (1990) noticed alteration in soil texture, bulk density and porosity. FA addition in sandy soil alters soil texture and increases micro-porosity (Ghodrati et al. 1995). Increase in porosity and decrease in bulk density in soil was also reported by Zibilski et al. (1995). Water holding capacity of soil increases with FA amendment in sunflower fields (Pani et al. 2015; Parab et al. 2012). FA amendment in clay soil improves infiltration whereas in the coarse soil it reduces infiltration as reported by Dhindsa et al. (2016).
The pH of soil (pH 6.65) increases with the addition of FA (pH 7.56) due to acid-neutralizing capacity of the latter one in presence of oxides of Ca and Mg in it. The soil becomes more alkaline with FA amendment in sunflower fields (Pani et al. 2015). Such increase in pH was also reported by Lee et al. (2006) and Sarkar et al. (2012). However, Sikka and Kansal (1995) reported no significant increase in pH with FA amendment. Electrical conductivity (EC) of the soil (281 dS cm−1) increases with the addition of FA (600 dS cm−1) in radish field, possibly due to precipitation of soluble cations (Singh et al. 2011a, b) and binding of metal ions to soil separates that facilitates ready availability of plant nutrients (Pani et al. 2015) in FA amended soils. However, elevated EC may suppress normal plant growth (Singh et al. 2011a, b). Organic carbon (OC) decreases with increase in FA concentration in radish (Sarkar et al. 2012) whereas in brinjal, the value of OC increases with FA (Singh et al. 2011a, b). FA improves nutrient levels in soil (Rautaray et al. 2003). Singh et al. (2011a, b) have observed increase in availability of N, P, K, Co, Ni, Cu, Zn, Mo, Al, V, Se, etc. as well as toxic metals such as Cr, Pb and As with addition of FA at different grades. Sarkar et al. (2012) have reported increase in availability of Na, K, Ca, Mg and Fe with significant reduction in total N, available P and OC under FA soil amendment. FA is also used to rectify B and S deficiencies in soil (Chang et al. 1977). P availability increases with the addition of FA (Lee et al. 2006). Reddy et al. (2010) have reported ‘the highest available N (224.6 kg ha−1), P (24.6 kg ha−1), K (366.7 kg ha−1), S (8.80 mg kg−1), Fe (10.62 mg kg−1) and Zn (0.95 mg kg−1) content after harvest of rice crop with application of FA at 15 t ha−1 + FYM at 10 t ha−1 (FA15 + FYM10), which were at par with FA10 + FYM10’. However, Sikka and Kansal (1995) reported no significant increase in available N and P in soil with the addition of FA whereas the available K increased.
The nematode population as observed in Chandrapura Thermal Power Station, reduced significantly (Singh et al. 2011a, b) with 40% FA amendment (Ahmad and Alam 1997; Khan et al. 1997) due to inhibitory effect (Khan et al. 1997; Tarannum et al. 2001) of FA. The carbon dioxide efflux from the soil as an indirect method of knowing soil biotic activities increased with 0–100 t ha−1 addition of FA than 400–700 t ha−1 amendments. Several metals present at potentially toxic levels in FA might have suppressed soil heterotrophic microbial activities at higher levels (Arthur et al. 1984).
10.4.2 FA in Agriculture
Direct use of FA in crop fields is not so promising due to poor bioavailability of plant nutrients such as C, N and P that inhibit mineralization through reduced microbial activities (Lazcano 2009/66). When applied to soil directly, it severely inhibits microbial process, N cycle and enzyme activity (Lazcano 2009). Pandey et al. (2009a) observed accumulation of Fe, Zn, Cu, Cd and Cr in Cajanas cajan when the soil was mixed with FA. FA amendment affects rice germination count in initial stage but after 115 h, it picks up again equalizing with the untreated soil. Such delay in germination could be due to increase in soil impedance/ strength (Kalra et al. 1997). However, no such inhibitory effect is noticed in green gram, golden gram and black gram at 0, 10, 20, 30, 40 and 50% FA amendment, except at 100%; possibly due to balance between growth promoters and inhibitors (Singh et al. 2011a, b). The highest rice seed germination is at 20 and 30% FA amendment (Adriano and Weber 2001) while the lowest is at 100% (Panda and Tikadar 2014). Germination of rice and maize in wet season is less sensitive to moderate FA than dry season (Kalra et al. 1998) whereas germination decreases with further increase in ash concentration (Panda and Tikadar 2014).
Shoot and root length of green gram, golden gram and black gram increase with application of FA and the maximum length occurs at 30–40% while in radish, FA shortens plant height (Singh et al. 2011a, b). Shoot length of Luffa cylindrica increases up to 180 t ha−1 FA but at higher dose, the plant shortens (Singh et al. 2011a, b). At 25% FA, taller rice plants with longer roots are observed compared to no or higher levels (Panda and Tikadar 2014). Tiller count in rice goes on increasing with the addition of FA up to 75 t ha−1 (Priatmadi et al. 2015) but on further addition, it declines (Sarkar et al. 2012). Chlorophyll a and b and carotenoid pigment concentration in chickpea, golden gram and black gram improves significantly at moderate levels of FA (120–180 t ha−1) but at 240 t ha−1, the pigmentation decreases (Singh et al. 2011a, b). Dry matter accumulation in rice seedlings reduces with increase in concentration of FA from 25 to 100% in rice nursery (Panda and Tikadar 2014). FA and FYM amendments enhance the rates of N transformation processes, plant available-N and paddy productivity (Singh and Pandey 2011) and can be used to enrich nutrient-poor soils for crop productivity and yields. The mixture of FA and press mud shows positive effect on crop growth, physicochemical, microbial and enzymatic activities of sodic soil (Singh et al. 2016a). The mixture of 40% soil + 20% FA + 40% vermicompost is proved as most promising blend for wet rice nursery raising and for remediating the coal FA in agricultural production system (Dwibedi et al. 2021). Recently, it is proved that phytoremediated FA can be used as a fertilizer up to 100% for peas farming as metal concentrations was reported either below detection limit or below the WHO permissible limit (Bhattacharya et al. 2021). The application of FA for agriculture production is explored in great depth using the facts of plants, amendments, FA doses range and remark (Pandey et al. 2009b).
10.5 What is Vermiremediation?
The term ‘vermiremediation’ has come from two Latin words: ‘vermis’ means ‘worm’ and ‘remedium’ means ‘correct’ or ‘remove an evil’ (Shi et al. 2020). The term was coined by Edward and Arancon (2006) while Rodriguez-Campos et al. (2014) first attempted to define it as ‘the use of earthworms for removing contaminants (Sinha et al. 2008) or not recyclable compounds (Gupta and Garg 2009) from the soil’. However, a better definition by Shi et al. (2020) has come up later which expresses ‘vermiremediation as an earthworm-based bioremediation technology that makes use of the earthworm’s life cycle (i.e. feeding, burrowing, metabolism and secretion) or their interaction with other abiotic and biotic factors to accumulate and extract, transform, or degrade contaminants in the soil environment’. As per this definition, few synonymous terms viz. vermiaccumulation and vermiextraction, vermitransformation, vermiconversion and drilodegradation or drilostimulation could be used to understand the mechanisms and processes of vermiremediation (Shi et al. 2020).
Vermiaccumulation and vermiextraction, similar to term phytoaccumulation, refer to the process of ingestion of contaminants (organic and inorganic) from the soil by earthworms and accumulation of pollutants in their body parts (Shi et al. 2020). Accumulation of contaminants occurs through preferential dermal or intestinal sequestration involving sub-organismic (preclitellum, clitellum, post-clitellum), tissue (body wall, gut, body fluids) and sub-cellular (intra and extracellular fractions) body parts of the earthworm (Shi et al. 2020). The process of biotransformation of contaminants by earthworms into harmless products by enzymes (such as peroxidases) and microbes (bacteria and fungi) in the alimentary canal and ultimately egested out as compost is known as vermitransformation or vermiconversion (Panda and Tikadar 2014). Drilodegradation or drilostimulation refers to the microbial decomposition, degradation or elimination of toxic materials by microbes present in the drilosphere, the 2 mm thick zone of earthworm burrow wall (Bouché 1975; Brown et al. 2000). Drilospheric soil is rich in earthworm mucus and casts that stimulate microbial growth which subsequently promotes the growth of protozoa and nematodes (Stromberger et al. 2012). Drilosphere, a habitat rich in energy and nutrients, mostly C and N, acts as hotspot for soil microbial communities (Kuzyakov and Blagodatskaya 2015). The nutrients are mixtures of low-molecular organic acids such as amino acids, nucleic acid derivatives, carbohydrates, phenolics and enzymes (Zhang et al. 2009). The labile organic carbon supply in drilosphere can sustain microbial communities that supplement utilizable sources of energy (Tiunov and Scheu 1999). And hence, drilospheric microorganisms have tremendous ability to remediate the potential pollutants (Shi et al. 2020).
10.5.1 Advantages of Vermiremediation
Vermiremediation is an emerging concept that needs rigorous investigation and exploration for gaining ecological milestones over conventional physicochemical methods. Primarily, it is one of the cheapest, easiest, efficient and in some cases, the fastest way of remediating the contaminated land without disturbing the topsoil. Furthermore, it is not substrate-specific, rather a useful technology for treating a wider range of hazardous pollutants. Synthetic insecticides, herbicides, polycyclic aromatic hydrocarbons (PAHs), polychlorinated biphenyls (PCBs), crude oil and FA in soil can be removed by engaging earthworms (Rodriguez-Campos et al. 2014). It is environmentally sustainable self-regenerating in-situ approach to remediate polluted land. Furthermore, vermiremediation enhances soil quality through addition of organic matter, supplementation of plant nutrients and proliferation of biodiversity (Sinha et al. 2008).
10.5.2 Limitations of Vermiremediation
Vermiremediation technology has its own limitations as it can only be applicable in moderately or slightly contaminated soils that allow survival of the earthworms. In severely contaminated soil, earthworms may not survive due to toxic effects of the pollutants (Rodriguez-Campos et al. 2014; Shi et al. 2019). Vermiremediation is also restricted to the earthworm habitats depending on the species used and ambient environmental conditions-beyond which its efficacy is limited. Earthworms are categorized into epigeic, anecic and endogeic groups (Fig. 10.1) depending on the species used, body size, mobility, fecundity, habitat, feeding and burrowing behaviour, casting activity, etc. (Lazcano et al. 2009) of the earthworm. Dendrobaena octaedra, Dendrobaena attemsi, Dendrodrilus rubidus, Eiseniella tetraedra, Heliodrilus oculatus, Lumbricus rubellus, Lumbricus castaneus, Lumbricus festivus, Lumbricus friendi, Lumbricus rubellus, Satchellius mammalis, Eisenia fetida and Eudrilus euginae live on the upper layer of the soil profile and feed mainly on organic debris and thus are classified as detritivores under epigeic group. Endogeic (means within the earth) earthworms such as Allolobophora chlorotica, Apporectodea caliginosa, Apporectodea icterica, Apporectodea rosea, Drawida grandis, Murchieona muldali, Octolasion cyaneum, Octolasion lacteum, Anecies longa, Anecies nocturna and Octochaectona thurstoni remain deep inside the soil and are geophagus in nature. Whereas anecics or anegeic (out of the earth) earthworms, e.g. Aporrectodea longa, Aporrectodea nocturna, Lumbricus friend, Lumbricus terrestris and Letmpito mauritii are sub-surface dwellers and are phyto-geophagus in nature (Brown and Sherlock 2021; Bhattacharya and Kim 2016). Vermiremediation potential is dependent on food abundance and feeding preference of earthworm species (Curry and Schmidt 2007). Earthworms are sensitive to temperature, moisture and other climatic and seasonal conditions that may inhibit their survivability thereby affecting the vermiremediation process (Butt and Lowe 2011). Additionally, accumulated contaminants in earthworms can become a potential threat if get transferred into food chain under mismanagement in disposal schedule (Shi et al. 2014).

Source Adapted and modified from Brown and Sherlock (2021)
Three major ecological groups of earthworms identified basing on feeding and burrowing behavior.
10.6 Biology of Earthworm and Its Functional Significance in Waste Degradation
Before getting into the process of vermiremediation in FA-contaminated soils, it is imperative to know the biology of earthworm and the mechanism of waste degradation with relation to soil health. They prefer moist and dark habitats with optimum moisture of 60–75% and their skin is permeable for which they need moist environment to prevent from drying out (Shi et al. 2020). Although they can survive temperature range of 5–35 °C, but the optimum is 20–25 °C. Most of them prefer neutral pH and C/N ratio of 2–8 (Sharma and Garg 2018). Within a life span of 220 days, they produce 300–400 offspring (Shi et al. 2020). They are bisexual and under ideal soil temperature, moisture, pH and food availability they can multiply 28 times in every six months (Shi et al. 2020). They mostly feed on detritus materials, living bacteria, fungi, protozoa, nematodes and many other microorganisms (Sharma and Garg 2018). Earthworms have digestive tubes housed inside their thick cylindrical muscular outer body tube (Berridge 2020). They swallow considerable amount of food materials along with soil through their mouth present at 1st segment and shred down by gizzard present at 8th or 8th to 9th segment. The elementary canal of earthworm includes mouth (1st), buccal cavity (2nd and 3rd), pharynx (3rd and 4th), esophagus (5th to 7th), gizzard (8th or 8th and 9th), stomach (9th or 10th to 14th), intestine (15th up to the last segment except anus) and anus (Aryal 2020). They also passively absorb dissolved chemicals through their body wall (Shi et al. 2020). These absorbed and eaten substrates are mixed with intestinal fluid and enzymes from microbes. Earthworm’s intestine acts as warehouse for microbes and enzymes such as lipase, amylase, nitrate reductase, protease, phosphatase, cellobiase, etc. that bioprocess disintegration of ingested foodstuffs.
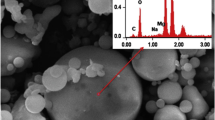
Earthworms maintain and improve soil quality parameters (Bhadauria and Saxena 2009) and act as bioindicators of soil quality (Fründ et al. 2011). Abundance and species composition of earthworms, their behaviour in contact with the soil, assimilation of chemicals in their body parts and biochemical or cytological stress markers can indicate soil quality (Fründ et al. 2011). Earthworms produce pores and aggregates (biostructures) in soil, thus influencing soil's physical properties, nutrient cycling and plant growth (Lal 1999; Scheu 2003). Anecic species make permanent burrows in mineral soils; they drag surface organic materials into the soil for food. Endogeic species are the ecosystem engineers who make nonpermanent burrows in the upper surface mineral layer through which other organisms get accessibility to underground resources (Jones et al. 1994). No till or minimal disturbance to the soil, as in conservation agriculture, enhances organic residues, thus creating ideal conditions for earthworm habitat (Labenz 2021). Mucus production associated with water excretion by earthworms enhances the activity of soil beneficial microorganisms that help in improving soil structure and aggregate stability. Earthworm’s excreta (cast) are rich in plant-available nutrients, thus concentration of N, P, K, Ca, Mg and many more trace elements in soil increases and toxic materials including heavy metals get accumulated in their gut (Usmani and Kumar 2017) which make them biologically potent for remediation of FA (Fig. 10.2).

[source Usmani and Kumar 2017 (Adapted and modified with the permission of the Publisher)]
Physicochemical transformations occurring in different compartments of earthworm illustrating heavy metal sequestration and nutrient assimilation on ingestion of FA
Metal accumulation mostly occurs in the chloragogenous tissue at the posterior end of the alimentary canal of earthworm (Usmani and Kumar 2017; Morgan and Morris 1982). On exposure to metals, earthworms synthesize metallothioneins (MT) that have low-molecular weight, cysteine-rich proteins with high affinity towards Cd, Cu and Zn (Dallinger 1994). These proteins protect organisms against toxic metal stress and thus can be used as indicator of soil pollution. While dealing the unnecessary heavy metals, earthworms detoxify their effects through interaction with many chemicals in the metabolic processes. Bioaccumulation of metals and organocomplex formation results in decline in the availability of heavy metals in soil as part of enzyme antioxidant systems such as superoxide dismutase (SOD) and MT (Li et al. 2008). As mentioned above, the highest metal accumulation occurs in the posterior alimentary canal (PAC) of the earthworm. Intracellular vesicles within PAC accumulate Pb and Zn and the superfluous metals interact with P ligands within the chloragosome matrix (Usmani and Kumar 2017; Morgan and Morgan 1990). The cation-exchange properties in chloragosomes (Fischer 1973, 1977) are considered as integral part for the physiological functioning of intracellular organelles (Morgan 1981; Fischer and Trombitts 1980). Microprobe X-ray analysis of air-dried chloragogenous tissue revealed Ca, Pb and Zn (in association with sulphur) accumulation in the chloragosomes while Cd was accumulated in an electron-lucent vesicular component called cadmosome (Usmani and Kumar 2017).
10.7 Process of Vermiremediation
Metal accumulation by earthworm (vermiremediation) may be in-situ or on-site treatment in the FA dumped sites (contaminated land), or it may be ex-situ through vermicomposting (Usmani and Kumar 2017). Eisenia fetida cannot tolerate 100% FA, thus addition of organic matter is essential (Niyazi and Chaurasia 2014). Considerable reduction in metal concentration occurs after vermiremedition. FA lacks N and C and thus organic matter addition is required to support microbial growth (Mupambw et al. 2015). Experiments on vermicomposting of cow dung with FA showed 30–50% reduction in heavy metal concentration up to 60% FA while 10–30% reduction was in 80% FA addition. Hence, 60% addition of FA with E. fetida was proposed to be a sustainable vermiremediation technique (Gupta et al. 2005). In another experiment, minimum mortality and maximum population growth were observed in 1:3 mixture of FA and cow dung. Significant reduction of heavy metals viz. Cu, Pb, Mn and Cr were also observed with vermiremediation at variable range of FA and cow dung mixtures. Vermistabilization resulted reduction in pH by 8–15.7%, EC by 16.2–53.6%, total organic carbon by 15.6–32.5% and C:N ratio by 43.2–97.4% (Singh et al. 2016b). A decline in heavy metal concentration in vermicompost was reported by Niyazi and Chaurasia (2014) like Anderson and Laursen (1982), Morgan and Morgan (1990) who observed variations in metal accumulation depending on inter-specific metal intake ability, worm age, their physiological utilization and transformation, season and many other factors (Usmani and Kumar 2017).
10.8 Strategies for Vermiremediation
Earthworm survival and mobility of contaminants are the two limiting factors in vermiremediation (Usmani and Kumar 2017). The performance of earthworms is affected by poor soil quality, environmental conditions and high concentration of pollutants (Sinha et al. 2008). Vermiremediation of FA-contaminated soils needs controlled mobility and bioavailability of toxicants and facilitated growth of earthworms under ameliorated soil environment. Nutrient and organic amendments and provisioning for better soil physical properties should be the prime management strategies for efficient and effective vermiremediation. The vermiremediation capacity of different earthworm species needs through assessment before their engagement in contaminated land reclamation. Suitability of crops to differential FA and organic residue amendments and bioaccumulation of toxic heavy metals across trophic levels need in-depth investigation for validation of the remediation technologies. Safe and timely evacuation of earthworms in vermiremediation is mostly lacking, which requires burning as specialized for hazardous waste (Sheoran et al. 2010; Ali et al. 2013). A brief account of different harvest methods of the earthworms used in vermiremediation is presented under Table 10.2. Vermiremediation can be facilitated through appropriate microbe-earthworm combined interactions as is evident in phosphorous solubilizing bacteria inoculated FA amendments (Lukashe et al. 2018). Harmonious integration of phytoremediation, vermiremediation and effective microorganisms has been far better option against any two of these remediation techniques to clean up residual contaminants (Deng and Zeng 2017). In heavily contaminated soils, vermiremediation can be used as polishing step after primary remedial treatment (Sinha et al. 2008). Another way of enhancement of vermiremediation is through quality food supplementation and optimization of the inoculation conditions (temperature, pH, aeration, moisture, etc.) that ultimately increase earthworm biomass and rate of uptake of contaminants as well (Curry and Schmidt 2007). Improvement of agronomic conditions such as soil texture, organic matter, hydraulic conductivity and homogenization of contaminants to avoid hotspots will certainly enhance vermiremediation (Gerhardt et al. 2017). Since it is impracticable and time-consuming to study individual species under all possible conditions, various models viz. empirical, rate, equilibrium-partition, mechanical and fugacity models predicting uptake and accumulation of toxic materials in earthworms need to be validated (Shi et al. 2020).
10.9 Conclusions and Prospects
Vermiremediation as an expanding, sustainable, ecofriendly and cost-effective technology available for treatment of polluted soils, including FA, has been well acknowledged widely. Unlike physiochemical remediation, vermiremediation is an environmental supportive and relatively cheaper, easier, effective and efficient technique that should be highlighted. Many researchers have studied vermiremediation of FA over past few decades thereby opening up an innovative scientific approach in remediating contaminated land. Vermiaccumulation and vermitransformation play important roles in vermiremediation of pollutants like heavy metals in FA. Furthermore, emphasis is to be given for enhancing bioavailability of organic residues and by providing congenial environment for optimum growth of earthworms. Integration of effective microorganisms, agronomic practices, phytoremediation, biomass enhancement, etc. has the potential to facilitate vermiremediation. Safe and timely harvest and disposal of contaminated earthworms could prevent biomagnification of pollutants in natural food chains which should be considered seriously. Available models for predicting uptake and accumulation in earthworms need to be validated so that the capacity, contribution and mechanism of different processes in vermiremediation are fully clarified.
References
Adriano DC, Page AL, Elseewi AA, Chang AC, Straughan I (1980) Utilization and disposal of fly ash and other coal residues in terrestrial Ecosystems: a review. J Environ Qual 9:333–344. https://doi.org/10.2134/jeq1980.00472425000900030001x
Ahmad A, Alam MM (1997) Utilization of fly ash and Paecilomyces lilacinus for the control of Meloidogyne incognita. Int J Nematol 7:162–164
Ali H, Khan E, Sajad MA (2013) Phytoremediation of heavy metals—concepts and applications. Chemosphere 91:869–881. https://doi.org/10.1016/j.chemosphere.2013.01.075
Anderson C, Laursen J (1982) Distribution of heavy metal in Lumbricus terrestris, Aporrectodea longa and A. rosea measured by atomic absoption and X ray fluoresecence spectrometry. Pedobiologia 24:347–356. https://inis.iaea.org/search/searchsinglerecord.aspx?recordsFor=SingleRecord&RN=14763698
Arthur MF, Zwick TC, Tolle DA, Voris PV (1984) Effects of fly ash on microbial CO2 evolution from an agricultural soil. Water Air Soil Pollut 22(2):209–211
Aryal S (2020) Digestive system of earthworm. The biology notes. Retrieved from https://thebiologynotes.com/digestive-system-earthworm/
Basu M, Pande M, Bhadoria PBS, Mahapatra SC (2009) Potential fly-ash utilization in agriculture a global review. Prog Nat Sci 19:1173–1186. https://doi.org/10.1016/j.pnsc.2008.12.006
Berridge L (2020) Earthworm biology. Earthworm Soc Britain, 20 March 2020. https://www.earthwormsoc.org.uk/earthworm-biology
Bhadauria T, Saxena KG (2009) Role of earthworms in soil fertility maintenance through the production of biogenic structures. Appl Environ Sci 2010:7 pages. Article ID 816073. https://doi.org/10.1155/2010/816073
Bhaisare B, Matte DB, Badole WP, Deshmukh A, Pillewan S (1999) Effect of fly ash on physicochemical properties of Vertsoils and yield of green gram. J Soils Crops 9:255–257
Bhattacharya SS, Kim KH (2016) Utilization of coal ash: is vermitechnology a sustainable avenue? Renew Sustain Energy Rev 58:1376–1386. https://doi.org/10.1016/J.RSER.2015.12.345
Bhattacharya T, Pandey SK, Pandey VC, Kumar A (2021) Potential and safe utilization of Fly ash as fertilizer for Pisum sativum L. Grown in phytoremediated and non-phytoremediated amendments. Environ Sci Pollut Res. https://doi.org/10.1007/s11356-021-14179-9
Bouché MB (1975) Action de la faune sur les états de lamatière organique dans les ecosystemes. In: Kilber-tius G, Reisinger O, Mourey A, Cancela da Fon-seca JA (eds) Humification et biodégradation. Pier-ron, Sarreguemines, France, pp 157–168
Brown GG, Barois I, Lavelle P (2000) Regulation of soil organic matter dynamics and microbial activity in the drilosphere and the role of interactions with other edaphic functional domains. Eur J Soil Biol 36:177–198
Brown KD, Sherlock E (2021) Earthworm ecology. The Earthworm Society of Britain: digging into the world of earthworms. Retrieved on 20 April 2021 from https://www.earthwormsoc.org.uk/earthworm-ecology
Butt KR, Lowe CN (2011) Controlled cultivation of endogeic and anecic earthworms. In: Karaca A (ed) Biology of earthworms. Springer, Berlin, Heidelberg, pp 107–121. https://doi.org/10.1007/978-3-642-14636-7_7
Carlson CL, Adriano DC (1993) Environmental impact of coal combustion residues. J Environ Qual 22:227–247. https://doi.org/10.2134/jeq1993.00472425002200020002x
Chan KY, Munro K (2001) Evaluating mustard extracts for earthworm sampling. Pedobiologia 45:272–278. https://doi.org/10.1078/0031-4056-00084
Chang AC, Lund LJ, Page AL, Warneke JE (1977) Physical properties of fly ash-amended soils. J Environ Qual 6:267–270
Čoja T, Zehetner K, Bruckner A, Watzinger A, Meyer E (2008) Efficacy and side effects of five sampling methods for soil earthworms (Annelida, Lumbricidae). Ecotoxicol Environ Saf 71:552–565. https://doi.org/10.1016/j.ecoenv.2007.08.002
Curry JP, Schmidt O (2007) The feeding ecology of earthworms—a review. Pedobiologia 50:463–477. https://doi.org/10.1016/j.pedobi.2006.09.001
Dallinger R (1994) Invertebrate organisms as biological indicators of heavy metal pollution. Human Press Inc 48(a):27–31. https://doi.org/10.1007/BF02825356
Deng S, Zeng D (2017) Removal of phenanthrene in contaminated soil by combination of alfalfa, white-rot fungus, and earthworms. Environ Sci Pollut Res 24:7565–7571. https://doi.org/10.1007/s11356-017-8466-y
Dhindsa HS, Sharma RD, Kuma R (2016) Role of fly ash in improving soil physical properties and yield of wheat (Triticum aestivum). Agri Sci Dig 36(2):97–101. https://doi.org/10.18805/asd.v36i2.10626
Dwibedi SK, Sahoo SK (2017) Effect of fly ash and vermicompost on the performance of rice seedlings and physicochemical properties of the soil in West Central Table Land Zone of Odisha. M. Tech. (Environmental Science and Engineering) thesis, Sambalpur University, Jyoti Vihar, Odisha, India
Dwibedi SK, Sahu SK, Pandey VC, Rout KK, Behera M (2021) Seedling growth and physicochemical transformations of rice nursery soil under varying levels of coal fly ash and vermicompost amendment. Environ Geochem Hlth. https://doi.org/10.1007/s10653-021-01074-y
Dwibedi SK (2018) Wetland management through agro-geo-special technology in Odisha. 11th science communicator’s meet. 105th Indian Science Congress, 19–20 March, Manipur University-795003, pp 27–28
East D, Knight D (1998) Sampling soil earthworm populations using household detergent and mustard. J Biol Educ 32:201–206. https://doi.org/10.1080/00219266.1998.9655621
Edwards CA (2004) Earthworm ecology. CRC Press, Boca Ratoon, FL
Edward CA, Arancon NQ (2006) The science of vermiculture: the use of earthworms in organic waste manangement. In: Guerrero RDIII, Guerrero-del Castillo MRA (eds) Vermi technologies for developing countries. Proceedings of the international symposium-workshop on vermi technologies for developing countries. Nov. 16–18, 2005
Eisenhauer N, Straube D, Scheu S (2008) Efficiency of two widespread non-destructive extraction methods under dry soil conditions for different ecological earthworm groups. Eur J Soil Biol 44(1):141–145. https://doi.org/10.1016/j.ejsobi.2007.10.002
Fischer E (1973) The chloragosomes of Lumbricidae as cation exchangers in vitro investigations. Acta Biol Acad Sci H 24:157–163
Fischer E (1977) The function of chloragosomes, the specific age pigment granules of annelids—a review. Exp Geront 12:69–74. https://doi.org/10.1016/0531-5565(77)90035-3
Fischer E, Trombitts K (1980) X-ray microprobe analysis of chloragosomes of untreated and EDTA-treated Lumbricus terrestris by using fresh air-dried smears. Acta Histochem 66:237–242. https://doi.org/10.1016/S0065-1281(80)80008-0
Fischer GL, Chang DPY, Brummer M (1978) Fly ash collected from electrostatic precipitator. Microcrystalline structures and the mystery of the spheres. Science 19:553. https://doi.org/10.1126/science.192.4239.553
Fründ HC, Graefe U, Tischer S (2011) Earthworms as bioindicators of soil quality. In: Karaca A (ed) Biology of earthworms. Soil Biol, vol 24. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-642-14636-7_16
Gerhardt KE, Gerwing PD, Greenberg BM (2017) Opinion: taking phytoremediation from proven technology to accepted practice. Plant Sci 256:170–185. https://doi.org/10.1016/j.plantsci.2016.11.016
Ghodrati M, Sims JT, Vasilas BS (1995) Evaluation of flyash as a soil amendment for the Atlantic coastal plain. I. Soil hydraulic properties and elemental leaching. J Water Soil Air Pollut 81:349–361. https://doi.org/10.1007/BF01104020
Goyal V, Angar MR, Srivastava DK (2002) Studies on the effect of fly ash treated soil on the increased protein contents in the seeds of Glycine max (soyabean). Asian J Chem 14:328–332
Gupta R, Garg VK (2009) Vermiremediation and nutrient recovery of non-recyclable paper waste employing Eisenia fetida. J Haz Mater 162(1):430–439. https://doi.org/10.1016/j.jhazmat.2008.05.055
Gupta SK, Tewari A, Srivastava R, Murthy RC, Chandra S (2005) Potential of Eisenia fetida for sustainable and efficient vermicomposting of fly ash. Water Air Pollut 163(1/4):293–302. https://doi.org/10.1007/s11270-005-0722-y
International Energy Agency (2020) Share of coal in global energy mix touches lowest level in 16 years, drops to 27 percent. IEA Clean Copal Centre. https://www.iea-coal.org/share-of-coal-in-global-energy-mix-touches-lowest-level-in-16-years-drops-to-27-per-cent/
Jones CG, Lawton JH, Shachak M (1994) Organisms as ecosystem engineers. Oikos 69(3):373–386. https://doi.org/10.1007/978-1-4612-4018-1_14
Kalra N, Jain M, Joshi H, Choudhry R, Harit R, Vasta B, Sharma S, Kumar V (1998) Fly ash as a soil conditioner and fertilizer. Biores Technol 64:163–167. https://doi.org/10.1016/S0960-8524(97)00187-9
Kalra N, Harit R, Sharma S (2000) Effect of fly ash incorporation on soil properties on texturally variant soils. Biores Technol 75:91–93. https://doi.org/10.1016/S0960-8524(00)00036-5
Khan MR, Khan MW, Singh K (1997) Management of root-knot disease of tomato by the application of fly ash in soil. Plant Pathol 46:33–43. https://doi.org/10.1046/j.1365-3059.1997.d01-199.x
Kuchawar OD, Matte DB, Kene DR (1997) Effect of fly ash application on physico-chemical properties of soil. J Soils Crops 7:73–75
Kumar V, Zacharia KA, Goswami G (2000) Fly ash use in agriculture: a perspective Proceedings of the 2nd international conference on fly ash disposal and utilization, vol 2, pp 1–13
Kuzyakov Y, Blagodatskaya E (2015) Microbial hotspots and hot moments in soil: concept & review. Soil Biol Biochem 83:184–199. https://doi.org/10.1016/j.soilbio.2015.01.025
Labenz AT (2021) Earthworm activity increases soil health. Natural Resources Conservation Service Hutchinson, Kansas. Retrieved on 20 March 2021. https://www.nrcs.usda.gov/wps/portal/nrcs/detail/ks/newsroom/features/?cid=stelprdb1242736
Lal R (1999) Soil conservation and biodiversity. In: Hawksworth DL (ed) The biodiversity of microorganisms and invertebrates: its role in sustainable agriculture. CAB International, Wallingford, UK, pp 89–103
Lawrence AP, Bowers MA (2002) A test of the ‘hot’ mustard extraction method of sampling earthworms. Soil Biol Biochem 34:549–552. https://doi.org/10.1016/S0038-0717(01)00211-5
Lazcano C, Arnold J, Tato A, Zaller JG (2009) Dominguez J (2009) Compost and vermicompost as nursery pot components: Effects on tomato plant growth and morphology. Span J Agric Res 7:944–951. https://doi.org/10.5424/sjar/2009074-1107
Lee H, Ha HS, Lee CH, Lee YB, Kim PJ (2006) Fly ash effect on improving soil properties and rice productivity in Korean paddy soils. Biores Technol 97:1490–1497. https://doi.org/10.1016/j.biortech.2005.06.020
Li LZ, Zhou DM, Wang P, Luo XS (2008) Subcellular distribution of Cd and Pb in earthworm Eisenia fetida as affected by Ca2+ ions and Cd-Pb interaction. Ecotoxicol Environ Saf 71:632–637. https://doi.org/10.1016/j.ecoenv.2008.04.001
Lukashe NS, Mupambwa HA, Green E, Mnkeni PNS (2018) Inoculation of fly ash amended vermicompost with phosphate solubilizing bacteria (Pseudomonas fluorescens) and its influence on vermi-degradation, nutrient release and biological activity. Waste Manage 83:14–22. https://doi.org/10.1016/j.wasman.2018.10.038
Morgan AJ (1981) A morphological and electron-microprobe study of the inorganic composition of the mineralized secretory products of the calciferous gland and chloragogenous tissue of the earthworm, Lumbricus terrestris L. The distribution of injected strontium. Cell Tissue Res 200:829–844. https://doi.org/10.1007/BF00210465
Morgan AJ, Morris B (1982) The accumulation and intracellular compartmentation of cadmium, lead, zinc and calcium in two earthworm species (Dendrobaena rubida and Lumbricus rubellus) living in highly contaminated soil. Histochem Cell Biol 75:269–285. https://doi.org/10.1007/BF00496017
Morgan JE, Morgan AJ (1990) The distribution of cadmium, copper, lead, zinc and calcium in the tissues of earthworm Lumbricus rubellus sampled from one uncontaminated and four polluted soils. Oecologia, (Berlin) 84:559–566. https://doi.org/10.1007/BF00328174
Mupambw HA, Dube E, Mnkeni PNS (2015) Fly ash composting to improve fertiliser value—a review. S Afr J Sci 111:7–8. https://doi.org/10.17159/SAJS.2015/20140103
Niyazi R, Chaurasia S (2014) Vermistabilization of fly ash amended with pressmud by employing Eisenia foetida. Int J Pharma Chem Biol Sci 4(1):85–95. http://www.ijpcbs.com/files/volume4-1-2014/13.pdf
Page AL, Elseewi AA, Straughan IR (1979) Physical and chemical properties of fly ash from coal-fired power plants with reference to environmental impacts. Residue Rev 71:83–120. https://springerlink.bibliotecabuap.elogim.com/chapter/. https://doi.org/10.1007/978-1-4612-6185-8_2
Panda D, Tikadar P (2014) Effect of fly ash incorporation in soil on germination and seedling characteristics of rice (Oryza sativa L.). Biolife J 2(3):800–807
Pandey VC, Singh N (2010) Impact of fly ash incorporation in soil systems. Agric Ecosyst Environ 136:16–27. https://doi.org/10.1016/j.agee.2009.11.013
Pandey VC, Abhilash PC, Singh N (2009a) The Indian perspective of utilizing fly ash in phytoremediation, phytomanagement and biomass production. J Environ Manage 90:2943–2958. https://doi.org/10.1016/j.jenvman.2009.05.001
Pandey VC, Abhilash PC, Upadhyay RN, Tewari DD (2009b) Application of fly ash on the growth performance and translocation of toxic heavy metals within Cajanus cajan L.: implication for safe utilization of fly ash for agricultural production. J Hazard Mater 166:255–259. https://doi.org/10.1016/j.jhazmat.2008.11.016
Pandey VC (2020a) Phytomanagement of fly ash. Elsevier (Authored book), p 334. ISBN: 9780128185445. https://doi.org/10.1016/C2018-0-01318-3
Pandey VC (2020b) Fly ash properties, multiple uses, threats, and management: an introduction. In: Phytomanagement of fly ash. Elsevier, Amsterdam, pp 1–34. https://doi.org/10.1016/B978-0-12-818544-5.00001-8
Pandey VC (2020c) Scope of fly ash use in agriculture–prospects and challenges. In: Phytomanagement of fly ash. Elsevier, Amsterdam, pp 63–101. https://doi.org/10.1016/B978-0-12-818544-5.00003-1
Pandey VC (2020d) Opportunities and challenges in fly ash–aided paddy agriculture. In: Phytomanagement of fly ash. Elsevier, Amsterdam, pp 103–139. https://doi.org/10.1016/B978-0-12-818544-5.00004-3
Pani NK, Samal P, Das R (2015) Effect of fly ash on soil properties, changes in bio-chemical parameters and heavy metal uptake in sunflower (Helianthus annus L.). Int J Sci Technol Manage 4(7):1–11. http://www.ijstm.com/images/short_pdf/1438321607_P174-184.pdf
Parab N, Mishra S, Bhonde SR (2012) Prospects of bulk utilization of fly ash in agriculture for integrated nutrient management. Bull Nat Inst Ecol 23:31–46. https://nieindia.org/Journal/index.php/niebull/article/view/162
Priatmadi BJ, Saidy AR, Septiana M (2015) Soil properties and growth performance of rice (Oryza sativa L.) grown in a fly-ash amended soil. Trop Wetland J 1(1):19–24
Rautaray SK, Ghosh BC, Mittra BN (2003) Effect of fly ash, organic wastes and chemical fertilizers on yield, nutrient uptake, heavy metal content and residual fertility in a rice-mustard cropping sequence under acid lateritic soils. Biores Technol 90:275–283. https://doi.org/10.1016/S0960-8524(03)00132-9
Reddy TP, Umadevi M, Rao PC (2010) Effect of fly ash and farm yard manure on soil properties and yield of rice grown on an inceptisol. Agric Sci Dig 30(4):281–285. https://www.indianjournals.com/ijor.aspx?target=ijor:asd&volume=30&issue=4&article=012
Rodriguez-Campos J, Dendooven L, Alvarez-Bernal D, Contreras-Ramos SM (2014) Potential of earthworms to accelerate removal of organic contaminants from soil: a review. Appl Soil Ecol 79:10–25. https://doi.org/10.1016/j.apsoil.2014.02.010
Sarkar A, Singh A, Agrawal SB (2012) Utilization of fly ash as soil amendments in agricultural fields of North-Eastern Gangetic plains of India: potential benefits and risks assessments. Bull Nat Inst Ecol 23:0-00. https://nieindia.org/Journal/index.php/niebull/article/view/150
Scheu S (2003) Effects of earthworms on plant growth: patterns and perspectives. Pedobiologia 47(5–6):846–856. https://doi.org/10.1078/0031-4056-00270
Sharma K, Garg VK (2018) Chapter 17—Solid-state fermentation for vermicomposting: a step toward sustainable and healthy soil. In: Pandey A, Larroche C, Soccol CR (eds) Current developments in biotechnology and bioengineering. Elsevier, pp 373–413. https://doi.org/10.1016/B978-0-444-63990-5.00017-7
Sharma SK, Kalra N (2006) Effect of fly ash incorporation on soil properties and plant productivity—a review. J Sci Ind Res 65:383–390. https://www.researchgate.net/publication/224319633_Effect_of_flyash_on_soil_properties_and_productivity_of_crops_A_review#:~:text=Flyash%20can%20be%20used%20for,macro%20and%20micronutrients%20for%20plants
Sheoran VA, Sheoran S, Poonia P (2010) Role of hyperaccumulators in phytoextraction of metals from contaminated mining sites: a review. Crit Rev Environ Sci Technol 41(2):168–214. https://doi.org/10.1080/10643380902718418
Shi ZM, Xu L, Hu F (2014) A hierarchic method for studying the distribution of phenanthrene in Eisenia fetida. Pedosphere 24:743–752. https://doi.org/10.1016/S1002-0160(14)60061-8
Shi Z, Tang Z, Wang C (2017) A brief review and evaluation of earthworm biomarkers in soil pollution assessment. Environ Sci Pollut Res 24:13284–13294. https://doi.org/10.1007/s11356-017-8784-0
Shi Z, Tang Z, Wang C (2019) Effect of phenanthrene on the physicochemical properties of earthworm casts in soil. Ecotoxicol Environ Saf 168:348–355. https://doi.org/10.1016/j.ecoenv.2018.10.032
Shi Z, Liu J, Tang Z, Zhao Y, Wang C (2020) Vermiremediation of organically contaminated soils: concepts, current status, and future perspectives. Appl Soil Ecol [Internet]. https://doi.org/10.1016/j.apsoil.2019.103377
Sikka R, Kansal BD (1995) Effect of fly ash application on yield and nutrient composition of rice, wheat and on pH and available nutrient status of soil. Biores Technol 51:199–203. https://doi.org/10.1016/0960-8524(94)00119-L
Singh SN, Kushreshtha K, Ahmed KJ (1997) Impact of fly ash soil amendment on seed germination, seedling growth and metal composition of Vicia faba L. Ecol Eng 9:203–208. https://doi.org/10.1016/S0925-8574(97)10004-0
Singh JS, Pandey VC (2013) Fly ash application in nutrient poor agriculture soils: impact on methanotrophs population dynamics and paddy yields. Ecotoxicol Environ Saf 89:43–51. https://doi.org/10.1016/j.ecoenv.2012.11.011
Singh S, Gond D, Pal A, Tewary BK, Sinha A (2011a) Performance of several crops grown in fly ash amended soil. In: World coal ash conference—May 9–12, 2011, Denver, CO, USA. http://www.flyash.info/
Singh JS, Pandey VC, Singh DP (2011b) Coal fly ash and farmyard manure amendments in dry-land paddy agriculture field: effect on N-dynamics and paddy productivity. Appl Soil Ecol 47:133–140. https://doi.org/10.1016/j.apsoil.2010.11.011
Singh K, Pandey VC, Singh B, Patra DD, Singh RP (2016a) Effect of fly ash on crop yield and physico-chemical, microbial and enzyme activities of sodic soils. Environ Eng Manage J 15(11):2433–2440
Singh S, Bhat SA, Singh J, Kaur R, Vig AP (2016b) Vermistabilization of thermal power plant fly ash using Eisenia fetida. J Ind Pollut Cont. Retrieved on 20 March 2021 from https://www.icontrolpollution.com/articles/vermistabilization-of-thermal-power-plant-fly-ash-using-eisenia-fetida-.php?aid=81970
Sinha RK, Bharambe G, Ryan D (2008) Converting wasteland into wonderland by earthworms-a low-cost nature’s technology for soil remediation: a case study of vermiremediation of PAHs contaminated soil. Environmentalist 28:466–475. https://doi.org/10.1007/s10669-008-9171-7
Sinha RK, Herat S, Valani D, Chauhan K (2010) Earthworms-The environmental engineers: review of vermiculture technologies for environmental management and resource development. Int J Glob Environ 10:265–292. https://doi.org/10.1504/IJGENVI.2010.037271
Steffen GPK, Antoniolli ZI, Steffen RB, Jacques RJS, dos Santos ML (2013) Earthworm extraction with onion solution. Agric Ecosyst Environ Appl Soil Ecol 69:28–31. https://doi.org/10.1016/j.apsoil.2012.12.013
Stromberger ME, Keith AM, Schmidt O (2012) Distinct microbial and faunal communities and translocated carbon in Lumbricus terrestris drilospheres. Soil Biol Biochem 46:155–162. https://doi.org/10.1016/j.soilbio.2011.11.024
Tarannum A, Khan AA, Diva I, Khan B (2001) Impact of fly ash on hatching, penetration and development of root-knot nematode, Meloidogyne javanica on chickpea (Cicer arietinum L.). Nematol Mediterr 29:215–218
Tiunov AV, Scheu S (1999) Microbial respiration, biomass, biovolume and nutrient status in burrow walls of Lumbricus terrestris L. (Lumbricidae). Soil Biol Biochem 31:2039–2048. https://doi.org/10.1016/S0038-0717(99)00127-3
Tripathy A, Sahu RK (1997) Effect of coal fly ash on growth and yield of wheat. J Environ Biol 18(2):131–135. https://www.osti.gov/etdeweb/biblio/488690
Usmani Z, Kumar V (2017) The implications of fly ash remediation through vermicomposting: a review. Nature Environ Pollut Technol 16(2):363–374. http://www.neptjournal.com/upload-images/NL-60-5-(3)B-3395com.pdf
Valckx J, Govers G, Hermy M, Muys B (2011) Optimizing earthworm sampling in ecosystems. In: Karaca A (ed) Biology of earthworms. Springer, Berlin, Heidelberg, pp 19–38
West TO, McBride AC (2005) The contribution of agricultural lime to carbon dioxide emissions in the United States: dissolution, transport and net emissions. Agric Ecosys Environ 108:145–154. https://doi.org/10.1016/j.agee.2005.01.002
Wong JWC, Wong MH (1990) Effects of fly ash on yields and elemental composition of two vegetables, Brassica parachinensis and B. chinensis. Agric Ecos Environ 30:251–264. https://doi.org/10.1016/0167-8809(90)90109-Q
Yeledhalli N, Prakash S, Gurumurthy S, Ravi M (2007) Coal fly ash as modifier of physico-chemical and biological properties of soil. Karnataka J Agric Sci 20(3):531–534. http://14.139.155.167/test5/index.php/kjas/article/viewFile/899/892
Zaborski ER (2003) Allyl isothiocyanate: an alternative chemical expellant for sampling earthworms. Appl Soil Ecol 22:87–95. https://doi.org/10.1016/S0929-1393(02)00106-3
Zhang S, Hu F, Li H, Li X (2009) Influence of earthworm mucus and amino acids on tomato seedling growth and cadmium accumulation. Environ Pollut 157:2737–2742. https://doi.org/10.1016/j.envpol.2009.04.027
Zibilski JJ, Alva AK, Sajwan KS (1995) Fly ash. In: Reckcigi JE (ed) Soil amendments and environmental quality. CRC press Inc., Boca Raton, FL, pp 327–363. https://journals.lww.com/soilsci/Citation/1996/07000/Soil_Amendments_and_Environmental_Quality.7.aspx
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2023 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Dwibedi, S.K., Pandey, V.C. (2023). Fly Ash Management Through Vermiremediation. In: Pandey, V.C. (eds) Bio-Inspired Land Remediation. Environmental Contamination Remediation and Management. Springer, Cham. https://doi.org/10.1007/978-3-031-04931-6_10
Download citation
DOI: https://doi.org/10.1007/978-3-031-04931-6_10
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-04930-9
Online ISBN: 978-3-031-04931-6
eBook Packages: Earth and Environmental ScienceEarth and Environmental Science (R0)