Abstract
The Hawaiian monk seal (Neomonachus schauinslandi) is among the world's rarest pinnipeds, with a population abundance of around 1,400 individuals. This species fills a unique ecological niche as the only tropical phocid. Since their listing as depleted under the Marine Mammal Protection Act and endangered under the Endangered Species Act, there has been substantial effort to recover the species. Ethological research has played an essential role in the conservation efforts for Hawaiian monk seals, contributing to our understanding of the seals' interactions with their environments and the threats they face. In some cases, behavioral patterns have increased monk seals' risk of harm, such as foraging in nearshore waters, increasing the potential for interactions with fishing gear. In other instances, management agencies have been able to exploit behavioral characteristics to design successful conservation interventions. For example, given their limited tendency for dispersal, Hawaiian monk seal pups can be safely translocated to areas that increase their chance of survival. This chapter reviews over four decades of research dedicated to Hawaiian monk seals, with particular attention to the ethological insights revealed in areas of reproductive behavior, social behavior, foraging behavior, and dispersal behavior. We highlight the conservation efforts ranging from animal rescues to translocations that have relied upon this information to work toward the recovery of this endangered seal.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Hawaiian monk seal
- Conservation
- Reproductive behavior
- Social behavior
- Foraging behavior
- Dispersal behavior
1 Overview and Conservation Status
Hawaiian monk seals (Neomonachus schauinslandi) are part of an ancient phocid lineage that includes the Mediterranean monk seal (Monachus monachus; endangered) and the Caribbean monk seal (Neomonachus tropicalis; extinct, last seen in 1952). It is estimated that the Hawaiian monk seal diverged from Caribbean seals between 3–11 million years ago after crossing over the submerged Isthmus of Panama into the Pacific Ocean (Fyler et al. 2005; LeDuc 2009). While the bulk of this chapter will focus on Hawaiian monk seals, we have included information about the Mediterranean monk seal in Appendix 16.7.
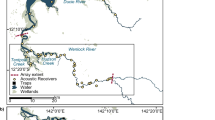
Hawaiian monk seals range throughout the Hawaiian Archipelago and are characterized as a metapopulation with semi-isolated subpopulations distributed among a number of islands and atolls (Antonelis et al. 2006). Across this >2,500 km archipelago, there is considerable variation in breeding sites inhabited by monk seals (Fig. 16.1). Six islands/atolls (Kure Atoll to French Frigate Shoals) in the Northwestern Hawaiian Islands (NWHI) comprise the most-studied breeding sites for Hawaiian monk seals. The largest of the NWHI landmasses, Laysan Island, has only 8 km of shoreline, whereas many of the atolls host several small islets totaling less than 1 km2 of land area. While the land area is small, these sites are surrounded by large lagoons and fringing reefs, and a series of submerged banks and seamounts extends the underwater habitat for monk seals. At the southeastern end of the NWHI lie Mokumanamana (Necker) Island and Nihoa Island, volcanic remnants with steep cliffs and small beach areas consisting of rock shelves and minimal sand beaches. These islands form something of a nexus between the primary breeding sites in the NWHI and the main Hawaiian Islands. The main Hawaiian Islands (MHI), the eight islands populated by humans, are large islands (50–430 km shoreline) with mountainous terrestrial terrain, beaches of sand and rock, and considerable human density (just under 1,000,000 people on O’ahu; 2010 US Census).
Monk seals likely inhabited the entire Hawaiian Archipelago prior to human settlement, with the population in the MHI being extirpated shortly after the arrival of Polynesians (~800–1200 CE). Archaeological evidence indicates that monk seals were present in the MHI prior to European contact in 1778, though their limited role in Hawaiian culture or legends suggests they were rare in the populated islands (Rosendahl 1994). Newspapers reference occasional monk seal sightings on the populated islands throughout the 1800–1900s (Watson et al. 2011). Commercial sealing expeditions reduced the remainder of the Hawaiian monk seal population in the NWHI to near extinction in the mid-1800s, and guano miners, bird hunters, and whalers further depleted the population by the early 1900s (Cobb 1902; Kenyon and Rice 1959).
1.1 Conservation Status and Threats
The first formal (aerial) surveys of the Hawaiian monk seal population counted a minimum of 1,013 seals on land in the summer of 1957 (Kenyon and Rice 1959); this likely represented a population of closer to 3,000 seals (accounting for haulout correction factors later developed; Harting et al. 2017). Follow-up surveys in the 1960s and 1970s suggested a 50% decline in abundance since the 1950s (Kenyon 1973; DeLong et al. 1976; Johnson et al. 1982). This decline led Hawaiian monk seals to be the first species designated depleted under the Marine Mammal Protection Act in 1976, as well as endangered under the Endangered Species Act. In more recent years, monk seal surveys have been standardized and conducted annually, revealing subtler variation in abundance trends over time. Population counts in the NWHI declined 4.2% per year from 1985–1993; this decline slowed to 1.9% per year from 1993–2003 (Carretta et al. 2004).
Meanwhile, monk seal sightings in the MHI remained relatively rare until the mid-1990s, but rebounded considerably in the early 2000s (Baker and Johanos 2004). By 2013, the population could be estimated across the entire archipelago, and was showing signs of a stabilizing trend (Baker et al. 2016b). Recent data have shown promising signs of positive trends, with the range-wide population increasing at a rate of 2% per year from 2013–2019 (NMFS unpublished). In light of this positive outlook and decisive management actions, Hawaiian monk seals were moved from critically endangered to endangered on the IUCN Red List in 2015 (IUCN 2015). The population in 2019 was just over 1,400 individuals with about 1,100 seals residing in the remote NWHI and about 300 in the MHI (Hawaiian Monk Seal Research Program 2019). Within the overall positive trend, local demographics and age-specific survival rates continue to vary between sites, with both juvenile survival and population growth rates generally lower in the NWHI than MHI (Baker et al. 2011b).
As with localized population trends, particular threats vary for monk seals across their range. Poor juvenile survival associated with prey limitation has been suggested as the primary proximate cause for the decline of monk seals in the NWHI in recent decades (Craig and Ragen 1999). Other significant threats in the NWHI include entanglement in marine debris (Henderson 2001), male aggression leading to female injury and death (particularly at Laysan Island; Hiruki et al. 1993b, Johanos et al. 2010), shark predation on seal pups (particularly at French Frigate Shoals; Gobush and Farry 2012), and the threat of island disappearance with sea-level rise (Baker et al. 2006). Meanwhile, threats in the MHI tend to be more anthropogenic in nature, with intentional seal killings, direct interactions with fisheries (hook ingestion or net entanglement), and disease (particularly infection with the parasite Toxoplasma gondii, spread by cats) comprising the leading causes of death in the MHI (Harting et al. 2020). Human disturbance and pollution also pose threats in the MHI (Baker et al. 2011b; Lopez et al. 2014). The current Recovery Plan for Hawaiian Monk Seals (a conservation planning document required for all ESA-listed species; National Marine Fisheries Service 2007) prioritizes activities to address threats throughout the monk seal range: (1) improve survivorship of females of all ages, particularly juveniles and yearlings in the NWHI, (2) maintain or expand existing field efforts, (3) ensure natural recovery of the Hawaiian monk seal in the MHI, and (4) reduce the probability of inadvertent introduction of infectious diseases into the Hawaiian monk seal population.
1.2 Modes of Research and Ethological Observation
Monk seals spend approximately one-third of their time hauled out on land for parturition, nursing, molting, and resting (Cahoon 2011; Harting et al. 2017, Wilson et al. 2017a). This time on land offers valuable opportunities for the observation that forms the backbone of population monitoring and ethological data (Fig. 16.2). The National Marine Fisheries Service Hawaiian Monk Seal Research Program (NMFS, HMSRP) began consistently tagging weaned monk seal pups in the 1980s (Antonelis et al. 2006). These tagged pups are resighted throughout their lifetimes using the applied flipper tags as well as natural markings or other identifiable characteristics documented in a digital photography database (Harting et al. 2004). Resighting efforts and methods vary across the monk seal range. In the remote NWHI, seals are surveyed over the course of 2–5-month field camps (established nearly every year since 1982; Baker and Johanos 2004; Johanos et al. 2014). Mokumanamana and Nihoa Islands are typically surveyed 1–4 times per year in conjunction with research missions to the NWHI. Unlike in the NWHI, where a relatively large number of seals are concentrated on small islands, in the MHI a small number of seals are distributed over extensive (and often inaccessible) coastlines. Thus, seals in the MHI are monitored by networks of volunteers and public seal watchers as well as biologists, yielding year-round coverage that provides a dataset sufficient to determine a minimum abundance in this region. Data from all sites and site-specific survey methods are combined for estimating population abundance and vital rates throughout the full Hawaiian monk seal range (Baker et al. 2016b).
In addition to tracking population trends, field observations are a primary means of collecting ethological data on monk seals. The timing of field camps is aligned to maximize the observation of pupping and breeding patterns to understand reproductive biology (Johanos et al. 1994). Throughout field camps in the NWHI, and some focused studies in the MHI, animal behavior and social interactions are observed at haulout areas (Johanos et al. 2010; Robinson et al. 2018). Many aspects of seal behavior require observation at sea. To understand diving and foraging behavior, we have employed a number of different technologies such as time-depth-recorders (DeLong et al. 1984), satellite and GPS tags (Stewart et al. 2006; Cahoon 2011), and seal-borne video cameras (Parrish and Littnan 2007; Wilson et al. 2017a).
An understanding of Hawaiian monk seals' behavior in the context of their ecology and survival threats has allowed the NMFS HMSRP to design conservation interventions aimed at enhancing the survival of individuals and ongoing recovery of the species. The cumulative impact of these intervention efforts has been one of the cornerstones of progress in Hawaiian monk seal recovery, with up to 32% of the population comprised of seals benefitting from such interventions (Harting et al. 2014). Throughout this chapter, we review the science related to Hawaiian monk seal behavior, with a particular focus on highlighting how insights from ethological research have informed conservation actions that benefit monk seals.
2 Reproductive Behavior—Influences on Reproduction and Survival
2.1 Reproductive Biology
Hawaiian monk seals typically give birth on beaches near shallow water that offers protection from rough seas and sharks. Females may pup at sites different from their most-used haulout locations, and typically show up to their pupping site immediately prior to pupping (Johanos et al. 1994). Monk seals exhibit asynchronous mating (Kenyon and Rice 1959; Johanos et al. 1994), a likely result of their tropical-subtropical environment lessening some of the pressures selecting for synchronous breeding in many other pinniped species. Resources are consistently available to support gestating females or newly weaned seals throughout the year, and seasonal thermal gradients are less extreme than in higher latitudes. While births have been observed throughout all months of the year, there is a (protracted) seasonality to monk seal breeding (Kenyon and Rice 1959; Johanos et al. 1994). The pupping season is concentrated from late February through early June in the NWHI (Johanos et al. 1994), and April–July in the MHI (Robinson et al. 2020). A similarly protracted breeding season occurs in the most southern population of Mediterranean monk seals (Appendix 16.7).
As capital breeders (Chap. 8), female Hawaiian monk seals remain hauled out or near shore with the pup and fast throughout the ~ 40 day nursing period, providing nutrients for pups to gain 50–80 kg (approximately tripling their birth mass). This is a contrast to Mediterranean monk seals, with females nursing for longer duration and leaving periodically to feed (Appendix 16.7). When the Hawaiian monk seal female's resources are depleted, weaning occurs abruptly as the female leaves the pup, swimming offshore to forage. Females in the MHI exhibit longer nursing periods than those in the NWHI (43.7 vs. 39.0 days in the NWHI, 12% longer nursing for MHI pups; Johanos et al. 1994; Robinson et al. 2020). Despite this longer nursing period, MHI females regain body condition more quickly and are able to undergo resource-intensive molting sooner after weaning than their NWHI counterparts (54 days post-weaning /98 days post-pupping vs. 66/105 days for northwestern females; Robinson et al. 2020). This increase in nursing period likely provides a significant nutritional benefit to MHI pups, consistent with reports of larger size-at-weaning and accelerated post-weaning growth in MHI versus NWHI pups (Baker and Johanos 2004; Baker et al. 2014).
Critical factors of the reproductive cycle, such as estrus and gestation, are less flexible than nursing and molting intervals. The mean inter-birth interval of 381–382 days has been recorded with remarkable consistency across studies spanning great geographic and temporal range (NWHI; Wirtz 1968, Johanos et al. 1994, MHI; Robinson et al. 2020). The consistency of inter-birth intervals suggests that polyestrous cycling described in a single captive monk seal (Pietraszek and Atkinson 1994) is not typical in wild seals. Estrus is believed to occur after weaning a pup and before molting (or ~ 1.5 month before molting for females without a pup in a given year). Because mating activity occurs offshore and is rarely observed, the timing of mating injuries (detailed in Sect. 16.2.3) is a critical factor in inferring time of estrus. Females hauled out on shore are typically attended by a male during the interval between weaning a pup and molting (Johanos et al. 1994). Injuries were observed an average of 26 days after the end of maternal investment, with the rate of injury decreasing as females approached molt, suggesting a consistent relationship with termination of nursing and onset of the next estrous cycle (Johanos et al. 1994).
Reproductive output varies across breeding sites and among individuals. Site-specific reproductive rates closely follow body condition and trends in growth, with age at primiparity (first pupping) aligning with the age at which females reach adult size (Harting et al. 2007). Females in the MHI tend to have robust body condition and pup at a younger age than NWHI females (Baker and Johanos 2004). The reproductive rate for MHI seals exceeds 0.70 for all ages 8–18 years (after accounting for unobserved pups; Robinson et al. 2020). Among closely studied NWHI sites, Laysan Island has the highest reproductive rate (0.73) and earliest mean age at primiparity among NWHI sites (7.4 years), whereas French Frigate Shoals females produce their first pups at an older age (~10 years) and exhibit lower reproductive rates (0.61). Prime reproductive years occur from ages 10–15, with reproductive senescence becoming evident around 15–20 years of age (Harting et al. 2007). On an individual level, some females consistently maintain either higher or lower reproductive rates throughout their lives, but we have found no relationship between reproductive investment and female survival (Harting et al. 2007).
2.2 Mom and Pup Interactions
Throughout the nursing period, mothers and pups spend time nursing and resting while hauled out and swim together in shallow waters near the birth site (Fig. 16.3). Swimming forays typically do not range far or deep, and there is no evidence of mother–pup pairs foraging together. Mothers and their offspring do not appear to maintain close interactions after weaning (NMFS HMSRP unpublished database).
Nursing female monk seals tend to be particularly asocial. Female monk seals occur in low density compared to most land-breeding seals, with densities as low as 1.5 females per 1000 m2 on East Island, a small sand islet within French Frigate Shoals, and just 0.5 females per 1000 m2 on Laysan, the largest of the NWHI sites (Boness et al. 1998). Even at such low densities, animals still showed avoidance of neighbors with daily nearest neighbor distances (mean 27.0 m; range 14.2–52.6 m) greater than expected by chance (Boness et al. 1998). Nursing females are intolerant when approached by other females sharing a nursing beach or males seeking attention (Johanos et al. 1994). When aggressive interactions occur between neighboring nursing mothers, it is common for the pups to be mixed up in the melee, often leading to switches in the mother–pup pairs (Boness 1990).
Fostering is common in Hawaiian monk seals and is described in a number of studies (Alcorn and Henderson 1984; Boness 1990; Johanos et al. 1994; Boness et al. 1998). While females begin nursing their biological pup after birth, many mothers foster one or more pups (in sequence, not simultaneously) and may not reunite and resume nursing their biological pup. An early study found fostering rates as high as 53% (Laysan Island) to 90% (East Island at French Frigate Shoals) (Boness et al. 1998). The average female spent 34% (range 1–91%) of her nursing period fostering an average of 2.3 pups (max observed 5; Boness 1990). While the most common cause of fostering is pup-switching between females, pups may also seek out a foster mother to nurse within a few days after being weaned. One extreme example of this is a pup, who after being weaned by its mother, proceeded to nurse from a foster mother that had lost her pup early, finally weaning after a total of 68 days of nursing from two mothers (Alcorn and Henderson 1984). Fostering is well documented in some pinniped species including Mediterranean monk seals (Appendix 16.7) and more distantly related species (elephant seals; Riedman and Le Boeuf 1982, harbor seals; Schaeff et al. 1999).
The frequency of pup-switching and fostering in Hawaiian monk seals calls into question the monk seal's ability to recognize individuals or distinguish close kin such as mothers or pups. Research on vocal recognition is inconclusive. An early study found that, despite significant differences in vocal attributes among pups, discriminant function analysis failed to distinguish individuals based on vocalizations (Job et al. 1995). Playback experiments in the field also indicated that females did not discriminate between the voices of their own pup versus another, leading to the conclusion that females were unable to recognize their pups by voice (Job et al. 1995). An ongoing study has found stronger distinction between vocal attributes, especially the “bah” and “mah” calls used by pups (Chaudun 2018). This study found that their discriminant function correctly classified 54% of individual calls, suggesting a better-than-random chance of vocally distinguishing individuals (Chaudun 2018). Further research may determine whether this discriminant ability is realized in the wild.
Because there is a strong relationship between girth at weaning and survival (Baker 2008), there can be winners and losers in a pup-switch. If pups of substantially different ages switch, the older pup may get an extended nursing period from the mother with more days left to nurse, while the younger pup might be weaned early by the mother whose resources are more depleted. A prematurely weaned pup that does not attain sufficient girth stands a poor chance of survival (Baker 2008). Given the importance of females in maximizing the reproductive potential of the population, it has been the policy of the HMSRP to intervene in cases where a female pup is disadvantaged by a male pup in a pup-switch (i.e., the female pup ends up with the mother with less milk left to give). Field biologists remove the switched pups from their respective foster mothers and place them back next to their original mothers. There may also be cases in which a mother and pup become separated, but no other nursing mother fosters the pup. In these cases, field biologists locate the mother and place the pup in her proximity. In many cases nursing resumes and the pup weans normally, but in some cases a mother may repeatedly reject her pup.
2.3 Male and Female Interactions
Hawaiian monk seals are polygamous, and mating occurs throughout the year. As a result, the breeding and pupping aggregations common in other pinniped species are not seen in monk seals. Males do not maintain specific breeding territories nor monopolize particular females over the long term, but they may haulout next to available females (presumably to attain a beneficial position to follow her to sea for mating), and spar with other males that approach. Mating occurs at sea and is rarely observed, but occasional observations have documented males holding a female with teeth and foreflippers in an attempt to mount her (Wirtz 1968; Hiruki et al. 1993a; Johanos et al. 1994). In some cases, termed multiple male aggression, more than one male may attempt to mount a single female (or an immature seal of either sex; Fig. 16.4a). Multiple male aggression events observed in nearshore waters have involved from 2–32 males, and have lasted from moments to >8 h (Johanos et al. 2010). Mating typically leaves females with injuries that can range from minor scratches or punctures to subcutaneous hemorrhage, or severe gaping wounds covering much of the dorsum (Fig. 16.4b), with the more severe injuries being associated with multiple male aggression incidents (Hiruki et al. 1993a). Wounds associated with mating can be an indicator of breeding activity, or an essential indicator of imbalances in social structure that pose severe risks to female survival.
Multiple male aggression reached particularly dangerous levels on Laysan Island in the 1980s. Approximately 70% of adult females on Laysan exhibited scars associated with injuries from multiple male aggression, and, over the same time, 45% of adult females at French Frigate Shoals had such scars (Hiruki et al. 1993a). The seasonal timing when females appear with severe wounds is similar to when females appear with more minor mating injuries (e.g., scratches), suggesting that both are associated with higher male attraction during estrus (Johanos et al. 1994). A study of hormone levels in captive male monk seals suggests that testosterone varies seasonally (peaking in summer, and declining when animals approach molt in the fall), which would support heightened male aggression during seasonal breeding (Atkinson and Gilmarti 1992). While seeking mating opportunities appears to be the impetus of multiple male aggression, animals other than adult females can become targets. During the peak years of aggression at Laysan, while 70% of observed mounting injuries were incurred by adult females, the other 30% were incurred by males or immature females for which estrus could not have been a factor (Johanos et al. 1994).
Injurious mating aggression has been documented in other phocids (Le Boeuf and Mesnick 1991; Mesnick and Le Boeuf 1991; Rose et al. 1991), but it seems to be particularly prevalent and problematic in Hawaiian monk seals. This could be due to differences in mating systems and female availability, or impacts may be exacerbated by small population sizes. The severity and prevalence of injuries from multiple male aggression has the potential to substantially impact the monk seal population by removing breeding females. During a peak period of aggressive activity on Laysan (1983–89), 87% of adult females that died had been seriously injured (Hiruki et al. 1993b). Post mortem examinations confirmed that the majority of females dying after male-inflicted injury were in estrus (Atkinson et al. 1994). Injuries that occur shortly prior to pupping or during nursing also have the potential to impact survival of the female's pup (Hiruki et al. 1993b). Females injured early in their breeding cycle typically had sufficient time to heal and regain body condition prior to pupping so that, provided mortality was avoided, wounding had little impact on female reproductive rate, nursing period, or pup survival (Hiruki et al. 1993b).
While monk seals do not have a rigid dominance hierarchy, males observed in aggressive interactions were likely to exhibit more subdominant behaviors. They were less likely to be sighted regularly hauled out, less likely to be sighted hauled out in proximity to a female, more likely to be sighted cruising (swimming along the shoreline, presumably looking for a female), and more likely to be sighted throughout many island sectors (wider/more varied space use). Only about 10–20% of males were classified as dominant (spending more time hauled out on land, and in proximity to adult females) in a given year. Given the association of multiple male aggression with male-biased sex ratios, it is likely that dominant males become overwhelmed by the more numerous competitors. During the peak period of aggression on Laysan Island, male:female ratios were ~2:1, but could be locally elevated due to animal haulout preferences. Space use differed between dominant and subdominant males, with dominant males hauling out primarily on the northeastern side of Laysan where adult females concentrated. Meanwhile, nondominant males had a more generalized distribution spanning northeast to southwest. Thus, the southwestern side of the island developed both an extreme male-biased sex ratio (as much as 25:1), and a high concentration of subdominant males. Females hauling out on these southwestern beaches were most likely to be observed with severe injuries (Johanos et al. 2010).
2.4 Conservation Interventions to Mitigate Threats of Male Aggression
Long-lasting population impacts of losing breeding females provided a strong impetus for human managers to mitigate the threat of female mortality from mounting injuries. One early avenue that was explored was the treatment of males with gonadotrophin-releasing hormone agonist to lower testosterone levels, and thus aggression. Atkinson et al. (1993) showed that testosterone was suppressed for 7–8 weeks after treatment, and recommended treatment at 2-month intervals to control male aggression, which was unrealistic in the field setting for wild populations. The association of multiple male aggression incidence with male-biased sex ratios and over-abundance of subdominant males led to the suggestion that adjustment of sex ratios to re-balance social structures within the population could provide a solution. Simulation models that evaluated different management scenarios according to outcomes for overall population size, number of breeding females, and sex ratio supported the removal of males to balance sex ratios (Starfield et al. 1995). Behavioral profiles were developed for males on Laysan Island to help identify males for removal that would (1) maximize the benefit of mitigating harmful injuries to females, and (2) be less disruptive to the social structure on the island (Johanos et al. 2010). From 1984–1994, 37 males were removed from Laysan Island (translocated either to Johnston Atoll or the main Hawaiian Islands), reducing the male sex ratio from 2.1 to 0.9. Following these translocations, female mortality decreased from 4.1% to 0.3% and non-lethal injury rates decreased from 10.9% to 2.3% (non-lethal injury rates went from 10.9%–2.3%; Johanos et al. 2010).
3 Social Behavior—Influences on Disease Spread and Mitigations
Hawaiian monk seals are not highly social (Kenyon and Rice 1959). Seals are often solitary, both when foraging and hauled out, which may in part be due to the fact that they naturally occur at low densities. An island or atoll in the NWHI may be shared by only 50–250 animals, even at the most populous breeding sites. At Laysan Island, the breeding population of ~250 seals is spread across just 8 km of coastline, resulting in densities of ~25 seals/km (NMFS HMSRP unpublished database). In the MHI, the population of ~300 seals is spread over >1500 km of coastline on eight islands, or just ~0.2 seals/km. A notable aggregation might be tens of animals hauled out on one beach in the NWHI, while in the MHI, well-known haulout “hotspots” might only host 3–6 seals (NMFS HMSRP unpublished database). While seals sometimes aggregate around a favorable haulout location, they appear to neither seek out interaction nor exhibit territorial behaviors (Kenyon and Rice 1959; though see reproductive behavior described in Sect. 15.2).
Observations of at-sea interactions are rare, but seal-mounted camera studies have recorded occasional interactions between seals underwater. Most of these interactions have been curious and brief, not demonstrating either agonistic or cooperative behavior (Parrish and Littnan 2007; Wilson et al. 2017a). Some video-sound recordings have detected underwater vocalizations, but it is unknown if the purpose of these vocalizations was to seek out nearby animals or deter them from a possible territory (CritterCam video footage; NMFS HMSRP unpublished instrument data). A number of telemetry studies have shown a considerable overlap in space use at sea in both the NWHI (Parrish and Abernathy 2006; Stewart et al. 2006; Curtice et al. 2011) and MHI (Littnan et al. 2006; Cahoon 2011, Wilson et al. 2017b), indicating the potential for interaction to occur.
Interactions among animals while hauled out have been more closely studied. Since the early 1980s, seals' behavior on Laysan Island and other sites has been recorded on breeding, aggression, and dominance interactions (Johanos et al. 1994; 2010). These data were later analyzed in a social network context to estimate the contact rates between seals (Baker et al. 2016a). Observations of seals on O’ahu were made to document types of contact and interaction rates in a lower-density population (Robinson et al. 2018). These studies revealed the subtle patterns of interactions within monk seals' low degree of sociality (Fig. 16.5).
Contact between Hawaiian monk seals may vary from merely lying in close proximity on a beach (a) to intensive interactions during play or aggressive incidents (b). A contact network of seals using one O’ahu beach shows the varying degree of contact between individuals, with male seals (blue) having the highest contact levels (line weight indicates proximity vs. intensive contact, line distance indicates relative frequency)
Most associations between monk seals are short-lived. On Laysan Island, most seal pairings (82%) occurred only once over a 5-month study period, and two seals were seldomly recorded together five or more times (2%, primarily mother–pup pairs; Baker et al. 2016a). The most common type of association between seals involves resting nearby (Fig. 16.5a); of the interactions observed on O’ahu, 78% of associations were based solely on haulout proximity (Robinson et al. 2018). Direct interactions were twice as likely to be casual sniffing or nudging (14.5% of total interactions), with aggressive interactions (sparring or biting) making up only 7.5% of the total (Fig. 16.5b; Robinson et al. 2018).
Seals do not mix randomly, but rather show preferential interaction among age and sex classes. Mothers and pups have a highly elevated rate of interaction throughout the nursing period (Johanos et al. 1994), but nursing females tend to avoid each other (Boness et al. 1998, Baker et al. 2016a). Males show more social interaction than females. For example, males exhibit a “cruising” behavior that involves swimming near the shoreline, apparently searching for females, and frequently come to shore and have brief interactions with other seals hauled out, which can raise their contact rate with numerous animals (Baker et al. 2016a). Males are likely to spar with one another, especially to attend a female; males may fight to displace one another or maintain their position near females (Johanos et al. 2010). On Laysan Island, subadult males were the most interactive, with interaction rates approaching mother–pup pairs (Baker et al. 2016a). On O’ahu, where there is a smaller population and fewer subadult males, adult males had the highest rate of interactions, and specifically aggressive interactions (Robinson et al. 2018). This difference is likely due to differences in sex ratio and the age structure of the given population. At the time of observation, Laysan Island had a highly male-biased sex ratio with numerous subadult and adult males, heightening the level of competitive behavior among (likely subdominant) subadult males. Meanwhile, on O’ahu, with few subadults in the local population, it appears that adult males showed more competitive interactions among themselves.
3.1 Implications for Disease Spread and Vaccination
For endangered species existing in small populations, such as Hawaiian monk seals, a disease outbreak could have disastrous consequences. Understanding social behavior in the context of potentially infectious contact rates is particularly important for assessing disease risk. While they do not show social aggregations common in some pinniped species, monk seals encounter and interact with enough seals to effectively spread disease through a small population. Hawaiian monk seal researchers have used behavioral observations and social network analysis to calculate contact rates, model disease spread, and design risk mitigations (Baker et al. 2016a; Baker et al. 2017; Robinson et al. 2018). Morbillivirus has been the focus of this work because viruses in this family, specifically phocine distemper virus (PDV) and canine distemper virus (CDV), have caused mass die-offs of other phocids (Grachev et al. 1989; Heide-Jørgensen et al. 1992; Kennedy et al. 2000; Jensen et al. 2002). Previous epidemiological screening has shown that Hawaiian monk seals were naïve to morbilliviruses (including canine distemper virus, phocine distemper virus, dolphin morbillivirus, porpoise morbillivirus) in both the MHI (Littnan et al. 2006) and NWHI (Aguirre et al. 2007). Due to their isolation in the Pacific and depauperate genetic diversity (Schultz et al. 2009), there is concern that Hawaiian monk seals could be highly susceptible if exposed to morbillivirus. The epidemiological models constructed for morbillivirus are likely to apply to other diseases directly spread between monk seals in close proximity (as by respiratory/aerosol substance). Monk seals face several disease threats other than morbillivirus (Barbieri et al. 2016; Harting et al. 2020) and some, like toxoplasmosis (transmitted by oocysts contaminating the environment via cat feces), are not related to direct contact among seals and cannot be prevented through vaccination.
Contact networks based on observed monk seal interactions indicated that networks were not subdivided into cliques despite heterogeneous contact structure. That is, all seals using an island were connected in a single component indicating pathogens could spread from one seal to another (Baker et al. 2016a; Robinson et al. 2018). Social connectivity was higher for seals in denser populations in the NWHI compared to those in the MHI, though perhaps not as different as expected given the disparity in seal density. Seals on Laysan had an average of 15 connections in the network of 250 seals using this island with 8 km of shoreline (Baker et al. 2016a), while seals on O’ahu had a mean of 10 contacts in a network of just 45 seals using 365 km of coastline (Robinson et al. 2018). The slight relationship between population density and contact is likely indicative of (a) numerous animals frequenting a few favored haulout locations, and (b) some animals such as subadult/adult males cruising the shoreline seeking out other seals, thus increasing connectivity throughout the population.
While connectivity was high among seals using a single island, the population's spatial subdivision works to limit contact among islands. Outbreaks simulated in a single admixed population grew in 91% of simulations, whereas in spatially subdivided populations (based on the MHI, with relatively frequent movement between nearby islands), the outbreak only grew in 38% of simulations (Baker et al. 2017). A recognition of the population's accurate spatial structure also helps in assessing the percentage of the population that needs to be vaccinated. Achieving herd immunity (in 95% of simulated scenarios) would require vaccinating 86% of the admixed population, but could be achieved after vaccinating just 60% of the spatially structured population (Baker et al. 2017).
3.2 Vaccination as a Conservation Intervention
Morbillivirus is one of the few pathogens for which vaccination is a management option. The American Association of Zoological Veterinarians recommended a recombinant vaccine to CDV (Purevax, Merial), which has been used extensively in zoological collections (Bronson et al. 2007) and has been tested for safety in Hawaiian monk seals (Yochem unpublished). Epidemic simulations showed that vaccination in response to an outbreak would have little efficacy in stemming the extent of the outbreak, primarily due to pathogens spreading faster than vaccines could be administered and confer immunity (Baker et al. 2017). Thus, in 2016, NOAA initiated a prophylactic vaccination program to protect Hawaiian monk seals against the possibility of morbillivirus exposure. During the pilot effort, 21 monk seals on O’ahu were successfully vaccinated (2 injections, 3–5 weeks apart), and showed no ill effects (Fig. 16.6; Robinson et al. 2018). Since that time, vaccination efforts have been expanded to all segments of the population in the NHI and NWHI, with work continuing to reach vaccination numbers sufficient for herd immunity. The Hawaiian monk seal system is particularly well-suited to protection through vaccination; nearly all pups are handled each year for flipper tagging, thus there is a good opportunity to vaccinate the majority of individuals in each new cohort of seals. After the 2019 field season, enough seals had been vaccinated to support herd immunity goals in 60–80% of simulations in the NWHI given the spatial structure of the population. This level of population protection was possible only after understanding social interactions and behavior relative to disease transmission and vaccination.
4 Foraging Behavior—Influences on Survival and Exposure to Risk of Fisheries Interactions
4.1 Diet
Hawaiian monk seals consume a wide variety of prey species of more than 40 families of fish, cephalopods, and crustaceans, many of which have a cryptic, benthic lifestyle. Prey include diurnal and nocturnal species that inhabit a range of habitats from the shallow reefs to subphotic depths (Goodman-Lowe 1998; Longenecker 2010; Iverson et al. 2011). Within this diversity of prey, there is considerable individual variation, with individual seals showing specialization on certain prey species or foraging areas (Parrish et al. 2000; Longenecker 2010; Iverson et al. 2011). Similarly, Mediterranean monk seals also consume a variety of predominantly benthic prey (Appendix 16.7).
Diet composition varies considerably depending on the method of diet analysis (Fig. 16.7). For example, fatty acid analysis (Iverson et al. 2011) indicates that snapper (Lutjanidae), octopus and squid (varied cephalopods) are significant parts of the monk seal diet; however, fecal remains analysis (Goodman-Lowe 1998; Cahoon et al. 2013) found these to be minor components, with triggerfishes (Balistidae) and crustaceans (varied) being the among the most frequent diet items. Both methods found tang and surgeonfishes (Acanthuridae) to be important diet items. Stable isotope analysis indicates that a subset of these prey families are in their diet (Thompson 2011). No one method is likely to provide a precise or complete picture of the monk seal diet, as each method is subject to its own biases. Fecal analysis is highly dependent on digestion rates and only provides a snapshot of the most recently consumed prey, whereas fatty acid and stable isotope analyses can be influenced by model assumptions and the prey libraries used to parameterize them.
Comparative studies have found differences in the diet composition of monk seals in the MHI versus NWHI. Based on fecal remains analysis, MHI seals consumed more fish and fewer cephalopods or crustaceans than NWHI seals (97.9% vs. 78.6% fish, 1.2% vs. 5.7% crustaceans, 0.9% vs. 15.7% cephalopods Cahoon et al. 2013). Further, the MHI monk seals' diet represented a subset (20 families; Cahoon et al. 2013) of the prey taxa included in the NWHI diet (31 families; Goodman-Lowe 1998). Fatty acid analysis found that the proportion of boarfish (Pentacerotidae), duckbill (Percophidae), squid (Ommastrephidae), and squirrelfish snapper (Etelis carbunculus) steadily decreased from the NWHI to the MHI, while the proportions of flower snapper (Pristipomoides zonatus) and surgeonfish (Acanthuridae) steadily increased (Iverson et al. 2011). Dietary differences have also been detected at a finer scale, among NWHI sites. For example, one study detected a greater proportion of octopi in the seals' diet at French Frigate Shoals, suggesting lesser fish availability, which could be linked to monk seals' especially poor condition and juvenile survival at this site (Goodman-Lowe 1998).
Ontogenetic shifts in monk seal diet show that foraging preferences or strategies evolve as animals mature, though there may not be consistent directions of change (Goodman-Lowe 1998; Iverson et al. 2011). When individuals were sampled as juveniles and adults using fatty acid analysis, individuals' diet compositions tended to change as they matured, but the prey species involved in the shifts were highly individual (Iverson et al. 2011). Analysis of fecal remains (but interestingly not fatty acids; Iverson et al. 2011), has consistently showed eel (Muraenidae and Ophichthidae) as an important diet item (Kenyon and Rice 1959; DeLong et al. 1984; Goodman-Lowe 1998; Longenecker et al. 2006; Longenecker 2010). Goodman-Lowe (1998) found that juveniles and subadults tended to have a higher percentage of nocturnal prey and eel in their diet, while adults consumed more wrasse (Labridae) and triggerfish. This has been suggested as a potential link between environment conditions and juvenile foraging success (Longenecker 2010), as both eel recruitment (Miller and McCleave 1994) and juvenile body condition appear to improve in response to oceanographic phenomena such as the periodic warming of the tropical Pacific, termed El Niño (Antonelis et al. 2003).
4.2 Habitat Use—Horizontal
Hawaiian monk seals appear to orient around submarine ridge systems, often following ridges from their home island/atoll to other foraging grounds (Abernathy 1999; Stewart et al. 2006). Areas of highest use tend to be concentrated within the 200 m bathymetric contour (Abernathy 1999; Littnan et al. 2006; Cahoon 2011). Home ranges of monk seals are typically on the order of 100 s of km2 in the MHI (Wilson et al. 2017b) to 1000 s of km2 in the NWHI (Curtice et al. 2011). This variation in home range size tends to reflect local topography and proximity of banks and seamounts used for foraging, but also may be indicative of the availability of prey in areas near the islands used for hauling out.
Monk seal home ranges often overlap, areas used by instrumented seals tend to overlap partially or entirely with others (Abernathy 1999), and seal-mounted video studies have shown tolerant interactions between animals underwater (Parrish et al. 2000). Seals from different sites often overlap at popular foraging locations. Seals from Midway and Kure Atolls use the Nero Seamount between the two atolls, seals from Laysan and Lisianski Islands overlap at the Hampton Seamounts between these sites, and animals from several islands in the MHI utilize Penguin Banks off the shore of Moloka'i (Stewart et al. 2006; Curtice et al. 2011; Cahoon et al. 2013, Wilson et al. 2017b). Individual seals may have favored foraging locations that they frequently visit, but the favored locations, and thus typical travel distances, are highly variable among individuals (Abernathy 1999, Wilson et al. 2017b). In addition to what might be the typical range for a given seal, most seals also show occasional exploratory movements well beyond their primary utilization distribution (Wilson et al. 2017b).
Differences in distance and duration of foraging trips highlight the extra effort dedicated to foraging in the NWHI, supporting the hypothesis that resources are more readily available for monk seals in the MHI. In the NWHI, seals typically use multiple foraging sites outside their home atoll or island (4 on average; Stewart et al. 2006), traveling 24–322 km to reach foraging sites on trips lasting an average of 17–20 days (Abernathy 1999). A notable exception in previous NWHI studies was Pearl and Hermes Reef, where animals seldom left the atoll to forage (throughout a study lasting one season), making home ranges and trip distances smaller for animals at this site (Stewart et al. 2006; Cahoon 2011). In the MHI, seals readily move between islands separated by as little as 15–100 km (~35–37% of individuals; Littnan et al. 2006; Wilson et al. 2017b), favoring foraging locations close to the island on which they commonly haulout. For example, seals from Kaua’i typically traveled between Kaua’i and neighboring Ni'ihau, while seals from Moloka’i often foraged at a submerged bank extending offshore (Cahoon 2011, Wilson et al. 2017b). Foraging trips in the MHI are typically much shorter than those in the NWHI, extending 10–50 km and lasting just 0.5–3.8 days (Cahoon 2011; Gobush et al. 2017).
In most cases, adult animals utilize larger areas and make longer foraging trips than juveniles. In the NWHI, younger animals were less likely than adults to leave their home atoll to forage at distant banks (Abernathy 1999; Stewart et al. 2006; Norris et al. 2017). Adults had more extensive home ranges than weaned pups and juveniles at all sites except Lisianski Island, where juveniles maintained the largest home ranges, followed by weaned pups, with adults having the smallest ranges (Curtice et al. 2011). In the MHI, like most of the NWHI, weaned pups used the smallest areas, followed by juveniles, with adults ranging the farthest.
4.3 Habitat Use—Vertical
Monk seals primarily dive to shallow depths (<50 m) for durations of 4–8 min (DeLong et al. 1984; Abernathy 1999; Stewart et al. 2006; Cahoon 2011, Wilson et al. 2017b). While the longest dive on record is ~20 min, dives lasting more than 10 min are rare (<5%; Abernathy 1999). Individual animals might diurnally shift their activity, showing a preference either for day or night foraging (Abernathy 1999), but there are not consistent or significant diurnal patterns in monk seal foraging activity (Parrish et al. 2000, Wilson et al. 2017a). Monk seals have also been recorded sleeping (34% of the time) and socializing with other seals (9% of the time) in waters as deep as 80 m (Parrish et al. 2000).
In the MHI, dives tend to be shallow, with a mean dive depth of 17.3 ± 16.8 m (Wilson et al. 2017a and 90% of dives <20 m (Cahoon 2011). Prey capture activity may be concentrated deeper than the average dive depth; in the NWHI, Parrish et al. (2000) found that prey searching and capture activity was concentrated at 50–60 m, while in the MHI, it occurred at a mean of 25.3 ± 16.2 m (Wilson et al. 2017a). Several studies have shown rare dives over 100 m and even >300 m, indicating what might be occasional explorations or directed deepwater foraging (DeLong et al. 1984; Parrish et al. 2002, Wilson et al. 2017a) as well as a few individuals with a focus on diving deeper banks (Parrish et al. 2000; Norris et al. 2017).
4.4 Foraging Behaviors
The Hawaiian monk seals' foraging strategy of focusing on cryptic prey leads to continuous foraging as seals move between small patches of habitat harboring individual prey (Wilson et al. 2017b). Seal-mounted cameras have been essential in revealing the ways that monk seals interact with their foraging habitat. Seals may swim in the upper water column (typically making shallow dives of 15–20 m) while transiting to an offshore bank, but most of their swimming time is focused near the seafloor where they can find demersal and benthic prey (Fig. 16.8; Parrish et al. 2000). Seals show the greatest foraging effort in benthic areas with moderate complexity, such as sand beds with occasional rocks or coral heads (Parrish et al. 2000, Wilson et al. 2017a). Uniform smooth or hard substrates likely offer fewer hiding spots to harbor the monk seals' prey, whereas highly complex areas such as extensive coral reefs likely make maneuvering and prey capture less efficient (Parrish et al. 2000).
When monk seals find a prey item, they typically invert their body, using their head to dig down into the sand or turn over rock or coral fragments to catch prey (Parrish et al. 2000, Wilson et al. 2017a). This characteristic and abrupt change in body position has made it possible to develop models that can successfully use sensors, such as triaxial accelerometers, to detect prey capture attempts from biologging instruments (Wilson et al. 2017a). These models have shown that the probability of prey capture attempts increases on long, deep dives with more time at the bottom and with increased body motion (Wilson et al. 2017a).
Monk seals often target small prey that are quickly consumed while continuing to swim and forage (80% of prey items <20 cm; Parrish et al. 2000). Large prey require substantially more handling time and are often consumed at the surface (Parrish et al. 2000). Handling time may also be impacted by the type of prey targeted; on average, seals spent three times longer digging out and eating burrowing prey vs. prey items camouflaged on the surface of the sand floor (Parrish et al. 2005). Individual seals can specialize in employing these different hunting tactics, with some focusing on sand fields where they adeptly dig burrowing prey from the sand, while others routinely search under large loose rocks for hiding prey (Parrish and Littnan 2007).
There is evidence that juvenile seals adjust their foraging habits over time, either developing physical strength, physiological capacity, or increasing local knowledge. Translocations (of animals after rehabilitation) have helped illustrate the learning process. Juvenile seals that were removed from the local environment for a period of captive rehabilitation showed wider exploratory movements upon release, whereas those that remained in their natural habitat had ranges more focused on particular foraging areas (Norris et al. 2011). Over time, the foraging range and diving habits of the post-rehabilitation seals converged with the controls as they learned the local foraging environment (Norris et al. 2011). Video-based studies at French Frigate Shoals found that juvenile seals used sand fields more extensively and targeted smaller prey (<10 cm) than adults (Parrish et al. 2005). Age-specific activity budgets (from video and triaxial accelerometry data; NMFS HMSRP unpublished instrument data) have shown that juvenile animals make more exploratory movements, use more energetically costly foraging techniques, and spend less time resting at sea than adults. This likely occurs because adults are more adept at finding profitable foraging areas, spending less time searching before making capture attempts, and using more energy-efficient prey capture techniques. These differences in foraging behavior illustrate the learning curve that young animals must overcome and provide clues to the mechanisms that may drive differential survival across age classes.
Interspecific competition is thought to be one factor underlying the nutritional limitation and poor body condition that is commonly seen in juvenile seals in the NWHI (National Marine Fisheries Service 2007). Seal-mounted videos have shown that predatory fish (most commonly jacks, Caranx spp., and sharks, Carcharhinus spp.) travel with foraging seals to exploit the seals' superior ability to flush cryptic prey from benthic habitats (Parrish et al. 2008). While escorts by such competitors were common in the NWHI (predatory fish followed seals for 17% of their foraging time; Parrish et al. 2008), they are much rarer in the MHI (few escorts observed in video footage; Wilson et al. 2017a) where predatory fish populations have been decreased by human fishing pressure. This difference in competitive pressure may be one factor leading to more robust nutritional status and improved survival of seals in the MHI (Baker et al. 2011b; Wilson et al. 2017a).
4.5 Interventions to Mitigate Food Limitation
Nutritional limitation leading to poor body condition and starvation takes a substantial toll on juvenile monk seals in the NWHI, with survival rates dipping as low as 0.20 at some sites (Baker and Thompson 2007). Ecological factors might impact nutritional condition including intra- and inter-specific competition (Parrish et al. 2008, Baker et al. 2011b), differences in prey type, abundance, or quality (Craig and Ragen 1999; Friedlander and Demartini 2002; Parrish et al. 2005), and accessibility and effort required to acquire food (Parrish et al. 2005; Cahoon 2011; Curtice et al. 2011). Because the juvenile survival bottleneck has lasting impacts on population dynamics and recovery potential, the HMSRP designates enhancing juvenile survival as a top priority (National Marine Fisheries Service 2007). While nutritional limitation may stem from ecosystem-level issues, interventions at the individual seal level have proven effective in boosting survival (Harting et al. 2014). Two key intervention strategies are used to mitigate nutritional limitation: translocating weaned pups from sites with the lowest survival rates to those with higher survival (see Sect. 16.5), and rehabilitation of young monk seals in poor body condition.
Pups tend to have insufficient maternal investment if they wean with an axillary girth <90 cm; these pups are typically found in the NWHI, and they have a little chance of first-year survival (Baker 2008). These undersized pups are prioritized for rehabilitation, and female pups in particular because they are the species' reproductive future. Juveniles (and one subadult) in poor body condition have also been rehabilitated. Early rehabilitation efforts included supplemental feeding in beach pens in the remote NWHI and rehabilitation at NMFS facilities and other facilities. While > 50 seals were successfully released back into the wild in the NWHI, both approaches had mixed results, with some animals dying or being deemed non-releasable (Ragen and Lavigne 1999; Norris et al. 2011). In 2014, The Marine Mammal Center opened a dedicated monk seal rehabilitation facility, Ke Kai Ola, in Kailua-Kona. Since this hospital's establishment, >30 NWHI seals have been rehabilitated, with all being released back into the wild. Young seals released from rehabilitation have survived at rates greater than would be expected without rehabilitation, given their poor body condition at the time of stranding (NMFS HMSRP unpublished database).
4.6 Interventions to Mitigate Fisheries Interactions
The foraging habits of monk seals in the MHI can bring seals into contact with anthropogenic threats. Because foraging in the MHI tends to focus mainly on shallower nearshore areas, much of the seals' space use overlaps with areas humans use for recreation and fishing. Accidental interactions with fishing gear, including ingestion or external lodging of fish hooks (“hookings”) and entanglement in lay nets, can pose a significant risk to monk seals. Net entanglements and hookings pose one of the greatest threats to seal survival in the MHI, depressing potential population growth by as much as 50% (Harting et al. 2020).
NOAA documented 232 monk seal–fisheries interactions in the MHI between 1976 and 2019 (Gobush et al. 2017, NMFS HMSRP unpublished database). These included 16 deaths linked to net drownings and 7 from ingested hooks (Harting et al. 2020). While net entanglements pose a greater immediate mortality risk, they are more cryptic than hookings, which are often more visible and, therefore, readily detected and mitigated. Between 1988 and 2014, approximately one quarter (26%) of monk seals in the MHI had at least one documented hooking (Gobush et al. 2017). Detected interactions likely represent only a portion of the complete number as detection is imperfect, and seals vary in their detectability based on their haulout areas' accessibility. Fisheries interactions have been the highest on the islands of Kaua’i and O’ahu where there is a high intersection of seal haulout use and human fishing effort in addition to much public surveillance (Gobush et al. 2017). The rates of seal–fisheries interactions have increased as the MHI seal population has grown (Gobush et al. 2017).
Mitigating threats from fisheries interactions involves a multifaceted approach, working with state agencies, local fishing communities, and the visiting and resident public to encourage wildlife awareness and responsible fishing, reporting, and finally, intervening to remove dangerous fishing gear from seals. HMSRP has successfully removed hooks from ~ 50% of seals that have been detected hooked, with 5% of these cases requiring surgery (NMFS HMSRP unpublished database).
5 Dispersal Behavior and Site Fidelity—Influences on the Success of Translocation as a Conservation Tool
The nature of the Hawaiian Archipelago, a string of atolls and islands separated by 10–100 s of kms, leads to fragmentation of island populations. Across the archipelago, subpopulations of Hawaiian monk seals at different sites (individual islands or atolls) are impacted by localized threats and exhibit variation in demographic rates (Baker and Thompson 2007). Despite local effects, Hawaiian monk seals move widely throughout the archipelago (Stewart et al. 2006; Johanos et al. 2014), and genetic analysis suggests that gene flow is sufficient to avoid genetic isolation (Schultz et al. 2011). An understanding of the degree of dispersal and population connectivity can be vital for informing conservation actions. Here we review research on monk seal dispersal and site fidelity and consider implications for translocation as a conservation tool.
5.1 Dispersal Patterns and Site Fidelity
The foundational knowledge regarding monk seal dispersal and site fidelity has come from the long history of visual monitoring as well as tracking through instrumentation. A 30-year retrospective study resighted 4,438 seals (4,320 NWHI and 118 MHI from 1981–2011), detecting 1,161 movements between sites (different islands or atolls) by 373 individuals (Johanos et al. 2014). Telemetry studies have documented many movements among sites and can capture more short-term movements (such as foraging trips) in addition to longer-term dispersals (Stewart et al. 2006).
The probability of a monk seal dispersing away from its natal site increases with age. Weaned pups spend much of their time near the beach where they weaned, and gradually increase their range in the months after weaning (Henderson and Johanos 1988; Norris et al. 2017). By the age of 10, 14% of seals had dispersed to a site other than their natal site, with an additional 28% making some brief forays before returning to their natal site (Johanos et al. 2014). Likewise, telemetry studies suggest that older animals are increasingly likely to move away from their home sites for longer foraging trips, and show that proportions of animals traveling between sites are likely even higher than what is recorded through visual observations (Stewart et al. 2006). Dispersal does not appear to be sex-biased, as both males and females appear equally likely to disperse from their natal sites (Johanos et al. 2014). Once females pup, they show high fidelity to their pupping site, with 95% of subsequent pups born at the same island or atoll as the female's first pup (Johanos et al. 2014).
Distance between sites influences connectivity and movement patterns, with closer sites exchanging more seals than those farther apart. Johanos et al. (2014) observed relatively high movement rates between sites separated by 100 km or less and very little between locations separated by >400 km (Johanos et al. 2014). In the MHI, where islands are separated by only 10 s of kms, over 30% of seals use multiple islands (Littnan et al. 2006; Wilson et al. 2017b). While less common, longer distance dispersals occur, including trips from the NWHI to the MHI, which would have been important in the early recovery of the MHI monk seal population (Johanos et al. 2014).
5.2 Translocation as a Conservation Intervention
Knowledge of monk seal movement patterns has informed (and been informed by) the use of translocation as a conservation intervention. For over 35 years, NMFS's HMSRP has been successfully utilizing translocation as a means to enhance the survival of Hawaiian monk seals. The primary purpose of Hawaiian monk seal translocations has been to improve the survival probability of young seals in the NWHI by moving weaned pups away from islets with especially heavy shark predation or by moving pups and rehabilitated yearlings from sites with lower survival to sites with higher survival and presumed favorable habitat (Fig. 16.9; Baker et al. 2011a, Baker et al. 2020). Translocation has also been used to mitigate various other survival threats. As discussed earlier, adult male monk seals were moved from Laysan Island to Johnston Atoll and the MHI to decrease the male-biased sex ratio to reduce the incidence of male aggression that led to high female mortality (Johanos et al. 2010, see Sect. 16.2). On occasion, seals have also been translocated within the MHI, or from the MHI to the NWHI to (a) move seals that have shown dangerous signs of habituation to human interaction, or (b) preempt habituation by moving weaned pups born on beaches with high human use to more remote locations (Baker et al. 2011a).
Translocating Hawaiian monk seals has proven safe overall, and seals have shown survival rates matching those of seals native to the destination sites (Baker et al. 2011a; Baker et al. 2020). However, the effectiveness of translocations has varied depending on the conservation goal and the target animal (Baker et al. 2011a). Upon reviewing 247 translocations taking place from 1984 through 2009, Baker et al. (2011a) found that weaned pups were the most amenable to translocation, while older animals were more likely to roam farther from their destination sites.
Because Hawaiian monk seal pups remain relatively sedentary during a post-weaning fast, they typically develop little foraging experience or strong ties to their home site prior to translocation (Henderson and Johanos 1988). Thus, weaned pups adapt remarkably well to their translocation site, typically remaining near their release beach for >40 days (Baker et al. 2011a). This has led to success in translocating weaned pups even small distances; for instance, at French Frigate Shoals, moving a pup from the islets with the heaviest shark predation to an islet just a few kms away can dramatically improve early survival (Baker et al. 2011a). When moved from one island/atoll to another, weaned pups typically remained at their new site until maturity (Baker et al. 2020). When tracked along with control animals at release sites, translocated pups developed similar foraging movements and maintained ranges similar to resident weaned pups (Norris et al. 2017).
In contrast, older seals (juveniles, subadults, adults) have already developed extensive home ranges (see Sect. 16.4) and have proven less likely to remain close to their site of translocation. Of the adult males translocated from the NWHI to the MHI, most resighted seals were seen on multiple islands within the MHI (Baker et al. 2011a) and one of these males made two round trips between the MHI and Nihoa (Johanos et al. 2014). One or more of the males translocated to Johnston Atoll remained there at least 2 years after release (though note low monitoring effort at this location; NMFS HMSRP unpublished database). While these males did not show strong fidelity to their new site, none are known to have returned to the NWHI. Thus, these translocations successfully mitigated a threat to the females at the origin site of Laysan Island.
Translocations of older seals showing signs of human habituation have been less successful (Baker et al. 2011a). In cases where seals were moved within the MHI, their range of normal foraging movements allowed them to come in contact with humans again quickly, and dangerous behaviors (interacting with swimmers or fishers) often resumed. In the few cases where MHI seals were moved to the NWHI to achieve a greater distance from human interaction, the seals failed to adapt to their new sites and disappeared soon after translocation, sometimes after making long exploratory movements (NMFS HMSRP unpublished instrument data). It is unknown whether this may be caused by a drive to return to the home site in the MHI, or lack of familiarity with the increased competition for resources with increased numbers of seals and other predators in the NWHI.
6 Conclusions and Future Directions
Behavioral ecology and ethological research have been essential in informing and evaluating conservation interventions, which are a crucial part of the effort to recover the Hawaiian monk seal. An integral part of the success of the HMSRP has been the extensive database collected from decades of fieldwork, which includes behavioral observations. This database has facilitated the ability to link monk seal ecology and behavior to survival threats and allowed successful interventions to help recover the population. In addition to collecting critical data, researchers' time in the field directly correlates with the ability to detect seals in need and conduct life-saving interventions (Harting et al. 2014).
Technology has dramatically enhanced the ability of researchers to study the underwater behavior of marine mammals. As technology continues to improve, future research directions will benefit from improvements such as smaller instruments, improved modes of data transmission, and longer battery life/solar charging capabilities. It is a consistent goal of NMFS HMSRP to collect data from animals with the least possible disturbance or impact on natural behaviors. In recent years, smaller instrument sizes have allowed us to expand our instrumentation of smaller seals, advancing our understanding of juvenile foraging ecology and the survival threats facing this vulnerable demographic class.
The recolonization of the MHI has provided a unique opportunity to study the influence of environment on monk seal behavior, reproductive success, and population dynamics. In light of population declines in the NWHI and expansion in the MHI, substantial research effort has focused on understanding the drivers underlying regional differences in population dynamics. Studies of foraging behavior have offered valuable clues regarding differences in activity budgets and interactions with competitors (Wilson et al. 2017a). Future research will explore how seals in the MHI versus NWHI may interact differently with their environments. While ecological conditions appear favorable in the MHI, concern has grown about a number of anthropogenic threats (Harting et al. 2020), and ethological research will take on new importance in the effort to understand how monk seal space use, diet, and behavior may impact individual exposure to risk factors.
The Hawaiian Archipelago, shaped dynamically over the past 30 + million years, has undoubtedly undergone substantial changes since monk seals first inhabited the Hawaiian Islands. With climate change altering ocean conditions, coral reef ecosystems, and coastal lands, monk seals are likely to experience more changes in the near future. In particular, severe weather events and sea-level rise have the potential to impact the low-lying sandy islands of the NWHI where the majority of seals reside. We have already seen shifts in favored pupping beaches as islets have shifted or disappeared at some atolls (Baker et al. 2006), and it will be essential to continue to assess monk seals' behavioral capacity to adapt to the changing environment. Further, safeguarding the MHI population in light of anthropogenic threats will become increasingly important to ensure safe refuge in the higher elevation islands.
References
Abernathy KJ (1999) Foraging ecology of Hawaiian monk seals at French Frigate Shoals. Thesis, University of Minnesota Hawaii
Aguirre AA, Keefe TJ, Reif JS, Kashinsky L, Yochem PK, Saliki JT, Stott JL, Goldstein T, Dubey J, Braun R (2007) Infectious disease monitoring of the endangered Hawaiian monk seal. J Wildl Dis 43:229–241
Alcorn D, Henderson J (1984) Resumption of nursing in “weaned” Hawaiian monk seal pups. Elepaio 45:11–12
Antonelis GA, Baker JD, Johanos TC, Braun R, Harting A (2006) Hawaiian monk seal (Monachus schauinslandi): status and conservation issues. Atoll Res Bull 543:75–101
Antonelis GA, Baker JD, Polovina JJ (2003) Improved body condition of weaned Hawaiian monk seal pups associated with El Nino events: potential benefits to an endangered species. Mar Mamm Sci 19:590–598
Atkinson S, Becker BL, Johanos TC, Pietraszek JR, Kuhn BC (1994) Reproductive morphology and status of female Hawaiian monk seals (Monachus schauinslandi) fatally injured by adult male seals. J Reprod Fertil 100:225–230
Atkinson S, Gilmarti W (1992) Seasonal testosterone pattern in Hawaiian monk seals (Monachus schauinslandi). J Reprod Fertil 96:35–39
Atkinson S, Gilmartin W, Lasley B (1993) Testosterone response to a gonadotrophin-releasing hormone agonist in Hawaiian monk seals (Monachus schauinslandi). J Reprod Fertil 97:35–38
Baker J, Becker B, Wurth T, Johanos T, Littnan C, Henderson J (2011a) Translocation as a tool for conservation of the Hawaiian monk seal. Biol Cons 144:2692–2701
Baker JD, Harting AL, Wurth TA, Johanos TC (2011b) Dramatic shifts in Hawaiian monk seal distribution predicted from divergent regional trends. Mar Mamm Sci 27:78–93
Baker JD (2008) Variation in the relationship between offspring size and survival provides insight into causes of mortality in Hawaiian monk seals. Endanger Species Res 5:55–64
Baker JD, Barbieri MM, Johanos TC, Littnan CL, Bohlander JL, Kaufman AC, Harting AL, Farry SC, Yoshinaga CH (2020) Conservation translocations of Hawaiian monk seals: accounting for variability in body condition improves evaluation of translocation efficacy. Marine Mammal Science submitted
Baker JD, Harting AL, Barbieri MM, Johanos TC, Robinson SJ, Littnan CL (2016a) Estimating contact rates of Hawaiian monk seals (Neomonachus schauinslandi) using social network analysis. J Wildl Dis 52:533–543
Baker JD, Harting AL, Johanos TC, Littnan CL (2016b) Estimating Hawaiian monk seal range-wide abundance and associated uncertainty. Endanger Species Res 31:317–324
Baker JD, Harting AL, Barbieri MM, Robinson SJ, Gulland FMD, Littnan CL (2017) Modeling a morbillivirus outbreak in Hawaiian Monk Seals to aid in the design of mitigation programs. J Wildl Dis 53:736–748
Baker JD, Johanos TC (2004) Abundance of the Hawaiian monk seal in the main Hawaiian Islands. Biol Cons 116:103–110
Baker JD, Johanos TC, Wurth TA, Littnan CL (2014) Body growth in Hawaiian monk seals. Mar Mamm Sci 30:259–271
Baker JD, Littnan CL, Johnston DW (2006) Potential effects of sea level rise on the terrestrial habitats of endangered and endemic megafauna in the Northwestern Hawaiian Islands. Endanger Species Res 4:1–10
Baker JD, Thompson PM (2007) Temporal and spatial variation in age-specific survival rates of a long-lived mammal, the Hawaiian monk seal. Proc R Soc B Biol Sci 274:407–415
Barbieri MM, Kashinsky L, Rotstein DS, Colegrove KM, Haman KH, Magargal SL, Sweeny AR, Kaufman AC, Grigg ME, Littnan CL (2016) Protozoal-related mortalities in endangered Hawaiian monk seals Neomonachus schauinslandi. Dis Aquat Org 121:85–95
Boness DJ (1990) Fostering behavior in Hawaiian monk seals: is there a reproductive cost? Behav Ecol Sociobiol 27:113–122
Boness DJ, Craig MP, Honigman L, Austin S (1998) Fostering behavior and the effect of female density in Hawaiian monk seals, Monachus schauinslandi. J Mammal 79:1060–1069
Bronson E, Deem SL, Sanchez C, Murray S (2007) Serologic response to a canarypox-vectored canine distemper virus vaccine in the giant panda (Ailuropoda melanoleuca). 38:363–366
Cahoon M (2011) The foraging ecology of monk seals in the main Hawaiian Islands. Thesis, University of Hawaii
Cahoon M, Littnan C, Longenecker K, Carpenter J (2013) Dietary comparison of two Hawaiian monk seal populations: the role of diet as a driver of divergent population trends. Endanger Species Res 20:137–146
Carretta JV, Forney KA, Muto MM, Barlow J, Baker J, Lowry M (2004) U. S. Pacific Marine Mammal Stock Assessments: 2003. NOAA-TM-NMFS-SWFSC-358291
Chaudun L (2018) Description du répertoire vocal aérien du phoque moine d’Hawaii (Neomonachus schauinslandi) et recherche de signatures vocales individuelles. Institut des Neurosciences Paris-Saclay, CNRS-UMR 9197.
Cobb JN (1902) Commercial fisheries of the Hawaiian Islands (extracted from the US Fish Commission Report for 1901). US Government Printing Office, Washington, DC, pp 353–499, Plates 21–27
Craig MP, Ragen TJ (1999) Body size, survival, and decline of juvenile Hawaiian monk seals, Monachus schauinslandi. Mar Mamm Sci 15:786–809
Curtice C, Schick RS, Dunn DC, Halpin PNJAM (2011) Home range analysis of Hawaiian Monk Seals (Monachus schauinslandi) based on colony, age, and sex. Aquat Mamm 37:360–371
DeLong R, Kooyman G, Gilmartin W, Loughlin T (1984) Hawaiian monk seal diving behavior. Acta Zoologica Fennica 172:129–131
DeLong RL, Fiscus CH, Kenyon KW (1976) Survey of monk seals (Monachus schauinslandi) populations of the Northwestern (Leeward) Hawaiian Islands (Internal report). NMFS, Seattle, WA
Friedlander AM, Demartini EE (2002) Contrasts in density, size and biomass of reef fishes: APEX predators. Mar Ecol Prog Ser 230:253–264
Fyler CA, Reeder TW, Berta A, Antonelis G, Aguilar A, Androukaki E (2005) Historical biogeography and phylogeny of monachine seals (Pinnipedia: Phocidae) based on mitochondrial and nuclear DNA data. J Biogeogr 32:1267–1279
Gobush K, Wurth T, Henderson J, Becker B, Littnan C (2017) Prevalence of interactions between Hawaiian monk seals (Nemonachus schauinslandi) and nearshore fisheries in the main Hawaiian Islands. Pac Conserv Biol 23:25–31
Gobush KS, Farry SC (2012) Non-lethal efforts to deter shark predation of Hawaiian monk seal pups. Aquat Conserv Mar Freshwat Ecosyst 22:751–761
Goodman-Lowe G (1998) Diet of the Hawaiian monk seal (Monachus schauinslandi) from the Northwestern Hawaiian islands during 1991 to 1994. Mar Biol 132:535–546
Grachev M, Kumarev V, Mamaev L et al (1989) Distemper virus in Baikal seals. Nature 338:209
Harting A, Baker J, Becker B (2004) Non-metrical digital photoidentification system for the Hawaiian monk seal. Mar Mamm Sci 20:886–895
Harting AL, Baker JD, Johanos TC (2007) Reproductive patterns of the Hawaiian monk seal. Mar Mamm Sci 23:553–573
Harting AL, Baker JD, Johanos TC (2017) Estimating population size for Hawaiian monk seals using haulout data. J Wildl Manag 81:1202–1209
Harting AL, Barbieri MM, Baker JD, Mercer T, Johanos TC, Robinson SJ, Littnan CL, Colegrove KM, Rotstein DS (2020) Population-level impacts of natural and anthropogenic causes-of-death for Hawaiian Monk Seals in the Main Hawaiian Islands. Ecological Applications submitted
Harting AL, Johanos TC, Littnan CL (2014) Benefits derived from opportunistic survival-enhancing interventions for the Hawaiian Monk Seal: the silver BB paradigm. Endanger Species Res 25:89–96
Hawaiian Monk Seal Research Program (2019) Population summary for Hawaiian Monk seals in 2019. Internal Report IR 20-001; Pacific Island Fisheries Science Center, Honolulu
Heide-Jørgensen MP, Härkönen T, Åberg PJA (1992) Long-term effects of epizootic in harbor seals in the Kattegat-Skagerak and adjacent areas. Ambio 21:511–516
Henderson JR (2001) A pre-and post-MARPOL Annex V summary of Hawaiian monk seal entanglements and marine debris accumulation in the Northwestern Hawaiian Islands, 1982–1998. Mar Pollut Bull 42:584–589
Henderson JR, Johanos TC (1988) Effects of tagging on weaned Hawaiian monk seal pups. Wildl Soc Bull 16:312–317
Hiruki LM, Gilmartin WG, Becker BL, Stirling I (1993) Wounding in Hawaiian monk seals (Monachus schauinslandi). Can J Zool 71:458–468
Hiruki LM, Stirling I, Gilmartin WG, Johanos TC, Becker BL (1993) Significance of wounding to female reproductive success in Hawaiian monk seals (Monachus schauinslandi) at Laysan Island. Can J Zool 71:469–474
Iverson S, Piché J, Blanchard W (2011) Hawaiian monk seals and their prey: assessing characteristics of prey species fatty acid signatures and consequences for estimating monk seal diets using Quantitative Fatty Acid Signature Analysis. NOAA Technical Memorandum NMFS-PIFSC-23
Jensen T, van de Bildt M, Dietz HH, Andersen TH, Hammer AS, Kuiken T, Osterhaus AJS (2002) Another phocine distemper outbreak in Europe. Science 297:209
Job DA, Boness DJ, Francis JM (1995) Individual variation in nursing vocalizations of Hawaiian monk seal pups, Monachus schauinslandi (Phocidae, Pinnipedia), and lack of maternal recognition. Can J Zool 73:975–983
Johanos TC, Becker BL, Baker JD, Ragen TJ, Gilmartin WG, Gerrodette T (2010) Impacts of sex ratio reduction on male aggression in the critically endangered Hawaiian monk seal Monachus schauinslandi. Endanger Species Res 11:123–132
Johanos TC, Becker BL, Ragen TJ (1994) Annual reproductive cycle of the female Hawaiian monk seal (Monachus schauinslandi). Mar Mamm Sci 10:13–30
Johanos TC, Harting AL, Wurth TA, Baker JD (2014) Range-wide movement patterns of Hawaiian monk seals. Mar Mamm Sci 30:165–1174
Johnson A, Delong RL, Fiscus CH, Kenyon KW (1982) Population status of the Hawaiian monk seal (Monachus schauinslandi), 1978. J Mammal 63:415–421
Kennedy S, Kuiken T, Jepson PD, Deaville R, Forsyth M, Barrett T, Van de Bildt M, Osterhaus A, Eybatov T, Duck C, Kydyrmanov A (2000) Mass die-Off of Caspian seals caused by canine distemper virus. Emerg Infect Dis 6:637
Kenyon KW (1973) The Hawaiian monk seal. International Union for the Conservation of Nature and Natural Resources, Publications, New Series, Supplemental Paper 39: 88–97
Kenyon KW, Rice DW (1959) Life history of the Hawaiian monk seal. Pac Sci 13:215–252
Le Boeuf BJ, Mesnick S (1991) Sexual behavior of male northern elephant seals: I lethal injuries to adult females. Behaviour 116:143–162
LeDuc R (2009) Biogeography. In: Perrin WF, Würsig B, Thewissen JGM (eds) Encyclopedia of marine mammals. Academic Press, San Diego, pp 112–115
Littnan CL, Stewart BS, Yochem PK, Braun R (2006) Survey for selected pathogens and evaluation of disease risk factors for endangered Hawaiian monk seals in the main Hawaiian Islands. EcoHealth 3:232–244
Longenecker K (2010) Fishes in the Hawaiian monk seal diet, based on regurgitate samples collected in the Northwestern Hawaiian Islands. Mar Mamm Sci 26:420–429
Longenecker K, Dollar RA, Cahoon M (2006) Increasing taxonomic resolution in dietary analysis of the Hawaiian monk seal. Atoll Res Bull 543:101–113
Lopez J, Hyrenbach KD, Littnan C, Ylitalo GM (2014) Geographic variation of persistent organic pollutants in Hawaiian monk seals Monachus schauinslandi in the main Hawaiian Islands. Endanger Species Res 24:249–262
Mesnick SL, Le Boeuf BJ (1991) Sexual Behavior of male northern elephant seals: II. Female response to potentially injurious encounters. Behaviour 117:262–280
Miller MJ, McCleave JD (1994) Species assemblages of leptocephali in the Subtropical Convergence Zone of the Sargasso Sea. J Mar Res 52:743–772
National Marine Fisheries Service (2007) Recovery Plan for the Hawaiian Monk Seal (Monachus schauinslandi): Revision
NMFS HMSRP. unpublished database. Hawaiian monk seal sightings database. https://inport.nmfs.noaa.gov
NMFS HMSRP. unpublished instrument data. Instrument deployments on Hawaiian monk seals.
Norris TA, Littnan CL, Gulland FMD (2011) Evaluation of the captive care and post-release behavior and survival of seven juvenile female Hawaiian monk seals (Monachus schauinslandi). Aquat Mamm 37:342–353
Norris TA, Littnan CL, Gulland FM, Baker JD, Harvey JT (2017) An integrated approach for assessing translocation as an effective conservation tool for Hawaiian monk seals. Endanger Species Res 32:103–115
Parrish FA, Abernathy K (2006) Movements of monk seals relative to ecological depth zones in the lower Northwestern Hawaiian Islands. Atoll Res Bull 543:115–130
Parrish FA, Abernathy K, Marshall GJ, Buhleier BM (2002) Hawaiian monk seals (Monachus schauinslandi) foraging in deep-water coral beds. Mar Mamm Sci 18:244–258
Parrish FA, Craig MP, Ragen TJ, Marshall GJ, Buhleier BM (2000) Identifying diurnal foraging habitat of endangered Hawaiian monk seals using a seal-mounted video camera. Mar Mamm Sci 16:392–412
Parrish FA, Littnan CL (2007) Changing perspectives in Hawaiian monk seal research using animal-borne imaging. Mar Technol Soc J 41:30–34
Parrish FA, Marshall GJ, Buhleier B, Antonelis GA (2008) Foraging interaction between monk seals and large predatory fish in the Northwestern Hawaiian Islands. Endanger Species Res 4:299
Parrish FA, Marshall GJ, Littnan C, Heithaus M, Canja S, Becker B, Braun R, Antoneijs GA (2005) Foraging of juvenile monk seals at French Frigate Shoals, Hawaii. Mar Mamm Sci 21:93–107
Pietraszek J, Atkinson S (1994) Concentrations of estrone sulfate and progesterone in plasma and saliva, vaginal cytology, and bioelectric impedance during the estrous cycle of the Hawaiian monk seal (Monachus schaunslandi). Mar Mamm Sci 10:430–441
Ragen TJ, Lavigne DM (1999) The Hawaiian monk seal: biology of an endangered species. In: Twiss JR, Reeves RR (eds) Conservation and management of marine mammals. Smithsonian Institution Press, Washington, DC, pp 224–245
Riedman ML, Le Boeuf BJ (1982) Mother-pup separation and adoption in northern elephant seals. Behav Ecol Sociobiol 11:203–215
Robinson S, Harting A, Mercer T, Johanos T, Baker J, Littnan C (2020) Sightings patterns reveal cryptic pupping events to revise reproductive rate estimates for Hawaiian monk seals in the Main Hawaiian Islands. Mar Mamm Sci submitted
Robinson SJ, Barbieri MM, Murphy S, Baker JD, Harting AL, Craft ME, Littnan CL (2018) Model recommendations meet management reality: implementation and evaluation of a network-informed vaccination effort for endangered Hawaiian monk seals. Proc R Soc B 285: 20171899
Rose NA, Le Boeuf BJ, Deutsch CJ (1991) Sexual behavior of male northern elephant seals: III. The mounting of weaned pups. Behaviour 119:171–192
Rosendahl PH (1994) Aboriginal Hawaiian structural remains and settlement patterns in the upland archeological zone at Lapakahi, Island of Hawaii. J Hawaii Archeol 3:15–70
Schaeff CM, Boness DJ, Bowen WD (1999) Female distribution, genetic relatedness, and fostering behaviour in harbour seals, Phoca vitulina. Anim Behav 427–434
Schultz JK, Baker JD, Toonen RJ, Bowen BW (2009) Extremely low genetic diversity in the endangered Hawaiian monk seal (Monachus schauinslandi). J Hered 100:25–33
Schultz JK, Baker JD, Toonen RJ, Harting AL, Bowen BW (2011) Range-wide genetic connectivity of the Hawaiian monk seal and implications for translocation. Conserv Biol 25:124–132
Starfield AM, Roth JD, Ralls K (1995) “Mobbing” in Hawaiian monk seals (Monachus schauinslani): The value of simulation modeling in the absence of apparently crucial data. Conserv Biol 9:166–174
Stewart BS, Antonelis GA, Baker JD, Yochem PK (2006) Foraging biogeography of Hawaiian monk seals in the northwestern Hawaiian Islands. Atoll Res Bull 543:1–145
Thompson NM (2011) Changes in Northwest Hawaiian Island monk seal (Monachus schauinslandi) populations as evidenced by stable isotope ratios. Nova Southeastern University, Thesis
Watson TK, Kittinger JN, Walters JS, Schofield TD (2011) Culture, conservation, and conflict: assessing the human dimensions of Hawaiian monk seal recovery. Aquat Mamm 37:386–396
Wilson K, Littnan C, Halpin P, Read A (2017a) Integrating multiple technologies to understand the foraging behaviour of Hawaiian monk seals. R Soc Open Sci 4:160703
Wilson K, Littnan C, Read A (2017b) Movements and home ranges of monk seals in the main Hawaiian Islands. Mar Mamm Sci 33:1080–1096
Wirtz WO (1968) Reproduction, growth and development, and juvenile mortality in the Hawaiian monk seal. J Mammal 49:229–238
Yochem PK (unpublished) Vaccination of Hawaiian monk seals with canarypox recombinant canine distemper vaccine (Merial Purevax Ferret Distemper Vaccine). Internal Report to Pacific Island Fisheries Science Center, Honolulu.
Acknowledgements
We are indebted to the entire Hawaiian Monk Seal Research Program, to the generations of biologists who dedicated their time in the remotest reaches of the Hawaiian Archipelago to build the strong data foundation upon which species recovery efforts have been built. We are likewise grateful to the staff of partner agencies, volunteers, and engaged members of the public that make our research and recovery activities possible. Many thanks to valuable comments in the early review from Jason Baker and Charles Littnan.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Appendices
16.7 Appendix
Mediterranean Monk Seal Monachus Monachus
By Joan Gonzalvo and Giuseppe Notarbartolo di Sciara; Tethys Research Institute, Milan, Italy.

Mediterranean monk seal, swimming (left) and hauled out on rocks in a sea cave (right). Photo credit: Joan Gonzalvo; Tethys Research Institute.
Mediterranean monk seals (Monachus monachus) were once widely and continuously distributed in the Mediterranean and Black Seas, and in North Atlantic waters from Morocco to Mauritania, including Cape Verde and the Canary Islands, Madeira, and the Azores (Johnson et al. 2006). Today fewer than 700 individuals are thought to survive in isolated subpopulations in the eastern Mediterranean, the archipelago of Madeira, and the Cabo Blanco area in the north-eastern Atlantic Ocean (Karamanlidis et al. 2015). The largest aggregations of Mediterranean monk seals are found near Cabo Blanco (González and Fernandez de Larrinoa 2012, Martínez-Jauregui et al. 2012). Principal sites in the Mediterranean are located in the Ionian and Aegean seas, including the National Marine Park of Alonissos (Trivourea et al. 2011) and the Gyaros Marine Protected Area (Dendrinos et al. 2008), both in Greece.
Mediterranean monk seals have teetered on the brink of extinction for about one-half century (Notarbartolo di Sciara and Kotomatas 2016). After being classified as Critically Endangered for almost two decades, their status was reassessed as Endangered on the IUCN’s Red List (Karamanlidis and Dendrinos 2015). Reassessment was based on the notion that monk seal numbers in specific locations (i.e., the Madeira archipelago, Western Sahara-Mauritania, and selected localities in the Aegean Sea) have been stable or may even be increasing. Moreover, there are recent indications that seals might be frequenting areas in the Mediterranean Basin within their historical range where they had been extirpated in previous decades (Bundone et al. 2019).
Historical evidence suggests that Mediterranean monk seals commonly hauled out on open beaches (Johnson and Lavigne 1999, González 2015). In more recent times—probably as an adaptation to increased human disturbance—they generally seek refuge in remote marine caves. These natural rocky shelters share common morphological characteristics, including one or more entrances above or below water level, an entrance corridor, an internal pool, and a beach that provides a dry haulout area (Dendrinos et al. 2007).
The monk seal populations at Cabo Blanco in the Atlantic and at Gyaros Island in the eastern Mediterranean are the only large extant aggregations of the species that still preserve the structure of a colony. The remaining subpopulations in the eastern Mediterranean are usually small, fragmented groups of <20 individuals (Karamanlidis et al. 2015). Most of our knowledge about Mediterranean monk seals’ mating system comes from observations made in the Cabo Blanco area. Mating has only been observed to occur in the water, where males, who are markedly larger than females (Samaranch and González 2000), patrol and defend clearly delineated long-lasting aquatic territories. Aggressive interactions with intruders are characterized by loud puffs (i.e., short, explosive burst of breath), chases, and subaquatic fights. This aquatic breeding strategy results in very low levels of polygyny, as evidenced by the observed low or null relatedness among pups belonging to the same cohort (Pastor et al. 2011).
Pupping periods show differences between locations; in the colony of Cabo Blanco, births are recorded from April to November (González et al. 2002), while elsewhere, births are concentrated between October and November (Littnan et al. 2018). For the first week after giving birth, mothers spend much time on land lying close to their newborns. After this time and throughout the nursing period, females leave their pups unattended for extended periods, lasting up to several hours to forage. As lactation progresses, pups begin to swim and engage in short, increasingly independent trips between caves. During mother–pup encounters, mothers use nuzzling and vocalization to recognize their pups (Aguilar et al. 2007, Gazo and Aguilar 2005). Fostering and milk stealing are common behavior patterns for lactating females and pups, respectively (Pires 2004, Aguilar et al. 2007). Weaning of pups gradually occurs at four to five months of age (Aguilar et al. 2007, Karamanlidis et al. 2015), when pups begin to forage on their own (Pastor and Aguilar 2003).
Mediterranean monk seals have a varied diet, consisting mainly of demersal fishes, cephalopods (the common octopus, Octopus vulgaris, being the most frequent prey item), and crustaceans (Salman et al. 2001, Karamanlidis et al. 2014, Pierce et al. 2011, Pinela et al. 2010, Kiraç and Ok 2019). Body parts of green turtles (Chelonia mydas) were also recently found in the stomach of an adult seal stranded in Turkey (Tonay et al. 2016). When foraging, monk seal pups typically dive to depths of a few tens of meters (mean depth = 11.6 ± 9.5 m), and their dive performance increases with age, in time spent diving and in dive parameters (Gazo et al. 2006). In Greece, a tagged rehabilitated monk seal performed 5–7 min dives with a mean maximum depth of 41 m and an overall maximum dive depth of 123 m (Dendrinos et al. 2007). Tagged seals have descended to the euphotic zone (Littnan et al. 2018) on dives lasting up to 18 min (Kiraç et al. 2002).
While at sea, Mediterranean monk seals have been reported sleeping, either at the surface floating (vertically or horizontally) with eyes closed, or resting underwater on the seafloor or over seagrass beds with eyes and nostrils shut (Karamanlidis et al. 2017, Mpougas et al. 2019). We have also observed similar episodes in the Ionian Sea (J. Gonzalvo, unpublished). On all occasions, seals woke up when approached by humans.
The main threats faced by Mediterranean monk seals include: (a) deliberate killings, mostly by artisanal fishers retaliating against net depredation and damage; (b) critical habitat deterioration, destruction, and fragmentation; (c) disturbance caused by tourists entering breeding caves during the reproductive season, as well as seal–boat interactions; and (d) bycatch in fishing gear, mainly of young inexperienced individuals (Güçlüsoy et al. 2004, Karamanlidis et al. 2020, Karamanlidis and Dendrinos 2015, Mpougas et al. 2019, Notarbartolo di Sciara and Kotomatas 2016). Other threats, such as disease outbreaks (e.g., morbillivirus), toxic algal blooms, prey depletion, and contamination by pollutants and oil spills in critical habitat, also have the potential of reducing monk seal survival (Karamanlidis et al. 2015). Inbreeding also raises concern, since the species appears to be “one of the most genetically depauperate mammals on Earth” (Karamanlidis et al. 2016), a condition believed to substantially increase the probability of extinction.
The Mediterranean monk seal is listed in Appendices I and II of the Convention on the Conservation of Migratory Species of Wild Animals (CMS), in Appendix I of the Convention on International Trade of Endangered Species (CITES), in Annex II of the Barcelona Convention Protocol on Specially Protected Areas and Biodiversity in the Mediterranean, and is a priority species of Community Interest, listed in Annexes II and IV of the EU Habitats Directive. The species is protected throughout its range through national laws. Parties to the Barcelona Convention have adopted a “Regional strategy for the conservation of Mediterranean monk seal” in 2013, revised and updated in 2019.
References
-
Aguilar A, Cappozzo LH, Gazo M, Pastor T, Forcada J, Grau E (2007) Lactation and mother–pup behaviour in the Mediterranean monk seal Monachus monachus: an unusual pattern for a phocid. J Mar Biol Assoc UK 87:93–99.
-
Bundone L, Panou A, Molinaroli E (2019) On sightings of (vagrant?) monk seals, Monachus monachus, in the Mediterranean Basin and their importance for the conservation of the species. Aquat Conserv Mar Freshw Ecosyst 29:554–563.
-
Dendrinos P, Karamanlidis AA, Kotomatas S, Legakis A, Tounta E, Matthiopoulos J (2007) Pupping habitat use in the Mediterranean monk seal: a long-term study. Mar Mamm Sci 23:615–628.
-
Dendrinos P, Karamanlidis AA, Kotomatas S, Paravas V, Adamantopoulou S (2008) Report of a new Mediterranean monk seal (Monachus monachus) breeding colony in the Aegean Sea, Greece. Aquat Mamm 34:355–361.
-
Gazo M, Aguilar A (2005) Maternal attendance and diving behavior of a lactating Mediterranean monk seal. Mar Mamm Sci 21:340–345.
-
Gazo M, Lydersen CV, Aguilar A (2006) Diving behaviour of Mediterranean monk seal pups during lactation and post weaning. Mar Ecol Progress Ser 308:303–309.
-
González LM, Cedenilla MA, Fernández de Larrinoa P, Layna JF, Aparicio F (2002) Changes in the breeding variables of the Mediterranean monk seal (Monachus monachus) colony of Cabo Blanco Peninsula after a mass mortality episode. Mammalia 6:173–182.
-
González LM, Fernandez de Larrinoa P (2012) Mediterranean monk seal Monachus monachus distribution and fisheries interactions in the Atlantic Sahara during the second half of the 20th century. Mammalia 77:41–49.
-
González LM (2015) Prehistoric and historic distributions of the critically endangered Mediterranean monk seal (Monachus monachus) in the eastern Atlantic. Mar Mamm Sci 31:1168–1192.
-
Güçlüsoy H, Kýraç CO, Veryeri NO, Savas Y (2004). Status of the Mediterranean monk seal, Monachus monachus (Hermann, 1779) in the coastal waters of Turkey. E.U. J Fish Aquat Sci 21(3–4):201–210.
-
Johnson WM, Karamanlidis AA, Dendrinos P, Fernández de Larrinoa P, Gazo M, González LM, Güçlüsoy H, Pires R, Schnellmann M (2006) Monk seal fact files. Biology, behaviour, status and conservation of the Mediterranean monk seal, Monachus monachus. The Monachus Guardian. http://www.monachus-guardian.org/factfiles/medit01.htm
-
Johnson WM, Lavigne DM (1999) Monk seals in antiquity. The Mediterranean monk seal (Monachus monachus) in ancient history and literature. Mededelingen 35:1–101.
-
Karamanlidis AA, Adamantopoulou S, Kallianiotis AA, Tounta E, Dendrinos P (2020) An interview‐based approach assessing interactions between seals and small‐scale fisheries informs the conservation strategy of the endangered Mediterranean monk seal. Aquat Conserv Mar Freshw Ecosyst 3:928–936.
-
Karamanlidis AA, Curtis PJ, Hirons AC, Psaradellis M, Dendrinos P, Hopkins III JB (2014) Stable isotopes confirm a coastal diet for critically endangered Mediterranean monk seals. Isot Environ Health Stud 50:1–11.
-
Karamanlidis AA, Dendrinos P (2015) Monachus monachus (errata version published in 2017). The IUCN Red List of Threatened Species 2015: e.T13653A117647375. https://dx.doi.org/10.2305/IUCN.UK.2015-4.RLTS.T13653A45227543.en. Downloaded on 09 June 2020.
-
Karamanlidis AA, Dendrinos P, Fernández de Larrinoa P, Gücü AC, Johnson WM, Kiraç CO, Pires R (2015) The Mediterranean monk seal Monachus monachus: status, biology, threats, and conservation priorities. Mamm Rev 46:92–105.
-
Karamanlidis AA, Gaughran S, Aguilar A, Dendrinos P, Huber D, Pires R, Schultz J, Skrbinsek T, Amato G (2016) Shaping species conservation strategies using mtDNA analysis: the case of the elusive Mediterranean monk seal (Monachus monachus). Biol Conserv 193:71–79.
-
Karamanlidis AA, Lyamin O, Adamantopoulou S, Dendrinos P (2017) First observations of aquatic sleep in the Mediterranean monk seal (Monachus monachus). Aquat Mamm 43(1):82–86. https://doi.org/10.1578/AM.43.1.2017.82.
-
Kiraç CO, Savas Y, Güçlüsoy H, Veryeri NO (2002) Observations on diving behaviour of free ranging monk seals Monachus monachus on the Turkish coasts. Monachus Guard. https://www.monachus-guardian.org/mguard09/09scien2.htm.
-
Kiraç OC, Ok M (2019) Diet of a Mediterranean monk seal Monachus monachus in a transitional post-weaning phase and its implications for the conservation of the species. Endanger Species Res 39:315–320.
-
Littnan C, Karamanlidis AA, Dendrinos P (2018) Monk seals. In: Würsig B, Thewissen JGM, Kovacs K (eds) Encyclopedia of marine mammals, 3rd edn. Academic Press, pp 622–653
-
Martínez-Jauregui M, Tavecchia G, Cedenilla MA, Coulson T, Fernández de Larrinoa P, Muñoz M, González LM (2012) Population resilience of the Mediterranean monk seal Monachus monachus at Cabo Blanco peninsula. Mar Ecol Progress Ser 461:273–281.
-
Mpougas E, Waggitt JJ, Dendrinos P, Adamantopoulou S, Karamanlidis AA (2019) Mediterranean Monk Seal (Monachus monachus) behavior at sea and interactions with boat traffic: implications for the conservation of the species in Greece. Aquat Mamm 45:419–424.
-
Notarbartolo di Sciara G, Kotomatas S (2016) Are Mediterranean monk seals, Monachus monachus, being left to save themselves from extinction? Adv Mar Biol 75:259–296.
-
Pastor T, Aguilar A (2003). Reproductive cycle of the female Mediterranean monk seal in the western Sahara. Mar Mamm Sci 19:318–330.
-
Pastor T, Cappozzo HL, Grau E, Amos W, Aguilar A (2011) The mating system of the Mediterranean monk seal in the Western Sahara. Mar Mamm Sci 27(4):E302–E320.
-
Pierce GJ, Hernandez-Milian G, Santos MB, Dendrinos P, Psaradellis M, Tounta E, Androukaki E, Edridge A (2011) Diet of the monk seal (Monachus monachus) in Greek waters. Aquat Mamm 37: 284–297.
-
Pinela AM, Borrell A, Cardona L, Aguilar A (2010) Stable isotope analysis reveals habitat partitioning among marine mammals off the NW African coast and unique trophic niches for two globally threatened species. Mar Ecol Progress Ser 416:295–306.
-
Pires R (2004) One pup –three mothers. Monachus Guard 7:33–34.
-
Salman A, Bilecenoglu M, Güçlüsoy H (2001) Stomach contents of two Mediterranean monk seals (Monachus monachus) from the Aegean Sea, Turkey. J Mar Biol Assoc UK 81:719–720.
-
Samaranch R, González LM (2000) Changes in morphology with age in Mediterranean monk seals (Monachus monachus). Mar Mamm Sci 16:141–157.
-
Tonay AM, Danyer E, Dede A, Öztürk B, Öztürk AA (2016) The stomach content of a Mediterranean Monk Seal (Monachus monachus): finding of Green Turtle (Chelonia mydas) remains. Zool Middle East 62:1–5.
-
Trivourea MN, Karamanlidis AA, Tounta E, Dendrinos P, Kotomatas S (2011) People and the Mediterranean monk seal (Monachus monachus): a study of the socioeconomic impacts of the National Marine Park of Alonissos, Northern Sporades, Greece. Aquat Mamm 37:305–318.
Rights and permissions
Copyright information
© 2022 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Robinson, S., Barbieri, M., Johanos, T. (2022). The Hawaiian Monk Seal: Ethology Applied to Endangered Species Conservation and Recovery. In: Costa, D.P., McHuron, E.A. (eds) Ethology and Behavioral Ecology of Phocids . Ethology and Behavioral Ecology of Marine Mammals. Springer, Cham. https://doi.org/10.1007/978-3-030-88923-4_16
Download citation
DOI: https://doi.org/10.1007/978-3-030-88923-4_16
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-88922-7
Online ISBN: 978-3-030-88923-4
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)